The QualityStocks Daily Stock List
- Voip-Pal.Com Inc. (VPLM)
- Kintara Therapeutics (KTRA)
- GlucoTrack Inc. (GCTK)
- EzFill Holdings (EZFL)
- Onconova Therapeutics (ONTX)
- Orogen Royalties (OGNRF)
- Intermap Technologies (ITMSF)
- Hestia Insight (HSTA)
- GoviEx Uranium (GVXXF)
- Polydex Pharmaceuticals (POLXF)
- Kandi Technologies Group Inc. (KNDI)
- HIVE Blockchain Technologies Ltd. (HIVE)
Voip-Pal.Com Inc. (VPLM)
QualityStocks, Equities.com, Mega Stock Pick, Pumps and Dumps, HotOTC, Stock Rich, Pick Alerts, BullRally, Buzz Stocks, Clutch Investments, CoolPennyStocks, equities Canada, Google Alerts, Monster OTC, Penny Stock Fever, StockEgg, MajorPennyStocks, Breaking Bulls, Bullish Stock Picks, Wallstreetbuzz, Bullseyestox.com, VC Stock Marketing, UndiscoveredEquities, TryBestPennyStocks.biz, TooNiceStocks, TheSUBWAY, FeedBlitz, Stockoutlaws, Penny Invest, HotStockProfits, SmallCapAllStars, MarketClub Analysis, Mega Stock Picks, Stock Twiter, NYC Marketing Inc, Best Stock Picks, Stock Traders Chat, WallstreetsHotteststocks, Premiumstockpicks, Promotion Stock Secrets, Stock Fortune Teller, SmallCapVoice and StockHotTips reported earlier on Voip-Pal.Com Inc. (VPLM), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Voip-Pal.Com Inc. (OTCQB: VPLM) is a broadband VoIP telecom firm which develops and owns a portfolio of VoIP services.
The firm has its headquarters in Waco, Texas and was incorporated in September 1997 by Emil Malak and Francis Assif. Prior to its name change in September 2006, the firm was known as VOIP MDI.com. It operates as part of the telecom services industry, under the communication services sector. The firm serves commercial and residential clients around the globe.
The company is focused on creating virtual bridges which span international communications. Its objective is to connect the world through the delivery of cost-efficient technology that enables high-volume voice, text and digital multi-media communications.
The enterprise provides routing and classification of communications geographically distributed nodes and over a number of networks; uninterrupted transmissions in the course of endpoint changes; mobile gateways; improved emergency calling support services; lawful interception of such communications; and billing and metering, including the resale of white label telecommunication services. It also offers a transactional billing platform that’s tailored to the air mile and points business. This is in addition to providing antivirus applications for smartphones. The enterprise serves wholesale and retail carriers, network suppliers and telephony system vendors.
The firm is focused on monetizing its VoIP (Voice over Internet Protocol) technology, which will help bring in revenues into the firm as well as attract additional investors. This is in addition to extending the firm’s consumer reach and global reach, which will bolster its growth and create revenue for its shareholders.
Voip-Pal.Com Inc. (VPLM), closed Wednesday's trading session at $0.083, up 110.1266%, on 30,526,619 volume. The average volume for the last 3 months is 770,486 and the stock's 52-week low/high is $0.01/$0.1144.
Kintara Therapeutics (KTRA)
QualityStocks, TradersPro, The Online Investor, The Stock Dork, StockEarnings, Profitable Trader Authority, PennyStockScholar, pennystockprophet, OTCtipReporter, MarketClub Analysis, MarketBeat, InvestorPlace and Early Bird reported earlier on Kintara Therapeutics (KTRA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Kintara Therapeutics, Inc. (NASDAQ: KTRA) (OTC: DMPWW) (FRA: 3DM) is a clinical stage drug development firm that is engaged in the development and commercialization of anti-cancer therapies for the treatment of cancer.
The firm has its headquarters in San Diego, California and was incorporated in 2009, on June 24th by William J. Garner, Dennis M. Brown and Jeffrey A. Bacha. Prior to its name change in August 2020, the firm was known as DelMar Pharmaceuticals Inc. It operates as part of the scientific research and development services industry and serves consumers around the globe. The firm has two companies in its corporate family.
The company is focused on identifying commercial and clinical-stage compounds and establishing a scientific rationale for developing orphan drug indications. It has teamed up with outstanding clinical research and academic institutions across the globe to support the development of its cancer treatments. It is party to a strategic collaboration with Guangxi Wuzhou Pharmaceutical Co. Ltd, which entails manufacturing and selling VAL-083 in China.
The enterprise’s product pipeline comprises of two late-stage therapeutics, including a photodynamic therapy dubbed REM-001 for the treatment of cutaneous metastatic breast cancer; and a DNA-targeting agent dubbed VAL-083 for the treatment of drug-resistant solid tumors like diffuse intrinsic pontine glioma, non-small cell lung cancer, ovarian cancer and glioblastoma multiforme.
The company recently announced its financial results for its fiscal year ended June 2021, with its CEO noting that the company was well-positioned both on its corporate and clinical development front, having entered into purchase agreements with investors to raise monies which will be used for the company’s corporate working capital needs and ongoing clinical studies.
Kintara Therapeutics (KTRA), closed Wednesday's trading session at $4.36, up 47.2973%, on 835,396 volume. The average volume for the last 3 months is 4.236M and the stock's 52-week low/high is $2.70/$20.00.
GlucoTrack Inc. (GCTK)
QualityStocks, PennyStockProphet, Money Wealth Matters, MarketClub Analysis, InvestorsUnderground and INO Market Report reported earlier on GlucoTrack Inc. (GCTK), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
GlucoTrack Inc. (NASDAQ: GCTK) is a medical device firm that is focused on designing, developing and commercializing glucose monitoring devices for use by individuals suffering from diabetes and pre-diabetics.
The firm has its headquarters in Or Yehuda, Israel and was incorporated in September 2001, by David Malka and Avner Gal. Prior to its name change in November 2021, the firm was known as Integrity Applications Inc. The firm serves consumers around the globe, with a focus on the Asia Pacific, the United States, Israel and Europe.
The enterprise’s products include the GlucoTrack model DF-F, a glucose monitoring device which uses a patented combination of thermal, electromagnetic and ultrasound technologies to obtain glucose readings for pre-diabetic and diabetic patients in less than a minute, through the use of a small sensor that is clipped onto the individual’s earlobe using a Personal Ear Clip. The sensor contains calibration electronics and is linked to a handheld display and control unit. The monitoring device has been designed to obtain glucose measurements without drawing interstitial fluid or blood, making the device pain-free and needle-free. The enterprise also develops a wireless module for the transmission of data of the glucose readings captured by the device to a cloud-based server.
The company recently completed its uplisting on Nasdaq, which will grow the company’s shareholder base, increase corporate visibility and allow both the company and its shareholders greater access to liquidity. This move comes after it appointed a new CEO, who has extensive experience working with new and innovative diabetes management devices.
GlucoTrack Inc. (GCTK), closed Wednesday's trading session at $0.4691, up 34.9152%, on 4,262,004 volume. The average volume for the last 3 months is 425,318 and the stock's 52-week low/high is $0.20755/$3.75.
EzFill Holdings (EZFL)
QualityStocks, Small Caps and MarketBeat reported earlier on EzFill Holdings (EZFL), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
EzFill Holdings Inc. (NASDAQ: EZFL) is a mobile fueling application-based firm that is engaged in the provision of on-demand mobile gas and fueling delivering services to businesses and consumers.
The firm has its headquarters in Miami, Florida and was incorporated in 2019, on March 28th by Michael Farkas. The firm serves consumers in the United States, with a focus on the state of Florida.
The company’s objective is to alter the gas station fueling model by offering businesses and consumers the touch-free, safety and convenient benefits of fueling services which are on-demand and are delivered to customer locations. Delivery on-site during downtimes for specialty and commercial consumers allows operators to start their daily operations with already fueled vehicles.
The enterprise offers fuel-delivery in subscription or on-demand in the specialty, commercial and consumer verticals. Its app-based interface offers customers the ability to select the location and time of their fueling needs, whether their services are structured in routine schedules or their service request is on demand. The enterprise streamlines its fuel purchasing and logistics with backend software, which mobilizes its fleet of delivery trucks and its customer accounts. The enterprise serves livery operators, rental car owners, boat owners at marinas and equipment rental companies, among others.
The company recently purchased an additional 33 fuel trucks, which has significantly expanded the size and capacity of its fleet and will in turn facilitate the expansion of the services in various cities in the U.S. Expanding its service offering will help extend the company’s consumer reach, which may bring in more investors and boost its growth.
EzFill Holdings (EZFL), closed Wednesday's trading session at $2.85, up 33.1713%, on 425,318 volume. The average volume for the last 3 months is 989,030 and the stock's 52-week low/high is $1.46/$7.84.
Onconova Therapeutics (ONTX)
MarketBeat, TraderPower, PCG Advisory, BUYINS.NET, StreetInsider, StockMarketWatch, QualityStocks, The Street, Channelchek, Broad Street, AwesomeStocks, Penny Stock 101, StockRockandRoll, OTCBB Journal, PennyStockLocks, The Online Investor, Investors Alley, MarketClub Analysis, Marketbeat.com, Money Morning, SeeThruEquity Research, Schaeffer's, InvestorPlace, Investing Futures, Daily Trade Alert, Zacks, TopPennyStockMovers, Trades Of The Day and Louis Navellier reported earlier on Onconova Therapeutics (ONTX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Onconova Therapeutics Inc. (NASDAQ: ONTX; FRA: OT22) is a biopharmaceutical firm which operates through the discovery and development of oncology therapeutics. Onconova is centered on finding and developing drug candidates to fight cancer.
The firm has its headquarters in Newton, Pennsylvania and was founded on December 22, 1998 by E. Premkumar Reddy and Ramesh Kumar. Onconova launched its IPO in 2013, which generated $78 million.
Onconova Therapeutics has various revenue-generating collaboration agreements with different companies. These include its collaboration and license agreement with HanX Biopharmaceuticals, a commercialization, development and license agreement with Pint International SA, and a SymBio Pharmaceuticals limited license agreement, for the registration, development and marketing of ON 123300.
Onconova Therapeutics Inc. has developed anti-cancer agents that work against particular cellular pathways vital to cancerous cells. The company’s drug candidates include both oral and intravenous formulations Rigosertib, which are in their Phase 2 and 3 clinical trials for lower and higher risk myelodysplastic syndromes as well as head, neck and pancreatic cancers. It’s other candidate Briciclib is undergoing a phase 1 dose-escalation multi-site trial for advanced solid tumors. Another drug candidate developed by the firm, Recilisib, recently concluded 4 phase 1 clinical trials for acute radiation syndromes.
The firm is focused on developing medications that better the outcomes of cancer patients, addresses the recurrence of the ailment and reduces the side effects brought about by the current cancer treatment methods. Rigosertib, which is Onconova’s lead candidate, has shown potential in doing so. If the product demonstrates promising results in its clinical trials, which result in an effective treatment, it may revolutionize how cancer, a disease that so many individuals succumb to annually, is managed and treated.
Onconova Therapeutics (ONTX), closed Wednesday's trading session at $1.39, up 29.9065%, on 993,101 volume. The average volume for the last 3 months is 7,902 and the stock's 52-week low/high is $0.62/$1.52.
Orogen Royalties (OGNRF)
We reported earlier on Orogen Royalties (OGNRF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Orogen Royalties Inc. (OTCQX: OGNRF) (CVE: OGN) (FRA: 5EV) is a mineral exploration firm that is focused on generating royalties through exploration.
The firm has its headquarters in Vancouver, Canada and was incorporated in 2005, on May 11th. Prior to its name change in August 2020, the firm was known as Evrim Resources Corp. It operates as part of the gold industry, under the basic materials sector. The firm serves consumers around the globe.
The company operates through the Mineral Royalties and the Mineral Exploration Project Generation segments. It owns a geological database covering parts of Mexico, the South Pacific, central Asia, western Canada, and the western United States. The company is well-financed with several projects actively being developed by joint venture partners.
The enterprise has a diverse portfolio of precious metal royalties and copper, gold, and silver exploration projects in the United States, Canada, Kenya, Mexico and Argentina. Its portfolio features gold and base metal projects in exploration districts, including Nevada, Northwestern British Columbia and Northwestern Mexico. Its flagship royalties include Silicon and Ermitano. The Silicon project is located in Bare Mountain mining district, Nye County, Nevada. The Ermitano property comprises of 165 km2 of contiguous mineral tenure located roughly 145 km from Hermosillo and only 3.5 km southeast of First Majestic’s Santa Elena gold-silver mine in Sonora, Mexico.
The firm, whose latest financial results show increases in its revenues, is committed to delivering value for its shareholders.
Orogen Royalties (OGNRF), closed Wednesday's trading session at $0.4361, up 2.3949%, on 7,902 volume. The average volume for the last 3 months is 1,500 and the stock's 52-week low/high is $0.2672/$0.4399.
Intermap Technologies (ITMSF)
We reported earlier on Intermap Technologies (ITMSF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Intermap Technologies Corp (OTCQX: ITMSF) (TSE: IMP) FRA: 19T1) is a geospatial intelligence firm that is engaged in the provision of a range of geospatial solutions and analytics.
The firm has its headquarters in Englewood, Colorado and was incorporated in 1919. It operates as part of the software-application industry, under the technology sector. The firm serves consumers around the globe, with a focus on those in the Asia Pacific, the United States and Europe.
The company is dedicated to innovation and a drive to see that its clients get the geospatial solutions needed to transform their businesses.
The enterprise’s Data-as-a-Service (DaaS) solutions include NEXTMap One, which provides precision, 3D geospatial data at an unprecedented 1-meter resolution; InsitePro, a configurable insurance underwriting software; and NEXTView, a configurable data solution that delivers terrain and obstacle awareness that enhance airborne safety. It also offers geospatial data acquisition and production services; software solutions and services; and value-added data licensing. Its geospatial solutions are used in various applications, including risk assessment, location-based information, engineering, utilities, geographic information systems, global positioning systems maps, renewable energy, oil and gas, environmental planning, hydrology, land management, transportation, wireless communications, 3D visualization and outdoor advertising.
The firm recently entered into a contract to supply 3D digital elevation models (DEMs) to reduce crop loss for a leading agricultural firm in Indonesia, a move that will bring in additional revenues for the firm but also open it up to new growth and investment opportunities.
Intermap Technologies (ITMSF), closed Wednesday's trading session at $0.4001, off by 1.0095%, on 1,500 volume. The average volume for the last 3 months is 1,000 and the stock's 52-week low/high is $0.2573/$0.7397.
Hestia Insight (HSTA)
We reported earlier on Hestia Insight (HSTA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Hestia Insight Inc. (OTCQB: HSTA) is engaged in the provision of strategic consulting and capital market advisory services for micro, small and medium-sized firms in the biotech, healthcare and fintech sectors.
The firm has its headquarters in Las Vegas, Nevada and was incorporated in 2003, on November 19th. Prior to its name change in May 2019, the firm was known as Luxshmi Investments Inc. It operates as part of the software-application industry, under the technology sector. The firm primary serves consumers in the United States.
The company mainly operates through its wholly owned operating subsidiaries; Hestia Investments Inc. (Hestia Investments) and HSTA Health Inc., doing business as Hestia Vending. Hestia Investments offers strategic consulting, marketing support, medical supply sales, management and capital markets advisory services for select firms. On the other hand, Hestia Vending operates within the beverage, healthy food and wellness products industry and the smart vending machine industry. The company makes strategic acquisitions of emerging growth companies with sciences and technologies. It also intends to pursue the acquisition and development of healthcare-related technologies in the healthcare and biotech sectors through acquisition, licensing, or joint ventures. The company provides sales and marketing guidance and services and capital markets advisory services to its clients.
The enterprise recently appointed an additional member to its advisory board, who has broad experience in healthcare industry startups, to assist in its growth. This move will not only help bolster its overall growth but also help create value for its shareholders.
Hestia Insight (HSTA), closed Wednesday's trading session at $0.32625, even for the day. The average volume for the last 3 months is 185,650 and the stock's 52-week low/high is $0.03/$3.10.
GoviEx Uranium (GVXXF)
We reported earlier on GoviEx Uranium (GVXXF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
GoviEx Uranium Inc. (OTCQX: GVXXF) (CVE: GXU) (FRA: 7GU) (LON: 0UYS) is a mineral resource firm focused on acquiring, exploring for and developing uranium projects in Africa.
The firm has its headquarters in Vancouver, Canada and was incorporated in 2006 by Govind Friedland. Prior to its name change in September 2008, the firm was known as Govi High Power Exploration Inc. It operates as part of the uranium industry, under the metals and mining sub-industry, in the energy sector. The firm serves consumers around the globe.
The company's flagship property is the Madaouela project located in north-central Niger. This project holds one of the biggest uranium resources in the world and is located roughly 10 km south of Arlit and Areva’s mining subsidiaries of Somair and Cominak, in north-central Niger. It also owns 100% interest in the Mutanga project, which comprises of 3 mine permits situated to the south of Lusaka, Zambia. This project is located about 200 km south of Lusaka, immediately north of Lake Kariba, at elevations of 500 meters to 960 meters. The Mutanga Project’s mining licenses include 13880-HQ-LML, 12634-HQ-LML and 13881-HQ-LML, encompassing about 720 km2. The company also owns 100% interest in the Falea project, a highly compelling and prospective polymetallic uranium, copper and silver deposit with surrounding gold. The project consists of 3 licenses; Madini, Bala and Falea.
The enterprise, which recently provided an update of its operations, remains focused on achieving its goal of becoming a leading producer of uranium while also making a positive impact for its stakeholders and harnessing technology to minimize its environmental impact.
GoviEx Uranium (GVXXF), closed Wednesday's trading session at $0.1156, off by 5.0903%, on 185,650 volume. The average volume for the last 3 months is 260 and the stock's 52-week low/high is $0.1052/$0.3049.
Polydex Pharmaceuticals (POLXF)
Zacks, MarketBeat, QualityStocks and Marketbeat.com reported earlier on Polydex Pharmaceuticals (POLXF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Polydex Pharmaceuticals Limited (OTC: POLXF) is a company focused on developing, manufacturing and marketing biotechnology-based products for the human pharmaceutical market globally.
The firm has its headquarters in Toronto, Canada and was incorporated in 1979, on June 14th by Thomas C. Usher. Prior to its name change in March 1984, the firm was known as Polydex Chemicals Ltd. It operates as part of the biotechnology industry, under the healthcare sector. The firm serves clients around the globe.
The enterprise manufactures bulk pharmaceutical intermediates for the veterinary pharmaceutical industry. It primarily provides dextran and derivative products, including Dextran Sulphate, a specialty chemical derivative of dextran used in biotechnology applications and the pharmaceutical industry; and Iron Dextran, a derivative of dextran that is injected into pigs at birth as a treatment for anemia. It also develops Ushercell, a high molecular weight cellulose sulphate for topical vaginal use primarily in the prevention and transmission of AIDS and other sexually transmitted diseases, as well as unplanned pregnancies. Further, the enterprise supplies ferric hydroxide and hydrogenated dextran solutions. It sells its products through independent wholesalers and distributors, as well as directly to companies. Geographically, the sale of its products can be seen amplifying in the regions of Canada, the United States and Europe. The United States region makes up the larger share of revenue.
The company recently entered into an arrangement agreement with BioSpectra Inc. and BioSpectra Canada Limited, which will generate value for its shareholders.
Polydex Pharmaceuticals (POLXF), closed Wednesday's trading session at $1.9, up 0.063198%, on 260 volume. The average volume for the last 3 months is 99,357 and the stock's 52-week low/high is $0.5501/$1.919.
Kandi Technologies Group Inc. (KNDI)
Green Car Stocks, MarketClub Analysis, InvestorPlace, QualityStocks, Schaeffer's, The Street, StockMarketWatch, Hit and Run Candle Sticks, StreetInsider, TraderPower, Greenbackers, Jason Bond, Alternative Energy, GreatStockPix, Wall Street Resources, China Stock Alerts, MarketBeat, BUYINS.NET, Investing Futures, Marketbeat.com, Money Morning, Penny Stock Rumble, ProfitableTrading, TradersPro, SmarTrend Newsletters, StreetAuthority Daily, Trades Of The Day, TradingMarkets, TopStockAnalysts, FeedBlitz, Energy and Capital, Dynamic Wealth Report, DrStockPick, Money and Markets, CRWEWallStreet, Street Insider, CRWEPicks, CRWEFinance, CoolPennyStocks, ChartAdvisor, Weekly Wizards, BullRally, BestOtc, Barchart, Daily Trade Alert, StockEgg, Profit Confidential, PennyTrader Publisher, PennyToBuck, PennyStockVille, PennyOmega, PennyInvest, SmallCapNetwork, SmallCapVoice, HotOTC, Stock Traders Chat, INO.com Market Report, MadPennyStocks, StockHotTips, InvestorsUnderground, Investors Alley, StockRich, InvestorGuide, Investor Ideas, Rick Saddler and Willy Wizard reported earlier on Kandi Technologies Group Inc. (KNDI), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
In last week’s first quarter earnings conference call, General Motors CEO Mary Barra announced that the company intends to discontinue making its electric Chevy Bolt vehicles prior to the year’s end. Most of General Motors’ electric car sales so far have been made up of the Chevy Bolt EV and EUV, a bigger version of the vehicle. The batteries in the cars, however, are an old design and chemistry compared to those found in the carmaker’s more recent electric models, such as the Cadillac Lyriq and GMC Hummer, which are built on GM’s Ultium platform.
Barra announced that an assembly facility in the suburbs of Detroit, where Chevy Bolts have been assembled since 2016, will be modified in order to start producing GMC Sierra trucks and the electric Chevy Silverado, both of which are scheduled to be manufactured beginning in 2024.
In 2016, General Motors put the Bolt in front of Tesla’s Model 3. It was expected that these two vehicles, with an initial cost of around $35,000, would be among the longest-range electric vehicles ever created for regular drivers.
But as the total number of electric vehicles sold decreased to negligible levels outside of Tesla, Bolt sales have failed to catch on, contrary to many executives’ hopes. In addition, the Bolt has been hit hard lately, with General Motors recalling every one of its Bolts manufactured because of an electrical malfunction in a supplier’s battery that resulted in some fires.
In a statement, Chevy spokesperson Cody Williams said the company would introduce a number of new electric vehicles this year on the basis of the Ultium platform in important market sectors, including the Silverado Blazer and Equinox electric vehicles. General Motors is expecting its forthcoming electric vehicles to be launched much more quickly than the luxury Hummer and Cadillac Lyriq models, which, in comparison to its conventional automobiles, have been rolled out at a very slow pace.
According to Barra, there would be almost three times the number of jobs and a production capacity to build 600,000 electric vehicles annually once the Orion facility in Michigan, which presently manufactures the Bolts, resumes and becomes fully operational.
In an effort to catch up with Tesla, the leader in the EV sector, General Motors has set itself the goal of producing a million electric vehicles in the United States and China each year.
The Chevrolet Bolt vehicles are the most affordable electric vehicles to be offered in America, with a starting price of below $30,000. General Motors’ forthcoming EV Equinox is anticipated to have a comparable starting price when it debuts this autumn.
With such an affordable starting prices for its EVs, GM has made its intentions known, and the onus is now on startups such as Kandi Technologies Group Inc. (NASDAQ: KNDI) to boost their production efficiencies so that they can compete with or even outcompete legacy carmakers like General Motors.
Kandi Technologies Group Inc. (KNDI), closed Wednesday's trading session at $3.16, even for the day, on 101,181 volume. The average volume for the last 3 months is 743,353 and the stock's 52-week low/high is $2.00/$3.30.
HIVE Blockchain Technologies Ltd. (HIVE)
QualityStocks, InvestorPlace, MarketClub Analysis, MarketBeat, StreetInsider, Early Bird, StockMarketWatch, Marketbeat.com, CryptoCurrencyWire, Greenbackers, Hit and Run Candle Sticks, Barchart, Stock Market Watch, WealthMakers, StockOodles, StreetAuthority Daily, The Night Owl, The Online Investor, TopStockAnalysts, Wall Street Resources and smartOTC reported earlier on HIVE Blockchain Technologies Ltd. (HIVE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
During a panel discussion at Consensus 2023, legal professionals emphasized the value of coordinating global regulatory frameworks for cryptocurrencies while also taking regional variations into account.
Marianne Bechara, a senior counsel at the IMF, pointed out that it would be unwise to treat all cryptocurrencies the same without considering the context. Brian Yeoh, director of fintech at Abu Dhabi Global Markets, noted there is some coordination, but he said there is room for local jurisdictions to address their specific circumstances.
For several years, regulators have been trying to figure out how to best coordinate global crypto regulations. As a result, the European Union parliament passed the MiCA legislation last month, which some analysts have said might serve as a model for other nations. The MiCA regulations, which apply to service providers and trade brokers, are designed to safeguard investors and consumers while promoting financial stability and innovation in the crypto sector. The rules will go into effect between the middle of 2024 and the beginning of 2025.
Nonetheless, the panelists noted that every nation or region has problems that are unique to that place and thus should develop crypto regulations that best suit its people. The IMF and FSB are expected to propose worldwide guidelines for cryptocurrency in September 2023. In the meantime, individual countries are moving ahead with crypto regulations.
The United States has so far taken a split stance on crypto regulation. While some legislators are pushing for a thorough new regulatory framework to be developed to allow the sector to flourish, financial market regulators are tightening up on worries over scams, money laundering and cybercrime.
In March 2022, the Biden administration issued an executive order requesting that federal officials evaluate the broad hazards and opportunities presented by cryptocurrency. Recently, SEC Chair Gary Gensler noted that investment advisors cannot use cryptocurrency platforms and lenders as qualified custodians if they are not subject to the same regulations as stock exchanges.
In the United Kingdom, the situation is the same. Rules have generally encouraged the development of the crypto industry, but authorities are starting to crack down due to worries about fraud and money laundering. The country’s main financial regulator, the FCA, even banned all activities by Binance, the largest cryptocurrency exchange in the world, in 2021.
The recent failures in the industry, such as the demise of FTX, the third-largest crypto exchange in the world, and the subsequent arrest of its CEO, Sam Bankman-Fried, have highlighted the need for regulation of the crypto industry.
The suggestion that the different authorities across the globe need to work together to develop regulations for the cryptocurrency space is one that could have the support of industry actors such as HIVE Blockchain Technologies Ltd. (NASDAQ: HIVE) (TSX.V: HIVE) since it would provide clarity regarding the rules that these companies must adhere to in the different markets in which they have an interest.
HIVE Blockchain Technologies Ltd. (HIVE), closed Wednesday's trading session at $3.02, off by 3.5144%, on 743,353 volume. The average volume for the last 3 months is 433,371 and the stock's 52-week low/high is $1.36/$8.00.
The QualityStocks Company Corner
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX)
- HeartBeam Inc. (NASDAQ: BEAT)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
- Aditxt Inc. (NASDAQ: ADTX)
- Advanced Container Technologies Inc. (OTC: ACTX)
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF)
- IGC Pharma Inc. (NYSE American: IGC)
- Mullen Automotive Inc. (NASDAQ: MULN)
- Genprex Inc. (NASDAQ: GNPX)
- Sharing Services Global Corporation (SHRG)
McEwen Mining Inc. (NYSE: MUX) (TSX: MUX)
The QualityStocks Daily Newsletter would like to spotlight McEwen Mining Inc. (NYSE: MUX) (TSX: MUX).
To view the event, visit https://ibn.fm/KRNjC
To view the full press release, visit https://ibn.fm/LeMbi
McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) is an asset rich diversified gold and silver producer in the Americas and has a large exposure to copper through its subsidiary, McEwen Copper, owner of the Los Azules copper deposit in Argentina, believed to be the 9th largest undeveloped copper resource in the world.
Led by a management team with a track record of success, MUX owns and operates mines in some of the most prolific gold producing regions in the Americas. In recent months, the company has undertaken strong actions to lower production costs and increase production across its portfolio of gold assets, driving some costs below the industry average. Gold and copper prices are forecast to enter a major uptrend over the next couple years. McEwen Mining is laying the groundwork to capitalize on this opportunity now.
Seldom is management so aligned with investors’ interests with a commitment to the company’s success. CEO Rob McEwen maintains a 17% ownership stake in McEwen Mining and a 15% ownership in McEwen Copper with a combined cost base of roughly $220 million. McEwen founded Goldcorp, where he took the company from a market capitalization of $50 million to over $8 billion, and that same vision led MUX to create McEwen Copper.
For McEwen Mining shareholders, the company’s 68% stake in McEwen Copper is expected to be a gamechanger, turbocharging MUX by creating the world’s next copper unicorn.
McEwen Copper
Most mined copper is currently used in infrastructure, with new critical demand emerging for use in the electrification of transportation and the global energy transformation. The price of copper rose from a low of about $2 per pound two years ago to over $4 per pound today, and strong demand is expected to continue to soar. A study by S&P Global, titled The Future of Copper: Will the Looming Supply Gap Short-circuit the Energy Transition?, projects global copper demand to nearly double over the next decade, from 25 million metric tons today to about 50 million metric tons by 2035. Based on current trends, S&P Global projects annual supply shortfalls to reach nearly 10 million metric tons in 2035.
McEwen Mining is a 68% shareholder in McEwen Copper, holder of a 100% interest in the Los Azules copper project in San Juan, Argentina, which was ranked the 9th largest undeveloped copper deposit in the world by Mining Intelligence (2022). Its current copper resources are estimated at 10.2 billion pounds at a grade of 0.48% Cu (Indicated category) and an additional 19.3 billion pounds at a grade of 0.33% Cu (Inferred category). McEwen Copper also owns a copper exploration project in Nevada, called Elder Creek.
In a 2017 Preliminary Economic Assessment (PEA), Los Azules was estimated to have a 36-year life, but indications are that the project could ultimately become an even larger mine, with a longer life, since in the assessment, only 55% of the known copper resources are to be mined. Numerous drill holes have shown strong copper mineralization extending below the PEA pit bottom. Its average annual production for its first 13 years was pegged at 415 million pounds of copper in the 2017 PEA – enough copper to supply 2.2 million electric vehicles per year.
In August 2022, McEwen Copper closed its non-brokered, private placement offering of $82 million, after securing a $25 million investment from mining giant Rio Tinto’s technology arm, Nuton LLC. This gives McEwen Copper an imputed value of $258 million, which would give McEwen Mining’s 68% interest a value of approximately $3.70/share. Additional value can be attributed to McEwen Mining’s 1.25% net smelter royalty on both the Los Azules and the Elder Creek projects.
“We completed an $82 million financing for McEwen Copper in a very tough equity market. Rio Tinto, the second largest mining company in the world, through its subsidiary Nuton, now owns 9.7% of McEwen Copper, a result of its investment of $25 million,” Rob McEwen stated in a news release. “Also, Nuton is testing the Los Azules copper mineralization to see if it can accelerate and increase copper recoveries. Another of Rio Tinto’s subsidiaries, Kennecott Exploration, signed an option to earn a 60% interest in McEwen Copper’s other copper project, Elder Creek, by spending $18 million on exploration.”
The Elder Creek project is prospective for porphyry copper and gold mineralization and is well situated in a district hosting several large copper and gold mines, including Marigold, Lone Tree and Phoenix. Kennecott Exploration will be the operator of the exploration program. McEwen Mining holds a 1.25% net smelter return (NSR) royalty on the Elder Creek property.
Following the capital raise, McEwen Copper is well-funded to advance its Los Azules Project. Publication of an updated PEA on the Los Azules copper project is planned for Q1 2023. In Q2, an IPO is planned, along with MUX completing a secondary offering, assuming no further private placements in the interim. MUX is strategically reducing its interest to increase its treasury, in order to reduce debt and fund the further development of its gold and silver mines.
McEwen Copper currently has an implied market cap of over $258 million, based on its most recently completed financing. However, when its Los Azules copper project is compared with other recent transactions and market valuations of copper projects in the same region, it appears very undervalued.
MUX’s management believes its ownership stake in McEwen Copper is not currently reflected in the share price of the company. In fact, it is management’s belief that the combined value of its 68% interest in McEwen Copper, plus its gold mines and portfolio of mineral royalties, represents a share value ranging from a low of $8 to a high of $30 per share. Rob McEwen provides a full breakdown of this valuation estimate in a news release detailing the company’s Q3 2022 results.
Gold & Silver Projects
The Fox Complex
McEwen Mining owns a 100% stake in the Fox Complex in the heart of a prolific gold district in Timmins, Canada.
“When MUX bought the Fox Complex, in late 2017, it was a distressed asset with a history of high operating cost/oz. While it has taken longer than I expected, the cost to produce an ounce of gold is significantly lower,” CEO Rob McEwen stated in a news release. “I am pleased to say that in Q3 our cash cost/oz at Fox fell to $774, our lowest since mid-2018. This is well below the industry average. With our mine operating much more efficiently, our next important area to improve at Fox is the process plant (mill). Specifically, we need to increase the throughput because our mine is now producing more ore than our mill can process. As a result, we have a large surface stockpile of ore equivalent to more than two months of production.”
This ore stockpile contains approximately 10,000 ounces of gold representing a potential source of $12 million in free cash flow.
Located in one of the most prolific gold production areas in the world, along the Destor-Porcupine Fault Zone within the Abitibi Greenstone Belt, the Fox Complex includes the Black Fox mine and Froome mine which together have, so far, produced in excess of 1,000,000 ounces of gold. Also, it includes the Grey Fox and Stock deposits that have an estimated additional 1,600,000 ounces in reserves and resources. The 2.7-billion-year-old Abitibi Greenstone Belt, formed by ancient volcanic activity, has proved to be one of the world’s richest and most abundant gold regions, boasting total gold content of over 300 million ounces.
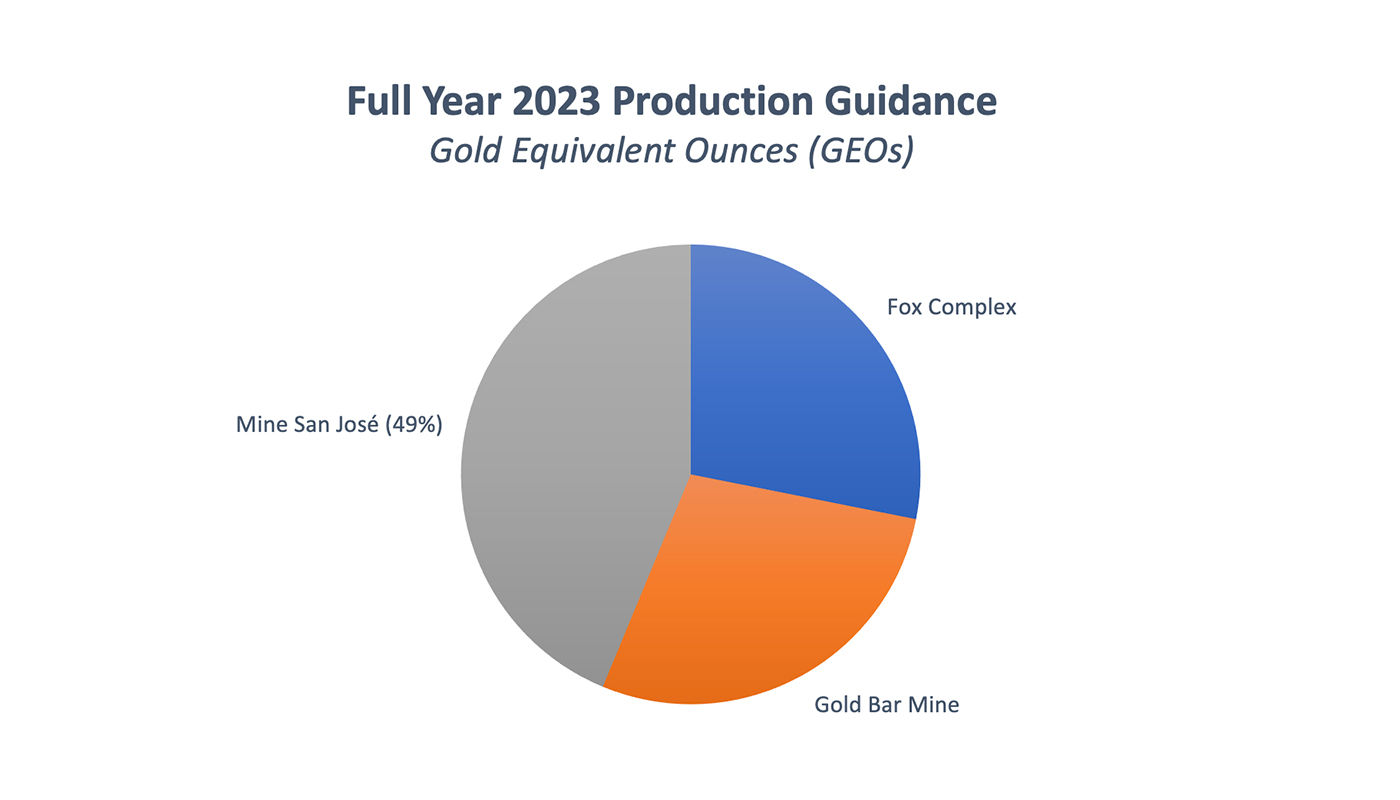
Full year 2023 guidance for The Fox Complex puts production estimates at 45,000 GEOs, 28% of MUX’s total production.
The Gold Bar Mine
McEwen Mining owns a 100% stake in the Gold Bar mine located in an area well known for gold production, the southern Roberts Mountains of the Battle Mountain-Eureka-Cortez gold trend in Eureka County, Central Nevada. The Gold Bar mine is on the same geological structure some 25 miles south of Nevada Gold Mines, a joint venture of Barrick and Newmont. This Cortez-Goldrush complex contains estimated reserves and resources of greater than 50 million gold ounces. Its annual gold production is 1,000,000 ounces.
Gold Bar had been previously mined, between 1991 and 1994, producing 134,000 gold ounces. MUX built a new facility in 2019. The open pit mine was expected to be a large contributor to MUX’s revenue and gold production, however operating challenges arose that reduced gold production and drove cost/oz unacceptably high. Mining activities have shifted recently to a nearby, satellite deposit called Gold Bar South (GBS). Going forward the expectations are higher gold production and lower operating cost/oz as a result of mining a higher ore grade (concentration of gold per ton) and having to move half the amount of material to capture an ounce of gold.
“At Gold Bar, we are looking forward to starting to mine our GBS deposit this quarter,” McEwen said in a November 2022 release. “We are expecting to have a much lower cost/oz than our YTD cost because we will be mining higher grade ore at GBS, with half the strip ratio and no problematic carbonaceous material.”
The Gold Bar Mine will account for approximately 28% of McEwen Mining’s 2023 total attributable production, with guidance pegged at 45,000 GEOs. Most of Gold Bar production in 2023 will be from GBS.
El Gallo/Fenix
Project Fenix is the proposed redevelopment plan for McEwen Mining’s El Gallo Complex in Mexico. There is a long history of mining in this region. MUX’s involvement began in 2013 operating it as an open pit, heap leach mine which produced 281,000 gold equivalent ounces at average cash cost of $655 per ounce. However, due to the transition to deeper sulfide mineralization that is not amenable to heap leaching, mining activities ceased in the second quarter of 2018. The redevelopment envisions constructing a mill at the existing mine site that will initially reprocess the existing heap leach material then transition to open pit mining and processing the sulphide mineralization. The company recently acquired a complete process plant on very advantageous terms that has considerably reduced the projected capital requirements for the project.
CEO Rob McEwen stated in a news release, “This acquisition has made Fenix more attractive to build and could provide a new long life mine for McEwen Mining.”
The initial development approach is to build a mill to reprocess the material on the heap leach pad and produce approximately 17,000 oz of gold annually for eight years. Construction of the Fenix project is expected to be completed by early 2024.
Mine San José
McEwen Mining is a 49% owner and non-operator of the San José gold and silver mine located in Santa Cruz province, Argentina. This high-grade underground mine has been operating since 2007 and currently has an expected life of six years with a reserve grade of 342 gpt silver and 5.7 gpt gold and a resource grade of 427 gpt silver and 7.0 g/t gold.
“The San José mine, where we have a 49% interest, put in a strong quarter and its exploration is continuing to extend its high-grade veins and discover new veins,” McEwen noted in a news release.
Production guidance for 2023 for MUX’s 49% is 70,000 GEOs, 44% of MUX’s total production. As a minority shareholder in the mine, MUX equity accounts for its investment in San Jose, and it receives 49% of the dividends from the mine’s free cash flow.
Market Outlook
Mining stocks took a beating in the wake of the COVID-19 pandemic. However, that could change, as many analysts are now forecasting a gold bull market in 2023.
“The operating challenges we faced in recent years have severely damaged our credibility with our shareholders and the market. As a result, few investors have taken a close look recently at our assets,” Rob McEwen said in a news release. “If they did, I believe some would see the potential value that I see today… I believe there is considerable potential value in MUX, and that is a big reason why I have a personal financial commitment of $220 million in MUX and McEwen Copper.”
Management Team
Robert R. McEwen is Chairman, CEO and Chief Owner of McEwen Mining. He has been associated with the gold industry all his career, with his first 18 years in the investment industry and, since 1990, as CEO of several gold mining companies. He founded Goldcorp and took that company from a $50 million market capitalization to more than $8 billion. He owns 17% of McEwen Mining and is in complete alignment with investors – the cost of his investment in MUX and McEwen Copper is $220 million and he takes an annual salary of only $1. He was awarded the Order of Canada and the Queen Elizabeth’s Diamond Jubilee Award, was inducted into the Mining Hall of Fame, was named an Ernst and Young Entrepreneur of the Year and has Honorary Doctor of Law degrees from York University and Western University.
William Shaver is interim COO and a Director of McEwen Mining. He has decades of management and executive experience in mine design, construction and operations. He was a founder of Dynatec Corporation, which became one of the leading contracting and mine operating groups in North America. In 2013, he was recognized as Ernst and Young Entrepreneur of the Year. Most recently, he served as COO of INV Metals. He is a Professional Engineer with a B.Sc. in Mining Engineering from Queens University.
Perry Ing is interim CFO at McEwen Mining. He has 25 years of experience in the Canadian mining industry. Over the past 15 years, he has held positions as CFO of Mountain Province Diamonds, Kirkland Lake Gold and McEwen Mining. Prior to that, he worked at Barrick Gold and Goldcorp and started his career in the mining practice at PwC. He has a Bachelor of Commerce from the University of Toronto and is a Chartered Professional Accountant in Canada and Certified Professional Accountant in the U.S.
Adrian Blanco S. is the company’s Director – America and Mexico Operations. He has extensive international experience in several industrial sectors and has held executive positions in Mexico, the United States, Peru and Argentina. He joined the McEwen Mining team in 2015 and has led a successful business transformation toward operational discipline, best business practices and financial profitability at subsidiaries Compañia Minera Pangea and McEwen Mining Nevada. He graduated from an Executive Management Program at IPADE and Harvard Business School.
Michael Meding is Vice President and General Manager of McEwen Copper. He has over 20 years of international experience, primarily with major mining companies such as Barrick Gold and Trafigura, including extensive experience with project development and operations in Argentina. While at Barrick Gold’s Veladero mine in Argentina, Mr. Meding played a key role in the turnaround, extension of the mine life and subsequent strategic partnering with Shandong Gold. He holds an MBA from Indiana University in Pennsylvania and an MBA from the Leipzig Graduate School of Management in Germany.
McEwen Mining Inc. (NYSE: MUX), closed Wednesday's trading session at $8.71, up 1.2791%, on 434,334 volume. The average volume for the last 3 months is 137,638 and the stock's 52-week low/high is $2.81/$10.00.
Recent News
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Announces Results Conference Call for Q1 2023
- MiningNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Positioned as Observers Expect Copper to Become the 'New Precious/Industrial Metal'
- MiningNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Eyes 'Exciting Results' from Exploration and Delineation Program at Los Azules Copper Project
HeartBeam Inc. (NASDAQ: BEAT)
The QualityStocks Daily Newsletter would like to spotlight HeartBeam Inc. (NASDAQ: BEAT) .
To view the full press release, visit https://ibn.fm/CRFN8
HeartBeam Inc. (NASDAQ: BEAT) is a cardiac technology company that has developed the first and only 3D-vector 12-lead electrocardiogram (ECG) platform for heart attack detection anytime, anywhere. The company’s proprietary ECG telehealth technology aims to redefine the way high risk cardiovascular patients are diagnosed in ambulatory and acute care settings. HeartBeam’s initial focus is on providing diagnostic data to help physicians with care management of patients with cardiovascular disease.
In August 2022, HeartBeam announced that it submitted its HeartBeam AIMI™ software for approval from the U.S. Food and Drug Administration (FDA). HeartBeam AIMI is a platform technology to improve the speed and accuracy of heart attack detection in acute care settings. The company expects FDA approval by the end of 2022, and a full commercial roll-out of HeartBeam AIMI is targeted for Q1 2023.
HeartBeam sees submission of its first product based on its platform technology as an important milestone toward commercialization, which underscores the company’s continued progress toward making the HeartBeam AIMI platform widely available to help emergency department physicians quickly and accurately identify a heart attack.
While the FDA conducts its regulatory review, HeartBeam will focus on executing key components of its commercialization plan and subscription revenue model. It will also continue to engage in discussions with strategic institutions, including academic centers, regional healthcare systems and regional community hospital systems that can utilize HeartBeam products.
The company is based in Santa Clara, California.
Products
HeartBeam’s development portfolio includes two products:
- HeartBeam AIMI is software that provides a 3D comparison of baseline and symptomatic 12-lead ECG to more accurately identify a heart attack in acute care settings and, as noted above, has been submitted for FDA approval; and
- HeartBeam AIMIGo™, the first and only credit card-sized 12-lead output ECG device coupled with a smartphone app and cloud-based diagnostic software system for remote heart attack detection.
HeartBeam is developing AIMIGo, a medical-grade detection and monitoring technology for use in remote heart attack detection, thereby allowing physicians to diagnose a patient’s heart attack as it occurs, even if the patient is not at a medical facility. The company’s system, once approved by the FDA, can be used by patients at home or almost anywhere and anytime to help their physicians assess whether chest pain is the result of a heart attack or another cause. While approximately 82% of chest pain ED visits are unnecessary, patients delay approximately 3 to 4 hours after symptoms begin, increasing mortality rates by 40%. The company’s goal is to shorten the time to treatment outside of the medical facility to improve patients’ well-being.
HeartBeam’s AIMIGo is a powerful, portable and easy-to-use prescription-based product. It comprises a smartphone app, a credit card-sized ECG device placed on a patient’s chest, the HeartBeam cloud platform, and a digital portal for the physician to view ECG results and direct patient action. For the first time outside of a medical setting, HeartBeam AIMIGo enables patients and their clinicians to determine if symptoms are due to a heart attack, quickly and easily, so care can be expedited, if needed.
Pending FDA clearance, AIMIGo is initially intended to be available by prescription, and is reimbursable under existing remote patient monitoring codes (RPM codes). This provides a new revenue stream to physicians who before did not have a way to monitor these high-risk patients. The RPM codes provide a monthly reoccurring revenue stream to the company, as well. On average, at current reimbursement rates, the practice will receive $1,300+ per year per patient they monitor, and the company will receive $600 per year per patient from this RPM reimbursement.
Market Overview
Adoption rates of telehealth services increased dramatically in recent years, with the COVID-19 pandemic serving as a major driver of growth. Among the areas seeing the greatest expansion are cardiology, radiology, behavioral health and online consultation.
Encouraging this growth, governments are actively developing new policies and reimbursement guidelines to promote the use of digital health platforms. The U.S. Centers for Medicare & Medicaid Services (CMS), for example, has recently expanded reimbursement for telehealth services. U.S. market growth is also being driven by the rising prevalence of chronic conditions and the growing geriatric population.
Remote heart attack detection is a previously unsolved problem with a massive and underserved market that is several times larger than the $2 billion total addressable market (TAM) in the U.S. for ECG cardiac arrhythmia monitoring.
Approximately 8 million Americans have suffered at least one heart attack, and a total of 18 million have been diagnosed with coronary artery disease (CAD). Based on these figures, HeartBeam projects a total addressable U.S. market TAM valued at $10 billion annually for its AIMIGo solution for remote heart attack monitoring of CAD.
Management Team
Branislav Vajdic, Ph.D., Chief Executive Officer and Founder of HeartBeam, Inc, combines over 30 years of experience in technology development and senior management positions. Dr. Vajdic has been deeply involved with the development of HeartBeam’s technology to fit his vision for the company. Prior to HeartBeam, from 2007 to 2010, Dr. Vajdic was CEO and Founder of NewCardio, a publicly traded company in the cardiovascular devices space. From 1984 to 2007, Dr. Vajdic was at Intel, where he held various senior management position. At Intel, Dr. Vajdic was the designer of first Flash memory and two key inventions that enabled Flash as a product and led engineering groups responsible for Pentium 1 through Pentium 4 designs. Dr. Vajdic was awarded two Intel Achievement Awards, the highest level of award for outstanding contributions to Intel. Dr. Vajdic is author of numerous patents and publications in the fields of cardiovascular devices, as well as chip design. Dr. Vajdic holds a Ph.D. in Electrical Engineering from the University of Minnesota.
Jon Hunt, Ph.D., has over 35 years’ experience in the medical/medical device industry with extensive domestic and international experience in general management, clinical/regulatory, sales and marketing. He also has diverse experience in Fortune 500 companies, as well as start-up environments. Dr. Hunt was the Vice President of Clinical Science and Technology, Medical Device Innovation Consortium, from July 2019 to July 2021, and Vice President of Clinical and Regulatory Affairs, Cryterion Medical from January 2018 to June 2019 (acquired by Boston Scientific Corporation in July 2018 for $202M). Dr. Hunt was the Founding President and CEO of Bardy Diagnostics, Inc. from October 2013 to November 2017 (acquired by Hill-Rom Holdings, Inc.). Prior to joining Bardy Diagnostics, Dr. Hunt spent the previous 11 years as the Vice President of Clinical & Regulatory Affairs with Cameron Health, Inc. (acquired by Boston Scientific Corporation). Dr. Hunt spent the previous 10 years with Cardiac Pacemakers, Inc., St. Jude Medical and Cardiac Pathways Corporation. Dr. Hunt began his career with Cardiac Pacemakers, Inc. (now Boston Scientific Corporation) as the Director of Clinical Programs. He subsequently held positions at St. Jude Medical in Clinical Affairs and as the Business Unit Director for the Cardiac Rhythm Management division for Europe, the Middle East and Africa. At Cardiac Pathways Corporation, Dr. Hunt held various executive positions as Vice President of International Sales and Marketing and Vice President of Worldwide Sales and Marketing (acquired by Boston Scientific Corporation). Dr. Hunt received his Ph.D. in Motor Control from The Pennsylvania State University, his Master’s from California State University, Long Beach and his undergraduate degree from Keele University in the United Kingdom.
Rick Brounstein, HeartBeam’s Chief Financial Officer, combines over 30 years of experience in health technology senior management. Since 2017, Mr. Brounstein has been and is currently a partner of Hardesty, LLC, a financial services firm, and Mr. Brounstein is currently a managing director of CTRLCFO, LLC, a firm Mr. Brounstein founded in 2016 to support funded start-ups in life science and technology. From 2008 to 2011, Mr. Brounstein was Chief Financial Officer of NewCardio, Inc., a microcap public company in the cardiology space, and, over his career, he has been with nine other companies in life science or technology, holding positions including Chief Financial Officer, Chief Operating Officer, Treasurer and Accounting Manager. From June 2001 through November 2007, Mr. Brounstein held several positions at Calypte Biomedical Corporation, a publicly traded medical device company, including Chief Financial Officer and Executive Vice President. In January 2007, Mr. Brounstein was appointed as the National Member Representative for the 2007 COSO Monitoring Project, which published new guidelines for monitoring internal financial controls in February 2009; Mr. Brounstein subsequently was a member of the FEI task force that issued the updated COSO Internal Control Framework in 2013. In March 2005, Mr. Brounstein was appointed to the SEC Advisory Committee on Smaller Public Companies. Mr. Brounstein earned his Certified Public Accountant (CPA) certification while working at Arthur Andersen LLP, formerly a public accounting firm. Mr. Brounstein holds a B.A. in accounting and an M.B.A. in finance, both from Michigan State University.
Ken Persen, HeartBeam’s Chief Technology Officer, combines over 28 years of experience in the medical device and digital health industries in engineering and senior management positions. Mr. Persen has been involved in several companies in Cardiac Rhythm Management, holding positions including Chief Executive Officer, Chief Technology Officer, Executive Vice President and Director of Engineering. Since 2016 and prior to joining HeartBeam, Mr. Persen was the Chief Technology Officer at LIVMOR, Inc., a digital health company. In addition, from 2016 through November 2021, he was also Chief Executive Officer of LIVMOR. Prior roles included Director of Engineering at Cameron Health (acquired by Boston Scientific), a late-stage medical device start up, and engineering and management positions at Guidant Corp. (acquired by Boston Scientific), a large medical device manufacturer. He has an undergraduate degree from University of Minnesota, Duluth, with a BA in Computer Science.
HeartBeam Inc. (NASDAQ: BEAT), closed Wednesday's trading session at $2.31, up 12.6829%, on 137,638 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $10.00/$.
Recent News
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Completes $25M Capital Raise to Achieve Upcoming Milestones, Extend Cash Runway
- BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Poised to Offer Timely Solution amid a 'Smoldering Public Health Crisis'
- BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Poised to Enable Expedited Care for Heart Attack Patients
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
To view the full press release, visit https://ibn.fm/Cz8C7
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Wednesday's trading session at $1.92, up 26.3158%, on 9,058,826 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.6105/$11.304.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Retains Analytics Firm, Investigates Possible Naked Short Selling
- New Gel Cures Brain Tumors in Mice
- InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) to Participate, Present at Upcoming Virtual Investor Spotlight Event
Aditxt Inc. (NASDAQ: ADTX)
The QualityStocks Daily Newsletter would like to spotlight Aditxt Inc. (NASDAQ: ADTX).
Data from the
Aditxt Inc. (NASDAQ: ADTX) is a biotech innovation company developing technologies focused on mapping and reprogramming the immune system. Aditxt’s immune mapping technologies are designed to provide a personalized immune profile. Aditxt’s immune reprogramming technologies, currently preclinical, are being developed to retrain the immune system to induce tolerance to address rejection of transplanted organs, autoimmune diseases, and allergies.
As further discussed below, the company’s first commercial product is an immune mapping technology, AditxtScore™, which is designed to provide a personalized profile of the immune system.
The company’s preclinical immune reprogramming technology, Apoptotic DNA Immunotherapy™ (“ADi™”), aims to retrain the immune system to induce tolerance, with the goal of addressing vast unmet needs in transplanted organ rejection, autoimmune diseases, and allergies. The company is developing specific ADi™ products for psoriasis, type 1 diabetes, and skin grafting.
Headquartered in Richmond, Virginia, Aditxt also operates locations in Silicon Valley and New York.
AditxtScore™
AditxtScore™ is a proprietary platform designed to provide a personalized, comprehensive profile of an individual’s immune system. The underlying technology, licensed from Stanford University through an exclusive worldwide agreement, offers a highly sensitive and accurate method of detecting and quantifying cellular responses, allowing greater specificity, quantification, and amplification of both clinical and commercial opportunities.
The company’s first commercial application of the platform, AditxtScore™ for COVID-19, delivers timely reports on vulnerability and immune status relating to SARS-CoV-2 and its known variants, giving consumers and physicians the data needed to make informed health decisions. Potential future applications will offer early detection of an array of conditions, including diabetes, cardio-metabolic maladies and hormonal imbalances.
Aditxt’s AditxtScore™ immune monitoring center in Richmond, Virginia, is operational and designed to support the anticipated increased demand for AditxtScore™ as well as related products and services. The company is currently scaling its capabilities at this location, with a goal of processing up to 10 million immune system tests/reports annually.
ADi™
ADi™ is Aditxt’s immune reprogramming platform addressing disease-causing immune responses while maintaining the immune system’s ability to combat pathogenic infection. The company is commercializing a nucleic acid-based technology called Apoptotic DNA Immunotherapy™ (ADi™) which utilizes a novel approach that mimics the way our bodies naturally induce tolerance to our own tissues (therapeutically induced immune tolerance). Aditxt believes its ADi™ technology platform can be engineered to address a wide variety of indications.
Aditxt is currently developing ADi™ products for psoriasis, type 1 diabetes and skin grafting.
Currently, immuno-tolerance is achievable through chimerism and cell-based therapy, but there is a clinical need for a more practical and cost-effective approach which:
- Can be made into a product
- Does not require additional hospitalization
- Is simple to produce and ship
Preclinical studies have demonstrated that ADi™ treatment significantly and substantially prolongs graft survival, in addition to successfully “reversing” other established immune-mediated inflammatory processes. ADi™ treatment is not expected to require hospitalization, instead being delivered as an injection in minute amounts into the skin.
IP Portfolio
Both AditxtScore™ and ADi™ are supported by a strong IP portfolio.
AditxtScore™, built upon initial technology invented, licensed from and used at Stanford University, is protected by U.S. patents encompassing methods, systems, and kits for detection and measurement of specific immune responses.
ADi™ technology is protected by seven patent families, including:
- 8 U.S. patents
- 4 pending U.S. patent applications
- 86 foreign patents and 14 pending foreign patent applications spanning the EU, Australia, Canada, Japan, China, India and Hong Kong
These patents are broadly categorized into three groups:
- Autoimmune diseases and Type 1 Diabetes
- Organ transplantation and a method of producing plasmid DNA to prevent immune activation
- Composition of matter for a tolerance delivery system for antigens of interest
Aditxt also possesses and/or in-licenses substantial know-how and trade secrets relating to the development and commercialization of its product candidates, including related manufacturing processes and technologies.
Market Overview
The potential market opportunities presented by immune monitoring and reprogramming are extensive, particularly as Aditxt continues to evaluate additional applications for the platforms.
The company’s initial focus on organ transplantation and related autoimmune response provides some insight into the potential of its approach. According to BCC Research, the global organ and tissue transplantation and alternatives market is on course to reach $120.3 billion by 2024, recording a CAGR of 7.4% from 2019. Industry data suggest that approximately 50% of all transplanted organs are rejected within 10-12 years, further highlighting the critical need for a practical, cost-effective solution to harmful autoimmune responses.
Through its focus on the COVID-19 testing market with AditxtScore™, Aditxt demonstrated the wide-ranging potential of its portfolio. Fortune Business Insights estimated the global COVID-19 diagnostics market at $48.64 billion for 2022. While demand for COVID-19 diagnostics is expected to lessen in the coming years, Aditxt will be uniquely positioned to leverage its existing infrastructure stemming from these operations as the company works to advance broader applications for the AditxtScore™ platform.
Leadership Team
Amro Albanna is the Co-Founder, Chairman, and CEO of Aditxt. He has founded multiple startups to commercialize innovations in various industries, including healthcare, enterprise software, telecommunications, nano technology, consumer health, and biotech. Mr. Albanna has led numerous M&A and going-public transactions as a founder, co-founder, and senior executive.
Shahrokh Shabahang, D.D.S., MS, Ph.D., is the company’s Co-Founder, Chief Innovation Officer, and a member of its board. He brings to the team more than 20 years of experience in developing and commercializing life science technologies focused on product and clinical development in the fields of microbiology and immunology.
Corinne Pankovcin, CPA, MBA, is the President of Aditxt. Prior to joining Aditxt, Ms. Pankovcin served as CFO for several world class organizations, including Business Development Corporation of America, Blackrock Kelso Capital and AIG Capital Partners. In these roles, Ms. Pankovcin was responsible for executing portfolio investments and managing significant M&A transactions.
Thomas Farley is the Chief Financial Officer of Aditxt. From December 2015 to June 2020, Mr. Farley was the Controller and Treasurer of Business Development Corporation of America (“BDCA”), a publicly listed business development company. Prior thereto, from January 2011 to August 2015, Mr. Farley was the Senior Controller of Blackrock Capital Investment Corporation (NASDAQ: BKCC). Prior to joining BlackRock Capital Investment Corporation, Mr. Farley was a Senior Controller for PineBridge Investments Emerging Markets practice. Mr. Farley was also an Accounting Manager for Bessemer Venture Partners prior to his tenue at PineBridge. Mr. Farley began his career with PricewaterhouseCoopers LLP, from 1996 to 2001. Mr. Farley earned his B.S. in Accounting from Long Island University and is a Certified Public Accountant.
Rowena Albanna is the company’s Chief Operating Officer. Ms. Albanna has over two decades of experience in senior leadership roles for both technology startups and public companies. Ms. Albanna’s experience spans a wide variety of industries, including biotechnology, insect control, nanotechnology, consumer electronics, financials, telecommunications, e-commerce, online marketing, medical, and defense.
Matthew Shatzkes is the Chief Legal Officer and General Counsel of Aditxt. As a former partner at an AM Law 50 law firm, Mr. Shatzkes advised a wide variety of healthcare related entities, including biotech companies, on corporate, regulatory, and strategic business matters. Mr. Shatzkes will oversee all aspects of the legal functions at Aditxt, including, providing advice and counsel on governance, regulatory matters, strategic alliances, mergers and acquisitions, and commercial transactions.
Aditxt Inc. (NASDAQ: ADTX), closed Wednesday's trading session at $0.6053, up 2.5932%, on 79,244 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.5804/$28.49.
Recent News
- Aditxt Inc. (NASDAQ: ADTX) - Stem Cell Transplantation Shows Promise in Treating Autoimmune Disorders
- Organ Donation, Transplantation: Challenges and How We Can Help More Individuals
- InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Closes on $1.9M Registered Direct Offering
Advanced Container Technologies Inc. (OTC: ACTX)
The QualityStocks Daily Newsletter would like to spotlight Advanced Container Technologies Inc. (OTC: ACTX).
A proposal that would enable
Advanced Container Technologies Inc. (OTC: ACTX) is in the business of selling and distributing self-contained, automated, indoor “micro-farms” called Grow Pods, along with related equipment and supplies. Additionally, the company designs and sells patented proprietary medical-grade plastic containers, known as the Medtainer®, that store and grind pharmaceuticals, herbs, teas and other solids or liquids.
ACTX is the leading distributor of Grow Pods. With a controlled environment, food and herbs can be grown without pesticides, harmful chemicals or risk of pathogen contamination, and with low energy consumption. Restaurants, grocery stores, non-profits, MSOs and entrepreneurs can use Grow Pods to ensure a fresh supply of ultra-clean produce year-round.
The company entered the Grow Pod business in October 2020 with its acquisition of all shares of Advanced Container Technologies Inc., a California corporation. As of February 28, 2022, ACTX is exploring the acquisition of the assets and the assumption of some or all of the liabilities of GP Solutions Inc., the developer and manufacturer of Grow Pods, for which ACTX is currently the sole U.S. distributor.
Because Grow Pods can be located almost anywhere, produce can be grown closer to the point of consumption and harvested at its peak, providing nutritious fruits and vegetables where needed. Indoor micro-farms, utilizing a practice known as vertical farming, have attracted the attention of governments and universities, which are now promoting vertical farming as a way to combat food insecurity and inequities.
The United States Department of Agriculture (USDA) has stated that vertical farming “is no longer a futuristic concept.” The department is enthusiastic about vertical farming, particularly those utilizing repurposed shipping containers, such as Grow Pods. Arizona State University reports that vertical farming reduces water use by 90 percent compared to conventional farming but produces 10 times the crop yield.
Products
Grow Pods
One of the company’s main business units is focused on selling advanced, self-contained hydroponic containers called Grow Pods. These unique and innovative automated systems are essentially micro-farms that can be placed virtually anywhere and, with their controlled and specially filtered environment, allow cultivation of a wide variety of crops, 365 days a year. The Grow Pod controlled environment offers major advantages for the production of high-value crops. The ability to grow year-round and the ability to cultivate in a smaller footprint using less water and power are some of the primary advantages of the system. Grow Pods offer constant temperature, humidity and airflow control, as well as automated watering and lighting schedules for optimal growth and minimal labor requirements, regardless of crop.
Containers
ACTX meets the needs of the pharmaceutical and medical markets, including the cannabis and hemp industries, with patented packaging systems. The company designs, customizes, brands and sells proprietary medical grade plastic containers that can store pharmaceuticals, herbs, teas and other solids or liquids, with a special built-in feature that can grind solids and shred herbs. The company’s flagship container product is the patented Medtainer®, a child resistant, medical-grade herb container and grinder that is water-tight, air-tight and smell proof. Packaging in the cannabis industry is critical, with numerous stringent regulations about how cannabis products must be packaged and labeled. ACTX also offers custom-branded, compliant vacuum seal bags and other retail container solutions.
Equipment and Supplies
ACTX markets and sells two principal products: Grow Pods, which are specially modified insulated shipping containers manufactured by GP Solutions Inc., in which plants, herbs and spices may be grown hydroponically in a controlled environment, and Medtainers®, which may be used to store pharmaceuticals, herbs, teas and other solids or liquids and can grind solids and shred herbs. The company also markets and sells various products related to Grow Pods and the Medtainer®, as well as providing private labeling and branding services for purchasers of Medtainers® and certain related products.
GP Solutions manufactures and sells other products, such as humidity controllers and LED lighting systems for vertical farming. The company’s specially designed lighting panels are programmed to emit the exact wavelength of light that each crop requires. The system has a daybreak-to-nightfall feature that gives plants the proper chromatic signals to grow rapidly and fruitfully. High efficiency LED light strips supply the crops with a red and blue light spectrum required for photosynthesis in the spectrum that plants need most.
Market Overview
The global vertical farming market is expected to reach $33.02 billion by 2030, according to a new report by Grand View Research. The market is forecast to expand at a CAGR of 25.5 percent from 2022 to 2030, according to Grand View. Escalating production of biopharmaceutical products, including cannabis, is anticipated to drive the market. The building-based segment of the market is expected to register a significant CAGR of 27.8 percent over the projected period. In addition, the climate control segment is expected to see high growth.
The global cannabis packaging market is expected to reach $14.34 billion by 2028, according to analysis by Reports and Data. The analysis forecasts 1,700 percent growth in cannabis users by the end of 2026, with packaging likely observing a whopping 26.42 percent growth in the forecast period. There are significant barriers to entry in the cannabis packaging market, giving an advantage to companies already established in the sector. These barriers include developing a thorough knowledge of the myriad regulations that govern cannabis packaging (which differ in each state), and child-resistance requirements.
Management Team
Douglas P. Heldoorn is the Founder and Chairman of Advanced Container Technologies Inc. He also holds the positions of President, CEO and COO at the company. Mr. Heldoorn has served on the Board of Directors since its inception in 2013. He has also previously held the position of Executive General Manager at Nissan Motor Corp.
Jeffory A. Carlson is CFO and Treasurer of ACTX. Mr. Carlson has also served as the company’s Corporate Controller since 2014.
Advanced Container Technologies Inc. (OTC: ACTX), closed Wednesday's trading session at $0.3, even for the day, on 1 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.073/$1.00.
Recent News
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Maine Legislator Files Anticipatory Bill to Facilitate Cannabis Interstate Commerce
- InvestorNewsBreaks - Advanced Container Technologies Inc. (ACTX) GrowPods Ideal Options for Academic Research, Education
- 420 with CNW - Australia's Medical Marijuana Market May Soon Surpass Canada's
Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF)
The QualityStocks Daily Newsletter would like to spotlight Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF).
A new archaeological study has found that Romans in 47 AD had
Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) is a mineral exploration company engaged in advancing precious and base metal deposits in the state of Arizona. Its flagship copper-gold-zinc-silver asset is the Kay Mine Project, located in Yavapai County. The company also owns Sugarloaf Peak gold project in La Paz County.
The company in October 2022 received permit approval from the Bureau of Land Management (BLM) for two new drill pads, located approximately 1,200 meters west of the Kay Mine Deposit. These new pads will allow for testing of the company’s Western Target, while also allowing for drilling of additional coincident anomalies located between the Central and Western Targets. Construction of the drill road for the Central Target (located 500 meters west of the Kay Mine Deposit) is currently underway, with drilling expected to begin in November 2022. Road construction for the Western Target will begin upon confirmation of BLM acceptance of the company’s posted bond, with drilling expected to commence in Q1 2023.
The company is fully funded, with $60 million in cash as of June 30, 2022, to complete the remaining 18,000 meters planned for the Phase 2 program at Kay, as well as an additional 76,000 meters in the Phase 3 program (budgeted at $27 million), which will be used to test the numerous parallel targets heading west of the Kay Deposit, as well as the northern and southern extensions of the Kay Deposit.
Arizona Metals Corp. is based in Toronto, Canada.
Projects
Arizona Metals Corp. owns 100% of the Kay Mine property in Yavapai County, which is located on a combination of patented and BLM claims totaling 1,300 acres that are not subject to any royalties. An historic estimate by Exxon Minerals in 1982 reported a “proven and probable reserve of 6.4 million short tons at a grade of 2.2% copper, 2.8 grams per ton gold, 3.03% zinc, and 55 grams per ton silver.” The historic estimate has not been verified as a current mineral resource. None of the key assumptions, parameters, and methods used to prepare the historic estimate were reported by Exxon, and no resource categories were used. Significant data compilation, re-drilling and data verification may be required by a “qualified person” (as defined in National Instrument 43-101 – Standards of Disclosure for Mineral Projects) before the historic estimate can be verified and upgraded to be a current mineral resource. A qualified person has not done sufficient work to classify it as a current mineral resource, and Arizona Metals is not treating the historic estimate as a current mineral resource.
The company also owns 100% of the Sugarloaf Peak Property in La Paz County, which is located on 4,400 acres of BLM claims. Sugarloaf is a heap-leach, open-pit target and has a historic estimate of “100 million tons containing 1.5 million ounces (of) gold” at a grade of 0.5 grams per ton. The historic estimate at the Sugarloaf Peak Property was reported by Westworld Resources in 1983. The historic estimate has not been verified as a current mineral resource. None of the key assumptions, parameters, and methods used to prepare the historic estimate were reported, and no resource categories were used. Significant data compilation, re-drilling and data verification may be required by a qualified person before the historic estimate can be verified and upgraded to a current mineral resource. A qualified person has not done sufficient work to classify it as a current mineral resource, and Arizona Metals is not treating the historic estimate as a current mineral resource.
Market Opportunity
The World Gold Council, an industry association representing gold producers with hundreds of mining operations in nearly 50 countries around the world, reports that global demand for gold during the first six months of 2022 was 2,189 tons, a 12% increase in demand over the same period in 2021. Demand came primarily from gold bar and coin investors, jewelry consumers, central bank purchases to bolster currency reserves and technology manufacturing.
The average price per ounce for the period was $1,871, marking a 1% year-over-year increase. The council reported gold mine production for the period was up 3% over 2021 at 1,764 tons. For the remainder of 2022 and into 2023, the council projects flat gold demand with possible slight increases in gold mine production. The council notes that unpredictable geopolitical factors, the Ukraine war for example, and likelihood of global economic slowdown could have significant near-term impact on gold demand and prices.
Management Team
Marc Pais is President and CEO of Arizona Metals. He previously founded and served as President of Telegraph Gold (listed as Castle Mountain Mining), which was acquired by Equinox Gold, a TSX-listed mining company. He has seven years of experience as a Mining Analyst, with a focus on precious metals development companies. He holds a B.Sc. in Geological Engineering (Mineral Exploration) from Queen’s University in Canada.
David Smith is the Vice President, Exploration of Arizona Metals. He has 30 years of global precious metals exploration experience, including codiscovery of the Solidaridad/La Sabila deposit in Mexico with deposits estimated at 1 million ounces of gold. His core areas of expertise are managing mineral projects from acquisition to exploration, resource modeling and mineral project development. He holds an M.Sc. from the University of Oregon and an MBA from Pinchot University/Presidio Graduate School.
Paul Reid is the Executive Chairman of Arizona Metals. He previously founded and served as Executive Chairman of Telegraph Gold (listed as Castle Mountain Mining), which was acquired by Equinox Gold, a TSX-listed mining company. Paul has extensive experience as an Investment Banking professional, involved in raising capital, go-public transactions, and advisory services.
Vision Energy Corp. (OTCQX: AZMCF), closed Wednesday's trading session at $2.79, off by 2.2767%, on 43,343 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.30/$4.73.
Recent News
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Research Shows Ancient Romans Missed Huge Silver Deposit in Germany
- Zinc Production to Recover in 2023 Though Scheduled Mine Closures Could Dampen Results
- InvestorNewsBreaks - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Intersects Broad Interval of VMS Mineralization in First Drill Hole at Western Target
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
•Zacks recently released a research report covering LexariaBioscience, a global innovator in drug delivery platforms
•The report discusses the company’s ongoing research and development(“R&D”) programs, forecasting penetration into global marketsfor hypertension, oral nicotine delivery, and antiviral productcategories
•Lexaria is targeting conditions with significant market potential,including hypertension, diabetes, dementia, and seizures
•These programs position the company for entry into collaborativeopportunities to support further growth and provide growth capital
Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing.
DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future.
Lexaria has a collaborative research agreement with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has filed for patent protection for specific delivery of nicotine, vitamins, NSAIDs, testosterone, estrogen, cannabinoids, terpenes, PDE5 inhibitors (with brand names like Viagra), tobacco and more.
Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats.
The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products.
DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions.
DehydraTECH Technology
Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following:
- Speeds up delivery – the effects of the product are felt by the subject in just minutes.
- Increases bioavailability – the technology is much more effective at delivering a drug or product into the bloodstream.
- Increases brain absorption – animal testing suggests significant improvement in the quantity of drug delivered across the blood-brain barrier.
- Improves drug potency – more of the ingested product is made available to the body, so lower doses are required to achieve the desired effect.
- Reduces drug administration cost – lower doses mean lower overall drug costs.
- Masks unwanted taste – the technology eliminates or reduces the need for sweeteners.
Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery.
Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine.
Market Outlook
Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments:
- Oral Cannabinoids – a market estimated to be worth $18.4 billion in 2021 and expected to reach $46.2 billion by 2025.
- Antivirals – an estimated $52.1 billion market in 2021 that’s expected to grow to $66.7 billion by 2025.
- Oral Mucosal Nicotine – smokeless tobacco products, a $13.6 billion market in 2018, is forecast to grow at 7.2 percent annually through 2025.
- Human Hormones – estrogen and testosterone replacement therapies represented a $21.9 billion market in 2019, with a forecast CAGR of 7.7 percent through 2027.
- Ibuprofen and Naproxen – NSAID sales totaled $15.6 billion globally in 2019 and are projected to reach $24.4 billion by 2027.
- Vitamin D3 – the global market size was $1.1 billion in 2021, growing at 7 percent per year and expected to reach $1.7 billion in 2026.
Management Team
Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations.
John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents.
Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations.
Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch.
Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia.
Lexaria Bioscience Corp. (LEXX), closed Wednesday's trading session at $1.62, off by 12.9032%, on 45,777 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.62/$3.60.
Recent News
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX), Featured in Recent Research Report by Zacks SCR
- Lexaria Bioscience Corp.'s (NASDAQ: LEXX) Successful DIAB-A22-1 Diabetes Study Indicates Just One of Several Potential Applications for DehydraTECH(TM) Drug Delivery Platform Technology
- InvestorNewsBreaks - Lexaria Bioscience Corp. (NASDAQ: LEXX) Awards CRO Contract for Upcoming Hypertension Study
Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF)
The QualityStocks Daily Newsletter would like to spotlight Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF).
Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) featuredamong companies participating inTHE Mining Investment Event of the North ("THE Event"), Québec City, June 19-21, 2023, which is to be heldat the Fairmont le Château Frontenac and Voltigeurs de QuébecArmoury. THE Mining Investment Event of the North is pleased to announce the launch of THE Student SponsorshipProgram, made possible through the generous support of Maple GoldMines and JDS Group of Companies. The program provides anunparalleled learning opportunity to 50 university or collegestudents studying in the fields of mining, finance, engineering,metallurgy, earth/social sciences and/or other related miningcourses over the course of three days at THE Event. Participatingstudents will have access to valuable insights from industryexperts, panels, and company presentations as well as theopportunity to network with professionals in all areas the miningindustry. The program also includes a special Meet and GreetDinner, where they will be able to engage directly with miningsector industry leaders. Students will also participate in a casestudy competition with a focus on innovation and sustainability inthe mining sector, with the winning team receiving a prize to beawarded on the final day of THE Event by the Sponsors.
Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) is advancing the next generation of nickel-cobalt sulfide projects to deliver the metals needed to power the electric vehicle (EV) revolution and feed the high growth stainless steel market. The company is one of only a few new sources of potential supply outside Indonesia and China.
Canada Nickel possesses industry leading nickel expertise and is focused on low risk, well established mining jurisdictions. The company has launched wholly owned subsidiary NetZero Metals Inc. to develop zero-carbon production of nickel, cobalt and iron and has applied in multiple jurisdictions to trademark the terms NetZero Nickel, NetZero Cobalt and NetZero Iron. Canada Nickel is also pursuing development of processes to allow net zero carbon production of these elements.
Canada Nickel is currently anchored by its 100% owned flagship Crawford Nickel-Cobalt Sulfide Project with large-scale potential located in the heart of Ontario’s prolific Timmins-Cochrane mining camp, adjacent to major infrastructure.
The company believes the EV industry and many other consumer sectors have an urgent need for zero-carbon metal this decade, not in 20-25 years as contemplated by some resource companies. Canada Nickel also believes that nickel supplies from Indonesia and other Pacific island nations, typically controlled by Chinese-owned companies, are not the answer for batteries needed by GM, Ford and the European automakers working to develop and manufacture EV models.
The company is headquartered in Toronto.
Crawford Nickel-Cobalt Sulfide Project
The Crawford Nickel-Cobalt Sulfide Project is the largest sulfide discovery since the early 1970s and contains the fifth-largest nickel sulfide resource in the world, based on Measured & Indicated resources, according to the latest update. The Crawford project is expected to be one of the largest base metal mines in Canada based on results of a Preliminary Economic Assessment. Early projections by Canada Nickel estimate that the project has the potential to produce 50,000 tons of nickel per year. The company is now in the final stages of completing the project’s feasibility study.
The project is projected to produce 2.8 tons of CO2 per ton of nickel equivalent production, which is 89% lower than the industry average of 34 tons of CO2 per ton of nickel equivalent production.
The company is taking significant steps toward developing the Crawford project as a net zero carbon producer. In addition to harnessing the natural ability of the project’s geology to act as a carbon sink through spontaneous reaction of the host rock once exposed to atmospheric conditions called mineral carbonation, Canada Nickel has discovered a new way to enhance carbon capture, termed In Process Tailings (IPT) Carbonation. This act of conditioning the tailings with a concentrated stream of carbon dioxide before deposition has been demonstrated at lab scale to achieve carbon capture at a rate 8-12 times faster than naturally occurring sequestration, achieving more than 60% of the capture that had previously taken six days.
These latest results move the company further toward production of Net Zero Nickel™ and generation of 21 tonnes of CO2 credits per tonne of nickel, which would produce an estimated average of 710,000 tonnes of CO2 credits annually and 18 million total tonnes of CO2 credits over the expected life of mine. IPT Carbonation does not require complex new technologies and major process modifications and could encourage the development of a net zero carbon industrial cluster centered around the Crawford project.
Canada Nickel in January 2023 announced that its latest test work results support the incorporation of carbon capture and storage into the Crawford project. The company believes that utilization of existing process streams should allow IPT to be efficiently engineered and incorporated into the project’s flowsheet, with an integrated feasibility study for the project expected in the second quarter of 2023.
In December 2022, Canada Nickel announced its engagement on Deutsche Bank Securities Inc. (“Deutsche Bank”) and Scotiabank – two of the world’s leading investment banks with a broad base of mining and industrial expertise – as financial advisors for the equity component of the project financing for the Crawford project. In the same release, the company announced the completion of another significant permitting milestone by filing the detailed project description with the Impact Assessment Agency of Canada. Canada Nickel targets receipt of permits by mid-2025, with construction to immediately follow.
Additional Projects
The Reid Nickel Property is located just 16 kilometers southwest of Crawford, or 37 kilometers northwest of Timmins, and contains an ultramafic body with a target geophysical footprint of 3.9 square kilometers. Preliminary assay results from Canada Nickel’s summer/fall drilling program confirm the presence of mineralized dunite, as well as currently undefined higher-grade sections. Partial assay results confirm expected nickel grades. Nickel mineralization in serpentinized dunite was found in all 16 holes drilled to date.
The Sothman Nickel Property is located 70 kilometers south of Timmins. Five drill holes on the eastern half of the target anomaly confirmed the continuation of ultramafic lithologies, primarily peridotite, with moderate to strong serpentinization and variable amounts of mineralization throughout.
The company in December 2022 announced positive drilling results from its ongoing regional exploration campaign at its Reid and Sothman properties. These latest results continue to reinforce the success of Canada Nickel’s geophysical targeting approach and increase the probability of success at the company’s other 20-plus properties within its 42 square kilometers of geophysical targets.
Building on this momentum, Canada Nickel in December 2022 announced its entry into a deal to acquire a 100% interest in the past producing Texmont property situated between the company’s properties south of Timmins. As noted in the news release, the acquisition of the Texmont property provides near-term smaller scale production potential and is highly complementary to the company’s large-scale Crawford and regional nickel sulphide projects.
Market Opportunity
Global demand leaves the market fundamentally short of nickel in the medium- and long-term. Global primary nickel demand will likely reach 3 million tons in 2022, up from 2.4 million tons in 2020, according to the International Nickel Study Group (INSG).
The INSG says primary nickel production is forecast to hit 3.1 million tons in 2022. Indonesia, the world’s largest nickel miner, halted exports of unprocessed nickel ore in January 2020, due to a government-imposed ban. Indonesia has floated the concept of a nickel cartel whose member nations would exert influence over world nickel supply and prices, similar to OPEC’s pricing power over oil.
Benchmark Minerals, a leading EV supply chain research firm, projects that, by 2035, world demand for nickel will double from current levels to 6 million tons annually. That growing demand represents a need for new nickel production equivalent to 70 mines the size of Canada Nickel’s Crawford Project.
Management Team
Mark Selby is Chairman, CEO and Director of Canada Nickel. He was formerly President and CEO of RNC Minerals, where he led a team that successfully raised over $100 million and advanced the Dumont nickel-cobalt project from initial resource to a fully permitted, construction-ready project. He has held senior management roles with Quadra Mining, Inco and Purolator Courier, and was a partner at Mercer Management Consulting. Since 2001, he has been recognized as one of the leading authorities on the nickel market. He graduated from Queen’s University with a Bachelor of Commerce.
Wendy Kaufman is CFO of Canada Nickel. She has 25 years of experience leading publicly listed mining companies in project financing, capital structuring, capital markets, accounting and internal controls, tax, and financial reporting and public disclosure. She was also previously CFO at Khiron Life Sciences Corp. and held CFO and senior finance positions at Pasinex Resources Limited, Primero Mining Corporation and Inmet Mining Corporation. She holds a Bachelor of Business Administration from Wilfrid Laurier University and is a Chartered Professional Accountant.
Steve Balch is VP Exploration at Canada Nickel. He is an Ontario registered geoscientist with 32 years of experience in geophysics, specializing in magnetic and electromagnetic methods. He founded Triumph Instruments and developed the AirTEM system, a multi-coil helicopter-borne EM system that is in use worldwide. He has also been active in borehole geophysics and helped develop new technologies including north-seeking gyros, temperature compensated induction conductivity probes, UAV-based magnetometers and high sensitivity magnetic gradiometers.
Christian Brousseau is VP Capital Projects at Canada Nickel. He is a professional engineer (P.Eng) with over 30 years of experience in engineering, design and construction in the Canadian mining industry, including six years as Project Director for the Dumont Project and three years as the Engineering and Construction Manager for Detour Gold. Prior to Detour, he held various construction management positions at Osisko’s Malartic Project and at Goldcorp’s Éléonore Project. He also spent eight years at Falconbridge supervising and managing various capital projects.
Canada Nickel Company Inc. (OTCQX: CNIKF), closed Monday's trading session at $2.05, up 3.0151%, on 73,251 volume with 375 trades. The average volume for the last 3 months is 57,207 and the stock's 52-week low/high is $1.04999995/$5.63000011.
Recent News
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - THE Mining Investment Event of the North - Quebec City, June 2023 Announces THE Student Sponsorship
- InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Featured in Bell2Bell Podcast
- THE Mining Investment Event of the North - Quebec City, June 2023
IGC Pharma Inc. (NYSE American: IGC)
The QualityStocks Daily Newsletter would like to spotlight IGC Pharma Inc. (NYSE American: IGC).
Even though some experts claim the
IGC Pharma Inc. (NYSE American: IGC), through subsidiary IGC Pharma, develops, patents, and markets advanced THC-based drug formulations for the treatment of symptoms related to various diseases including but not limited to Alzheimer’s disease, Tourette syndrome, chronic pain, and pet seizures.
IGC’s leading drug candidate, IGC-AD1, has completed Phase 1 of a safety and tolerability trial and entered Phase 2 trials for treating agitation in patients with Alzheimer’s dementia, the first study in humans of a natural tetrahydrocannabinol (THC) compound plus another molecule (www.clinicaltrials.gov). As of September 2022, the IGC trial is the only ongoing Phase 2 trial of a natural THC-based formulation on Alzheimer’s patients.
The company’s other drug candidate, TGR-63, is an enzyme inhibitor that has shown in preclinical trials the potential to reduce neurotoxicity in Alzheimer’s cell lines. Both drug candidates have shown their ability to ameliorate beta amyloid plaques in Alzheimer’s cell lines and improve memory in Alzheimer’s mouse models. Beta amyloid plaques are a key hallmark of Alzheimer’s and an important target of Alzheimer’s pharmaceutical drug development.
Neuro Psychiatric Symptoms (NPS) are not only debilitating for Alzheimer’s patients; they also place an immense emotional burden on their caregivers. Beyond reducing symptoms, IGC-AD1’s active molecules and TGR-63 have also shown promise in preclinical trials to reduce important hallmarks of Alzheimer’s including plaques and tangles, as well as improving the treatment of memory loss.
Over the past eight years, the IGC team has amassed a deep knowledge of cannabinoid science, including extraction, isolation, purification, and development. The company’s strategy is to leverage its unique end-to-end capabilities, platform, and expertise to develop a class-leading program and bring it to market quickly and cost efficiently to treat neurodegenerative diseases such as Alzheimer’s.
The company also has a family of cannabidiol (CBD)-based consumer products (www.Holief.com) such as pain relief creams, pain relief gels, purpose gummies, tinctures, and capsules targeting women’s wellness, with a particular focus on premenstrual syndrome (PMS) and dysmenorrhea (period cramps). In addition, the company targets individuals that need sleep-aids with its specially formulated low melatonin cannabinoid gummies.
IGC has also introduced a low-calorie CBD- and caffeine-infused energy beverage brand (www.SundaySeltzer.com) that is currently available for purchase. The company’s brands are founded on the belief that effective natural solutions should be affordable and accessible to everyone. As the demand for natural products targeting women’s wellness and energy drinks continue to grow, these products are seeing strong traction in the market.
The company operates three facilities – a large GMP (Good Manufacturing Production Standards) certified facility that includes extraction, distillation, and manufacturing, in Washington State; a GMP-211 (pharmaceutical) grade facility in Maryland; and a facility licensed for controlled substances including cannabis in Bogota, Colombia, with complete access to legal licensed cannabis where the company conducts its testing.
In addition, the company’s development under Magistral Formulations is approved by INVIMA (Colombia National Food and Drug Surveillance Institute) to treat neurological disorders, non-oncological chronic pain, and mental disorders.
IGC’s intellectual property (IP) portfolio comprises of eight patents that it controls and seven patent applications. The portfolio includes #11,446,276, a patent for extreme low dose THC treatment of Alzheimer’s that was granted in September 2022.
The company is headquartered in Potomac, Maryland.
IGC-AD1
IGC-AD1 is the company’s leading drug candidate for the treatment and relief of Alzheimer’s symptoms. A significant amount of research on Alzheimer’s cell lines has shown that the active agents in IGC-AD1 reduce plaques and neurofibrillary tangles that are the hallmarks of Alzheimer’s. Further, micro-dosing of THC, as shown in cell lines, could increase the functioning of mitochondria and potentially promote the growth of new neural pathways (neurogenesis). The research shows that micro-dosing of THC affects the brain radically differently from the normal higher dosing of THC.
While there is a significant body of research showing that THC is neuro-toxic at normal levels of dosing, micro-dosing of THC has been shown to be non-toxic to neurons. With the results of these preclinical studies, the company developed an oral formulation, IGC-AD1. The company recently completed a safety and tolerability Phase 1 trial on Alzheimer’s patients and has initiated a Phase 2, multi-site, double-blind, randomized, placebo-controlled trial of the safety and efficacy of IGC-AD1 on agitation in participants with dementia due to Alzheimer’s disease at sites in the U.S. and Canada. IGC expects the Phase 2 trial to take between 9 and 12 months to complete, barring unknown factors such as, for example, a resurgence of COVID and the enforcement of lockdowns and travel restrictions.
With further successful trials and FDA approvals, IGC hopes to bring a drug based on natural THC as an effective treatment for agitation in Alzheimer’s to market.
TGR-63
The company’s other molecule, TGR-63, has been shown to reduce the neurotoxicity that impacts memory loss in preclinical trials with mice. On a dose dependent manner, transgenic Alzheimer’s mice treated with TGR-63 showed improvement in memory relative to control.
Both drug candidates, IGC-AD1 and TGR-63, have shown their ability to reduce the brain plaques associated with memory loss in Alzheimer’s in mice.
With further successful trials and FDA approvals, IGC hopes to bring TGR-63 as a treatment for Alzheimer’s disease to market.
Market Opportunity
Alzheimer’s disease impacts over 55 million people worldwide and about 5.5 million individuals in the U.S. Over 70% of these patients face debilitating symptoms, including anxiety, depression, and agitation (Mendez, 2021). Agitation in dementia patients can include excessive physical movement and verbal activity, restlessness, pacing, belligerence, aggression, screaming, crying, and wandering.
In 2020, the estimated healthcare costs for Alzheimer’s disease in the U.S. were $305 billion. Medicare and Medicaid covered about 70% of those costs, leaving considerable burden on patients and families. At the current rate of growth of Alzheimer’s and other dementia diagnoses, those costs are estimated to reach over $1 trillion by 2050.
Currently, there are no FDA-approved medications to alleviate the symptoms of dementia due to Alzheimer’s disease, providing a tremendous opportunity for formulations that can have an impact on quality of life and disease progression.
Management Team
Richard Prins has been chairman at IGC since 2012 and served as an independent director since 2007. From March 1996 to 2008, he was the Director of Investment Banking at Ferris, Baker Watts, Incorporated. Prins served in a consulting role to RBC until January 2009. He currently volunteers full time with a non-profit organization, Advancing Native Missions, and is a private investor. Since February 2003, he has been on the board of Amphastar Pharmaceuticals Inc. He holds a bachelor’s degree from Colgate University and an MBA from Oral Roberts University.
Ram Mukunda is CEO and President of IGC. He has been the chief inventor and architect of most of the company’s patent filings and is responsible for the company’s strategic positioning. Prior to IGC, he was founder and CEO of Startec Global Communications, which he took public in 1997. He served as Strategic Planning Advisor at Intelsat, a communications satellite services provider. From 2001 to 2003, he was a Council Member at Harvard’s Kennedy School of Government, Belfer Center of Science and International Affairs. He was named the 1998 Ernst & Young Entrepreneur of the Year. He holds bachelor’s degrees in electrical engineering and mathematics, and a master’s degree in engineering from the University of Maryland.
Dr. Jagadeesh Rao is the company’s Principal Scientist. His career spans two decades in the public sector and product R&D for Johnson & Johnson. He leads IGC’s scientists in the development of pharmaceutical and OTC products. He worked for the federal National Institutes of Health, and for the National Institute on Drug Abuse. His Ph.D. in Neurochemistry is from the National Institute of Mental Health & Neurosciences in India. He did postdoctoral training at the University of Illinois-Chicago.
Claudia Grimaldi is a Director, Vice President, Principal Financial Officer, and Chief Compliance Officer for IGC. She also serves as a Director/Manager Director for some of the company’s subsidiaries. She graduated with highest honors from Javeriana University in Colombia with a bachelor’s degree in psychology. She holds an MBA, graduating with highest honors, from Meredith College in North Carolina. In addition, she has attended the Darden School of Business Financial Management Executives program and the Corporate Governance Program at Columbia Business School. She is currently pursuing her Directorship Certification with the National Association of Corporate Directors. She is fluent in both English and Spanish.
IGC Pharma Inc. (NYSE American: IGC), closed Wednesday's trading session at $0.332, off by 3.4884%, on 77,544 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.2785/$0.8432.
Recent News
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Study Finds No Elevated Risk of Psychosis Resulting from Marijuana Use
- 420 with CNW - Coalition Urges Biden Administration to End Federal Prohibition, Deschedule Marijuana
- 420 with CNW - New Jersey's Marijuana Market Still Facing Issues Following Launch
Mullen Automotive Inc. (NASDAQ: MULN)
The QualityStocks Daily Newsletter would like to spotlight Mullen Automotive Inc. (MULN).
To view the full press release, visit https://ibn.fm/v6mGI
Mullen Automotive Inc. (NASDAQ: MULN) is a Southern California-based automotive company that owns and partners with several synergistic businesses working toward the unified goal of creating clean and scalable energy solutions. Mullen has evolved over the past decade in sync with consumers and technology trends. Today, the company is working diligently to provide exciting EV options built entirely in the United States and made to fit perfectly into the American consumer’s life. Mullen strives to make EVs more accessible than ever by building an end-to-end ecosystem that takes care of all aspects of EV ownership.
Commencement of Trading on Nasdaq
On November 5, 2021, Mullen announced its commencement of trading on the Nasdaq Capital Market.
“Today is a monumental day for Mullen Automotive. I am especially proud of our team, investors and all who have believed in Mullen and taken us to this point as a publicly traded company on the Nasdaq Capital Market,” David Michery, CEO and Chairman of Mullen Automotive, stated in the news release. “Trading on Nasdaq now opens us up to new investors, both institutional and retail shareholders, and broadens our awareness and company profile, while increasing awareness of Mullen and our technology platform and opening new opportunities in EV and beyond. The road ahead has never been brighter for Mullen, and I am proud to lead us into the future.”
The milestone came in the wake of the company’s stock-for-stock merger with Net Element Inc.
The Mullen FIVE
The Mullen FIVE EV Crossover, debuting at the Los Angeles International Auto Show (LAIAS) on November 17, 2021, embodies Mullen’s Southern California roots with an inspired design focused on two complementary Golden State themes – California landscape and California urban.
The FIVE is built on an EV Crossover skateboard platform that offers multiple powertrain configurations and trim levels in a svelte design that is Strikingly Different™ and exciting to experience in person.
Prior to the start of LAIAS, the Mullen FIVE was selected as a finalist by the LA Auto Show for Top EV SUV in the ZEVA “People’s Choice” Awards.
LAIAS provides Mullen an opportunity to display multiple variants of the FIVE model while also showcasing its powertrain, battery and charging technology. The company intends to bring the FIVE to market in 2024, and reservations are currently open here.
Mullen’s development portfolio also includes EV Fleet Vans, which it intends to bring to market in Q2 2022, and the pure electric, high performance Mullen DragonFLY.
Expansion of Manufacturing Capacity
On November 2, 2021, Mullen announced plans to expand its facility in Robinsonville, Mississippi.
Mullen’s Advanced Manufacturing and Engineering Facility (AMEC) currently occupies 124,000 square feet of manufacturing space. The total available land on the property is over 100 acres, and Mullen is moving ahead with plans to build out another 1.2 million square feet of manufacturing space to support class 1 and class 2 EV cargo vans and the Mullen FIVE EV Crossover.
On the expanded site, Mullen plans to build a body shop, a fully automated paint shop and a general assembly shop.
EV Market Outlook
The global EV market was reported to consist of 3,269,671 units in 2019, a figure that is expected to grow at a CAGR of 21.1% through 2030 to a total of 26,951,318 units worldwide. This market’s monetary value was estimated at $162.34 billion in 2019 and is expected to grow at a CAGR of 22.6%, resulting in an approximate value of $802.81 billion by 2027. The primary driver for this exponential growth is a worldwide increase in vehicle emissions regulations.
Management Team
David Michery is the CEO and Founder of Mullen and has been leading the company and its divisions since inception in 2014. With over 25 years of executive management, marketing, distressed assets, and business restructuring experience, Mr. Michery brings a wealth of relevant knowledge and expertise to the Mullen brand. He has notably created 12 trademarks so far to develop the company brand and vision.
Mr. Michery is working toward a sustainable future accessible to all by creating a suite of clean-energy electric vehicles at varied price points. With entirely U.S.-based manufacturing and operations, he is also determined to have Mullen Technologies play a role in shaping a self-sustaining local economy by creating more jobs in America.
Mr. Michery manages risks and company expectations as a pathway to success and has personally overseen several businesses that totaled over $1 billion in transactions. His key strength is the ability to be fiscally responsible and lead teams to complete projects on time and within budget. As a seasoned professional in this space, Mr. Michery has demonstrated skill in building businesses from the ground up and into successful entities that subsequently sold for hundreds of millions of dollars.
Mullen Automotive Inc. (MULN), closed Wednesday's trading session at $0.0639, off by 21.1111%, on 1,163,770,680 volume. The average volume for the last 3 months is 1.164B and the stock's 52-week low/high is $0.06/$1.71.
Recent News
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Receives Follow-Up Fleet Order from UNC Charlotte
- BDEW Says Germany Boosted EV Charging Network by 35% in 2022
- InvestorNewsBreaks - Mullen Automotive Inc.'s (NASDAQ: MULN) MAEO to Execute $680K Contract with the District of Columbia
Genprex Inc. (NASDAQ: GNPX)
The QualityStocks Daily Newsletter would like to spotlight Genprex Inc. (NASDAQ: GNPX).
Genprex Inc. (GNPX) is a clinical-stage gene therapy company developing potentially life-changing technologies for cancer patients based upon a unique proprietary technology platform, including Genprex’s initial product candidate, Oncoprex™ immunogene therapy for non-small cell lung cancer (NSCLC). Genprex’s platform technologies are designed to administer cancer-fighting genes by encapsulating them into nanoscale hollow spheres called nanovesicles, which are then administered intravenously and taken up by tumor cells where they express proteins that are missing or found in low quantities.
Research and Development
Genprex holds a portfolio of 30 issued and two pending patents covering its technologies and targeted molecular therapies. The company’s research and development program is focused on identifying and developing leading-edge gene therapies that can be used alone or in combination with other therapies for treatment of cancer.
Genprex’s initial product candidate is Oncoprex™, an immunogene therapy for the treatment of non-small cell lung cancer (NSCLC). Oncoprex works by interrupting cell signaling pathways that cause replication and proliferation of cancer cells, re-establishes pathways for apoptosis (or programmed cell death) in cancer cells, and modulates the immune response against cancer cells. Oncoprex has also been shown to block mechanisms that create drug resistance.
Preclinical research is being conducted with the goal of developing Oncoprex to be administered with targeted therapies in other solid tumors, and with immunotherapies in NSCLC and other solid tumors. In addition, Genprex has conducted and plans to continue research into other tumor suppressor genes associated with chromosome 3p21.3, as well as other potential applications of the company’s immunogene therapy platform.
Clinical Trials
Genprex is currently conducting the second phase of a phase I/II clinical trial at the University of Texas MD Anderson Cancer Center in Houston. The company plans to expand its clinical program by adding a new clinical study evaluating Oncoprex™ in combination with a checkpoint inhibitor for treatment of Stage IV or recurrent NSCLC. In research presented at the 2017 Annual Meeting of the American Association of Cancer Research in Washington, D.C., Genprex’s collaborators showed that TUSC2 in combination with PD-1 checkpoint inhibition has a significantly greater anti-tumor effect in lung cancer than either agent alone. The research also shows that TUSC2 in combination with PD-1 blockade has synergistic activity in upregulating natural killer (NK) cells, correlating with prolonged survival in mice.
TUSC2 (Tumor Suppressor Candidate 2) is a tumor suppressor gene that is absent or deficient in cancer cells of many different cancer types.
The Market
Genprex technologies seek to bridge a critical gap by combining with targeted therapies and immunotherapies to provide treatments to large patient populations who would otherwise not be candidates for those therapies or who have become resistant to them. Genprex technologies are being developed to overcome genomic limitations which are inherent in targeted therapies and immunotherapies in order to provide new treatment solutions to large cancer populations, such as those with lung cancer.
Each year, more people die of lung cancer than of colon, breast and prostate cancers combined. NSCLC is the most common type of lung cancer, accounting for about 85 percent of all lung cancers, according to the American Cancer Society (“ACS”). Despite radical advances in drug development and novel therapeutic standards, survival for late stage lung cancer has not improved significantly in the past 25 years.
Senior Management
Chairman and Chief Executive Officer J. Rodney Varner, JD, is a co-founder of Genprex and has served in these roles since August 2012. He has more than 35 years of legal experience with large and small law firms and as outside general counsel of a Nasdaq-listed company. Varner has served as counsel in company formation, mergers and acquisitions, capital raising, other business transactions, protection of trade secrets and other intellectual property, real estate, and business litigation. He is a member of the State Bar of Texas and has been admitted to practice before the U.S. Court of Appeals for the Fifth Court and the U.S. Tax Court.
Julien L. Pham, M.D., MPH, is president and chief operating officer of Genprex. In March 2013, Dr. Pham co-founded RubiconMD, a healthcare IT company that connects primary care providers to specialists for additional guidance and opinions on medical cases and served as its chief medical officer. He has served on the faculty at Harvard Medical School’s Brigham and Women’s Hospital and is a board-certified internal medicine doctor and nephrologist.
Ryan M. Confer, MS, has served as Genprex chief financial officer since September 2016. Confer has more than 10 years of executive experience in planning, launching, developing, and growing emerging technology companies and has served in the chief operating and chief financial roles for non-profit and for-profit entities since 2008. Confer has also served as an international business development consultant for the University of Texas at Austin’s IC2 Institute, where he focused on evaluating the commercialization potential of nascent technologies in domestic and international markets applicable to technology incubator programs associated with the University. Confer holds a BS in finance and legal studies from Bloomsburg University of Pennsylvania and an MS in technology commercialization from the McCombs School of Business at the University of Texas at Austin.
Jan Stevens, RN, is vice president of Clinical Operations. Stevens has nearly 20 years of comprehensive clinical operations experience in the biopharma industry and a specialization in early-to-late stage oncology companies. Stevens joined the company to help support the various clinical development programs for Oncoprex™.
Genprex Inc. (NASDAQ: GNPX), closed Wednesday's trading session at $0.801, up 2.666%, on 242,555 volume. The average volume for the last 3 months is 241,219 and the stock's 52-week low/high is $0.7358/$2.67.
Recent News
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Positive Data Supporting Therapeutic Potential of Its Non-Viral Delivery System
- InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Positive Research Findings, Additional Support for REQORSA(R) and Its Therapeutic Potential
- InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Provides Statement Regarding FDA Initiative to Accelerate Approval Process for Gene Therapies
Sharing Services Global Corporation (SHRG)
The QualityStocks Daily Newsletter would like to spotlight Sharing Services Global Corporation (SHRG).
Sharing Services Global Corporation (SHRG), formerly Sharing Services Inc., is a diversified company dedicated to maximizing shareholder value, operating through two primary subsidiaries: Elepreneurs Holdings, a direct-selling company, and Elevacity Holdings, a products company. Headquartered in Plano, Texas, SHRG markets and distributes Elevate-branded health and wellness products through an independent sales force of distributors called Elepreneurs.
Proprietary Products
SHRG’s current exclusive Elevate product offerings are marketed under the Elevacity brand, so named to signify the company’s commitment to elevating lives.
The Elevate health and wellness product line consists of nutraceutical products that SHRG refers to as D.O.S.E., which stands for dopamine, oxytocin, serotonin and endorphins – all of which are key hormones proven to promote happiness and well-being.
Elevacity brand products are carefully formulated, chosen and designed to support a single objective: elevate the happiness and well-being of the consumer.
Global Network of Elepreneurs
Elevacity products are shared and sold by a growing international network of home-based entrepreneurs, called Elepreneurs, operated by Elepreneurs Holdings. This SHRG subsidiary provides basic and advanced programs for both new and experienced entrepreneurs who are focusing on their direct-sales careers.
SHRG’s high-performing independent sales force follows the company’s Blue Ocean selling strategy, an approach that encourages individuals to seek new markets, lead, and to “stop competing and start creating.” The Blue Ocean strategy is based on the book, “Blue Ocean Strategy,” written by Professor Renée Mauborgne, who notes that “the lesson here is that the best defense is offense, and the best offense… is to make a blue ocean shift and create your own blue ocean.”
Following this selling strategy, SHRG’s Elepreneurs are taught that, rather than competing directly in a competitive, direct-selling market, they should focus on making competitors irrelevant and succeeding in an uncontested marketplace.
In addition, SHRG’s Elepreneurs use the interactive, video-based VERB sales-marketing platform developed by Verb Technology Company Inc. The app utilizes proprietary interactive video data collection and analysis technology and provides next-generation customer relationship management, lead generation, and video marketing software applications.
Continued Momentum as Industry Leader
These selling strategies have resulted in sharp and consistent revenue gains. In the company’s 10-Q filed with the SEC for the three months ended Oct. 31, 2019, SHRG reported sales of $38.8 million for fiscal Q2 2019, an increase of 116% over sales of $17.9 million reported for the comparable quarter of 2018. Consolidated gross profit jumped by $16.2 million to $27.4 million for the same period compared to Q2 2018.
SHRG’s consolidated operating earnings were $3.9 million in the fiscal quarter ended Oct. 31, 2019, compared to $866,802 for the comparable period the prior year. Consolidated gross margin also grew 70.9% for the three months ended Oct. 31, 2019, compared to 62.2% the prior year.
These numbers are continuing a trend established over the past two years. In fiscal Q1 2019, SHRG achieved revenues of $35.4 million, more than double that of the comparable period in 2018. Even earlier, the company reported sales of $85.9 million for fiscal year ended April 30, 2019. This represents a nine-fold increase, or $77.5 million jump, over the company’s revenues of $8.4 million the prior year.
These numbers bring SHRG’s sales revenues since December 2017 — when the company’s Elevate product line was released — to an impressive cumulative total of $169 million.
Preparing for Success
SHRG is well prepared to continue and accommodate for this growth. The company recently expanded its corporate footprint by moving to a 10,000-square-foot facility in Plano, Texas, that offers ample room to expand as the company grows and flourish. The larger corporate locale provides space for a growing customer service department, product fulfillment, opportunity and training rooms, as well as a video production suite.
In addition, the company has a seasoned, expert leadership team in place, led by John “JT” Thatch. Thatch was appointed president and CEO of SHRG in March 2018, bringing to the company his expertise obtained from successfully starting, owning and operating several businesses in various industries. His experience with corporate growth, acquisitions, financing and negotiation in fast-paced and flexible environments will significantly assist SHRG as the company aims to expand and increase revenues.
Contact
469.304.9400 x 201
[email protected]
http://www.SHRGinc.com
Sharing Services Global Corporation (SHRG), closed the day's trading session at $1.01, up 23.17%, on 276,351 volume with 217 trades. The average volume for the last 3 months is 279,074 and the stock's 52-week low/high is $0.27/$2.54.
Recent News
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG), DSS Release Date of Share Distribution
- InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Part of DSS Common Stock Distribution Plan
- DSS Announces Letter to Shareholders
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Closes on $3M Registered Direct Offering, Concurrent Private Placement
- Aditxt Inc. (NASDAQ: ADTX) - Stem Cell Transplantation Shows Promise in Treating Autoimmune Disorders
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Maine Legislator Files Anticipatory Bill to Facilitate Cannabis Interstate Commerce
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Renews Partnership with Michigan Works! Southeast
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Closes on $3M Registered Direct Offering
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Research Shows Ancient Romans Missed Huge Silver Deposit in Germany
- Augmedix Inc. (NASDAQ: AUGX) - InvestorNewsBreaks - Augmedix Inc. (NASDAQ: AUGX) Executes Strategic Financing, Partners with Leading Healthcare Provider to Develop AI-Enabled Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Featured in Bell2Bell Podcast
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Study Shows that Skin Conditions May Indicate Presence of Rheumatic Disease
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF), IMAC Execute Strategic 'Merger-of-Equals' LOI
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - THE Mining Investment Event of the North - Quebec City, June 2023 Announces THE Student Sponsorship
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - TechMediaBreaks - CISO Global Inc. (NASDAQ: CISO) Achieves Widely Recognized SOC 2 Certification
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Retains Analytics Firm, Investigates Possible Naked Short Selling
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) CEO to Participate at Upcoming Milken Institute Global Conference
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Private Placement Pricing, Announces Credit Facility Refinance
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Continues Winning Streak; Expands Across Financial Sector
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Brings Power of Quantum Computing to Manufacturing and Logistics
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Challenges Facing European Psychedelics Industry
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Inks New Sales Orders, Anticipates More Sales from New 'Orientable' System
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Bill Pearson Provides Market with Corporate Update
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Germans' Debate on Nuclear vs. Renewable Energy Continues Amid Station Shutdown
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Boasts Unique Position Within RNG Space; Put Its Foot on the Pedal for Development Projects
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Enters Strategic Partnership with Tier-1 Global Motor Manufacturer
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Retires All Outstanding Institutional Convertible Debt
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Why Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Is 'One to Watch'
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Anomalous Silver Intercepts at Flagship Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Planned 1-for-30 ADS Ratio Change
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Additional Funding to Support Growth
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Approaching Finish Line on First Farm-Ready FP300 System
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Positioned to Gain from Shifting Tide as Renewable Electricity Sources Become Cheaper Than Traditional
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Positive Data Supporting Therapeutic Potential of Its Non-Viral Delivery System
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces Engagement of Kingswood Capital Partners LLC for coyni Spin-Off
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Completes $25M Capital Raise to Achieve Upcoming Milestones, Extend Cash Runway
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Schedules Release of Q1 2023 Earnings Results, Conference Call
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Announces Closing of Second Tranche of Non-Brokered Private Placement
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Introduces Transparent Orderbooks with Goal of Bringing Transparency to Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - When Ideanomics Inc ($IDEX) Moves Investors should Listen
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Study Finds No Elevated Risk of Psychosis Resulting from Marijuana Use
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Files 2022 Annual Report on Form 20-F
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Concludes Enrollment for Phase 2 Trial Studying INM-755 Cannabinol Cream
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Research Finds Taking Vitamin D Supplements While Pregnant Reduces Eczema Risk in Infants
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Announces Site of Upcoming Robot Roadshow, Deploys ASR to Bay-Area Biotech
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Subsidiary Applies for Patent Covering Revolutionary Teeth Whitening Method
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX), Featured in Recent Research Report by Zacks SCR
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Announces Results Conference Call for Q1 2023
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Positive Preclinical Results on MYCO-006 Series
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participating in EF Hutton Fireside Chat
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Receives Follow-Up Fleet Order from UNC Charlotte
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Adds Good Planet Foods to eCommerce Partner Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - Playgon Announces Upsize and Closing of Brokered Offering and Anchor Financing
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp.'s (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Subsidiary Issues Update on NRG Fueling Station Plans in East San Bernardino County
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Appoints Seasoned Veteran as New Board Member
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces Rapid Progress on Limited Offering of Exclusive THC Licenses for UST(TM) Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Report on Gastroparesis Results at Annual Digestive Disease Meeting
- Progressive Care Inc. (OTCQB: RXMD) - Progressive Care Inc. (RXMD) Technology Solutions May Spark Rapid Expansion
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Granted European Patent for Clinical-Stage Radiopharmaceutical Drug Candidate
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Names New CFO, Corporate Secretary
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) Aims to Meet Surging Graphite Demand Amid Global Electrification Push
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Announces FY 2022 Performance, 240% Revenue Growth
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) CEO Releases Stockholder Letter
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG), DSS Release Date of Share Distribution
- SideChannel Inc. (OTCQB: SDCH) - Cybersecurity Solutions Provider SideChannel Inc. (SDCH) Helps Clients Remain Vigilant Against The Growing Threat of Network Attacks
- Sigma Additive Solutions (NASDAQ: SASI) - Sigma Additive Solutions Reports Fourth Quarter and Full Year 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Announces Intent to Acquire BitGift as Potential Blockchain Payment Processor for Oregon Psilocybin
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Expands Footprint in Education Vertical with > $900K Order
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Inks New Deal with Stein Distributing for All Splash Brands
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation at Upcoming Echelon Partners Lithium Summit
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Inc. (STCB) Is 'One to Watch'
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces the Televised Lineups for Shows 461, 462, and 463; Airings on Three Networks Will Begin on Saturday, April 29, 2023, at 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Releases Q1 2023 Financial Results
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Flagship Product Added to Cold-Vault Schematics of Store Chain
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Files Audited Financial Statements, Information for Recently Acquired Tingo Foods
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Closes on AUD $2.4M Oversubscribed Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Announces Scheduled Release of Q1 2023 Financial Results, Conference Call
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Eyeing Opportunity in Multibillion Rare Oxide Market
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Files Report on Saskatchewan-Based Roughrider Project
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - Vistagen Presents New Fasedienol (PH94B) Research at 2023 Anxiety and Depression Association of America Conference
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Reports on 'Game-Changing' Appliance Treatment for Migraines
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies Announces Sponsorship of the Professional Networking Symposium Event at the Professionalizing Law Enforcement Community Engagement Training (PLECET) National Conference
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Closes on $3M Registered Direct Offering, Concurrent Private Placement
- Aditxt Inc. (NASDAQ: ADTX) - Stem Cell Transplantation Shows Promise in Treating Autoimmune Disorders
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Maine Legislator Files Anticipatory Bill to Facilitate Cannabis Interstate Commerce
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Renews Partnership with Michigan Works! Southeast
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Closes on $3M Registered Direct Offering
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Research Shows Ancient Romans Missed Huge Silver Deposit in Germany
- Augmedix Inc. (NASDAQ: AUGX) - InvestorNewsBreaks - Augmedix Inc. (NASDAQ: AUGX) Executes Strategic Financing, Partners with Leading Healthcare Provider to Develop AI-Enabled Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Featured in Bell2Bell Podcast
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Study Shows that Skin Conditions May Indicate Presence of Rheumatic Disease
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF), IMAC Execute Strategic 'Merger-of-Equals' LOI
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - THE Mining Investment Event of the North - Quebec City, June 2023 Announces THE Student Sponsorship
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - TechMediaBreaks - CISO Global Inc. (NASDAQ: CISO) Achieves Widely Recognized SOC 2 Certification
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Retains Analytics Firm, Investigates Possible Naked Short Selling
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) CEO to Participate at Upcoming Milken Institute Global Conference
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Private Placement Pricing, Announces Credit Facility Refinance
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Continues Winning Streak; Expands Across Financial Sector
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Brings Power of Quantum Computing to Manufacturing and Logistics
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Challenges Facing European Psychedelics Industry
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Inks New Sales Orders, Anticipates More Sales from New 'Orientable' System
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Bill Pearson Provides Market with Corporate Update
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Germans' Debate on Nuclear vs. Renewable Energy Continues Amid Station Shutdown
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Boasts Unique Position Within RNG Space; Put Its Foot on the Pedal for Development Projects
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Enters Strategic Partnership with Tier-1 Global Motor Manufacturer
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Retires All Outstanding Institutional Convertible Debt
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Why Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Is 'One to Watch'
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Anomalous Silver Intercepts at Flagship Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Planned 1-for-30 ADS Ratio Change
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Additional Funding to Support Growth
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Approaching Finish Line on First Farm-Ready FP300 System
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Positioned to Gain from Shifting Tide as Renewable Electricity Sources Become Cheaper Than Traditional
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Positive Data Supporting Therapeutic Potential of Its Non-Viral Delivery System
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces Engagement of Kingswood Capital Partners LLC for coyni Spin-Off
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Completes $25M Capital Raise to Achieve Upcoming Milestones, Extend Cash Runway
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Schedules Release of Q1 2023 Earnings Results, Conference Call
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Announces Closing of Second Tranche of Non-Brokered Private Placement
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Introduces Transparent Orderbooks with Goal of Bringing Transparency to Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - When Ideanomics Inc ($IDEX) Moves Investors should Listen
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Study Finds No Elevated Risk of Psychosis Resulting from Marijuana Use
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Files 2022 Annual Report on Form 20-F
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Concludes Enrollment for Phase 2 Trial Studying INM-755 Cannabinol Cream
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Research Finds Taking Vitamin D Supplements While Pregnant Reduces Eczema Risk in Infants
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Announces Site of Upcoming Robot Roadshow, Deploys ASR to Bay-Area Biotech
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Subsidiary Applies for Patent Covering Revolutionary Teeth Whitening Method
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX), Featured in Recent Research Report by Zacks SCR
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Announces Results Conference Call for Q1 2023
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Positive Preclinical Results on MYCO-006 Series
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participating in EF Hutton Fireside Chat
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Receives Follow-Up Fleet Order from UNC Charlotte
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Adds Good Planet Foods to eCommerce Partner Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - Playgon Announces Upsize and Closing of Brokered Offering and Anchor Financing
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp.'s (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Subsidiary Issues Update on NRG Fueling Station Plans in East San Bernardino County
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Appoints Seasoned Veteran as New Board Member
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces Rapid Progress on Limited Offering of Exclusive THC Licenses for UST(TM) Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Report on Gastroparesis Results at Annual Digestive Disease Meeting
- Progressive Care Inc. (OTCQB: RXMD) - Progressive Care Inc. (RXMD) Technology Solutions May Spark Rapid Expansion
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Granted European Patent for Clinical-Stage Radiopharmaceutical Drug Candidate
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Names New CFO, Corporate Secretary
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) Aims to Meet Surging Graphite Demand Amid Global Electrification Push
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Announces FY 2022 Performance, 240% Revenue Growth
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) CEO Releases Stockholder Letter
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG), DSS Release Date of Share Distribution
- SideChannel Inc. (OTCQB: SDCH) - Cybersecurity Solutions Provider SideChannel Inc. (SDCH) Helps Clients Remain Vigilant Against The Growing Threat of Network Attacks
- Sigma Additive Solutions (NASDAQ: SASI) - Sigma Additive Solutions Reports Fourth Quarter and Full Year 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Announces Intent to Acquire BitGift as Potential Blockchain Payment Processor for Oregon Psilocybin
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Expands Footprint in Education Vertical with > $900K Order
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Inks New Deal with Stein Distributing for All Splash Brands
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation at Upcoming Echelon Partners Lithium Summit
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Inc. (STCB) Is 'One to Watch'
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces the Televised Lineups for Shows 461, 462, and 463; Airings on Three Networks Will Begin on Saturday, April 29, 2023, at 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Releases Q1 2023 Financial Results
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Flagship Product Added to Cold-Vault Schematics of Store Chain
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Files Audited Financial Statements, Information for Recently Acquired Tingo Foods
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Closes on AUD $2.4M Oversubscribed Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Announces Scheduled Release of Q1 2023 Financial Results, Conference Call
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Eyeing Opportunity in Multibillion Rare Oxide Market
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Files Report on Saskatchewan-Based Roughrider Project
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - Vistagen Presents New Fasedienol (PH94B) Research at 2023 Anxiety and Depression Association of America Conference
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Reports on 'Game-Changing' Appliance Treatment for Migraines
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies Announces Sponsorship of the Professional Networking Symposium Event at the Professionalizing Law Enforcement Community Engagement Training (PLECET) National Conference
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Closes on $3M Registered Direct Offering, Concurrent Private Placement
- Aditxt Inc. (NASDAQ: ADTX) - Stem Cell Transplantation Shows Promise in Treating Autoimmune Disorders
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Maine Legislator Files Anticipatory Bill to Facilitate Cannabis Interstate Commerce
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Renews Partnership with Michigan Works! Southeast
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Closes on $3M Registered Direct Offering
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Research Shows Ancient Romans Missed Huge Silver Deposit in Germany
- Augmedix Inc. (NASDAQ: AUGX) - InvestorNewsBreaks - Augmedix Inc. (NASDAQ: AUGX) Executes Strategic Financing, Partners with Leading Healthcare Provider to Develop AI-Enabled Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Featured in Bell2Bell Podcast
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Study Shows that Skin Conditions May Indicate Presence of Rheumatic Disease
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF), IMAC Execute Strategic 'Merger-of-Equals' LOI
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - THE Mining Investment Event of the North - Quebec City, June 2023 Announces THE Student Sponsorship
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - TechMediaBreaks - CISO Global Inc. (NASDAQ: CISO) Achieves Widely Recognized SOC 2 Certification
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Retains Analytics Firm, Investigates Possible Naked Short Selling
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) CEO to Participate at Upcoming Milken Institute Global Conference
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Private Placement Pricing, Announces Credit Facility Refinance
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Continues Winning Streak; Expands Across Financial Sector
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Brings Power of Quantum Computing to Manufacturing and Logistics
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Challenges Facing European Psychedelics Industry
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Inks New Sales Orders, Anticipates More Sales from New 'Orientable' System
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Bill Pearson Provides Market with Corporate Update
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Germans' Debate on Nuclear vs. Renewable Energy Continues Amid Station Shutdown
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Boasts Unique Position Within RNG Space; Put Its Foot on the Pedal for Development Projects
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Enters Strategic Partnership with Tier-1 Global Motor Manufacturer
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Retires All Outstanding Institutional Convertible Debt
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Why Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Is 'One to Watch'
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Anomalous Silver Intercepts at Flagship Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Planned 1-for-30 ADS Ratio Change
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Additional Funding to Support Growth
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Approaching Finish Line on First Farm-Ready FP300 System
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Positioned to Gain from Shifting Tide as Renewable Electricity Sources Become Cheaper Than Traditional
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Positive Data Supporting Therapeutic Potential of Its Non-Viral Delivery System
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces Engagement of Kingswood Capital Partners LLC for coyni Spin-Off
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Completes $25M Capital Raise to Achieve Upcoming Milestones, Extend Cash Runway
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Schedules Release of Q1 2023 Earnings Results, Conference Call
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Announces Closing of Second Tranche of Non-Brokered Private Placement
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Introduces Transparent Orderbooks with Goal of Bringing Transparency to Trading
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - When Ideanomics Inc ($IDEX) Moves Investors should Listen
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Study Finds No Elevated Risk of Psychosis Resulting from Marijuana Use
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Files 2022 Annual Report on Form 20-F
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Concludes Enrollment for Phase 2 Trial Studying INM-755 Cannabinol Cream
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Research Finds Taking Vitamin D Supplements While Pregnant Reduces Eczema Risk in Infants
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Announces Site of Upcoming Robot Roadshow, Deploys ASR to Bay-Area Biotech
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Subsidiary Applies for Patent Covering Revolutionary Teeth Whitening Method
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX), Featured in Recent Research Report by Zacks SCR
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Announces Results Conference Call for Q1 2023
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Positive Preclinical Results on MYCO-006 Series
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participating in EF Hutton Fireside Chat
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Receives Follow-Up Fleet Order from UNC Charlotte
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Adds Good Planet Foods to eCommerce Partner Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - Playgon Announces Upsize and Closing of Brokered Offering and Anchor Financing
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp.'s (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Subsidiary Issues Update on NRG Fueling Station Plans in East San Bernardino County
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Appoints Seasoned Veteran as New Board Member
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces Rapid Progress on Limited Offering of Exclusive THC Licenses for UST(TM) Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Report on Gastroparesis Results at Annual Digestive Disease Meeting
- Progressive Care Inc. (OTCQB: RXMD) - Progressive Care Inc. (RXMD) Technology Solutions May Spark Rapid Expansion
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Granted European Patent for Clinical-Stage Radiopharmaceutical Drug Candidate
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Names New CFO, Corporate Secretary
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) Aims to Meet Surging Graphite Demand Amid Global Electrification Push
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Announces FY 2022 Performance, 240% Revenue Growth
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) CEO Releases Stockholder Letter
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG), DSS Release Date of Share Distribution
- SideChannel Inc. (OTCQB: SDCH) - Cybersecurity Solutions Provider SideChannel Inc. (SDCH) Helps Clients Remain Vigilant Against The Growing Threat of Network Attacks
- Sigma Additive Solutions (NASDAQ: SASI) - Sigma Additive Solutions Reports Fourth Quarter and Full Year 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Announces Intent to Acquire BitGift as Potential Blockchain Payment Processor for Oregon Psilocybin
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Expands Footprint in Education Vertical with > $900K Order
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Inks New Deal with Stein Distributing for All Splash Brands
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation at Upcoming Echelon Partners Lithium Summit
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Inc. (STCB) Is 'One to Watch'
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces the Televised Lineups for Shows 461, 462, and 463; Airings on Three Networks Will Begin on Saturday, April 29, 2023, at 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Releases Q1 2023 Financial Results
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Flagship Product Added to Cold-Vault Schematics of Store Chain
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Files Audited Financial Statements, Information for Recently Acquired Tingo Foods
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Closes on AUD $2.4M Oversubscribed Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Announces Scheduled Release of Q1 2023 Financial Results, Conference Call
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Eyeing Opportunity in Multibillion Rare Oxide Market
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Files Report on Saskatchewan-Based Roughrider Project
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - Vistagen Presents New Fasedienol (PH94B) Research at 2023 Anxiety and Depression Association of America Conference
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Reports on 'Game-Changing' Appliance Treatment for Migraines
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies Announces Sponsorship of the Professional Networking Symposium Event at the Professionalizing Law Enforcement Community Engagement Training (PLECET) National Conference
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































