The QualityStocks Daily Stock List
- SilverSun Technologies (SSNT)
- Happy Belly Food Group (VGANF)
- Inovalis Real Estate Investment Trust (IVREF)
- Sintana Energy (SEUSF)
- ParcelPal Logistics (PTNYF)
- Graphano Energy (GELEF)
- Stronghold Digital Mining Inc. (SDIG)
- Curaleaf Holdings Inc. (CURLF)
- Seelos Therapeutics Inc. (SEEL)
- Lucid Motors (LCID)
- Cronos Group Inc. (CRON)
- SOBR Safe Inc.’s (SOBR)
SilverSun Technologies (SSNT)
QualityStocks, PennyStocks24, StreetInsider, TradersPro, StockMarketWatch, MarketClub Analysis, FeedBlitz, Bull Trends, FreeRealTime, MarketBeat, SeriousTraders, The Stock Dork, PoliticsAndMyPortfolio, Schaeffer's, InvestorPlace, INO Market Report, FutureMoneyTrends.com, Daily Trade Alert, comcast.net, BUYINS.NET and Penny Stock Rumble reported earlier on SilverSun Technologies (SSNT), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
SilverSun Technologies, Inc. (NASDAQ: SSNT) provides software solutions. The Company develops business application software for accounting and business management, financial reporting, enterprise resource planning, warehouse management systems, and customer relationship management, as well as offering consulting services for programming, training, and technical support.
It is engaged in providing transformative business management solutions, technologies and professional consulting services to small and medium-sized businesses in the manufacturing, distribution and service industries. The company was founded in 1988 and is headquartered in East Hanover, NJ. It mainly serves clients in the U.S. The company was formerly known as Trey Resources, Inc. and changed its name to SilverSun Technologies, Inc. in June 2011.
The firm is a business application, technology, and consulting company. It is engaged in providing strategies and solutions to meet information, technology and business management needs. Its services and technologies enable customers to manage, protect and monetize their enterprise assets in the cloud. The company has a network services practice, which provides managed services, hosting, business continuity, cloud, e-mail, and Web services.
The company has an information technology (IT) network services practice that provides managed services, cybersecurity, application hosting, disaster recovery, business continuity, cloud and other services. Its value-added services focus on consulting and professional services, specialized programming, training, and technical support.
In addition, the company provides and manages cybersecurity, business continuity, disaster recovery, data back-up, network maintenance and service upgrades, and application hosting services. It serves small and medium-sized businesses primarily in the manufacturing, distribution, and service industries.
The firm’s recently released financial results show an increase in its cybersecurity client base, as well as showing growth in other segments of the company’s operations. This will encourage more investors to inject capital into the firm and boost its growth.
SilverSun Technologies (SSNT), closed Friday's trading session at $21.98, up 18.8108%, on 776,682 volume. The average volume for the last 3 months is 9,272 and the stock's 52-week low/high is $2.71/$22.47.
Happy Belly Food Group (VGANF)
We reported earlier on Happy Belly Food Group (VGANF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Happy Belly Food Group Inc. (OTCQB: VGANF) (CNSX: HBFG) (FRA: W3J) is a health and wellness firm engaged in the provision of food products.
The firm has its headquarters in Vancouver, Canada and was incorporated in 2014, on November 24th. Prior to its name change in September 2022, the firm was known as Plant & Co. Brands Limited. It operates as part of the packaged foods industry, under the consumer defensive sector. The firm mainly serves consumers in Canada.
The enterprise operates through two segments, Consumer Product Goods and Quick Restaurant Services. Its Consumer Product Goods segment comprises of 2 brands; Lumber Heads, which produces and sells snack foods; and Holy Crap, which produces and sells breakfast cereal and related food products. The company provides its products under these brand names, which are gluten-free, organic, kosher and plant-based. It markets its products through retailers as well as online retailers and its own Website. On the other hand, the Quick Restaurant Services segment includes brands, such as Rosie's Burgers, Heal Lifestyles, Lettuce Love, Phiro Fresh Greek Grill and JoeyTurks Island Grill. Rosie's Burgers is a boutique QSR restaurant brand that serves original recipe smash burgers, poutine, onion rings, milkshakes, and others.
The company, through its Next Level Partners division, recently signed a strategic consulting agreement with Coho Collective Kitchens Inc., a leading player in the culinary industry. This move will allow both companies to capitalize on the numerous opportunities that lie ahead while also creating new avenues to generate value for their stakeholders.
Happy Belly Food Group (VGANF), closed Friday's trading session at $0.2025, up 12.6878%, on 33,597 volume. The average volume for the last 3 months is 4,651 and the stock's 52-week low/high is $0.055/$0.2025.
Inovalis Real Estate Investment Trust (IVREF)
MarketBeat reported earlier on Inovalis Real Estate Investment Trust (IVREF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Inovalis Real Estate Investment Trust (OTC: IVREF) is an open-ended real estate investment trust focused on acquiring and owning office properties.
The firm has its headquarters in Toronto, Canada and was founded in 2013, on February 8th by Stephane Amine. It operates as part of the REIT-Office industry, under the real estate sector. The firm serves consumers in Canada.
The company was formed for the purpose of acquiring and owning office properties primarily located in Spain, France, and Germany but also opportunistically in other European countries where assets meet the REIT’s investment criteria. Its objective is to convert its extensive knowledge of the French, German and Spanish real estate market into innovative investment products and quality advice for our institutional and private investors. The company acquires (indirectly) real estate properties via CanCorPEurope, authorized Alternative Investment Fund (AIF) and managed by INOVALIS S.A.
The REIT’s Spain property is Delgado while its France properties include Sabliere, Baldi, Delizy, Gaia, Metropolitain and Arcueil. Its Germany properties include Trio, Kosching, Neu Isenburg, Stuttgart, Bad Homburg and Duisburg. The majority of its revenue is generated from rental income in France.As at December 31st 2022, the REIT Properties totaling thirteen office properties in France, Germany and Spain made up roughly 1,540,000ft2 of GLA.
The firm recently announced its latest financial results, with its CEO noting that they remained focused on providing the REIT with greater financial strength and flexibility while also advancing their short and long-term objectives. This may encourage investments into the firm.
Inovalis Real Estate Investment Trust (IVREF), closed Friday's trading session at $1.25, even for the day, on 67 volume. The average volume for the last 3 months is 96,870 and the stock's 52-week low/high is $0.494145/$3.089751.
Sintana Energy (SEUSF)
We reported earlier on Sintana Energy (SEUSF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Sintana Energy Inc. (OTCQB: SEUSF) (CVE: SEI) is an oil and gas firm focused on exploring for, developing and producing crude oil and natural gas resources in Colombia.
The firm has its headquarters in Toronto, Canada and was incorporated in 1994, on February 22nd. It operates as part of the oil and gas E&P industry, under the energy sector. The firm serves consumers in North and South America.
The enterprise’s exploration strategy is to acquire, explore, develop, and produce assets with significant reserves potential. It is engaged in petroleum and natural gas exploration and development activities in 5 large onshore and offshore petroleum exploration licenses in Namibia, as well as in Colombia's Middle Magdalena Basin. Its primary assets are held through its 49% interest in Inter Oil (Pty) Ltd (Inter Oil). Inter Oil is a private firm which indirectly holds a portfolio of offshore petroleum exploration license (PEL) including a 15% carried interest in PEL 87; a 10% carried interest in each of PELs 82 and 83, and a 10% carried interest in PEL 90. In addition, it holds private participation interests of 25% unconventional and 100% conventional in the potential hydrocarbon resources of the 43,158 acres Valle Medio Magdalena 37 (VMM-37) Block in Colombia. The Middle Magdalena Basin is located in north-central Colombia between the Central and Eastern Ranges of the Andes, covering an area of 34,000km2 (13,000m2).
The company remains committed to advancing its exploration efforts at the Orange Basin while also generating additional value for its stakeholders.
Sintana Energy (SEUSF), closed Friday's trading session at $0.2435, off by 1.0239%, on 74,983 volume. The average volume for the last 3 months is 96,045 and the stock's 52-week low/high is $0.073/$0.297.
ParcelPal Logistics (PTNYF)
We reported earlier on ParcelPal Logistics (PTNYF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
ParcelPal Logistics Inc. (OTCQB: PTNYF) (CNSX: PKG) (FRA: PT0A) is a courier and logistics firm engaged in the provision of last-mile delivery services and logistics solutions.
The firm has its headquarters in Vancouver, Canada and was incorporated in 1997, on March 10th by Dylan McRobert and Kelly Abbott. Prior to its name change in June 2021, the firm was known as ParcelPal Technology Inc. It operates as part of the integrated freight and logistics industry, under the industrials sector. The firm mainly serves consumers in Canada.
The enterprise’s mission is to be the world’s most convenient delivery service. It connects people and businesses through its network of couriers in major Canadian cities, including Vancouver, Calgary, and Toronto, as well as in the western region of the United States. Some of its verticals include pharmacy and health, meal kit deliveries, retail, groceries and more. It offers a range of solutions, including warehousing solutions, next day delivery, same day delivery and e-commerce integration. The enterprise provides logistic management services and supply chain solutions. Under warehousing solutions, it streamlines its transport products with warehousing to assist in meeting storage requirements. The enterprise offers delivery and logistics solutions for business to business, business to customer, and any other tailored creative solution its partners may require.
The company recently announced its latest financial results, which show another consecutive month of operating profitability. Its CEO noted that they remained focused on future growth in different geographic regions. This may open the company up to new growth and investment opportunities while also bringing in additional revenues.
ParcelPal Logistics (PTNYF), closed Friday's trading session at $0.0098, up 4.2553%, on 500 volume. The average volume for the last 3 months is 1,838 and the stock's 52-week low/high is $0.0055/$0.0263.
Graphano Energy (GELEF)
We reported earlier on Graphano Energy (GELEF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Graphano Energy Ltd (OTCQB: GELEF) (CVE: GEL) (FRA: 97G0) is a mining firm focused on the evaluation, acquisition and development of graphite resources.
The firm has its headquarters in Vancouver, Canada and was incorporated in 2020, on October 5th. It operates as part of the other industrial metals and mining industry, under the basic materials sector. The firm mainly serves consumers in Canada.
The enterprise’s mineral property includes Lac Aux Bouleaux Graphite Property (LAB Graphite Property). It intends to focus on exploration and development of the LAB Graphite Property. The LAB Graphite Property is made up of 14 mineral claims in one block totaling approximately 738.12 hectares of land on NTS 31J05, near the town of Mont-Laurier in southern Quebec. It lies adjacent to the south of TIMCAL’s Lac des Iles graphite mine in Quebec. The LAB Graphite property is situated under Precambrian age rocks of the Grenville Series consisting of quartzo feldspathic, garnetiferous paragneiss and limestone or marble beds. Paragneiss is fine to medium grained with a range of compositions, such as quartzo-feldspathic gneiss, biotite gneiss, biotite-garnet gneiss, biotite-garnet-silliminite gneiss and biotite-hornblende gneiss. The enterprise also holds an option agreement to acquire 100% interest in the Standard Graphite Mine comprising of 6 claims covering an area of approximately 355 hectares.
The company recently announced the commencement of drilling activity at its Standard Mine and LAB Graphite properties in an effort to propel its projects into production and help fortify international markets against the expected surge in demand driven by electric vehicles. This may, in turn, bring in addition revenues into the company while generating value for its shareholders.
Graphano Energy (GELEF), closed Friday's trading session at $0.08485, even for the day, on 53 volume. The average volume for the last 3 months is 833,785 and the stock's 52-week low/high is $0.021/$0.3659.
Stronghold Digital Mining Inc. (SDIG)
QualityStocks, RedChip, MarketBeat, SmallCapVoice, Real Pennies, InvestorPlace, The Online Investor, StocksEarning, StockPicksNYC, StockEarnings, OTC Markets Group, InsiderTrades and Early Bird reported earlier on Stronghold Digital Mining Inc. (SDIG), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The Financial Intelligence Unit (FIU) of India has asked the Ministry of Electronics and Information Technology to restrict the website URLs of nine cryptocurrency exchanges. The aim is to restrict access to platforms such as MEXC Global, Bitfinex, Bitstamp, Binance, Gate.io, Kraken, Bittrex, Huobi and Kucoin, citing their alleged “illegal operations” and noncompliance with the Money Laundering Prevention Act.
The FIU, serving as the central national agency, is tasked with receiving, processing, analyzing and disseminating information related to suspect financial transactions, including terror financing and money laundering, to enforcement agencies worldwide.
India has been actively working to integrate the cryptocurrency sector with its conventional financial landscape. In March, the country mandated that cryptocurrency companies acquire know-your-customer (KYC) data, requiring them to register with the agency. Any virtual asset service provider (VASP) operating in India, whether offshore or domestically, is obligated to register with the FIU as a reporting entity and adhere to the Prevention of Money Laundering Act.
While the FIU now has 31 firms registered, the agency noted that several offshore entities that serve a sizable percentage of Indian users were not abiding by the laws pertaining to antimoney laundering (AML) and counterfeiting (CFT). The FIU issued show-cause notifications to the nine blocked VASPs, though the specific repercussions beyond URL blocking remain unclear.
In August, India Prime Minister Narendra Modi emphasized the necessity of global cryptocurrency regulation, asserting that regulations and frameworks should extend beyond individual countries or regional groups. India, however, has displayed a cautious approach to crypto, introducing high capital profits taxes, imposing a substantial 30% tax — surpassing rates applied to equities and other investments — and a 1% tax deducted at source (TDS). Offenders or platforms failing to adhere to TDS may face penalties, including fines or imprisonment ranging from three months to seven years, under an amendment to Section 271C of the Income Tax Act.
In October, the Financial Action Task Force (FATF) advocated for member nations to implement a travel rule tracking crypto transactions to combat terrorist financing and illicit activities. India’s stance on enforcing the travel rule remains uncertain.
India tops the globe in the adoption of cryptocurrencies among the general public, according to a Chainalysis analysis, despite the country’s central bank governor dismissing cryptocurrencies as speculative gaming opportunities. Though there are legislative obstacles, the nation has the second-largest raw estimated transaction volume in the world for cryptocurrencies.
Established crypto companies such as Stronghold Digital Mining Inc. (NASDAQ: SDIG) hope that major jurisdictions around the world enact stable crypto laws so that industry actors can be certain about the regulatory environment in which they operate.
Stronghold Digital Mining Inc. (SDIG), closed Friday's trading session at $7.19, off by 3.6193%, on 1,625,204 volume. The average volume for the last 3 months is 540,279 and the stock's 52-week low/high is $3.46/$12.50.
Curaleaf Holdings Inc. (CURLF)
InvestorPlace, Kiplinger Today, MarketBeat, QualityStocks, Cabot Wealth, Daily Trade Alert, Top Pros' Top Picks, MarketClub Analysis, Profit Trends, The Online Investor, StreetInsider, Wealth Insider Alert, Early Bird, Trades Of The Day, Trading For Keeps, The Street, Investment U, Daily Profit, CFN Media Group, StreetAuthority Daily, Zacks, TradersPro, Wyatt Investment Research, wyatt research newsletter and Schaeffer's reported earlier on Curaleaf Holdings Inc. (CURLF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
A Canadian study involving high schoolers has found that cannabis legalization made it harder for minors to access the controversial plant. According to the newly published study, the portion of students who said they had easy access to marijuana has fallen recently.
This period of reduced minor access to cannabis also coincides with the Canadian government legalizing marijuana consumption for eligible adults and authorizing cannabis retail outlets across Canada. The study contradicts cannabis legalization opponents who claimed that removing criminal penalties for possessing and consuming cannabis would make it significantly easier for youth and young children to access cannabis.
Researchers used data from a multitopic student survey called the COMPASS Study to determine whether or not students have had increased access to cannabis post-legalization. The survey polled students from grades 9 to 12 in secondary schools in Alberta, Quebec, British Columbia and Ontario. The research team considered cannabis-related questions posted to the students during three separate time periods: 2018–19, 2019–20 and 2020–21.
The research team looked at the responses to the following two questions: “In the last 12 months, how often did you use marijuana or cannabis?” and “Do you think it would be difficult or easy for you to get marijuana if you wanted some?”
Team members discovered that the frequency among students who reported having easy access to cannabis dropped by 26.7% from 51.0% to 37.4% while rates of past-month cannabis use fell from 12.7% in the 2018–19 period to 7.5% in 2020–21, despite the country legalizing cannabis and launching legal sales.
Study participants were more likely to report gaining easier access to marijuana as they grew older, especially the participants who used cannabis as minors. Furthermore, the research team noted that students with a history of marijuana use had a higher likelihood of reporting having easy access to the drug post-legalization.
The initial coronavirus pandemic period “slightly impeded” ease of access to cannabis, but the issue went away as the pandemic continued.
Researchers say that while self-reported access to marijuana among minors has decreased since Canada legalized the drug and during the pandemic period, the chances of underage students reporting increased ease of access to cannabis rise as they grow older and advance through high school, especially among the third wave of participants in the most recent COMPASS survey data. Consequently, the researchers say, there is still a lot of room for improving cannabis control efforts and maximizing their impact to limit minor access to marijuana.
This research provides yet more evidence that legalizing marijuana and licensing companies such as Curaleaf Holdings Inc. (CSE: CURA) (OTCQX: CURLF) goes a long way in preventing minors from accessing the substance.
Curaleaf Holdings Inc. (CURLF), closed Friday's trading session at $4.66, up 6.8807%, on 886,624 volume. The average volume for the last 3 months is 600,482 and the stock's 52-week low/high is $2.19/$5.80.
Seelos Therapeutics Inc. (SEEL)
QualityStocks, MarketBeat, StockEarnings, StockMarketWatch, MarketClub Analysis, TradersPro, Schaeffer's, BUYINS.NET, Trades Of The Day, Prism MarketView and INO Market Report reported earlier on Seelos Therapeutics Inc. (SEEL), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The U.S. Food and Drug Administration (FDA) issued two warnings regarding compounded ketamine use on Feb. 16, 2022, and a year and a half later on Oct. 10, 2023. Ketamine is a dissociative anesthetic that is typically used to induce loss of consciousness and treat pain in both humans and animals. It has some hallucinogenic properties and is often called a dissociative anesthetic hallucinogen because it can make patients and recreational users feel detached from physical pain and the environment.
Ketamine can also induce amnesia, a sense of sedation and immobility, making it the drug of choice for bad actors looking to perpetuate sexual assault.
The FDA’s February 2022 warning was titled “FDA Alerts Health Care Professionals of Potential Risks Associated with Compounded Ketamine Nasal Spray,” while the 2023 warning was titles “FDA Warns Patients and Health Care Providers About Potential Risks Associated with Compounded Ketamine Products, Including Oral Formulations, for the Treatment of Psychiatric Disorders.”
While both warnings seem to cover a lot of the same ground, it seems the 2022 warning did not have the intended effect, necessitating the release of a second warning more than a year later. The federal agency noted that although it has approved a ketamine-containing nasal spray called esketamine (Spravato) for certain conditions, it warned that ketamine is not approved for psychiatric use.
Esketamine is currently part of the agency’s Risk Evaluation and Mitigation Strategy (REMS) program, which requires that the medication is “dispensed and administered in health care settings.” According to the FDA, esketamine cannot be provided outside of certified medical settings, and patients must remain under observation in a healthcare setting for at least two hours after they take the medication to ensure they are safe to leave.
This program is designed to house approved medications, such as Spravato, that can have severe side effects when taken outside of strict medical settings. As such, the FDA said, ketamine and compounded ketamine, which is ketamine with different dosage levels, “are not approved for the treatment of any psychiatric disorder.”
The FDA issued its first warning after it combed through April 2011 to January 2022 data from the FDA Adverse Event Reporting System and found five cases where compounded ketamine use was associated with dissociation, visual hallucinations, delusions, misuse and abuse.
Following this warning, the FDA issued another warning last October with some of the same information contained in the February 2022 warning. This warning noted that there was increased public interest in compounded ketamine as a treatment for behavioral health disorders, especially with the proliferation of telehealth providers that provide in-home ketamine services.
It stressed that “ketamine is not safe and effective” as a psychiatric disorder treatment and that the FDA wasn’t aware of any evidence suggesting that the dissociative psychedelic is safer, more effective or more fast acting than FDA-approved psychiatric treatments.
The concerns of the FDA support the position of startups such as Seelos Therapeutics Inc. (NASDAQ: SEEL) to the effect that psychedelic treatments should be used in only clinical settings where patients can be assessed prior to and after a treatment session to minimize the likelihood of adverse events.
Seelos Therapeutics Inc. (SEEL), closed Friday's trading session at $1.335, up 0.37594%, on 215,268 volume. The average volume for the last 3 months is 32.884M and the stock's 52-week low/high is $1.04/$49.80.
Lucid Motors (LCID)
Green Car Stocks, InvestorPlace, Schaeffer's, StockEarnings, QualityStocks, The Street, MarketBeat, Early Bird, MarketClub Analysis, Investopedia, StocksEarning, INO Market Report, GreenCarStocks, Kiplinger Today, The Online Investor, Daily Trade Alert, Money Wealth Matters, Trades Of The Day, Louis Navellier, The Wealth Report, The Night Owl, DividendStocks, Cabot Wealth, Green Energy Stocks, Wealth Whisperer, InsiderTrades, InvestorsUnderground, AllPennyStocks, Zacks, The Stock Dork, Smartmoneytrading and Top Pros’ Top Picks reported earlier on Lucid Motors (LCID), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Last year was a mixed bag of a year for the nascent electric vehicle industry. Major companies such as Tesla broke sales records while Chinese automakers made major inroads into international markets. However, surging interest rates and high prices depressed electric vehicle demand as 2023 drew to a close, forcing many automakers to reconsider their ambitious plans for electrification. Even so, 2023 was a significant year for the EV industry.
Carmakers began switching to Tesla’s proprietary connector in 2023 after the Texas-based automaker allowed other carmakers to adopt its charging connector. With the exception of Stellantis, every major carmaker has formalized the switch to Tesla’s North American Charging Standard (NACS). This move grants the majority of non-Tesla EVs access to Tesla’s massive network of supercharging stations and could help increase consumer confidence in EV charging.
Automakers and retailers got increasingly interested in EV charging. Both groups began to scale up their EV charging commitments in 2021 and 2022 with the aim of emulating Tesla’s wildly successful Supercharger Network. Mercedes-Benz will begin developing a fast-charging network in America while seven major automakers are funding a charging network dubbed We Charge that would rival the Tesla Supercharger Network in both size and scope.
General Motors also partnered with Pilot Travel Centers to open more than a dozen charging stations across 13 states, and convenience store chain 7-Eleven has plans to develop the largest public EV charging network in the country.
Electric vehicle tax credits were still a mess in 2023 amid a new price cap and household income requirements, an EV-leasing loophole and the planned shift to a dealer-based rebate system. Imprecise language in EPA consumer information and from the Department of Treasury also made it difficult for EV buyers to know if the cars they intended to purchase were eligible for tax credits.
The Treasury didn’t release full guidance on EV tax credits for 2024 until Dec. 1, 2023, leaving many buyers, automakers and even the EPA unsure of the EV models that qualified for tax credits.
EV prices finally dropped in 2023 after automakers spent a large portion of the previous year price-gouging electric cars. Tesla prices dropped by an average of 20% as the automaker cut prices for its Model 3 and Model Y. Coupled with an influx of vehicles into the secondhand market, prices for used Teslas also went down. Non-Tesla electric cars such as the Ford F-150 Lightning, Ford Mustang Mach-E, Lucid Air from Lucid Motors (NASDAQ: LCID) and the Hyundai Ioniq 6 also experienced notable price reductions.
Overambitious software rollouts nearly hindered EV adoption last year. Software has always been an integral component of the electric car, and EV software has made significant advancements in recent years. However, cases of software updates bricking infotainment systems and automakers temporarily halting EV deliveries or recalling hundreds of thousands of EVs due to software issues were rampant in 2023.
Lucid Motors (LCID), closed Friday's trading session at $3.78, off by 2.8278%, on 29,498,913 volume. The average volume for the last 3 months is 1.795M and the stock's 52-week low/high is $3.62/$17.81.
Cronos Group Inc. (CRON)
InvestorPlace, Schaeffer's, Kiplinger Today, MarketClub Analysis, The Street, StocksEarning, MarketBeat, Daily Trade Alert, Trades Of The Day, Wealth Insider Alert, The Online Investor, QualityStocks, Market Intelligence Center Alert, StockMarketWatch, StreetInsider, StockEarnings, BUYINS.NET, Zacks, The Wealth Report, Top Pros' Top Picks, Stock Up Featured, Investopedia, Cabot Wealth, Daily Profit, InvestmentHouse, Early Bird, InsiderTrades, Jim Cramer, The Rich Investor, InvestorsObserver Team, VectorVest, Money Morning, TheTradingReport, 24/7 Trader, Wall Street Window, Small Cap Firm, Stock Gumshoe and InvestorsUnderground reported earlier on Cronos Group Inc. (CRON), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The year 2023 witnessed a range of notable federal developments on the cannabis landscape. Among these were significant milestones such as the Biden administration’s proposal to reschedule cannabis, an expansion of pardons for federal cannabis possession offenses and setbacks in the congressional journey toward marijuana banking reform.
As Congress takes a recess for the holiday season, stakeholders and advocates are reflecting on the highs and lows of the past year, intending to advance their efforts in 2024.
A pivotal moment in 2023 was the U.S. Department of Health & Human Sciences recommending the reclassification of cannabis from Schedule I to Schedule III under the Controlled Substances Act (CSA). This recommendation, arising from President Biden’s directive, is undergoing the administrative process led by the U. S. Drug Enforcement Agency (DEA). If implemented, this move would symbolically acknowledge that marijuana is not a dangerous drug lacking medical value; it would also potentially ease research barriers and allow state-licensed businesses to claim federal tax deductions.
Biden also made headlines by extending and broadening pardons for individuals with federal cannabis possession offenses, aligning with his renewed focus on marijuana reform. However, these actions fall short of his initial campaign promises, which included federal legalization of medical marijuana, marijuana decriminalization, and preventing incarceration for nonviolent marijuana-related offenses.
Marijuana banking reform faced challenges in the Senate, with the Secure and Fair Enforcement Regulation (SAFER) Banking Bill stalling despite passing through the Banking Committee. Senate Majority Leader Chuck Schumer remains committed to advancing the bill in 2024, emphasizing the need for bipartisanship in a divided Congress.
Public support for cannabis legalization reached an all-time high of 70%, according to a Gallup poll released in November. This bipartisan support extended across states with varying marijuana legalization statuses, reflecting a growing consensus on the need for reform. In congressional legislation, various bipartisan bills were introduced in 2023, addressing different aspects of federal cannabis policy. Notable bills included the STATES 2.0 Act, the MORE Act and the HOPE Act, each proposing reforms ranging from ending federal prohibition in legal states to expunging prior marijuana convictions.
While multiple drug-policy reform amendments were blocked by the GOP-controlled Rules Committee, some amendments, such as allowing VA doctors to recommend medical marijuana to veterans, did find success in the House. In addition, several federal agencies engaged with marijuana-related issues in 2023. The DEA announced plans to increase domestic production of cannabinoids for research purposes while also proposing a federal ban on two psychedelic compounds. The U.S. Food & Drug Administration stirred controversy by refraining from regulating hemp-derived CBD in the food supply, and the VA reaffirmed its prohibition on doctors recommending medical cannabis to veterans.
The Justice Department faced legal challenges defending the federal ban preventing cannabis consumers from buying or possessing firearms. Despite court rulings deeming the ban unconstitutional, the government continues to appeal.
Looking ahead to 2024, the marijuana landscape remains uncertain. The DEA’s decision on cannabis scheduling and the fate of the banking bill in the Senate will be key focal points. With a presidential election year on the horizon, Biden may continue to emphasize his marijuana pardon actions, though the extent of his commitment to broader legalization remains to be seen. Despite the challenges, the momentum for marijuana reform persists, providing stakeholders and advocates with a compelling force for the future.
Established cannabis entities such as Cronos Group Inc. (NASDAQ: CRON) (TSX: CRON) will certainly take a keen interest on how the regulatory landscape evolves this year.
Cronos Group Inc. (CRON), closed Friday's trading session at $2.18, up 2.3474%, on 2,124,204 volume. The average volume for the last 3 months is 84,114 and the stock's 52-week low/high is $1.64/$2.64.
SOBR Safe Inc.’s (SOBR)
MarketClub Analysis, QualityStocks, Broad Street, StockWireNews, StockStreetWire, Small Cap Firm, RedChip, ProTrader, INO Market Report and Fierce Analyst reported earlier on SOBR Safe Inc.’s (SOBR), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
SOBR Safe (NASDAQ: SOBR) (“SOBRsafe”), a provider of next-generation transdermal alcohol detection solutions, today announced that current customer Oceanfront Recovery has expanded its use of SOBRcheck(TM) to its two inpatient treatment facilities. Laguna Beach, California-based Oceanfront previously installed SOBRcheck at its headquarters, and deployed SOBRsure(TM) for continuous client monitoring.
“We have had SOBRcheck installed at our headquarters since September, and it has been an invaluable tool for client compliance check-in,” said Oceanfront Recovery President and COO Keenen Diamond. “It empowers us to verify the absence of alcohol in a rapid, accurate way, replacing breathalyzers for our everyday use. Based on the success of this initial implementation, we are excited to roll the SOBRcheck technology out to our treatment centers and ensure safe and alcohol-free environments of care.”
To view the full press release, visit https://ibn.fm/QKQdm
About SOBRsafe(TM)
Alcohol misuse is the fourth leading cause of preventable death in America, and the seventh worldwide. Yet prevention and monitoring solutions have not kept pace with this epidemic. Legacy technologies are invasive and inefficient, unhygienic and unconnected. There has to be a better way. Enter SOBRsafe(TM), where advanced transdermal (touch-based) technology detects and instantaneously reports the presence of alcohol as emitted through a user’s skin – no breath, blood or urine sample is required. With a powerful backend data platform, SOBRsafe provides next-generation, passive detection technology for the behavioral health, transportation, oil and gas, judicial and consumer markets. The SOBRsafe technology is commercially available for access control (“SOBRcheck(TM)”), wearable use (“SOBRsure (TM)”) and licensing/white labeling. SOBRsafe is creating a culture of prevention and support. To learn more, visit www.SOBRsafe.com.
SOBR Safe Inc.’s (SOBR), closed Friday's trading session at $0.416999, off by 16.4331%, on 239,527 volume. The average volume for the last 3 months is 34,303 and the stock's 52-week low/high is $0.2004/$3.16.
The QualityStocks Company Corner
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF)
- D-Wave Quantum Inc. (NYSE: QBTS)
- Electronic Servitor Publication Network Inc. (OTCQB: XESP)
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF)
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI)
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF)
- Longeveron Inc. (NASDAQ: LGVN)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- InMed Pharmaceuticals Inc. (NASDAQ: INM)
- Freight Technologies Inc. (NASDAQ: FRGT)
- Astrotech Corp. (NASDAQ: ASTC)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF)
The QualityStocks Daily Newsletter would like to spotlight Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF).
Ucore, a company engaged in the exploration for and separation and scalable production of REEs in Canada and the US, is in the process of perfecting and ultimately commercializing the separation and purification of critical metals
This is in a move to slash reliance on Chinese rare earths technologies and its toxic by-products
The company has made incredible headway over the years and is on track to commercially separate U.S.-friendly sources of REES, using its RapidSX(TM) technology, by 2025
The commissioning of its RapidSX Demonstration plant in Kingston, Ontario, earlier this year affirms its commitment to actualizing the goal of re-establishing a North American rare earths supply chain
Ucore Rare Metals (TSX.V: UCU) (OTCQX: UURAF), a company engaged in the exploration for and separation and scalable production of rare earth elements ("REEs") in Canada and the U.S., understands the current state of the rare earths market, currently dominated by China. As a nation that has spent 30 years mastering solvent extraction, China has gotten the upper hand in the industry, leaving the West playing catch up in what appears to be a challenging position for players therein. Ucore, however, is seen as being ahead of the game, through having developed a more efficient processing technology for extracting REE products (https://ibn.fm/g4S5L).
Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) is a critical metals (“CM”) separation technology company executing an ESG-centered plan toward establishing a comprehensive North American critical metals supply chain. The company has developed a transformative commercial-ready technology, RapidSX™, for separating and purifying critical metals. Ucore intends to deploy this technology in pursuit of a CM supply chain independent of China for Western original equipment manufacturers (“OEMs”), most notably in the automotive and renewable energy industries.
Ucore’s vision is to become a leading advanced technology company providing best-in-class metal separation products and services to the mining and mineral extraction industry. Its initial focus is on processing heavy and light rare earth elements (“REEs”), disrupting a supply chain that is dominated by China.
China currently controls about 80% of the world’s access to REE mining projects and over 90% of the world’s REE processing capabilities, and it produces about 95% of the goods containing REE components.
Ucore is working to scale Western supply needs by establishing REE separation and rare earth oxide (“REO”) production capabilities in cooperation with strategic upstream supply and downstream offtake partnerships. The company, along with its industry partners, aims to unlock access to Western REEs for current consumer, energy, manufacturing and military sectors.
By 2025, Ucore expects to commercially separate U.S.-friendly sources of REEs and supply OEMs with REOs required to produce rare earth permanent magnets (“REPMs”) – the essential component of electric motors and generators required to support the world’s transition to electrification and sustainable energy sources.
The company intends to contribute to this initiative through the near-term development of a heavy and light rare-earth processing facility in Louisiana and subsequent development of Strategic Metals Complexes (SMCs) in Alaska and Canada, as well as through the longer-term development of its 100%-owned Heavy Rare Earth Element (HREE) mineral resource property at Bokan Mountain on Prince of Wales Island, Alaska.
Ucore is headquartered in Halifax, Nova Scotia.
Projects & Technology
RapidSX™ Demonstration Plant
The Kingston, Ontario, RapidSX™ Demonstration Plant commissioning process is underway. Once commissioned, the plant is designed to demonstrate the commercial capabilities of the RapidSX technology platform.

The RapidSX demo plant will show:
- The techno-economic advantages of the RapidSX technology platform
- The processing of tens of tons of heavy and light mixed rare earth element concentrates in a simulated production environment
- The platform’s ability to operate for thousands of semi-continuous run-time hours
- Production of high-purity NdPr, praseodymium, neodymium, terbium and dysprosium rare earth elements for early OEM product qualification trials
The demo plant is located within Ucore’s 5,000-square-foot RapidSX Commercialization and Demonstration Facility and is run by its laboratory partner, Kingston Process Metallurgy Inc. (“KPM”).
RapidSX™ Technology
Innovation Metals Corp., acquired by the company in 2020, developed the RapidSX separation technology platform with early-stage assistance from the United States Department of Defense, later resulting in the production of commercial-grade, separated rare earth elements at pilot scale.
RapidSX combines the time-proven chemistry of conventional solvent extraction (SX) with a new column-based platform that significantly reduces time to completion and plant footprint, as well as potentially lowering capital and operating costs. SX is the international REE industry’s standard commercial separation technology and is currently used by all REE producers worldwide for bulk commercial separation of both heavy and light REEs.
Utilizing similar chemistry to conventional SX, RapidSX is not a “new” technology, but it represents a significant improvement on the well-established, well-understood, proven conventional SX separation technology preferred by REE producers.
Strategic Metals Complex
Ucore, engineering partner Mech-Chem Associates Inc. and KPM are developing the full-scale engineering for the company’s first Strategic Metals Complex (SMC). The SMC is a planned REE separation and rare earth oxide production plant slated to commence construction in Louisiana in 2023. It is scheduled to initially process 2,000 tons of total rare earth oxides by the end of 2024, increasing to 5,000 tons in 2026.
The company has three initial U.S.-friendly feedstock agreements in place for the Louisiana complex, along with multiple developing offtake agreements. It received a C$16 million+ incentive package offer from Louisiana Economic Development to support construction of the SMC.
Bokan-Dotson Ridge REE Deposit

Ucore has invested over C$35 million to establish and validate the Bokan-Dotson Ridge resource in preparation for mine design and permitting. Initial drilling is complete, and a Preliminary Economic Assessment has been issued. Next steps for the project include a feasibility study, detailed mine design and permit acquisition. The project can be “near shovel ready” for construction in less than 30 months after receipt of the next stage of development funding.
Market Opportunity
According to a report by Grand View Research, the global rare earth elements market was valued at $2.8 billion in 2018 and is forecast to reach a value of $5.6 billion by 2025, achieving a CAGR of 10.4% during the period. Market growth is driven by increasing demand for these elements in the manufacturing of magnets and catalysts for the automotive industry. Rising demand for electric vehicles to reduce CO2 emissions is expected to propel the use of permanent magnets in the production of EV batteries.
China is the major producer and consumer of REEs. To maintain self-sufficiency and to meet future demand, China has been raising the export tariffs on rare earth elements shipped to various countries, including the U.S., Japan, India, Brazil and the European Union. This led to the current supply-demand gap in these countries, as they rely on imports from China.
China reduced the exports of REEs by 72% in the second half of 2010 to preserve its reserves of these elements and continues to export REEs at reduced levels, thereby affecting industries such as automotive, oil and gas, and electronics, which require an ample amount of rare earth elements.
Management Team
Pat Ryan, P.Eng., is Chairman and CEO of Ucore Rare Metals. He began as a director with the company when he developed a heightened interest in critical metals. Before joining Ucore, he founded and led a multimillion-dollar automotive OEM design and lean manufacturing company. His understanding of complex supply chains across international markets has led to a prime positioning as the global auto industry transitions to vehicle electrification. He holds a Bachelor of Engineering degree from Dalhousie University.
Peter Manuel is Vice President and CFO of Ucore. Prior to joining the company, he practiced as a Chartered Accountant for more than 17 years, providing consulting services to companies in a range of industries, with a focus on the financial services and resource sectors. He spent 10 years in England and Ireland providing assurance, strategic planning, corporate finance and other consulting services to a portfolio of both public and private entities. He holds a Bachelor of Commerce Degree from Dalhousie University.
Michael Schrider, MEng, P.E., is Vice President and COO of Ucore. He is a multidisciplinary engineer who has been involved in manufacturing, engineering and managing complex structural and mechanical systems projects since 1989. He was the Founder, President and Chief Engineer of Schrider & Associates and Alton Bay Design, both engineering services firms. He holds a bachelor’s degree in naval architecture and marine engineering from the University of New Orleans and a master’s degree in mining, geological and geophysical engineering from the University of Arizona.
Mark MacDonald is Vice President of Investor Relations at Ucore. He has over 25 years of experience implementing award winning business development and marketing programs at regional and national levels. As Vice President of Sales, he was responsible for Mediapro Communication’s growth as AT&T Canada’s leading B2B sales partner. He subsequently became Atlantic Regional Vice President of AT&T Canada Corp. He holds a Bachelor of Commerce degree from Dalhousie University.
Ucore Rare Metals Inc. (UURAF), closed Friday's trading session at $0.6636, off by 0.450045%, on 28,890 volume. The average volume for the last 3 months is 1.505M and the stock's 52-week low/high is $0.395/$1.15.
Recent News
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) the West’s Strategy to Weaken China’s Minerals Dominance with its RapidSX(TM) Technology
- InvestorNewsBreaks — Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Announces Acquisition of Louisiana Facility for Future REE Processing Plant
- InvestorNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Comments on China Ban on Export of REE Tech, Notes Company's REE Focus Is of 'Increasing Strategic Importance'
D-Wave Quantum Inc. (NYSE: QBTS)
The QualityStocks Daily Newsletter would like to spotlight D-Wave Quantum Inc. (NYSE: QBTS).
National Defense Authorization Act ("NDAA") signed into law on December 22, 2023
NDAA includes quantum pilot program dedicated to near-term quantum computing applications, which D-Wave has already been developing, and calls for consideration of all quantum technologies (annealing, hybrid and gate model)
Legislation promotes cultivating and strengthening relationships between the Department of Defense, academic institutions, small enterprises, and unconventional defense contractors
D-Wave Quantum (NYSE: QBTS), the world's first commercial supplier of quantum computers, software, and services, commends U.S. policymakers for including a groundbreaking quantum pilot program, dedicated to near-term quantum computing applications, as part of the National Defense Authorization Act ("NDAA") signed into law December 22, 2023 (https://ibn.fm/5teyZ). D-Wave Quantum Inc. (NYSE: QBTS) is a leader in quantum computing systems, software and services focused on delivering customer value via practical quantum applications for problems such as logistics, artificial intelligence, materials sciences, drug discovery, scheduling, fault detection and financial modeling. As the only provider building both annealing and gate-model quantum computers, the company is unlocking commercial use cases in optimization today, while building the technologies that will enable new solutions tomorrow. D-Wave is a pioneer in quantum computing, with a history of delivering the world’s first commercial quantum computer; the first real-time quantum cloud service; countless hardware and software product and research milestones; and the planned first cross-platform quantum solution which will deliver both annealing and gate-model quantum computers to customers via an integrated platform. Its current commercial product offerings include: Advantage™ (fifth generation quantum computer), Leap™ (quantum cloud service), Launch™ (quantum computing onboarding service) and Ocean™ (full suite of open-source programming tools). D-Wave’s relentless pursuit of practical quantum computing has resulted in the technology being used today by some of the world’s most advanced enterprises – more than 25 of the Forbes Global 2000 use D-Wave. D-Wave’s commercial customers include blue-chip industry leaders like Volkswagen, Accenture, BBVA, NEC Corporation, Save-On-Foods, DENSO and Lockheed Martin. The company boasts an extensive IP portfolio featuring more than 200 issued U.S. patents and over 100 peer-reviewed papers published in leading scientific journals. Founded in 1999, D-Wave is the world’s first commercial supplier of quantum computers. With headquarters and the Quantum Engineering Center of Excellence based near Vancouver, Canada, D-Wave’s U.S. operations are based in Palo Alto, California. Advantage™ Quantum Computer With the Advantage™ Quantum Computer, D-Wave has incorporated two decades of experience and over 10 years of customer feedback to create the first and only quantum computer designed for business. The platform features a new processor architecture with over 5,000 qubits and 15-way qubit connectivity. This is 2.5x more connections and more than double the number of qubits than the company’s previous generation quantum computer. D-Wave’s quantum computers, first located in its facilities in British Columbia, have been available to North American users through its Leap™ quantum cloud service since 2018. It has since introduced new Advantage systems in Julich, Germany, and most recently, Marina Del Rey, California, which marked the availability of the first Advantage quantum computer physically located in the United States. That new deployment is part of the USC-Lockheed Martin Quantum Computing Center (QCC) hosted at USC’s Information Sciences Institute (ISI), a unit of the University of Southern California’s prestigious Viterbi School of Engineering. Additionally, Amazon Web Services (AWS) and D-Wave announced that the U.S.-based system is available for use in Amazon 2racket, expanding the number to three different D-Wave quantum systems available to AWS users. Leap Quantum Cloud Service D-Wave’s customers interface with its systems through the Leap™ quantum cloud service. Leap delivers immediate, real-time access to the company’s Advantage quantum computer and quantum hybrid solver service, all with enterprise-class performance and scalability. Leap allows developers proficient in Python to get started building and running quantum applications. Through a seamless and secure cloud-based connection, users can easily start solving complex problems of up to 1 million variables and 100,000 constraints. Using Leap, D-Wave customers have developed quantum hybrid applications for use cases in manufacturing, logistics, financial services, life sciences, materials science, retail and transportation. By eliminating the need to wait hours, days or weeks to get good answers to a broad array of problems, D-Wave is helping businesses move forward. D-Wave Launch D-Wave Launch™ is the company’s onboarding platform aimed at helping businesses easily start their quantum journey. Through this program, D-Wave’s team of experts and partners aid enterprises in identifying best use cases for quantum and work with them to develop a proof of concept and production pilot. From there, the team coordinates with customers to get their hybrid quantum applications up and running, providing ongoing Leap quantum cloud access to ensure the application is operating smoothly and delivering real business value. Target Verticals While the potential applications for quantum computing are effectively limitless, D-Wave has identified a number of industry verticals as key areas of focus for its quantum architecture, providing case studies for each. These include: Market Opportunity The quantum computing total addressable market is projected to grow between $450 billion and $850 billion over the next 15 to 30 years, with between $5 billion and $10 billion of anticipated TAM growth coming in the next three to five years, according to Boston Consulting Group. Driving factors behind this growth include rising investments in quantum computing tech by governments and an increasing number of commercial use-cases. Forward-thinking organizations see quantum as an opportunity to move ahead of the competition. From finding efficiencies and reducing waste to decreasing time to solution and solving problems abandoned due to complexity, the business value is real. According to data from 451 Research, 40% of large enterprises are already experimenting with quantum computing. D-Wave is strategically positioned – in an industry with significant barriers to entry – as evident by a decades-long track record serving a roster of blue-chip customers. The company is singularly focused on helping its customers achieve clear value by leveraging quantum computing in practical business applications. With a full stack of systems, software, developer tools and services, D-Wave is working to enable enterprises, governments, developers and researchers to access the power of quantum computing, thereby providing an intriguing opportunity for prospective investors. D-Wave’s current investor base includes PSP Investments, Goldman Sachs, BDC Capital, NEC Corporation, Aegis Group Partners and In-Q-Tel. Leadership Team Dr. Alan Baratz has served as the CEO of D-Wave since 2020. Previously, as Executive Vice President of R&D and Chief Product Officer, he drove the development, delivery, and support of all of D-Wave’s products, technologies, and applications. Dr. Baratz has over 25 years of experience in product development and bringing new products to market at leading technology companies and software startups. As the first president of JavaSoft at Sun Microsystems, he oversaw the growth and adoption of the Java platform from its infancy to a robust platform supporting mission-critical applications in nearly 80 percent of Fortune 1000 companies. He has also held executive positions at Symphony, Avaya, Cisco, and IBM. Dr. Baratz holds a doctorate in computer science from the Massachusetts Institute of Technology. John Markovich is the company’s CFO. He brings to D-Wave over three decades of experience working with rapidly growing private and public technology companies across all stages of development. Mr. Markovich has directed the finance, accounting, tax, treasury, M&A, legal, operations, customer service, IR, HR, and IT functions for companies ranging from privately held pre-revenue startups to an NYSE-listed Fortune 500 multi-national company with over $1.2 billion in annual revenue. During his career, he has negotiated and closed over 150 debt, equity, M&A, and joint venture transactions exceeding $2.5 billion in value; over a dozen private placements; nearly a dozen M&A transactions; and several international joint ventures. Mr. Markovich holds a BS in Business from Miami University and an MBA from the Michigan State Graduate School of Business. D-Wave Quantum Inc. (NYSE: QBTS), closed Friday's trading session at $0.8213, off by 6.2657%, on 862,632 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.15/$.
Recent News
Electronic Servitor Publication Network Inc. (OTCQB: XESP)
The QualityStocks Daily Newsletter would like to spotlight Electronic Servitor Publication Network Inc. (OTCQB: XESP).
The Digital Engagement Engine(TM) allows companies to maintain complete control of their content while creating meaningful relationships with new customers and revenue streams
XESP focuses on three important digital elements to improve client outcomes – connecting with the audience, relating to the community, and influencing positive customer relationships
XESP plans to expand business development planning and communications through channel partners and consultants, and fully engage in efforts to create rapid expansion and adoption of its managed service product across multiple verticals
Electronic Servitor Publication Network (OTCQB: XESP), a digital engagement company, recently announced the finalization of its purchase of assets of PhiTech Management, LLC, a digital activation and engagement technology company. PhiTech provides customer and content workflow applications to connect people with customized content through dynamic content provisioning, creating relevant digital relationships (https://ibn.fm/w4lQ3).
Electronic Servitor Publication Network Inc. (OTCQB: XESP) is a digital engagement company offering a managed service which provides digital activation and engagement solutions to companies that seek to optimize their growth. Its managed service is powered by a proven, proprietary technology – the Digital Engagement Engine™. This technology provides intelligent interaction management, dynamic content provisioning, and a logic-driven workflow, which creates digital experiences that accelerate an audience from awareness to action – driving growth.
Electronic Servitor Publication Network’s services are designed to drive growth for both established and developing organizations. Through the optimization of digital interactions within current and new communities, the Digital Engagement Engine™ ensures that client content is relevant, reaches the right audience, and connects with the intended person at the right time.
The company calls it ‘Growth as a Service’.
Client implementation is nearly effortless, since the solution is completely managed by the Electronic Servitor Publication Network team. This business model allows clients to focus on their brands, core product offerings, and content creation, while the company manages the technology and outcome.
The company is headquartered in Minneapolis, Minnesota.
Technology
Electronic Servitor Publication Network’s Digital Engagement Engine™ utilizes a combination of automation, unique data management, and a modern workflow built on a microservices architecture to achieve greater reach and lift. Using sophisticated data analysis and smart technology, the Digital Engagement Engine™ provides companies with the ability to maintain complete control of their content while creating meaningful relationships with new customers and revenue streams.
The Digital Engagement Engine™ isn’t just another marketing or technology tool; it’s a way to develop real connections with target markets.
Market Outlook
According to a report by ReportLinker.com, an award-winning market research firm, the global customer engagement solutions market was estimated at $19.3 billion in 2022 and is forecast to grow to $32.2 billion by 2027, achieving a CAGR of 10.8% during the forecast period.
The report notes that these engagement solutions are vital to companies seeking to widen their customer bases, reduce customer churn rates and increase customer retention. These perceived benefits of customer engagement solutions are likely to drive their growing adoption around the globe during the forecast period, according to the report.
Management Team
Peter Hager is President and CEO of Electronic Servitor. He joined the company from Pointward Inc., a medtech customer engagement agency that provided solutions to drive market entry, growth, and commercialization for Fortune 500 health care brands and medtech startups. He has founded and managed multiple technology, professional services and medtech organizations throughout his career. Mr. Hager holds a bachelor’s degree from Macalester College in St. Paul, Minnesota, with concentrations in economics and psychology.
Jim Kellogg is CFO of Electronic Servitor. He has served as the principal of J. Kellogg & Company Inc., a business and tax consultant, since 2005. He has provided legal support to clients’ business valuations, business interruption and divorce property valuations. He has worked as a professional tax adviser since 1983. Mr. Kellogg obtained his JD with emphasis on taxation from Western State University College of Law and was certified as a financial planner by the College for Financial Planning in 1990.
Thomas (Denny) Spruce, RPh, is COO of Electronic Servitor. He oversees company infrastructure, regulatory reporting, and strategic partner relationships, among other roles and responsibilities. He joined the company in March 2022 and, since that time, has implemented foundational support processes, developed contractual relationships with service providers, managed financial and regulatory reporting and overseen contract development and management with the legal team. Mr. Spruce obtained a BS in Pharmacy from the University of Arkansas.
Electronic Servitor Publication Network Inc. (XESP), closed Friday's trading session at $0.128325, off by 25.6087%, on 6,111 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.05/$0.22.
Recent News
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - Electronic Servitor Publication Network Inc. (XESP) Finalizes PhiTech Management, LLC Asset Purchase Including Proprietary Digital Engagement Engine(TM)
- InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Completes Pointward Inc. Merger, Adds to 'Formidable' Portfolio
- InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Acquires Assets of PhiTech Management LLC
Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF)
The QualityStocks Daily Newsletter would like to spotlight Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) .
After seeing its prices rise significantly in late 2023, gold seems to be maintaining its upward trajectory in 2024. The precious metal surged in value significantly as a confluence of global factors increased its safe-haven appeal and drew tons of investors looking to protect their portfolios from potential economic disaster.
With several armed conflicts raging on across the world, U.S. Treasury Yields dropping to their lowest levels since August, gold prices have surged to historic highs in recent weeks. Gold closed December's opening trading day at $2,071 after the U.S. Fed chair addressed a college in Atlanta and went against investor expectations, breaking a price record set in August 2020. While the market expected the Fed chair to go against loosening financial conditions, he instead said that the Fed's monetary policy was "well into restrictive territory." Exploration companies such as Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) could become emboldened from the bullish outlook for gold over the coming months and years.
Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) is a leading gold explorer in the Guiana Shield, South America. In early 2021, the Company announced an exciting new greenfield gold discovery at its Oko West project in Guyana, where, after 22 months of resource definition drilling, the Company has announced an initial Mineral Resource Estimate (MRE) containing 2.475 Moz of gold in Indicated resources at 1.84 g/t and 1.762 Moz of gold in inferred resources at 2.02 g/t contained within a pit shell outline. Preliminary metallurgy results performed by the company, consisting of 8 bottle roll tests obtained strong results, averaging just under 90% recoveries on average. The Company is continuing with additional development activities at Oko West, including environmental base line studies and additional metallurgical work relating to the delivery of a PEA by year end 2023. In addition, Reunion Gold is currently exploring several priority targets in the Oko West project area on which the company feels there is good potential to add additional resource ounces. This includes the opportunity to grow the initial mineral resource estimate (MRE) released on June 13, 2023, and to discover additional gold ounces at Oko West outside of the resource area.
The Guiana Shield remains one of the most prospective exploration locations globally for the discovery of world class orogenic gold deposits. The shield, including both Guyana and Surinam, contain large relatively underexplored greenstone belts, from which Reunion Gold expects many more significant gold discoveries could emerge in the coming years.
Oko West Project
Reunion Gold’s Oko West Project is a brand-new gold discovery in northwest Guyana located within the historical Oko gold district. Alluvial gold has been mined from the Oko district since the turn of the century, but very little primary gold has been mined or even explored for to the best of the company’s knowledge. The project comprises a prospecting license with an area of approximately 44 square kilometers and is 100% held by Reunion’s Guyanese subsidiary.
In 2020, Reunion Gold’s geochemical survey, trenching and initial 1000 m drill program discovered and confirmed the presence of gold mineralization in this Orogenic gold system. The gold occurs in the eastern edge of the project area, along a 6km long sheared contact between a granitoid intrusion and a meta volcanic-meta sedimentary rock package. The MRE is located within the Kairuni zone, which represents the northern most 1.9 km of the 6 km long contact.
“We are advancing our Oko West project along two tracks. The first is to advance the exploration programs outside of the Kairuni zone, aimed at outlining and discovering additional gold mineralization within our Prospecting License. On this front, I am very excited by the results from the initial Scout RC Geochem drill program that is defining new targets west of our Kairuni zone,” Rick Howes, President and CEO of Reunion Gold, stated in a recent news release. In addition to the targets west of the Kairuni zone, the company has also commenced exploration work on the southern ~ 4 km of the same sheared contact that hosts the Kairuni zone MRE. In addition, the company feels that the MRE marks the size of the Kairuni resource at a point in time and that there is good potential to continue to grow the resource. The MRE remains open at depth below the resource pit outline in the block 4 area and also to depth and along strike in the block 5 and 6 areas. In addition to the exploration programs, the second strategic track is to rapidly advance the Kairuni zone along the path to development. To that end the company is moving forward with the engineering and other studies, including more detailed metallurgical studies, that will support the release a PEA on the Kairuni zone by year end 2023 The company feels that the rapid advancement of development of Kairuni zone MRE, while in parallel continuing to explore for additional ounces on the project, is the best path to try and maximize shareholder value in the shortest period of time.
Guyana
Guyana boasts a long history of mining gold, bauxite, diamonds and manganese. Still, the greenstone belts of the Guiana Shield remain relatively unexplored when compared to the analogous regions of the West African Shield (Birimian), which, according to geological evidence, was once connected to the Guiana Shield, forming a contiguous craton prior to the Mesozoic period.
Despite a historical lack of accessibility and low exploration intensity, several significant large-scale projects have emerged in the Guiana Shield, including Aurora, Oko West, Oko Main, Toroparu and Omai. Guyana is English speaking with a British based parliamentary and legal system and boasts the world’s fastest growing economy on the back of significant offshore oil discoveries by Exxon and its partners. It is expected that a significant amount of the revenues from oil production will be invested in improving the infrastructure, education and health care and agriculture within the country.
Market Opportunity
The World Gold Council, an industry association representing gold producers with hundreds of mining operations in nearly 50 countries around the world, called 2022 the “strongest year for gold demand in over a decade.” Annual gold demand jumped 18% YoY due to “colossal central bank purchases, aided by vigorous retail investor buying and slower ETF outflows.”
Despite this spike in demand, total annual gold supply increased by just 2% in 2022, halting two years of successive declines but failing to challenge 2018 highs. This supply-demand imbalance could provide a favorable market environment as Reunion Gold continues to advance drilling programs at its 100%-owned Oko West Project.
Management Team
Successful exploration and the discovery of significant deposits in any given region require immense amounts of local knowledge and experience. This is the principle around which Reunion Gold has built its management team. In total, the company’s leadership boasts over 225 years of combined experience in the Guiana Shield.
David Fennell is the Executive Chairman of Reunion Gold, a position he has held since the company’s inception in 2003. He has 40 years of experience in the mining industry. He received a law degree from the University of Alberta in 1979 and practiced law until he founded Golden Star Resources Ltd. in 1983. During his term as President and CEO, Golden Star became one of the largest and most successful exploration companies. While at Golden Star, he was instrumental in the discovery and development of the Omai Gold Mine in Guyana and the Gross Rosebel Mine in Suriname. In 1998, Mr. Fennell became Chairman and CEO of Hope Bay Gold Corporation. He held this position through the merger of Hope Bay and Miramar Mining Corporation and remained as Executive Vice-Chairman and Director for the combined entity until its takeover by Newmont Mining Corporation in 2008. Mr. Fennell is currently a member of the board of directors of G Mining Ventures Corp. and Sabina Gold & Silver Corp.
Rick Howes, P.Eng., is the company’s President and CEO. He is a seasoned mining executive with over 39 years of experience in the mining industry, most recently as CEO of Dundee Precious Metals. Mr. Howes has extensive operating, technical and project development experience in both underground and open pit mines throughout Canada and internationally. In 2009, Mr. Howes joined Dundee Precious Metals, where, as VP and General Manager, he led the transformation of the Chelopech Mine in Bulgaria to reach world-class levels of performance. He became COO in 2011 and oversaw several significant growth capital development projects, including the expansion of the Chelopech Mine, the upgrade and expansion of the Tsumeb Smelter in Namibia and the development of the greenfield Ada Tepe open pit gold mine in Bulgaria. He was appointed CEO in April 2013, leading Dundee’s transformation from a junior gold producer to a multi-asset mid-tier gold producer generating strong free cashflow and solid returns to shareholders. Mr. Howes has been recognized as a visionary leader in mining, organizational innovation and transformation and was recognized as the Outstanding Innovator of 2016 by the International Mining Technology Hall of Fame.
Alain Krushnisky is the CFO of Reunion Gold. He brings to the company years of experience in the mining sector, including 10 years with Cambior Inc. (now IAMGOLD) in capacities such as Vice-President and Controller. Since 2004, Mr. Krushnisky has been doing consulting work for various publicly listed exploration and mining companies. He graduated from the University of Ottawa in 1983 with a bachelor’s degree in commerce and is a Chartered Professional Accountant.
Justin van der Toorn is the company’s VP Exploration. He is an exploration geologist with 18 years’ experience in the minerals industry, leading and managing exploration teams from grassroots activities through to discovery and resource definition drilling. Mr. van der Toorn’s previous experience has been in a range of commodity and deposit styles, including extensive work in Carlin-style gold, low- and high-sulphidation epithermal, porphyry and orogenic gold systems. He holds an MSci degree in Geological Sciences from the Royal School of Mines, Imperial College London. He is registered as a Chartered Geologist (CGeol) of the Geological Society, and a European Geologist (EurGeol) by the European Federation of Geologists.
Doug Flegg is the company’s business Development advisor. Doug has over 35 years’ experience in mining and mining finance with senior positions in research, portfolio management and global equity sales. Previously, Mr. Flegg was Managing Director Global Mining Sales with BMO Capital Markets where he was involved in raising $35 billion in over 200 corporate financings. Since 2016 he has been providing business development, strategic, and financing advice to corporate mining clients. Mr. Flegg also has a B.Sc. in Geology, work experience as a geologist and an MBA from Queens University.
Reunion Gold Corp. (OTCQX: RGDFF), closed Friday's trading session at $0.299, even for the day, on 431,000 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.2123/$0.46.
Recent News
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - As 2024 Gets Underway, Gold Seems Set on Upward Trajectory
- InvestorNewsBreaks - Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) Announces Settlement Agreement over Termination of Strategic Alliance
- Reunion Gold Announces Settlement Agreement With Barrick Gold Corporation
Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI)
The QualityStocks Daily Newsletter would like to spotlight Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI).
Scinai Immunotherapeutics (NASDAQ: SCNI), a biopharmaceutical company focused both on development of inflammation and immunology ("I&I") biological therapeutic products and provision of boutique CDMO services, has closed on its previously announced exercise of certain outstanding warrants. The exercise involved the purchase of up to 2,606,552 American Depositary Shares ("ADSs"), with each share representing 400 ordinary shares. According to the announcement, the shares had exercise prices ranging from $1.16 to $5 with terms ranging from five and one-half years to three years. The announcement noted that in consideration for the immediate exercise of the warrants for cash, Scinai issued new unregistered warrants to purchase up to 5,213,104 ADSs. With an exercise price of $0.65 per ADS, the new warrants are immediately exercisable and have a term of exercise equal to three years or five and one-half years from the date of issuance. The exercise of warrants resulted in an estimated $1.69 million in gross proceeds for Scinai. The company anticipates using the funds from the offering for general working capital, R&D and general corporate purposes. H.C. Wainwright & Co. acted as the exclusive placement agent for the offering.
To view the full press release, visit https://ibn.fm/PLrJH
Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) is a biotechnology company focused on developing, manufacturing, and commercializing innovative immunotherapeutic products primarily for the treatment of infectious diseases and autoimmune diseases.
In collaboration with the prestigious Max Planck Institute for Multidisciplinary Sciences (MPG) and the University Medical Center Göttingen (UMG), both in Germany, Scinai is developing a pipeline of innovative nanosized antibody (NanoAb) therapies addressing diseases underserved by current treatments and with large and growing markets, such as COVID-19, asthma and psoriasis.
NanoAbs, also known as VHH-antibodies or Nanobodies, are alpaca-derived nanosized antibodies that exhibit multiple significant competitive advantages over existing antibody therapies, including stability at high temperatures, superior binding affinity, more effective and convenient routes of administration and efficient production. Scinai is uniquely positioned to advance nanosized antibody innovation from R&D through commercialization.
The company’s highly experienced and successful pharmaceutical industry leadership team includes former senior executives from Novartis, GSK and Bristol-Myers Squibb.
Since its founding, Scinai has executed eight clinical trials, including a seven-country, 12,400-participant Phase 3 trial of a prior influenza vaccine candidate, and it built, owns and operates a 20,000 sq. ft. state-of-the-art GMP biologics manufacturing facility housing its laboratories, production facilities and offices.
Lead Candidate: Inhaled COVID-19 NanoAb
In December 2021, Scinai signed definitive agreements with the Max Planck Society – parent organization of the Max Planck Institute for Multidisciplinary Sciences– and the UMG to enter a strategic collaboration for the development and commercialization of innovative COVID-19 NanoAbs.
The company is planning a rapid development path that leverages its expertise and capabilities in biological drug development and manufacturing. Scinai anticipates preclinical proof-of-concept results for an inhaled COVID-19 NanoAb by the end of 2022, with initial Phase 1/2a human clinical trial results expected in 2023.
The intended inhaled mechanism of delivery of Scinai’s COVID-19 NanoAb formulation may serve as a significant differentiator when compared to approved monoclonal antibodies, which are injected. Inhaled delivery has shown to be cheaper, more convenient and likely safer for patients and providers.
NanoAb Pipeline: Psoriasis, Asthma and More
The COVID-19 NanoAb development agreement is part of a broader five-year research collaboration agreement signed in March 2022 covering discovery, development and commercialization of NanoAbs for several other disease indications with large market medical needs, including asthma, psoriasis, macular degeneration and psoriatic arthritis.
Scinai has an exclusive worldwide license for development and commercialization of COVID-19 NanoAbs and exclusive options for similar worldwide licenses for NanoAbs for the above mentioned additional large market disorders currently underserved by approved therapeutic antibodies.
Academic research teams from MPG and UMG have verified strong affinity by the new NanoAbs to their biological target molecules and high thermostability. They have also demonstrated strong neutralization by several NanoAb candidates of their respective target molecules. Neutralization studies of the other NanoAbs are expected to begin later in 2022.
Based on the promising results, Scinai will focus development efforts beginning with the following NanoAbs:
- NanoAbs targeting IL-17 as drug candidates for the potential treatment of psoriasis and psoriatic arthritis
- NanoAbs targeting IL-13 and NanoAbs targeting TSLP as drug candidates for the potential treatment of asthma
These are conditions for which the antibody target is validated by existing treatments and the mechanism of action is well understood. Both represent large medical needs and growing markets. Scinai anticipates preclinical proof-of-concept for at least one of these NanoAbs in 2023. This is in addition to the aforementioned human clinical Phase 1/2a for the inhaled COVID-19 NanoAb therapy, which is also anticipated in 2023.
CDMO Services
While NanoAb pipeline development is Scinai’s core focus, the company also offers its cGMP manufacturing facility, aseptic fill and finish suite, laboratories and experienced professionals for contract development and manufacturing organization (CDMO) services. This offering is designed to keep the Scinai team abreast of the latest industry developments and trends while building experience and generating revenue to support the company’s NanoAb pipeline development.
Market Opportunity
COVID-19 treatment, target of the company’s lead NanoAb therapy candidate, had an estimated market size of $22 billion in 2021.
Future Scinai drug candidates will target conditions with large markets growing at attractive CAGRs.
The global asthma treatment market was valued at $18.08 billion in 2019 and is projected to reach $26.01 billion by 2027, exhibiting a CAGR of 4.5% during the forecast period, according to Fortune Business Insights. The research firm predicts that the global psoriasis treatment market will grow from $26.37 billion in 2022 to $47.24 billion by 2029, exhibiting a CAGR of 8.7% over the forecast period.
Management Team
Amir Reichman is Scinai’s CEO. He previously was Head of Global Vaccines Engineering Core Technologies at GSK Vaccines in Belgium. Prior to that, he held leadership roles at Novartis Vaccines’ Global Vaccines Supply Chain Management organization. He was the first employee of NeuroDerm Ltd., a company focused on transdermal drug delivery, and served as Senior Scientist until his departure in 2009. He earned a M.Sc. in Biotechnology Engineering from Ben-Gurion University and an MBA in Finance and Health Care Management from the University of Pennsylvania’s Wharton School.
Tamar Ben-Yedidia, Ph.D., is Chief Science Officer at Scinai. She has more than 30 years of experience in immunology, with specific expertise in the development of vaccines. She began her career with Biotechnology General Ltd., working on development of a recombinant Hepatitis-B vaccine. She later joined the Weizmann Institute of Science, working on the design of a peptide-based vaccine against several pathogens. She is widely published, with numerous refereed articles and invited reviews in various scientific journals. She received her Ph.D. from the Weizmann Institute.
Elad Mark is COO at Scinai. He has over 15 years of biotechnology industry experience encompassing diverse project stages including feasibility studies, conceptual and detailed design, commissioning, qualification and process validation. Prior to joining Scinai, he led Novartis’s $800 million investment in a biologics facility in Singapore. With Biopharmax and Antero, both global pharmaceutical engineering companies, he successfully led projects in Israel, China and Singapore. He holds a BSc. in Engineering from the Afeka Tel Aviv Academic College of Engineering and an MBA from the Open University of Israel.
Uri Ben-Or is CFO at Scinai. He has served as CFO with public life science companies traded on the TASE, OTC and Nasdaq. Ben-Or provides his services to Scinai through CFO Direct, a company he founded and for which he serves as CEO. He served as the VP of Finance of Glycominds, a leading biotechnology company, and as CFO of a spin-off from Telrad Networks. He also served as a Corporate Controller at Menorah Capital Markets and as an Auditor at PWC. He holds a B.A. in Business from the College of Administration, an MBA from Bar-Ilan University, and is a CPA.
Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI), closed Friday's trading session at $0.58, off by 5.8442%, on 27,442 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.501/$3.60.
Recent News
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks – Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Closes on $1.69M Exercise of Outstanding Warrants
- InvestorNewsBreaks – Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) CEO Releases Shareholder Letter
- Shining Spotlight on How Vitiligo Could Be Connected to Other Autoimmune Disorders
Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF)
The QualityStocks Daily Newsletter would like to spotlight Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) .
Prospera (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) has announced that 2023 was a highly successful year for the restructured company. Among the highlights, PEI reportedly drilled, completed and tied-in nine horizontal wells in heavy oil reservoir with initial production rates exceeding expectations at 100+bpd per well. The ninth well was rig released on Dec. 24 and was brought online on Dec. 28. Prospera also drilled a directional well accessing medium-light oil flowing at 500+ Bpd. The company attained gross peak rates of 1,800 boepd prior to year-end, excluding the production of the recently drilled and completed horizontals, and the shut-in production for drilling. This continued production resulted in a monthly average of approximately 1,300 gross boepd. Currently, Prospera's total production capability is 2,200 boepd that will be optimized in the next few weeks barring any extreme winter weather conditions. In addition, the company was effectively able to fund the 2023 development plan with minimal dilution. PEI's future development goals are substantiated and encouraged by the 2023 development drilling and production results. Therefore, Prospera is proposing robust development and acquisition plans to attain year end production rates of 5,000 boepd in 2024. Further, PEI intends to pilot and implement enhanced oil recovery ("EOR") methods and add to existing production levels. The strategic acquisitions are to expand the core reserve base and diversify the product mix. Prospera will facilitate funding for its 2024 development program through cash flow generated from existing operations, debt and equity financing through private placement.
To view the full press release, visit https://ibn.fm/xqP3B
Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) is a public oil and gas exploration, exploitation and development company focusing on conventional oil and gas reservoirs in Western Canada. The company uses its experience to develop, acquire and drill assets with potential for primary and secondary recovery.
Prospera is primarily focused on optimizing hydrocarbon recovery from legacy fields through environmentally safe and efficient reservoir development methods and production practices. It is in the midst of a three-stage restructuring process aimed at prioritizing cost effective operations while appreciating production capacity and reducing liabilities.
The company is based in Calgary, Alberta, Canada.
Operations
Prospera’s core properties include more than 42,000 cumulative acres across Cuthbert, Luseland and Heart Hills in Saskatchewan and Red Earth and Pouce Coupe in Alberta. In total, the company estimates that there are half a billion barrels of oil in place at these sites accounting for 20+ years of forward project lifespan, with as little as 8% of total reserves having been recovered via legacy vertical well technology.
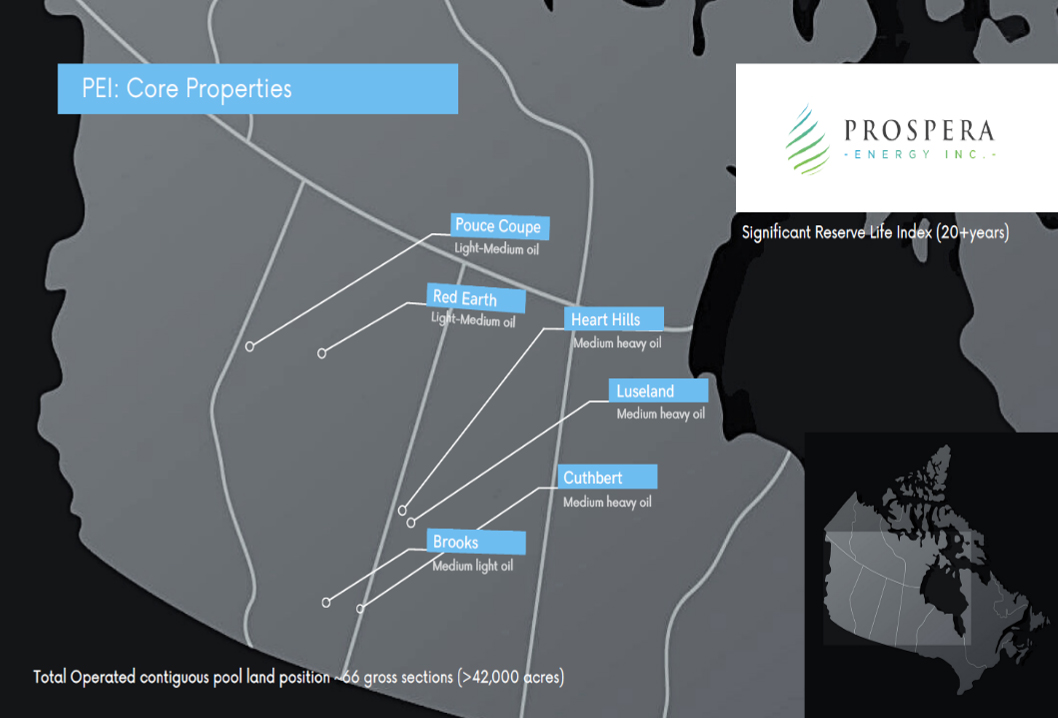
Restructuring Initiative
In 2021, Prospera enacted a top-down reorganization. The early results of these efforts were on display in May 2023, when the company reported a three-fold year-over-year increase in annual revenue for 2022 alongside drastically reduced operating costs and record-high cash flow from operations.
Prospera noted in the news release that it has positioned itself in 2023 to execute the second phase of its development plan aimed at increasing production through medium-oil development in Alberta and leveraging horizontal wells to capture the significant remaining reserves in Saskatchewan.
During the company’s investor summit in August 2023, Prospera CEO Samuel David provided more information regarding this three-phase strategy:
Phase I
Prospera completed the first phase of its restructuring by optimizing operations at its existing assets and addressing legacy arrears and non-compliance issues.
At the beginning of this transformation, the company was producing just 80 barrels of oil equivalent (BOE) per day. In Q4 of last year, Prospera peaked at nearly 1,200 BOE per day. Its breakeven is around 500 barrels per day, illustrating the opportunity for free cash flow. This prospect has driven Prospera’s capital development and optimization in recent quarters.
After a temporary slowdown in production due to harsh winter conditions, Prospera is currently producing about 800 BOE per day and anticipates an additional 300-500 barrels of daily production following the completion of ongoing site maintenance work.
This sustained increase has pushed the company’s NPV from roughly $3 million prior to the restructuring efforts to approximately $72 million today.
In an effort to build on this progress and maximize its available resources, Prospera piloted two horizontal reentries to assess a potential horizontal well transformation at its properties.
Phase II
Following up on the optimization efforts of Phase I, Prospera aims to commence a horizontal well transformation at its properties in the coming months. Based on its pilot wells from Phase I, the company has proposed 10 horizontal well locations at its Cuthbert and Heart Hills properties.
Prospera has likewise proposed eight medium light oil direction wells at its Alberta property, and it is exploring strategic acquisitions aimed at expanding its core area and diversifying its product mix.
Other facets of Phase II include piloting an enhanced oil recovery (EOR) application and continuing to execute its liability management goals and ESG initiatives. Prospera has already abandoned 60 vertical wells as part of its three-year LMR plan to reclaim surface land and reduce the environmental footprint of its operations.
Phase III
Beginning next year, Phase III of Prospera’s corporate redevelopment strategy will focus on continuing the company’s horizontal modular development to appreciate production and optimize recovery of remaining reserves. Prospera intends to implement full-scale EOR applications based on the results of its Phase II pilot program, which is forecast to optimize recovery by greater than 10%.
Prospera also intends to continue its acquisition strategy to diversify its product mix. Its goal, as detailed by in August 2023 investor summit, is to attain 50% light oil, 40% heavy oil and 10% gas – all while continuing to eliminate carbon emissions as part of its existing ESG initiatives.
Poised for Growth
Following its transformational efforts in 2022, Prospera is poised to achieve record growth in 2023. The company has forecast significant reductions in production costs through 2024, alongside sizable increases in daily production.
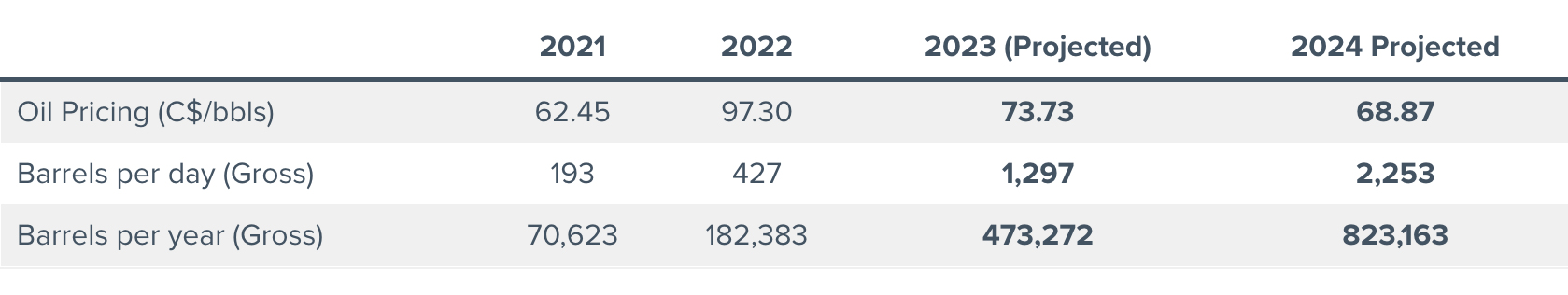
Prospera is currently exploring strategic acquisition targets to potentially increase its production beyond 5,000 BPD while expanding its reserve base to a billion barrels.
Market Opportunity
While the oil and gas industry faces long-term geopolitical and macroeconomic uncertainty, there is a clear trend to secure supply in the short term. According to Deloitte, the global upstream industry ended 2022 with some of the highest free cash flows on record, driving reinvestment in hydrocarbons and overall investment in clean energy.
The Energy Information Administration recently forecast a dip in global oil inventories over each of the next five quarters, placing upward pressure on oil prices. The agency further forecasts a YoY increase in fuel consumption, exacerbating the effects of OPEC+ production cuts that are set to remain in place through 2024.
For Prospera, these forecasts are promising. The company aims to build on its recent financial growth in the coming months (Prospera reported a three-fold YoY increase in revenue to $13.9 million in 2022), hitting a projected $57 million in total revenue by the end of 2024 while working to expand its core area holdings through accretive M&A transactions.
Leadership Team
Prospera is led by a team with extensive, diverse petroleum industry experience spanning both reservoir management and operations of oil and gas assets. The team boasts a proven track record of reorganizing companies, structuring financing arrangements and positioning for growth.
Samuel David is the company’s President and CEO. He brings to Prospera over 32 years of experience in operation, development and management of oil and gas assets and companies. Mr. David holds a B.Sc. in Mechanical Engineering and a B.A. in Economics from the University of Calgary. His background consists of both engineering and executive management experience with majors Petro-Canada, AEC Oil & Gas (now EnCana / Cenovus) and Husky Energy, as well as founding and operating juniors Ventura Energy and First West Petroleum. Mr. David has proven expertise in corporate planning, production, reservoir engineering, depletion strategies, EOR, property evaluations, acquisitions and divestitures.
George Magarian is VP Subsurface for Prospera. He is a professional petroleum geologist (APEGA) with over 36 years’ experience in the Western Canada Sedimentary Basin. After graduating with an Honors B.Sc. degree in Earth Science from the University of Waterloo, Mr. Magarian spearheaded many successful exploration programs, conducted evaluations for improved recovery schemes and assessed/exploited unconventional oil reservoir opportunities. He has held roles of increasing responsibility, from exploration geologist at oil industry major Petro-Canada and intermediates Anderson Exploration and Jordan Petroleum, to geoscience manager and VP exploration at junior companies Ionic Energy, Gentry Resources and Westfire Energy.
Chris Ludtke is the company’s VP Finance & Accounting. He is a high functioning finance leader with extensive expertise in finance, budgets and planning, accounting, economic evaluation, management, governance and sound decision making. Mr. Ludtke has 20 years of experience within the oil and gas, clean energy and renewables industries, including 12+ years working for Husky Energy before moving into an executive role in the junior oil and gas and hydrogen space. He graduated from the University of Lethbridge (Bachelor of Management) and is a Chartered Professional Accountant in the Province of Alberta.
Matthew Kenna is the CFO of Prospera. He has over 30 years’ experience leading organizations and helping them expand, drive efficiencies and grow profitability. Mr. Kenna is a professional accountant (CPA, CMA) and spent 15 years heading up the financial and operating departments at KUDU Industries, where he fostered financing arrangements, client relationships and manufacturing teams to take the organization from $35M to $150M in revenue. He has extensive experience turning companies around, growing them and building efficient organizations.
Prospera Energy Inc. (OTC: GXRFF), closed Friday's trading session at $0.07, up 7.8915%, on 77,220 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.056/$0.134.
Recent News
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks – Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Recaps Successful Year, Eyes Robust 2024 Development Plans
- MiningNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Looking to Triple Daily Crude Output with Launch of Second Phase of Drilling Program
- InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Releases Per-Day Production Report
Longeveron Inc. (NASDAQ: LGVN)
The QualityStocks Daily Newsletter would like to spotlight Longeveron Inc. (NASDAQ: LGVN) .
Last year was a major year for Alzheimer's disease research. After spending decades studying the debilitating neurological condition, 2023 saw scientists make significant advancements that opened the door to an entirely new era of Alzheimer's treatments.
The condition affects an estimated 6.7 million Americans and is projected to affect close to 13 million Americans by mid-century. It typically affects people older than 65 years of age and compromises an individual's thinking, memory, judgment, problem-solving, language and personality as it progresses.
The clinical trials being conducted by numerous enterprises such as Longeveron Inc. (NASDAQ: LGVN) could yield breakthroughs that slow the progression of Alzheimer's or even reverse its symptoms so that patients can have a new lease of life.
Longeveron Inc. (NASDAQ: LGVN) is a clinical-stage biotechnology company developing regenerative medicines to address unmet medical needs for specific aging-related and life-threatening conditions. The Company’s research and therapies are aimed at improving the outcome of infants born with a life-threatening heart condition, as well as improving the healthspan for the aging population – the number of years a person is expected to live in relatively good health, free of chronic disease and disabilities of aging, with function and ability to perform activities of daily living.
Longeveron is involved in clinical trials in the following indications: Hypoplastic left heart syndrome (HLHS), Alzheimer’s disease, and Aging-related Frailty.
The Company’s philosophy revolves around the idea that regenerative medicine may hold the potential to improve certain rare medical conditions and contribute to healthy aging. While there has been a remarkable rise in life expectancy over the last century due to medical and public health advancements, this increase in longevity has not been paralleled by the number of years a person is expected to live in relatively good health, free of chronic disease and disabilities of aging.
Longeveron’s lead investigational product is Lomecel-B™, an allogeneic Medicinal Signaling Cell therapy product isolated from the bone marrow of young, healthy adult donors. As humans age, they experience a decrease in immune system function, a decline in blood vessel functioning, chronic inflammation, and other issues. Clinical data has suggested that Lomecel-B™ may address these conditions through multiple mechanisms of action (MOA) that simultaneously target key aging-related processes.
The Company is headquartered in Miami, Florida.
Lomecel-B™
Lomecel-B™ is being evaluated in multiple clinical trials for aging-related chronic diseases and other life-threatening conditions under U.S. FDA-approved Investigational New Drug applications. Lomecel-B™ has multiple potential mechanisms of action encompassing pro-vascular, pro-regenerative, anti-inflammatory, and tissue repair and healing effects with broad potential applications across a spectrum of disease areas.
The drug is made from special living cells called Medicinal Signaling Cells (MSCs) that are isolated from fresh bone marrow tissue that has been donated by adult donors aged 18 to 45. Once the MSCs have been isolated from the fresh bone marrow through a careful selection process, the cells are culture-expanded (allowed to replicate under controlled laboratory conditions) into the billions using specialized techniques and processes. After a specific number of expansion cycles, called “passages,” the cells are harvested, separated into specific doses (e.g., 50 million cells), and cryopreserved until future use.
These cells have been shown to have characteristics that allow them to be transplanted from a donor to host without triggering a harmful immune response in the recipient, and they can be administered on an outpatient basis in as little as 40 minutes after thawing. Because of these characteristics, Lomecel-B™ is considered an “off-the-shelf” product.
In some trials, such as for Alzheimer’s disease and Aging-related Frailty, Lomecel-B™ is administered via peripheral intravenous infusion, while, in the Company’s HLHS trial, Lomecel-B™ is administered via direct injection into the heart tissue.
Market Opportunity
Longeveron estimates the potential market size for Lomecel-B™ in the treatment of HLHS to be up to $1 billion annually, globally.
U.S. patients suffering from Aging-related Frailty are estimated using U.S. Census Bureau statistics to be approximately 8.1 million. That population potentially represents a market for Lomecel-B™ of between $4 billion and $8 billion globally per year, according to Company estimates.
Additionally, the Alzheimer’s Association puts the number of Americans with that disease at 5.1 million, highlighting another potentially addressable market for Lomecel-B™, that’s worth $5 billion to $10 billion annually.
Management Team
Wa’el Hashad is CEO of Longeveron. He has more than 35 years of experience in the pharmaceutical and biotech industries. He has launched several successful brands in the U.S. and worldwide markets. Prior to joining Longeveron, he was president and CEO of Avanir Pharmaceuticals. Before Avanir, he was the chief commercial officer of Seres Therapeutics. He also has held senior leadership positions at Amgen, Boehringer Ingelheim, and Eli Lilly and Company. He holds a bachelor’s degree in pharmacy from Cairo University and an MBA from the University of Akron.
Joshua M. Hare, M.D., FACC, FAHA, is Co-Founder, Chief Science Officer and Chairman of Longeveron. He is a double board-certified cardiologist and is the founding director of the Interdisciplinary Stem Cell Institute at the University of Miami’s Miller School of Medicine. He is a recipient of the Paul Beeson Physician Faculty Scholar in Aging Research Award and is an elected member of the American Association of Physicians and The American Society for Clinical Investigation. He is also an elected Fellow of the American Heart Association. He received a bachelor’s degree from the University of Pennsylvania and his M.D. from The Johns Hopkins University School of Medicine.
Lisa Locklear is CFO at Longeveron. She previously served as the senior vice president and CFO for Avanir Pharmaceuticals. Prior to Avanir, she held senior financial roles at GSN Games, CoreLogic, Ingram Micro, the Walt Disney Company, and Price Waterhouse, with assignments in Paris and London. She holds a bachelor’s degree in plant science from the University of California, Davis, and an MBA from the University of California, Irvine. She is a licensed CPA (inactive) and is a member of the American Institute of Certified Public Accountants, the California Society of CPAs, and Financial Executives International.
Dr. Nataliya Agafonova, M.D., is the Chief Medical Officer at Longeveron. She previously served as clinical development lead, senior medical director, and product development chair at Otsuka Pharmaceuticals. Before that, she was the clinical development lead and senior medical director at Bristol-Myers Squibb. She previously held senior leadership positions at Ardea Bioscience, Biogen, Amgen, and Genzyme Corporation. She earned an M.D. from the Ukrainian National Medical University and completed her internal medicine residency at Kharkov State University Hospital in Ukraine.
Certain statements in this corporate profile that are not historical facts are forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, which reflect management’s current expectations, assumptions, and estimates of future operations, performance and economic conditions, and involve risks and uncertainties that could cause actual results to differ materially from those anticipated by the statements made herein. Forward-looking statements are generally identifiable by the use of forward-looking terminology such as “believe,” “expects,” “may,” “looks to,” “will,” “should,” “plan,” “intend,” “on condition,” “target,” “see,” “potential,” “estimates,” “preliminary,” or “anticipates” or the negative thereof or comparable terminology, or by discussion of strategy or goals or other future events, circumstances, or effects. Factors that could cause actual results to differ materially from those expressed or implied in any forward-looking statements in this release include, but are not limited to, statements regarding the offer and sale of securities, the terms of the offering, about the ability of Longeveron’s clinical trials to demonstrate safety and efficacy of the Company’s product candidates, and other positive results; the timing and focus of the Company’s ongoing and future preclinical studies and clinical trials and the reporting of data from those studies and trials; the size of the market opportunity for the Company’s product candidates, including its estimates of the number of patients who suffer from the diseases being targeted; the success of competing therapies that are or may become available; the beneficial characteristics, safety, efficacy and therapeutic effects of the Company’s product candidates; the Company’s ability to obtain and maintain regulatory approval of its product candidates in the U.S., Japan and other jurisdictions; the Company’s plans relating to the further development of its product candidates, including additional disease states or indications it may pursue; the Company’s plans and ability to obtain or protect intellectual property rights, including extensions of existing patent terms where available and its ability to avoid infringing the intellectual property rights of others; the need to hire additional personnel and the Company’s ability to attract and retain such personnel; the Company’s estimates regarding expenses, future revenue, capital requirements and needs for additional financing; the Company’s need to raise additional capital, and the difficulties it may face in obtaining access to capital, and the dilutive impact it may have on its investors; the Company’s financial performance and ability to continue as a going concern, and the period over which it estimates its existing cash and cash equivalents will be sufficient to fund its future operating expenses and capital expenditure requirements. Additionally, Longeveron makes no assurance that any public offering of its securities as described herein will occur on the timelines, in the manner or on the terms anticipated due to numerous factors. Further information relating to factors that may impact the Company’s results and forward-looking statements are disclosed in the Company’s filings with the Securities and Exchange Commission, including Longeveron’s Annual Report on Form 10-K for the year ended December 31, 2022, filed with the Securities and Exchange Commission on March 14, 2023 and its Quarterly Report on Form 10-Q for the second quarter of 2023 filed with the SEC on August 11, 2023. The forward-looking statements contained in this corporate profile are made as of the date of this corporate profile, and the Company disclaims any intention or obligation, other than imposed by law, to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise.
Investor Contact
Mike Moyer
LifeSci Advisors
Tel: 617-308-4306
Email: [email protected]
Date prepared: August 31, 2023
Longeveron Inc. (NASDAQ: LGVN), closed Friday's trading session at $1.29, off by 3.7313%, on 67,551 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.15/$4.5793.
Recent News
- Longeveron Inc. (NASDAQ: LGVN) - Five Significant Strides Made in Alzheimer’s Research in 2023
- Studies Suggest Fasting Could Tone Down Alzheimer's Signs
- InvestorNewsBreaks - Longeveron Inc. (NASDAQ: LGVN) Enters Definitive Agreement for $2.36M Registered Direct Offering
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing.
DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future.
Lexaria has also collaborated with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has been granted patent protection for specific delivery of nicotine, vitamins, NSAIDs, antiviral drugs, cannabinoids and more.
Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats.
The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products.
DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions.
DehydraTECH Technology
Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following:
- Speeds up delivery – the effects of the product are felt by the subject in just minutes.
- Increases bioavailability – the technology is much more effective at delivering a drug or product into the bloodstream.
- Increases brain absorption – animal testing suggests significant improvement in the quantity of drug delivered across the blood-brain barrier.
- Improves drug potency – more of the ingested product is made available to the body, so lower doses are required to achieve the desired effect.
- Reduces drug administration cost – lower doses mean lower overall drug costs.
- Masks unwanted taste – the technology eliminates or reduces the need for sweeteners.
Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery.
Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine.
Market Outlook
Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments:
- Oral Cannabinoids – a market estimated to be worth $18.4 billion in 2021 and expected to reach $46.2 billion by 2025.
- Antivirals – an estimated $52.1 billion market in 2021 that’s expected to grow to $66.7 billion by 2025.
- Oral Mucosal Nicotine – smokeless tobacco products, a $13.6 billion market in 2018, is forecast to grow at 7.2 percent annually through 2025.
- Human Hormones – estrogen and testosterone replacement therapies represented a $21.9 billion market in 2019, with a forecast CAGR of 7.7 percent through 2027.
- Ibuprofen and Naproxen – NSAID sales totaled $15.6 billion globally in 2019 and are projected to reach $24.4 billion by 2027.
- Vitamin D3 – the global market size was $1.1 billion in 2021, growing at 7 percent per year and expected to reach $1.7 billion in 2026.
Management Team
Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations.
John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents.
Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations.
Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch.
Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia.
Lexaria Bioscience Corp. (LEXX), closed Friday's trading session at $1.35, off by 2.8777%, on 77,337 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.6488/$3.5953.
Recent News
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - InvestorNewsBreaks – Lexaria Bioscience Corp.’s (NASDAQ: LEXX) DehydraTECH(TM) Shown to Improve Oral Performance of Rybelsus(R)-Branded GLP-1 Drug Semaglutide
- Lexaria Bioscience Corp. (NASDAQ: LEXX) Provides Superior Drug and API Delivery with Patented DehydraTECH Technology
- TinyGemsBreaks - Lexaria Bioscience Corp. (NASDAQ: LEXX) Leveraging Commercial Viability of DehydraTECH(TM) in Aggressive Patent Application Push
InMed Pharmaceuticals Inc. (NASDAQ: INM)
The QualityStocks Daily Newsletter would like to spotlight InMed Pharmaceuticals Inc. (NASDAQ: INM).
InMed Pharmaceuticals Inc. (NASDAQ: INM) is a global leader in the manufacturing and clinical development of rare cannabinoids. InMed is a clinical stage company developing cannabinoid-based pharmaceutical drug candidates, as well as manufacturing technologies for pharmaceutical-grade rare cannabinoids.
The company is dedicated to delivering new therapeutic alternatives to treat conditions with high unmet medical needs. The company is also developing a proprietary manufacturing technology to produce pharmaceutical-grade rare cannabinoids in the lab and has recently announced an LOI to acquire a leading rare cannabinoid manufacturer.
Research and Technology
There are more than 100 rare cannabinoids found in only trace amounts in the cannabis plant, together making up less than 1% of the plant’s biomass. InMed is initially focused on the therapeutic benefits of cannabinol (CBN) in diseases with high unmet medical need. Preclinical studies of CBN demonstrated an excellent safety profile and showed CBN has potential for therapeutic benefit over other cannabinoids such as tetrahydrocannabinol (THC) and cannabidiol (CBD).
Evidence suggests there may be great therapeutic potential in rare cannabinoids. Each has a specific chemical structure, and different cannabinoids have been observed to have distinct physiological properties in humans, including therapeutic potential for specific diseases as well as unique safety profiles. CBN is the active pharmaceutical ingredient (API) in InMed’s two lead programs for dermatological and ocular diseases.
InMed’s most advanced compound, INM-755, is a CBN topical cream under clinical development for the treatment of epidermolysis bullosa, a severe genetic skin disorder. To date, INM-755 has been evaluated in two Phase 1 clinical trials in healthy volunteers. InMed has filed Clinical Trial Applications in several countries as part of a global Phase 2 clinical trial of INM-755 (cannabinol) cream in epidermolysis bullosa. Responses from the National Competent Authorities and Ethics Committees are expected throughout the summer of 2021.
InMed is also involved in developing INM-088, an ocular CBN formulation being researched for the treatment of glaucoma, the second leading cause of blindness in the developed world. InMed is currently evaluating several formulations to deliver CBN into the eye to address issues of dosing frequency, side effects and treatment penetration. INM-088 is being designed for topical delivery to the eye. This localized delivery results in very little drug being absorbed or migrating into the bloodstream, thus minimizing potential adverse side effects. INM-088 shows promise to reduce intraocular pressure and provide neuroprotection of the eye.
Manufacturing
The limited availability of rare cannabinoids like CBN makes them economically impractical to extract directly from the plant for pharmaceutical use. InMed is developing IntegraSyn, a cannabinoid synthesis manufacturing system to create rare cannabinoids in the lab that are bioidentical to the compounds derived from the cannabis plant. IntegraSyn uses multiple standard pharmaceutical processes and has achieved a cannabinoid yield of 5 grams per liter, surpassing commercial viability and significantly exceeding currently reported industry yields. InMed is now focusing on manufacturing scale-up to larger batch sizes while continuing process optimization, targeting increased cannabinoid yield and further reducing overall cost of goods.
BayMedica Inc. Acquisition
On June 29, 2021, InMed announced it had entered into a non-binding letter of intent to acquire BayMedica Inc., a private company based in Nevada and California that specializes in the manufacture and commercialization of rare cannabinoids.
As noted in the news release, BayMedica is a revenue-stage biotechnology company leveraging its significant expertise in synthetic biology and pharmaceutical chemistry to develop efficient, scalable and proprietary manufacturing approaches to produce high quality, regulatory-compliant rare cannabinoids for consumer applications. BayMedica is currently commercializing the rare cannabinoid CBC (cannabichromene) as a B2B supplier to distributors and manufacturers marketing products in the health and wellness sector. BayMedica is planning additional rare cannabinoid launches for the coming year.
Pursuant to the indicative terms of the LOI, InMed and BayMedica intend to negotiate and enter into a definitive agreement under which InMed would acquire 100% of BayMedica in exchange for 1.6 million InMed common shares to be issued to BayMedica’s equity and convertible debt holders, with any such issued InMed common shares being subject to a six-month contractual hold period.
Market Outlook
There is a rapidly growing demand for rare cannabinoids. However, their low natural concentration makes traditional harvesting of these compounds cost prohibitive. Biosynthesis allows production of rare cannabinoids in the lab that are bioidentical to compounds found in nature, with significantly higher yields which reduce costs. Biosynthesis can produce pharmaceutical-grade, bioidentical, THC-free compounds at a cost that’s 70 to 90 percent less than wholesale prices of naturally harvested rare cannabinoids.
Cannabinoid-based pharmaceuticals are expected to overtake the market as rare cannabinoids become less expensive and more available. According to Statista, the value of the consumer market for cannabinoid-based pharmaceuticals in the United States is forecast to grow to $25 billion by 2025 and to $50 billion by 2029, with cannabinoid-based pharmaceuticals used to treat health conditions including pain, respiratory conditions, autoimmune conditions and more.
Management Team
Eric A. Adams has been CEO and president of InMed since June 2016. He has more than 25 years of experience in establishing corporate entities, capital formation, global market development, mergers and acquisitions, licensing and corporate governance. He previously served as CEO at enGene Inc. Prior to enGene, he held senior positions in global market development with QLT Inc. (Vancouver), Advanced Tissue Sciences Inc. (La Jolla, CA), Abbott Laboratories (Chicago, IL) and Fresenius AG (Germany).
Bruce S. Colwill is InMed’s CFO. He has more than 25 years of financial leadership experience in public and private companies. Prior to InMed, he served as CFO of General Fusion Inc., a private clean energy company. He was also CFO at Entrée Resources Inc., a mineral exploration company, from 2011 to 2016. He has held CFO roles at Neuromed Pharmaceuticals Ltd., Response Biomedical Corp, Forbes Medi-Tech Inc. and Euronet Worldwide Inc.
Alexandra D.J. Mancini is Senior Vice President, Clinical and Regulatory Affairs at InMed. She has more than 30 years of global biopharmaceutical research and development experience. She has been an executive with numerous biotech companies, including senior vice president of Clinical and Regulatory Affairs at Sirius Genomics; senior vice president of Clinical and Regulatory Affairs at INEX Pharmaceuticals; and vice president of Regulatory Affairs at QLT Inc.
Eric C. Hsu is Senior Vice President, Pre-Clinical Research and Development at InMed. He joined InMed with more than 18 years of scientific leadership experience in the field of gene therapy. He has held various positions within enGene Inc., including vice president of Research and vice president of Scientific Affairs and Operations. He received his Doctorate from the Department of Medical Biophysics at the University of Toronto.
Michael Woudenberg is Vice President, Chemistry, Manufacturing and Controls at InMed. He has more than 20 years of successful drug development, process engineering, GMP manufacturing and leadership experience. He has held positions with 3M, Cardiome Pharma, Arbutus Biopharma and, most recently, was Managing Director of Phyton Biotech LLC.
InMed Pharmaceuticals Inc. (INM), closed Friday's trading session at $0.37, off by 1.5957%, on 139,547 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.2913/$2.77.
Recent News
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Approves Election of Directors During 2023 Annual General Meeting
- InMed Announces Results of 2023 Annual General Meeting
- InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Advancing INM-089 to Target Treatment of Age-Related Macular Degeneration
Freight Technologies Inc. (NASDAQ: FRGT)
The QualityStocks Daily Newsletter would like to spotlight Freight Technologies Inc. (NASDAQ: FRGT).
Freight Technologies Inc. (NASDAQ: FRGT) (“Fr8Tech”) is a technology company developing solutions to optimize and automate the supply chain process, providing a platform for B2B cross-border shipping in the NAFTA region. The company’s mission is to revolutionize cross-border shipping by providing carriers with increased growth opportunities and shippers with flexibility, visibility and simplicity for the once-complex process of international over-the-road shipping.
Freight Technologies, formerly known as Hudson Capital Inc., assumed its current name and ticker symbol on May 27, 2022. Its primary operating subsidiary and its marketplace are known as Fr8App, and it conducts operations throughout North America under the names of Fr8App and/or Freight App. The company is headquartered in Houston, Texas, with multiple locations across the U.S. and Mexico.
The Fr8Tech Solutions Suite
Fr8Tech leverages artificial intelligence to provide cloud-based platforms aimed at automating the over-the-road transportation process, effectively reducing human touch points and expediting load booking times. The company’s suite of solutions includes:
- Fr8app – A B2B marketplace powered by AI and Machine Learning offering a real-time broker portal to connect shippers with qualified carriers
- Fr8Radar – A tracking solution providing shippers and carriers real-time locational data via Fr8app’s mobile solution or through integration with third-party GPS alternatives
- Fr8TMS – A transportation management system designed to help shippers manage their freight and all of the documents involved in shipping transactions, including invoices, customs documents, confirmation rates and proof of deliveries
- Fr8FMS – A fleet management system allowing transportation companies to better manage their fleets, reduce operational costs and provide better service to their customers
- Fr8Data – A data solution offering real-time dashboards and reports to shippers and carriers in an effort to increase visibility and control while supporting better business decisions
- Fr8Fleet – A platform that provides private fleet management, enabling large corporate shippers to purchase dedicated capacity secured by Fr8app in exchange for a fixed fee
Commitment to the Environment
Through its core focus on technology, Fr8Tech seeks to reduce the carbon footprint of the logistics industry. Its solutions aim to minimize empty miles for transportation firms and reduce overall paper consumption.
Fr8University
Fr8University is an educational program offering classroom and on-the-job training for Fr8Tech team members. Through the program, employees learn in-depth business fundamentals and applications along the truckload freight industry value chain.
Led by corporate educator Mario Mena, Fr8University is designed as an investment in the company’s human capital, providing an opportunity to communicate Fr8Tech’s corporate culture while accelerating operational growth.
Market Outlook
Fr8Tech’s established foothold in Mexico is key to its current efforts to promote sustainable growth in the cross-border shipping industry. Ongoing disruption in U.S.-Chinese trade relations have strengthened Mexico’s status as the largest trading partner of the U.S., with cross-border annual freight spending estimated at $385 billion according to data from the U.S. Department of Transportation. Annual domestic shipping in Mexico is estimated at $34 billion, while annual domestic shipping in the U.S. is estimated to total $732 billion.
Despite the size of this industry, fragmentation and inefficiencies prevail in the space. Thousands of legacy brokers, tens of thousands of shippers and hundreds of thousands of carriers still rely on outdated systems to arrange transport, spending hours on the phone negotiating pricing, waiting days to find trucks and drivers, preparing and printing forms, and operating without tracking or visibility. Add in cross-border complexity relating to customs and additional paperwork, and you have an industry ripe for technological disruption.
Fr8Tech’s recent revenue growth trends have highlighted the company’s efforts to capitalize on this opportunity. In 2021, Fr8Tech achieved revenues of $21.5 million, marking a year-over-year increase of 134%. The company issued revenue guidance for fiscal 2022 of $40 million in a February 9, 2022, press release, which would account for a further 86% year-over-year increase.
Management Team
Javier Selgas is CEO and a Director of Freight Technologies Inc. and Freight App Inc. He brings to the company over 15 years of experience developing technology and digital marketing strategies, including serving as Country Manager for Osigu, Spain, and as head of AJEgroup’s IT division for the Asia-Pacific region. Prior to joining Fr8Tech, Mr. Selgas founded digital marketing agency Lanzadera Online. He has also served as an IT consultant to major corporations, including Endesa and Ibermatica.
Mike Flinker is President of Fr8Tech. He has over four decades of experience in the transportation industry, with 30+ years focused on cross-border logistics. Prior to joining Fr8Tech, Mr. Flinker founded FLS Transportation, the largest cross-border logistics company in Canada. He also previously held positions with Clarke Transport Inc., Canadian Pacific and Reimer Express Inc. (a division of Roadway Express).
Paul Freudenthaler is the company’s CFO and Secretary to the company Board. He has over 30 years of financial expertise, having previously served as CFO for several leading companies across multiple countries, including Macquarie in Mexico, Old Mutual in Latin America and Ascentium Capital in the U.S. Mr. Freudenthaler’s experience include leadership roles from which he guided IPOs and M&A transactions.
Luisa Lopez is COO of Fr8Tech. She brings to the company 25+ years of management experience in logistics, supply chain, operations and customer service. Ms. Lopez previously served as a Director of Landstar, where she was responsible for commercial and client development strategies in the Mexican market. Additionally, she managed more than 2,000 transport units specialized in staff and school mobility while with Traxion in Mexico.
Freight Technologies Inc. (NASDAQ: FRGT), closed Friday's trading session at $0.3251, off by 6.2031%, on 851,155 volume. The average volume for the last 3 months is 4.139M and the stock's 52-week low/high is $0.252/$6.70.
Recent News
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Agrees to Terms with Amazon Mexico for Domestic Shipping for the Second Consecutive Year
- InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Investors Convert $3.55M Debt to Equity
- Freight Technologies, Inc. Announces Conversion of $3.55M Convertible Debt to Equity
Astrotech Corp. (NASDAQ: ASTC)
The QualityStocks Daily Newsletter would like to spotlight Astrotech Corp. (NASDAQ: ASTC).
Astrotech Corp. (NASDAQ: ASTC) is an instrumentation company that designs, manufactures and commercializes solutions. Its solutions include mass spectrometry, process controls, chemical detectors and medical disease detection.
The company was established in 1984 and, prior to 2009, was known as SPACEHAB Inc., a NASA contractor offering technology originally developed for NASA to monitor air quality on the International Space Station. When the Space Shuttle program ended, the company focused on its satellite processing and mass spectrometer instrumentation units and adopted the Astrotech name.
In 2014, Astrotech sold its satellite subsidiary to focus on its Astrotech Technology Inc. (ATi) mass spectrometry solutions, which offer a number of advantages over competing platforms. Notably, Astrotech’s ATi technology is ruggedized, rapid, simple to use and customizable, with hands-free calibration and tuning.
Between 2016 and 2019, the company secured U.S. patents for its technology and achieved European Union (ECAC) certification for the TRACER 1000™, the world’s first mass-spec Explosives Trace Detector (ETD) used in airports worldwide. Astrotech continues to innovate and add to its suite of products, including AgLAB-1000, a process control system, and the BreathTest 1000, a disease detection solution.
Astrotech is headquartered in Austin, Texas.
Subsidiaries
Astrotech Technologies Inc.
Astrotech Technologies Inc. (ATi) owns and licenses the platform mass spectrometry technology originally developed by 1st Detect. This technology is designed to be less expensive, smaller and easier to use than traditional mass spectrometers.
Unlike other technologies, ATi works under high vacuum, which eliminates competing molecules, yielding higher resolution and fewer false alarms. The company’s intellectual property includes 18 granted patents, along with extensive trade secrets.
ATi exclusively licenses the Astrotech Mass Spectrometer Technology to the three wholly owned subsidiaries of Astrotech.
1st Detect Corp.
1st Detect Corp. developed the TRACER 1000, the world’s first mass spectrometry-based explosives and narcotics trace detector. 1st Detect ETDs were developed for use at airports, cargo facilities and other secured locations and borders worldwide.
1st Detect’s commercial sales of the TRACER 1000 ETD, consumables and recurring maintenance services brought in $750,000 in total revenue during the fiscal year ended June 30, 2023. The Astrotech subsidiary recently secured two orders for a total of 24 Tracer 1000 units from two Romanian security and telecommunications companies, to be delivered during calendar 2023.
AgLAB Inc.
AgLAB Inc. is developing a series of mass spectrometers for use in the hemp and cannabis market, with an initial focus on optimizing yields in the distillation processes.
AgLAB, which uses the company’s proprietary AgLAB 1000-D2™ mass spectrometer, has been proven to improve distillation oil yields and bottom-line profits for hemp and cannabis producers. During field trials, AgLAB was able to improve ending-weight yields by an average of 24%.
BreathTech Corp.
BreathTech is developing the BreathTest-1000™, a breath analysis tool to screen for volatile organic compound (“VOC”) metabolites found in a person’s breath that could indicate they may have a compromised condition including but not limited to a bacterial or viral infection. The company believes that new tools to aid in the battle against COVID-19 and other diseases remain of the utmost importance to help more quickly identify that an infection may be present.
Market Opportunity
A report by Mordor Intelligence, a research and advisory firm, put the global mass spectrometry market at $6.37 billion in 2023. The market is forecast to grow to $8.63 billion by 2028, achieving a CAGR of 6.25% during the forecast period.
One of the major driving factors for the growth of the mass spectrometry market is technological advancements in mass spectrometer devices, the report states. Key market players are continuously working toward advancing their existing products and launching innovative and advanced mass spectrometer devices.
Another major factor that is expected to boost market growth is increasing research and development expenditure by both government and private entities, according to the report. Mass spectrometry devices are also being used in the detection and analysis of COVID-19 and other disease samples, which may have a positive impact on the market.
Management Team
The Astrotech leadership team includes management executives, as well as industry and technology experts. The company continues to actively expand its talent pool to meet evolving demands.
Thomas B. Pickens III is Chairman, CEO and Chief Technology Officer of Astrotech Corp. He also serves as CEO of Astrotech subsidiaries ATi, 1st Detect, AgLAB Inc. and BreathTech Corp. Previously, he was the founder and president of Beta Computer Systems Inc. and T.B. Pickens & Co. He was founder and general partner of Grace Pickens Acquisition Partners L.P and managing partner of Sumpter Partners. He also served as CEO of Catalyst Energy Corporation and United Thermal Corporation and as president of Golden Bear Corp., United Hydro Inc. and Slate Creek Corp. He received a B.A. in Economics, Computer Science and Engineering from Southern Methodist University.
Jaime Hinojosa, CPA, is CFO at Astrotech Corp. He joined the company in 2015 and has served as its Corporate Controller since 2019. His previous roles with the company include Director of Finance, from 2017 to 2019, and Assistant Controller, from 2015 to 2017. Prior to joining Astrotech, Mr. Hinojosa worked as an Accounting Manager for O’Reilly Auto Parts and gained public accounting experience as an Audit Manager at Burton McCumber & Cortez LLP.
Astrotech Corp. (NASDAQ: ASTC), closed Friday's trading session at $8.3, even for the day, on 524 volume. The average volume for the last 3 months is 3,916 and the stock's 52-week low/high is $7.00/$15.11.
Recent News
- Astrotech Corp. (NASDAQ: ASTC) - InvestorNewsBreaks - Astrotech Corp. (NASDAQ: ASTC) Creates New Subsidiary, Announces Application of New Technology
- Astrotech Corp. (NASDAQ: ASTC) Is 'One to Watch'
- InvestorNewsBreaks - Astrotech Corporation (NASDAQ: ASTC) Unveils AgLAB MVP TM - a Game Changer for Cannabinoid Industry
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Friday's trading session at $1.19, up 3.4783%, on 9,341 volume. The average volume for the last 3 months is 107,029 and the stock's 52-week low/high is $0.6105/$4.40.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Light, Viruses Could Transform Brain-Tumor Treatment
- InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Announces DSMB Recommendation to Continue Pivotal GBM Study without Modification
- Researchers Discuss How Brain Cancer Radiotherapy Side Effects Can Be Reduced
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Announces Engagement of Alliance Global Partners
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks – Aditxt Inc. (NASDAQ: ADTX) Secures $6M in Private Placement
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks – Amesite Inc. (NASDAQ: AMST), CMU Collaborate to Offer New Asynchronous Implicit Bias Course for Healthcare Professionals
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - Appia Rare Earths & Uranium - A Year in Review
- Fresh2 Group Ltd. (NASDAQ: FRES) - Appointment of New Independent Director
- Astiva Health - InvestorNewsBreaks – Astiva Health Files Statement for Proposed IPO
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp Identifies New Priority Drill Targets at its Kay Mine Project
- Astrotech Corp. (NASDAQ: ASTC) - InvestorNewsBreaks - Astrotech Corp. (NASDAQ: ASTC) Creates New Subsidiary, Announces Application of New Technology
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Google Cloud to Bring Medically-Tuned AI Technology to Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - Battery Mineral Resources Corp. Extends Closing Date on its Announced Offering of up to US$6M in Unsecured Convertible Debentures
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks – Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Closes on $1.69M Exercise of Outstanding Warrants
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Announces Completion of Cladribine ODF Toxicity Study and Financing
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Reports Filing BFS for Crawford Nickel Sulphide Project
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - New Study Highlights Role of Mitochondria in Connecting Cancer to Dietary Fat
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Light, Viruses Could Transform Brain-Tumor Treatment
- Correlate Energy Corp. (OTCQB: CIPI) - Correlate Energy Corp. (CIPI) Completes Two Solar Projects and Commences a Third, Promoting Sustainability and Carbon Footprint Reduction
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks – Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Eyes Enormous Opportunities Across Its Clinical Development Programs
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp.'s (NASDAQ: DRIO) Solutions Selected by National Employer
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks – Diamond Lake Minerals Inc. (DLMI) Partners with Online Tree-Planting Platform to Plant Trees in Kenya
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Demonstrates Powerful Quantum-Hybrid Applications Through New Customer Success Stories
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - Electronic Servitor Publication Network Inc. (XESP) Finalizes PhiTech Management, LLC Asset Purchase Including Proprietary Digital Engagement Engine(TM)
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Reports 'Encouraging' Results from Definition Drill Program at Iska Iska Project
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Commences Production at Three US Uranium Mines
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF), Completes Fraser Valley Biogas Expansion in Company 'Milestone'
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Releases Shareholder Letter Outlining Operational Update
- FingerMotion Inc. (NASDAQ: FNGR) - Lind Partners Statement on FingerMotion, Inc.
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Provides Update on Assay Results and Data Analysis at Deer Horn
- Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) - Fathom Nickel sees multiple intercepts following successful drill program at Gochager Lake
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) Reports on Assay Results of Soil Geochemical Survey, Indicates New Target Areas at Augustus Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Enters Commercialization Agreement with Leading Global Defense Company
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Agrees to Terms with Amazon Mexico for Domestic Shipping for the Second Consecutive Year
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - Indians Recycle Jewelry as Gold Prices Surge
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Issues Update on 2023 Milestones, 2024 Path Forward
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - Genprex Inc. (NASDAQ: GNPX) Granted Orphan Drug Designation in Small Cell Lung Cancer by FDA, Presented Data at Two October Events Focused on Lung Cancer
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Regains Compliance with Nasdaq Listing Requirements
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Combines AI, Proprietary Technology to Deliver Unprecedented Cardiac Insights
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Receives Indication of Interest from Koito Manufacturing Co.
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FSE: 7HI) Appoints Finance, Accounting Veteran as CFO
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks — Upstream Experiences Key Achievements in Pivotal Year as App ‘Propels’ Itself Forward into Future of Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Releases Unaudited Financial Results for 2023 Half-Year
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Announces Receipt of Nasdaq Notice Regarding Late Form 10-Q and Subsequent Compliance Determination
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Receives 'Milestone' Patent for Drug Formulation Used to Treat Agitation in Alzheimer's
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Inks New Business Contract with IT System Company
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Approves Election of Directors During 2023 Annual General Meeting
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot Inc.'s (NASDAQ: SHOT) Patented Beverage Sells Out First Day on DTC Website
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks – Knightscope, Inc.’s (NASDAQ: KSCP) Solid Execution, Revenue Growth Potential Featured in Ascendiant Report
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Five Significant Strides Made in Alzheimer’s Research in 2023
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - InvestorNewsBreaks – Lexaria Bioscience Corp.’s (NASDAQ: LEXX) DehydraTECH(TM) Shown to Improve Oral Performance of Rybelsus(R)-Branded GLP-1 Drug Semaglutide
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Announces Closing on $16.1M Private Placement Financing
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - Mydecine Innovations Group Receives Notice of Allowance from USPTO for its MYCO-005 Compound
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Seeks to Raise Up To $10 Million from Regulation A Offering
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Positive Nasdaq Listing Decision
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Highlights from Earnings Results of Major e-Commerce Sector Players
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks — Mullen Automotive Inc. (NASDAQ: MULN) Invoices $12M-Plus for Class 1, Class 3 EVs in 2023
- PaxMedica Inc. (NASDAQ: PXMD) - Israeli Study Offers Hope to Sleeping Sickness Patients
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTC: PLTXF) (Frankfurt: WNT1) Expands Ecommerce Fulfillment Platform to Add JUST Egg
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Results of Annual and Special Meeting Voting
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Biweekly Status Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q3 2023 Financial Results, Business Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc.'s (PBIO) UltraShear(TM) Platform Featured in Food Technology Article
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Provides Interim Analysis from Phase 1b NGC-Cap Trial
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc.'s (RXMD) PharmcoRx Collaborates to Expand Medicare Advantage OTC Benefits
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Inks Acquisition Term Sheet with Telix Pharmaceuticals
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks – Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Recaps Successful Year, Eyes Robust 2024 Development Plans
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Reports Status Update on Aleafia Transaction
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Oncologist Explores Whether Including Fiber in Diets Boosts Immunotherapy Outcomes
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Nickel Market at Decisive Point Between Demand, Supply
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - As 2024 Gets Underway, Gold Seems Set on Upward Trajectory
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Subsidiary Begins Controlled Trial to Study Reduction of Greenhouse Gas Emissions
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Software Effective Solutions Corp. (OTC: SFWJ) - InvestorNewsBreaks - Software Effective Solutions Corp. (SFWJ) Engages IBN for Corporate Communications Expertise
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Announces Filing of Definitive Proxy Statement, Details for Annual Meeting
- Sigyn Therapeutics Inc. (OTCQB: SIGY) - InvestorNewsBreaks – Sigyn Therapeutics Inc. (SIGY) CEO Releases Shareholder Letter Outlining Opportunities, Challenges, Processes
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks – Silo Pharma Inc. (NASDAQ: SILO) Successfully Completes First Phase of Dose-Ranging SPC-15 Study
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Offers Robust Offender Monitoring Solutions, Meeting the Emerging Challenges for both Agency and Private Operators
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces First SD7 Handset Order from Institution of Higher Education
- SOHM Inc. (OTC: SHMN) - SOHM Inc. (SHMN) Releases New Product Line and Expands Manufacturing Operations in Carlsbad, California
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Two Brands to be Spotlighted by AMPM's Thorntons Convenience Stores
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) CEO to Participate in Upcoming Investor Conferences
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Celebrates Unprecedented Organic Growth of Winona Pure® Popcorn Spray
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - Vision Marine Technologies Inc. (NASDAQ: VMAR) Partners with Leading Florida Boat Rental in Win-Win-Win Expansion
- SenesTech Inc. (NASDAQ: SNES) - SenesTech Inc. (NASDAQ: SNES) Reports Major Ramp-Up for its Latest Rodent Birth Control Product Amid Strong Demand for Non-lethal Pest Control
- Sekur Private Data Ltd. (FRA: GDT0) (CSE: SKUR) (OTCQB: SWISF) - Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FRA: GDT0) Suite of Services Protects Enterprises From Cybersecurity Breaches
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Receives Nasdaq Notice on Late Filing of Its Form 10-Q
- Tingo Group Inc. (NASDAQ: TIO) - Tingo Group Announces Interim Co-CEO to Temporarily Step Down by Mutual Agreement
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks – Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Announces Dosing of First Patient in Phase 2a TRP-8802 Clinical Trial
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TRxADE Announces Receipt of Notification of Deficiency from Nasdaq Regarding Requirement to Timely File Quarterly Report on Form 10-Q.
- Turbo Energy S.A. (NASDAQ: TURB) - InvestorNewsBreaks - Turbo Energy S.A. (NASDAQ: TURB) Announces Interim Results for Period Ended June 30, 2023, Transition in Management Structure
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) the West’s Strategy to Weaken China’s Minerals Dominance with its RapidSX(TM) Technology
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Enters Strategic MOU to Collaborate on Domestic Uranium Fuel Supply
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - Vision Marine Technologies Inc. (NASDAQ: VMAR) Partners with Leading Florida Boat Rental in Win-Win-Win Expansion
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Releases Q2 2024 Financial Report, Corporate Update
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) to Host Pioneering Conference for Women in Medical, Dental Sectors
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Bolsters BolaWrap® 150 Deployments, Promoting Safer Communities Nationwide
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Announces Engagement of Alliance Global Partners
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks – Aditxt Inc. (NASDAQ: ADTX) Secures $6M in Private Placement
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks – Amesite Inc. (NASDAQ: AMST), CMU Collaborate to Offer New Asynchronous Implicit Bias Course for Healthcare Professionals
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - Appia Rare Earths & Uranium - A Year in Review
- Fresh2 Group Ltd. (NASDAQ: FRES) - Appointment of New Independent Director
- Astiva Health - InvestorNewsBreaks – Astiva Health Files Statement for Proposed IPO
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp Identifies New Priority Drill Targets at its Kay Mine Project
- Astrotech Corp. (NASDAQ: ASTC) - InvestorNewsBreaks - Astrotech Corp. (NASDAQ: ASTC) Creates New Subsidiary, Announces Application of New Technology
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Google Cloud to Bring Medically-Tuned AI Technology to Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - Battery Mineral Resources Corp. Extends Closing Date on its Announced Offering of up to US$6M in Unsecured Convertible Debentures
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks – Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Closes on $1.69M Exercise of Outstanding Warrants
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Announces Completion of Cladribine ODF Toxicity Study and Financing
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Reports Filing BFS for Crawford Nickel Sulphide Project
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - New Study Highlights Role of Mitochondria in Connecting Cancer to Dietary Fat
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Light, Viruses Could Transform Brain-Tumor Treatment
- Correlate Energy Corp. (OTCQB: CIPI) - Correlate Energy Corp. (CIPI) Completes Two Solar Projects and Commences a Third, Promoting Sustainability and Carbon Footprint Reduction
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks – Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Eyes Enormous Opportunities Across Its Clinical Development Programs
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp.'s (NASDAQ: DRIO) Solutions Selected by National Employer
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks – Diamond Lake Minerals Inc. (DLMI) Partners with Online Tree-Planting Platform to Plant Trees in Kenya
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Demonstrates Powerful Quantum-Hybrid Applications Through New Customer Success Stories
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - Electronic Servitor Publication Network Inc. (XESP) Finalizes PhiTech Management, LLC Asset Purchase Including Proprietary Digital Engagement Engine(TM)
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Reports 'Encouraging' Results from Definition Drill Program at Iska Iska Project
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Commences Production at Three US Uranium Mines
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF), Completes Fraser Valley Biogas Expansion in Company 'Milestone'
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Releases Shareholder Letter Outlining Operational Update
- FingerMotion Inc. (NASDAQ: FNGR) - Lind Partners Statement on FingerMotion, Inc.
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Provides Update on Assay Results and Data Analysis at Deer Horn
- Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) - Fathom Nickel sees multiple intercepts following successful drill program at Gochager Lake
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) Reports on Assay Results of Soil Geochemical Survey, Indicates New Target Areas at Augustus Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Enters Commercialization Agreement with Leading Global Defense Company
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Agrees to Terms with Amazon Mexico for Domestic Shipping for the Second Consecutive Year
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - Indians Recycle Jewelry as Gold Prices Surge
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Issues Update on 2023 Milestones, 2024 Path Forward
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - Genprex Inc. (NASDAQ: GNPX) Granted Orphan Drug Designation in Small Cell Lung Cancer by FDA, Presented Data at Two October Events Focused on Lung Cancer
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Regains Compliance with Nasdaq Listing Requirements
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Combines AI, Proprietary Technology to Deliver Unprecedented Cardiac Insights
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Receives Indication of Interest from Koito Manufacturing Co.
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FSE: 7HI) Appoints Finance, Accounting Veteran as CFO
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks — Upstream Experiences Key Achievements in Pivotal Year as App ‘Propels’ Itself Forward into Future of Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Releases Unaudited Financial Results for 2023 Half-Year
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Announces Receipt of Nasdaq Notice Regarding Late Form 10-Q and Subsequent Compliance Determination
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Receives 'Milestone' Patent for Drug Formulation Used to Treat Agitation in Alzheimer's
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Inks New Business Contract with IT System Company
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Approves Election of Directors During 2023 Annual General Meeting
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot Inc.'s (NASDAQ: SHOT) Patented Beverage Sells Out First Day on DTC Website
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks – Knightscope, Inc.’s (NASDAQ: KSCP) Solid Execution, Revenue Growth Potential Featured in Ascendiant Report
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Five Significant Strides Made in Alzheimer’s Research in 2023
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - InvestorNewsBreaks – Lexaria Bioscience Corp.’s (NASDAQ: LEXX) DehydraTECH(TM) Shown to Improve Oral Performance of Rybelsus(R)-Branded GLP-1 Drug Semaglutide
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Announces Closing on $16.1M Private Placement Financing
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - Mydecine Innovations Group Receives Notice of Allowance from USPTO for its MYCO-005 Compound
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Seeks to Raise Up To $10 Million from Regulation A Offering
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Positive Nasdaq Listing Decision
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Highlights from Earnings Results of Major e-Commerce Sector Players
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks — Mullen Automotive Inc. (NASDAQ: MULN) Invoices $12M-Plus for Class 1, Class 3 EVs in 2023
- PaxMedica Inc. (NASDAQ: PXMD) - Israeli Study Offers Hope to Sleeping Sickness Patients
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTC: PLTXF) (Frankfurt: WNT1) Expands Ecommerce Fulfillment Platform to Add JUST Egg
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Results of Annual and Special Meeting Voting
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Biweekly Status Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q3 2023 Financial Results, Business Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc.'s (PBIO) UltraShear(TM) Platform Featured in Food Technology Article
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Provides Interim Analysis from Phase 1b NGC-Cap Trial
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc.'s (RXMD) PharmcoRx Collaborates to Expand Medicare Advantage OTC Benefits
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Inks Acquisition Term Sheet with Telix Pharmaceuticals
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks – Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Recaps Successful Year, Eyes Robust 2024 Development Plans
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Reports Status Update on Aleafia Transaction
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Oncologist Explores Whether Including Fiber in Diets Boosts Immunotherapy Outcomes
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Nickel Market at Decisive Point Between Demand, Supply
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - As 2024 Gets Underway, Gold Seems Set on Upward Trajectory
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Subsidiary Begins Controlled Trial to Study Reduction of Greenhouse Gas Emissions
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Software Effective Solutions Corp. (OTC: SFWJ) - InvestorNewsBreaks - Software Effective Solutions Corp. (SFWJ) Engages IBN for Corporate Communications Expertise
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Announces Filing of Definitive Proxy Statement, Details for Annual Meeting
- Sigyn Therapeutics Inc. (OTCQB: SIGY) - InvestorNewsBreaks – Sigyn Therapeutics Inc. (SIGY) CEO Releases Shareholder Letter Outlining Opportunities, Challenges, Processes
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks – Silo Pharma Inc. (NASDAQ: SILO) Successfully Completes First Phase of Dose-Ranging SPC-15 Study
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Offers Robust Offender Monitoring Solutions, Meeting the Emerging Challenges for both Agency and Private Operators
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces First SD7 Handset Order from Institution of Higher Education
- SOHM Inc. (OTC: SHMN) - SOHM Inc. (SHMN) Releases New Product Line and Expands Manufacturing Operations in Carlsbad, California
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Two Brands to be Spotlighted by AMPM's Thorntons Convenience Stores
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) CEO to Participate in Upcoming Investor Conferences
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Celebrates Unprecedented Organic Growth of Winona Pure® Popcorn Spray
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - Vision Marine Technologies Inc. (NASDAQ: VMAR) Partners with Leading Florida Boat Rental in Win-Win-Win Expansion
- SenesTech Inc. (NASDAQ: SNES) - SenesTech Inc. (NASDAQ: SNES) Reports Major Ramp-Up for its Latest Rodent Birth Control Product Amid Strong Demand for Non-lethal Pest Control
- Sekur Private Data Ltd. (FRA: GDT0) (CSE: SKUR) (OTCQB: SWISF) - Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FRA: GDT0) Suite of Services Protects Enterprises From Cybersecurity Breaches
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Receives Nasdaq Notice on Late Filing of Its Form 10-Q
- Tingo Group Inc. (NASDAQ: TIO) - Tingo Group Announces Interim Co-CEO to Temporarily Step Down by Mutual Agreement
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks – Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Announces Dosing of First Patient in Phase 2a TRP-8802 Clinical Trial
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TRxADE Announces Receipt of Notification of Deficiency from Nasdaq Regarding Requirement to Timely File Quarterly Report on Form 10-Q.
- Turbo Energy S.A. (NASDAQ: TURB) - InvestorNewsBreaks - Turbo Energy S.A. (NASDAQ: TURB) Announces Interim Results for Period Ended June 30, 2023, Transition in Management Structure
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) the West’s Strategy to Weaken China’s Minerals Dominance with its RapidSX(TM) Technology
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Enters Strategic MOU to Collaborate on Domestic Uranium Fuel Supply
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - Vision Marine Technologies Inc. (NASDAQ: VMAR) Partners with Leading Florida Boat Rental in Win-Win-Win Expansion
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Releases Q2 2024 Financial Report, Corporate Update
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) to Host Pioneering Conference for Women in Medical, Dental Sectors
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Bolsters BolaWrap® 150 Deployments, Promoting Safer Communities Nationwide
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Announces Engagement of Alliance Global Partners
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks – Aditxt Inc. (NASDAQ: ADTX) Secures $6M in Private Placement
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks – Amesite Inc. (NASDAQ: AMST), CMU Collaborate to Offer New Asynchronous Implicit Bias Course for Healthcare Professionals
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - Appia Rare Earths & Uranium - A Year in Review
- Fresh2 Group Ltd. (NASDAQ: FRES) - Appointment of New Independent Director
- Astiva Health - InvestorNewsBreaks – Astiva Health Files Statement for Proposed IPO
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp Identifies New Priority Drill Targets at its Kay Mine Project
- Astrotech Corp. (NASDAQ: ASTC) - InvestorNewsBreaks - Astrotech Corp. (NASDAQ: ASTC) Creates New Subsidiary, Announces Application of New Technology
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Google Cloud to Bring Medically-Tuned AI Technology to Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - Battery Mineral Resources Corp. Extends Closing Date on its Announced Offering of up to US$6M in Unsecured Convertible Debentures
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks – Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Closes on $1.69M Exercise of Outstanding Warrants
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Announces Completion of Cladribine ODF Toxicity Study and Financing
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Reports Filing BFS for Crawford Nickel Sulphide Project
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - New Study Highlights Role of Mitochondria in Connecting Cancer to Dietary Fat
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Light, Viruses Could Transform Brain-Tumor Treatment
- Correlate Energy Corp. (OTCQB: CIPI) - Correlate Energy Corp. (CIPI) Completes Two Solar Projects and Commences a Third, Promoting Sustainability and Carbon Footprint Reduction
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks – Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Eyes Enormous Opportunities Across Its Clinical Development Programs
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp.'s (NASDAQ: DRIO) Solutions Selected by National Employer
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks – Diamond Lake Minerals Inc. (DLMI) Partners with Online Tree-Planting Platform to Plant Trees in Kenya
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Demonstrates Powerful Quantum-Hybrid Applications Through New Customer Success Stories
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - Electronic Servitor Publication Network Inc. (XESP) Finalizes PhiTech Management, LLC Asset Purchase Including Proprietary Digital Engagement Engine(TM)
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Reports 'Encouraging' Results from Definition Drill Program at Iska Iska Project
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Commences Production at Three US Uranium Mines
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF), Completes Fraser Valley Biogas Expansion in Company 'Milestone'
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Releases Shareholder Letter Outlining Operational Update
- FingerMotion Inc. (NASDAQ: FNGR) - Lind Partners Statement on FingerMotion, Inc.
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Provides Update on Assay Results and Data Analysis at Deer Horn
- Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) - Fathom Nickel sees multiple intercepts following successful drill program at Gochager Lake
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) Reports on Assay Results of Soil Geochemical Survey, Indicates New Target Areas at Augustus Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Enters Commercialization Agreement with Leading Global Defense Company
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Agrees to Terms with Amazon Mexico for Domestic Shipping for the Second Consecutive Year
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - Indians Recycle Jewelry as Gold Prices Surge
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Issues Update on 2023 Milestones, 2024 Path Forward
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - Genprex Inc. (NASDAQ: GNPX) Granted Orphan Drug Designation in Small Cell Lung Cancer by FDA, Presented Data at Two October Events Focused on Lung Cancer
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Regains Compliance with Nasdaq Listing Requirements
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Combines AI, Proprietary Technology to Deliver Unprecedented Cardiac Insights
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Receives Indication of Interest from Koito Manufacturing Co.
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FSE: 7HI) Appoints Finance, Accounting Veteran as CFO
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks — Upstream Experiences Key Achievements in Pivotal Year as App ‘Propels’ Itself Forward into Future of Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Releases Unaudited Financial Results for 2023 Half-Year
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Announces Receipt of Nasdaq Notice Regarding Late Form 10-Q and Subsequent Compliance Determination
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Receives 'Milestone' Patent for Drug Formulation Used to Treat Agitation in Alzheimer's
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Inks New Business Contract with IT System Company
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Approves Election of Directors During 2023 Annual General Meeting
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot Inc.'s (NASDAQ: SHOT) Patented Beverage Sells Out First Day on DTC Website
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks – Knightscope, Inc.’s (NASDAQ: KSCP) Solid Execution, Revenue Growth Potential Featured in Ascendiant Report
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Five Significant Strides Made in Alzheimer’s Research in 2023
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - InvestorNewsBreaks – Lexaria Bioscience Corp.’s (NASDAQ: LEXX) DehydraTECH(TM) Shown to Improve Oral Performance of Rybelsus(R)-Branded GLP-1 Drug Semaglutide
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Announces Closing on $16.1M Private Placement Financing
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - Mydecine Innovations Group Receives Notice of Allowance from USPTO for its MYCO-005 Compound
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Seeks to Raise Up To $10 Million from Regulation A Offering
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Positive Nasdaq Listing Decision
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Highlights from Earnings Results of Major e-Commerce Sector Players
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks — Mullen Automotive Inc. (NASDAQ: MULN) Invoices $12M-Plus for Class 1, Class 3 EVs in 2023
- PaxMedica Inc. (NASDAQ: PXMD) - Israeli Study Offers Hope to Sleeping Sickness Patients
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTC: PLTXF) (Frankfurt: WNT1) Expands Ecommerce Fulfillment Platform to Add JUST Egg
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Results of Annual and Special Meeting Voting
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Biweekly Status Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q3 2023 Financial Results, Business Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc.'s (PBIO) UltraShear(TM) Platform Featured in Food Technology Article
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Provides Interim Analysis from Phase 1b NGC-Cap Trial
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc.'s (RXMD) PharmcoRx Collaborates to Expand Medicare Advantage OTC Benefits
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Inks Acquisition Term Sheet with Telix Pharmaceuticals
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks – Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Recaps Successful Year, Eyes Robust 2024 Development Plans
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Reports Status Update on Aleafia Transaction
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Oncologist Explores Whether Including Fiber in Diets Boosts Immunotherapy Outcomes
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Nickel Market at Decisive Point Between Demand, Supply
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - As 2024 Gets Underway, Gold Seems Set on Upward Trajectory
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Subsidiary Begins Controlled Trial to Study Reduction of Greenhouse Gas Emissions
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Software Effective Solutions Corp. (OTC: SFWJ) - InvestorNewsBreaks - Software Effective Solutions Corp. (SFWJ) Engages IBN for Corporate Communications Expertise
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Announces Filing of Definitive Proxy Statement, Details for Annual Meeting
- Sigyn Therapeutics Inc. (OTCQB: SIGY) - InvestorNewsBreaks – Sigyn Therapeutics Inc. (SIGY) CEO Releases Shareholder Letter Outlining Opportunities, Challenges, Processes
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks – Silo Pharma Inc. (NASDAQ: SILO) Successfully Completes First Phase of Dose-Ranging SPC-15 Study
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Offers Robust Offender Monitoring Solutions, Meeting the Emerging Challenges for both Agency and Private Operators
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces First SD7 Handset Order from Institution of Higher Education
- SOHM Inc. (OTC: SHMN) - SOHM Inc. (SHMN) Releases New Product Line and Expands Manufacturing Operations in Carlsbad, California
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Two Brands to be Spotlighted by AMPM's Thorntons Convenience Stores
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) CEO to Participate in Upcoming Investor Conferences
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Celebrates Unprecedented Organic Growth of Winona Pure® Popcorn Spray
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - Vision Marine Technologies Inc. (NASDAQ: VMAR) Partners with Leading Florida Boat Rental in Win-Win-Win Expansion
- SenesTech Inc. (NASDAQ: SNES) - SenesTech Inc. (NASDAQ: SNES) Reports Major Ramp-Up for its Latest Rodent Birth Control Product Amid Strong Demand for Non-lethal Pest Control
- Sekur Private Data Ltd. (FRA: GDT0) (CSE: SKUR) (OTCQB: SWISF) - Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FRA: GDT0) Suite of Services Protects Enterprises From Cybersecurity Breaches
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Receives Nasdaq Notice on Late Filing of Its Form 10-Q
- Tingo Group Inc. (NASDAQ: TIO) - Tingo Group Announces Interim Co-CEO to Temporarily Step Down by Mutual Agreement
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks – Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Announces Dosing of First Patient in Phase 2a TRP-8802 Clinical Trial
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TRxADE Announces Receipt of Notification of Deficiency from Nasdaq Regarding Requirement to Timely File Quarterly Report on Form 10-Q.
- Turbo Energy S.A. (NASDAQ: TURB) - InvestorNewsBreaks - Turbo Energy S.A. (NASDAQ: TURB) Announces Interim Results for Period Ended June 30, 2023, Transition in Management Structure
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) the West’s Strategy to Weaken China’s Minerals Dominance with its RapidSX(TM) Technology
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Enters Strategic MOU to Collaborate on Domestic Uranium Fuel Supply
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - Vision Marine Technologies Inc. (NASDAQ: VMAR) Partners with Leading Florida Boat Rental in Win-Win-Win Expansion
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Releases Q2 2024 Financial Report, Corporate Update
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) to Host Pioneering Conference for Women in Medical, Dental Sectors
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Bolsters BolaWrap® 150 Deployments, Promoting Safer Communities Nationwide
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































