The QualityStocks Daily Stock List
- NuCana (NCNA)
- Aptinyx (APTX)
- Invacare Corporation (IVC)
- Achieve Life Sciences (ACHV)
- POET Technologies (POET)
- FGI Industries (FGI)
- EC Healthcare (UNHLF)
- Longeveron Inc. (LGVN)
- Medirom Healthcare Technologies (MRM)
- Microbot Medical (MBOT)
- Lucid Motors (LCID)
- Southern Copper Corporation (SCCO)
NuCana (NCNA)
StockMarketWatch, QualityStocks, MarketClub Analysis, MarketBeat, TraderPower, Wealth Insider Alert, TradersPro, Schaeffer's, InvestorPlace, FreeRealTime and BUYINS.NET reported earlier on NuCana (NCNA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
NuCana PLC (NASDAQ: NCNA; FRA: NO4A) is a clinical-stage biotechnology firm which develops new treatments for treating breast, ovarian, pancreatic, biliary, hematology and colorectal cancers, which may better how chemotherapy treatments fight cancer.
The biopharmaceutical firm uses ProTide, its proprietary technology platform to manufacture cancer medications that are better tolerated by patients as well as more effective. This technology was developed by the late Dr. Christopher McGuigan at Cardiff University.
NuCana PLC was founded on January 28, 1997 by Christopher B. Wood and Hugh S. Griffith, who is its current CEO. The firm, which was formerly known as NuCana BioMed Limited, changed its name in August 2017 before launching its IPO shortly after, in September. The company’s IPO generated $115 million. NuCana PLC serves customers in the United States as well as the U.K. and has its headquarters in Edinburgh, United Kingdom.
The company, which has a license agreement, collaboration and research with University College Cardiff Consultants Ltd and Cardiff University, has two primary ProTide technology candidates; NUC-3373 and Acelarin. The firm recently begun clinical trials for these candidates, where the former is in its Phase 1 clinical trial for the treatment of advanced solid tumors while the latter is in different phases of various clinical trials to examine the candidates’ efficacy in treating metastatic pancreatic cancer, biliary cancer, platinum-resistant ovarian cancer and biliary tract cancer. NuCana PLC is also conducting a phase 1 clinical trial for NUC-7738, its candidate designed to treat hematological tumors as well as advanced solid tumors.
Cancer is among the leading causes of death in the world. Research for a cancer cure has been ongoing for decades with no luck yet. However, the clinical trials being conducted by the firm may soon bear fruit. If NuCana does succeed in finding effective alternative treatments for various cancers, not only will their research be worth a lot, so will their stocks.
NuCana (NCNA), closed Tuesday's trading session at $1.49, up 21.1382%, on 299,562 volume. The average volume for the last 3 months is 597,916 and the stock's 52-week low/high is $0.5171/$3.3197.
Aptinyx (APTX)
MarketBeat, StockMarketWatch, TradersPro, The Online Investor, StreetInsider, MarketClub Analysis, Top Pros' Top Picks, Schaeffer's, QualityStocks, InvestorPlace and Zacks reported earlier on Aptinyx (APTX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Aptinyx Inc. (NASDAQ: APTX) is a clinical-stage biopharmaceutical firm that is engaged in discovering, developing and commercializing new, synthetic and proprietary small molecules for treating nervous system and brain disorders.
The firm has its headquarters in Evanston, Illinois and was incorporated in June 2015 by Joseph R. Moskal and Norbert G. Riedel. It operates as part of the biotechnology research services industry, under the health care sector, in the biotech and pharma sub-industry. The firm serves consumers in the U.S. and has two companies in its corporate family.
The enterprise has a platform used for the discovery of proprietary compounds that work via NMDA receptor modulation, which improves pathways involved in nerve cell communication. It is party to a research collaboration agreement with Allergan Plc.
The company’s product pipeline is made up of an NMDA receptor modulator dubbed NYX-458, which is currently undergoing phase 1 clinical trials to evaluate its effectiveness in treating Parkinson’s disease cognitive impairment; and an NMDA receptor modulator known as NYX-783, which has been indicated for the treatment of post-traumatic stress disorder. In addition to this, it also develops NYX-2925, which has been indicated for treating painful diabetic peripheral neuropathy. The company also develops the AGN-241751 program.
The firm’s NYX-2925 was recently granted Fast Track Designation by the FDA for the treatment of pain related to diabetic peripheral neuropathy. This move will not only benefit patients who suffer from neuropathic pain linked to diabetic peripheral neuropathy if the treatment is approved by the FDA but also encourage more investments into the firm, which will in turn boost growth.
Aptinyx (APTX), closed Tuesday's trading session at $0.4301, up 18.4848%, on 599,049 volume. The average volume for the last 3 months is 488,783 and the stock's 52-week low/high is $0.20/$3.7599.
Invacare Corporation (IVC)
MarketBeat, QualityStocks, StreetInsider, StockMarketWatch, BUYINS.NET, InvestorPlace, Zacks, MarketClub Analysis, PoliticsAndMyPortfolio, Street Insider, StreetAuthority Daily, The Stock Dork, The Street, MicroCap Press, TopPennyStockMovers, Marketbeat.com, TradersPro, Kiplinger Today, Trades Of The Day, First Penny Picks, Dynamic Wealth Report and SmarTrend Newsletters reported earlier on Invacare Corporation (IVC), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Invacare Corporation (NYSE: IVC) (FRA: IV3) is focused on manufacturing, distributing and exporting medical equipment used in extended care, retail and healthcare markets globally.
The firm has its headquarters in Elyria, Ohio and was incorporated in 1979 by Aaron Malachi Mixon III. It serves consumers across the globe.
The company operates through the Asia/Pacific, Europe and North America geographical segments, which comprise of the institutional products group and home medical equipment segments. It sells its products mainly to government health service customers, dealers and residential care operators via its sales force, as well as to home medical equipment providers, via e-commerce and retail channels. The majority of the company’s revenue is derived from the international markets.
Under the PinDot brand, the enterprise provides custom molded seat modules while under the Invacare brands, it provides seating and positioning products, custom manual wheelchairs and mobility and seating products, like power wheelchairs. It also offers lifestyle products, which include institutional and residential care beds and accessories; safe patient handling products under the ISA, Evo and Birdie brands; and pressure relieving mattress systems and overlays under the Invacare microAIR and Softform brands. The enterprise also provides respiratory therapy products, which include HomeFill oxygen systems, portable oxygen concentrators and stationary oxygen concentrators.
The firm is focused on positioning the company for long-term success and growth through its detailed growth strategy, which involves the expansion of new products in all of its segments. It is currently focused on improving its profitability, optimizing its business and meeting its consumers’ demands.
Invacare Corporation (IVC), closed Tuesday's trading session at $0.6308, up 20.1524%, on 488,941 volume. The average volume for the last 3 months is 385,249 and the stock's 52-week low/high is $0.33/$2.845.
Achieve Life Sciences (ACHV)
StockMarketWatch, MarketBeat, BUYINS.NET, QualityStocks, Schaeffer's, MarketClub Analysis, TraderPower, StreetInsider, The Online Investor, Small Cap Firm, InvestorsUnderground and INO Market Report reported earlier on Achieve Life Sciences (ACHV), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Achieve Life Sciences Inc. (NASDAQ: ACHV) (FRA: SP4P) is a clinical-stage pharmaceutical firm that is focused on commercializing cytisinicline for nicotine addiction and smoking cessation.
The firm has its headquarters in Vancouver, Canada and was incorporated in October 1991. It operates as part of the pharmaceutical and medicine manufacturing industry, under the health care sector. The firm has six companies in its corporate family and serves consumers around the globe, with a focus on North America.
The company’s objective is to address the international smoking health epidemic through cytisinicline commercialization. It is committed to the advancement of cytisinicline as a widely available treatment option for people with nicotine addiction. The company is party to license agreements with the University of Bristol and Sopharma AD.
The enterprise’s products include cytisinicline, which is a plant-based alkaloid extracted from the seeds of a plant known as Laburnum anagyroides. It interacts with nicotine receptors in the brain to reduce the satisfaction and reward linked to nicotine through antagonistic properties and decreases nicotine withdrawal symptoms severity through agonistic effects on nicotine receptors. The enterprise also develops Apatorsen, which blocks the production of a cell-survival protein known as heat shock protein 27, which is expressed in different types of cancer, including pancreatic, breast and prostate cancers.
The firm recently released its third quarter financial results for 2021, with its CEO noting that they were focused on meeting the firm’s key development milestones. This comes after the FDA accepted its Investigation New Drug application for its cytisinicline product for smoking cessation.
Achieve Life Sciences (ACHV), closed Tuesday's trading session at $3.43, up 22.5%, on 385,249 volume. The average volume for the last 3 months is 300,832 and the stock's 52-week low/high is $2.00/$8.60.
POET Technologies (POET)
MarketBeat reported earlier on POET Technologies (POET), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
POET Technologies Inc. (NASDAQ: POET) is a technology development firm that is focused on designing, developing, manufacturing and selling integrated and discrete opto-electronic solutions.
The firm has its headquarters in Toronto, Canada and was incorporated in 1985, on November 14th. Prior to its name change in June 2013, the firm was known as Opel Technologies Inc. The firm mainly serves consumers in Singapore, the United States and Canada.
The company’s vision is to be an international leader in chip-scale photonic solutions by using its technology to enable the seamless integration of electronics and photonics for an extensive range of vertical market applications. It operates through one segment which involves the manufacture and sale of semiconductor products for commercial applications. The company serves the on-board optic, automotive LIDAR, industrial sensing and Internet of Things, telecommunications and data center markets.
The enterprise provides integration solutions based on its Optical interposer technology which integrates photonic and electronic devices into one multi-chip module through the use of advanced wafer-level semiconductor manufacturing methods and packaging techniques. Its interposer platform does so at the lowest cost, in addition to offering maximum scalability and flexibility. The enterprise is also involved in the development of photonic integrated components.
The company recently announced its second quarter financial results which show that it achieved significant milestones during the period. It is currently focused on growing product development activity, executing its strategic plan and realizing its vision to become an international leader in chip-scale photonic solutions, which will be good for investments into the company.
POET Technologies (POET), closed Tuesday's trading session at $4.58, up 40.4908%, on 300,832 volume. The average volume for the last 3 months is 14,643 and the stock's 52-week low/high is $2.34/$11.00.
FGI Industries (FGI)
MarketBeat reported earlier on FGI Industries (FGI), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
FGI Industries Ltd (NASDAQ: FGI) is a business to business (B2B) company focused on supplying kitchen and bath products.
The firm has its headquarters in East Hanover, New Jersey and was incorporated in 2021, on May 26th. It operates as part of the furnishings, fixtures and appliances industry, under the consumer cyclical sector. The firm serves consumers around the globe, with a focus on those in Canada, the United States and Europe.
The company is focused on servicing its retail, wholesale and commercial channel customers globally and offering an extensive range of products to serve their needs. It operates as a Foremost Groups Limited subsidiary.
The enterprise’s products fall under the Sanitaryware, Bath Furniture and Others categories. It sells a range of sanitaryware products, including sinks, toilets, toilet seats and pedestals. It also sells various products under its bath furniture category, including wood and wood-substitute furniture for bathrooms, like mirrors, vanities, medicine cabinets, laundry and other storage systems; and shower doors and shower systems. Products under the Others category include customer kitchen cabinetry and other accessory items under the Avenue, Foremost, Rosenberg, Craft + Main, Covered Bridge Cabinetry, contrac, Jetcoat, FGI Germany and Covered Bridge Cabinetry brand names. It sells its products through online retailers, mass retail centers, wholesale and commercial distributors, independent dealers, and distributors.
The firm, which recently released its latest financial results, remains focused on expanding its geographic footprint into Australia and the United Kingdom. This will help grow the firm’s global presence while also opening it up to new growth and investment opportunities.
FGI Industries (FGI), closed Tuesday's trading session at $2.38, up 5.7778%, on 14,643 volume. The average volume for the last 3 months is 200 and the stock's 52-week low/high is $1.9483/$6.25.
EC Healthcare (UNHLF)
We reported earlier on EC Healthcare (UNHLF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
EC Healthcare (OTC: UNHLF) (FRA: TUM0) (HKG: 2138) is an investment holding firm focused on providing medical and healthcare services.
The firm has its headquarters in Mong Kok, Hong Kong and was incorporated in 2005, on November 23rd by Chi Fai Tang. Prior to its name change in April 2021, the firm was known as Union Medical Healthcare Ltd. It operates as part of the household and personal products industry, under the consumer defensive sector. The firm serves consumers in Macau, the People’s Republic of China and Hong Kong.
The company operates through the Medical, Aesthetic medical and Beauty and wellness, and Others segments. The Medical segment is engaged in the provision of medical services while the Aesthetic medical and Beauty and wellness segment offers various services, including medical aesthetics and dental services, as well as traditional beauty, hair care and supplemental wellness services. This segment also sells beauty, skincare and healthcare products. The Other segment offers marketing services and veterinary services. The medical segment generates the majority of the company’s revenue.
The enterprise also provides administrative, management, laboratory testing, performance marketing, marketing, and travel agency services; and operates in healthcare machines and property investment holding businesses. The enterprise provides its products and services primarily under private-label brands, such as Swissline, PRODERMA LAB, Suissebeaute, and re:HEALTH.
The company remains focused on becoming the leading and client-centric one-stop medical healthcare service provider in Asia. This will allow it to occupy a larger share of the market and help generate value for its shareholders.
EC Healthcare (UNHLF), closed Tuesday's trading session at $1.14, even for the day. The average volume for the last 3 months is 52,213 and the stock's 52-week low/high is $0.67/$1.14.
Longeveron Inc. (LGVN)
MarketClub Analysis, InvestorPlace, QualityStocks, ProTrader, MarketBeat, TradersPro, StockStreetWire, StocksEarning, Schaeffer's and MicroCapDaily reported earlier on Longeveron Inc. (LGVN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Longeveron Inc. (NASDAQ: LGVN) is a clinical stage biotechnology firm that is focused on the development of cellular therapies for life-threatening and aging-related conditions.
The firm has its headquarters in Miami, Florida and was incorporated in 2014, on October 9th by Donald M. Soffers and Joshua M. Hare. It operates as part of the biotechnology industry, under the healthcare sector. The firm serves consumers around the globe.
The company hopes to improve healthy aging through regenerative medicine approaches. It sponsors clinical trials in various indications, including Aging Frailty, Alzheimer's disease, Metabolic Syndrome, Acute Respiratory Distress Syndrome (ARDS), and Hypoplastic left heart syndrome (HLHS). The company has concluded 2 multi-center trials in the U.S. for aging frailty.
The enterprise’s lead investigational product, dubbed the LOMECEL-B (LMSC), is a cell-based therapy product that has been derived from culture-expanded medicinal signaling cells (MSCs) that are sourced from the bone marrow of young, healthy, adult donors. It is evaluating Lomecel-B as a therapy for aging frailty and is being designed to reduce inflammation associated with aging frailty and to promote an anti-inflammatory state by releasing anti-inflammatory molecules, which can promote physiological restoration to a more normal state.
The company recently released a corporate update, with its CEO noting that they remained focused on building on their progress in HLHS and Alzheimer’s. The success and approval of its formulations will not only help meet the currently unmet needs of patients with various indications but also encourage more investments into the company.
Longeveron Inc. (LGVN), closed Tuesday's trading session at $3.35, up 1.5152%, on 52,213 volume. The average volume for the last 3 months is 576 and the stock's 52-week low/high is $2.80/$16.77.
Medirom Healthcare Technologies (MRM)
SmallCapVoice, QualityStocks, MarketClub Analysis, MarketBeat, InvestorPlace and Greenbackers reported earlier on Medirom Healthcare Technologies (MRM), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Medirom Healthcare Technologies Inc. (NASDAQ: MRM) is a health services provider engaged in the provision of holistic healthcare services.
The firm has its headquarters in Tokyo, Japan and was incorporated in July 2000 by Koji Eguchi. Prior to its name change in March 2020, the firm was known as Medirom Inc. It operates as part of the personal services industry, under the consumer cyclical sector. The firm primarily serves consumers in Japan.
The enterprise operates through the Relaxation Salon, Digital Preventative Healthcare, and Luxury Beauty segments. The Relaxation Salon segment owns and franchises relaxation salons, which offer stretch therapy, finger-pressure style bodywork therapy, and posture and joint alignment, as well as physical therapy elements; and different individual services, including athletic support therapy, reflexology, anti-fatigue therapy and slim-down therapy. As of December 2021, this segment operated 312 relaxation salons under the Ruam Ruam and Re.Ra.Ku brand. The Digital Preventative Healthcare segment provides government-sponsored Specific Health Guidance programs, using Lav, its internally developed on-demand health monitoring smartphone application; MOTHER Tracker for fitness applications; and preventative healthcare services using its digital application and devices. On the other hand, the Luxury Beauty segment manages and operates hair salons under the ZACC brand name.
The company remains committed to increasing its sales, serving even more customers and expanding its footprint through the launch of new locations. This will generate additional revenue while opening the company up to new growth and investment opportunities which will positively influence shareholder value.
Medirom Healthcare Technologies (MRM), closed Tuesday's trading session at $4.72, off by 0.033887%, on 576 volume. The average volume for the last 3 months is 12,632 and the stock's 52-week low/high is $4.0101/$9.80.
Microbot Medical (MBOT)
MarketBeat, StockMarketWatch, MarketClub Analysis, QualityStocks, TraderPower, StocksEarning, BUYINS.NET, Schaeffer's, Jason Bond, PennyStockScholar, Buzz Stocks, HotOTC, OTCtipReporter, Penny Pick Finders, PennyStockProphet, PoliticsAndMyPortfolio, Profitable Trader Authority, Promotion Stock Secrets, StockOnion and Planet Penny Stocks reported earlier on Microbot Medical (MBOT), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Microbot Medical Inc. (NASDAQ: MBOT) (FRA: CY9D) is a pre-clinical medical device firm that is focused on conducting research on, designing and developing robotic endoluminal surgery devices that target the minimally invasive surgery space.
The firm has its headquarters in Hingham, Massachusetts and was incorporated in 1988, on August 2nd by Yosseph Bornstein, Moshe Shoham and Harel Gadot. It operates as part of the medical instruments and supplies industry, under the healthcare sector. The firm serves consumers in the United States.
The company is primarily focused on leveraging its micro-robotic technologies with the goal of improving surgical outcomes for patients. It is party to a strategic collaboration agreement with Stryker Corp for technology co-development.
The enterprise carries out its operations through the ViRob and TipCat platforms. The ViRob platform technology is an autonomous crawling micro-robot that can be controlled remotely or within the body. It uses the Self-Cleaning Shunt, a robotic system designed as the ventricular catheter portion of a Cerebrospinal Fluid shunt system and is mainly used for the treatment of hydrocephalus and Normal Pressure Hydrocephalus. The TipCat platform is a self-propelling, flexible, and semi-disposable endoscope, provides see-and-treat capabilities within tubular lumens in the human body such as the colon, blood vessels, and the urinary tract. It is also focused on the development of a multi generation pipeline portfolio. The enterprise has 42 issued/allowed patents and 23 patent applications pending worldwide.
The firm remains focused on strengthening its intellectual property portfolio and advancing its global commercialization strategy. Its success will bring in additional revenues and investments into the firm while also generating value for its shareholders.
Microbot Medical (MBOT), closed Tuesday's trading session at $3.55, up 1.1396%, on 12,991 volume. The average volume for the last 3 months is 25.981M and the stock's 52-week low/high is $3.00/$7.6399.
Lucid Motors (LCID)
Green Car Stocks, InvestorPlace, Schaeffer's, QualityStocks, MarketClub Analysis, The Street, MarketBeat, StocksEarning, Early Bird, Trades Of The Day, Daily Trade Alert, Kiplinger Today, The Online Investor, Investopedia, The Wealth Report and Louis Navellier reported earlier on Lucid Motors (LCID), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Automakers in China are able to make an EV for 10,000 fewer euros, or $10,618, which is below what their competitors in Europe can charge. The difference is due to their low R&D and capital expenditure costs, as well as low labor costs when compared to their European rivals. According to Patrick Koller, CEO of Forvia, a supplier of vehicles, this denotes massive cost savings, which will pressurize the European car makers in their local markets.
Speaking las week at the CES convention in Las Vegas, Koller acknowledged that the cars made in China were good and that it was going to be difficult for Europe to stop importing them since consumers in Europe continue to look for affordable EVs. He added that the situation is worse in Europe as compared to the United States because China currently accounts for 5.8% of all electric vehicle sales in Europe; those numbers are according to Inovev, a French automotive consultancy, which forecasts a sharp increase over the next few years as brands in China continue to produce more affordable models. China’s share of the U.S. auto market, on the other hand, has remained minimal due to high import taxes in the United States on vehicles made in China.
According to a JATO Dynamics study that gives industry trending analysis, since the year 2015, the average cost of an EV in China has decreased from $70,203 to $33,440, making it significantly less expensive than the cost of a gasoline-powered vehicle. In contrast, the cost of EVs has increased since 2015 in Europe from $51,424 to $58,652 and from $53,038 to $63,864 in the United States.
China’s automakers are in a rush to increase vehicle deliveries in Europe, as are other global automakers such as Tesla, because Europe is largely open to importing China-made cars. Chinese products have been rated positively by the regulators in Europe, dispelling the myth that their vehicles are of low quality.
According to Koller, the prospect for the demand of vehicles worldwide in 2023 is unclear. A resolution to the Ukrainian conflict could well improve the situation, but a longer, more intense conflict could result in the worst possible outcome.
According to Koller, Forvia will increase its investments in the United States in order to benefit from the federal incentives offered by the Inflation Reduction Act, which was passed into law in August. He further stated that he anticipates the introduction of a hydrogen-powered pickup in the United States by 2025, because Forvia foresees the possibility of equipping pickup trucks in North America with hydrogen power technology.
The increasing competition from Chinese EV manufacturers should give startups from other regions such as Lucid Motors (NASDAQ: LCID) plenty to think about as they make their long-term expansion plans.
Lucid Motors (LCID), closed Tuesday's trading session at $7.19, up 6.0472%, on 25,980,588 volume. The average volume for the last 3 months is 1.86M and the stock's 52-week low/high is $6.09/$47.59.
Southern Copper Corporation (SCCO)
SmarTrend Newsletters, MarketBeat, InvestorPlace, The Street, Louis Navellier, The Online Investor, Daily Wealth, QualityStocks, Daily Trade Alert, The Wealth Report, TopStockAnalysts, Trades Of The Day, StreetAuthority Daily, Marketbeat.com, Zacks, Barchart, Money Morning, TheStockAdvisor, Early Bird, Schaeffer's, Market Intelligence Center Alert, Kiplinger Today, Uncommon Wisdom, The Growth Stock Wire, Investopedia, Market Authority, Top Pros' Top Picks, MarketClub Analysis, TheStockAdvisors, ChartAdvisor, The Stock Enthusiast, INO.com Market Report, StreetInsider, InvestmentHouse, Investiv, Investing Futures, Greenbackers, Forbes, Investor Update, Dividend Opportunities, CRWEWallStreet, CRWEPicks, CRWEFinance, BestOtc, AllPennyStocks, DrStockPick, StockLockandLoad, Wealth Insider Alert, Wealth Daily, Vantage Wire, TradingMarkets, TradingAuthority Daily, The Tycoon Report, The Trading Report, The Motley Fool, MarketDNA, StockRockandRoll, InvestorGuide, StockHotTips, Profit Confidential, PennyToBuck, PennyOmega, Navellier Growth, Money and Markets, 24/7 Trader, InvestorsObserver Team, InvestorIntel and Streetwise Reports reported earlier on Southern Copper Corporation (SCCO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Precious metals trader Heraeus Group predicts that several geopolitical and economic factors will play a major role in silver, platinum and gold markets through 2023. Last year proved to be extremely volatile, with the Russia-Ukraine war slowing down the already weak global economy and lockdowns in China crippling the country’s economy and impacting supply chains around the world. Prices for commodities such as gold, palladium and silver rallied in the wake of the Ukraine invasion but saw their prices settle at lower levels as the war waged on.
With most of the world still grappling with rising inflation and increased cost of living, experts predict slower economic growth in 2023. Signs point to a recession in major markets, including the United States and Europe, this year amid slowed economic growth; precious metals traders believe this will likely weigh on the commodities markets.
Gold prices, for starters, have been kept in check by a strong greenback despite decade-high inflation levels that would have traditionally increased gold’s haven metal appeal and raised its prices. This is due to the fact that the U.S. Federal Reserve has consistently raised benchmark interest rates for several months to forestall inflation, increasing the opportunity cost of holding nonyielding bullion and decreasing its appeal to investors because it doesn’t earn any interest.
If the Fed continues with its aggressively tight monetary policy into the new year, more investors may move away from gold to other assets that can allow them to leverage the higher interest rates and secure a greater ROI. On the other hand, Heraeus says, the Fed may loosen its stance amid a weakening economy, causing the dollar to lose some of its strength and increasing the value of gold and silver holdings.
According to Heraeus, investors may see a record high return on their gold holdings “in euro terms” if the Fed keeps benchmark interest rates stable or reduces them. Estimates from the Germany-based precious metals trader show that the Federal Bank’s actions could cause gold and silver to trade between $1,620 per ounce to $1,920 per ounce and $17 per ounce and $25 per ounce respectively in 2023.
As with gold, platinum also saw its prices drop as the dollar surged in late 2022; the metal should see an upward trend if the greenback weakens this year. However, the platinum market remains oversupplied by more than 400,000 ounces, and the metal is predicted to trade at between $800 to $1,150 per ounce.
The market movements in the commodities industry are likely to impact the short-term performance of extraction firms such as Southern Copper Corporation (NYSE: SCCO), especially if a recession takes root.
Southern Copper Corporation (SCCO), closed Tuesday's trading session at $71.82, up 3.8161%, on 1,880,220 volume. The average volume for the last 3 months is 310 and the stock's 52-week low/high is $42.42/$79.315.
The QualityStocks Company Corner
- QSAM Biosciences Inc. (OTCQB: QSAM)
- InMed Pharmaceuticals Inc. (NASDAQ: INM)
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF)
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF)
- Aditxt Inc. (NASDAQ: ADTX)
- Data443 Risk Mitigation Inc. (OTC: ATDS)
- India Globalization Capital Inc. (NYSE American: IGC)
- MetAlert Inc. (OTC: MLRT)
- Cub Crafters Inc.
- REZYFi, Inc.
- Silo Pharma Inc. (OTCQB: SILO)
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO)
QSAM Biosciences Inc. (OTCQB: QSAM)
The QualityStocks Daily Newsletter would like to spotlight QSAM Biosciences Inc. (OTCQB: QSAM).
The letter outlined goals for 2023 to complete the phase 1 study,secure capital through an underwritten offering and a concurrentNASDAQ uplisting. “In reaching these goals, shareholders shouldexpect to see progress through several nearer-term milestones, suchas: securing additional trial sites and starting enrollment ofpatients at those sites; completing patient cohorts (groupings) inour phase 1 trial, which will lead to escalating doses insubsequent cohorts; establishing a more robust supply chain throughthe qualification of additional nuclear reactors and other keyvendors; and other important achievements,” said the QSAMexecutives in the shareholder letter. “We are looking forward toprogress in 2023, which we expect will create fundamental value forour shareholders. While we cannot predict the tide of externalmarket forces, we are confident in our team, our technology, andour ability to advance CycloSam through the FDA process.Ultimately, however, our primary mission is to help the hundreds ofthousands of adults and children each year suffering from bonecancer.”
To view the full letter, visit https://ibn.fm/Ij7dL
QSAM Biosciences Inc. (OTCQB: QSAM) is a clinical stage biotechnology company focused on bringing to market targeted therapeutic radiopharmaceuticals. The company is committed to advancing the fight against cancer through the discovery, development and delivery of effective treatment options for adult and pediatric patients.
QSAM Biosciences was founded in 2020 by Executive Chairman Dr. C. Richard Piazza and CEO Douglas Baum. It is headquartered in Austin, Texas.
CycloSam®
CycloSam®, QSAM Biosciences’ initial technology, is a clinical-stage bone targeting radiopharmaceutical invented by world-renowned scientists at IsoTherapeutics Group LLC. By leveraging a patented, low specific activity form of Samarium-153 (resulting in far less undesirable europium impurity) and what management believes to be a superior chelating agent in DOTMP, CycloSam is designed to selectively target sites of high bone mineral turnover to deliver a prescribed tumor-killing dose of radiation to the bone tumor sites while minimizing radiation exposure to nearby healthy tissue. These parameters are currently being tested in an FDA-cleared clinical trial.
CycloSam® has been shown in laboratory testing to cause significantly less (30x less) buildup of long-lived radionuclidic impurities than prior FDA-approved drugs, which management believes will enable the ability to safely administer therapeutic doses via higher and multiple-dose regimens and effectively expand its potential clinical utility to therapeutic uses in areas of high unmet medical needs.
The indications for CycloSam® currently being evaluated by QSAM Biosciences include:
- Metastatic Bone Cancers – On April 28, 2022, QSAM Biosciences announced that the first patient had commenced treatment in its clinical trial evaluating CycloSam in patients with metastatic bone cancer. As noted in the release, the study is a Phase 1 open-label, dose-escalation trial to evaluate the safety, tolerability, dosimetry, and preliminary efficacy of CycloSam®.
- Pediatric Osteosarcoma/Ewing’s Sarcoma – On February 2, 2022, the company announced that the U.S. FDA has granted Rare Pediatric Disease Designation to CycloSam for the treatment of osteosarcoma. Combined with a previously granted orphan drug designation for osteosarcoma received in 2021, this milestone “may allow QSAM to potentially bring CycloSam® to market more rapidly through additional incentives and eligibilities,” according to CEO Douglas Baum.
- Bone Marrow Ablation – In a 2020 single patient Investigational New Drug (IND) study, an investigator concluded that high-dose CycloSam® can be administered safely to ablate bone marrow in advance of a stem cell transplant with no apparent renal toxicity and no unexpected adverse events attributable to the drug.
QSAM Biosciences’ preclinical and clinical development pipeline is supported by a strong IP portfolio. The company has secured 14 patents across three distinct patent families spanning the U.S., Japan, Canada and the European Union.
Market Outlook
Through its ongoing development of CycloSam®, QSAM Biosciences is targeting multiple large and underserved market opportunities. According to the American Cancer Society, roughly 400,000 new cases of malignant bone metastasis are diagnosed annually in the U.S. alone. Additionally, QSAM will pursue indications for osteosarcoma and Ewing’s sarcoma that are the most common primary malignancies of bone tissues in children.
Despite this pressing need, the current standard of care for bone cancer is aggressive and suboptimal, leading to marginal success with significant side effects and poor long-term survival prognosis. As a result, QSAM Biosciences estimates a sizable market opportunity for its development pipeline.
- Bone Metastasis has an estimated total addressable market of $20 billion in the U.S. based on total new cases and comparable drug pricing.
- Osteosarcoma/Ewing’s Sarcoma have a total addressable market of roughly $125 million in the U.S. based on approximately 1,000 new cases in 2021.
- The total addressable market for Bone Marrow Ablation is projected at $1 billion, with an estimated 32,000 procedures completed annually.
The company anticipates that the ability to administer CycloSam® for higher and multiple-dose regimens may expand its clinical utility for therapeutic uses in additional areas of high unmet medical needs.
Management Team
QSAM Biosciences is led by an experienced management team and board with an extensive record of FDA approvals, big pharma partnerships and M&A transactions.
Dr. C. Richard Piazza is the Executive Chairman of QSAM Biosciences. Since 2017, he has also served as President and CEO of IGL Pharma Inc., the licensor of CycloSam®, and as a consultant to IsoTherapeutics Group LLC, the inventors of the technology. Dr. Piazza also currently serves on the board of directors of NovaScan LLC, a privately held cancer detection and diagnostics company. He has more than 48 years of health care experience in both medical devices and pharmaceutical/biotech and has led several technology companies to market success, including numerous FDA approvals in both sectors. Dr. Piazza obtained a BS in Economics and a BS in Speech Pathology from the State University of New York and an MA & PhD in Economics from the University of Buffalo and Leeds University.
Douglas R. Baum is the company’s CEO and Director. He brings to QSAM Biosciences over 30 years of experience in the bioscience and biotech industries, including development, FDA/EMA approval and commercialization of multiple drugs and medical devices. Mr. Baum has overseen 15 product approvals through the FDA and EMA and raised over $85 million in capital to fund breakthrough technologies. From 2017 to 2020, he consulted with multiple medical schools and biotech and pharmaceutical companies, and, from 2012 to 2017, he served as President, Chief Executive Officer and Director of Xeris Pharmaceuticals Inc. Mr. Baum holds a Master of Science in Technology Commercialization and a BBA in International Business and Marketing from the University of Texas.
Adam King is the CFO of QSAM Biosciences. He is also the Founder and CEO of King Consulting Group, where he provides a range of financial and reporting services for clients. Before founding King Consulting Group in January 2021, Mr. King was the CFO for Netsertive, a venture-backed digital marketing company. From 2016 to 2018, he was the Office Managing Audit Director for BDO’s Greenville, South Carolina, office, in addition to serving as Audit Director in Raleigh, North Carolina, and Boston, Massachusetts. While at BDO, Mr. King worked with various clients, from tech and life science start-ups to billion-dollar publicly traded companies. He holds a Bachelor of Science in Accounting from Elon University and is a CPA in Raleigh, North Carolina.
QSAM Biosciences Inc. (OTCQB: QSAM), closed Tuesday's trading session at $5, up 1.2658%, on 310 volume. The average volume for the last 3 months is 4.692M and the stock's 52-week low/high is $3.50/$14.00.
Recent News
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Releases Shareholder Letter Outlining 2022 Updates, 2023 Milestone Goals
- New Treatment Approach for Cancer Combines Bacterial, Pharmaceutical Therapy
- Research Suggests That Vegetarian Diets Decrease Prostate Cancer Risk
InMed Pharmaceuticals Inc. (NASDAQ: INM)
The QualityStocks Daily Newsletter would like to spotlight InMed Pharmaceuticals Inc. (NASDAQ: INM).
To view the full press release, visit https://ibn.fm/gex4Q
InMed Pharmaceuticals Inc. (NASDAQ: INM) is a global leader in the manufacturing and clinical development of rare cannabinoids. InMed is a clinical stage company developing cannabinoid-based pharmaceutical drug candidates, as well as manufacturing technologies for pharmaceutical-grade rare cannabinoids.
The company is dedicated to delivering new therapeutic alternatives to treat conditions with high unmet medical needs. The company is also developing a proprietary manufacturing technology to produce pharmaceutical-grade rare cannabinoids in the lab and has recently announced an LOI to acquire a leading rare cannabinoid manufacturer.
Research and Technology
There are more than 100 rare cannabinoids found in only trace amounts in the cannabis plant, together making up less than 1% of the plant’s biomass. InMed is initially focused on the therapeutic benefits of cannabinol (CBN) in diseases with high unmet medical need. Preclinical studies of CBN demonstrated an excellent safety profile and showed CBN has potential for therapeutic benefit over other cannabinoids such as tetrahydrocannabinol (THC) and cannabidiol (CBD).
Evidence suggests there may be great therapeutic potential in rare cannabinoids. Each has a specific chemical structure, and different cannabinoids have been observed to have distinct physiological properties in humans, including therapeutic potential for specific diseases as well as unique safety profiles. CBN is the active pharmaceutical ingredient (API) in InMed’s two lead programs for dermatological and ocular diseases.
InMed’s most advanced compound, INM-755, is a CBN topical cream under clinical development for the treatment of epidermolysis bullosa, a severe genetic skin disorder. To date, INM-755 has been evaluated in two Phase 1 clinical trials in healthy volunteers. InMed has filed Clinical Trial Applications in several countries as part of a global Phase 2 clinical trial of INM-755 (cannabinol) cream in epidermolysis bullosa. Responses from the National Competent Authorities and Ethics Committees are expected throughout the summer of 2021.
InMed is also involved in developing INM-088, an ocular CBN formulation being researched for the treatment of glaucoma, the second leading cause of blindness in the developed world. InMed is currently evaluating several formulations to deliver CBN into the eye to address issues of dosing frequency, side effects and treatment penetration. INM-088 is being designed for topical delivery to the eye. This localized delivery results in very little drug being absorbed or migrating into the bloodstream, thus minimizing potential adverse side effects. INM-088 shows promise to reduce intraocular pressure and provide neuroprotection of the eye.
Manufacturing
The limited availability of rare cannabinoids like CBN makes them economically impractical to extract directly from the plant for pharmaceutical use. InMed is developing IntegraSyn, a cannabinoid synthesis manufacturing system to create rare cannabinoids in the lab that are bioidentical to the compounds derived from the cannabis plant. IntegraSyn uses multiple standard pharmaceutical processes and has achieved a cannabinoid yield of 5 grams per liter, surpassing commercial viability and significantly exceeding currently reported industry yields. InMed is now focusing on manufacturing scale-up to larger batch sizes while continuing process optimization, targeting increased cannabinoid yield and further reducing overall cost of goods.
BayMedica Inc. Acquisition
On June 29, 2021, InMed announced it had entered into a non-binding letter of intent to acquire BayMedica Inc., a private company based in Nevada and California that specializes in the manufacture and commercialization of rare cannabinoids.
As noted in the news release, BayMedica is a revenue-stage biotechnology company leveraging its significant expertise in synthetic biology and pharmaceutical chemistry to develop efficient, scalable and proprietary manufacturing approaches to produce high quality, regulatory-compliant rare cannabinoids for consumer applications. BayMedica is currently commercializing the rare cannabinoid CBC (cannabichromene) as a B2B supplier to distributors and manufacturers marketing products in the health and wellness sector. BayMedica is planning additional rare cannabinoid launches for the coming year.
Pursuant to the indicative terms of the LOI, InMed and BayMedica intend to negotiate and enter into a definitive agreement under which InMed would acquire 100% of BayMedica in exchange for 1.6 million InMed common shares to be issued to BayMedica’s equity and convertible debt holders, with any such issued InMed common shares being subject to a six-month contractual hold period.
Market Outlook
There is a rapidly growing demand for rare cannabinoids. However, their low natural concentration makes traditional harvesting of these compounds cost prohibitive. Biosynthesis allows production of rare cannabinoids in the lab that are bioidentical to compounds found in nature, with significantly higher yields which reduce costs. Biosynthesis can produce pharmaceutical-grade, bioidentical, THC-free compounds at a cost that’s 70 to 90 percent less than wholesale prices of naturally harvested rare cannabinoids.
Cannabinoid-based pharmaceuticals are expected to overtake the market as rare cannabinoids become less expensive and more available. According to Statista, the value of the consumer market for cannabinoid-based pharmaceuticals in the United States is forecast to grow to $25 billion by 2025 and to $50 billion by 2029, with cannabinoid-based pharmaceuticals used to treat health conditions including pain, respiratory conditions, autoimmune conditions and more.
Management Team
Eric A. Adams has been CEO and president of InMed since June 2016. He has more than 25 years of experience in establishing corporate entities, capital formation, global market development, mergers and acquisitions, licensing and corporate governance. He previously served as CEO at enGene Inc. Prior to enGene, he held senior positions in global market development with QLT Inc. (Vancouver), Advanced Tissue Sciences Inc. (La Jolla, CA), Abbott Laboratories (Chicago, IL) and Fresenius AG (Germany).
Bruce S. Colwill is InMed’s CFO. He has more than 25 years of financial leadership experience in public and private companies. Prior to InMed, he served as CFO of General Fusion Inc., a private clean energy company. He was also CFO at Entrée Resources Inc., a mineral exploration company, from 2011 to 2016. He has held CFO roles at Neuromed Pharmaceuticals Ltd., Response Biomedical Corp, Forbes Medi-Tech Inc. and Euronet Worldwide Inc.
Alexandra D.J. Mancini is Senior Vice President, Clinical and Regulatory Affairs at InMed. She has more than 30 years of global biopharmaceutical research and development experience. She has been an executive with numerous biotech companies, including senior vice president of Clinical and Regulatory Affairs at Sirius Genomics; senior vice president of Clinical and Regulatory Affairs at INEX Pharmaceuticals; and vice president of Regulatory Affairs at QLT Inc.
Eric C. Hsu is Senior Vice President, Pre-Clinical Research and Development at InMed. He joined InMed with more than 18 years of scientific leadership experience in the field of gene therapy. He has held various positions within enGene Inc., including vice president of Research and vice president of Scientific Affairs and Operations. He received his Doctorate from the Department of Medical Biophysics at the University of Toronto.
Michael Woudenberg is Vice President, Chemistry, Manufacturing and Controls at InMed. He has more than 20 years of successful drug development, process engineering, GMP manufacturing and leadership experience. He has held positions with 3M, Cardiome Pharma, Arbutus Biopharma and, most recently, was Managing Director of Phyton Biotech LLC.
InMed Pharmaceuticals Inc. (INM), closed Tuesday's trading session at $2.3, up 36.0947%, on 4,691,933 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $14.00/$.
Recent News
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Key Milestones, 2023 Business Outlook
- InvestorNewsBreaks – InMed Pharmaceuticals Inc. (NASDAQ: INM) Shares Annual General Meeting Results
- InMed Announces Results of 2022 Annual General Meeting
EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF)
The QualityStocks Daily Newsletter would like to spotlight EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF).
•Methane, such as that in livestock waste, is up to 50% more potentthan carbon dioxide in holding heat in the atmosphere
•EverGen is an Infrastructure platform that specializes in renewablenatural gas (“RNG”) projects converting diverse types of waste intousable, green alternatives to fossil fuels
•EverGen’s RNG capacity will exceed 2 million gigajoules annuallyonce its projects and expansions are complete in B.C., Alberta, andOntario
We are surrounded by untapped, atmosphere-friendly energy. The sunand wind are two popular sources today, but the problem is that itisn’t always sunny or windy. For
EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) is developing Canada’s Renewable Natural Gas Infrastructure Platform, starting on the west coast in British Columbia. The company is combating climate change and helping communities contribute to a sustainable future by acquiring, developing, building, owning and operating a portfolio of renewable natural gas (RNG), waste-to-energy, and related infrastructure projects.
While EverGen is currently focused on British Columbia, its continued growth is expected across other regions of North America. RNG is produced differently than conventional natural gas, without drilling wells. RNG is derived from biogas, which is captured from decomposing organic waste in landfills, food waste, agricultural waste matter and wastewater from treatment facilities. This waste feedstock is supplied to an anaerobic digester which contains bacteria that breaks down organic matter in the absence of oxygen. The resulting biogas is captured and cleaned to create carbon neutral or carbon negative RNG to be used by the existing North American gas pipeline grid. By capturing these emissions and transforming them into RNG, then combusting into CO2, the overall greenhouse gases (GHG) impact is materially less potent than allowing natural decomposition to release methane into the atmosphere. Liquid and solid digestate matter is a byproduct of the RNG production process and is used as fertilizer and in other applications.
EverGen operates three projects in British Columbia. The company was incorporated in 2020 and went public in 2021, with its common shares listed on the TSX Venture Exchange under ticker symbol ‘EVGN’. In February 2022, EverGen’s common shares began trading on the OTCQB Venture Market in the U.S. under ticker symbol ‘EVGIF’. The company is headquartered in Vancouver.
Portfolio Projects
Fraser Valley Biogas is one of three projects in EverGen’s portfolio. Located in Abbotsford, British Columbia, the facility has been digesting manure and off-farm organics since 2011 and was the first agricultural digester in Canada to produce RNG. The RNG generated through this project is part of a FortisBC program to supply renewable gas to homes, businesses and other customers. Fraser Valley Biogas also provides Abbotsford farms with renewable fertilizer via the digestate produced. EverGen acquired Fraser Valley Biogas early in 2021 and is currently enhancing and expanding the facility. These optimization projects resulted in record production during the month of September 2021, supporting the growing demand for RNG in British Columbia. Optimization activities contributed an additional 18% of RNG production for September and a 9% higher year-to-date production compared to the previous year. The facility produces approximately 80,000 gigajoules of RNG, enough to heat more than 1,000 homes for a year.
Net Zero Waste Abbotsford, a wholly owned EverGen subsidiary and portfolio project, is an existing composting and organic processing facility and RNG expansion project. The British Columbia Utilities Commission recently approved a 20-year offtake agreement between the facility and FortisBC, an electricity and gas utility. Under this agreement, FortisBC will purchase up to 173,000 gigajoules of RNG annually for injection into its natural gas system upon completion of an anaerobic digester project at Net Zero Waste Abbotsford. Once construction is complete, this project is expected to produce enough energy to meet the needs of more than 1,900 homes.
Sea to Sky Soils, a wholly owned EverGen subsidiary and portfolio project, is an existing composting and organic processing facility and potential future RNG expansion project which has been operating near Pemberton, British Columbia, on Lil’wat Nation land since 2012. The Lil’wat Nation is a key partner and supporter of the facility, which has employed a majority of its staff from the First Nation since inception. The Sea to Sky Soils facility processed approximately 160 percent of its forecast tonnage in the second half of 2021. In total, Sea to Sky Soils processed approximately 36,000 tons of organic waste in 2021. The facility is working with the Ministry of Environment to expand its operational capacity in 2022. EverGen has partnered with local municipalities – including Metro Vancouver and the municipality of Pemberton – for the delivery of additional organic waste to the facility. The facility is an important part of EverGen’s RNG infrastructure platform and serves as a source of valuable feedstock to support the company’s existing and future operations.
Market Outlook
A report from Global Market Insights states that the biogas market is projected to see significant growth over the next few years, driven by a shifting preference to utilize biogas to reduce emission levels from traditional fuels. Escalating RNG usage by gas utilities as a sustainable and low carbon alternative to supply heat and electricity in industries and buildings will further stimulate growth. RNG is increasingly deployed across the transport sector, especially for heavy vehicles and vessels, to abate GHG emissions.
Many North American gas utilities have set RNG targets of 5% to 15% of production by volume in 2030, compared to less than 1% by volume in 2020. FortisBC has a goal of including 15% RNG in its gas supply by 2030. EverGen believes this presents a potential C$16 billion+ opportunity for RNG producers.
Management Team
Chase Edgelow is co-founder and CEO at EverGen. He has over 15 years of specialized private investment, finance, and technical expertise in the energy and infrastructure sectors. His background is as a Facilities Engineer with Petro-Canada, independently managing energy infrastructure capital projects located in western Canada. He holds a Professional Engineer designation from the province of Alberta.
Mischa Zajtmann is co-founder and President at Evergen. He has 15 years of experience providing consulting and management for Canadian and American companies in the natural resources and energy space. He is a corporate securities lawyer who began his career at Blake, Cassels & Graydon LLP. His J.D. is from the University of Saskatchewan Law School. He’s a member of the British Columbia Bar.
Sean Mezei is COO at EverGen. He has 20 years of experience in the RNG industry, having served previously as the president of Greenlane Biogas and as a senior manager at QuestAir, and founder and president of Dekany Consulting. He was a co-chairman of the American Biogas Council’s RNG working group for six years. He has been a Registered Professional Engineer in the province of British Columbia since 1994.
Natasha Monk is CFO at EverGen. She is a CPA with 12 years accounting, financial reporting, and tax experience in public practice and industry. She is currently a partner at Affirm LLP, where she advises and consults to a wide variety of companies in multiple industries across public and private sectors. Prior to joining EverGen, she worked at KPMG. She graduated from the University of Calgary.
EverGen Infrastructure Corp. (OTCQX: EVGIF), closed Tuesday's trading session at $2.035, up 12.4309%, on 4,700 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.365/$4.00.
Recent News
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) Signs $31M Definitive Agreement with Roynat Capital, Export Development Canada
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Capitalizing on the Abundance of Low Carbon Energy Sources
- EverGen Infrastructure Announces Definitive Agreement for $31 Million Senior Term Loan with Roynat Capital & Export Development Canada
Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF)
The QualityStocks Daily Newsletter would like to spotlight Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF).
•Canada Nickel in December 2022 announced positive drilling resultsfrom exploration at its Reid and Sothman properties
•The company in November 2022 announced recovery of valuableby-products from a processing pilot test, including cobalt,palladium, platinum and chromium
•Canada Nickel announced in November 2022 improvements to itsaccelerated CO2 capture process
•Canada Nickel announced in October 2022 it closed a secured loanwith Auramet International of $10 million
•Canada Nickel has engaged Deutsche Bank and Scotiabank as financialadvisors for the Crawford Nickel Sulphide Project
•Projections indicate that, by 2035, world demand for nickel willdouble from current levels to 6 million tons annually
Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) is advancing the next generation of nickel-cobalt sulfide projects to deliver the metals needed to power the electric vehicle (EV) revolution and feed the high growth stainless steel market. The company is one of only a few new sources of potential supply outside Indonesia and China.
Canada Nickel possesses industry leading nickel expertise and is focused on low risk, well established mining jurisdictions. The company has launched wholly owned subsidiary NetZero Metals Inc. to develop zero-carbon production of nickel, cobalt and iron and has applied in multiple jurisdictions to trademark the terms NetZero Nickel, NetZero Cobalt and NetZero Iron. Canada Nickel is also pursuing development of processes to allow net zero carbon production of these elements.
Canada Nickel is currently anchored by its 100% owned flagship Crawford Nickel-Cobalt Sulfide Project with large-scale potential located in the heart of Ontario’s prolific Timmins-Cochrane mining camp, adjacent to major infrastructure.
The company believes the EV industry and many other consumer sectors have an urgent need for zero-carbon metal this decade, not in 20-25 years as contemplated by some resource companies. Canada Nickel also believes that nickel supplies from Indonesia and other Pacific island nations, typically controlled by Chinese-owned companies, are not the answer for batteries needed by GM, Ford and the European automakers working to develop and manufacture EV models.
The company is headquartered in Toronto.
Crawford Nickel-Cobalt Sulfide Project
The Crawford Nickel-Cobalt Sulfide Project is the largest sulfide discovery since the early 1970s and contains the fifth-largest nickel sulfide resource in the world, based on Measured & Indicated resources, according to the latest update. The Crawford project is expected to be one of the largest base metal mines in Canada based on results of a Preliminary Economic Assessment. Early projections by Canada Nickel estimate that the project has the potential to produce 50,000 tons of nickel per year. The company is now in the final stages of completing the project’s feasibility study.
The project is projected to produce 2.8 tons of CO2 per ton of nickel equivalent production, which is 89% lower than the industry average of 34 tons of CO2 per ton of nickel equivalent production.
The company is taking significant steps toward developing the Crawford project as a net zero carbon producer. In addition to harnessing the natural ability of the project’s geology to act as a carbon sink through spontaneous reaction of the host rock once exposed to atmospheric conditions called mineral carbonation, Canada Nickel has discovered a new way to enhance carbon capture, termed In Process Tailings (IPT) Carbonation. This act of conditioning the tailings with a concentrated stream of carbon dioxide before deposition has been demonstrated at lab scale to achieve carbon capture at a rate 8-12 times faster than naturally occurring sequestration, achieving more than 60% of the capture that had previously taken six days.
These latest results move the company further toward production of Net Zero Nickel™ and generation of 21 tonnes of CO2 credits per tonne of nickel, which would produce an estimated average of 710,000 tonnes of CO2 credits annually and 18 million total tonnes of CO2 credits over the expected life of mine. IPT Carbonation does not require complex new technologies and major process modifications and could encourage the development of a net zero carbon industrial cluster centered around the Crawford project.
Canada Nickel in January 2023 announced that its latest test work results support the incorporation of carbon capture and storage into the Crawford project. The company believes that utilization of existing process streams should allow IPT to be efficiently engineered and incorporated into the project’s flowsheet, with an integrated feasibility study for the project expected in the second quarter of 2023.
In December 2022, Canada Nickel announced its engagement on Deutsche Bank Securities Inc. (“Deutsche Bank”) and Scotiabank – two of the world’s leading investment banks with a broad base of mining and industrial expertise – as financial advisors for the equity component of the project financing for the Crawford project. In the same release, the company announced the completion of another significant permitting milestone by filing the detailed project description with the Impact Assessment Agency of Canada. Canada Nickel targets receipt of permits by mid-2025, with construction to immediately follow.
Additional Projects
The Reid Nickel Property is located just 16 kilometers southwest of Crawford, or 37 kilometers northwest of Timmins, and contains an ultramafic body with a target geophysical footprint of 3.9 square kilometers. Preliminary assay results from Canada Nickel’s summer/fall drilling program confirm the presence of mineralized dunite, as well as currently undefined higher-grade sections. Partial assay results confirm expected nickel grades. Nickel mineralization in serpentinized dunite was found in all 16 holes drilled to date.
The Sothman Nickel Property is located 70 kilometers south of Timmins. Five drill holes on the eastern half of the target anomaly confirmed the continuation of ultramafic lithologies, primarily peridotite, with moderate to strong serpentinization and variable amounts of mineralization throughout.
The company in December 2022 announced positive drilling results from its ongoing regional exploration campaign at its Reid and Sothman properties. These latest results continue to reinforce the success of Canada Nickel’s geophysical targeting approach and increase the probability of success at the company’s other 20-plus properties within its 42 square kilometers of geophysical targets.
Building on this momentum, Canada Nickel in December 2022 announced its entry into a deal to acquire a 100% interest in the past producing Texmont property situated between the company’s properties south of Timmins. As noted in the news release, the acquisition of the Texmont property provides near-term smaller scale production potential and is highly complementary to the company’s large-scale Crawford and regional nickel sulphide projects.
Market Opportunity
Global demand leaves the market fundamentally short of nickel in the medium- and long-term. Global primary nickel demand will likely reach 3 million tons in 2022, up from 2.4 million tons in 2020, according to the International Nickel Study Group (INSG).
The INSG says primary nickel production is forecast to hit 3.1 million tons in 2022. Indonesia, the world’s largest nickel miner, halted exports of unprocessed nickel ore in January 2020, due to a government-imposed ban. Indonesia has floated the concept of a nickel cartel whose member nations would exert influence over world nickel supply and prices, similar to OPEC’s pricing power over oil.
Benchmark Minerals, a leading EV supply chain research firm, projects that, by 2035, world demand for nickel will double from current levels to 6 million tons annually. That growing demand represents a need for new nickel production equivalent to 70 mines the size of Canada Nickel’s Crawford Project.
Management Team
Mark Selby is Chairman, CEO and Director of Canada Nickel. He was formerly President and CEO of RNC Minerals, where he led a team that successfully raised over $100 million and advanced the Dumont nickel-cobalt project from initial resource to a fully permitted, construction-ready project. He has held senior management roles with Quadra Mining, Inco and Purolator Courier, and was a partner at Mercer Management Consulting. Since 2001, he has been recognized as one of the leading authorities on the nickel market. He graduated from Queen’s University with a Bachelor of Commerce.
Wendy Kaufman is CFO of Canada Nickel. She has 25 years of experience leading publicly listed mining companies in project financing, capital structuring, capital markets, accounting and internal controls, tax, and financial reporting and public disclosure. She was also previously CFO at Khiron Life Sciences Corp. and held CFO and senior finance positions at Pasinex Resources Limited, Primero Mining Corporation and Inmet Mining Corporation. She holds a Bachelor of Business Administration from Wilfrid Laurier University and is a Chartered Professional Accountant.
Steve Balch is VP Exploration at Canada Nickel. He is an Ontario registered geoscientist with 32 years of experience in geophysics, specializing in magnetic and electromagnetic methods. He founded Triumph Instruments and developed the AirTEM system, a multi-coil helicopter-borne EM system that is in use worldwide. He has also been active in borehole geophysics and helped develop new technologies including north-seeking gyros, temperature compensated induction conductivity probes, UAV-based magnetometers and high sensitivity magnetic gradiometers.
Christian Brousseau is VP Capital Projects at Canada Nickel. He is a professional engineer (P.Eng) with over 30 years of experience in engineering, design and construction in the Canadian mining industry, including six years as Project Director for the Dumont Project and three years as the Engineering and Construction Manager for Detour Gold. Prior to Detour, he held various construction management positions at Osisko’s Malartic Project and at Goldcorp’s Éléonore Project. He also spent eight years at Falconbridge supervising and managing various capital projects.
Canada Nickel Company Inc. (OTCQX: CNIKF), closed Monday's trading session at $2.05, up 3.0151%, on 73,251 volume with 375 trades. The average volume for the last 3 months is 57,207 and the stock's 52-week low/high is $1.04999995/$5.63000011.
Recent News
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Is 'One to Watch'
- InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Announces Integration of IPT Carbonation Process into Crawford Project
- InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Announced Acquisition Deal, Releases Update on Texmont Drill Program
Aditxt Inc. (NASDAQ: ADTX)
The QualityStocks Daily Newsletter would like to spotlight Aditxt Inc. (NASDAQ: ADTX).
•Aditxt recently announced the formation of a U.S.-based whollyowned subsidiary, Adimune, Inc., and appointed Joachim-FriedrichKapp, M.D., Ph.D., a 30-year veteran of the pharmaceuticalindustry, as its Chairman and CEO
•Dr. Kapp will be tasked with leading and developing the ApoptoticDNA Immunotherapy(TM) (“ADI(TM)”) technology toward clinical trials
•Aditxt also plans to submit a Clinical Trial Application (“CTA”)for immunotherapeutic technology drug candidate ADI(TM)-100,seeking approval for Adimune(TM) to conduct the first-in-humanstudy in psoriasis patients, beginning in the second half of 2023
•ADI(TM) is a nucleic acid-based technology platform that utilizes anovel antigen-specific approach to induce immune tolerance bymimicking the way the body naturally induces tolerance to its owntissues
Aditxt Inc. (NASDAQ: ADTX) is a biotech innovation company developing technologies focused on mapping and reprogramming the immune system. Aditxt’s immune mapping technologies are designed to provide a personalized immune profile. Aditxt’s immune reprogramming technologies, currently preclinical, are being developed to retrain the immune system to induce tolerance to address rejection of transplanted organs, autoimmune diseases, and allergies.
As further discussed below, the company’s first commercial product is an immune mapping technology, AditxtScore™, which is designed to provide a personalized profile of the immune system.
The company’s preclinical immune reprogramming technology, Apoptotic DNA Immunotherapy™ (“ADi™”), aims to retrain the immune system to induce tolerance, with the goal of addressing vast unmet needs in transplanted organ rejection, autoimmune diseases, and allergies. The company is developing specific ADi™ products for psoriasis, type 1 diabetes, and skin grafting.
Headquartered in Richmond, Virginia, Aditxt also operates locations in Silicon Valley and New York.
AditxtScore™
AditxtScore™ is a proprietary platform designed to provide a personalized, comprehensive profile of an individual’s immune system. The underlying technology, licensed from Stanford University through an exclusive worldwide agreement, offers a highly sensitive and accurate method of detecting and quantifying cellular responses, allowing greater specificity, quantification, and amplification of both clinical and commercial opportunities.
The company’s first commercial application of the platform, AditxtScore™ for COVID-19, delivers timely reports on vulnerability and immune status relating to SARS-CoV-2 and its known variants, giving consumers and physicians the data needed to make informed health decisions. Potential future applications will offer early detection of an array of conditions, including diabetes, cardio-metabolic maladies and hormonal imbalances.
Aditxt’s AditxtScore™ immune monitoring center in Richmond, Virginia, is operational and designed to support the anticipated increased demand for AditxtScore™ as well as related products and services. The company is currently scaling its capabilities at this location, with a goal of processing up to 10 million immune system tests/reports annually.
ADi™
ADi™ is Aditxt’s immune reprogramming platform addressing disease-causing immune responses while maintaining the immune system’s ability to combat pathogenic infection. The company is commercializing a nucleic acid-based technology called Apoptotic DNA Immunotherapy™ (ADi™) which utilizes a novel approach that mimics the way our bodies naturally induce tolerance to our own tissues (therapeutically induced immune tolerance). Aditxt believes its ADi™ technology platform can be engineered to address a wide variety of indications.
Aditxt is currently developing ADi™ products for psoriasis, type 1 diabetes and skin grafting.
Currently, immuno-tolerance is achievable through chimerism and cell-based therapy, but there is a clinical need for a more practical and cost-effective approach which:
- Can be made into a product
- Does not require additional hospitalization
- Is simple to produce and ship
Preclinical studies have demonstrated that ADi™ treatment significantly and substantially prolongs graft survival, in addition to successfully “reversing” other established immune-mediated inflammatory processes. ADi™ treatment is not expected to require hospitalization, instead being delivered as an injection in minute amounts into the skin.
IP Portfolio
Both AditxtScore™ and ADi™ are supported by a strong IP portfolio.
AditxtScore™, built upon initial technology invented, licensed from and used at Stanford University, is protected by U.S. patents encompassing methods, systems, and kits for detection and measurement of specific immune responses.
ADi™ technology is protected by seven patent families, including:
- 8 U.S. patents
- 4 pending U.S. patent applications
- 86 foreign patents and 14 pending foreign patent applications spanning the EU, Australia, Canada, Japan, China, India and Hong Kong
These patents are broadly categorized into three groups:
- Autoimmune diseases and Type 1 Diabetes
- Organ transplantation and a method of producing plasmid DNA to prevent immune activation
- Composition of matter for a tolerance delivery system for antigens of interest
Aditxt also possesses and/or in-licenses substantial know-how and trade secrets relating to the development and commercialization of its product candidates, including related manufacturing processes and technologies.
Market Overview
The potential market opportunities presented by immune monitoring and reprogramming are extensive, particularly as Aditxt continues to evaluate additional applications for the platforms.
The company’s initial focus on organ transplantation and related autoimmune response provides some insight into the potential of its approach. According to BCC Research, the global organ and tissue transplantation and alternatives market is on course to reach $120.3 billion by 2024, recording a CAGR of 7.4% from 2019. Industry data suggest that approximately 50% of all transplanted organs are rejected within 10-12 years, further highlighting the critical need for a practical, cost-effective solution to harmful autoimmune responses.
Through its focus on the COVID-19 testing market with AditxtScore™, Aditxt demonstrated the wide-ranging potential of its portfolio. Fortune Business Insights estimated the global COVID-19 diagnostics market at $48.64 billion for 2022. While demand for COVID-19 diagnostics is expected to lessen in the coming years, Aditxt will be uniquely positioned to leverage its existing infrastructure stemming from these operations as the company works to advance broader applications for the AditxtScore™ platform.
Leadership Team
Amro Albanna is the Co-Founder, Chairman, and CEO of Aditxt. He has founded multiple startups to commercialize innovations in various industries, including healthcare, enterprise software, telecommunications, nano technology, consumer health, and biotech. Mr. Albanna has led numerous M&A and going-public transactions as a founder, co-founder, and senior executive.
Shahrokh Shabahang, D.D.S., MS, Ph.D., is the company’s Co-Founder, Chief Innovation Officer, and a member of its board. He brings to the team more than 20 years of experience in developing and commercializing life science technologies focused on product and clinical development in the fields of microbiology and immunology.
Corinne Pankovcin, CPA, MBA, is the President of Aditxt. Prior to joining Aditxt, Ms. Pankovcin served as CFO for several world class organizations, including Business Development Corporation of America, Blackrock Kelso Capital and AIG Capital Partners. In these roles, Ms. Pankovcin was responsible for executing portfolio investments and managing significant M&A transactions.
Thomas Farley is the Chief Financial Officer of Aditxt. From December 2015 to June 2020, Mr. Farley was the Controller and Treasurer of Business Development Corporation of America (“BDCA”), a publicly listed business development company. Prior thereto, from January 2011 to August 2015, Mr. Farley was the Senior Controller of Blackrock Capital Investment Corporation (NASDAQ: BKCC). Prior to joining BlackRock Capital Investment Corporation, Mr. Farley was a Senior Controller for PineBridge Investments Emerging Markets practice. Mr. Farley was also an Accounting Manager for Bessemer Venture Partners prior to his tenue at PineBridge. Mr. Farley began his career with PricewaterhouseCoopers LLP, from 1996 to 2001. Mr. Farley earned his B.S. in Accounting from Long Island University and is a Certified Public Accountant.
Rowena Albanna is the company’s Chief Operating Officer. Ms. Albanna has over two decades of experience in senior leadership roles for both technology startups and public companies. Ms. Albanna’s experience spans a wide variety of industries, including biotechnology, insect control, nanotechnology, consumer electronics, financials, telecommunications, e-commerce, online marketing, medical, and defense.
Matthew Shatzkes is the Chief Legal Officer and General Counsel of Aditxt. As a former partner at an AM Law 50 law firm, Mr. Shatzkes advised a wide variety of healthcare related entities, including biotech companies, on corporate, regulatory, and strategic business matters. Mr. Shatzkes will oversee all aspects of the legal functions at Aditxt, including, providing advice and counsel on governance, regulatory matters, strategic alliances, mergers and acquisitions, and commercial transactions.
Aditxt Inc. (NASDAQ: ADTX), closed Tuesday's trading session at $1.37, up 6.2016%, on 186,736 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.00/$28.49.
Recent News
- Aditxt Inc. (NASDAQ: ADTX) - Aditxt, Inc. (NASDAQ: ADTX) Bolsters Goal to Conduct First-In-Human Clinical Trial Evaluating ADI(TM) Tech with Formation of Adimune, Inc. Subsidiary; Appoints Industry Veteran as Subsidiary Chairman and CEO
- InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Forms Subsidiary with Focus on Immune Modulation Therapies
- Aditxt Inc. (NASDAQ: ADTX) Announces Goal of First-in-Human Trials for Drug Candidate ADI(TM)-100 with a Focus on Psoriasis
Data443 Risk Mitigation Inc. (OTC: ATDS)
The QualityStocks Daily Newsletter would like to spotlight Data443 Risk Mitigation Inc. (OTC: ATDS).
To view the full press release, visit https://ibn.fm/3U7Tf
Data443 Risk Mitigation Inc. (OTC: ATDS) is a data security and privacy software company for ALL THINGS DATA SECURITY™. The company is committed to organizing the world’s information by identifying and protecting all sensitive data regardless of location, platform or format.
Data443 provides software and services to enable secure data across devices and databases – at rest and in transit – locally, on a network, or in the cloud. With over 10,000 customers in more than 100 countries, Data443 provides a modern approach to data governance and security. The company’s framework helps customers prioritize risk, identify security gaps, and implement effective data protection and privacy management strategies.
Data443 derives revenue primarily from contracts for subscriptions to access its SaaS platforms, and ancillary services provided in connection with its subscription services. In today’s ever-changing environment with unique and complex requirements for data privacy, governance and hybrid workforces, every organization needs to know where all their data is, who has access to it and how sensitive it is. Data443 provides the tools needed to give companies control over their data processing activities, with capabilities for identifying, reporting and migrating or deleting sensitive data.
The company is headquartered in Research Triangle Park, North Carolina.
Products
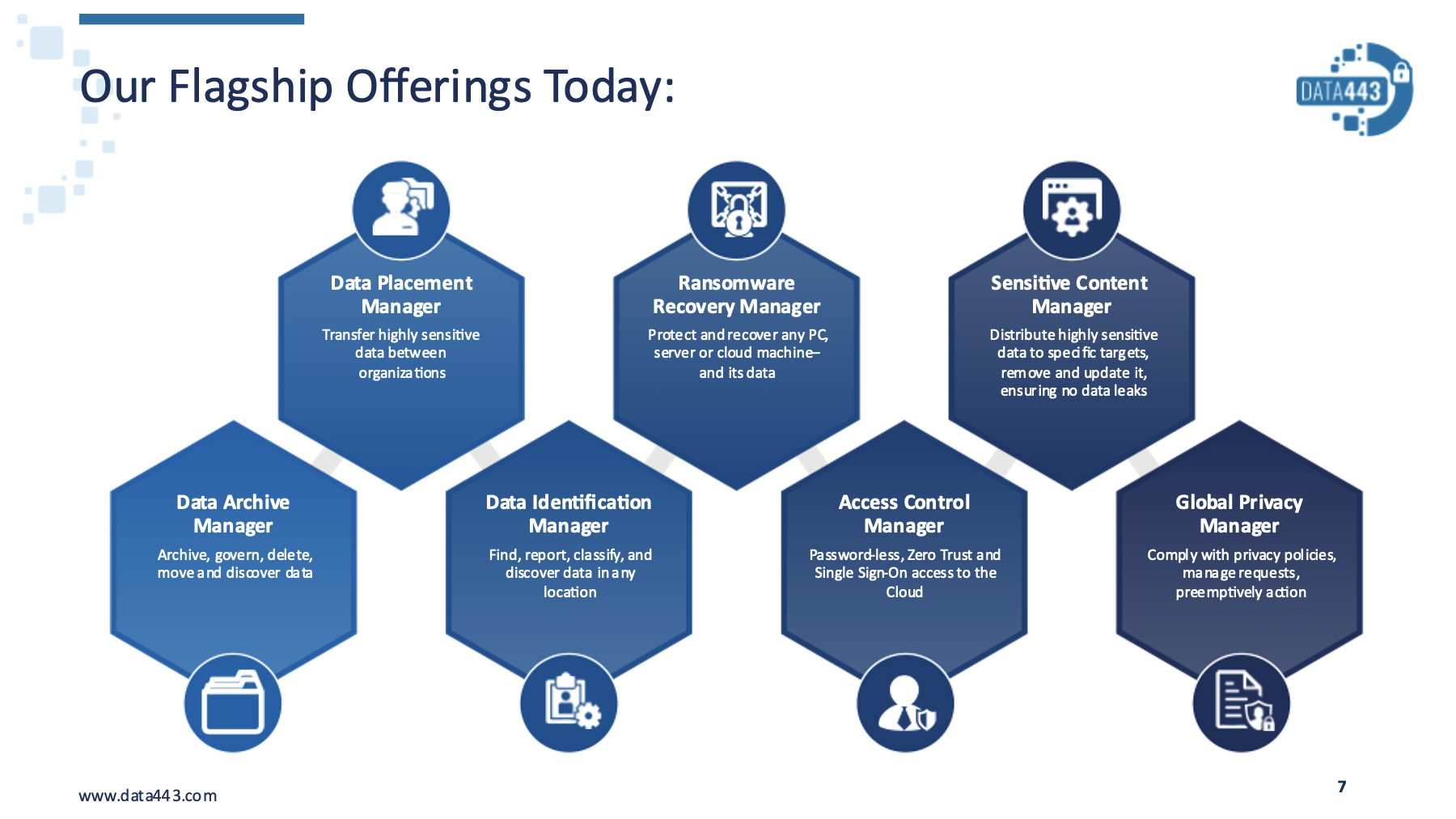
Focused on data security with a privacy-forward methodology, the Data443 product suite delivers solutions designed to securely manage data and data privacy needs on-premises, in the cloud and in hybrid environments. Offerings include:
- Data Identification Manager reduces risk by shining a light on dark data across cloud, on-premises and hybrid environments. From a centralized dashboard, Data Identification Manager provides the ability to automatically inventory all data repositories, classify and tag all data, and enable global search and discovery – all through an agentless deployment.
- Data Placement Manager quickly and securely transfers sensitive data over any public or private network. Available as an HP Nonstop server-based application and for Windows, Linux or any public cloud provider, Data Placement Manager enables the scheduling, routing, formatting and transfer of business-critical data.
- Data Archive Manager is an “all information, anywhere” archiving solution designed to handle and manage all types of privacy requests across cloud, on-premises and hybrid environments. With over 15 years operational history and hundreds of clients managing millions of mailboxes, the platform is purpose-built for information archiving, retention and privacy request management.
- Data Hound™ is a data discovery, classification and capture toolset that enables organizations to perform quick scans, detailed reporting and subsequent data actions based on policy.
- Ransomware Recovery Manager is the only industry solution that actively recovers the device, operating system and data with a simple reboot. Using patented, proven technology, the product produces 100% effectiveness for the whole device and datasets.
- Access Control Manager provides user ID and passwordless access to quickly enable trust across an organization’s entire ecosystem. Its unique architecture allows it to leverage multiple distributed authoritative sources to understand and resolve a typical access request – with the ability to enable or deny the action on the fly.
- Global Privacy Manager provides organizations one comprehensive view, for all privacy requirements, across all enterprise data, all at once. This unmatched visibility into an organization’s data assets ensures that all private and sensitive data can be identified and protected and that enterprises can obey all relevant privacy laws in any jurisdiction.
- Sensitive Content Manager is a security-centric collaboration service designed to give organizations the tools needed for successful content sharing, collaboration and safe distribution with full enterprise management in mind. With a continuous sync feature, encrypted data is automatically downloaded and updated in real time – regardless of location – ensuring that users have the most accurate data available.
Market Outlook
A report from Allied Market Research estimates that the global data security market was worth about $19 billion in 2021 and is projected to reach a value of $54.23 billion by 2027. That represents a CAGR of more than 18% for the forecast period, making data security one of the hottest areas within IT.
Separately, Fortune Business Insights estimates the global data privacy software market is valued at $2.36 billion in 2022 and projects it will grow to $25.85 billion by 2029. That represents a CAGR of 40.8% over the forecast period.
Management Team
Jason Remillard is President, CEO and Founder of Data443. He is responsible for overseeing global expansion, management, execution and corporate development. With over 25 years in global enterprise and B2C software sales and marketing, he brings deep leadership and technical experience, having spent previous time at Fortune 500 companies such as Deutsche Bank, TD Bank, IBM & Merrill Lynch.
Greg McCraw is CFO at Data443. He has over 25 years of experience helping businesses strengthen their accounting and finance operations. He previously served as Vice President of Finance for a dental services organization active in acquisitions, and, prior to that, he was managing director of a boutique accounting and finance consulting firm advising Fortune 500 clients in pharmaceutical, financial services, and private equity sectors on how to execute on regulatory and compliance solutions.
Bennett Pursell is Data443’s Chief Technology Officer. He has over 20 years of experience in IT architecture, security governance and systems integration. Prior to his role at Data443, he served as Head of Technology Architecture at Moody’s Investor Services and was Vice President and Technical Architect of Cloud Computing at Deutsche Bank, along with a host of technical and project management roles dating back to 2006, after starting his career as a web developer with a few startups and running research labs.
Kirill Kashigin is Chief Software Architect at Data443. He leads the development and quality teams, and serves as technical adviser and subject matter expert, bringing vast technical knowledge on privacy management and data security. Formerly the CTO of FileFacets, he has nearly 20 years in development of high-performance systems and deployment.
Data443 Risk Mitigation Inc. (OTC: ATDS), closed Tuesday's trading session at $0.37, up 34.521%, on 44,273 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.27005/$6.99.
Recent News
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Secures VB100 Certification for Antivirus Product
- Data443 Risk Mitigation Inc. (ATDS) Seeks to Capitalize on Expanding Data Protection Market
- Data443 Risk Mitigation Inc. (ATDS) Lands $350,000 Add-On Contract, Closing 2022 on a High Note
India Globalization Capital Inc. (NYSE American: IGC)
The QualityStocks Daily Newsletter would like to spotlight India Globalization Capital Inc. (NYSE American: IGC).
The International Association for the Study of Pain estimates that
India Globalization Capital Inc. (NYSE American: IGC), through subsidiary IGC Pharma, develops, patents, and markets advanced THC-based drug formulations for the treatment of symptoms related to various diseases including but not limited to Alzheimer’s disease, Tourette syndrome, chronic pain, and pet seizures.
IGC’s leading drug candidate, IGC-AD1, has completed Phase 1 of a safety and tolerability trial and entered Phase 2 trials for treating agitation in patients with Alzheimer’s dementia, the first study in humans of a natural tetrahydrocannabinol (THC) compound plus another molecule (www.clinicaltrials.gov). As of September 2022, the IGC trial is the only ongoing Phase 2 trial of a natural THC-based formulation on Alzheimer’s patients.
The company’s other drug candidate, TGR-63, is an enzyme inhibitor that has shown in preclinical trials the potential to reduce neurotoxicity in Alzheimer’s cell lines. Both drug candidates have shown their ability to ameliorate beta amyloid plaques in Alzheimer’s cell lines and improve memory in Alzheimer’s mouse models. Beta amyloid plaques are a key hallmark of Alzheimer’s and an important target of Alzheimer’s pharmaceutical drug development.
Neuro Psychiatric Symptoms (NPS) are not only debilitating for Alzheimer’s patients; they also place an immense emotional burden on their caregivers. Beyond reducing symptoms, IGC-AD1’s active molecules and TGR-63 have also shown promise in preclinical trials to reduce important hallmarks of Alzheimer’s including plaques and tangles, as well as improving the treatment of memory loss.
Over the past eight years, the IGC team has amassed a deep knowledge of cannabinoid science, including extraction, isolation, purification, and development. The company’s strategy is to leverage its unique end-to-end capabilities, platform, and expertise to develop a class-leading program and bring it to market quickly and cost efficiently to treat neurodegenerative diseases such as Alzheimer’s.
The company also has a family of cannabidiol (CBD)-based consumer products (www.Holief.com) such as pain relief creams, pain relief gels, purpose gummies, tinctures, and capsules targeting women’s wellness, with a particular focus on premenstrual syndrome (PMS) and dysmenorrhea (period cramps). In addition, the company targets individuals that need sleep-aids with its specially formulated low melatonin cannabinoid gummies.
IGC has also introduced a low-calorie CBD- and caffeine-infused energy beverage brand (www.SundaySeltzer.com) that is currently available for purchase. The company’s brands are founded on the belief that effective natural solutions should be affordable and accessible to everyone. As the demand for natural products targeting women’s wellness and energy drinks continue to grow, these products are seeing strong traction in the market.
The company operates three facilities – a large GMP (Good Manufacturing Production Standards) certified facility that includes extraction, distillation, and manufacturing, in Washington State; a GMP-211 (pharmaceutical) grade facility in Maryland; and a facility licensed for controlled substances including cannabis in Bogota, Colombia, with complete access to legal licensed cannabis where the company conducts its testing.
In addition, the company’s development under Magistral Formulations is approved by INVIMA (Colombia National Food and Drug Surveillance Institute) to treat neurological disorders, non-oncological chronic pain, and mental disorders.
IGC’s intellectual property (IP) portfolio comprises of eight patents that it controls and seven patent applications. The portfolio includes #11,446,276, a patent for extreme low dose THC treatment of Alzheimer’s that was granted in September 2022.
The company is headquartered in Potomac, Maryland.
IGC-AD1
IGC-AD1 is the company’s leading drug candidate for the treatment and relief of Alzheimer’s symptoms. A significant amount of research on Alzheimer’s cell lines has shown that the active agents in IGC-AD1 reduce plaques and neurofibrillary tangles that are the hallmarks of Alzheimer’s. Further, micro-dosing of THC, as shown in cell lines, could increase the functioning of mitochondria and potentially promote the growth of new neural pathways (neurogenesis). The research shows that micro-dosing of THC affects the brain radically differently from the normal higher dosing of THC.
While there is a significant body of research showing that THC is neuro-toxic at normal levels of dosing, micro-dosing of THC has been shown to be non-toxic to neurons. With the results of these preclinical studies, the company developed an oral formulation, IGC-AD1. The company recently completed a safety and tolerability Phase 1 trial on Alzheimer’s patients and has initiated a Phase 2, multi-site, double-blind, randomized, placebo-controlled trial of the safety and efficacy of IGC-AD1 on agitation in participants with dementia due to Alzheimer’s disease at sites in the U.S. and Canada. IGC expects the Phase 2 trial to take between 9 and 12 months to complete, barring unknown factors such as, for example, a resurgence of COVID and the enforcement of lockdowns and travel restrictions.
With further successful trials and FDA approvals, IGC hopes to bring a drug based on natural THC as an effective treatment for agitation in Alzheimer’s to market.
TGR-63
The company’s other molecule, TGR-63, has been shown to reduce the neurotoxicity that impacts memory loss in preclinical trials with mice. On a dose dependent manner, transgenic Alzheimer’s mice treated with TGR-63 showed improvement in memory relative to control.
Both drug candidates, IGC-AD1 and TGR-63, have shown their ability to reduce the brain plaques associated with memory loss in Alzheimer’s in mice.
With further successful trials and FDA approvals, IGC hopes to bring TGR-63 as a treatment for Alzheimer’s disease to market.
Market Opportunity
Alzheimer’s disease impacts over 55 million people worldwide and about 5.5 million individuals in the U.S. Over 70% of these patients face debilitating symptoms, including anxiety, depression, and agitation (Mendez, 2021). Agitation in dementia patients can include excessive physical movement and verbal activity, restlessness, pacing, belligerence, aggression, screaming, crying, and wandering.
In 2020, the estimated healthcare costs for Alzheimer’s disease in the U.S. were $305 billion. Medicare and Medicaid covered about 70% of those costs, leaving considerable burden on patients and families. At the current rate of growth of Alzheimer’s and other dementia diagnoses, those costs are estimated to reach over $1 trillion by 2050.
Currently, there are no FDA-approved medications to alleviate the symptoms of dementia due to Alzheimer’s disease, providing a tremendous opportunity for formulations that can have an impact on quality of life and disease progression.
Management Team
Richard Prins has been chairman at IGC since 2012 and served as an independent director since 2007. From March 1996 to 2008, he was the Director of Investment Banking at Ferris, Baker Watts, Incorporated. Prins served in a consulting role to RBC until January 2009. He currently volunteers full time with a non-profit organization, Advancing Native Missions, and is a private investor. Since February 2003, he has been on the board of Amphastar Pharmaceuticals Inc. He holds a bachelor’s degree from Colgate University and an MBA from Oral Roberts University.
Ram Mukunda is CEO and President of IGC. He has been the chief inventor and architect of most of the company’s patent filings and is responsible for the company’s strategic positioning. Prior to IGC, he was founder and CEO of Startec Global Communications, which he took public in 1997. He served as Strategic Planning Advisor at Intelsat, a communications satellite services provider. From 2001 to 2003, he was a Council Member at Harvard’s Kennedy School of Government, Belfer Center of Science and International Affairs. He was named the 1998 Ernst & Young Entrepreneur of the Year. He holds bachelor’s degrees in electrical engineering and mathematics, and a master’s degree in engineering from the University of Maryland.
Dr. Jagadeesh Rao is the company’s Principal Scientist. His career spans two decades in the public sector and product R&D for Johnson & Johnson. He leads IGC’s scientists in the development of pharmaceutical and OTC products. He worked for the federal National Institutes of Health, and for the National Institute on Drug Abuse. His Ph.D. in Neurochemistry is from the National Institute of Mental Health & Neurosciences in India. He did postdoctoral training at the University of Illinois-Chicago.
Claudia Grimaldi is a Director, Vice President, Principal Financial Officer, and Chief Compliance Officer for IGC. She also serves as a Director/Manager Director for some of the company’s subsidiaries. She graduated with highest honors from Javeriana University in Colombia with a bachelor’s degree in psychology. She holds an MBA, graduating with highest honors, from Meredith College in North Carolina. In addition, she has attended the Darden School of Business Financial Management Executives program and the Corporate Governance Program at Columbia Business School. She is currently pursuing her Directorship Certification with the National Association of Corporate Directors. She is fluent in both English and Spanish.
India Globalization Capital Inc. (NYSE American: IGC), closed Tuesday's trading session at $0.3473, up 3.5171%, on 191,494 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.2785/$1.16.
Recent News
- India Globalization Capital Inc. (NYSE American: IGC) - Scientific Review Highlights Stigma Directed at People Living with Chronic Pain
- 420 with CNW - New York Launches First Legal Adult-Use Cannabis Dispensary
- Why a Dog's Teeth May Chatter
MetAlert Inc. (OTC: MLRT)
The QualityStocks Daily Newsletter would like to spotlight MetAlert Inc. (OTC: MLRT).
•MetAlert is a pioneer in location-sensitive health monitoringdevices and wearable technology products targeting the Alzheimer’s,Dementia, and Autism (“ADA”) market
•The company is looking to provide cutting-edge telehealthtechnologies and is continuously working to roll out improved andmore innovative products for its users
•Its SmartSole(R) product facilitates discreet tracking and remotemonitoring of ADA patients
•The company has also launched SmartSole plus(R), which addsBluetooth and Wi-Fi capabilities for better tracking
•MetAlert also distributes RoomMate, a wall-mounted 3D infraredsupervision product that enables caregivers to look after patientswithout intruding on their personal space
The policy changes spearheaded by the US government at the heightof the COVID-19 pandemic boosted telehealth, includingtelemedicine. As a result, by April 2020, telehealth servicesconstituted 32% of Medicare claims, up from less than 1% before thepandemic, and by July 2021, the figure had plateaued to between 13%and 17% (https://ibn.fm/QtTZL). Against this backdrop, a recent article (https://ibn.fm/9deW5) in Equities asks: “But will its momentum continue now that thepandemic has lost its urgency?”
MetAlert Inc. (OTC: MLRT) is a pioneer in location sensitive health monitoring devices (estimated $47 billion industry in 2021) and wearable technology products (industry forecast to reach $174 billion by 2030).
With over 20 years of experience and an extensive patent portfolio (30+), MetAlert is a leader for consumers/patients afflicted with Alzheimer’s, dementia, and autism (ADA). This market represents approximately 2.9% of the world’s population (approximately 34 million people in 24 developed countries). Due to specific behaviors (problems with memory, adversity to wearing unknown items, etc.) of consumers/patients in this market segment, traditional products, such as an iPhone or Fitbit, are not a practical solution. This has created a significant market with very few competitors for MetAlert.
MetAlert and its subsidiaries are engaged in designing, developing, manufacturing, distributing, and selling products and services in GPS/BLE wearable technology, personal location, wandering assistive technology, and health data collection and monitoring. The company offers a global end-to-end hardware, software, and connectivity solution, in addition to developing two-way tracking technologies, which seamlessly integrate with consumer products and enterprise applications.
Using its award-winning, patented GPS SmartSole® as a hub for collecting and transmitting data to the cloud in real-time, MetAlert is expanding its value proposition to consumers and increasing its revenue per user (RPU) while creating the largest database of health statistics for ADA consumers/patients. MetAlert generates revenue from product sales, recurring subscriptions, intellectual property (IP) licensing, and professional services. The company has international distributors servicing customers in over 35 countries and is an approved U.S. military government contractor. Its customers include public health authorities and municipalities, emergency and law enforcement, private schools, assisted living facilities, NGOs, small business enterprises, senior care homes and consumers.
The company is headquartered in Los Angeles, California, with a sales office in London, England, and distributors across the globe.
Products
- GPS SmartSoles® HUB (launched Q4 2022) is a GPS/BLE-equipped insole that allows remote monitoring, data collection, and encrypted data transmission to the cloud.
- Telehealth (available Q4 2022) allows access remotely to doctors and other health professionals on an as-needed basis. This service will also function as the prescribing doctor once Medicare reimbursement codes are established.
- Concierge (available Q4 2022) provides 24/7/365 enhanced emergency response that coordinates with all relevant parties to quickly detect false alarms and escalate response as needed.
- Bluetooth Enabled Devices (available Q1 2023) include third-party devices that collect vitals and other health data and connect with the GPS Smartsoles® HUB.
- Artificial Intelligence (available Q1 2023) software will evaluate the Teradata of health information identifying trends and respond to preestablished alert thresholds.
- Take-Along Tracker is a small GPS tracking device – less than three inches long – that works with 4G cellular service and will have the same “HUB” functionality as the GPS Smartsoles®. This versatile and affordable mini tracker boasts super long battery life, with up to 14 days of operation per charge.
- RoomMate™ is a wall-mounted alert system that detects and alerts caregivers about patient behavior that could lead to falls and injuries. The system features 3D infrared and wall-mounted sensors, eliminating the need for any other physical installation or wearables. RoomMate™ offers patient privacy by design. Images are not stored, but all actions are logged. It’s a unique solution for looking after patients without intruding on their personal space.
Market Outlook
According to Grand View Research (Patient Monitoring Devices Market Size & Share Report, 2030), the global patient monitoring devices market size was valued at $47.0 billion in 2021 and is expected to expand at a compound annual growth rate (CAGR) of 7.8% from 2022 to 2030. The expansion of the industry can be attributed to the rise in demand for monitoring devices used to measure, distribute, record, and display a variety of biometric data, including blood pressure, temperature, and blood oxygen saturation level.
The growing number of chronic disorders, such as diabetes, stroke, and kidney disease, are driving the demand for patient monitoring devices. For instance, according to the World Health Organization (WHO), about 422 million people globally have diabetes. Likewise, the number of asthma and chronic obstructive pulmonary disease patients (COPD) is increasing rapidly.
According to the WHO, around 235 million people suffer from asthma. As a result, peak flow meters, which are used to gauge respiration rate, are increasingly used. The market for patient monitoring devices is driven by the simplicity with which it is handled, transported, and remotely accessible. Major market players are engaging in a variety of tactics to expand the industry, including partnerships, cooperation, innovation, launches, and mergers.
During the COVID-19 outbreak, social segregation and quarantining procedures were put into place worldwide. Many people avoided regular hospital visits as a result. Many people now need routine home temperature and oxygen level monitoring to maintain track of their health, thereby demanding monitoring devices at home.
Various government programs are supporting the pandemic outbreak. The FDA has granted Emergency Use Authorizations (EUAs) for a few wearables and patient monitoring devices to improve access to medicines, monitor patients more closely, and lessen the risk of SARS-CoV-2 exposure to medical professionals during the COVID-19 pandemic.
The growing popularity of wearable and remote patient monitoring devices is another factor fueling the market’s expansion. By fusing clinical symptomology with vital indicators, wearable technology helps in the diagnosis of many chronic diseases. Thus, there has been a dramatic rise in the usage of wearable technology to combat COVID-19.
The wearable medical device market is anticipated to reach $174.48 Billion by 2030, expanding at a 27.1% CAGR during the forecast period (2022-2030), according to Market Research Future.
MetAlert identifies the total addressable market for its wearable patient monitoring tech for those with Alzheimer’s, dementia, and autism at more than 34 million potential patients in North America, Europe, South Africa, and Asia.
Management Team
Patrick E. Bertagna is Founder, CEO and Chairman at MetAlert. He began his career in apparel sales in 1983 and was promoted to national sales manager within two years. In 1986, he founded his first company importing apparel from Europe and selling to U.S. retailers from JCPenney to Neiman Marcus. He has founded several technology and apparel companies, including MetAlert in 2002, which he took public in 2008. He attended Cal State University Northridge with a business major and a psychology minor.
Louis Rosenbaum is COO of MetAlert. He co-founded Global Trek Xploration and was an initial investor in MetAlert. He has successfully started companies in multiple industries, including apparel, environmental services, and the music industry, achieving annual revenues in the multi-millions of dollars. He previously was president of Elements, a women’s apparel company, and of Advanced Environmental Services.
Alex McKean is CFO at MetAlert. He is also the CFO of Encore Brands Inc., a position he has held since 2009. He has held positions as Controller and VP of Finance at 24:7 Film and InternetStudios.com, Director of FP&A/SVP at Franchise Mortgage Acceptance Company, Corporate Accounting Manager/Treasurer of Polygram Filmed Entertainment and Assistant Treasurer/Controller for State Street Bank. He holds an International MBA from Thunderbird School of Global Management and undergraduate degrees in business and political science from Trinity University.
MetAlert Inc. (OTC: MLRT), closed Tuesday's trading session at $0.32, up 33.3333%, on 13,439 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.06/$1.00.
Recent News
- MetAlert Inc. (OTC: MLRT) - InvestorNewsBreaks - MetAlert Inc. (MLRT) Featured on 'Gametime with Sequire'
- MetAlert, Inc. (MLRT) Developing Innovative Location-Sensitive Health Monitoring and Supervision Products as It Targets Market Leadership in Telehealth Space
- InvestorNewsBreaks - MetAlert Inc. (MLRT) Featured in Ludlow Research Analyst Brief
Cub Crafters Inc.
The QualityStocks Daily Newsletter would like to spotlight Cub Crafters Inc.
To view the full article, visit https://ibn.fm/4A3wF
Cub Crafters Inc. (typically styled CubCrafters) is an OEM aircraft manufacturer based at McAllister Field Airport in Yakima, Washington. The company was founded in 1980 to build parts and supplementary type certificate (STC) improvement modifications, which were used to establish it as the preeminent center for rebuilding the classic Piper PA-18 Super Cub light aircraft. CubCrafters went on to advance the market with its own, newly manufactured aircraft models and holds an approved Federal Aviation Administration (FAA) Production Certificate. Yakima-based operations include an engineering design-test-certification center, aircraft parts and assembly production facilities, and an MRO maintenance service and overhaul facility.
The first newly manufactured aircraft by the company, the CC18-180 Top Cub, was Federal Aviation Administration (FAA) type certified in December 2004. The Top Cub was also granted type certificates (TC) by Transport Canada in July 2008, followed by Australian certification in August of that same year. With the FAA’s release of the new Light Sport Aircraft (LSA) class, CubCrafters created a brand-new model in 2008, the CC11-100 Sport Cub, similarly based on the original Piper J-3 Cub’s appearance, which it validated to ASTM international standards as an LSA. This model advanced to become known as the Carbon Cub, the bestselling LSA of all time in the U.S.
CubCrafters focuses on four main product lines, including the Carbon Cub SS, Carbon Cub FX, XCub, and the Top Cub under license. Some models are built to be lightweight and powerful for quicker flights, while others are built for longer missions in unforgiving backcountry environments.
CubCrafters has a service and overhaul facility for PA-18 Super Cubs and other Cub derivative designs at its Yakima headquarters. The company sells aircraft kits as well as finished aircraft.
Aircraft
The Carbon Cub is available in three variants: Carbon Cub SS (production Light Sport Aircraft), Carbon Cub FX (an innovative Builder Assist E/A-B aircraft) and Carbon Cub EX (E/A-B aircraft kit). Carbon Cub has been designed for off-airport operation with a powerful engine, strong lightweight airframe and nimble low-speed manners. The Carbon Cub has taken the fundamentally superior design of the Piper Super Cub and reinvented it using 21st century materials and computer-aided design. Superior engineering results include the Carbon Cub having 50% fewer parts and weighing more than 300 pounds less than a similarly equipped Super Cub. Now in its third generation of innovation advancements, there are over 1,000 Carbon Cubs flying.
The CubCrafters CC19-180 XCub, FAA Certified and introduced in June 2016, is supplied complete and ready-to-fly. The XCub is a further scaled development of the CubCrafters Carbon Cub, which the company continues to supply, but with higher performance and incorporating more structural carbon fiber. The XCub was developed over a six-year period and not publicly announced until FAA TC had been completed and issued. The process was completed organically using company resources and did not involve any venture capital, loans nor any advanced customer deposits. XCub is built on a wholly original fuselage design. The CNC-milled 4130 chromoly steel frame meets the latest FAA Part 23 certification standards for 2,300-pound gross weight aircraft. XCub’s useful load is as high as 1,084 pounds. Current Part 23 certification requirements ensure this is the strongest Cub ever produced. It can fly farther, providing greater comfort. It is an airplane that has taken the best from the past and, using the very latest in design, material and manufacturing technology, has established a new standard.
The XCub was approved by the FAA for seaplane operations in December 2017. That same month, EASA approved the XCub design and issued a new type certificate. Four international type certificates have been gained: EASA Dec-2017, Canada Feb-2018, Japan April-2018, and Australia Aug-2018.
CubCrafters increased the horsepower of the XCub line in 2019, offering two new models: the CC19-215 FAA Certified version and the CCX-2300 Builder Assist, both powered by the new CC393i 215 HP engine built by Lycoming.
In December 2021, CubCrafters gained FAA Certification of a new nose wheel version of the XCub, branded the NXCub.
Market Overview
According to a 2022 analysis by research firm Expert Market Research (“EMR”), the global ultralight and light aircraft market was valued at $7.63 billion in 2021. The EMR report says the market is expected to grow at a CAGR of 4.5% in the forecast period of 2022-2027 to reach a value of $9.93 billion. Ultralight and light aircraft are small aircraft with on-board pilot (and perhaps passengers) designed for use in recreation, sports, pilot training, aerial surveys, mapping, research and agriculture, humanitarian backcountry access, and special military missions, as well as business and personal travel.
CubCrafters currently enjoys a dominant market share of the rugged adventure airplane market.
Management Team
Patrick Horgan is President and CEO at CubCrafters. Before he assumed that role, he was the company’s Vice President/Director of Engineering & Product Development for three years, when he led the FAA Part 23 type certificate approval and production certificate approval of CubCrafters’ newest flagship, the XCub. Mr. Horgan also directed the breakthrough certification that authorized the use of experimental avionics in FAA-certified production aircraft, a first in aviation history. He brings over 30 years’ aircraft development and manufacturing experience in general aviation, commercial, and military industries. Prior to service at the company, he was the General Manager at WACO Classic Aircraft Corporation in Battle Creek, Michigan, and was the commercial aircraft manager of the Boeing 777 wheel and brake program for Goodrich Aerospace in Troy, Ohio. He was also a designer on the F/A-18 Super Hornet at McDonnell Douglas (now Boeing) in St. Louis, Missouri. Mr. Horgan holds degrees in aeronautical and astronautical engineering from the University of Illinois, and a certificate in Disruptive Strategy from Harvard Business School. He serves as a member of the Board of Directors of the General Aviation Manufacturers Association and on ASTM aircraft standards committees.
Brad Damm is Vice President at CubCrafters. He has overseen CubCrafters’ sales, marketing, and brand management operations since 2018. Since first joining CubCrafters in 2013, Mr. Damm has served as Factory Direct Sales Manager, the Director of Sales Support, the Global Director of Sales, and the Vice President of Sales and Marketing. During his tenure, the company has seen new sales records year after year across all of CubCrafters new aircraft and kit product lines, and the CubCrafters brand has risen to new levels of awareness and respect with aviation consumers worldwide. Prior to joining the company, he served for over 10 years as the Business Development Manager for one of the largest commercial concrete contractors in the Pacific Northwest, driving the sales and revenue growth that allowed the company to expand from a few dozen to hundreds of employees.
Rick Johnson is the Director of Finance at CubCrafters and has been with the company since 2017. He has 27 years of previous experience as controller and CFO for fruit packing and timber operations in the Pacific Northwest. He holds a Bachelor of Science in Business Administration from Central Washington University.
Christopher Matus is Production Manager at CubCrafters and joined the company in 2011. Before taking that post, he held positions as Fabrication Plant Manager, Machine Shop Manager and CNC Machinist for the company. He has also served as a Combat Engineer in the Washington Army National Guard, deploying to Afghanistan and to natural disasters including the 2014 Oso Mudslide.
Justin Jansky is the Administrative Manager at CubCrafters. He joined the company in 2015 and has a demonstrated history of successful collaboration on major FAA type certification projects in the general aviation industry, specifically under 14 CFR Parts 21 and 23. He is responsible for process management, document control, facilitating FAA certification processes, coordination with FAA delegates and documenting compliance testing. He holds a bachelor’s degree in technology and applied design.
Recent News
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. Merging High Performance, Latest Technology to Bring Unique Perspective to Access the World
- InvestorNewsBreaks – CubCrafters Inc.’s Distinctiveness in Backcountry Aviation Increases Demand for Its Aircraft
- CubCrafters Inc.’s Extension of Long-Term Relationship with US Government Demonstrates Capabilities of Newest Generation of Its XCub Aircraft

REZYFi, Inc.
The QualityStocks Daily Newsletter would like to spotlight REZYFi, Inc.
REZYFi, Inc. is a cannabis mortgage bank servicing the needs of both traditional and non-traditional consumers and businesses. Its target markets include licensed and permitted cannabis companies, owners of real estate who lease to cannabis companies, and companies and individual homeowners seeking a variety of real estate-related first and additional mortgage-based financing and project-specific financings, such as solar installations and real estate development projects.
Headquartered in Miami, Florida, REZYFi operates through two wholly owned subsidiaries – REZYFi Lending, which primarily addresses emerging real estate-related financing opportunities, and ResMac Inc., the company’s traditional mortgage origination, correspondent and servicing operation. REZYFi is currently licensed in 34 U.S. states, with plans to expand to all remaining states later this year.
REZYFi is positioned as one of first cannabis mortgage bankers in the U.S., while most traditional lenders are still reticent to serve the state-licensed cannabis industry.
Operations
REZYFi Lending
REZYFi Lending leverages a wide network to offer options such as 15- and 30-year fixed-rate loans, FHA loans, VA loans, reverse mortgages, jumbo loans and adjustable-rate mortgages.
Looking ahead, the company expects increased funding in marketing and loan agents to drive significant origination growth over the next two years, further supported by the planned launch of a high-margin cannabis division later this year.
ResMac Inc.
ResMac has been in operation for 13 years, having closed more than 20,000 loans for more than 15,000 clients. The company expects to accumulate $285 million in retail origination in 2023, alongside $250 million in wholesale origination for the same period. ResMac is further targeting $600 million in origination through its mortgage correspondent operations for 2023.
Through its ResMac subsidiary, REZYFi operates as a direct lender and originator of residential mortgages, with active mortgage correspondent and mortgage servicing operations. Through its correspondent segment, ResMac primarily purchases and aggregates residential mortgages from trusted third-party originators.
The company intends to harvest the database of customers within its mortgage servicing operations as an essential source of additional growth, especially relative to the new alternative residential loan programs being offered.
Corporate Strengths
- Experience – REZYFi is led by a seasoned management team with significant expertise spanning a wide range of real estate and financing subsectors. The team also has extensive experience in the cannabis and hemp marketplace, which the company intends to leverage as it navigates the changing landscape of the cannabis industry while sourcing the best opportunities in the sector.
- Network of Independent Brokers – Over the past five years, REZYFi has developed an extensive network of independent mortgage-related brokers and licensed loan officers. The company is currently training the network members on its new service offerings, with many already launching sales efforts. REZYFi believes this network will be a vital asset moving forward as other firms in the sector terminate relationships in the face of slowing mortgage business in a rising interest rate environment.
- Proprietary Technology – REZYFi has invested heavily in designing, building and implementing proprietary automated/machine learning technology to shorten loan processing timeframes and increase efficiencies, allowing it to operate its legacy business at staffing levels meaningfully below those of its competitors.
Market Overview
REZYFi’s diversified approach to the real estate lending sector positions it to capitalize on growth in multiple verticals in the years to come.
In the first quarter of 2022, lenders issued 2.71 million residential loans, with the average balance for a first mortgage climbing to a record high of $298,324 in 2021, according to the Mortgage Bankers Association. This trend is expected to continue, with Freddie Mac forecasting a 10.4 percent increase in home prices in 2022 and a 5.0 percent bump in 2023. Growth prospects in the cannabis industry paint a similar picture.
The National Association of Realtors® issued a report in April 2021 examining the correlation between cannabis legalization and real estate demand. In states where prescription and recreational cannabis use is legal, more than a third of surveyed agents reported an increase in demand for warehouses. Likewise, 23 percent of those surveyed reported an increase in demand for storefronts, and 28 percent observed increased demand for land. As other states look to join the 19 that have embraced full cannabis legalization, this rising demand could create an opportunity for REZYFi’s cannabis-focused initiatives.
In total, an analysis by market research firm Business Research Insights projects the global loan servicing market to reach a value of nearly $1.5 billion by 2028, up from $680.8 million in 2021. Those figures represent a CAGR of 11.0 percent during the forecast period of 2022-2028.
Management Team
John Vu, Esq., is CEO of REZYFi, Inc. He has more than two decades of experience in the mortgage and commercial banking industry. He has filled many senior and executive management positions in high-producing mortgage banks, including C-level assignments. He has also served as general counsel for a nationally associated commercial bank. Mr. Vu brings considerable cannabis industry expertise to REZYFi. He has served as a corporate attorney to multiple cannabis cultivators, manufacturers and retailers.
Ji Ji Zhang, Esq., is CFO of REZYFi, Inc. He is a multifaceted entrepreneur who owns a law firm, a portfolio of hotels and a high-producing mortgage bank. Mr. Zhang is also an investor in the development of a cannabis business park. He brings more than five years of experience in mortgage banking to REZYFi, having developed Freddie Mac and HUD licenses and amassed a managed portfolio valued at over $300 million.
Kevin Heckemeyer is President of REZYFi, Inc. He has more than 25 years of experience in mortgage banking. He has built and sold several high producing mortgage businesses. In his current roles with ResMac, he is responsible for production and operations.
Spencer Dang is Chief Credit Officer of REZYFi, Inc. He has more than a decade of experience in mortgage operations. He is a direct endorsement underwriter for HUD and has specialized in non-QM underwriting. Under his watch as an underwriter, he has never had a single repurchase.
Recent News
- REZYFi, Inc. - 420 with CNW — US Border Patrol Sees 95% Drop in Cannabis Seizures as Legalization Spreads
- USDA’s Inclusion of Hemp Data in the 2022 Census for Agriculture is seen as Another Boost to the Cannabis Sector, and REZYFi, Inc. is Set to Benefit from It
- 420 with CNW — Connecticut Clears Some 43,000 Cannabis Records Before Recreational Sales Begin

Silo Pharma Inc. (OTCQB: SILO)
The QualityStocks Daily Newsletter would like to spotlight Silo Pharma Inc. (OTCQB: SILO).
To view the full press release, visit https://ibn.fm/8sNNu
Silo Pharma Inc. (OTCQB: SILO), a developmental stage biopharmaceutical company, is focused on merging traditional therapeutics with psychedelic research for people suffering from indications such as post-traumatic stress disorder (PTSD), fibromyalgia, Alzheimer’s disease, Parkinson’s disease, and other rare neurological disorders. Silo’s mission is to identify assets to license and fund research that the company believes will be transformative to the wellbeing of patients and the health care industry.
Silo is committed to developing innovative solutions to address a variety of underserved conditions. Combining Silo’s resources with world-class medical research partners, the company looks to make significant advances in the medical and psychedelic space.
Silo works to identify and partner with leading medical universities, providing the needed financial resources to develop safe therapeutic treatments while moving cutting-edge research through the clinical stage and into commercialization. The company is well-capitalized with access to additional funds as opportunities present themselves.
Silo recently engaged Donohoe Advisory Associates LLC for consulting and advisory services in connection with the potential uplisting of Silo’s common shares to the Nasdaq Stock Market.
Research
Silo has entered into research agreements and partnerships with multiple leading medical universities.
The company is involved in a sponsored study with Maastricht University utilizing repeated low doses of ketamine and psilocybin to examine the effects on cognitive and emotional dysfunctions in Parkinson’s disease and to understand its mechanism of action. The investigator in the Netherlands is acquiring the substances for the study and will then finalize the documentation to submit to the ethics committee.
Additionally, in June 2021, Silo announced its entry into a scientific research agreement with the University of California San Francisco (UCSF). The agreement will leverage four other clinical trials being planned by the university to determine the effects of psilocybin on inflammation. The study will take place at The Translational Psychedelic Research (TrPR) Program at UCSF.
Silo also recently extended its exclusive option agreement with the University of Maryland, Baltimore (UMB) to explore a novel invention generally known as joint-homing peptides. These peptides are being developed for use in the investigation and treatment of arthritogenic processes and can be used for enhanced targeting of therapeutic agents.
This agreement includes the study of two separate peptides. The first is an option and study for the treatment of arthritis. The second is a patented licensed peptide for the central nervous system, with an initial study for MS autoimmune diseases, in addition to rheumatoid arthritis. Animal studies are underway for both initial indications relating to the UMB agreement, with the potential for studies evaluating additional indications in the future.
Finally, Silo signed an agreement with Columbia University granting it an option to license certain assets currently under development, including an Alzheimer’s disease formulation targeting NDMARs and 5-HT4Rs, as well as a prophylactic treatment for stress-induced disorders and PTSD. Both candidates are currently being tested in mice and have already provided early data.
In addition to its university partnerships, Silo entered a joint venture agreement with Zylo Therapeutics Inc. (“ZTI”) focused on the development of ketamine and psilocybin using ZTI’s Z-Pod™ technology for the transdermal time released delivery of therapeutics. In November 2021, the company announced ZTI’s reception of its first ketamine shipment and initiation of loading ketamine into its Z-Pod technology. In a news release, Eric Weisblum, CEO of Silo, called the development an “important milestone” that will help the company “study the benefits of slow-release transdermal release of Ketamine.”
Market Overview
According to Coherent Market Insights, the fibromyalgia treatment market was valued at $2.78 billion in 2018 and has a projected CAGR of 3.3% over the forecast period 2018 to 2026. Fibromyalgia is a condition that causes pain all over the body, sleep problems, fatigue, and emotional and mental distress.
The global PTSD therapeutics market is expected to reach $10.68 billion by 2026 with a CAGR of 4.5% during the forecast period from 2018 to 2026, according to a report by Credence Research. Growing prevalence of PTSD is the chief factor driving the global treatment market. Increases in events such as wars, combat, and interpersonal violence has been a major contributing factor. Other factors like growing emphasis on rehabilitation initiatives by governments for treating their war veterans has also been facilitating the increase in demand for PTSD therapeutics.
Fortune Business Insights reports the global Parkinson’s disease treatment market is predicted to grow to $8.38 billion by 2026, with a CAGR of 8.1% during the forecast period. Parkinson’s is a neurodegenerative disease of the central nervous system which primarily affects the brain, causing uncontrollable shaking and tremors, difficulties in balance and restricted body movement making it difficult for the person to function or perform a daily routine.
Management Team
Eric Weisblum is CEO and founder of Silo Pharma. He has over 25 years of Wall Street experience, most recently in the biotechnology sector. He has served on the board of Aikido Pharma and was the president of Sableridge Capital. He has a proven track record in licensing therapeutic assets and assisting in their development. He brings to the company nearly 20 years of expertise in structuring and trading financial instruments. He holds a bachelor’s degree from the University of Hartford’s Barney School of Business.
Dr. Kevin Muñoz was appointed to the Silo board of directors in October 2020. He teaches biomedical sciences and medical intervention for the Passaic County Technical Institute. He previously served as Director of Operations at Physical Medicine and Rehabilitation. He began his career with Harlem Health Promotion Center in New York City as a research assistant. He earned a bachelor’s degree from the University of Michigan and a Doctor of Medicine from Xavier University School of Medicine.
Josh Woolley, M.D., Ph.D., is a Scientific Advisor for Silo. He is an associate professor in the Department of Psychiatry and Behavioral Sciences at the University of California, San Francisco. He is also a psychiatrist on staff at the San Francisco Veterans Affairs Medical Center. He is the director and founder of the Bonding and Attunement in Neuropsychiatric Disorders Laboratory. He received both his M.D. and his Ph.D. in Neuroscience from UCSF, where he completed his psychiatry residency training.
Charles Nemeroff, M.D., Ph.D., is a Scientific Advisor for Silo Pharma. He directs the Institute for Early Life Adversity Research within the Department of Psychiatry and Behavioral Sciences as part of the Mulva Clinic for the Neurosciences. He was chair of the Department of Psychiatry and Behavioral Sciences and clinical director of the Center on Aging at the University of Miami Miller School of Medicine. He received his M.D. and Ph.D. in neurobiology from the University of North Carolina School of Medicine.
Silo Pharma Inc. (OTCQB: SILO), closed Tuesday's trading session at $2.89, off by 3.0201%, on 297,222 volume. The average volume for the last 3 months is 296,180 and the stock's 52-week low/high is $2.35/$12.445.
Recent News
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Advances Clinical Study Examining Psilocybin's Effect on Inflammation
- InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Milestone in IND-Enabling Study of Time-Released Ketamine Formulation
- New Psychedelic Journal to Focus on Publishing Psychedelic Research
Cerberus Cyber Sentinel Corp. (NASDAQ: CISO)
The QualityStocks Daily Newsletter would like to spotlight Cerberus Cyber Sentinel Corp. (NASDAQ: CISO).
To view the full press release, visit https://ibn.fm/yEMvQ
Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) is an industry leader in cybersecurity and compliance services. The company leverages an integrated approach to reduce noise and bridge common silos that often limit the effectiveness of cybersecurity programs. Pulling disparate technologies, teams, and vendors together, Cerberus helps its clients enjoy a simpler and more successful journey to cyber resilience. Since 2019, Cerberus Sentinel has worked to rapidly expand by acquiring world-class cybersecurity and compliance businesses with top-tier talent who utilize the latest technology to create innovative protection solutions.
The Cerberus Sentinel workforce is comprised of cybersecurity experts spanning not only global geographies, but also specialties, industries, regulatory frameworks and focus areas. Its team includes audit and compliance specialists, certified forensics experts, ethical hackers, IEEE® certified biometric professionals, security engineers, around-the-clock analysts, and more – all backed by the most respected credentials in the industry. On an ongoing basis, the company works to identify cyber talent that is culturally aligned and that offers operating leverage through both existing customer revenue and relationships.
Cerberus Sentinel has invested in enterprise solutions and executive talent to integrate its different organizations into an ecosystem that works together to provide complete cybersecurity through cross-pollination of solutions that begin at the network level and extend through technologies, people, policy, and practices. This ecosystem is intended to foster additional growth opportunities and drive overall recurring revenue. Once engaged, the company strives to become trusted advisors for customers’ cybersecurity and compliance demands by providing tailored security solutions based upon their organizational needs.
While cyber resilience requires cycles of continuous improvement, it is a journey that few in the current business and security climate seem to understand. With its deep bench of seasoned experts, Cerberus Sentinel works to simplify that journey for its growing customer base, straightening out the curves and speeding up the process to resilience along the way.
Cybersecurity is a Culture, Not a Product
Integrating compliance and security, including principles of security by design, Cerberus Sentinel helps its clients create an organization-wide culture of cybersecurity. Its offerings include audit and compliance, security operations center services, security engineering, virtual Chief Information Security Officer services, incident response, certified forensics, technical assessments and cybersecurity training.
In contrast to the majority of cybersecurity firms that specialize in a specific technology or service, Cerberus Sentinel seeks to differentiate itself by remaining technology agnostic, focusing on accumulating highly sought-after subject matter experts. Cerberus Sentinel believes that bringing together a world-class team of technological experts with multi-faceted proficiency in the critical aspects of cybersecurity is key to providing technology agnostic solutions to its clients in a business ecosystem that suffers from a chronic lack of highly skilled professionals.
Cerberus Sentinel’s goal is to create a culture of security and to help quantify, define and capture a return on investment from information technology and cybersecurity spending. Its end-to-end, holistic process covers every aspect of clients’ cybersecurity and compliance requirements in an effort to promote greater efficiency and strengthen awareness about the integral role of internal team members in the cybersecurity culture of an organization.
As a result of this strategy, Cerberus Sentinel customers receive an efficient engagement from a single partner that covers a wide range of their needs – addressing challenges more thoroughly and resolving problems more rapidly when compared to working with a host of vendors.
Market Outlook
According to an analysis by the firm Research and Markets, the global managed security services market was valued at $22.45 billion in 2020 and is projected to reach $77.01 billion by 2030, growing at a CAGR of 12.8% through the forecast period.
An expected increase in cybercrime, cost effectiveness of provided solutions and stringent mandatory government regulations aimed at protecting corporate data will drive the global managed security services market for the foreseeable future.
In addition, the documented and growing use of mobile devices in the workplace and the rise in captured and stored digital data serve to fuel market growth. Moreover, growing awareness about the critical nature of data security, the growing importance of e-business and demand for customized services is expected to offer ample opportunities for expansion of the market during the forecast period.
Management Team
David Jemmett is CEO and founder of Cerberus Sentinel. He has more than 35 years of executive management and technology experience with telecommunications, managed services, and cybersecurity consulting services. He previously held positions as CEO of GenResults, a leading provider of security consulting services and technology solutions, and as CTO and founder at ClearData Networks, a HIPAA-compliant HealthDATA cloud hosting platform.
Dave Bennett is COO at Cerberus Sentinel. Since 2015, he has served on the President’s STEM Advisory Board of Grand Canyon University. Before joining Cerberus Sentinel, he served as Chief Product Officer at Experian Health and as Senior Vice President, Product for Gainwell Technologies. He has also held positions as Vice President and Worldwide Head of Build, Healthcare and Life Sciences at DXC Technology, and as EVP, Product and Strategy at Orion Health.
Ashley Devoto is President and Chief Information Security Officer at Cerberus Sentinel. Over the past 17 years, Devoto has worked with the cybersecurity elite to design, build, and operate world-class cybersecurity programs for large, diverse organizations in both government and commercial enterprises. Prior to joining Cerberus, Devoto served as CISO for Booz Allen Hamilton, as business information security officer (BISO) at Bank of America, and as a cyberspace operations officer in the United States Air Force.
Deb Smith is CFO at Cerberus Sentinel. Prior to assuming that position, she was the company’s EVP, Finance and Accounting. She has also served as SVP, Global Accounting at International Cruise and Excursions Inc., and as Chief Accounting Officer for BeyondTrust, an information security software company. She has also held the positions of Corporate Controller at Aspect Software and Assistant Controller at JDA Software.
Cerberus Cyber Sentinel Corp. (NASDAQ: CISO), closed Tuesday's trading session at $1.96, off by 5.314%, on 162,916 volume. The average volume for the last 3 months is 160,759 and the stock's 52-week low/high is $1.88/$49.00.
Recent News
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Inks Deal to Acquire South America-Based Cybersecurity Company
- InvestorNewsBreaks - Cerberus Cyber Sentinel Corporation (NASDAQ: CISO) Announces Corporate Rebrand to Reflect Global Organization
- InvestorNewsBreaks - Cerberus Cyber Sentinel Corporation (NASDAQ: CISO) Poised with Solutions as Costs for Cybercrime Expected to Hit $10.5T by 2025
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - Newman Ferrara LLP Investigating Officers and Directors of 180 Life Sciences Corp. (ATNF)
- Aditxt Inc. (NASDAQ: ADTX) - Aditxt, Inc. (NASDAQ: ADTX) Bolsters Goal to Conduct First-In-Human Clinical Trial Evaluating ADI(TM) Tech with Formation of Adimune, Inc. Subsidiary; Appoints Industry Veteran as Subsidiary Chairman and CEO
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Six Marijuana Trends to Watch for in 2023
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) CEO Discusses Promising Future in Learning on Benzinga Podcast
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Releases Report Noting 48.8% Decrease in 2022 Net Loss
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Taps into Arizona's Rich Mining History
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Exits Calendar 2022 with Expected $35 Million in Annual Recurring Revenue
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - America Faces Shortage of Infectious Disease Experts
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - Brain Scientific Signs Agreement with MVAP Medical Supplies to Go After Rapidly Growing Pediatric Neurodiagnostic Space
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Is 'One to Watch'
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Inks Deal to Acquire South America-Based Cybersecurity Company
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - TinyGemsBreaks - CNS Pharmaceuticals Inc.'s (NASDAQ: CNSP) Berubicin Poised to Potentially Change History
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Playing a Critical Role in Helping Companies Use Energy More Efficiently, with Fewer Carbon Emissions
- Coyuchi Inc. - With Sustainability in Mind, Coyuchi Inc. Joins C4 Ecosystem for Developing Climate-Beneficial, Farm-Forward Textiles
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. Merging High Performance, Latest Technology to Bring Unique Perspective to Access the World
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Releases Status Update on Clinical Development Programs
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Reports on New Research in Published Journal Article
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Secures VB100 Certification for Antivirus Product
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Secures Multi-Year Reseller Agreement with Davidson Technologies Inc.
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Ketamine, Smiling Faces Combo Hold Promise in Treating Depression
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Subsidiary Shares Statement on 2023 Golf Cart, LSV Markets
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Analysts Discuss Gold 2023 Outlook
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Three US Firms Win Contracts to Supply Uranium Strategic Reserve
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) Signs $31M Definitive Agreement with Roynat Capital, Export Development Canada
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces Major Project and Corporate Milestone
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Looks for Strong Numbers in Upcoming Quarter
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Enters Option, JV Agreement Regarding Lithium Claims
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - GOP Congresswoman Files Measure to Amend IRS Code So Cannabis Businesses Can Obtain Tax Relief
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Unveils Virtual Sensor for Auto Industry at CES 2023
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Stream of Steady Business on Fr8App Platform
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Poised to Benefit as the Inflation Reduction Act Encourages Homeowners to Go Green
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Exclusive Distribution Agreement
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - Green Hygienics Holdings Inc. Provides Corporate Update
- RYVYL Inc. (NASDAQ: GBOX) - RYVYL Inc Issues Shareholder Letter
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Receives Key Patent from European Patent Office
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc.'s (NASDAQ: CPTN) Vista-X120 Plus Featured in FreightWaves Article
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Eyes Transition of ZVS Prototype from Lab to Real-World Demonstrations
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces US$5 Million Share Repurchase Program
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Inc IDEX Trading Report
- India Globalization Capital Inc. (NYSE American: IGC) - Scientific Review Highlights Stigma Directed at People Living with Chronic Pain
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Key Milestones, 2023 Business Outlook
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Begins Trading on Next-Gen Marketplace
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Kicks Off New Year with Multiple New Contracts, Announces Planned Path to Profitability
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) DehydraTECH(TM)-Processed CBD Can be Administered More Effectively at Lower Levels than Other CBD, Boosting Hope for Hypertension Patients
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - InvestorNewsBreaks - MetAlert Inc. (MLRT) Featured on 'Gametime with Sequire'
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - Mind Cure Health to Acquire South American Utility Company
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Closes Sale of Mindleap Health Inc.
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) CEO to Present at Planet MicroCap Showcase: Virtual 2022
- Mullen Automotive Inc. (NASDAQ: MULN) - Why Mullen (MULN) Stock Is Shaking Off Tesla's EV Plunge TodayMore From InvestorPlace
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTCQB: PLTXF) (Frankfurt: WNT1) Announces Preliminary Q3 2022 Financial Results
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Anticipates Completion of Private Placement of Unsecured Convertible Debentures, Shares
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Issues Corporate Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Expands Scientific Advisory Board to Appoint Genomics Expert
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Releases Q4 2022 Guidance, Projections for 2023
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Receives Approval Recommendation for Retail Cannabis Outlet by San Diego Planning Group
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Releases Shareholder Letter Outlining 2022 Updates, 2023 Milestone Goals
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Board Votes to Eliminate Millions in Debt
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - InvestorNewsBreaks - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF), Snowflake Partner to Help Fill Gap in US Data Market
- REZYFi, Inc. - 420 with CNW — US Border Patrol Sees 95% Drop in Cannabis Seizures as Legalization Spreads
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - SEDAR filing of the unaudited Interim Financial Statements to March 31, and June 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Inc. Joins Digital DJ Pool
- Save Foods Inc. (NASDAQ: SVFD) - This Company Anticipates Massive Results As It Works To Address Food Waste, Loss And Food Safety Challenges
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks – Sharing Services Global Corporation’s (SHRG) Hapi Travel Destinations Providing Max Savings, Travel Perks
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) Helps Safeguard Vital Company Data Through vCISO Contracts That Offer Affordable Alternative to In-house Staffing, Critical for Cannabis and other SMB Operations
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Advances Clinical Study Examining Psilocybin's Effect on Inflammation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Significant Expansion of Addressable Market for Rugged SD7 Device
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Issues Corporate Update
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Receives Notice of Allowance for Final US Patent Covering DLE Technology
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street TV Announces Five Corporate Interviews on its 423rd Show Airing on the Fox Business Network, Tonight, Monday, January 09, 2023, at 10:30 PM PT
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Released End-of-Year Status Update
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces New DSD Partnerships, Expanded Distribution for Alkaline88(R)
- Tingo Inc. (OTC: TMNA) - Four Possible Crypto Trends to Watch in 2023
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Uranium Energy Corp. (NYSE American: UEC) - Top Picks 2023: Uranium Energy (UEC)
- Vision Energy Corp. (OTCQB: VENG) - InvestorNewsBreaks - Vision Energy Corp. (VENG) Incorporates Dutch Subsidiaries
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Completes 4-Week Treatment Protocol for Final Patient in Phase 2 AjDA Trial
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Closing of $8M Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Technology Acquisition and Partnership with Lumeto, Inc. to Expand its Wrap Reality Training Platform
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - Newman Ferrara LLP Investigating Officers and Directors of 180 Life Sciences Corp. (ATNF)
- Aditxt Inc. (NASDAQ: ADTX) - Aditxt, Inc. (NASDAQ: ADTX) Bolsters Goal to Conduct First-In-Human Clinical Trial Evaluating ADI(TM) Tech with Formation of Adimune, Inc. Subsidiary; Appoints Industry Veteran as Subsidiary Chairman and CEO
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Six Marijuana Trends to Watch for in 2023
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) CEO Discusses Promising Future in Learning on Benzinga Podcast
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Releases Report Noting 48.8% Decrease in 2022 Net Loss
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Taps into Arizona's Rich Mining History
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Exits Calendar 2022 with Expected $35 Million in Annual Recurring Revenue
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - America Faces Shortage of Infectious Disease Experts
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - Brain Scientific Signs Agreement with MVAP Medical Supplies to Go After Rapidly Growing Pediatric Neurodiagnostic Space
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Is 'One to Watch'
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Inks Deal to Acquire South America-Based Cybersecurity Company
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - TinyGemsBreaks - CNS Pharmaceuticals Inc.'s (NASDAQ: CNSP) Berubicin Poised to Potentially Change History
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Playing a Critical Role in Helping Companies Use Energy More Efficiently, with Fewer Carbon Emissions
- Coyuchi Inc. - With Sustainability in Mind, Coyuchi Inc. Joins C4 Ecosystem for Developing Climate-Beneficial, Farm-Forward Textiles
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. Merging High Performance, Latest Technology to Bring Unique Perspective to Access the World
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Releases Status Update on Clinical Development Programs
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Reports on New Research in Published Journal Article
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Secures VB100 Certification for Antivirus Product
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Secures Multi-Year Reseller Agreement with Davidson Technologies Inc.
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Ketamine, Smiling Faces Combo Hold Promise in Treating Depression
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Subsidiary Shares Statement on 2023 Golf Cart, LSV Markets
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Analysts Discuss Gold 2023 Outlook
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Three US Firms Win Contracts to Supply Uranium Strategic Reserve
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) Signs $31M Definitive Agreement with Roynat Capital, Export Development Canada
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces Major Project and Corporate Milestone
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Looks for Strong Numbers in Upcoming Quarter
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Enters Option, JV Agreement Regarding Lithium Claims
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - GOP Congresswoman Files Measure to Amend IRS Code So Cannabis Businesses Can Obtain Tax Relief
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Unveils Virtual Sensor for Auto Industry at CES 2023
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Stream of Steady Business on Fr8App Platform
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Poised to Benefit as the Inflation Reduction Act Encourages Homeowners to Go Green
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Exclusive Distribution Agreement
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - Green Hygienics Holdings Inc. Provides Corporate Update
- RYVYL Inc. (NASDAQ: GBOX) - RYVYL Inc Issues Shareholder Letter
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Receives Key Patent from European Patent Office
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc.'s (NASDAQ: CPTN) Vista-X120 Plus Featured in FreightWaves Article
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Eyes Transition of ZVS Prototype from Lab to Real-World Demonstrations
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces US$5 Million Share Repurchase Program
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Inc IDEX Trading Report
- India Globalization Capital Inc. (NYSE American: IGC) - Scientific Review Highlights Stigma Directed at People Living with Chronic Pain
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Key Milestones, 2023 Business Outlook
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Begins Trading on Next-Gen Marketplace
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Kicks Off New Year with Multiple New Contracts, Announces Planned Path to Profitability
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) DehydraTECH(TM)-Processed CBD Can be Administered More Effectively at Lower Levels than Other CBD, Boosting Hope for Hypertension Patients
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - InvestorNewsBreaks - MetAlert Inc. (MLRT) Featured on 'Gametime with Sequire'
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - Mind Cure Health to Acquire South American Utility Company
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Closes Sale of Mindleap Health Inc.
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) CEO to Present at Planet MicroCap Showcase: Virtual 2022
- Mullen Automotive Inc. (NASDAQ: MULN) - Why Mullen (MULN) Stock Is Shaking Off Tesla's EV Plunge TodayMore From InvestorPlace
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTCQB: PLTXF) (Frankfurt: WNT1) Announces Preliminary Q3 2022 Financial Results
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Anticipates Completion of Private Placement of Unsecured Convertible Debentures, Shares
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Issues Corporate Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Expands Scientific Advisory Board to Appoint Genomics Expert
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Releases Q4 2022 Guidance, Projections for 2023
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Receives Approval Recommendation for Retail Cannabis Outlet by San Diego Planning Group
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Releases Shareholder Letter Outlining 2022 Updates, 2023 Milestone Goals
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Board Votes to Eliminate Millions in Debt
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - InvestorNewsBreaks - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF), Snowflake Partner to Help Fill Gap in US Data Market
- REZYFi, Inc. - 420 with CNW — US Border Patrol Sees 95% Drop in Cannabis Seizures as Legalization Spreads
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - SEDAR filing of the unaudited Interim Financial Statements to March 31, and June 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Inc. Joins Digital DJ Pool
- Save Foods Inc. (NASDAQ: SVFD) - This Company Anticipates Massive Results As It Works To Address Food Waste, Loss And Food Safety Challenges
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks – Sharing Services Global Corporation’s (SHRG) Hapi Travel Destinations Providing Max Savings, Travel Perks
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) Helps Safeguard Vital Company Data Through vCISO Contracts That Offer Affordable Alternative to In-house Staffing, Critical for Cannabis and other SMB Operations
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Advances Clinical Study Examining Psilocybin's Effect on Inflammation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Significant Expansion of Addressable Market for Rugged SD7 Device
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Issues Corporate Update
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Receives Notice of Allowance for Final US Patent Covering DLE Technology
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street TV Announces Five Corporate Interviews on its 423rd Show Airing on the Fox Business Network, Tonight, Monday, January 09, 2023, at 10:30 PM PT
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Released End-of-Year Status Update
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces New DSD Partnerships, Expanded Distribution for Alkaline88(R)
- Tingo Inc. (OTC: TMNA) - Four Possible Crypto Trends to Watch in 2023
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Uranium Energy Corp. (NYSE American: UEC) - Top Picks 2023: Uranium Energy (UEC)
- Vision Energy Corp. (OTCQB: VENG) - InvestorNewsBreaks - Vision Energy Corp. (VENG) Incorporates Dutch Subsidiaries
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Completes 4-Week Treatment Protocol for Final Patient in Phase 2 AjDA Trial
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Closing of $8M Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Technology Acquisition and Partnership with Lumeto, Inc. to Expand its Wrap Reality Training Platform
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - Newman Ferrara LLP Investigating Officers and Directors of 180 Life Sciences Corp. (ATNF)
- Aditxt Inc. (NASDAQ: ADTX) - Aditxt, Inc. (NASDAQ: ADTX) Bolsters Goal to Conduct First-In-Human Clinical Trial Evaluating ADI(TM) Tech with Formation of Adimune, Inc. Subsidiary; Appoints Industry Veteran as Subsidiary Chairman and CEO
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Six Marijuana Trends to Watch for in 2023
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) CEO Discusses Promising Future in Learning on Benzinga Podcast
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Releases Report Noting 48.8% Decrease in 2022 Net Loss
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Taps into Arizona's Rich Mining History
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Exits Calendar 2022 with Expected $35 Million in Annual Recurring Revenue
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - America Faces Shortage of Infectious Disease Experts
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - Brain Scientific Signs Agreement with MVAP Medical Supplies to Go After Rapidly Growing Pediatric Neurodiagnostic Space
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Is 'One to Watch'
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Inks Deal to Acquire South America-Based Cybersecurity Company
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - TinyGemsBreaks - CNS Pharmaceuticals Inc.'s (NASDAQ: CNSP) Berubicin Poised to Potentially Change History
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Playing a Critical Role in Helping Companies Use Energy More Efficiently, with Fewer Carbon Emissions
- Coyuchi Inc. - With Sustainability in Mind, Coyuchi Inc. Joins C4 Ecosystem for Developing Climate-Beneficial, Farm-Forward Textiles
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. Merging High Performance, Latest Technology to Bring Unique Perspective to Access the World
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Releases Status Update on Clinical Development Programs
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Reports on New Research in Published Journal Article
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Secures VB100 Certification for Antivirus Product
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Secures Multi-Year Reseller Agreement with Davidson Technologies Inc.
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Ketamine, Smiling Faces Combo Hold Promise in Treating Depression
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Subsidiary Shares Statement on 2023 Golf Cart, LSV Markets
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Analysts Discuss Gold 2023 Outlook
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Three US Firms Win Contracts to Supply Uranium Strategic Reserve
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) Signs $31M Definitive Agreement with Roynat Capital, Export Development Canada
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces Major Project and Corporate Milestone
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Looks for Strong Numbers in Upcoming Quarter
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Enters Option, JV Agreement Regarding Lithium Claims
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - GOP Congresswoman Files Measure to Amend IRS Code So Cannabis Businesses Can Obtain Tax Relief
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Unveils Virtual Sensor for Auto Industry at CES 2023
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Stream of Steady Business on Fr8App Platform
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Poised to Benefit as the Inflation Reduction Act Encourages Homeowners to Go Green
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Exclusive Distribution Agreement
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - Green Hygienics Holdings Inc. Provides Corporate Update
- RYVYL Inc. (NASDAQ: GBOX) - RYVYL Inc Issues Shareholder Letter
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Receives Key Patent from European Patent Office
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc.'s (NASDAQ: CPTN) Vista-X120 Plus Featured in FreightWaves Article
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Eyes Transition of ZVS Prototype from Lab to Real-World Demonstrations
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces US$5 Million Share Repurchase Program
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Inc IDEX Trading Report
- India Globalization Capital Inc. (NYSE American: IGC) - Scientific Review Highlights Stigma Directed at People Living with Chronic Pain
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Key Milestones, 2023 Business Outlook
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Begins Trading on Next-Gen Marketplace
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Kicks Off New Year with Multiple New Contracts, Announces Planned Path to Profitability
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) DehydraTECH(TM)-Processed CBD Can be Administered More Effectively at Lower Levels than Other CBD, Boosting Hope for Hypertension Patients
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - InvestorNewsBreaks - MetAlert Inc. (MLRT) Featured on 'Gametime with Sequire'
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - Mind Cure Health to Acquire South American Utility Company
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Closes Sale of Mindleap Health Inc.
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) CEO to Present at Planet MicroCap Showcase: Virtual 2022
- Mullen Automotive Inc. (NASDAQ: MULN) - Why Mullen (MULN) Stock Is Shaking Off Tesla's EV Plunge TodayMore From InvestorPlace
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTCQB: PLTXF) (Frankfurt: WNT1) Announces Preliminary Q3 2022 Financial Results
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Anticipates Completion of Private Placement of Unsecured Convertible Debentures, Shares
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Issues Corporate Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Expands Scientific Advisory Board to Appoint Genomics Expert
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Releases Q4 2022 Guidance, Projections for 2023
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Receives Approval Recommendation for Retail Cannabis Outlet by San Diego Planning Group
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Releases Shareholder Letter Outlining 2022 Updates, 2023 Milestone Goals
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Board Votes to Eliminate Millions in Debt
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - InvestorNewsBreaks - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF), Snowflake Partner to Help Fill Gap in US Data Market
- REZYFi, Inc. - 420 with CNW — US Border Patrol Sees 95% Drop in Cannabis Seizures as Legalization Spreads
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - SEDAR filing of the unaudited Interim Financial Statements to March 31, and June 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Inc. Joins Digital DJ Pool
- Save Foods Inc. (NASDAQ: SVFD) - This Company Anticipates Massive Results As It Works To Address Food Waste, Loss And Food Safety Challenges
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks – Sharing Services Global Corporation’s (SHRG) Hapi Travel Destinations Providing Max Savings, Travel Perks
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) Helps Safeguard Vital Company Data Through vCISO Contracts That Offer Affordable Alternative to In-house Staffing, Critical for Cannabis and other SMB Operations
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Advances Clinical Study Examining Psilocybin's Effect on Inflammation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Significant Expansion of Addressable Market for Rugged SD7 Device
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Issues Corporate Update
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Receives Notice of Allowance for Final US Patent Covering DLE Technology
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street TV Announces Five Corporate Interviews on its 423rd Show Airing on the Fox Business Network, Tonight, Monday, January 09, 2023, at 10:30 PM PT
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Released End-of-Year Status Update
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces New DSD Partnerships, Expanded Distribution for Alkaline88(R)
- Tingo Inc. (OTC: TMNA) - Four Possible Crypto Trends to Watch in 2023
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Uranium Energy Corp. (NYSE American: UEC) - Top Picks 2023: Uranium Energy (UEC)
- Vision Energy Corp. (OTCQB: VENG) - InvestorNewsBreaks - Vision Energy Corp. (VENG) Incorporates Dutch Subsidiaries
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Completes 4-Week Treatment Protocol for Final Patient in Phase 2 AjDA Trial
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Closing of $8M Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Technology Acquisition and Partnership with Lumeto, Inc. to Expand its Wrap Reality Training Platform
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































