The QualityStocks Daily Stock List
- Cazoo Group (CZOO)
- Tenax Therapeutics (TENX)
- Resolute Mining (RMGGF)
- Petrus Resources (PTRUF)
- Precinct Properties New Zealand (AOTUF)
- Anadolu Efes Biracilik Ve Malt Sanayii (AEBZY)
- Focus Universal (FCUV)
- Stronghold Digital Mining Inc. (SDIG)
- Grapefruit USA Inc. (GPFT)
- Seelos Therapeutics Inc. (SEEL)
- QuantumScape Corp. (QS)
- Freeport-McMoRan Inc. (FCX)
Hycroft Mining Holding Corporation (HYMC)
QualityStocks, MarketBeat, InvestorPlace, The Street, The Stock Dork, Schaeffer's, PennyPro, InsiderTrades and Broad Street reported earlier on Hycroft Mining Holding Corporation (HYMC), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Hycroft Mining Holding Corporation (NASDAQ: HYMC) is a silver and gold producer that is focused on the exploration, mining, operation and development of mines and silver and gold properties.
The firm has its headquarters in Denver, Colorado and was incorporated in 2017, on August 28th. Prior to its name change, the firm was known as Mudrick Capital Acquisition Corp. It operates as part of the metal ore mining industry and mainly serves consumers in the United States.
The company holds interests in the Hycroft mine, which is an open-pit operation that hosts silver and gold deposits. The mine is ranked among the top twenty biggest primary gold deposits in the world. It is also the second biggest gold deposit in the United States. The Hycroft mine is located roughly 50 miles west of Winnemucca, in the state of Nevada. This is near the Kamma Mountains in the Range and Basin physiographic province of northwest Nevada. The heap leach mine’s deposit is broken into 6 major zones, based on alteration, mineralization and geology. The zones include Came, Boneyard, Bay, Central, Vortex and Brimstone. The Hycroft has proven and probable mineral reserves of 479 million ounces of silver and roughly 12 million ounces of gold.
The enterprise is well positioned to unlock the value of its world-class mineral endowment through the ongoing improvement program, its technical plans and the initiatives implemented at the site. This will have a positive effect on the enterprise’s growth and encourage more investments into the firm.
Hycroft Mining Holding Corporation (HYMC), closed Monday's trading session at $0.3991, up 28.7419%, on 41,798,224 volume. The average volume for the last 3 months is 623,948 and the stock's 52-week low/high is $0.31/$3.10.
Aveanna Healthcare (AVAH)
StocksEarning, QualityStocks and MarketBeat reported earlier on Aveanna Healthcare (AVAH), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Aveanna Healthcare Holdings Inc. (NASDAQ: AVAH) is a diversified home care platform firm that is focused on the provision of enteral nutrition, home-based pediatric therapy, adult home health and hospice and private duty nursing services.
The firm has its headquarters in Atlanta, Georgia and was incorporated in 2016, on November 30th. It serves consumers in the United States.
The company’s patient-centered care delivery platform allows patients to minimize overusing high-cost care settings like hospitals by remaining in their homes. It operates through the Medical solutions, Home Health and Hospice and Private Duty Services segments.
The enterprise’s medical solutions segment provides enteral nutrition supplies and durable medical equipment to children and adults, which are delivered on an as-needed or periodic basis. The home health segment offers hospice services for individuals and their families when a life-threatening disease no longer responds to treatments; medical social and aide services; speech, occupational and physical therapy services; and home health services, which include in-home skilled nursing services. On the other hand, its private duty service segment provides home-based and in-clinic pediatric therapy services like occupational, physical and speech services; employer of record support services; unskilled nursing services; services to patients in their pediatric day healthcare centers; nursing services in school settings, which involves caregivers accompanying patients to school; and private duty nursing services, which include in-home skilled nursing services to medically fragile kids.
The firm recently released its latest financial results, with its CEO noting that it successfully refinanced its remaining debt balances, which offered it enhanced financial and operational flexibility.
Aveanna Healthcare (AVAH), closed Monday's trading session at $1.55, up 18.3206%, on 623,948 volume. The average volume for the last 3 months is 1.882M and the stock's 52-week low/high is $0.6664/$6.00.
HIVE Blockchain Technologies (HIVE)
QualityStocks, InvestorPlace, MarketClub Analysis, MarketBeat, StreetInsider, Early Bird, Marketbeat.com, StockMarketWatch, Greenbackers, Hit and Run Candle Sticks, Barchart, Stock Market Watch, WealthMakers, StockOodles, StreetAuthority Daily, The Night Owl, The Online Investor, TopStockAnalysts, Wall Street Resources and smartOTC reported earlier on HIVE Blockchain Technologies (HIVE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
HIVE Blockchain Technologies Ltd (NASDAQ: HIVE) (CVE: HIVE) (FRA: HBF) is a crypto currency mining firm that is focused on building bridges between the blockchain sector and traditional capital markets.
The firm has its headquarters in Vancouver, Canada and was incorporated in 1987, on June 24th. Prior to its name change in September 2017, the firm was known as Leeta Gold Corp. It serves consumers around the globe.
The company is focused on being the leading listed blockchain infrastructure firm by accelerating the development of the blockchain sector. It operates through the Norway, Switzerland, Iceland, Sweden and Canada geographical segments. The majority of the company’s revenue is generated from Canada. The company offers its shareholders a portfolio of crypto-coins as well as operating margins of digital currency mining.
The enterprise is involved in the sale and mining of digital currencies. It validates transactions on blockchain networks. It also owns state-of-the-art green energy-powered data center facilities in Iceland, Sweden and Canada which generate minted digital currencies like Ethereum and Bitcoin on the cloud, around the clock. The enterprise’s projects include Iceland Crypto currency mining. In addition to this, it also owns a data center campus in New Brunswick.
The company recently released its annual report for the year ended March 2021 which shows significant increases in the company’s net income and gross mining margin. It is now focused on improving its profitability and efficiency across its mining operations by maximizing its infrastructure and electrical capacity through the installation of new mining equipment, lowering its costs and optimizing crypto currency mining output.
HIVE Blockchain Technologies (HIVE), closed Monday's trading session at $2.71, up 15.3191%, on 1,888,899 volume. The average volume for the last 3 months is 54.443M and the stock's 52-week low/high is $1.36/$11.70.
Tenax Therapeutics (TENX)
QualityStocks, TraderPower, StockMarketWatch, BUYINS.NET, TradersPro, Schaeffer's, StocksImpossible, OTCBB Journal, MarketClub Analysis, First Penny Picks, MarketBeat, StreetInsider, The Online Investor, The Stock Dork, PennyStockScholar, PennyStockProphet and RedChip reported earlier on Tenax Therapeutics (TENX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Tenax Therapeutics Inc. (NASDAQ: TENX) (FRA: YBOC) is a specialty pharmaceutical firm which is focused on the identification, development and commercialization of products which address unmet medical needs for pulmonary and cardiovascular ailments in Canada and the United States.
The firm has a remarkable scientific advisory team, which includes recognized international experts in pulmonary hypertension. It is based in Morrisville, North Carolina and was incorporated in 1967, on May 26. Before changing its name to Tenax Therapeutics Inc., the firm was known as Oxygen Biotherapeutics Inc.
The company is also engaged in the cosmetic industry and has developed the DERMACYTE line of skin care products, which addresses various common aging issues, including blemishes, skin tone, puffiness, wrinkles, fine lines and more. This is in addition to developing a PFC-based topical cream for use in dermatological indications and wounds.
Additionally, it owns the rights to commercialize and develop levosimendan in North America, which was developed as a pulmonary hypertension treatment associated with preserved ejection fraction and heart failure. Apart from levosimendan, the firm is also engaged in the development of a tyrosine kinase inhibitor indicated for treating pulmonary arterial hypertension, known as Imatinib.
The company recently reported progress in its phase II study of its candidate levosimendan as a treatment for heart failure and pulmonary hypertension. Their statement noted that the drug effectively decreased pulmonary capillary wedge pressure and was the first drug to show improved cardiovascular hemodynamics. Given that pulmonary hypertension and heart failure with preserved ejection fraction has no effective medical treatments, the company’s candidate may well be on its way to becoming the first approved treatment for this disease. This will help address a previously unmet clinical need, which will help patients who suffer from this ailment, while also benefitting the firm and its shareholders.
Tenax Therapeutics (TENX), closed Monday's trading session at $0.6542, up 14.8122%, on 54,442,608 volume. The average volume for the last 3 months is 545,559 and the stock's 52-week low/high is $0.452/$17.00.
Zynex, Inc. (ZYXI)
TaglichBrothers, QualityStocks, MarketBeat, MarketClub Analysis, Trades Of The Day, SmarTrend Newsletters, Daily Trade Alert, The Wealth Report, BUYINS.NET, Zacks, FeedBlitz, InvestorPlace, SeeThruEquity Research, SmallCapVoice, StockMarketWatch, FNNO Newsletters, The Street, TradersPro, Daily Markets and Schaeffer's reported earlier on Zynex, Inc. (ZYXI), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Zynex, Inc. is a medical technology company listed on the OTCQB. It specializes in the manufacture and sale of non-invasive medical devices for pain management, stroke rehabilitation, neuro diagnostics, cardiac and blood volume monitoring. In addition, the Company is developing a new blood volume monitor (non-invasive Blood Volume Monitor, CM-1500) for use in hospitals and surgery centers. Zynex was founded by Mr. Thomas Sandgaard, the Company’s current Chief Executive Officer and Chairman. Zynex has its corporate office in Englewood, Colorado.
The Company’s product lines are completely developed, Food and Drug Administration (FDA)-cleared, and commercially sold internationally. Zynex engineers, manufactures, markets, and sells its own design of medical devices in three subsidiaries.
Zynex Medical is a provider of electrotherapy products for home use. Zynex Monitoring Solutions develops products for cardiac monitoring for use in hospitals. Zynex NeuroDiagnostics develops devices for EMG and EEG diagnostic purposes in the neurology clinic markets.
The Company’s belief is that its non-invasive Blood Volume Monitor, CM-1500, will be the first device to provide an indication of fluid balance and blood loss in the operating room or potential post-surgical internal bleeding in recovery. Zynex markets and sells its own design of electrotherapy medical devices used for pain management and rehabilitation.
Furthermore, Zynex markets and sells its proprietary NeuroMove device designed to help recovery of stroke and spinal cord injury patients. The design of the NeuroMove™ device is to assist stroke survivors in regaining movement using the brain's ability to rewire itself, also known as "neuro-plasticity".
Zynex, Inc. (ZYXI), closed Monday's trading session at $10.94, up 14.7954%, on 547,509 volume. The average volume for the last 3 months is 48,421 and the stock's 52-week low/high is $4.97/$17.25.
Caravelle International (CACO)
We reported earlier on Caravelle International (CACO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Caravelle International Group (NASDAQ: CACO) is a global ocean technology firm engaged in the provision of ocean transportation services.
The firm has its headquarters in Singapore and was incorporated in 2021. It operates as part of the marine shipping industry, under the industrials sector. The firm serves consumers around the globe, with a focus on those in Singapore.
The company’s goal is to achieve the net-zero emission standard. Its segments include ocean transportation and heating business. Its business comprises of two sectors; the traditional business in international shipping, operated by the Topsheen Companies (Topsheen Shipping Group Corporation (Samoa) and its subsidiaries) and the new CO-Tech business, under Singapore Garden Technology Pte. Ltd. The CO-Tech business is a new development building upon the existing shipping business. It enables wood desiccation during the maritime shipping process, with full utilization of the shipping time, space, and the waste heat of exhaust gas from the shipping vessels. This replaces the traditional wood desiccation model using onshore drying kilns, which burns fossil fuels and emits greenhouse gases and other pollutants. The company also provides transportation services under voyage contracts and vessels services for and on behalf of ship owners.
The enterprise recently entered into a partnership with a U.S. corporation, which will involve the manufacture of wood products in Gabon for export to the U.S. This will generate additional revenues into the firm while also opening it up to new growth and investment opportunities.
Caravelle International (CACO), closed Monday's trading session at $0.939599, up 2.2748%, on 48,421 volume. The average volume for the last 3 months is 2.886M and the stock's 52-week low/high is $0.842201/$1.60.
Atara Biotherapeutics (ATRA)
MarketBeat, StreetInsider, TraderPower, InvestorPlace, Kiplinger Today, MarketClub Analysis, StockMarketWatch, Trades Of The Day, The Street, Schaeffer's, StreetAuthority Daily, Barchart, Marketbeat.com, Wall Street Daily, Market Intelligence Center Alert, Daily Trade Alert, BUYINS.NET, Stock Up Featured, Street Insider, Zeke Truligio, The Online Investor, Wealth Insider Alert, Zacks, Investing Daily and Equities.com reported earlier on Atara Biotherapeutics (ATRA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Atara Biotherapeutics Inc. (NASDAQ: ATRA) (FRA: AT2) (LON: 0H1V) is an allogeneic T-cell immunotherapy firm that is focused on the development of treatments for patients with hematologic cancers, solid tumors and autoimmune illnesses.
The firm has its headquarters in Thousand Oaks, California and was incorporated in 2012, on August 22nd by Isaac E. Ciechanover. It operates as part of the biotechnology industry, under the healthcare sector. The firm primarily serves consumers in the United States.
The company harnesses the natural biology of T cells—our body’s most effective weapon in fighting disease—to develop transformative therapies. It is party to a license agreement with Memorial Sloan Kettering Cancer Center, as well as a strategic collaboration with H. Lee Moffitt Cancer Center. The company is also party to a license and research and development collaboration agreement with QIMR Berghofer Medical Research Institute.
The enterprise’s product pipeline is comprised of a T-cell immunotherapy program dubbed Ebvallo (tabelecleucel), which is in Phase III clinical trials for the treatment of epstein-barr virus (EBV) driven post-transplant lymphoproliferative disease. This formulation’s effectiveness in treating nasopharyngeal carcinomais is also being tested. The enterprise’s CAR T immunotherapy pipeline products include ATA3271 and ATA2271, to treat mesothelin; and ATA3219 for the treatment of B-cell malignancies. It also develops ATA188, which is in Phase II trials to treat multiple sclerosis.
The firm’s Ebvallo formulation recently received authorization for marketing from the European Commission for the treatment of relapsed or refractory Epstein-Barr virus. This move will not only benefit patients with this illness but also encourage more investments into the firm, while also bolstering its overall growth.
Atara Biotherapeutics (ATRA), closed Monday's trading session at $3.12, up 10.2473%, on 2,886,203 volume. The average volume for the last 3 months is 758,849 and the stock's 52-week low/high is $2.66/$10.01.
CaesarStone (CSTE)
Zacks, The Online Investor, MarketBeat, StreetInsider, MarketClub Analysis, Hit and Run Candle Sticks, InvestorPlace, Daily Trade Alert, Marketbeat.com, The Street, StreetAuthority Daily, Top Pros' Top Picks, Trades Of The Day, Louis Navellier, Investors Alley, Wealth Insider Alert, WealthMakers, Greenbackers, FreeRealTime and Rick Saddler reported earlier on CaesarStone (CSTE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
CaesarStone Ltd (NASDAQ: CSTE) is a company focused on the development, manufacture and marketing of engineered quartz and other surfaces.
The firm has its headquarters in Menashe, Israel and was incorporated in 1987. Prior to its name change in June 2016, the firm was known as CaesarstoneSdot Yam Limited. It operates as part of the building products and equipment industry, under the industrials sector. The firm serves consumers across the globe, with a focus on those in Israel, Australia, the United States, Latin America, Canada, Asia, Africa, Europe and the Middle East.
The enterprise’s engineered quartz slabs are primarily used as outdoor and indoor kitchen countertops in the remodeling and renovation construction end markets. Its products are also used in other applications, including wall panels, vanity tops, floor tiles, back splashes, stairs, furniture, and other interior and exterior surfaces that are used in a range of residential and non-residential applications. The enterprise also provides porcelain products under the Lioli brand for flooring and cladding applications, as well as resells natural stones, various ancillary fabrication tools, and installation accessories. This is in addition to selling sinks and materials. It sells its products directly to fabricators, sub-distributors, and resellers; and through a direct sales force and indirect network of independent distributors, under the Caesarstone brand.
The company recently announced its latest financial results showing increases in its annual revenue. It remains focused on executing its growth strategy while meeting the growing demand of its products from consumers. This will generate additional revenues while also creating shareholder value.
CaesarStone (CSTE), closed Monday's trading session at $4.73, off by 4.6371%, on 762,544 volume. The average volume for the last 3 months is 678,462 and the stock's 52-week low/high is $4.52/$11.66.
Sunworks (SUNW)
MarketClub Analysis, TradersPro, InvestorPlace, MarketBeat, Trading Concepts, Trades Of The Day, Broad Street, StockMarketWatch, QualityStocks, Market Intelligence Center Alert, OTCBB Journal, PoliticsAndMyPortfolio, StocksImpossible, The Bowser Report, TopPennyStockMovers, Zacks, StocksEarning, StreetInsider, Marketbeat.com, The Street, InvestorsUnderground, Wealth Insider Alert, Daily Trade Alert, Cabot Wealth, BUYINS.NET and StockEarnings reported earlier on Sunworks (SUNW), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Sunworks Inc. (NASDAQ: SUNW) (FRA: 7SL2) is a company engaged in the provision of photovoltaic and battery-based power and storage systems for the agricultural, commercial, industrial, public works and residential markets.
The firm has its headquarters in Provo, Utah and was incorporated in 1983 by Christopher T. Kleveland, Mark P. Harris and Roland F. Bryan. Prior to its name change in March 2016, the firm was known as Solar3D Inc. It operates as part of the solar industry, under the technology sector. The firm serves consumers in North America.
The company’s mission is to help homeowners and businesses take control of their electric costs through solar. It operates through the Solcius and Sunworks business segments. The Solcius segment mainly focuses on residential projects. Here, the company designs, arranges financing, integrates, installs and manages systems, primarily for residential homeowners. On the other hand, the Sunworks segment is involved in the commercial projects including commercial, agricultural, industrial, and public works projects. Here, the company designs, arranges financing, integrates, installs, and manages systems ranging in size from approximately 2 kilowatts for residential projects to multi-megawatt (MW) systems for larger ACI and public works projects. ACI installations have included installations at manufacturing plants, office buildings, warehouses, churches, service stations and agricultural facilities such as farms, dairies and wineries.
The enterprise is focused on advocating for the advancement of solar power, which will support the transition to a greener and more sustainable future. This is in addition to generating revenues for the firm as well as boosting shareholder value.
Sunworks (SUNW), closed Monday's trading session at $1.67, off by 5.6497%, on 678,462 volume. The average volume for the last 3 months is 3,000 and the stock's 52-week low/high is $1.23/$4.72.
Robex Resources (RSRBF)
We reported earlier on Robex Resources (RSRBF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Robex Resources Inc. (OTC: RSRBF) (CVE: RBX) (FRA: RB4) is a junior operation and exploration mining firm focused on exploring for, developing and producing gold from mineral properties.
The firm has its headquarters in Québec, Canada and was incorporated in 1985, on June 14th. It operates as part of the gold industry, under the basic materials sector. The firm serves consumers around the globe.
The company operates the Nampala mining permit located in southern Mali. It also holds 5 exploration permits, including Mininko and Kamasso exploration permits located in southern Mali; Sanoula and Diangounte exploration permits situated in west Mali. Its Mininko exploration permits cover over 62 km2 and is located around 57 km to the southwest of the town of Sikasso and 21 km south of Niena village. Its Kamasso exploration permits cover over 100 km2 and is located about 74 km southwest of Sikasso and 35 km south of Niena village. Its Gladie exploration permits cover over 52 km2. Its Sanoula exploration permit covers approximately 31.5 km2. It is located around 58 km north-northwest of the town of Kenieba and 120 km south of the city of Kaye. Its Diangounte license covers approximately 52.14 km2.
The enterprise, which recently announced their latest financial results, remains focused on having a significant positive environmental and social impact while also meeting its strategic goals. This will encourage more investments into the firm and bolster its overall growth. In addition to this, it is strengthening its teams to lead the Kiniero project until it enters into operation.
Robex Resources (RSRBF), closed Monday's trading session at $0.2104, even for the day. The average volume for the last 3 months is 16.532M and the stock's 52-week low/high is $0.1801/$0.34105.
Newmont Corporation (NEM)
MarketClub Analysis, InvestorPlace, The Street, Kiplinger Today, Schaeffer's, StocksEarning, MarketBeat, INO.com Market Report, The Online Investor, Barchart, StreetAuthority Daily, Daily Trade Alert, TopStockAnalysts, Top Pros' Top Picks, StreetInsider, Louis Navellier, Streetwise Reports, Daily Wealth, Investopedia, SmarTrend Newsletters, Zacks, TradingMarkets, Uncommon Wisdom, Money Morning, Marketbeat.com, PROFIT CONFIDENTIAL, The Growth Stock Wire, Trades Of The Day, Wealth Daily, TheStockAdvisor, The Wealth Report, Lebed.biz, ProfitableTrading, QualityStocks, Wall Street Grand, Cabot Wealth, Investing Signal, Dividend Opportunities, TheStockAdvisors, The Best Newsletters, Market Intelligence Center Alert, Trading Markets, National Inflation Association, All about trends, InvestmentHouse, Market FN, Energy and Capital, Wyatt Investment Research, Money and Markets, Darwin Investing Network, Stockhouse, AllPennyStocks, Buttonwood Research, Wall Street Daily, OTC Stock Pick, TradingAuthority Daily, InvestorsObserver Team, Daily Markets, Investment House, Street Insider, Wall Street Greek, Investors Alley, StreetAlerts, The Tycoon Report, Trading Tips, Investiv, Dynamic Wealth Report, StockEarnings, Daily Profit, Investment U, Trader Jack, Investing Futures, InvestorIntel, FutureMoneyTrends.com, Trading Concepts, The Stock Enthusiast, Market Authority, Wealth Insider Alert, Short Term Wealth, MarketWatch, DividendStocks, 24/7 Trader, StockTwits, FNNO Newsletters, Global Equity Alert, Investing Daily, equities Canada, Insider Wealth Alert, Candle Stick Forum, CrushTheStreet.com, CNBC Breaking News, Early Bird, Global Equity Report, ChartAdvisor, INO Market Report, Profits Run, Weekly Wizards, Wall Street Window, VectorVest, Vantage Wire, UndiscoveredEquities, Trade of the Week, Total Wealth, The Weekly Options Trader, The Wall Street Transcript, The Street Report, The Motley Fool, Super Stock Picker, SmallCap Network, One Hot Stock, OTCPicks, Investor Update, Investors Insights, Jim Cramer, Leeb's Market Forecast, MarketClub, Small Cap Firm, Normandy Investment Research, Seeking Alpha, Penny Detectives, Penny Stock Buzz, PennyStockProphet, Power Profit Trades, Profitable Trader Authority, Investor Guide and Navellier Growth reported earlier on Newmont Corporation (NEM), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Chairman emeritus of Franco-Nevada and CEO of Fireside Investments Pierre Lassonde believes that gold will trade at as high as $2,400 per ounce by the year 2028. The precious metal will see its value soar as nations such as Russia get rid of the U.S. dollars they hold after the greenback was weaponized following the Kremlin’s invasion of Ukraine, Lassonde said.
The former chairman of gold-focused royalty and streaming company Franco-Nevada Corporation suggested that we could see the emergence of a dual currency system by 2028.
Speaking with Kitco News lead anchor and editor-in-chief Michelle Makori at the BMO Metals, Mining & Critical Metals Conference, Lassonde said that he saw gold trading at around $2,300 to $2,400 over the next five years. He noted that the development of a dual system of currency and payment as well as high gold prices will be mainly precipitated by the ongoing Russia-Ukraine war.
The surge in gold prices began in 2022 after central banks around the world started buying up gold to add to their reserves, causing gold demand to skyrocket. Last year saw the most central bank gold purchases since 1950 and represented the 13th consecutive year of net gold purchases by central banks.
Lassonde suggested that central banks in Brazil, Russia, India, China and South Africa (BRICS) have beefed up their gold purchases as part of efforts to replace the dollar as a reserve currency. He said that although he didn’t know if these central banks will begin using gold as a new currency, he believed that the end result would be a dual system of currency and payments.
Nations such as China, Russia and India are particularly worried about the United States influencing their internal affairs, Lassonde said, and separating themselves from the dollar and the control it grants the U.S. will likely grant them more autonomy. Central banks continued purchasing gold into 2023, collectively adding 31 tons to global gold reserves, and they are expected to remain net gold purchasers throughout 2023.
Furthermore, Lassonde predicted a recession and stock market crash this year, adding that the economic upheaval would ultimately benefit gold. America’s economy will likely enter a recession over the next two quarters, he said, resulting in a “fairly significant recession” that will bring the S&P down by 30%.
Action by the U.S. Federal Reserve would come in too late, and the U.S. dollar would go down while investors overwhelmingly turned their attention to gold.
According to Lassonde, gold equities would continue functioning well under pressure and remain steady as they carry dividend yields.
The expected rise in the price of gold over the coming years is likely to boost the balance sheets of major extraction companies such as Newmont Corporation (NYSE: NEM) if they keep their costs down and optimize all their operations during this inflationary period.
Newmont Corporation (NEM), closed Monday's trading session at $45.12, up 7.0209%, on 16,698,437 volume. The average volume for the last 3 months is 17.903M and the stock's 52-week low/high is $37.45/$86.37.
Lucid Motors (LCID)
Green Car Stocks, InvestorPlace, Schaeffer's, The Street, QualityStocks, MarketClub Analysis, Early Bird, StockEarnings, MarketBeat, StocksEarning, Investopedia, The Online Investor, Daily Trade Alert, Trades Of The Day, Kiplinger Today, INO Market Report, Louis Navellier, GreenCarStocks, The Wealth Report, InvestorsUnderground, AllPennyStocks, Green Energy Stocks, The Stock Dork, Wealth Whisperer and Zacks reported earlier on Lucid Motors (LCID), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
When electric cars began to proliferate in the automobile industry at a quicker pace, safety was and has always been a top concern for potential buyers. The Insurance Institute for Highway Safety (IIHS), a well-known vehicle safety organization, presented evidence that electric vehicles have become just as safe as traditional ones. Actually, according to a review of insurance evidence, accident claims are less common for electric vehicles.
This information is two years old. A sizable number of electric vehicles received the reputable IIHS Top Overall Pick honors in 2022 through 2023. Those who won underwent meticulous crash tests, much like conventional vehicles, and demonstrated their safety in endless accidents, different overlapping collisions and additional IIHS-required trials.
Here’s a list of the most secure electric vehicles for 2022 through 2023, according to crash study results.
2023 Volkswagen ID.4
The safe electric vehicle to buy in 2023 is the VW ID.4. The 2023 Best Safety Choice+ distinction was given by IIHS to this all-around outstanding ride, with the exception of the headlights. Also, to be noted, the 2023 Volkswagen ID.4 is highly rated for forward collision avoidance (vehicle-to-pedestrian, all the time) as opposed to an outstanding rating. NHTSA rated the VW ID.4 as the best safety choice from the test results, although it lost one star for rollover-rated vehicles.
2023 Tesla Model Y
The Tesla Model Y is another vehicle among the 2023 best safety choices with the + designation. The majority of its crash testing yielded outstanding results for this electric vehicle, with only the headlamp (on certain trims) and LATCH (ease of use) sections receiving acceptable ratings. The NHTSA accorded the Model Y in its earlier manufacturing years all-star safety ratings.
2023 Subaru Solterra
The 2023 Subaru Solterra receives the best green scores across areas from the IIHS, earning it the 2023 Best Safety Choice title. Nonetheless, three instances where the electric vehicle received a lower rate have been highlighted.
2023 Audi E-Tron
This is a gorgeous electric vehicle with heart-pounding performance in all trims that also excelled in the crash tests. IIHS awarded this vehicle top marks in every crash test with the exception of the headlights, designating it the best safety choice for 2022. The vehicle has outstanding safety ratings from the NHTSA as well, despite losing a star because of a frontal collision.
2023 Kia EV6
This 2023 Kia EV6 is simple to like. While being brand new to the automotive industry, the vehicle offers exceptional performance, is practical and comfortable, and attractive inside and out. On top of this, the IIHS labels this car as the best safety choice for 2022. The design and safety cages are the only components of the electric vehicle that received a low grade.
The test results above don’t in any way imply that other models from manufacturers such as Lucid Motors (NASDAQ: LCID) aren’t safe or didn’t pass crash tests. Rather, the ranking brings to attention those models that excelled during the tests.
Lucid Motors (LCID), closed Monday's trading session at $7.84, off by 0.381194%, on 17,990,516 volume. The average volume for the last 3 months is 775,506 and the stock's 52-week low/high is $6.09/$28.135.
The QualityStocks Company Corner
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX)
- HeartBeam Inc. (NASDAQ: BEAT)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
- InMed Pharmaceuticals Inc. (NASDAQ: INM)
- Tingo Inc. (OTC: TMNA)
- Prime Harvest Inc.
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
- Silo Pharma Inc. (OTCQB: SILO)
- CISO Global, Inc. (NASDAQ: CISO)
- India Globalization Capital Inc. (NYSE American: IGC)
- Advanced Container Technologies Inc. (OTC: ACTX)
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF)
McEwen Mining Inc. (NYSE: MUX) (TSX: MUX)
The QualityStocks Daily Newsletter would like to spotlight McEwen Mining Inc. (NYSE: MUX) (TSX: MUX).
•McEwen Mining subsidiary McEwen Copper has been gaining increasedfinancial support as it develops a major copper mine opportunity inArgentina described as one of the 10 largest undeveloped depositsin the world
•Mining giant Rio Tinto’s venture Nuton LLC has boosted last year’s$25 million investment into McEwen Copper with a new $30 millionaddition, all on top of the recent Stellantis investment ofapproximately $155 million
•The project in Argentina’s San Juan province benefits from amining-friendly government and population
•The demand for copper is expected to continue to grow as a resultof its importance to green energy transition efforts and ongoingchallenges the metal’s supply stream faces
The strength of metals explorer McEwen Copper’s profile and of itsparent company
McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) is an asset rich diversified gold and silver producer in the Americas and has a large exposure to copper through its subsidiary, McEwen Copper, owner of the Los Azules copper deposit in Argentina, believed to be the 9th largest undeveloped copper resource in the world.
Led by a management team with a track record of success, MUX owns and operates mines in some of the most prolific gold producing regions in the Americas. In recent months, the company has undertaken strong actions to lower production costs and increase production across its portfolio of gold assets, driving some costs below the industry average. Gold and copper prices are forecast to enter a major uptrend over the next couple years. McEwen Mining is laying the groundwork to capitalize on this opportunity now.
Seldom is management so aligned with investors’ interests with a commitment to the company’s success. CEO Rob McEwen maintains a 17% ownership stake in McEwen Mining and a 15% ownership in McEwen Copper with a combined cost base of roughly $220 million. McEwen founded Goldcorp, where he took the company from a market capitalization of $50 million to over $8 billion, and that same vision led MUX to create McEwen Copper.
For McEwen Mining shareholders, the company’s 68% stake in McEwen Copper is expected to be a gamechanger, turbocharging MUX by creating the world’s next copper unicorn.
McEwen Copper
Most mined copper is currently used in infrastructure, with new critical demand emerging for use in the electrification of transportation and the global energy transformation. The price of copper rose from a low of about $2 per pound two years ago to over $4 per pound today, and strong demand is expected to continue to soar. A study by S&P Global, titled The Future of Copper: Will the Looming Supply Gap Short-circuit the Energy Transition?, projects global copper demand to nearly double over the next decade, from 25 million metric tons today to about 50 million metric tons by 2035. Based on current trends, S&P Global projects annual supply shortfalls to reach nearly 10 million metric tons in 2035.
McEwen Mining is a 68% shareholder in McEwen Copper, holder of a 100% interest in the Los Azules copper project in San Juan, Argentina, which was ranked the 9th largest undeveloped copper deposit in the world by Mining Intelligence (2022). Its current copper resources are estimated at 10.2 billion pounds at a grade of 0.48% Cu (Indicated category) and an additional 19.3 billion pounds at a grade of 0.33% Cu (Inferred category). McEwen Copper also owns a copper exploration project in Nevada, called Elder Creek.
In a 2017 Preliminary Economic Assessment (PEA), Los Azules was estimated to have a 36-year life, but indications are that the project could ultimately become an even larger mine, with a longer life, since in the assessment, only 55% of the known copper resources are to be mined. Numerous drill holes have shown strong copper mineralization extending below the PEA pit bottom. Its average annual production for its first 13 years was pegged at 415 million pounds of copper in the 2017 PEA – enough copper to supply 2.2 million electric vehicles per year.
In August 2022, McEwen Copper closed its non-brokered, private placement offering of $82 million, after securing a $25 million investment from mining giant Rio Tinto’s technology arm, Nuton LLC. This gives McEwen Copper an imputed value of $258 million, which would give McEwen Mining’s 68% interest a value of approximately $3.70/share. Additional value can be attributed to McEwen Mining’s 1.25% net smelter royalty on both the Los Azules and the Elder Creek projects.
“We completed an $82 million financing for McEwen Copper in a very tough equity market. Rio Tinto, the second largest mining company in the world, through its subsidiary Nuton, now owns 9.7% of McEwen Copper, a result of its investment of $25 million,” Rob McEwen stated in a news release. “Also, Nuton is testing the Los Azules copper mineralization to see if it can accelerate and increase copper recoveries. Another of Rio Tinto’s subsidiaries, Kennecott Exploration, signed an option to earn a 60% interest in McEwen Copper’s other copper project, Elder Creek, by spending $18 million on exploration.”
The Elder Creek project is prospective for porphyry copper and gold mineralization and is well situated in a district hosting several large copper and gold mines, including Marigold, Lone Tree and Phoenix. Kennecott Exploration will be the operator of the exploration program. McEwen Mining holds a 1.25% net smelter return (NSR) royalty on the Elder Creek property.
Following the capital raise, McEwen Copper is well-funded to advance its Los Azules Project. Publication of an updated PEA on the Los Azules copper project is planned for Q1 2023. In Q2, an IPO is planned, along with MUX completing a secondary offering, assuming no further private placements in the interim. MUX is strategically reducing its interest to increase its treasury, in order to reduce debt and fund the further development of its gold and silver mines.
McEwen Copper currently has an implied market cap of over $258 million, based on its most recently completed financing. However, when its Los Azules copper project is compared with other recent transactions and market valuations of copper projects in the same region, it appears very undervalued.
MUX’s management believes its ownership stake in McEwen Copper is not currently reflected in the share price of the company. In fact, it is management’s belief that the combined value of its 68% interest in McEwen Copper, plus its gold mines and portfolio of mineral royalties, represents a share value ranging from a low of $8 to a high of $30 per share. Rob McEwen provides a full breakdown of this valuation estimate in a news release detailing the company’s Q3 2022 results.
Gold & Silver Projects
The Fox Complex
McEwen Mining owns a 100% stake in the Fox Complex in the heart of a prolific gold district in Timmins, Canada.
“When MUX bought the Fox Complex, in late 2017, it was a distressed asset with a history of high operating cost/oz. While it has taken longer than I expected, the cost to produce an ounce of gold is significantly lower,” CEO Rob McEwen stated in a news release. “I am pleased to say that in Q3 our cash cost/oz at Fox fell to $774, our lowest since mid-2018. This is well below the industry average. With our mine operating much more efficiently, our next important area to improve at Fox is the process plant (mill). Specifically, we need to increase the throughput because our mine is now producing more ore than our mill can process. As a result, we have a large surface stockpile of ore equivalent to more than two months of production.”
This ore stockpile contains approximately 10,000 ounces of gold representing a potential source of $12 million in free cash flow.
Located in one of the most prolific gold production areas in the world, along the Destor-Porcupine Fault Zone within the Abitibi Greenstone Belt, the Fox Complex includes the Black Fox mine and Froome mine which together have, so far, produced in excess of 1,000,000 ounces of gold. Also, it includes the Grey Fox and Stock deposits that have an estimated additional 1,600,000 ounces in reserves and resources. The 2.7-billion-year-old Abitibi Greenstone Belt, formed by ancient volcanic activity, has proved to be one of the world’s richest and most abundant gold regions, boasting total gold content of over 300 million ounces.
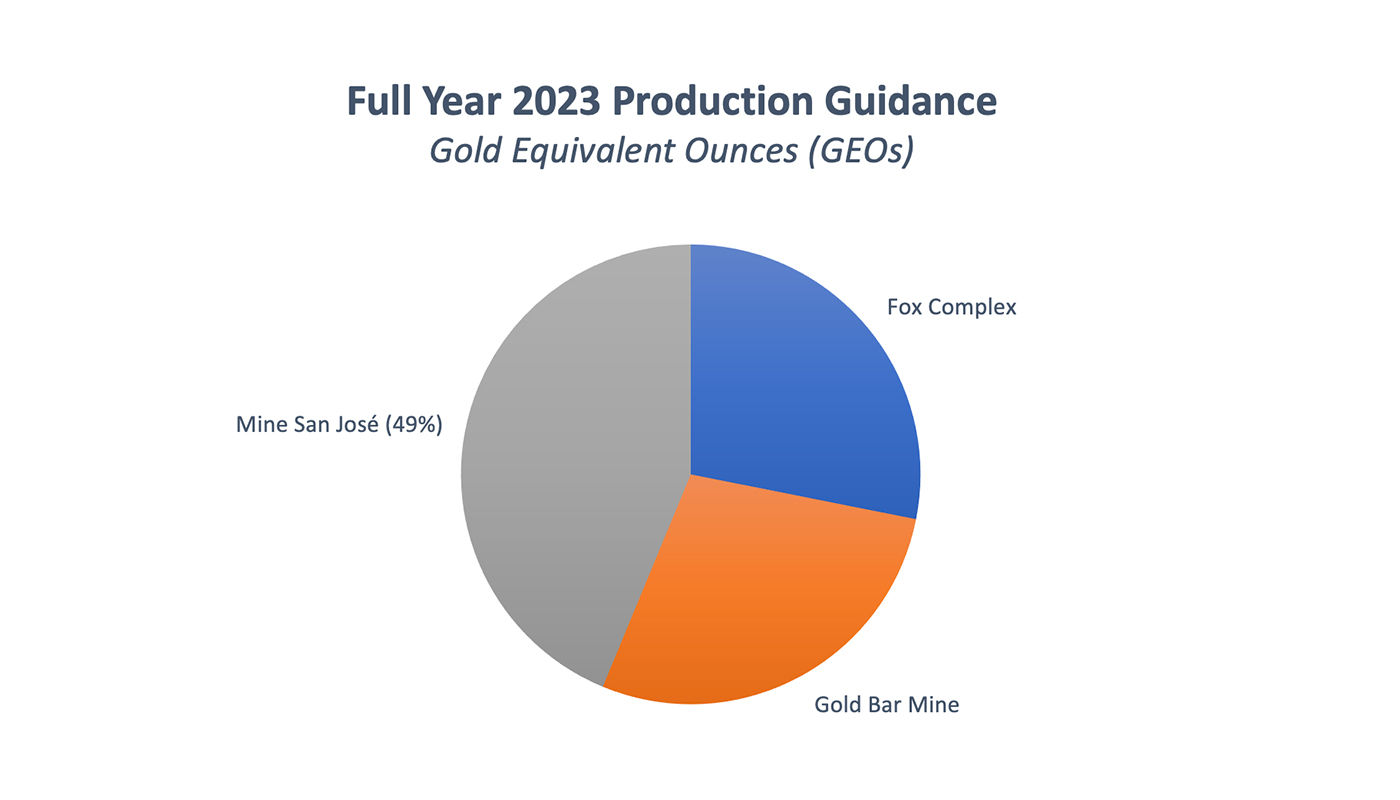
Full year 2023 guidance for The Fox Complex puts production estimates at 45,000 GEOs, 28% of MUX’s total production.
The Gold Bar Mine
McEwen Mining owns a 100% stake in the Gold Bar mine located in an area well known for gold production, the southern Roberts Mountains of the Battle Mountain-Eureka-Cortez gold trend in Eureka County, Central Nevada. The Gold Bar mine is on the same geological structure some 25 miles south of Nevada Gold Mines, a joint venture of Barrick and Newmont. This Cortez-Goldrush complex contains estimated reserves and resources of greater than 50 million gold ounces. Its annual gold production is 1,000,000 ounces.
Gold Bar had been previously mined, between 1991 and 1994, producing 134,000 gold ounces. MUX built a new facility in 2019. The open pit mine was expected to be a large contributor to MUX’s revenue and gold production, however operating challenges arose that reduced gold production and drove cost/oz unacceptably high. Mining activities have shifted recently to a nearby, satellite deposit called Gold Bar South (GBS). Going forward the expectations are higher gold production and lower operating cost/oz as a result of mining a higher ore grade (concentration of gold per ton) and having to move half the amount of material to capture an ounce of gold.
“At Gold Bar, we are looking forward to starting to mine our GBS deposit this quarter,” McEwen said in a November 2022 release. “We are expecting to have a much lower cost/oz than our YTD cost because we will be mining higher grade ore at GBS, with half the strip ratio and no problematic carbonaceous material.”
The Gold Bar Mine will account for approximately 28% of McEwen Mining’s 2023 total attributable production, with guidance pegged at 45,000 GEOs. Most of Gold Bar production in 2023 will be from GBS.
El Gallo/Fenix
Project Fenix is the proposed redevelopment plan for McEwen Mining’s El Gallo Complex in Mexico. There is a long history of mining in this region. MUX’s involvement began in 2013 operating it as an open pit, heap leach mine which produced 281,000 gold equivalent ounces at average cash cost of $655 per ounce. However, due to the transition to deeper sulfide mineralization that is not amenable to heap leaching, mining activities ceased in the second quarter of 2018. The redevelopment envisions constructing a mill at the existing mine site that will initially reprocess the existing heap leach material then transition to open pit mining and processing the sulphide mineralization. The company recently acquired a complete process plant on very advantageous terms that has considerably reduced the projected capital requirements for the project.
CEO Rob McEwen stated in a news release, “This acquisition has made Fenix more attractive to build and could provide a new long life mine for McEwen Mining.”
The initial development approach is to build a mill to reprocess the material on the heap leach pad and produce approximately 17,000 oz of gold annually for eight years. Construction of the Fenix project is expected to be completed by early 2024.
Mine San José
McEwen Mining is a 49% owner and non-operator of the San José gold and silver mine located in Santa Cruz province, Argentina. This high-grade underground mine has been operating since 2007 and currently has an expected life of six years with a reserve grade of 342 gpt silver and 5.7 gpt gold and a resource grade of 427 gpt silver and 7.0 g/t gold.
“The San José mine, where we have a 49% interest, put in a strong quarter and its exploration is continuing to extend its high-grade veins and discover new veins,” McEwen noted in a news release.
Production guidance for 2023 for MUX’s 49% is 70,000 GEOs, 44% of MUX’s total production. As a minority shareholder in the mine, MUX equity accounts for its investment in San Jose, and it receives 49% of the dividends from the mine’s free cash flow.
Market Outlook
Mining stocks took a beating in the wake of the COVID-19 pandemic. However, that could change, as many analysts are now forecasting a gold bull market in 2023.
“The operating challenges we faced in recent years have severely damaged our credibility with our shareholders and the market. As a result, few investors have taken a close look recently at our assets,” Rob McEwen said in a news release. “If they did, I believe some would see the potential value that I see today… I believe there is considerable potential value in MUX, and that is a big reason why I have a personal financial commitment of $220 million in MUX and McEwen Copper.”
Management Team
Robert R. McEwen is Chairman, CEO and Chief Owner of McEwen Mining. He has been associated with the gold industry all his career, with his first 18 years in the investment industry and, since 1990, as CEO of several gold mining companies. He founded Goldcorp and took that company from a $50 million market capitalization to more than $8 billion. He owns 17% of McEwen Mining and is in complete alignment with investors – the cost of his investment in MUX and McEwen Copper is $220 million and he takes an annual salary of only $1. He was awarded the Order of Canada and the Queen Elizabeth’s Diamond Jubilee Award, was inducted into the Mining Hall of Fame, was named an Ernst and Young Entrepreneur of the Year and has Honorary Doctor of Law degrees from York University and Western University.
William Shaver is interim COO and a Director of McEwen Mining. He has decades of management and executive experience in mine design, construction and operations. He was a founder of Dynatec Corporation, which became one of the leading contracting and mine operating groups in North America. In 2013, he was recognized as Ernst and Young Entrepreneur of the Year. Most recently, he served as COO of INV Metals. He is a Professional Engineer with a B.Sc. in Mining Engineering from Queens University.
Perry Ing is interim CFO at McEwen Mining. He has 25 years of experience in the Canadian mining industry. Over the past 15 years, he has held positions as CFO of Mountain Province Diamonds, Kirkland Lake Gold and McEwen Mining. Prior to that, he worked at Barrick Gold and Goldcorp and started his career in the mining practice at PwC. He has a Bachelor of Commerce from the University of Toronto and is a Chartered Professional Accountant in Canada and Certified Professional Accountant in the U.S.
Adrian Blanco S. is the company’s Director – America and Mexico Operations. He has extensive international experience in several industrial sectors and has held executive positions in Mexico, the United States, Peru and Argentina. He joined the McEwen Mining team in 2015 and has led a successful business transformation toward operational discipline, best business practices and financial profitability at subsidiaries Compañia Minera Pangea and McEwen Mining Nevada. He graduated from an Executive Management Program at IPADE and Harvard Business School.
Michael Meding is Vice President and General Manager of McEwen Copper. He has over 20 years of international experience, primarily with major mining companies such as Barrick Gold and Trafigura, including extensive experience with project development and operations in Argentina. While at Barrick Gold’s Veladero mine in Argentina, Mr. Meding played a key role in the turnaround, extension of the mine life and subsequent strategic partnering with Shandong Gold. He holds an MBA from Indiana University in Pennsylvania and an MBA from the Leipzig Graduate School of Management in Germany.
McEwen Mining Inc. (NYSE: MUX), closed Monday's trading session at $7.71, up 7.6816%, on 775,760 volume. The average volume for the last 3 months is 28,883 and the stock's 52-week low/high is $2.81/$9.18899.
Recent News
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) subsidiary McEwen Copper adds $30 Million Boost from Nuton LLC to recent Stellantis investment of $155 Million, Plans for subsidiary IPO
- InvestorNewsBreaks - McEwen Mining Inc.'s (NYSE: MUX) (TSX: MUX) McEwen Copper Inc. Announces Strong Exploration Drilling Results at Los Azules
- McEwen Mining Inc.'s (NYSE: MUX) (TSX: MUX) Commitment to Electrifying Transport Receives Boost Following Subsidiary's Closure of ARS $30 Billion Investment by Automotive Multinational Stellantis
HeartBeam Inc. (NASDAQ: BEAT)
The QualityStocks Daily Newsletter would like to spotlight HeartBeam Inc. (NASDAQ: BEAT) .
To view the full article, visit https://ibn.fm/LvVnj
HeartBeam Inc. (NASDAQ: BEAT) is a cardiac technology company that has developed the first and only 3D-vector 12-lead electrocardiogram (ECG) platform for heart attack detection anytime, anywhere. The company’s proprietary ECG telehealth technology aims to redefine the way high risk cardiovascular patients are diagnosed in ambulatory and acute care settings. HeartBeam’s initial focus is on providing diagnostic data to help physicians with care management of patients with cardiovascular disease.
In August 2022, HeartBeam announced that it submitted its HeartBeam AIMI™ software for approval from the U.S. Food and Drug Administration (FDA). HeartBeam AIMI is a platform technology to improve the speed and accuracy of heart attack detection in acute care settings. The company expects FDA approval by the end of 2022, and a full commercial roll-out of HeartBeam AIMI is targeted for Q1 2023.
HeartBeam sees submission of its first product based on its platform technology as an important milestone toward commercialization, which underscores the company’s continued progress toward making the HeartBeam AIMI platform widely available to help emergency department physicians quickly and accurately identify a heart attack.
While the FDA conducts its regulatory review, HeartBeam will focus on executing key components of its commercialization plan and subscription revenue model. It will also continue to engage in discussions with strategic institutions, including academic centers, regional healthcare systems and regional community hospital systems that can utilize HeartBeam products.
The company is based in Santa Clara, California.
Products
HeartBeam’s development portfolio includes two products:
- HeartBeam AIMI is software that provides a 3D comparison of baseline and symptomatic 12-lead ECG to more accurately identify a heart attack in acute care settings and, as noted above, has been submitted for FDA approval; and
- HeartBeam AIMIGo™, the first and only credit card-sized 12-lead output ECG device coupled with a smartphone app and cloud-based diagnostic software system for remote heart attack detection.
HeartBeam is developing AIMIGo, a medical-grade detection and monitoring technology for use in remote heart attack detection, thereby allowing physicians to diagnose a patient’s heart attack as it occurs, even if the patient is not at a medical facility. The company’s system, once approved by the FDA, can be used by patients at home or almost anywhere and anytime to help their physicians assess whether chest pain is the result of a heart attack or another cause. While approximately 82% of chest pain ED visits are unnecessary, patients delay approximately 3 to 4 hours after symptoms begin, increasing mortality rates by 40%. The company’s goal is to shorten the time to treatment outside of the medical facility to improve patients’ well-being.
HeartBeam’s AIMIGo is a powerful, portable and easy-to-use prescription-based product. It comprises a smartphone app, a credit card-sized ECG device placed on a patient’s chest, the HeartBeam cloud platform, and a digital portal for the physician to view ECG results and direct patient action. For the first time outside of a medical setting, HeartBeam AIMIGo enables patients and their clinicians to determine if symptoms are due to a heart attack, quickly and easily, so care can be expedited, if needed.
Pending FDA clearance, AIMIGo is initially intended to be available by prescription, and is reimbursable under existing remote patient monitoring codes (RPM codes). This provides a new revenue stream to physicians who before did not have a way to monitor these high-risk patients. The RPM codes provide a monthly reoccurring revenue stream to the company, as well. On average, at current reimbursement rates, the practice will receive $1,300+ per year per patient they monitor, and the company will receive $600 per year per patient from this RPM reimbursement.
Market Overview
Adoption rates of telehealth services increased dramatically in recent years, with the COVID-19 pandemic serving as a major driver of growth. Among the areas seeing the greatest expansion are cardiology, radiology, behavioral health and online consultation.
Encouraging this growth, governments are actively developing new policies and reimbursement guidelines to promote the use of digital health platforms. The U.S. Centers for Medicare & Medicaid Services (CMS), for example, has recently expanded reimbursement for telehealth services. U.S. market growth is also being driven by the rising prevalence of chronic conditions and the growing geriatric population.
Remote heart attack detection is a previously unsolved problem with a massive and underserved market that is several times larger than the $2 billion total addressable market (TAM) in the U.S. for ECG cardiac arrhythmia monitoring.
Approximately 8 million Americans have suffered at least one heart attack, and a total of 18 million have been diagnosed with coronary artery disease (CAD). Based on these figures, HeartBeam projects a total addressable U.S. market TAM valued at $10 billion annually for its AIMIGo solution for remote heart attack monitoring of CAD.
Management Team
Branislav Vajdic, Ph.D., Chief Executive Officer and Founder of HeartBeam, Inc, combines over 30 years of experience in technology development and senior management positions. Dr. Vajdic has been deeply involved with the development of HeartBeam’s technology to fit his vision for the company. Prior to HeartBeam, from 2007 to 2010, Dr. Vajdic was CEO and Founder of NewCardio, a publicly traded company in the cardiovascular devices space. From 1984 to 2007, Dr. Vajdic was at Intel, where he held various senior management position. At Intel, Dr. Vajdic was the designer of first Flash memory and two key inventions that enabled Flash as a product and led engineering groups responsible for Pentium 1 through Pentium 4 designs. Dr. Vajdic was awarded two Intel Achievement Awards, the highest level of award for outstanding contributions to Intel. Dr. Vajdic is author of numerous patents and publications in the fields of cardiovascular devices, as well as chip design. Dr. Vajdic holds a Ph.D. in Electrical Engineering from the University of Minnesota.
Jon Hunt, Ph.D., has over 35 years’ experience in the medical/medical device industry with extensive domestic and international experience in general management, clinical/regulatory, sales and marketing. He also has diverse experience in Fortune 500 companies, as well as start-up environments. Dr. Hunt was the Vice President of Clinical Science and Technology, Medical Device Innovation Consortium, from July 2019 to July 2021, and Vice President of Clinical and Regulatory Affairs, Cryterion Medical from January 2018 to June 2019 (acquired by Boston Scientific Corporation in July 2018 for $202M). Dr. Hunt was the Founding President and CEO of Bardy Diagnostics, Inc. from October 2013 to November 2017 (acquired by Hill-Rom Holdings, Inc.). Prior to joining Bardy Diagnostics, Dr. Hunt spent the previous 11 years as the Vice President of Clinical & Regulatory Affairs with Cameron Health, Inc. (acquired by Boston Scientific Corporation). Dr. Hunt spent the previous 10 years with Cardiac Pacemakers, Inc., St. Jude Medical and Cardiac Pathways Corporation. Dr. Hunt began his career with Cardiac Pacemakers, Inc. (now Boston Scientific Corporation) as the Director of Clinical Programs. He subsequently held positions at St. Jude Medical in Clinical Affairs and as the Business Unit Director for the Cardiac Rhythm Management division for Europe, the Middle East and Africa. At Cardiac Pathways Corporation, Dr. Hunt held various executive positions as Vice President of International Sales and Marketing and Vice President of Worldwide Sales and Marketing (acquired by Boston Scientific Corporation). Dr. Hunt received his Ph.D. in Motor Control from The Pennsylvania State University, his Master’s from California State University, Long Beach and his undergraduate degree from Keele University in the United Kingdom.
Rick Brounstein, HeartBeam’s Chief Financial Officer, combines over 30 years of experience in health technology senior management. Since 2017, Mr. Brounstein has been and is currently a partner of Hardesty, LLC, a financial services firm, and Mr. Brounstein is currently a managing director of CTRLCFO, LLC, a firm Mr. Brounstein founded in 2016 to support funded start-ups in life science and technology. From 2008 to 2011, Mr. Brounstein was Chief Financial Officer of NewCardio, Inc., a microcap public company in the cardiology space, and, over his career, he has been with nine other companies in life science or technology, holding positions including Chief Financial Officer, Chief Operating Officer, Treasurer and Accounting Manager. From June 2001 through November 2007, Mr. Brounstein held several positions at Calypte Biomedical Corporation, a publicly traded medical device company, including Chief Financial Officer and Executive Vice President. In January 2007, Mr. Brounstein was appointed as the National Member Representative for the 2007 COSO Monitoring Project, which published new guidelines for monitoring internal financial controls in February 2009; Mr. Brounstein subsequently was a member of the FEI task force that issued the updated COSO Internal Control Framework in 2013. In March 2005, Mr. Brounstein was appointed to the SEC Advisory Committee on Smaller Public Companies. Mr. Brounstein earned his Certified Public Accountant (CPA) certification while working at Arthur Andersen LLP, formerly a public accounting firm. Mr. Brounstein holds a B.A. in accounting and an M.B.A. in finance, both from Michigan State University.
Ken Persen, HeartBeam’s Chief Technology Officer, combines over 28 years of experience in the medical device and digital health industries in engineering and senior management positions. Mr. Persen has been involved in several companies in Cardiac Rhythm Management, holding positions including Chief Executive Officer, Chief Technology Officer, Executive Vice President and Director of Engineering. Since 2016 and prior to joining HeartBeam, Mr. Persen was the Chief Technology Officer at LIVMOR, Inc., a digital health company. In addition, from 2016 through November 2021, he was also Chief Executive Officer of LIVMOR. Prior roles included Director of Engineering at Cameron Health (acquired by Boston Scientific), a late-stage medical device start up, and engineering and management positions at Guidant Corp. (acquired by Boston Scientific), a large medical device manufacturer. He has an undergraduate degree from University of Minnesota, Duluth, with a BA in Computer Science.
HeartBeam Inc. (NASDAQ: BEAT), closed Monday's trading session at $3.15, up 3.2787%, on 28,883 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $9.18899/$.
Recent News
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) On Track to Advance Innovative Products
- InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Makes Changes in Senior Leadership Roles
- HeartBeam Inc. (NASDAQ: BEAT) CEO Spotlighted in Interview on Making a Positive Social Impact
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
To view the full press release, visit https://ibn.fm/IYAUA
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Monday's trading session at $1.33, up 6.4%, on 27,701 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.12/$13.425.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Announces It Has No Exposure to Silicon Valley Bank Issues
- Study Documents Dangers of Halting Opioid Treatments for Chronic Pain
- InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Enrolls First Patient in Switzerland in Potentially Pivotal Berubicin Trial
InMed Pharmaceuticals Inc. (NASDAQ: INM)
The QualityStocks Daily Newsletter would like to spotlight InMed Pharmaceuticals Inc. (NASDAQ: INM).
To view the full study results, visit https://ibn.fm/wjPrK
To view the full press release, visit https://ibn.fm/Ctc8c
InMed Pharmaceuticals Inc. (NASDAQ: INM) is a global leader in the manufacturing and clinical development of rare cannabinoids. InMed is a clinical stage company developing cannabinoid-based pharmaceutical drug candidates, as well as manufacturing technologies for pharmaceutical-grade rare cannabinoids.
The company is dedicated to delivering new therapeutic alternatives to treat conditions with high unmet medical needs. The company is also developing a proprietary manufacturing technology to produce pharmaceutical-grade rare cannabinoids in the lab and has recently announced an LOI to acquire a leading rare cannabinoid manufacturer.
Research and Technology
There are more than 100 rare cannabinoids found in only trace amounts in the cannabis plant, together making up less than 1% of the plant’s biomass. InMed is initially focused on the therapeutic benefits of cannabinol (CBN) in diseases with high unmet medical need. Preclinical studies of CBN demonstrated an excellent safety profile and showed CBN has potential for therapeutic benefit over other cannabinoids such as tetrahydrocannabinol (THC) and cannabidiol (CBD).
Evidence suggests there may be great therapeutic potential in rare cannabinoids. Each has a specific chemical structure, and different cannabinoids have been observed to have distinct physiological properties in humans, including therapeutic potential for specific diseases as well as unique safety profiles. CBN is the active pharmaceutical ingredient (API) in InMed’s two lead programs for dermatological and ocular diseases.
InMed’s most advanced compound, INM-755, is a CBN topical cream under clinical development for the treatment of epidermolysis bullosa, a severe genetic skin disorder. To date, INM-755 has been evaluated in two Phase 1 clinical trials in healthy volunteers. InMed has filed Clinical Trial Applications in several countries as part of a global Phase 2 clinical trial of INM-755 (cannabinol) cream in epidermolysis bullosa. Responses from the National Competent Authorities and Ethics Committees are expected throughout the summer of 2021.
InMed is also involved in developing INM-088, an ocular CBN formulation being researched for the treatment of glaucoma, the second leading cause of blindness in the developed world. InMed is currently evaluating several formulations to deliver CBN into the eye to address issues of dosing frequency, side effects and treatment penetration. INM-088 is being designed for topical delivery to the eye. This localized delivery results in very little drug being absorbed or migrating into the bloodstream, thus minimizing potential adverse side effects. INM-088 shows promise to reduce intraocular pressure and provide neuroprotection of the eye.
Manufacturing
The limited availability of rare cannabinoids like CBN makes them economically impractical to extract directly from the plant for pharmaceutical use. InMed is developing IntegraSyn, a cannabinoid synthesis manufacturing system to create rare cannabinoids in the lab that are bioidentical to the compounds derived from the cannabis plant. IntegraSyn uses multiple standard pharmaceutical processes and has achieved a cannabinoid yield of 5 grams per liter, surpassing commercial viability and significantly exceeding currently reported industry yields. InMed is now focusing on manufacturing scale-up to larger batch sizes while continuing process optimization, targeting increased cannabinoid yield and further reducing overall cost of goods.
BayMedica Inc. Acquisition
On June 29, 2021, InMed announced it had entered into a non-binding letter of intent to acquire BayMedica Inc., a private company based in Nevada and California that specializes in the manufacture and commercialization of rare cannabinoids.
As noted in the news release, BayMedica is a revenue-stage biotechnology company leveraging its significant expertise in synthetic biology and pharmaceutical chemistry to develop efficient, scalable and proprietary manufacturing approaches to produce high quality, regulatory-compliant rare cannabinoids for consumer applications. BayMedica is currently commercializing the rare cannabinoid CBC (cannabichromene) as a B2B supplier to distributors and manufacturers marketing products in the health and wellness sector. BayMedica is planning additional rare cannabinoid launches for the coming year.
Pursuant to the indicative terms of the LOI, InMed and BayMedica intend to negotiate and enter into a definitive agreement under which InMed would acquire 100% of BayMedica in exchange for 1.6 million InMed common shares to be issued to BayMedica’s equity and convertible debt holders, with any such issued InMed common shares being subject to a six-month contractual hold period.
Market Outlook
There is a rapidly growing demand for rare cannabinoids. However, their low natural concentration makes traditional harvesting of these compounds cost prohibitive. Biosynthesis allows production of rare cannabinoids in the lab that are bioidentical to compounds found in nature, with significantly higher yields which reduce costs. Biosynthesis can produce pharmaceutical-grade, bioidentical, THC-free compounds at a cost that’s 70 to 90 percent less than wholesale prices of naturally harvested rare cannabinoids.
Cannabinoid-based pharmaceuticals are expected to overtake the market as rare cannabinoids become less expensive and more available. According to Statista, the value of the consumer market for cannabinoid-based pharmaceuticals in the United States is forecast to grow to $25 billion by 2025 and to $50 billion by 2029, with cannabinoid-based pharmaceuticals used to treat health conditions including pain, respiratory conditions, autoimmune conditions and more.
Management Team
Eric A. Adams has been CEO and president of InMed since June 2016. He has more than 25 years of experience in establishing corporate entities, capital formation, global market development, mergers and acquisitions, licensing and corporate governance. He previously served as CEO at enGene Inc. Prior to enGene, he held senior positions in global market development with QLT Inc. (Vancouver), Advanced Tissue Sciences Inc. (La Jolla, CA), Abbott Laboratories (Chicago, IL) and Fresenius AG (Germany).
Bruce S. Colwill is InMed’s CFO. He has more than 25 years of financial leadership experience in public and private companies. Prior to InMed, he served as CFO of General Fusion Inc., a private clean energy company. He was also CFO at Entrée Resources Inc., a mineral exploration company, from 2011 to 2016. He has held CFO roles at Neuromed Pharmaceuticals Ltd., Response Biomedical Corp, Forbes Medi-Tech Inc. and Euronet Worldwide Inc.
Alexandra D.J. Mancini is Senior Vice President, Clinical and Regulatory Affairs at InMed. She has more than 30 years of global biopharmaceutical research and development experience. She has been an executive with numerous biotech companies, including senior vice president of Clinical and Regulatory Affairs at Sirius Genomics; senior vice president of Clinical and Regulatory Affairs at INEX Pharmaceuticals; and vice president of Regulatory Affairs at QLT Inc.
Eric C. Hsu is Senior Vice President, Pre-Clinical Research and Development at InMed. He joined InMed with more than 18 years of scientific leadership experience in the field of gene therapy. He has held various positions within enGene Inc., including vice president of Research and vice president of Scientific Affairs and Operations. He received his Doctorate from the Department of Medical Biophysics at the University of Toronto.
Michael Woudenberg is Vice President, Chemistry, Manufacturing and Controls at InMed. He has more than 20 years of successful drug development, process engineering, GMP manufacturing and leadership experience. He has held positions with 3M, Cardiome Pharma, Arbutus Biopharma and, most recently, was Managing Director of Phyton Biotech LLC.
InMed Pharmaceuticals Inc. (INM), closed Monday's trading session at $1.13, up 7.619%, on 1,308,192 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.9213/$35.50.
Recent News
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Peer-Reviewed Study Results Published
- Life Sciences Virtual Investor Forum: Presentations Now Available for Online Viewing
- InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) CEO to Present at Two March Investor Events
Tingo Inc. (OTCQB: TMNA)
The QualityStocks Daily Newsletter would like to spotlight Tingo Inc. (TMNA).
Many people are in favor of blockchain games, and this year appearsto be the year that could bring about high-quality online gamesusing the
Tingo Inc. (OTCQB: TMNA) is a digital service agri-fintech technology company focused on foundation-level agriculture and related financial services in Africa. The company aims to be Africa’s leading agri-fintech player, transforming rural farming communities to connect through its proprietary platform to meet their complete needs – from inputs and agronomy to off take and marketplace – and deliver sustainable income in an impactful way. The company’s vision is to build complete digitally inclusive ecosystems that promote financial inclusion and deliver disruptive micro-finance solutions, empower societies, produce social upliftment in rural communities and open international opportunities.
Tingo believes that a truly connected world will help contribute to a better global society. The company’s core focus areas are telecoms, financial services/fintech and agritech. Tingo’s goal is to provide a best-in-class customer experience, support the domestic economies of its host countries and support technological and financial inclusion to end the poverty premium. Through this, Tingo hopes to deliver attractive returns to shareholders while investing in the long-term future of the company and its subsidiaries.
Global climate change is challenging sustainable production and food security. Tingo’s strategy and market execution provide an opportunity for Africa to be a core focal point to solve a number of key areas of concern, including food security, gender equality, financial inclusion and poverty alleviation, to name a few. Disruption of micro finance through the use of DeFi-based stable coins and smart contracts will give agri-communities access to capital markets-driven digital finance solutions that make them more competitive and sustainable economically, striking a good balance of returns between digital asset providers and Tingo as the service partner. This innovation will deliver significant access to much needed finance at ‘Grassroot’ levels, delivering tangible social upliftment and GDP growth in the African markets served by Tingo.
Tingo Mobile, with more than nine million subscribers, is Nigeria’s leading technology and device-as-a-service platform aimed at accelerating digital commerce, especially in the country’s agritech and fintech verticals. The company helps farmers acquire mobile phones through a unique leasing plan, connecting them to mobile and data networks through its own virtual mobile network. Tingo also connects farmers to markets, services and resources via Nwassa, its digital agritech marketplace platform that commenced operations in 2020. The company has also launched a beta version of TingoPay – a B2B and B2C fintech app aimed at providing financial services to users inside and outside of the agriculture value chain. Among the services offered are mobile wallets, payment processing and access to specialist lenders, insurers and pension products.
Tingo will soon announce its innovative blockchain-based solution for use of digital stable coins to empower frictionless trade across borders in Africa. The company’s market-proven model in Nigeria is its core foundation, enabling Tingo to deliver the same service model across Africa to become the continent’s leading agri-fintech business powered through smartphone technology.
The African Continental Free Trade (ACFT) plan will be a key framework to prepare the company to be the leading intra-Africa trading hub for trade flows across Africa in the medium term, when it is likely the agreement will be executed into tangible activity. Tingo is well positioned to easily transform the goals of the ACFT into reality when finally implemented by the African Union and the various African countries that have not signed up.
Tingo posted total revenue of $594 million in 2020, with $212 million EBITDA. As of December 31, 2020, Tingo has 9,344,000 subscribers. The company is confident that these figures will grow through its expansion across Africa and natural progression of business in Nigeria.
Businesses
Tingo has four core businesses:
- Mobile Phone Leasing – Tingo has distributed almost 30 million mobile handsets since 2014 and will continue to replace the devices of its installed customer base every three years. Tingo Mobile provides the latest mobile phone handsets at an affordable price point and allows customers to spread payments over 36 months.
- Mobile Voice and Data Service – Through a mobile virtual network, Tingo provides its customers with voice and data services, allowing customers to communicate effectively, both inside and outside the agricultural ecosystem.
- Nwassa Marketplace Platform – Nwassa is Tingo’s proprietary agritech platform which provides Africa’s farmers with access to global markets to secure more competitive pricing for their crops. The platform processes 500,000 daily transactions with a value of over $8 million. A select group of trusted partners can assist smallholder farmers and agricultural cooperatives with packaging, warehousing, and dry and wet cargo logistics, as well as up-to-date information from the global agricultural sector. Tingo provides its customers with digital wallet services, which enable them to send and receive domestic payments, monitor cash flow in real time and securely hold money. The company also provides access to other services, such as utility bill payment, virtual airtime top-up, insurance services and alternative lending solutions.
- TingoPay – Since the launch of the Nwassa platform, Tingo has been a dominant player in the B2B fintech vertical. After many successful months of operating Nwassa, Tingo entered the fintech B2C vertical to extend its B2B offering to a broader market beyond agriculture.
TingoPay is still in its beta phase and will launch in 2021 with a comprehensive marketing campaign. TingoPay offers the following services:
- Tingo Wallet top-up
- Peer to Peer payments, inclusive of merchant payments at the stores
- Utility payments – airtime, broadband, cable, electricity, water, hotel, flights etc.
- Pension payments
- QR code payment services
Market Opportunity
Africa is the second-largest continent by population. It is also the youngest by far, with a median age of 18 for its 1.3 billion people. Tingo believes the building blocks for growth in Africa’s agriculture industry are in place and that the company is well positioned to participate in the upside. Sub-Saharan Africa’s population is growing at a rate of 2.7 percent per year. At the current growth rate, the continent’s population will double by 2050. Africa’s youthfulness represents a significant opportunity for material growth in demand for agricultural commodities. This younger generation is also being born into a digital world and is comfortable using technology.
Africa’s governments are improving business conditions for entrepreneurs and small businesses. Sub-Saharan Africa’s World Bank Doing Business rank has improved from 45 in 2004 to 65 in 2020. Tingo believes this trend will continue and encourage establishment of more new ventures across all economic sectors, including agriculture.
Africa attracted $407 billion of Foreign Direct Investments (“FDI”) between 2014 and 2018. Investments are increasingly focused on services and industrial sectors. Only 20 percent of investments are in extractive industries – a clear reversal from 2008, when 55 percent of FDI was aimed at resource extraction. Tingo believes FDI into Africa will help resolve significant infrastructure constraints and create value for agribusiness.
Management Team
Dozy Mmobuosi is the CEO of Tingo. He cofounded Tingo Mobile PLC (Nigeria) in 2001 and led the design and launch of Nigeria’s first SMS banking solution, which is still in use in the country today. He also headed a team of more than 120 Chinese and Nigerian engineers in the construction of two mobile phone assembly plants in Nigeria, which have produced and distributed 20 million phones across the country. He has led Tingo’s growth to more than $600 million in revenue annually. He holds a Ph.D. in Rural Advancement from UPM Malaysia.
Dakshesh Patel is the CFO of Tingo. He was formerly CFO of NatWest’s Global Debt and Investment Banking division. He has served as a Director at Gerken Capital Associates, a San Francisco-based alternative asset fund manager. He also led the restructure of Lloyds Banking Group (last financial crisis); managed integration of two leading shipping groups’ global treasury function to create world-leading shipping group Maersk Shipping; built three fintech companies; and exited one to Worldpay. Mr. Patel has strong banking experience, with a focus on Africa. He is a chartered accountant.
Chris Cleverly is president of Tingo. He has served as CEO of the Made in Africa Foundation, and as CEO of blockchain payments gateway startup Kamari. He has been a board member of several companies, both public and private, in the UK, India, China and Africa. He has advised multiple UK companies on their entrance into African markets, and regularly advises the UK Government on development issues and African governments on investment issues.
Clarence Simms is the Chief Technology Officer at Tingo. He has 25 years of IT and IT management experience. He has worked in IT Shared Services Technical Operations and IT Program Management for Huawei Technologies and MTN. As an entrepreneur, he created Africaprepay.com, a service that allows African Diaspora travelers to send airtime, pay bills, send mobile money and transfer money to a bank account from anyplace in the world.
Rory Bowen is the Chief of Staff at Tingo. Mr. Bowen started his career in traditional capital and derivatives markets working for Moneycorp and Tradition UK in European and emerging markets across FX, interest rate derivative and government bond markets. He has also spent time with one of Europe’s fastest growing fintech’s banking circles. Before joining Tingo, he was Chief of Staff at FinTech Alliance, an organization established in partnership with the UK Government Department for International Trade to foster innovation, growth and foreign direct investment (FDI) in the financial services sector and facilitate greater public/private cooperation.
Tingo Inc. (OTCQB: TMNA), closed Monday's trading session at $0.268, up 0.374532%, on 55,914 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.01/$4.00.
Recent News
- Tingo Inc. (OTC: TMNA) - 2023 Is Year of High-Quality Blockchain Games - Polygon
- Crypto Loudly Absent at This Year's Super Bowl
- Five Industries Making Use of Blockchain Tech
Prime Harvest Inc.
The QualityStocks Daily Newsletter would like to spotlight Prime Harvest Inc.
To view the full article, visit https://ibn.fm/4YFF5
Prime Harvest Inc., based in San Diego, California, is a technology-focused, full-service cannabis company with horizontally diversified operations spanning various segments of the cannabis value chain, from licensing acquisition and compliance management to direct-to-consumer operations. The company is leveraging a long-term strategy of investing in the growth and scale of licensed assets anchored by the power of data-driven technology to expand its footprint throughout California.
Sustainability is key to Prime Harvest’s corporate vision. The company aims to ensure that the communities it serves capture their fair share of the fruits of the industry’s growth, including financial profit, employment opportunities, environmental enrichment and impactful innovation through R&D and education.
The company’s mission is to appeal to the ethos of the cannabis consumer by setting a new operational standard emphasizing accountability, sustainability and community. With this commitment, Prime Harvest continues to work toward positively affecting millions of lives through the creation of a world-class platform that caters to strengthening the commercial cannabis pipeline.
Jaxx Cannabis
Jaxx Cannabis is the flagship brand in Prime Harvest’s portfolio. Through Jaxx Cannabis, the company aims to use technology to facilitate a true customer-centric culture while enhancing the overall craft cannabis experience. Jaxx features an expertly curated selection of premium products from some of the most respected brands in the thriving California market.
Key values serving as the foundation of Jaxx Cannabis include:
- Creating and nurturing a welcoming culture for all
- Unlocking the true potential of customer value
- Being innovative in uncovering new ways to grow both the company and the industry
- Meeting the wants and needs of consumers to promote profitability
- Remaining accountable for the results of its operations
It is these values that differentiate Prime Harvest and Jaxx Cannabis in the California cannabis sector.
Brand Partnerships
Prime Harvest works diligently to establish strong alliances with complementary brands that are in alignment with its culture and values. Through a combination of deliberate foresight and strategic action, the company seeks to grow existing cannabis brands and continuously discover new, high-potential performers that are primed for long-term success.
These partnerships enhance Prime Harvest’s efforts to transform the world’s cannabis access and bring its consumers high-quality products that are fair for both people and the planet.
Responsibility
Prime Harvest remains committed to the goal of creating a more sustainable environment, now and in the future. Concern for human beings and the environment can be observed in every facet of its operations, including its ongoing R&D activities dedicated to exploring methods of reducing and repurposing waste into composite materials and exploring the potential of the hemp plant for industrial and wellness contributions.
The company is a proud member of the Community Alliance Program, a foundation that seeks to make a difference in local communities by providing financial assistance for educational programs, housing homeless veterans, creating urban farms, and holding local arts initiatives for children and adults. The program also helps explore the natural healing attributes of medical cannabis through research, development, clinical trials, and advocating for the safe access of cannabis to those in need.
Market Overview
Ongoing changes in U.S. state government policies toward cannabis are expected to cause demand for legal marijuana to surge. In addition, the number of indications for which medical marijuana is prescribed continues to increase. These factors are expected to rapidly boost legal sales of cannabis products.
Legal sales across the U.S. hit a record of $17.5 billion in 2020, marking an increase of 46% over 2019, according to Forbes. This strong growth is expected to continue. According to a Grand View Research report, the global legal marijuana market is forecast to grow at a CAGR of 26.7 percent from 2021 to 2028.
California – Prime Harvest’s home state – has consistently led the pack in terms of U.S. cannabis sales. The Motley Fool pegged cannabis spending in the Golden State at $3.8 billion in 2020, more than doubling the second state on its list.
Leadership Team
The Prime Harvest team is composed of true experts in their respective fields focused on building a world-class organization capable of driving the cannabis industry and movement forward.
E. Duane Alexander is the company’s Founder and CEO. He brings to the team more than 25 years of real-world, hands-on cannabis retail, marketing and commercial operations experience. Mr. Alexander has championed 40+ cannabis license applications throughout the western U.S. to date.
John Wilczak is the COO of Prime Harvest. He has 30+ years of executive management, strategy development & configuration experience with GE, pharmaceutical and agriculture companies. Mr. Wilczak is a Brown & Columbia MBA with vast knowledge of technology driven intellectual properties.
Andrea Jenson is the Chief Financial Officer of Prime Harvest. As CFO, she is responsible for all the company’s financial functions, including accounting, corporate finance and investor relations. Her career spans more than 20 years of varied experience in financial management, business leadership and financial strategy.
John Kazanjian is the VP of Business Development of Prime Harvest. He has worked over 40 years in business operations, brand marketing, sales and investor/lender communications. Mr. Kazanjian earned his B.S. from Rutgers University and his MBA from Harvard University.
Johann Balbuena is the Chief Marketing Officer of Prime Harvest. She has more than six years of experience in California cannabis licensing acquisition and compliance management. Ms. Balbuena has led multimedia production and content marketing efforts for the likes of the Social Club TV app, The Emerald Cup, High Times, Weedmaps and Synergy.
Recent News
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Secures Reg A+ Qualification, Eyes New Heights of Opportunity
- Prime Harvest Inc. Reveals That Their Reg A+ Equity Offering Has Been Granted Approval to Go Ahead
- InvestorNewsBreaks – Prime Harvest Inc. Delivering Excellence in Legal Access to Cannabis, Best-in-Class Experiences

BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
The QualityStocks Daily Newsletter would like to spotlight BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV).
•BiondVax is a biopharmaceutical company focused on developing,manufacturing, and commercializing innovative immunotherapeuticproducts, primarily for the treatment of infectious and autoimmunediseases
•The company is led by CEO Amir Reichman, with over 20 years ofinternational experience as a biopharma expert
•Amir took company reins in 2021 and pivoted the mission and visionincluding diversifying risks
•With new focus and opportunity, BiondVax now developing aninnovative alpaca-derived nanosized antibody (“NanoAb”) pipeline
•BiondVax is aggressively advancing new NanoAb therapeutic andprophylactic technologies owing to exclusive collaborations,previous pharma development experience and a state-of-the-art GMPmanufacturing facility
Leveraging top-tier pharmaceutical industry leadership, extensivedrug development expertise, a GMP biologics manufacturing facility,and strategic collaborations with Max Planck Institute forMultidisciplinary Sciences (“MPI”) and the University MedicalCenter Göttingen (“UMG”),
Asthma is a respiratory disease caused by chronic inflammation inthe lung airways and is characterized by shortness of breath, chesttightness, wheezing and coughing. It affects an estimated 8.3% ofAmericans (26.5 million) with
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) is a biopharmaceutical company focused on developing, manufacturing and commercializing innovative products for the prevention and treatment of infectious diseases and other illnesses.
In collaboration with the prestigious Max Planck Institute for Multidisciplinary Sciences (MPG) and the University Medical Center Göttingen (UMG), both in Germany, BiondVax is developing a pipeline of innovative nanosized antibody (NanoAb) therapies addressing diseases underserved by current treatments and with large and growing markets, such as COVID-19, asthma and psoriasis.
NanoAbs, also known as VHH-antibodies or Nanobodies, are alpaca-derived nanosized antibodies that exhibit multiple significant competitive advantages over existing antibody therapies, including stability at high temperatures, superior binding affinity, more effective and convenient routes of administration and efficient production. BiondVax is uniquely positioned to advance nanosized antibody innovation from R&D through commercialization.
The company’s highly experienced and successful pharmaceutical industry leadership team includes former senior executives from Novartis, GSK and Bristol-Myers Squibb.
Since its founding, BiondVax has executed eight clinical trials, including a seven-country, 12,400-participant Phase 3 trial of a prior influenza vaccine candidate, and it built, owns and operates a 20,000 sq. ft. state-of-the-art GMP biologics manufacturing facility housing its laboratories, production facilities and offices.
Lead Candidate: Inhaled COVID-19 NanoAb
In December 2021, BiondVax signed definitive agreements with the Max Planck Society – parent organization of the Max Planck Institute for Multidisciplinary Sciences– and the UMG to enter a strategic collaboration for the development and commercialization of innovative COVID-19 NanoAbs.
The company is planning a rapid development path that leverages its expertise and capabilities in biological drug development and manufacturing. BiondVax anticipates preclinical proof-of-concept results for an inhaled COVID-19 NanoAb by the end of 2022, with initial Phase 1/2a human clinical trial results expected in 2023.
The intended inhaled mechanism of delivery of BiondVax’s COVID-19 NanoAb formulation may serve as a significant differentiator when compared to approved monoclonal antibodies, which are injected. Inhaled delivery has shown to be cheaper, more convenient and likely safer for patients and providers.
NanoAb Pipeline: Psoriasis, Asthma and More
The COVID-19 NanoAb development agreement is part of a broader five-year research collaboration agreement signed in March 2022 covering discovery, development and commercialization of NanoAbs for several other disease indications with large market medical needs, including asthma, psoriasis, macular degeneration and psoriatic arthritis.
BiondVax has an exclusive worldwide license for development and commercialization of COVID-19 NanoAbs and exclusive options for similar worldwide licenses for NanoAbs for the above mentioned additional large market disorders currently underserved by approved therapeutic antibodies.
Academic research teams from MPG and UMG have verified strong affinity by the new NanoAbs to their biological target molecules and high thermostability. They have also demonstrated strong neutralization by several NanoAb candidates of their respective target molecules. Neutralization studies of the other NanoAbs are expected to begin later in 2022.
Based on the promising results, BiondVax will focus development efforts beginning with the following NanoAbs:
- NanoAbs targeting IL-17 as drug candidates for the potential treatment of psoriasis and psoriatic arthritis
- NanoAbs targeting IL-13 and NanoAbs targeting TSLP as drug candidates for the potential treatment of asthma
These are conditions for which the antibody target is validated by existing treatments and the mechanism of action is well understood. Both represent large medical needs and growing markets. BiondVax anticipates preclinical proof-of-concept for at least one of these NanoAbs in 2023. This is in addition to the aforementioned human clinical Phase 1/2a for the inhaled COVID-19 NanoAb therapy, which is also anticipated in 2023.
Market Opportunity
COVID-19 treatment, target of the company’s lead NanoAb therapy candidate, had an estimated market size of $22 billion in 2021.
Future BiondVax drug candidates will target conditions with large markets growing at attractive CAGRs.
The global asthma treatment market was valued at $18.08 billion in 2019 and is projected to reach $26.01 billion by 2027, exhibiting a CAGR of 4.5% during the forecast period, according to Fortune Business Insights. The research firm predicts that the global psoriasis treatment market will grow from $26.37 billion in 2022 to $47.24 billion by 2029, exhibiting a CAGR of 8.7% over the forecast period.
Management Team
Amir Reichman is BiondVax’s CEO. He previously was Head of Global Vaccines Engineering Core Technologies at GSK Vaccines in Belgium. Prior to that, he held leadership roles at Novartis Vaccines’ Global Vaccines Supply Chain Management organization. He was the first employee of NeuroDerm Ltd., a company focused on transdermal drug delivery, and served as Chief Engineer and Senior Scientist until his departure in 2009. He earned a M.Sc. in Biotechnology Engineering from Ben-Gurion University and an MBA in Finance and Health Care Management from the University of Pennsylvania’s Wharton School.
Tamar Ben-Yedidia, Ph.D., is Chief Science Officer at BiondVax. She has more than 30 years of experience in immunology, with specific expertise in the development of vaccines. She began her career with Biotechnology General Ltd., working on development of a recombinant Hepatitis-B vaccine. She later joined the Weizmann Institute of Science, working on the design of a peptide-based vaccine against several pathogens. She is widely published, with numerous refereed articles and invited reviews in various scientific journals. She received her Ph.D. from the Weizmann Institute.
Elad Mark is COO at BiondVax. He has over 15 years of biotechnology industry experience encompassing diverse project stages including feasibility studies, conceptual and detailed design, commissioning, qualification and process validation. Prior to joining BiondVax, he led Novartis’s $800 million investment in a biologics facility in Singapore. With Biopharmax and Antero, both global pharmaceutical engineering companies, he successfully led projects in Israel, China and Singapore. He holds a BSc. in Engineering from the Afeka Tel Aviv Academic College of Engineering and an MBA from the Open University of Israel.
Uri Ben-Or is CFO at BiondVax. He has served as CFO with public life science companies traded on the TASE, OTC and Nasdaq. Ben-Or provides his services to BiondVax through CFO Direct, a company he founded and for which he serves as CEO. He served as the VP of Finance of Glycominds, a leading biotechnology company, and as CFO of a spin-off from Telrad Networks. He also served as a Corporate Controller at Menorah Capital Markets and as an Auditor at PWC. He holds a B.A. in Business from the College of Administration, an MBA from Bar-Ilan University, and is a CPA.
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV), closed Monday's trading session at $2.1, off by 2.2346%, on 50,975 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.08/$22.90.
Recent News
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Study Shows Air Pollution in Urban Areas Worsens Lung Function in Kids
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Leverages Top-Tier Leadership and GMP Facility to Advance Rapid Development of New Therapeutic and Prophylactic Technologies for Multiple Diseases and Disorders
- Bionxt Solutions Announces Non-Brokered Private Placement
Silo Pharma Inc. (OTCQB: SILO)
The QualityStocks Daily Newsletter would like to spotlight Silo Pharma Inc. (OTCQB: SILO).
•A recent medical study found that psychedelic drugs showed promiseas a treatment alternative for hard-to-treat depression
•Psychedelics were found to activate a greater number of serotonin2A receptors then traditional antidepressant drugs, largely becauseof their ability to penetrated through cells’ fatty outer membranes
•The study reinforces the potential use of psychedelic drugs, andpsilocybin in particular, as a treatment methodology for a varietyof mental health related conditions
•Silo Pharma, a developmental stage biopharmaceutical company haslong focused around merging traditional therapeutics withpsychedelic research
In a study carried out between 2013 to 2016, researchers found that8.1% of American adults aged 20 and over suffered from depressionduring any given 2-week period, with women nearly twice as likelythan men to have had acute depression (https://ibn.fm/ygq4x). Whilst depression has long been treated with a combination ofself-help, talking therapies and medicines, studies have previouslyfound anti-depressant drug therapy to not be as effective astraditionally assumed. As a result, a recent study which found thatpsychedelic drugs could show promise as a therapeutic alternativein treating drug-resistant depression was greeted with someoptimism within the sector (https://ibn.fm/cXz8A). Alternative treatments for depression have emerged thatattempted to help the brain create new connections more efficiently– most notably ketamine and psychedelic therapy; initial findingshave found these alternative treatments to be about as effective asantidepressants, improving depression in roughly 60 percent of thepeople who try them. It is precisely this issue which
Silo Pharma Inc. (OTCQB: SILO), a developmental stage biopharmaceutical company, is focused on merging traditional therapeutics with psychedelic research for people suffering from indications such as post-traumatic stress disorder (PTSD), fibromyalgia, Alzheimer’s disease, Parkinson’s disease, and other rare neurological disorders. Silo’s mission is to identify assets to license and fund research that the company believes will be transformative to the wellbeing of patients and the health care industry.
Silo is committed to developing innovative solutions to address a variety of underserved conditions. Combining Silo’s resources with world-class medical research partners, the company looks to make significant advances in the medical and psychedelic space.
Silo works to identify and partner with leading medical universities, providing the needed financial resources to develop safe therapeutic treatments while moving cutting-edge research through the clinical stage and into commercialization. The company is well-capitalized with access to additional funds as opportunities present themselves.
Silo recently engaged Donohoe Advisory Associates LLC for consulting and advisory services in connection with the potential uplisting of Silo’s common shares to the Nasdaq Stock Market.
Research
Silo has entered into research agreements and partnerships with multiple leading medical universities.
The company is involved in a sponsored study with Maastricht University utilizing repeated low doses of ketamine and psilocybin to examine the effects on cognitive and emotional dysfunctions in Parkinson’s disease and to understand its mechanism of action. The investigator in the Netherlands is acquiring the substances for the study and will then finalize the documentation to submit to the ethics committee.
Additionally, in June 2021, Silo announced its entry into a scientific research agreement with the University of California San Francisco (UCSF). The agreement will leverage four other clinical trials being planned by the university to determine the effects of psilocybin on inflammation. The study will take place at The Translational Psychedelic Research (TrPR) Program at UCSF.
Silo also recently extended its exclusive option agreement with the University of Maryland, Baltimore (UMB) to explore a novel invention generally known as joint-homing peptides. These peptides are being developed for use in the investigation and treatment of arthritogenic processes and can be used for enhanced targeting of therapeutic agents.
This agreement includes the study of two separate peptides. The first is an option and study for the treatment of arthritis. The second is a patented licensed peptide for the central nervous system, with an initial study for MS autoimmune diseases, in addition to rheumatoid arthritis. Animal studies are underway for both initial indications relating to the UMB agreement, with the potential for studies evaluating additional indications in the future.
Finally, Silo signed an agreement with Columbia University granting it an option to license certain assets currently under development, including an Alzheimer’s disease formulation targeting NDMARs and 5-HT4Rs, as well as a prophylactic treatment for stress-induced disorders and PTSD. Both candidates are currently being tested in mice and have already provided early data.
In addition to its university partnerships, Silo entered a joint venture agreement with Zylo Therapeutics Inc. (“ZTI”) focused on the development of ketamine and psilocybin using ZTI’s Z-Pod™ technology for the transdermal time released delivery of therapeutics. In November 2021, the company announced ZTI’s reception of its first ketamine shipment and initiation of loading ketamine into its Z-Pod technology. In a news release, Eric Weisblum, CEO of Silo, called the development an “important milestone” that will help the company “study the benefits of slow-release transdermal release of Ketamine.”
Market Overview
According to Coherent Market Insights, the fibromyalgia treatment market was valued at $2.78 billion in 2018 and has a projected CAGR of 3.3% over the forecast period 2018 to 2026. Fibromyalgia is a condition that causes pain all over the body, sleep problems, fatigue, and emotional and mental distress.
The global PTSD therapeutics market is expected to reach $10.68 billion by 2026 with a CAGR of 4.5% during the forecast period from 2018 to 2026, according to a report by Credence Research. Growing prevalence of PTSD is the chief factor driving the global treatment market. Increases in events such as wars, combat, and interpersonal violence has been a major contributing factor. Other factors like growing emphasis on rehabilitation initiatives by governments for treating their war veterans has also been facilitating the increase in demand for PTSD therapeutics.
Fortune Business Insights reports the global Parkinson’s disease treatment market is predicted to grow to $8.38 billion by 2026, with a CAGR of 8.1% during the forecast period. Parkinson’s is a neurodegenerative disease of the central nervous system which primarily affects the brain, causing uncontrollable shaking and tremors, difficulties in balance and restricted body movement making it difficult for the person to function or perform a daily routine.
Management Team
Eric Weisblum is CEO and founder of Silo Pharma. He has over 25 years of Wall Street experience, most recently in the biotechnology sector. He has served on the board of Aikido Pharma and was the president of Sableridge Capital. He has a proven track record in licensing therapeutic assets and assisting in their development. He brings to the company nearly 20 years of expertise in structuring and trading financial instruments. He holds a bachelor’s degree from the University of Hartford’s Barney School of Business.
Dr. Kevin Muñoz was appointed to the Silo board of directors in October 2020. He teaches biomedical sciences and medical intervention for the Passaic County Technical Institute. He previously served as Director of Operations at Physical Medicine and Rehabilitation. He began his career with Harlem Health Promotion Center in New York City as a research assistant. He earned a bachelor’s degree from the University of Michigan and a Doctor of Medicine from Xavier University School of Medicine.
Josh Woolley, M.D., Ph.D., is a Scientific Advisor for Silo. He is an associate professor in the Department of Psychiatry and Behavioral Sciences at the University of California, San Francisco. He is also a psychiatrist on staff at the San Francisco Veterans Affairs Medical Center. He is the director and founder of the Bonding and Attunement in Neuropsychiatric Disorders Laboratory. He received both his M.D. and his Ph.D. in Neuroscience from UCSF, where he completed his psychiatry residency training.
Charles Nemeroff, M.D., Ph.D., is a Scientific Advisor for Silo Pharma. He directs the Institute for Early Life Adversity Research within the Department of Psychiatry and Behavioral Sciences as part of the Mulva Clinic for the Neurosciences. He was chair of the Department of Psychiatry and Behavioral Sciences and clinical director of the Center on Aging at the University of Miami Miller School of Medicine. He received his M.D. and Ph.D. in neurobiology from the University of North Carolina School of Medicine.
Silo Pharma Inc. (OTCQB: SILO), closed Monday's trading session at $2.07, off by 5.3368%, on 17,438 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.05/$12.445.
Recent News
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Converts over $1 Million in Debt to Equity This Week with Another $119k Conversion; Update on Annual Filings Delay
- Silo Pharma Inc. (NASDAQ: SILO) Granted US Patent For SPC-15 to Prevent and Treat Stress-Induced Disorders
- Silo Wellness Enters LOI with Largest Creditor, Exchanging $1.06M Debt for Interest-Free Note Convertible at 36% Premium
CISO Global, Inc. (NASDAQ: CISO)
The QualityStocks Daily Newsletter would like to spotlight CISO Global, Inc. (NASDAQ: CISO).
To view the full press release, visit https://ibn.fm/Kw6b4
CISO Global, Inc. (NASDAQ: CISO) is an industry leader in cybersecurity and compliance services. The company leverages an integrated approach to reduce noise and bridge common silos that often limit the effectiveness of cybersecurity programs. Pulling disparate technologies, teams, and vendors together, CISO helps its clients enjoy a simpler and more successful journey to cyber resilience. Since 2019, CISO Global has worked to rapidly expand by acquiring world-class cybersecurity and compliance businesses with top-tier talent who utilize the latest technology to create innovative protection solutions.
The CISO Global workforce is comprised of cybersecurity experts spanning not only global geographies, but also specialties, industries, regulatory frameworks and focus areas. Its team includes audit and compliance specialists, certified forensics experts, ethical hackers, IEEE® certified biometric professionals, security engineers, around-the-clock analysts, and more – all backed by the most respected credentials in the industry. On an ongoing basis, the company works to identify cyber talent that is culturally aligned and that offers operating leverage through both existing customer revenue and relationships.
CISO Global has invested in enterprise solutions and executive talent to integrate its different organizations into an ecosystem that works together to provide complete cybersecurity through cross-pollination of solutions that begin at the network level and extend through technologies, people, policy, and practices. This ecosystem is intended to foster additional growth opportunities and drive overall recurring revenue. Once engaged, the company strives to become trusted advisors for customers’ cybersecurity and compliance demands by providing tailored security solutions based upon their organizational needs.
While cyber resilience requires cycles of continuous improvement, it is a journey that few in the current business and security climate seem to understand. With its deep bench of seasoned experts, CISO Global works to simplify that journey for its growing customer base, straightening out the curves and speeding up the process to resilience along the way.
Cybersecurity is a Culture, Not a Product
Integrating compliance and security, including principles of security by design, CISO Global helps its clients create an organization-wide culture of cybersecurity. Its offerings include audit and compliance, security operations center services, security engineering, virtual Chief Information Security Officer services, incident response, certified forensics, technical assessments and cybersecurity training.
In contrast to the majority of cybersecurity firms that specialize in a specific technology or service, CISO Global seeks to differentiate itself by remaining technology agnostic, focusing on accumulating highly sought-after subject matter experts. CISO Global believes that bringing together a world-class team of technological experts with multi-faceted proficiency in the critical aspects of cybersecurity is key to providing technology agnostic solutions to its clients in a business ecosystem that suffers from a chronic lack of highly skilled professionals.
CISO Global’s goal is to create a culture of security and to help quantify, define and capture a return on investment from information technology and cybersecurity spending. Its end-to-end, holistic process covers every aspect of clients’ cybersecurity and compliance requirements in an effort to promote greater efficiency and strengthen awareness about the integral role of internal team members in the cybersecurity culture of an organization.
As a result of this strategy, CISO Global customers receive an efficient engagement from a single partner that covers a wide range of their needs – addressing challenges more thoroughly and resolving problems more rapidly when compared to working with a host of vendors.

Market Outlook
According to an analysis by the firm Research and Markets, the global managed security services market was valued at $22.45 billion in 2020 and is projected to reach $77.01 billion by 2030, growing at a CAGR of 12.8% through the forecast period.
An expected increase in cybercrime, cost effectiveness of provided solutions and stringent mandatory government regulations aimed at protecting corporate data will drive the global managed security services market for the foreseeable future.
In addition, the documented and growing use of mobile devices in the workplace and the rise in captured and stored digital data serve to fuel market growth. Moreover, growing awareness about the critical nature of data security, the growing importance of e-business and demand for customized services is expected to offer ample opportunities for expansion of the market during the forecast period.
Management Team
David Jemmett is CEO and founder of CISO Global. He has more than 35 years of executive management and technology experience with telecommunications, managed services, and cybersecurity consulting services. He previously held positions as CEO of GenResults, a leading provider of security consulting services and technology solutions, and as CTO and founder at ClearData Networks, a HIPAA-compliant HealthDATA cloud hosting platform.
Dave Bennett is COO at CISO Global. Since 2015, he has served on the President’s STEM Advisory Board of Grand Canyon University. Before joining CISO Global, he served as Chief Product Officer at Experian Health and as Senior Vice President, Product for Gainwell Technologies. He has also held positions as Vice President and Worldwide Head of Build, Healthcare and Life Sciences at DXC Technology, and as EVP, Product and Strategy at Orion Health.
Ashley Devoto is President and Chief Information Security Officer at CISO Global. Over the past 17 years, Devoto has worked with the cybersecurity elite to design, build, and operate world-class cybersecurity programs for large, diverse organizations in both government and commercial enterprises. Prior to joining CISO, Devoto served as CISO for Booz Allen Hamilton, as business information security officer (BISO) at Bank of America, and as a cyberspace operations officer in the United States Air Force.
Deb Smith is CFO at CISO Global. Prior to assuming that position, she was the company’s EVP, Finance and Accounting. She has also served as SVP, Global Accounting at International Cruise and Excursions Inc., and as Chief Accounting Officer for BeyondTrust, an information security software company. She has also held the positions of Corporate Controller at Aspect Software and Assistant Controller at JDA Software.
CISO Global, Inc. (NASDAQ: CISO), closed Monday's trading session at $2.05, up 3.0151%, on 73,251 volume with 375 trades. The average volume for the last 3 months is 57,207 and the stock's 52-week low/high is $1.04999995/$5.63000011.
Recent News
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Announces Participation in Upcoming Roth Conference
- InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Accelerating Clients' Journey to Cyber Resilience
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Featured in Coverage of the 35th Annual Roth Conference
India Globalization Capital Inc. (NYSE American: IGC)
The QualityStocks Daily Newsletter would like to spotlight India Globalization Capital Inc. (NYSE American: IGC).
Missouri has expunged the records of more than 7,500 people who hadprior cannabis-related convictions. The state
India Globalization Capital Inc. (NYSE American: IGC), through subsidiary IGC Pharma, develops, patents, and markets advanced THC-based drug formulations for the treatment of symptoms related to various diseases including but not limited to Alzheimer’s disease, Tourette syndrome, chronic pain, and pet seizures.
IGC’s leading drug candidate, IGC-AD1, has completed Phase 1 of a safety and tolerability trial and entered Phase 2 trials for treating agitation in patients with Alzheimer’s dementia, the first study in humans of a natural tetrahydrocannabinol (THC) compound plus another molecule (www.clinicaltrials.gov). As of September 2022, the IGC trial is the only ongoing Phase 2 trial of a natural THC-based formulation on Alzheimer’s patients.
The company’s other drug candidate, TGR-63, is an enzyme inhibitor that has shown in preclinical trials the potential to reduce neurotoxicity in Alzheimer’s cell lines. Both drug candidates have shown their ability to ameliorate beta amyloid plaques in Alzheimer’s cell lines and improve memory in Alzheimer’s mouse models. Beta amyloid plaques are a key hallmark of Alzheimer’s and an important target of Alzheimer’s pharmaceutical drug development.
Neuro Psychiatric Symptoms (NPS) are not only debilitating for Alzheimer’s patients; they also place an immense emotional burden on their caregivers. Beyond reducing symptoms, IGC-AD1’s active molecules and TGR-63 have also shown promise in preclinical trials to reduce important hallmarks of Alzheimer’s including plaques and tangles, as well as improving the treatment of memory loss.
Over the past eight years, the IGC team has amassed a deep knowledge of cannabinoid science, including extraction, isolation, purification, and development. The company’s strategy is to leverage its unique end-to-end capabilities, platform, and expertise to develop a class-leading program and bring it to market quickly and cost efficiently to treat neurodegenerative diseases such as Alzheimer’s.
The company also has a family of cannabidiol (CBD)-based consumer products (www.Holief.com) such as pain relief creams, pain relief gels, purpose gummies, tinctures, and capsules targeting women’s wellness, with a particular focus on premenstrual syndrome (PMS) and dysmenorrhea (period cramps). In addition, the company targets individuals that need sleep-aids with its specially formulated low melatonin cannabinoid gummies.
IGC has also introduced a low-calorie CBD- and caffeine-infused energy beverage brand (www.SundaySeltzer.com) that is currently available for purchase. The company’s brands are founded on the belief that effective natural solutions should be affordable and accessible to everyone. As the demand for natural products targeting women’s wellness and energy drinks continue to grow, these products are seeing strong traction in the market.
The company operates three facilities – a large GMP (Good Manufacturing Production Standards) certified facility that includes extraction, distillation, and manufacturing, in Washington State; a GMP-211 (pharmaceutical) grade facility in Maryland; and a facility licensed for controlled substances including cannabis in Bogota, Colombia, with complete access to legal licensed cannabis where the company conducts its testing.
In addition, the company’s development under Magistral Formulations is approved by INVIMA (Colombia National Food and Drug Surveillance Institute) to treat neurological disorders, non-oncological chronic pain, and mental disorders.
IGC’s intellectual property (IP) portfolio comprises of eight patents that it controls and seven patent applications. The portfolio includes #11,446,276, a patent for extreme low dose THC treatment of Alzheimer’s that was granted in September 2022.
The company is headquartered in Potomac, Maryland.
IGC-AD1
IGC-AD1 is the company’s leading drug candidate for the treatment and relief of Alzheimer’s symptoms. A significant amount of research on Alzheimer’s cell lines has shown that the active agents in IGC-AD1 reduce plaques and neurofibrillary tangles that are the hallmarks of Alzheimer’s. Further, micro-dosing of THC, as shown in cell lines, could increase the functioning of mitochondria and potentially promote the growth of new neural pathways (neurogenesis). The research shows that micro-dosing of THC affects the brain radically differently from the normal higher dosing of THC.
While there is a significant body of research showing that THC is neuro-toxic at normal levels of dosing, micro-dosing of THC has been shown to be non-toxic to neurons. With the results of these preclinical studies, the company developed an oral formulation, IGC-AD1. The company recently completed a safety and tolerability Phase 1 trial on Alzheimer’s patients and has initiated a Phase 2, multi-site, double-blind, randomized, placebo-controlled trial of the safety and efficacy of IGC-AD1 on agitation in participants with dementia due to Alzheimer’s disease at sites in the U.S. and Canada. IGC expects the Phase 2 trial to take between 9 and 12 months to complete, barring unknown factors such as, for example, a resurgence of COVID and the enforcement of lockdowns and travel restrictions.
With further successful trials and FDA approvals, IGC hopes to bring a drug based on natural THC as an effective treatment for agitation in Alzheimer’s to market.
TGR-63
The company’s other molecule, TGR-63, has been shown to reduce the neurotoxicity that impacts memory loss in preclinical trials with mice. On a dose dependent manner, transgenic Alzheimer’s mice treated with TGR-63 showed improvement in memory relative to control.
Both drug candidates, IGC-AD1 and TGR-63, have shown their ability to reduce the brain plaques associated with memory loss in Alzheimer’s in mice.
With further successful trials and FDA approvals, IGC hopes to bring TGR-63 as a treatment for Alzheimer’s disease to market.
Market Opportunity
Alzheimer’s disease impacts over 55 million people worldwide and about 5.5 million individuals in the U.S. Over 70% of these patients face debilitating symptoms, including anxiety, depression, and agitation (Mendez, 2021). Agitation in dementia patients can include excessive physical movement and verbal activity, restlessness, pacing, belligerence, aggression, screaming, crying, and wandering.
In 2020, the estimated healthcare costs for Alzheimer’s disease in the U.S. were $305 billion. Medicare and Medicaid covered about 70% of those costs, leaving considerable burden on patients and families. At the current rate of growth of Alzheimer’s and other dementia diagnoses, those costs are estimated to reach over $1 trillion by 2050.
Currently, there are no FDA-approved medications to alleviate the symptoms of dementia due to Alzheimer’s disease, providing a tremendous opportunity for formulations that can have an impact on quality of life and disease progression.
Management Team
Richard Prins has been chairman at IGC since 2012 and served as an independent director since 2007. From March 1996 to 2008, he was the Director of Investment Banking at Ferris, Baker Watts, Incorporated. Prins served in a consulting role to RBC until January 2009. He currently volunteers full time with a non-profit organization, Advancing Native Missions, and is a private investor. Since February 2003, he has been on the board of Amphastar Pharmaceuticals Inc. He holds a bachelor’s degree from Colgate University and an MBA from Oral Roberts University.
Ram Mukunda is CEO and President of IGC. He has been the chief inventor and architect of most of the company’s patent filings and is responsible for the company’s strategic positioning. Prior to IGC, he was founder and CEO of Startec Global Communications, which he took public in 1997. He served as Strategic Planning Advisor at Intelsat, a communications satellite services provider. From 2001 to 2003, he was a Council Member at Harvard’s Kennedy School of Government, Belfer Center of Science and International Affairs. He was named the 1998 Ernst & Young Entrepreneur of the Year. He holds bachelor’s degrees in electrical engineering and mathematics, and a master’s degree in engineering from the University of Maryland.
Dr. Jagadeesh Rao is the company’s Principal Scientist. His career spans two decades in the public sector and product R&D for Johnson & Johnson. He leads IGC’s scientists in the development of pharmaceutical and OTC products. He worked for the federal National Institutes of Health, and for the National Institute on Drug Abuse. His Ph.D. in Neurochemistry is from the National Institute of Mental Health & Neurosciences in India. He did postdoctoral training at the University of Illinois-Chicago.
Claudia Grimaldi is a Director, Vice President, Principal Financial Officer, and Chief Compliance Officer for IGC. She also serves as a Director/Manager Director for some of the company’s subsidiaries. She graduated with highest honors from Javeriana University in Colombia with a bachelor’s degree in psychology. She holds an MBA, graduating with highest honors, from Meredith College in North Carolina. In addition, she has attended the Darden School of Business Financial Management Executives program and the Corporate Governance Program at Columbia Business School. She is currently pursuing her Directorship Certification with the National Association of Corporate Directors. She is fluent in both English and Spanish.
India Globalization Capital Inc. (NYSE American: IGC), closed Monday's trading session at $0.358, off by 3.2168%, on 168,771 volume. The average volume for the last 3 months is 165,712 and the stock's 52-week low/high is $0.2785/$1.16.
Recent News
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - Missouri Expunges at Least 7,500 Cannabis Convictions
- CannabisNewsBreaks - India Globalization Capital Inc. (NYSE American: IGC) Bringing Hope for Alzheimer's Treatment
- 420 with CNW - Two Cannabis Legalization Measures in Wyoming Fail to Make 2024 Ballot
Advanced Container Technologies Inc. (OTC: ACTX)
The QualityStocks Daily Newsletter would like to spotlight Advanced Container Technologies Inc. (OTC: ACTX).
There were a lot of concerns when
Advanced Container Technologies Inc. (OTC: ACTX) is in the business of selling and distributing self-contained, automated, indoor “micro-farms” called Grow Pods, along with related equipment and supplies. Additionally, the company designs and sells patented proprietary medical-grade plastic containers, known as the Medtainer®, that store and grind pharmaceuticals, herbs, teas and other solids or liquids.
ACTX is the leading distributor of Grow Pods. With a controlled environment, food and herbs can be grown without pesticides, harmful chemicals or risk of pathogen contamination, and with low energy consumption. Restaurants, grocery stores, non-profits, MSOs and entrepreneurs can use Grow Pods to ensure a fresh supply of ultra-clean produce year-round.
The company entered the Grow Pod business in October 2020 with its acquisition of all shares of Advanced Container Technologies Inc., a California corporation. As of February 28, 2022, ACTX is exploring the acquisition of the assets and the assumption of some or all of the liabilities of GP Solutions Inc., the developer and manufacturer of Grow Pods, for which ACTX is currently the sole U.S. distributor.
Because Grow Pods can be located almost anywhere, produce can be grown closer to the point of consumption and harvested at its peak, providing nutritious fruits and vegetables where needed. Indoor micro-farms, utilizing a practice known as vertical farming, have attracted the attention of governments and universities, which are now promoting vertical farming as a way to combat food insecurity and inequities.
The United States Department of Agriculture (USDA) has stated that vertical farming “is no longer a futuristic concept.” The department is enthusiastic about vertical farming, particularly those utilizing repurposed shipping containers, such as Grow Pods. Arizona State University reports that vertical farming reduces water use by 90 percent compared to conventional farming but produces 10 times the crop yield.
Products
Grow Pods
One of the company’s main business units is focused on selling advanced, self-contained hydroponic containers called Grow Pods. These unique and innovative automated systems are essentially micro-farms that can be placed virtually anywhere and, with their controlled and specially filtered environment, allow cultivation of a wide variety of crops, 365 days a year. The Grow Pod controlled environment offers major advantages for the production of high-value crops. The ability to grow year-round and the ability to cultivate in a smaller footprint using less water and power are some of the primary advantages of the system. Grow Pods offer constant temperature, humidity and airflow control, as well as automated watering and lighting schedules for optimal growth and minimal labor requirements, regardless of crop.
Containers
ACTX meets the needs of the pharmaceutical and medical markets, including the cannabis and hemp industries, with patented packaging systems. The company designs, customizes, brands and sells proprietary medical grade plastic containers that can store pharmaceuticals, herbs, teas and other solids or liquids, with a special built-in feature that can grind solids and shred herbs. The company’s flagship container product is the patented Medtainer®, a child resistant, medical-grade herb container and grinder that is water-tight, air-tight and smell proof. Packaging in the cannabis industry is critical, with numerous stringent regulations about how cannabis products must be packaged and labeled. ACTX also offers custom-branded, compliant vacuum seal bags and other retail container solutions.
Equipment and Supplies
ACTX markets and sells two principal products: Grow Pods, which are specially modified insulated shipping containers manufactured by GP Solutions Inc., in which plants, herbs and spices may be grown hydroponically in a controlled environment, and Medtainers®, which may be used to store pharmaceuticals, herbs, teas and other solids or liquids and can grind solids and shred herbs. The company also markets and sells various products related to Grow Pods and the Medtainer®, as well as providing private labeling and branding services for purchasers of Medtainers® and certain related products.
GP Solutions manufactures and sells other products, such as humidity controllers and LED lighting systems for vertical farming. The company’s specially designed lighting panels are programmed to emit the exact wavelength of light that each crop requires. The system has a daybreak-to-nightfall feature that gives plants the proper chromatic signals to grow rapidly and fruitfully. High efficiency LED light strips supply the crops with a red and blue light spectrum required for photosynthesis in the spectrum that plants need most.
Market Overview
The global vertical farming market is expected to reach $33.02 billion by 2030, according to a new report by Grand View Research. The market is forecast to expand at a CAGR of 25.5 percent from 2022 to 2030, according to Grand View. Escalating production of biopharmaceutical products, including cannabis, is anticipated to drive the market. The building-based segment of the market is expected to register a significant CAGR of 27.8 percent over the projected period. In addition, the climate control segment is expected to see high growth.
The global cannabis packaging market is expected to reach $14.34 billion by 2028, according to analysis by Reports and Data. The analysis forecasts 1,700 percent growth in cannabis users by the end of 2026, with packaging likely observing a whopping 26.42 percent growth in the forecast period. There are significant barriers to entry in the cannabis packaging market, giving an advantage to companies already established in the sector. These barriers include developing a thorough knowledge of the myriad regulations that govern cannabis packaging (which differ in each state), and child-resistance requirements.
Management Team
Douglas P. Heldoorn is the Founder and Chairman of Advanced Container Technologies Inc. He also holds the positions of President, CEO and COO at the company. Mr. Heldoorn has served on the Board of Directors since its inception in 2013. He has also previously held the position of Executive General Manager at Nissan Motor Corp.
Jeffory A. Carlson is CFO and Treasurer of ACTX. Mr. Carlson has also served as the company’s Corporate Controller since 2014.
Advanced Container Technologies Inc. (OTC: ACTX), closed Monday's trading session at $0.321, off by 8.2857%, on 200 volume. The average volume for the last 3 months is 200 and the stock's 52-week low/high is $0.2005/$1.25.
Recent News
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - How Oklahoma's Marijuana Landscape Could Evolve Once Adult-Use Cannabis is Legalized
- 420 with CNW - DOJ Launches Application for Federal Cannabis Pardon Certificates
- 420 with CNW - Virginia's Recreational Marijuana Launch Stalls Indefinitely
Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF)
The QualityStocks Daily Newsletter would like to spotlight Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF).
Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) is engaged in Rare Earth Element (REE) resource development and in commercializing its critical metals separation technology, RapidSX™, for the mining and metals extraction industry. The company is guided by principles of environmental, social and corporate governance (ESG) with a focus on disrupting China’s current dominance of the U.S. REE supply chain.
Ucore’s vision is to become a leading advanced technology company providing best-in-class metal separation products and services to the mining and mineral extraction industry. It plans to aid in the development, through strategic partnerships, of a North American REE supply chain controlled by the U.S. and its allies.
The company intends to contribute to this initiative through the near-term development of a heavy and light rare-earth processing facility in Louisiana and subsequent development of Strategic Metals Complexes (SMCs) in Alaska and Canada, as well as through the longer-term development of its 100%-owned Heavy Rare Earth Element (HREE) mineral resource property at Bokan Mountain on Prince of Wales Island, Alaska.
Ucore is headquartered in Halifax, Nova Scotia.
Projects & Technology
RapidSX™ Demonstration Plant
The Kingston, Ontario, RapidSX™ Demonstration Plant commissioning process is underway. Once commissioned, the plant is designed to demonstrate the commercial capabilities of the RapidSX technology platform.
The RapidSX demo plant will show:
- The techno-economic advantages of the RapidSX technology platform
- The processing of tens of tons of heavy and light mixed rare earth element concentrates in a simulated production environment
- The platform’s ability to operate for thousands of semi-continuous run-time hours
- Production of high-purity NdPr, praseodymium, neodymium, terbium and dysprosium rare earth elements for early OEM product qualification trials
The demo plant is located within Ucore’s 5,000-square-foot RapidSX Commercialization and Demonstration Facility and is run by its laboratory partner, Kingston Process Metallurgy Inc. (“KPM”).
RapidSX™ Technology
Innovation Metals Corp., acquired by the company in 2020, developed the RapidSX separation technology platform with early-stage assistance from the United States Department of Defense, later resulting in the production of commercial-grade, separated rare earth elements at pilot scale.
RapidSX combines the time-proven chemistry of conventional solvent extraction (SX) with a new column-based platform that significantly reduces time to completion and plant footprint, as well as potentially lowering capital and operating costs. SX is the international REE industry’s standard commercial separation technology and is currently used by all REE producers worldwide for bulk commercial separation of both heavy and light REEs.
Utilizing similar chemistry to conventional SX, RapidSX is not a “new” technology, but it represents a significant improvement on the well-established, well-understood, proven conventional SX separation technology preferred by REE producers.
Strategic Metals Complex
Ucore, engineering partner Mech-Chem Associates Inc. and KPM are developing the full-scale engineering for the company’s first Strategic Metals Complex (SMC). The SMC is a planned REE separation and rare earth oxide production plant slated to commence construction in Louisiana in 2023. It is scheduled to initially process 2,000 tons of total rare earth oxides by the end of 2024, increasing to 5,000 tons in 2026.
Bokan-Dotson Ridge REE Deposit
Ucore has invested over C$35 million to establish and validate the Bokan-Dotson Ridge resource in preparation for mine design and permitting. Initial drilling is complete, and a Preliminary Economic Assessment has been issued. Next steps for the project include a feasibility study, detailed mine design and permit acquisition. The project can be “near shovel ready” for construction in less than 30 months after receipt of the next stage of development funding.
Market Opportunity
According to a report by Grand View Research, the global rare earth elements market was valued at $2.8 billion in 2018 and is forecast to reach a value of $5.6 billion by 2025, achieving a CAGR of 10.4% during the period. Market growth is driven by increasing demand for these elements in the manufacturing of magnets and catalysts for the automotive industry. Rising demand for electric vehicles to reduce CO2 emissions is expected to propel the use of permanent magnets in the production of EV batteries.
China is the major producer and consumer of REEs. To maintain self-sufficiency and to meet future demand, China has been raising the export tariffs on rare earth elements shipped to various countries, including the U.S., Japan, India, Brazil and the European Union. This led to the current supply-demand gap in these countries, as they rely on imports from China.
China reduced the exports of REEs by 72% in the second half of 2010 to preserve its reserves of these elements and continues to export REEs at reduced levels, thereby affecting industries such as automotive, oil and gas, and electronics, which require an ample amount of rare earth elements.
Management Team
Pat Ryan, P.Eng., is Chairman and CEO of Ucore Rare Metals. He began as a director with the company when he developed a heightened interest in critical metals. Before joining Ucore, he founded and led a multimillion-dollar automotive OEM design and lean manufacturing company. His understanding of complex supply chains across international markets has led to a prime positioning as the global auto industry transitions to vehicle electrification. He holds a Bachelor of Engineering degree from Dalhousie University.
Peter Manuel is Vice President and CFO of Ucore. Prior to joining the company, he practiced as a Chartered Accountant for more than 17 years, providing consulting services to companies in a range of industries, with a focus on the financial services and resource sectors. He spent 10 years in England and Ireland providing assurance, strategic planning, corporate finance and other consulting services to a portfolio of both public and private entities. He holds a Bachelor of Commerce Degree from Dalhousie University.
Michael Schrider, MEng, P.E., is Vice President and COO of Ucore. He is a multidisciplinary engineer who has been involved in manufacturing, engineering and managing complex structural and mechanical systems projects since 1989. He was the Founder, President and Chief Engineer of Schrider & Associates and Alton Bay Design, both engineering services firms. He holds a bachelor’s degree in naval architecture and marine engineering from the University of New Orleans and a master’s degree in mining, geological and geophysical engineering from the University of Arizona.
Mark MacDonald is Vice President of Investor Relations at Ucore. He has over 25 years of experience implementing award winning business development and marketing programs at regional and national levels. As Vice President of Sales, he was responsible for Mediapro Communication’s growth as AT&T Canada’s leading B2B sales partner. He subsequently became Atlantic Regional Vice President of AT&T Canada Corp. He holds a Bachelor of Commerce degree from Dalhousie University.
Ucore Rare Metals Inc. (UURAF), closed Monday's trading session at $0.94, up 1.5887%, on 29,979 volume. The average volume for the last 3 months is 29,979 and the stock's 52-week low/high is $0.40/$1.15.
Recent News
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc.'s (TSX.V: UCU) (OTCQX: UURAF) RapidSX(TM) Combining Time-Proven SX with New Platform to Process REEs
- UK Government to Avail $18M Funding for Rare Earths Recycling Research
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Develops North American Rare Earth Element Technology Amidst Tensions with China
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) to Present at Summer Research Conference
- Aditxt Inc. (NASDAQ: ADTX) - Amesite Inc. (NASDAQ: ADTX) Announces That it Has Regained Compliance with Nasdaq Listing Requirements
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - How Oklahoma's Marijuana Landscape Could Evolve Once Adult-Use Cannabis is Legalized
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Regains Compliance with NASDAQ Minimum Bid Price, Other Listing Requirements
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Announces US$5M Private Placement
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - MiningNewsBreaks - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Leveraging Decades of Industry Experience to Develop World-Class Exploration Projects
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Names Rod O'Reilly as Non-Executive Chairman of the Board of Directors
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Study Shows Air Pollution in Urban Areas Worsens Lung Function in Kids
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Enters New Distribution Deal with Major India Distributor, Expands in Global Market
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Assay Results Confirms Potential at Texmont
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Announces Participation in Upcoming Roth Conference
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Announces It Has No Exposure to Silicon Valley Bank Issues
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Experiencing Record Growth as It Pushes Burgeoning Organic Textile Market Forward
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) CEO to Participate at Oppenheimer 33rd Annual Health Care Conference
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Q4 and FY 2022 Results
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Files 2022 Annual Report, Well Positioned to Deliver High-Grade Data Security and Protection Services as Data Breaches Go Mainstream
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Schedules Q4, FY 2022 Earnings Report and Call
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Risks of Buying Psilocybin Mushrooms from Online Suppliers
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) New Line of Golf Carts Featured in Proactive Investor Video
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Report Reveals Central Banks Are Still Stockpiling Gold
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Energy Fuels Inc. (UUUU) Q4 2022 Earnings Call Transcript
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - ESGBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Ideally Poised as Increased RNG Use Expected to Catalyze Growth
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Inks Strategic Partnership with Global Automotive Industry Supplier
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - Creatd Inc. (NASDAQ: CRTD) Creates Advisory Board, Names Inaugural Member
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Releases Update on Drilling at Augustus Lithium Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Enters POC Project with Global Japanese Vehicle Manufacturer
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Strong YTD Results, Projects ~40% Annual Revenue Growth in 2023
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Initiates Testing on Green Ammonia Production System
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Tapping Opportunity to Make SmartGreen(TM) a Household Name
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces $4M Registered Direct Offering
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Issues Update on Restatement of Previously Issued Financial Statements
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) On Track to Advance Innovative Products
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Schedules Release of Q4 and FY 2022 Financial Results, Earnings Call
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Targets Clean Energy Sector with ZVS Power Inverter Architecture and Platform
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Hits 5K Milestone, Continues Welcoming the Future of Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Manufacturing of New Round of Zero-Emission Street Sweepers
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - Missouri Expunges at Least 7,500 Cannabis Convictions
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces $5M Registered Direct Offering, Private Placement
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Peer-Reviewed Study Results Published
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc.'s (INND) Now Offering OTC Rechargeable Hearing Aids on Target.com, Celebrates World Hearing Day
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Simple Skin Swab Could Help Predict Future Eczema Risks in Kids
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Deploys 'Optimus' ASR for Fifth Year Running
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Releases Additional Findings From Human Clinical Study, Demonstrating DehydraTECH(TM)-CBD Effects on Hypertension
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) subsidiary McEwen Copper adds $30 Million Boost from Nuton LLC to recent Stellantis investment of $155 Million, Plans for subsidiary IPO
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Reports on Debt Settlement Agreements
- Nemaura Medical Inc. (NASDAQ: NMRD) - Nemaura Medical Announces Receipt of Notice from Nasdaq
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Schedules Q4 and 2022 Results Conference Call
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTCQB: PLTXF) (Frankfurt: WNT1) Expands eCommerce Platform to Add 'No Evil Foods'
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Reports Success at ICE London iGaming Event
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Releases Update on Corporate Activities, Subsidiary Highlights
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Updates Shareholders, Provides Company Overview and Plans for Future
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Largest Investor Exchanges $10M-Plus Debt into Non-Secured Equity
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Secures Reg A+ Qualification, Eyes New Heights of Opportunity
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Prioritizes Development of Next Generation Chemotherapies
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Featured in New Goldman Small Cap Research Report
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Top Marijuana Stocks Right Now In 2023
- REZYFi, Inc. - 420 with CNW — Voters in Oklahoma to Decide Whether to Legalize Recreational Marijuana
- RYAH Group Inc. (CSE: RYAH) - Failure-to File Cease Trade Order- Answer to the Second Comment Letter from the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - Are These Relatively Little-Known Organic And Earth-Friendly Food Stocks On Your Watchlist?
- Sharing Services Global Corporation (SHRG) - DSS Announces Letter to Shareholders
- SideChannel Inc. (OTCQB: SDCH) - InvestorNewsBreaks - SideChannel Inc. (SDCH), Virgin Voyages Team up to Enhance Data Protection from Ship to Shore
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Collaborates with DyeMansion to Expand into Post Processing
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Converts over $1 Million in Debt to Equity This Week with Another $119k Conversion; Update on Annual Filings Delay
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Deploys SD7 MCPTT Solution for 2023 Special Olympics
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc.'s (NYSE American: SBEV) Copa di Vino, Pulpoloco and TapouT to Hit Shelves Across North Carolina
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation Schedule for Upcoming Investor Conferences
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces its TV Corporate Interviews Airing as Episodes 447 and 448 with Shows Starting on Saturday, March 11, 2023, 3:30 PM ET
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Enters Agreement to Acquire Smoking Cessation App
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Release Q3 Financial Report, Update
- Tingo Inc. (OTC: TMNA) - 2023 Is Year of High-Quality Blockchain Games - Polygon
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc.'s (TSX.V: UCU) (OTCQX: UURAF) RapidSX(TM) Combining Time-Proven SX with New Platform to Process REEs
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Result 'Demonstrates Potential' of, Expands Christie Lake Project
- Utopia VR - InvestorNewsBreaks – Utopia VR Disrupting Future of Enterprise, Education and Corporate Remote Offerings with Distinct Metaverse Solution
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Provides Update on Phase 1 Clinical Trial of Proprietary PH10
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Expands Product Portfolio, Revenue Potential Through Strategic Acquisition
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Reports Fourth Quarter and Full Year 2022 Results
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) to Present at Summer Research Conference
- Aditxt Inc. (NASDAQ: ADTX) - Amesite Inc. (NASDAQ: ADTX) Announces That it Has Regained Compliance with Nasdaq Listing Requirements
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - How Oklahoma's Marijuana Landscape Could Evolve Once Adult-Use Cannabis is Legalized
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Regains Compliance with NASDAQ Minimum Bid Price, Other Listing Requirements
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Announces US$5M Private Placement
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - MiningNewsBreaks - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Leveraging Decades of Industry Experience to Develop World-Class Exploration Projects
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Names Rod O'Reilly as Non-Executive Chairman of the Board of Directors
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Study Shows Air Pollution in Urban Areas Worsens Lung Function in Kids
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Enters New Distribution Deal with Major India Distributor, Expands in Global Market
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Assay Results Confirms Potential at Texmont
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Announces Participation in Upcoming Roth Conference
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Announces It Has No Exposure to Silicon Valley Bank Issues
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Experiencing Record Growth as It Pushes Burgeoning Organic Textile Market Forward
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) CEO to Participate at Oppenheimer 33rd Annual Health Care Conference
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Q4 and FY 2022 Results
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Files 2022 Annual Report, Well Positioned to Deliver High-Grade Data Security and Protection Services as Data Breaches Go Mainstream
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Schedules Q4, FY 2022 Earnings Report and Call
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Risks of Buying Psilocybin Mushrooms from Online Suppliers
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) New Line of Golf Carts Featured in Proactive Investor Video
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Report Reveals Central Banks Are Still Stockpiling Gold
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Energy Fuels Inc. (UUUU) Q4 2022 Earnings Call Transcript
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - ESGBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Ideally Poised as Increased RNG Use Expected to Catalyze Growth
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Inks Strategic Partnership with Global Automotive Industry Supplier
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - Creatd Inc. (NASDAQ: CRTD) Creates Advisory Board, Names Inaugural Member
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Releases Update on Drilling at Augustus Lithium Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Enters POC Project with Global Japanese Vehicle Manufacturer
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Strong YTD Results, Projects ~40% Annual Revenue Growth in 2023
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Initiates Testing on Green Ammonia Production System
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Tapping Opportunity to Make SmartGreen(TM) a Household Name
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces $4M Registered Direct Offering
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Issues Update on Restatement of Previously Issued Financial Statements
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) On Track to Advance Innovative Products
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Schedules Release of Q4 and FY 2022 Financial Results, Earnings Call
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Targets Clean Energy Sector with ZVS Power Inverter Architecture and Platform
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Hits 5K Milestone, Continues Welcoming the Future of Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Manufacturing of New Round of Zero-Emission Street Sweepers
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - Missouri Expunges at Least 7,500 Cannabis Convictions
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces $5M Registered Direct Offering, Private Placement
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Peer-Reviewed Study Results Published
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc.'s (INND) Now Offering OTC Rechargeable Hearing Aids on Target.com, Celebrates World Hearing Day
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Simple Skin Swab Could Help Predict Future Eczema Risks in Kids
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Deploys 'Optimus' ASR for Fifth Year Running
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Releases Additional Findings From Human Clinical Study, Demonstrating DehydraTECH(TM)-CBD Effects on Hypertension
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) subsidiary McEwen Copper adds $30 Million Boost from Nuton LLC to recent Stellantis investment of $155 Million, Plans for subsidiary IPO
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Reports on Debt Settlement Agreements
- Nemaura Medical Inc. (NASDAQ: NMRD) - Nemaura Medical Announces Receipt of Notice from Nasdaq
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Schedules Q4 and 2022 Results Conference Call
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTCQB: PLTXF) (Frankfurt: WNT1) Expands eCommerce Platform to Add 'No Evil Foods'
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Reports Success at ICE London iGaming Event
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Releases Update on Corporate Activities, Subsidiary Highlights
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Updates Shareholders, Provides Company Overview and Plans for Future
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Largest Investor Exchanges $10M-Plus Debt into Non-Secured Equity
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Secures Reg A+ Qualification, Eyes New Heights of Opportunity
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Prioritizes Development of Next Generation Chemotherapies
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Featured in New Goldman Small Cap Research Report
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Top Marijuana Stocks Right Now In 2023
- REZYFi, Inc. - 420 with CNW — Voters in Oklahoma to Decide Whether to Legalize Recreational Marijuana
- RYAH Group Inc. (CSE: RYAH) - Failure-to File Cease Trade Order- Answer to the Second Comment Letter from the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - Are These Relatively Little-Known Organic And Earth-Friendly Food Stocks On Your Watchlist?
- Sharing Services Global Corporation (SHRG) - DSS Announces Letter to Shareholders
- SideChannel Inc. (OTCQB: SDCH) - InvestorNewsBreaks - SideChannel Inc. (SDCH), Virgin Voyages Team up to Enhance Data Protection from Ship to Shore
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Collaborates with DyeMansion to Expand into Post Processing
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Converts over $1 Million in Debt to Equity This Week with Another $119k Conversion; Update on Annual Filings Delay
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Deploys SD7 MCPTT Solution for 2023 Special Olympics
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc.'s (NYSE American: SBEV) Copa di Vino, Pulpoloco and TapouT to Hit Shelves Across North Carolina
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation Schedule for Upcoming Investor Conferences
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces its TV Corporate Interviews Airing as Episodes 447 and 448 with Shows Starting on Saturday, March 11, 2023, 3:30 PM ET
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Enters Agreement to Acquire Smoking Cessation App
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Release Q3 Financial Report, Update
- Tingo Inc. (OTC: TMNA) - 2023 Is Year of High-Quality Blockchain Games - Polygon
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc.'s (TSX.V: UCU) (OTCQX: UURAF) RapidSX(TM) Combining Time-Proven SX with New Platform to Process REEs
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Result 'Demonstrates Potential' of, Expands Christie Lake Project
- Utopia VR - InvestorNewsBreaks – Utopia VR Disrupting Future of Enterprise, Education and Corporate Remote Offerings with Distinct Metaverse Solution
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Provides Update on Phase 1 Clinical Trial of Proprietary PH10
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Expands Product Portfolio, Revenue Potential Through Strategic Acquisition
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Reports Fourth Quarter and Full Year 2022 Results
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) to Present at Summer Research Conference
- Aditxt Inc. (NASDAQ: ADTX) - Amesite Inc. (NASDAQ: ADTX) Announces That it Has Regained Compliance with Nasdaq Listing Requirements
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - How Oklahoma's Marijuana Landscape Could Evolve Once Adult-Use Cannabis is Legalized
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Regains Compliance with NASDAQ Minimum Bid Price, Other Listing Requirements
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Announces US$5M Private Placement
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - MiningNewsBreaks - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Leveraging Decades of Industry Experience to Develop World-Class Exploration Projects
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Names Rod O'Reilly as Non-Executive Chairman of the Board of Directors
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Study Shows Air Pollution in Urban Areas Worsens Lung Function in Kids
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Enters New Distribution Deal with Major India Distributor, Expands in Global Market
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Assay Results Confirms Potential at Texmont
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Announces Participation in Upcoming Roth Conference
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Announces It Has No Exposure to Silicon Valley Bank Issues
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Experiencing Record Growth as It Pushes Burgeoning Organic Textile Market Forward
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) CEO to Participate at Oppenheimer 33rd Annual Health Care Conference
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Q4 and FY 2022 Results
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Files 2022 Annual Report, Well Positioned to Deliver High-Grade Data Security and Protection Services as Data Breaches Go Mainstream
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Schedules Q4, FY 2022 Earnings Report and Call
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Risks of Buying Psilocybin Mushrooms from Online Suppliers
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) New Line of Golf Carts Featured in Proactive Investor Video
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Report Reveals Central Banks Are Still Stockpiling Gold
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Energy Fuels Inc. (UUUU) Q4 2022 Earnings Call Transcript
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - ESGBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Ideally Poised as Increased RNG Use Expected to Catalyze Growth
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Inks Strategic Partnership with Global Automotive Industry Supplier
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - Creatd Inc. (NASDAQ: CRTD) Creates Advisory Board, Names Inaugural Member
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Releases Update on Drilling at Augustus Lithium Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Enters POC Project with Global Japanese Vehicle Manufacturer
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Strong YTD Results, Projects ~40% Annual Revenue Growth in 2023
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Initiates Testing on Green Ammonia Production System
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Tapping Opportunity to Make SmartGreen(TM) a Household Name
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces $4M Registered Direct Offering
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Issues Update on Restatement of Previously Issued Financial Statements
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) On Track to Advance Innovative Products
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Schedules Release of Q4 and FY 2022 Financial Results, Earnings Call
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Targets Clean Energy Sector with ZVS Power Inverter Architecture and Platform
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Hits 5K Milestone, Continues Welcoming the Future of Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Manufacturing of New Round of Zero-Emission Street Sweepers
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - Missouri Expunges at Least 7,500 Cannabis Convictions
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces $5M Registered Direct Offering, Private Placement
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Peer-Reviewed Study Results Published
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc.'s (INND) Now Offering OTC Rechargeable Hearing Aids on Target.com, Celebrates World Hearing Day
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Simple Skin Swab Could Help Predict Future Eczema Risks in Kids
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Deploys 'Optimus' ASR for Fifth Year Running
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Releases Additional Findings From Human Clinical Study, Demonstrating DehydraTECH(TM)-CBD Effects on Hypertension
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) subsidiary McEwen Copper adds $30 Million Boost from Nuton LLC to recent Stellantis investment of $155 Million, Plans for subsidiary IPO
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Reports on Debt Settlement Agreements
- Nemaura Medical Inc. (NASDAQ: NMRD) - Nemaura Medical Announces Receipt of Notice from Nasdaq
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Schedules Q4 and 2022 Results Conference Call
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTCQB: PLTXF) (Frankfurt: WNT1) Expands eCommerce Platform to Add 'No Evil Foods'
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Reports Success at ICE London iGaming Event
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Releases Update on Corporate Activities, Subsidiary Highlights
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Updates Shareholders, Provides Company Overview and Plans for Future
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Largest Investor Exchanges $10M-Plus Debt into Non-Secured Equity
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Secures Reg A+ Qualification, Eyes New Heights of Opportunity
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Prioritizes Development of Next Generation Chemotherapies
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Featured in New Goldman Small Cap Research Report
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Top Marijuana Stocks Right Now In 2023
- REZYFi, Inc. - 420 with CNW — Voters in Oklahoma to Decide Whether to Legalize Recreational Marijuana
- RYAH Group Inc. (CSE: RYAH) - Failure-to File Cease Trade Order- Answer to the Second Comment Letter from the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - Are These Relatively Little-Known Organic And Earth-Friendly Food Stocks On Your Watchlist?
- Sharing Services Global Corporation (SHRG) - DSS Announces Letter to Shareholders
- SideChannel Inc. (OTCQB: SDCH) - InvestorNewsBreaks - SideChannel Inc. (SDCH), Virgin Voyages Team up to Enhance Data Protection from Ship to Shore
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Collaborates with DyeMansion to Expand into Post Processing
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Converts over $1 Million in Debt to Equity This Week with Another $119k Conversion; Update on Annual Filings Delay
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Deploys SD7 MCPTT Solution for 2023 Special Olympics
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc.'s (NYSE American: SBEV) Copa di Vino, Pulpoloco and TapouT to Hit Shelves Across North Carolina
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation Schedule for Upcoming Investor Conferences
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces its TV Corporate Interviews Airing as Episodes 447 and 448 with Shows Starting on Saturday, March 11, 2023, 3:30 PM ET
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Enters Agreement to Acquire Smoking Cessation App
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Release Q3 Financial Report, Update
- Tingo Inc. (OTC: TMNA) - 2023 Is Year of High-Quality Blockchain Games - Polygon
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc.'s (TSX.V: UCU) (OTCQX: UURAF) RapidSX(TM) Combining Time-Proven SX with New Platform to Process REEs
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Result 'Demonstrates Potential' of, Expands Christie Lake Project
- Utopia VR - InvestorNewsBreaks – Utopia VR Disrupting Future of Enterprise, Education and Corporate Remote Offerings with Distinct Metaverse Solution
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Provides Update on Phase 1 Clinical Trial of Proprietary PH10
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Expands Product Portfolio, Revenue Potential Through Strategic Acquisition
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Reports Fourth Quarter and Full Year 2022 Results
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































