The QualityStocks Daily Stock List
- Greene Concepts (INKW)
- NanoViricides (NNVC)
- Artificial Intelligence Technology Solutions (AITX)
- OZOP Energy Solutions (OZSC)
- DiaMedica Therapeutics (DMAC)
- Trulieve Cannabis Corp. (TCNNF)
- Aileron Therapeutics Inc. (ALRN)
- Cemtrex Inc. (CETX)
- Zevra Therapeutics (ZVRA)
- Curaleaf Holdings Inc. (CURLF)
- Nikola Corporation (NKLA)
- Hecla Mining Company (HL)
Greene Concepts (INKW)
QualityStocks, PennyTrader, Penny Picks, HotOTC, Damn Good Penny Picks, Wall Street Mover, PREPUMP STOCKS, Penny Stock Newsletter, MicroCapDaily, MegaPennyStocks and MarketClub Analysis reported earlier on Greene Concepts (INKW), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Greene Concepts, Inc. (OTC: INKW) manages a beverage and bottling firm in Marion, North Carolina through Mammoth Ventures Inc., its wholly-owned subsidiary.
The company is based in Clovis, California and was founded in 1952, on August 18th by Leonard M. Greene. Its product line is made up of enhanced athletic drinks, beverage offerings, artesian and spring water as well as ketogenic and CBD-infused beverages. The company is focused on delivering water to its consumers directly.
Its Marion plant is a 60,000 ft2 bottling and beverage facility that is found within the Pisgah National Forest boundaries. The plant’s water source is a combination of 7 artesian and spring wells that are fed from a natural aquifer found in the Pisgah forest. Mammoth Ventures is also a 3rd party bottler and producer of ‘white label’ water and beverage products. These services are offered to clients that would like to market their own product formulations, labeling and brand name while outsourcing the bottling and production of their products to Mammoth. Through its Water Club Inc. subsidiary, the company is pursuing subscription-based delivery of scientifically formulated beverages and water directly to the consumer’s markets and homes.
The firm recently hired an auditing company to audit its financials, in preparation for an up list to the NASDAQ. This could not only lead to a change in its stock symbol but also attract more investors, which will boost both the firm’s share prices as well as its growth.
Greene Concepts (INKW), closed Monday's trading session at $0.005, up 42.8571%, on 27,670,512 volume. The average volume for the last 3 months is 29,680 and the stock's 52-week low/high is $0.00164/$0.0066.
NanoViricides (NNVC)
Tip.us, Wall Street Resources, QualityStocks, StockMarketWatch, Stock Preacher, Beacon Equity Research, InvestorSoup, PennyStocks24, MarketClub Analysis, BUYINS.NET, StockProfessors, Jason Bond, TradersPro, SuperStockTips, Penny Stocks Finder, StockHideout, Penny Stock Craze, MissionIR, SmarTrend Newsletters, Stock Roach, MarketBeat, PennyStockShark, LightningStockPicks, Stock Analyzer, Standout Stocks, FeedBlitz, USA Market News, CoolPennyStocks, ProTrader, Penny Stock Finder, Market Wrap Daily, OTCPicks, StreetAuthority Daily, Tiny Gems, HotOTC, TopStockAnalysts, Broad Street, Small Caps, PennyOmega, CRWEPicks, The Street, The Online Investor, InvestorPlace, InvestorsUnderground, HotStockChat, Greenbackers, CRWEWallStreet, CRWEFinance, BullRally, BestOtc, AllPennyStocks, All about trends, DrStockPick, Agora Financial, Stock Market Watch, SmallCapVoice, Round Up the Bulls, RedChip, Real Pennies, Stock Rich, ProfitableTrading, PennyTrader Publisher, PennyToBuck, MegaPennyStocks, Stock Source, StreetInsider, StockEgg, StockHotTips, Penny Pick Finders, Penny Invest, OTCReporter, Stockwire, MicrocapVoice, Stock Beast, MicroCapDaily, MarketClub and Zacks reported earlier on NanoViricides (NNVC), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
NanoViricides Inc. (NYSE American: NNVC) (FRA: NV3P) is a nano-biopharmaceutical firm that is engaged in the discovery, development and commercialization of therapies for treating viral infections and helping advance the care of patients with these life-threatening ailments.
The firm has its headquarters in Shelton, Connecticut and was incorporated in 2005, on April 1st by Anil R. Diwan. It operates in the health care sector, under the biotech and pharma sub-industry.
The company centers its clinical programs and research on certain anti-viral therapies and is working on adding to them its current portfolio of products via an in-licensing strategy and its clinical development and internal discovery programs. It is currently engaged in the application of Nano medicine technologies to the intricate issues of viral illnesses.
The enterprise’s product pipeline is made up of an anti-HIV nanoviricide dubbed HIVCide; DengueCide developed for treating all Dengue viruses; Nanoviricide eye drops for viral ailments that affect the external eye; and an injectable and an anti-influenza oral broad-spectrum nanoviricide known as FluCide, developed for both outpatients and hospitalized patients. The enterprise also develops a treatment for viral acute necrosis dubbed HerpeCide IntraOcular Injection and a formulation indicated for the treatment of ocular herpes keratitis, genital herpes, recurrent herpes labialis, herpes, chickenpox, PHN and shingles, dubbed HerpeCide Dermal Topical and Eye Drops. In addition to this, it is also engaged in HerpeCide program expansion drug projects for various herpes viruses; and researching and developing other nanoviricides for treating different indications and viruses.
The firm recently entered into an agreement with TheraCour Pharma Inc. to target encephalitis, conjunctivitis, dengue and Ebola. The development of cures for these ailments, which presently have no cures, will not only significantly boost the growth of both companies but also improve share prices and encourage more investments into the companies.
NanoViricides (NNVC), closed Monday's trading session at $1.4, up 23.8938%, on 1,067,879 volume. The average volume for the last 3 months is 90.719M and the stock's 52-week low/high is $1.00/$2.00.
Artificial Intelligence Technology Solutions (AITX)
QualityStocks, MarketClub Analysis, The Street Report, Investors Alley and InvestorPlace reported earlier on Artificial Intelligence Technology Solutions (AITX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Artificial Intelligence Technology Solutions Inc. (OTC: AITX) is focused on delivering robotic and artificial intelligence solutions for monitoring, security and operational needs. The company’s AI technologies are supported by custom cloud and software services and paired with multi-use hardware, which helps to intelligently integrate and automate operational, concierge and security tasks.
The firm has its headquarters in Reno, Nevada and was founded by John B. Crawford in 2010, on March 25. Before changing its name in August 2018 to Artificial Intelligence Technology Solutions Inc., the firm was known as On The Move Systems Corp. The company serves consumers in the U.S. and in particular, automobile fleet administrators and vehicle dealerships.
The company operates through its wholly-owned subsidiaries, one of which is Robotic Assistance Devices Inc. The subsidiary develops unique technology, specifically; mobileware, cloudware, software and hardware, which are used in the development of powerful tools like RADGuard, RADPMC and RADSOC as well as to improve performance in companies.
The firm is also engaged in the provision of mobile electronic services and offers after-market video, audio and electronic upgrades for boats, recreational vehicles and automobiles.
The company recently entered into an agreement with EAGL Technology Inc. for the provision of the latter firm’s gunshot detection system in all present and future RAD devices. The integration of EAGL solutions into the innovative products will not only make the products more popular but also expand their reach into the markets they serve, which will be good for investments as well as the firm’s stock.
Artificial Intelligence Technology Solutions (AITX), closed Monday's trading session at $0.0067, up 21.8625%, on 196,809,028 volume. The average volume for the last 3 months is 25.153M and the stock's 52-week low/high is $0.0019/$0.0146.
OZOP Energy Solutions (OZSC)
QualityStocks, MarketClub Analysis and InvestorPlace reported earlier on OZOP Energy Solutions (OZSC), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
OZOP Energy Solutions Inc. (OTC: OZSC) is focused on the design, manufacture and distribution of power electronics and endoscopic instruments.
The firm invents and develops power supplies, converters, inverters and ultra-high power chargers for various applications in the maritime, aircraft ground support, heavy industrial and defense sectors. OZOP Energy Solutions Inc. serves consumers across the globe.
The firm, which was known as OZOP Surgical Corp before changing its name, has its headquarters in Warwick, New York. OZOP Energy Solutions Inc. was established on July 17, 2015 and operates through the Hong Kong and United States geographical segments.
OZOP Energy Solutions also provides surgical devices, instrumentation and implants, which focus on neurological, spine and pain management specialties and procedures. The firm’s AC (alternating current) and DC (direct current) power supplies, which are available in ratings from 5KW (kilowatt) to 20 MW (megawatt), are utilized in multi-pulse silicon control rectifier, insulated gated bipolar transistor design. They provide low output noise, precise regulation, low ripple and also help achieve efficiency. The firm also offers a frequency converter for any military, industrial or commercial application with power ranges from 4KVA to 500 KVA (kilovolt-ampere). This is in addition to providing 400 hertz aircraft ground support equipment and manufacturing all types of power inverters, including electrostatic precipitators, power transmissions, solar cell power stations, AC/AC inverters, DC/AC inverters and DC/DC inverters.
OZOP Energy Solutions Inc. recently announced that they’d be developing the first contract energy systems for non-grid solutions. This move aligns with the firm’s expansion plan and with energy use in the U.S. set to increase significantly, the energy production industry is bound to need help in meeting the population’s needs, which is where the firm is set to gain hefty returns for its shareholders.
OZOP Energy Solutions (OZSC), closed Monday's trading session at $0.00135, up 17.3913%, on 85,015,486 volume. The average volume for the last 3 months is 37,656 and the stock's 52-week low/high is $0.0007/$0.0085.
DiaMedica Therapeutics (DMAC)
MarketBeat, TradersPro, QualityStocks, StockMarketWatch, InsiderTrades, StreetInsider, Schaeffer's, MarketClub Analysis and BUYINS.NET reported earlier on DiaMedica Therapeutics (DMAC), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
DiaMedica Therapeutics Inc. (NASDAQ: DMAC) (FRA: F2KA) is a clinical stage biopharmaceutical firm that is engaged in the development of therapies for kidney and neurological ailments.
The firm has its headquarters in Minneapolis, Minnesota and was incorporated in 2000, on January 21st by Wayne Lautt. Prior to its name change in December 2016, the firm was known as DiaMedica Inc. It operates as part of the scientific research and development services industry under the health care sector in the biotech and pharma sub-industry. The firm serves consumers in Canada and the U.S. and has three companies in its corporate family.
The company is focused on developing treatments for clinically unmet needs or where no treatments are available, its main focus being on acute ischemic stroke and chronic kidney disease.
The enterprise’s product pipeline is made of a recombinant human tissue kallikrein-1 protein dubbed DM199, which concluded phase 2b REMEDY trial evaluating its efficacy in treating patients with acute ischemic stroke as well as phase 1b clinical trials evaluating its effectiveness in treating patients with severe or moderate chronic kidney disease caused by Type II or I diabetes. The DM199 formulation has also been indicated for the treatment of vascular dementia. The enterprise is working on advancing its kallikrein-1 recombinant into clinical testing to evaluate its efficacy in treating stroke.
The company recently revealed that the FDA had approved its Investigational New Drug Application for its DM199 formulation, which brings it one step closer to providing a much needed treatment option for individuals who experience acute ischemic stroke each year. The success of the formulation will not only be beneficial to patients but will also bring in more investments into the firm.
DiaMedica Therapeutics (DMAC), closed Monday's trading session at $3.5, up 16.6667%, on 143,897 volume. The average volume for the last 3 months is 558,838 and the stock's 52-week low/high is $1.58/$4.75.
Trulieve Cannabis Corp. (TCNNF)
InvestorPlace, QualityStocks, MarketBeat, Wealth Insider Alert, Daily Trade Alert, Top Pros' Top Picks, Cabot Wealth, The Street, Trades Of The Day, Profit Trends, TradersPro, The Online Investor, StreetInsider and Prism MarketView reported earlier on Trulieve Cannabis Corp. (TCNNF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Germany saw a significant surge in its marijuana imports for scientific and medical purposes last year, reaching a record high, indicating a growing interest from international businesses eyeing opportunities in Europe’s largest federally regulated medical cannabis market. According to the latest data from the Federal Institute for Drugs & Medical Devices (BfArM), the country imported 36.4 tons (31,398 kilograms) of marijuana products last year. This marks a notable increase of 26.2% compared to the previous year’s import volume of 24,876 kilograms.
The trend of increasing imports has been consistent through the years. In 2022, the country saw imports of 27.4 tons (24,876 kilograms) of marijuana, reflecting a 19.8% increase from the previous year. The year 2021 saw imports of 22.9 tons (20,771 kilograms), representing a significant surge of 77% over the previous year.
In 2020, imports totaled 12.8 tons (11,746 kilograms), indicating a 46% increase from 2019, while in 2019, Germany imported 8.9 tons (8,057 kilograms) of marijuana, which was an 80% increase from the previous year’s imports. While some of these imports are re-exported to other European Union countries annually, experts suggest that the data underscores the rapid growth of the industry.
Only three businesses were authorized to grow medical cannabis in 2019; these companies were given strict output targets that would last for four years. Due to this restriction, cannabis had to be imported from overseas to meet demand. However, changes in marijuana law have the potential to reduce its dependence on imports in the future. The new law eliminates the quota system, allowing companies to apply for permits to grow medical cannabis through the agency.
Furthermore, the recent revision of the marijuana law, effective April 1, 2024, has reclassified the substance, removing it from the list of narcotics. This regulatory change simplifies the process for patients seeking medical marijuana because they will no longer be required to have a narcotic prescription form. Standard prescriptions will now suffice, likely stimulating demand in the market.
Despite the optimism for increased domestic cultivation, Germany is expected to continue relying on significant imports in the foreseeable future. German-based Demecan’s managing director, Von der Groeben, pointed out Article 21 of the UN Single Convention on Narcotic Drugs, which suggests that domestic demand must be met before imports can be halted. However, he acknowledges that achieving self-sufficiency in marijuana production may take several years.
Canada remained the primary supplier of marijuana to the German market last year, accounting for approximately 50% of imports. Portugal came in second, while the Netherlands was the third-largest supplier.
The further easing of marijuana laws in Germany could create additional opportunities that major players in the North American cannabis landscape, such as Trulieve Cannabis Corp. (CSE: TRUL) (OTCQX: TCNNF), may look to exploit as they expand their footprint in different legal markets.
Trulieve Cannabis Corp. (TCNNF), closed Monday's trading session at $11.31, off by 8.7903%, on 436,086 volume. The average volume for the last 3 months is 86,101 and the stock's 52-week low/high is $3.42/$14.50.
Aileron Therapeutics Inc. (ALRN)
TradersPro, MarketBeat, StreetInsider, QualityStocks, InvestorPlace, BUYINS.NET, WiseAlerts, The Online Investor, StockMarketWatch, Money Wealth Matters and Barchart reported earlier on Aileron Therapeutics Inc. (ALRN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Aileron Therapeutics (NASDAQ: ALRN) is a biopharmaceutical company advancing a novel pipeline of first-in-class medicines to address significant unmet medical needs in orphan pulmonary and fibrosis indications. The company has announced the closing of its underwritten registered direct offering priced at-the-market under Nasdaq rules of 4,273,505 shares of its common stock and accompanying warrants to purchase an aggregate of 4,273,505 shares of common stock. Aileron secured aggregate gross proceeds of approximately $20 million, before deducting underwriting discounts and commissions and other offering expenses and excluding any proceeds that may be received from exercise of the warrants. Titan Partners Group, a division of American Capital Partners, acted as sole book-running manager for the offering.
To view the full press release, visit https://ibn.fm/RCTHH
About Aileron Therapeutics
Aileron is a biopharmaceutical company advancing a pipeline of first-in-class medicines to address significant unmet medical needs in orphan pulmonary and fibrosis indications. Aileron’s lead product candidate, LTI-03, is a novel, synthetic peptide with a dual mechanism targeting alveolar epithelial cell survival as well as inhibition of profibrotic signaling. Currently, LTI-03 is being evaluated in a Phase 1b clinical trial for the treatment of idiopathic pulmonary fibrosis. Aileron’s second product candidate, LTI-01, is a proenzyme that has completed Phase 1b and Phase 2a clinical trials for the treatment of loculated pleural effusions. LTI-01 has received Orphan Drug Designation in the U.S. and EU and Fast Track Designation in the U.S.
Aileron Therapeutics Inc. (ALRN), closed Monday's trading session at $3.95, up 5.0532%, on 87,757 volume. The average volume for the last 3 months is 476,501 and the stock's 52-week low/high is $1.01/$7.42.
Cemtrex Inc. (CETX)
TaglichBrothers, StockMarketWatch, TraderPower, Broad Street, BUYINS.NET, InvestorPlace, QualityStocks, MarketBeat, OTCBB Journal, StocksImpossible, The Bowser Report, Small Cap Firm, Jason Bond, StockOodles, The Street, Stock Commander, Market FN, Profitable Trader Authority, OTCtipReporter, Penny Pick Finders, PennyStockProphet, PennyStockScholar, AwesomeStocks, MarketClub Analysis, StockOnion, Shiznit Stocks, Buzz Stocks, HotOTC, Penny Stock General, Investing Futures, StockHideout, Today's Stock Tip, The Best Newsletters, StockRunway, OTC Markets Group, Wall Street Resources, TopPennyStockMovers, Fierce Analyst, StockWireNews, Prime Tech Stocks, StockRockandRoll, Weekly Wizards, StockPicksNYC, Penny Stock 101, PennyStockAlertsNYC, PennyStockLocks, Small Caps, SeeThruEquity Research, Schaeffer's and Marketbeat.com reported earlier on Cemtrex Inc. (CETX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Cemtrex (NASDAQ: CETX), an advanced-security technology and industrial services company, has closed on a firm-commitment underwritten public offering. According to the announcement, the company will receive an estimated $10 million in gross proceeds from the offering, before standard deductions and expenses. Upsized from an original $9 million commitment, the base offering consisted of 11,764,705 common units or prefunded units; the purchase price of each common unit was $0.85, and the purchase price of each prefunded unit was $0.849. Cemtrex anticipates using the net proceeds from the offering for operations as well as to increase marketing efforts and invest in existing business initiatives and products; funds may also be used for partial repayment of indebtedness or to acquire or invest in complementary businesses, products or technologies. The company also granted Aegis Capital Corp., the sole book-running manager for the offering, a 45-day option to purchase additional common shares and/or warrants to cover any overallotments. The offering closed on May 3, 2024.
To view the full press release, visit https://ibn.fm/LDSCh
About Cemtrex Inc.
Cemtrex is a company that owns two operating subsidiaries: Vicon Industries Inc. and Advanced Industrial Services Inc. (“AIS”). Vicon is a global leader in advanced-security and surveillance technology to safeguard businesses, schools, municipalities, hospitals and cities. Since 1967, Vicon has delivered mission-critical security surveillance systems, specializing in engineering complete security solutions that simplify deployment, operation and ongoing maintenance. Vicon provides security solutions for some of the largest municipalities and businesses in the United States and around the world, offering a wide range of cutting-edge and compliant security technologies, from artificial intelligence (“AI”)-driven video analytics to fully integrated access control solutions. AIS is a premier provider of industrial contracting services including millwrighting, rigging, piping, electrical and welding. AIS installs high-precision equipment in a wide variety of industrial markets including automotive, printing and graphics, industrial automation, packaging and chemicals. AIS owns and operates a modern fleet of custom-designed specialty equipment to assure safe and quick installation of production equipment. AIS staff participates in recurring instructional training, provided to ensure that the most current industry methods are being utilized to provide an efficient and safe working environment. For more information about the company, visit www.Cemtrex.com.
Cemtrex Inc. (CETX), closed Monday's trading session at $0.2701, off by 2.3146%, on 1,401,185 volume. The average volume for the last 3 months is 252,353 and the stock's 52-week low/high is $0.25/$11.3635.
Zevra Therapeutics (ZVRA)
MarketBeat and QualityStocks reported earlier on Zevra Therapeutics (ZVRA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Zevra Therapeutics (NASDAQ: ZVRA), a rare disease therapeutics company, uniquely combines data-driven techniques, patient needs and outside the box strategies while challenging moribund drug development protocols to bring new therapeutic relief to millions of people suffering with rare diseases. With a team of expert scientists, patient advocates, development strategists, commercialization specialists, and business development gurus – Zevra intends to become a global leader in developing and commercializing treatments for rare diseases that have limited or no therapeutic options. The company’s recent Letter to Stockholders reveals what looks like a launch code for success and suggests the company is on a fast track to drive value for all its stakeholders – patients and caregivers, stockholders, investors, and employees.
In the letter, Neil F. McFarlane, President and CEO, highlighted recent achievements and, importantly, outlined the vision for the company’s future. McFarlane began his tenure by instilling a couple of prime directives that have become guiding principles for the Zevra team. The first directive has become the company mantra, “What’s best for the patient?”, and patient outcomes now guide daily decisions. The second directive McFarlane instilled, “We must execute.”, drives value not just for patients but for all stakeholders – he believes that by defining a clear strategy, creating effective tactics to reach strategic objectives, then executing at the highest level will create and drive value, and help propel Zevra into global leadership in developing and commercializing rare disease therapeutics. Hard to argue with his directives, Zevra is fast making huge strides in reaching objectives.
Zevra has already made significant progress advancing its rare disease portfolio and made demonstrable progress in adroitly executing key strategic priorities. In November 2023, the company completed a shrewd, capital-efficient acquisition of Acer Therapeutics. That acquisition allowed Zerva to immediately start delivering value to patients by commercializing OLPRUVA(R) for treatment of urea cycle disorders (“UCDs”). UCDs are related to defects of enzymes involved in the urea cycle which can lead to high toxic features of ammonia-like irreversible brain damage, coma, and death.
Immediately after the acquisition, Zerva deployed a systematic strategy to build awareness with physicians across the country about OLPRUVA for people living with UCDs. In the three months since launch, Zerva’s team also engaged with over 90% of its customers. The company concurrently achieved meaningful growth in reimbursement coverage, from ~55% of U.S. covered lives at the time of acquisition, to now more than 70%.
The full commercial launch of OLPRUVA at the end of January 2024 was designed to provide scale and cost synergies to support and accelerate the launch and commercialization of arimoclomol to treat Niemann-Pick Disease. Affecting mostly children, Niemann-Pick is a group of rare genetic conditions that affect the body’s ability to break down and use fats, such as cholesterol and lipids, inside cells, and over time, the cells die worsening the function of the nerves, brain, and other organs. With no known cure and sometimes fatal, Zerva’s arimoclomol could be the lifeline so many children need.
Exemplifying the prime directives, the entire Zerva team put forth enormous effort to submit a comprehensive data set to the U.S. Food and Drug Administration (“FDA”) supporting the resubmission of the New Drug Application (“NDA”) for arimoclomol. In March, Zerva announced that the FDA had extended the review period for the NDA, resulting in a revised Prescription Drug User Fee Act (“PDUFA”) date of September 21, 2024.
Also in March, Zerva reported positive top-line data from its Phase 2 study of KP1077 in patients with idiopathic hypersomnia (“IH”). Initial results show that KP1077 is well tolerated and demonstrates early signs of differentiated and meaningful clinical benefits. Topline data from the completed trial are expected in the first half of 2024 and represent another potential Zevra commercial candidate.
Zerva fully intends to bring life-changing therapeutics to people living with rare diseases through expertise, elegant solutions, and transparent data narratives which have already produced a late-stage rare disease clinical pipeline and two partnered commercial products.
But blockbusters and breakthroughs don’t happen in a vacuum. It takes a dedicated and committed team of expert scientists, patient advocates, development strategists, commercialization specialists, and business development gurus with a proven record of bringing new therapies to patients…. and Zerva is loaded with talent.
Zerva is building a team of talented professionals committed to using science and data-driven development approaches to create and deliver new therapies to address critical and often overlooked unmet needs. Since announcing the strategic objective of becoming a leading rare disease company in the first half of 2022, Zerva has grown its team of skilled professionals through both recruiting and acquisitions, from a team of 24 to 81. It’s important to note that this personnel ramp-up includes the buildout of targeted customer-facing commercial and medical affairs teams with decades of experience in rare diseases. The company also added Adrian Quartel, M.D. as Zevra’s Chief Medical Officer, who brings more than 20 years of experience with a track record of success in clinical development, pharmacovigilance and medical affairs addressing the needs of the rare disease community. Such powerful commercial and clinical muscle are likely to really drive value and bode well for the future of the company.
Zevra Therapeutics leverages decades upon decades of collective experience in drug development and regulatory machinations to develop therapies faster and overcome challenging regulatory situations. Rare diseases may finally be reigned-in and held at bay by combining science, data, and patient needs with a team of dedicated professionals to create transformational therapies and bring life-changing therapeutics to millions of people living with rare diseases. Look for more exciting news from Zerva in the months to come.
For more information, please visit www.Zevra.com.
Zevra Therapeutics (ZVRA), closed Monday's trading session at $5.155, up 7.1726%, on 310,448 volume. The average volume for the last 3 months is 865,178 and the stock's 52-week low/high is $3.89/$7.28.
Curaleaf Holdings Inc. (CURLF)
InvestorPlace, QualityStocks, Kiplinger Today, MarketBeat, Cabot Wealth, Daily Trade Alert, Top Pros' Top Picks, The Online Investor, MarketClub Analysis, Profit Trends, Wealth Insider Alert, StreetInsider, Early Bird, Trading For Keeps, Trades Of The Day, The Street, Prism MarketView, TradersPro, Zacks, Schaeffer's, Investment U, StreetAuthority Daily, wyatt research newsletter, Daily Profit, CFN Media Group and Wyatt Investment Research reported earlier on Curaleaf Holdings Inc. (CURLF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Smart Approaches to Marijuana leaders believe the U.S. Drug Enforcement Administration (DEA) may soon make an announcement on marijuana rescheduling. The DEA has spent the last few months chewing over a recommendation made by the U.S. Department of Health and Human Services (HSS) to move marijuana to Schedule III from its current classification.
Now officials of Smart Approaches to Marijuana (SAM), led by president Kevin Sabet, believe the agency will announce its results soon, following chatter from different sources. This has been heard for some months now, with sources mostly leaning toward pro-reform.
In the interview, Sabet also revealed that he was skeptical about the revised FDA review process on drug scheduling. This has been reiterated by a number of Republican legislators. This is in addition to discussing prior scheduling petitions that took years to conclude, only for the federal government to deny proposals to recategorize cannabis under the Controlled Substances Act.
In this case, however, the scheduling review was issued as a directive by President Joseph Biden himself.
Additionally, one DEA official recently stated that at times, it took up to six months for the agency to conclude an analysis of recommendations given by health officials. As it stands, this is less than the amount of time it has been since the DEA started its current evaluation of marijuana’s scheduling.
The prohibition group has long been against cannabis reform and has worked with Republican legislators to discourage the agency from rescheduling the drug. Additionally, SAM has urged its supporters to talk to their representatives in congress about opposing cannabis banking reforms, rescheduling and proposals to legalize the drug.
A week ago, a caucus of 21 congressional legislators asked that the DEA remove cannabis from its current classification and recognized that the DEA could be dealing with disagreements on the issue at an internal scale. This point references a recent report, which noted that officials at the agency were at loggerheads with the current administration over the scheduling review. Thus far, the agency has offered no on-the-record comment about the status of the review, with the White House also declining to give a statement on the timeline.
In other news, the FDA’s head stated that there was no reason for the Drug Enforcement Administration to “put off” making a decision on cannabis scheduling. In March, HSS secretary Xavier Becerra defended the recommendation made by his department during a committee hearing at the senate. In his statement, Becerra told one marijuana lobbyist that he needed to pay the Drug Enforcement Administration a visit and ask officials about the timing of the decision.
The entire marijuana industry, including major players such as Curaleaf Holdings Inc. (CSE: CURA) (OTCQX: CURLF), eagerly awaits the DEA’s announcement of its decision on the classification of marijuana in the Controlled Substances Act. The comment period that will follow is likely to uncover more information about what the industry thinks of the decision that the federal agency will have made.
Curaleaf Holdings Inc. (CURLF), closed Monday's trading session at $5.63, off by 0.3539823%, on 970,443 volume. The average volume for the last 3 months is 95.189M and the stock's 52-week low/high is $2.36/$6.40.
Nikola Corporation (NKLA)
Green Car Stocks, Schaeffer's, InvestorPlace, StockEarnings, QualityStocks, MarketClub Analysis, StocksEarning, MarketBeat, The Street, Early Bird, Kiplinger Today, Trades Of The Day, StreetInsider, Daily Trade Alert, The Online Investor, Zacks, GreenCarStocks, Cabot Wealth, Louis Navellier, CNBC Breaking News, Wealth Insider Alert, Investopedia, INO Market Report, InvestorsUnderground, MarketTamer, StockMarketWatch, The Wealth Report, Earnings360, AllPennyStocks, DividendStocks, Daily Profit, Outsider Club, Prism MarketView, BillionDollarClub and Green Energy Stocks reported earlier on Nikola Corporation (NKLA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The Chinese automotive industry has run headlong into an issue that may prevent it from competing effectively in foreign markets: buyers are not buying electric cars manufactured in China. The east Asian nation’s auto industry has come a significantly long way over the past several decades, evolving from a sector that essentially produced clones of Western vehicles to one that’s now producing some of the best vehicles on the globe.
Years of major investments by Beijing have helped car manufacturers in China cut vehicle production costs by a wide margin, allowing them to price their cars at much lower price points compared to Western automakers. As such, Chinese offerings tend to be more affordable, despite being of a similar or even higher quality to cars manufactured in the West. This lower price point should give these Chinese companies a hypothetical leg up in a market pressured by high electric vehicle prices and high borrowing costs.
Indeed, the European Commission has even launched a probe into the cheap Chinese EVs streaming into Europe to determine whether the Chinese government used subsidies to lower its automakers’ electric vehicle prices. However, a growing number of Chinese electric cars are piling up at European ports, and sellers are struggling to find buyers, despite their lower prices. Some China-made electric cars have spent up to 18 months parked at port car parks because their manufacturers have failed to rouse buyer interest.
Although Chinese EVs have received plenty of popular reviews and typically feature high-quality technology, their makers aren’t having an easy time breaking into the century-old-plus automotive industry. Challenging the leaders of a sector as established as car making, where brand recognition is everything, is an uphill task. Challengers have little to no brand image and have to deal with trade protectionism as well as buyer wariness and limited buyer faith.
China’s automotive sector draws a comparison to Japan in the 1960s and 1970s when the country was attempting to move into Western car markets. Japanese carmakers offered commendable products that unfortunately lacked the longevity, design and finesse of Western-made cars. Many American buyers were also unwilling to buy cars made in the nation responsible for the Pearl Harbor attacks. Even so, by focusing on consistently producing reliable, affordable and increasingly well-designed vehicles, Japan became an automotive giant in the 1990s and 2000s.
China is currently in a similar position, facing plenty of suspicion from Western buyers who are wary of Chinese products and still remember all the illegal and legal clones of Western cars developed in China. The nation’s financial might may have helped its carmakers hire the best car-making experts on the globe and slash production costs, but China has been unable to buy customers’ loyalty so far. As with Japan, attracting and retaining customers will take consistently high-quality products combined with time.
The existing unease about Chinese EVs should give manufacturers such as Nikola Corporation (NASDAQ: NKLA) an advantage, which they should exploit and get a foothold in the European market for EVs. That advantage may not last for an unlimited time, so speed may be of the essence.
Nikola Corporation (NKLA), closed Monday's trading session at $0.6374, off by 3.057%, on 89,108,418 volume. The average volume for the last 3 months is 8.571M and the stock's 52-week low/high is $0.521/$3.71.
Hecla Mining Company (HL)
MarketClub Analysis, QualityStocks, SmarTrend Newsletters, Schaeffer's, InvestorPlace, StocksEarning, Wyatt Investment Research, MarketBeat, Lebed.biz, Top Pros' Top Picks, TopStockAnalysts, StreetAuthority Daily, DividendStocks, The Street, INO.com Market Report, StockEarnings, Money Morning, Zacks, Marketbeat.com, Jason Bond, Daily Trade Alert, Kiplinger Today, Wall Street Grand, StreetInsider, Today's Financial News, Trades Of The Day, MiningNewsWire, TradersPro, StockOodles, INO Market Report, Streetwise Reports, TheStockAdvisors, The Wealth Report, Gryphon Digest, Stockhouse, Penny Detectives, SureMoney, TradingAuthority Daily, Early Bird, National Inflation Association, Darwin Investing Network, ChartAdvisor, Options Elite, PennyStockLive, Penny Sleuth, Profit Confidential, ProfitableTrading, Wall Street Daily, The Growth Stock Wire, TraderPower, Daily Markets, Traders For Cash Flow, Greenbackers, Forbes, TradingMarkets, DrStockPick, Wealth Insider Alert, WealthMakers, Investopedia, CustomerService, CRWEWallStreet, CRWEPicks, CRWEFinance, Weiss Research, BestOtc, Barchart, Daily Wealth, StockHotTips, AllPennyStocks, PennyToBuck, MonsterStocksPicks, Money and Markets, Residual Income Report, Rockwell Trading, SmallCapVoice, Investing Futures, Stock Stars, Trade of the Week, The Online Investor, InvestorIntel, InvestorGuide, Investor Update, Investor Guide, Investing Lab, PennyOmega and MarketArmor.com reported earlier on Hecla Mining Company (HL), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Gold, silver and copper have been performing well recently, with their prices increasing as each day goes by. Investors looking to enrich their portfolios and capitalize on higher metal prices may benefit from injecting money into companies that are developing or exploring porphyries.
This is primarily because these deposits house a lot of copper, among other minerals. For instance, the Escondida mine in Chile is the biggest copper mine globally, with 40-year mine life.
It is expected that copper, silver and gold motherlodes will bring in huge returns if a commodity super cycle occurs. Motherlodes are principal zones or veins of silver or gold ore that are common along the west coast of North and South America. They are formed when dense oceanic plates subduct and collide below the continental crust, compressing the earth’s crust to form a chain of mountains that runs to Alaska, all the way from Patagonia.
It is this subduction that gives rise to huge porphyry deposits, where magmatic rocks combine with mineralized fluids. While silver and gold are considered byproducts of these zones, their quantities are sometimes so vast that they exceed total quantities at standalone mines.
For instance, Argentina’s Filo Del Sol project holds more than four million gold ounces as a by-product. This is large enough to be considered a gold mine on its own. Surprisingly, however, the company mainly focuses on copper, holding 3.2 billion pounds of the red metal. This is in addition to more than 150 million ounces of silver.
This and other porphyry deposits are an exciting opportunity for investors, and with the current mood for commodities shifting, a super cycle is bound to occur. Copper, in particular, is set to experience a boom in the commodity market. Increasing prices of the red metal may see prices of other base metals such as aluminum and nickel also rise.
This is why investors need to observe the copper market for any clues of an upcoming super cycle. Its highest price stands at $4.26 a pound, as of January 2023. The metal’s price going past $4.80 a pound may indicate a new rally. Currently, the metal still has some obstacles to overcome. When it does and if it reaches new highs as gold did, it will indicate the start of a new commodity super cycle.
Investors are advised to focus on companies holding geological assets of high quality, particularly those with porphyry deposits. For those looking to buy stock in companies focusing on only one of the trio, Hecla Mining Company (NYSE: HL) can be subjected to adequate due diligence by investors looking for exposure to silver.
Hecla Mining Company (HL), closed Monday's trading session at $5, up 5.4852%, on 10,983,802 volume. The average volume for the last 3 months is 3.141M and the stock's 52-week low/high is $3.33/$6.23.
The QualityStocks Company Corner
- Mullen Automotive Inc. (NASDAQ: MULN)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- Torr Metals Inc. (TSX.V: TMET)
- D-Wave Quantum Inc. (NYSE: QBTS)
- HealthLynked Corp. (OTCQB: HLYK)
- Horizon Fintex | Upstream
- Nutriband Inc. (NASDAQ: NTRB)
- Cepton Inc. (NASDAQ: CPTN)
- Turbo Energy S.A. (NASDAQ: TURB)
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF)
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI)
- Btab Ecommerce Group Inc. (OTC: BBTT)
Mullen Automotive Inc. (NASDAQ: MULN)
The QualityStocks Daily Newsletter would like to spotlight Mullen Automotive Inc. (MULN).
Mullen Automotive (NASDAQ: MULN), an electric vehicle ("EV") manufacturer, has added a commercial dealer to its growing list of dealerships. The company announced that National Auto Fleet Group ("NAFG") will offer Mullen's full line of all-electric commercial vehicles, specifically in Northern and Southern California. NAFG is a Sourcewell contract awardee and provides fleet vehicle purchasing to state, local government and educational institutions; Sourcewell is a government agency that has fulfilled bidding requirements and holds hundreds of competitively solicited contracts ready for use. "Everything you see, smell or touch was moved by truck at one time or another," said National Fleet Auto Group's Clarke Cooper in the press release. "With Mullen, we will offer municipal and government fleets the opportunity to work more efficiently, with a stronger TCO and with zero emissions. We believe Mullen's all-electric line provides municipalities and government fleets with the option they need to transition to electric. Further, the fact that these vehicles are assembled in the United States by Americans upholds the goals we value when we work with public fleets and Sourcewell."
In addition, Mullen announced that its board of directors unanimously adopted a limited duration stockholder rights plan. The announcement noted that the rights agreement is not a financing transaction and no additional shares of common stock or preferred stock or warrants or convertible notes are currently being issued in connection with the agreement. According to the announcement, the plan is designed to promote the fair and equal treatment of all stockholders and is much like rights plans adopted by other publicly held companies. Effective immediately, the plan will expire on May 1, 2025.
To view the full press releases, visit https://ibn.fm/DmlkG and https://ibn.fm/AzjQD
Mullen Automotive Inc. (NASDAQ: MULN) is a Southern California-based automotive company that owns and partners with several synergistic businesses working toward the unified goal of creating clean and scalable energy solutions. Mullen has evolved over the past decade in sync with consumers and technology trends. Today, the company is working diligently to provide exciting EV options built entirely in the United States and made to fit perfectly into the American consumer’s life. Mullen strives to make EVs more accessible than ever by building an end-to-end ecosystem that takes care of all aspects of EV ownership.
Commencement of Trading on Nasdaq
On November 5, 2021, Mullen announced its commencement of trading on the Nasdaq Capital Market.
“Today is a monumental day for Mullen Automotive. I am especially proud of our team, investors and all who have believed in Mullen and taken us to this point as a publicly traded company on the Nasdaq Capital Market,” David Michery, CEO and Chairman of Mullen Automotive, stated in the news release. “Trading on Nasdaq now opens us up to new investors, both institutional and retail shareholders, and broadens our awareness and company profile, while increasing awareness of Mullen and our technology platform and opening new opportunities in EV and beyond. The road ahead has never been brighter for Mullen, and I am proud to lead us into the future.”
The milestone came in the wake of the company’s stock-for-stock merger with Net Element Inc.
The Mullen FIVE
The Mullen FIVE EV Crossover, debuting at the Los Angeles International Auto Show (LAIAS) on November 17, 2021, embodies Mullen’s Southern California roots with an inspired design focused on two complementary Golden State themes – California landscape and California urban.
The FIVE is built on an EV Crossover skateboard platform that offers multiple powertrain configurations and trim levels in a svelte design that is Strikingly Different™ and exciting to experience in person.
Prior to the start of LAIAS, the Mullen FIVE was selected as a finalist by the LA Auto Show for Top EV SUV in the ZEVA “People’s Choice” Awards.
LAIAS provides Mullen an opportunity to display multiple variants of the FIVE model while also showcasing its powertrain, battery and charging technology. The company intends to bring the FIVE to market in 2024, and reservations are currently open here.
Mullen’s development portfolio also includes EV Fleet Vans, which it intends to bring to market in Q2 2022, and the pure electric, high performance Mullen DragonFLY.
Expansion of Manufacturing Capacity
On November 2, 2021, Mullen announced plans to expand its facility in Robinsonville, Mississippi.
Mullen’s Advanced Manufacturing and Engineering Facility (AMEC) currently occupies 124,000 square feet of manufacturing space. The total available land on the property is over 100 acres, and Mullen is moving ahead with plans to build out another 1.2 million square feet of manufacturing space to support class 1 and class 2 EV cargo vans and the Mullen FIVE EV Crossover.
On the expanded site, Mullen plans to build a body shop, a fully automated paint shop and a general assembly shop.
EV Market Outlook
The global EV market was reported to consist of 3,269,671 units in 2019, a figure that is expected to grow at a CAGR of 21.1% through 2030 to a total of 26,951,318 units worldwide. This market’s monetary value was estimated at $162.34 billion in 2019 and is expected to grow at a CAGR of 22.6%, resulting in an approximate value of $802.81 billion by 2027. The primary driver for this exponential growth is a worldwide increase in vehicle emissions regulations.
Management Team
David Michery is the CEO and Founder of Mullen and has been leading the company and its divisions since inception in 2014. With over 25 years of executive management, marketing, distressed assets, and business restructuring experience, Mr. Michery brings a wealth of relevant knowledge and expertise to the Mullen brand. He has notably created 12 trademarks so far to develop the company brand and vision.
Mr. Michery is working toward a sustainable future accessible to all by creating a suite of clean-energy electric vehicles at varied price points. With entirely U.S.-based manufacturing and operations, he is also determined to have Mullen Technologies play a role in shaping a self-sustaining local economy by creating more jobs in America.
Mr. Michery manages risks and company expectations as a pathway to success and has personally overseen several businesses that totaled over $1 billion in transactions. His key strength is the ability to be fiscally responsible and lead teams to complete projects on time and within budget. As a seasoned professional in this space, Mr. Michery has demonstrated skill in building businesses from the ground up and into successful entities that subsequently sold for hundreds of millions of dollars.
Mullen Automotive Inc. (MULN), closed Monday's trading session at $6.03, up 44.7779%, on 26,266,278 volume. The average volume for the last 3 months is 507,105 and the stock's 52-week low/high is $2.3565/$1458.00.
Recent News
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc.'s (NASDAQ: MULN) Inks Agreement with NAFG, Board Adopts Limited Duration Stockholder Rights Plan
- InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Partners with Pritchard EV, MRSS to Target 'Great Opportunity' within Roadside Service Market
- InvestorNewsBreaks - Mullen Automotive Inc.'s (NASDAQ: MULN) Announces Newest Franchise Partner as Pritchard EV Joins Dealer Network
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
Lexaria Bioscience (NASDAQ: LEXX, LEXXW), a global innovator in drug delivery platforms, has announced an applied research program to evaluate certain molecular characteristics of DehydraTECH processed with the glucagon-peptide 1 ("GLP-1") drug, semaglutide, related to its mode of action and performance. According to the announcement, the research will be conducted in partnership with the National Research Council of Canada ("NRC"). "We are excited about our partnership with the NRC for the mode of action molecular characterization of DehydraTECH-GLP-1 drugs. This work program could potentially provide data to assist Lexaria's efforts in partnering with the pharmaceutical industry for the most rapid introduction possible of DehydraTECH with GLP-1 drugs," said John Docherty, president of Lexaria. "The NRC was selected for this important work program because of its high-caliber research facilities and capabilities, further building upon the previous history of successful research projects between Lexaria and the NRC."
To view the full press release, visit https://ibn.fm/FoHC7 Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing. DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future. Lexaria has also collaborated with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has been granted patent protection for specific delivery of nicotine, vitamins, NSAIDs, antiviral drugs, cannabinoids and more. Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats. The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products. DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions. DehydraTECH Technology Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following: Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery. Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine. Market Outlook Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments: Management Team Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations. John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents. Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations. Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch. Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia. Lexaria Bioscience Corp. (LEXX), closed Monday's trading session at $3.41, up 8.9457%, on 294,182 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1458.00/$.
Recent News
Torr Metals Inc. (TSX.V: TMET)
The QualityStocks Daily Newsletter would like to spotlightFathom Torr Metals Inc. (TSX.V: TMET) .
REMOVE
Torr Metals Inc. (TSX.V: TMET) operates as a mineral exploration company focusing on the identification, acquisition, and advancement of mineral properties. With full 100% ownership of over 1,000 square kilometers of gold and copper projects strategically positioned in premier low-cost mining jurisdictions, Torr is poised for substantial returns across various promising regions.
The company’s extensive portfolio encompasses multiple district-scale projects, including the Filion Gold Project in northern Ontario, the Kolos Copper-Gold Project in south-central British Columbia, and the Latham Copper-Gold Project in northern British Columbia. These projects are all located in prolific mining regions with paved highway access, robust support infrastructure, and favorable geological conditions offering significant potential for new discoveries.
Headquartered in Vancouver, British Columbia, Torr Metals is ideally situated to leverage its expertise and resources for continued exploration and growth.
Projects
Kolos Copper-Gold Project
Situated within British Columbia’s prime copper-producing belt, the 140-square-kilometer Kolos Copper-Gold Project exhibits Nicola Belt geology similar to notable porphyry mines, including Copper Mountain and Highland Valley, respectively situated 106 kilometers to the south and 30 kilometers to the northwest.
With field operations based in the nearby city of Merritt and year-round access provided via Highway 5, the Kolos Project showcases substantial discovery upside potential with five defined large-scale copper-gold-molybdenum anomalies untested by drilling.
Torr Metals’ primary focus lies in unlocking the potential for major new discoveries at the Kolos Copper-Gold Project, with recent surface geochemical results marking a significant milestone positioning the company as a new key player in the region.
Filion Gold Project
The 261-square-kilometer Filion Project is situated within a largely unexplored greenstone belt where gold was initially discovered in the 1930s. With a comparable geological setting to regional orogenic gold deposits and multiple newly identified and undrilled gold trends in surficial geochemistry, the Filion Project holds significant district-scale exploration promise.
The Filion Project benefits from unparalleled infrastructure access, with direct drive-on access from the Trans-Canada Highway, as well as a regional railway and power grid four kilometers to the south. Additionally, the nearby town of Kapuskasing, with a population of 8,300, provides essential support services.
This strategic positioning ensures the Filion Project’s viability for cost-effective, year-round operations in an area poised for untapped discovery potential.
Latham Copper-Gold Project
Situated in British Columbia’s renowned Golden Triangle, the Latham Project spans a vast 689-square-kilometer district, offering immense potential for multiple major discoveries. Accessible year-round via Highway 37, just 20 kilometers south of the town of Dease Lake, the site is strategically located amidst established mining infrastructure, including the active Red Chris mine to the southeast and upcoming major porphyry projects at Schaft Creek and Galore Creek along-trend to the southwest.
Highlighted by the Gnat Pass copper-gold porphyry deposit dating back to the 1960s, the Latham Project presents a compelling opportunity for significant expansion and potential discovery. A non-compliant indicated resource at the Gnat Pass deposit includes 33 million tonnes at 0.39% copper, open beyond 200 meters vertical depth, alongside six drill-ready kilometer-scale copper-gold exploration targets.
Moreover, the Latham Project’s appeal corresponds to the region being an attractive destination for major asset acquisitions and takeovers. Recent transactions within a 40-kilometer radius include Newmont’s 2021 acquisition of the Saddle North copper-gold porphyry deposit for $311 million and Newcrest’s investment in the Red Chris copper-gold porphyry deposit in 2019 for $804 million, underscoring industry acknowledgment of the region’s potential.
Market Opportunity
The World Gold Council, the industry association for the world’s gold producers, estimated in 2023 that the physical financial gold market, which is made up of bars, coins, gold ETFs and central bank reserves, is worth nearly $5 trillion.
The council reports that gold mine production adds approximately 3,500 tons of the precious metal to the world’s supply annually, equivalent to about 2% growth. This historical scarcity and relatively slow production of new supply, as compared to other commodities, is a primary reason gold has retained its value for millennia, according to the council.
Likewise, a report from Acumen Research and Consulting, a global provider of market intelligence and consulting services, valued the global copper market at $304.1 billion in 2022 and forecast that it will reach a market size of $496.8 billion by 2032, growing at a CAGR of 5.1% over the forecast period.
The report identifies a growing demand for copper in the electronics industry, as well as an expanding copper supply due to increasing production from existing mines and the rising number of mine development projects in developing nations, as driving factors in the rising value of the copper market.
Management Team
Malcolm Dorsey, P.Geo., is President, CEO and Director of Torr Metals. He brings over a decade of expertise as a seasoned exploration geologist and project developer, having been pivotal in driving the success of numerous diverse projects across North, Central, and South America. His comprehensive background spans early-stage exploration through to resource development and project acquisitions. His academic credentials include an M.Sc. in Geology and Geophysics from the University of Calgary, where his research characterized the district-scale structural influences affecting copper and gold mineralizing events in western British Columbia. Prior to his current role, he served as Senior Geologist for Benchmark Metals, where his contributions were instrumental in advancing the company’s gold equivalent resource from approximately 80,000 ounces to a maiden resource estimate of 2.92 million ounces.
John Williamson, P. Geol., is Chairman and Director of Torr Metals. He is a mining executive and investor with more than 30 years of experience as a founder, promoter and leader in the formation, financing and operation of private and public companies with exploration and mining interests worldwide. On more than one occasion his team’s efforts have been recognized for excellence by being named to the TSX Venture 50. He holds a B.Sc. in Geology and is a registered Professional Geologist (P.Geol.) with the Association of Professional Engineers and Geoscientists (APEGA) and the Geological Association of Canada.
Torr Metals Inc. (TSX.V: TMET), closed Monday's trading session at $28.935, up 0.9595255%, on 425 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $24.01/$29.38.
Recent News
- Torr Metals Inc. (TSX.V: TMET) - Unlocking the Riches: Torr Metals Inc. (TSX.V: TMET) Aims to Test Untapped Copper and Gold Potential in British Columbia and Ontario, Canada
- InvestorNewsBreaks - Torr Metals Inc. (TSXV: TMET) Recent Kolos Copper-Gold Project Assay Results Spotlighted in Syndicated Broadcast
- InvestorNewsBreaks - Torr Metals Inc. (TSXV: TMET) CEO Discusses Preliminary Geophysical Survey Results in Latest Proactive Interview
D-Wave Quantum Inc. (NYSE: QBTS)
The QualityStocks Daily Newsletter would like to spotlight D-Wave Quantum Inc. (NYSE: QBTS).
Leading experts in the fields of genomics and quantum computing have teamed up to create new methods and algorithms for processing biological data using next-generation computing. For many genome experts, mapping out the human genome is a dream that has been out of reach since the genome was first identified. Analyzing and fully mapping the genome and accounting for diversity could superboost the understanding of DNA and lead to the development of personalized medicines. Humanity's limited technological capabilities in the face of something as complex as mapping out the entire human genome has kept us from achieving this lofty dream so far, however. Fortunately, consistent advances in computing technology have steadily brought us closer and closer to analyzing and understanding the human genome in its entirety. With the new project drawing leading experts in both genomics and quantum computing, it could potentially lead to a breakthrough that helps researchers leverage the power of quantum computing in genomics and expand their understanding of human DNA. Most importantly, such a breakthrough could open the door to the eventual development of personalized treatments for conditions that don't typically respond to conventional medicines. With the increasing availability of quantum computers from entities such as D-Wave Quantum Inc. (NYSE: QBTS), quantum computing could make inroads into many more medical research functions and help to improve our understanding of conditions that have remained a mystery for decades.
D-Wave Quantum Inc. (NYSE: QBTS) is a leader in quantum computing systems, software and services focused on delivering customer value via practical quantum applications for problems such as logistics, artificial intelligence, materials sciences, drug discovery, scheduling, fault detection and financial modeling. As the only provider building both annealing and gate-model quantum computers, the company is unlocking commercial use cases in optimization today, while building the technologies that will enable new solutions tomorrow.
D-Wave is a pioneer in quantum computing, with a history of delivering the world’s first commercial quantum computer; the first real-time quantum cloud service; countless hardware and software product and research milestones; and the planned first cross-platform quantum solution which will deliver both annealing and gate-model quantum computers to customers via an integrated platform. Its current commercial product offerings include: Advantage™ (fifth generation quantum computer), Leap™ (quantum cloud service), Launch™ (quantum computing onboarding service) and Ocean™ (full suite of open-source programming tools).
D-Wave’s relentless pursuit of practical quantum computing has resulted in the technology being used today by some of the world’s most advanced enterprises – more than 25 of the Forbes Global 2000 use D-Wave.
D-Wave’s commercial customers include blue-chip industry leaders like Volkswagen, Accenture, BBVA, NEC Corporation, Save-On-Foods, DENSO and Lockheed Martin. The company boasts an extensive IP portfolio featuring more than 200 issued U.S. patents and over 100 peer-reviewed papers published in leading scientific journals.
Founded in 1999, D-Wave is the world’s first commercial supplier of quantum computers. With headquarters and the Quantum Engineering Center of Excellence based near Vancouver, Canada, D-Wave’s U.S. operations are based in Palo Alto, California.
Advantage™ Quantum Computer
With the Advantage™ Quantum Computer, D-Wave has incorporated two decades of experience and over 10 years of customer feedback to create the first and only quantum computer designed for business. The platform features a new processor architecture with over 5,000 qubits and 15-way qubit connectivity. This is 2.5x more connections and more than double the number of qubits than the company’s previous generation quantum computer.
D-Wave’s quantum computers, first located in its facilities in British Columbia, have been available to North American users through its Leap™ quantum cloud service since 2018. It has since introduced new Advantage systems in Julich, Germany, and most recently, Marina Del Rey, California, which marked the availability of the first Advantage quantum computer physically located in the United States.
That new deployment is part of the USC-Lockheed Martin Quantum Computing Center (QCC) hosted at USC’s Information Sciences Institute (ISI), a unit of the University of Southern California’s prestigious Viterbi School of Engineering. Additionally, Amazon Web Services (AWS) and D-Wave announced that the U.S.-based system is available for use in Amazon 2racket, expanding the number to three different D-Wave quantum systems available to AWS users.
Leap Quantum Cloud Service
D-Wave’s customers interface with its systems through the Leap™ quantum cloud service. Leap delivers immediate, real-time access to the company’s Advantage quantum computer and quantum hybrid solver service, all with enterprise-class performance and scalability.
Leap allows developers proficient in Python to get started building and running quantum applications. Through a seamless and secure cloud-based connection, users can easily start solving complex problems of up to 1 million variables and 100,000 constraints.
Using Leap, D-Wave customers have developed quantum hybrid applications for use cases in manufacturing, logistics, financial services, life sciences, materials science, retail and transportation. By eliminating the need to wait hours, days or weeks to get good answers to a broad array of problems, D-Wave is helping businesses move forward.
D-Wave Launch
D-Wave Launch™ is the company’s onboarding platform aimed at helping businesses easily start their quantum journey. Through this program, D-Wave’s team of experts and partners aid enterprises in identifying best use cases for quantum and work with them to develop a proof of concept and production pilot.
From there, the team coordinates with customers to get their hybrid quantum applications up and running, providing ongoing Leap quantum cloud access to ensure the application is operating smoothly and delivering real business value.
Target Verticals
While the potential applications for quantum computing are effectively limitless, D-Wave has identified a number of industry verticals as key areas of focus for its quantum architecture, providing case studies for each. These include:
- Manufacturing – D-Wave worked with Volkswagen to identify a commercial optimization application, the binary paint shop problem, which was run on D-Wave’s hybrid solver service. The solver outperformed four purely classical methods on problem sizes at commercial scale (N=3,000). In a separate project, similar inputs were tested using a leading ion trap system, which failed to find any commercial solution.
- Life Sciences – Menten AI makes use of D-Wave quantum computing to assist in the design of novel therapeutic peptides—short strings of amino acids that can act as potent drugs. With the rise of COVID-19, D-Wave’s Advantage system made it possible to identify molecules that might be especially well-suited for binding and inhibiting the related spike protein, producing several promising peptide designs.
- Finance – Multiverse Computing, a leader in developing quantum solutions for the financial sector, leveraged D-Wave’s hybrid solver service in a collaboration with BBVA, one of the world’s largest financial institutions. Multiverse demonstrated management strategies that far exceeded the granularity of traditional returns in a fraction of the time, helping BBVA identify a low-risk portfolio for investment.
Market Opportunity
The quantum computing total addressable market is projected to grow between $450 billion and $850 billion over the next 15 to 30 years, with between $5 billion and $10 billion of anticipated TAM growth coming in the next three to five years, according to Boston Consulting Group. Driving factors behind this growth include rising investments in quantum computing tech by governments and an increasing number of commercial use-cases.
Forward-thinking organizations see quantum as an opportunity to move ahead of the competition. From finding efficiencies and reducing waste to decreasing time to solution and solving problems abandoned due to complexity, the business value is real. According to data from 451 Research, 40% of large enterprises are already experimenting with quantum computing.
D-Wave is strategically positioned – in an industry with significant barriers to entry – as evident by a decades-long track record serving a roster of blue-chip customers. The company is singularly focused on helping its customers achieve clear value by leveraging quantum computing in practical business applications. With a full stack of systems, software, developer tools and services, D-Wave is working to enable enterprises, governments, developers and researchers to access the power of quantum computing, thereby providing an intriguing opportunity for prospective investors.
D-Wave’s current investor base includes PSP Investments, Goldman Sachs, BDC Capital, NEC Corporation, Aegis Group Partners and In-Q-Tel.
Leadership Team
Dr. Alan Baratz has served as the CEO of D-Wave since 2020. Previously, as Executive Vice President of R&D and Chief Product Officer, he drove the development, delivery, and support of all of D-Wave’s products, technologies, and applications. Dr. Baratz has over 25 years of experience in product development and bringing new products to market at leading technology companies and software startups. As the first president of JavaSoft at Sun Microsystems, he oversaw the growth and adoption of the Java platform from its infancy to a robust platform supporting mission-critical applications in nearly 80 percent of Fortune 1000 companies. He has also held executive positions at Symphony, Avaya, Cisco, and IBM. Dr. Baratz holds a doctorate in computer science from the Massachusetts Institute of Technology.
John Markovich is the company’s CFO. He brings to D-Wave over three decades of experience working with rapidly growing private and public technology companies across all stages of development. Mr. Markovich has directed the finance, accounting, tax, treasury, M&A, legal, operations, customer service, IR, HR, and IT functions for companies ranging from privately held pre-revenue startups to an NYSE-listed Fortune 500 multi-national company with over $1.2 billion in annual revenue. During his career, he has negotiated and closed over 150 debt, equity, M&A, and joint venture transactions exceeding $2.5 billion in value; over a dozen private placements; nearly a dozen M&A transactions; and several international joint ventures. Mr. Markovich holds a BS in Business from Miami University and an MBA from the Michigan State Graduate School of Business.
D-Wave Quantum Inc. (NYSE: QBTS), closed Monday's trading session at $1.41, up 0.7142857%, on 4,456,060 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.3962/$3.20.
Recent News
- D-Wave Quantum Inc. (NYSE: QBTS) - Quantum Computing Could Make Genomic Analysis Hyper-Fast
- D-Wave Quantum Inc. (NYSE: QBTS) Announces Fast-Anneal Feature Availability in Leap(TM) Quantum Cloud Service
- InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Announces Date for Q1 2024 Earnings
HealthLynked Corp. (OTCQB: HLYK)
The QualityStocks Daily Newsletter would like to spotlightFathom HealthLynked Corp. (OTCQB: HLYK) .
HealthLynked achieved significant optimization in operational expenses, resulting in a 70% reduction in net loss for the fourth quarter and highlighting a period of strategic positioning for future growth
HealthLynked's full-year 2023 financial results mark technological advancements, with a focus on core technologies and profitability
The company anticipates further performance improvements in the second half of 2024, positioning itself as a leader in healthcare technology
HealthLynked (OTCQB: HLYK), a leader in healthcare networking and technological innovation, recently announced its financial results for the fourth quarter and full year ending December 31, 2023. HealthLynked achieved a significant 70% reduction in net loss for the fourth quarter, highlighting a period of strategic advancements and focus on healthcare technologies plus networking capabilities (https://ibn.fm/ycgvs).
HealthLynked Corp. (OTCQB: HLYK) is at the forefront of a transformative movement in healthcare, utilizing its extensive collection of health data to improve care for all. With a commitment to leveraging its advanced technology platforms, HealthLynked employs a sophisticated, cloud-based network that serves as a comprehensive repository for personal health data. This system not only simplifies the management and archiving of medical records but also enables the application of AI to deliver personalized healthcare insights. Through deep analysis of this data, HealthLynked’s AI capabilities help identify the root causes of diseases, tailor healthcare solutions to individual needs, and accelerate medical discoveries.
In addition to these capabilities, HealthLynked provides a user-friendly platform for booking healthcare appointments, similar to how OpenTable operates for restaurant reservations. This feature allows patients to conveniently book appointments with healthcare providers across the country, including options for telemedicine consultations, enhancing accessibility and efficiency in healthcare service delivery.
Strategically headquartered in Naples, Florida, HealthLynked operates through three primary divisions: Health Services, Digital Healthcare, and Medical Distribution. Each division supports the company’s mission to revolutionize patient care and health management. Positioned as a potential leader in healthcare AI, HealthLynked is dedicated to shaping the future of the industry over the next 20 years, driving significant advancements in healthcare accessibility and effectiveness through innovation and technology.
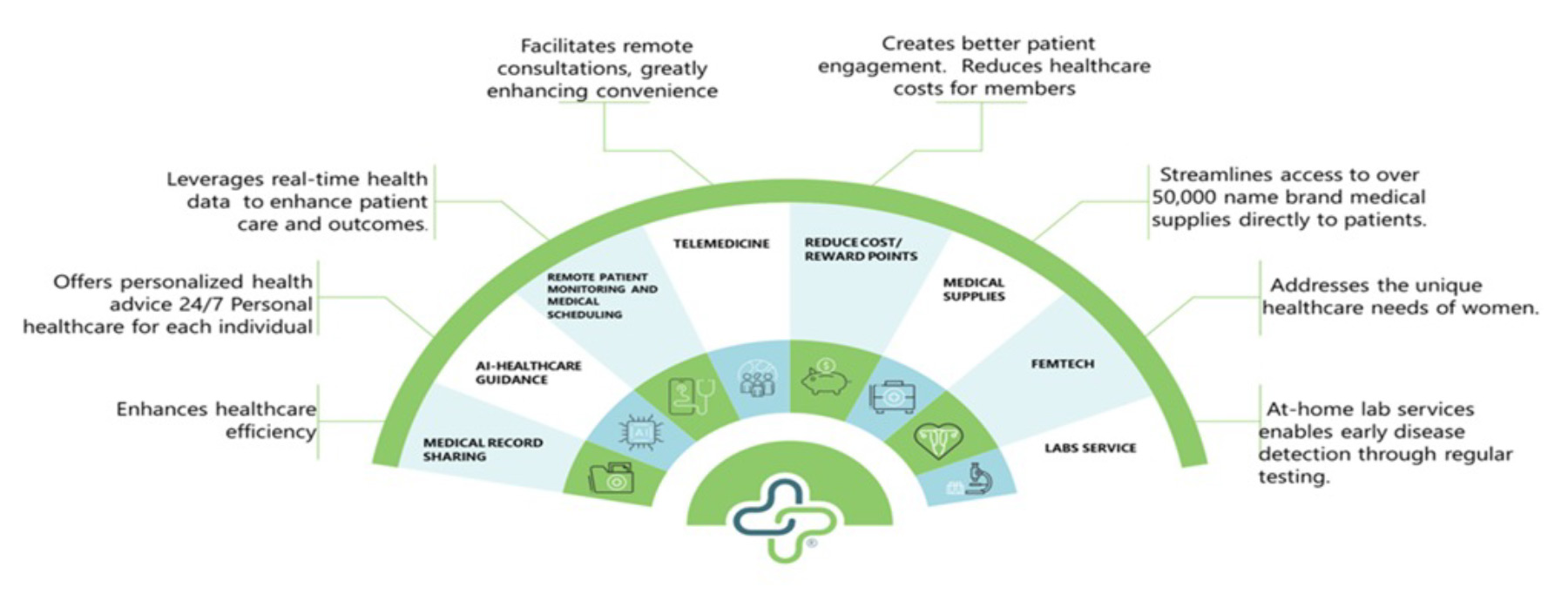
Strategic Initiatives and Operational Highlights
The company’s commitment to enhancing global health is evident in its dual goals: transforming healthcare through advanced technology and creating a patient-centric network that accelerates medical discoveries and the development of disease cures.
HealthLynked’s intellectual property portfolio is robust and strategically developed to enhance healthcare delivery and management. In March 2023, HealthLynked was granted a patent for a groundbreaking healthcare-specific wireless access point, known as the “Patient Access Hub.” This technology significantly improves the efficiency of healthcare practices by enabling real-time monitoring of patient flow within facilities. It intelligently determines patients waiting in exam rooms and calculates wait times, alongside other critical practice metrics. This system not only enhances patient experience by reducing unnecessary wait times but also optimizes resource allocation within healthcare settings.
Additionally, in October 2023, HealthLynked filed a patent application for its advanced AI program, ARI (Augmented Real-time Interface). ARI acts as a virtual doctor for patients, capable of performing medical intake, booking appointments, and providing personalized medical recommendations based on a patient’s medical history. By integrating these tasks, ARI streamlines the healthcare process, reducing the administrative burden on healthcare providers and ensuring that patients receive timely and tailored healthcare advice. This AI-driven interface enhances the accessibility and personalization of healthcare, embodying HealthLynked’s commitment to leveraging technology for better health outcomes. The company recently launched HealthLynked 3.2.0, an advanced version of its application, incorporating telemedicine, AI-driven personal healthcare guidance, and remote patient monitoring – setting a new standard in healthcare technology.
Market Position and Future Outlook
According to Facts and Figures Research, a research and consulting firm, the global market for patient-centric healthcare applications is projected to reach $41.6 billion by 2030, growing at a CAGR of 18.77% from 2022. HealthLynked’s offerings align perfectly with this expansive market opportunity, especially with increasing demands for digital health solutions and data management in healthcare.
HealthLynked’s strategic direction, spearheaded by its seasoned management team, is designed to leverage these market dynamics, enhancing patient engagement and healthcare efficiency on a global scale.
Management Team
Michael T. Dent, M.D., Founder, CEO, and Chairman, brings extensive experience from his foundational role at NeoGenomics and leadership in various healthcare and technology sector companies.
David Rosal, CFO, with previous senior roles at Teradata and McDonald’s Corporation, brings a wealth of expertise in financial and business integration strategies essential for growth and operational efficiency.
Chris Hall, CTO, with a strong background in global technology development from his time at Siemens and several patents to his name, is instrumental in driving the innovation and technological advancement at HealthLynked.
Bill Crupi, Operations Manager, has a proven track record in streamlining operations and enhancing productivity across multiple sectors within the healthcare industry. His expertise is crucial in maintaining the operational excellence that HealthLynked is known for.
Michael Paisan, Director of Investor Relations, leverages his extensive experience in finance and communications to enhance HealthLynked’s relationships with investors and stakeholders, ensuring transparent and effective communication of the company’s value and growth strategy.
Gagan Babber, Manager of Software Development, oversees the HealthLynked development teams based in the U.S. and India. With a robust background in engineering and software development, he plays a critical role in guiding the technological direction of HealthLynked’s products. His expertise in developing scalable, innovative software solutions is essential for driving the company’s technical initiatives forward and ensuring that HealthLynked stays at the forefront of digital healthcare technology.
HealthLynked Corp. (OTCQB: HLYK), closed Monday's trading session at $0.058, up 1.1334%, on 1,738 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.033/$0.0999.
Recent News
- HealthLynked Corp. (OTCQB: HLYK) - HealthLynked Corp. (HLYK) Q4, FY2023 Financial Results; Show Bottom-line Improvements, Core Network Application Ramp-Up Expected in 2024
- Simplifying Healthcare Appointments: How HealthLynked Corp. (HLYK) is Revolutionizing Online Booking with a Patient-Focused Approach
- InvestorNewsBreaks - HealthLynked Corp. (HLYK) Announces Strategic Partnership with IBN
Horizon Fintex | Upstream
The QualityStocks Daily Newsletter would like to spotlight Horizon Fintex | Upstream
Upstream, a MERJ Exchange market and trading app, recently published a blog written by Fernanda De La Torre and Vanessa Malone. The piece reiterates strategic benefits that dual listing offers to companies aiming to appeal to international investors and shifts focus to the essential factors that issuers should consider when contemplating dual listing and how Upstream addresses these challenges.
"Dual listing continues to be used as a growth strategy for issuers as they go global. It's important to think about regulatory compliance, costs and management resources inherent in this process," the blog reads. "With our streamlined approach and comprehensive support services, companies can leverage dual listing as a strategic tool to access global capital markets and drive sustainable growth."
To view the full blog, visit https://ibn.fm/2be80
Horizon Fintex is a software business specializing in compliant securities solutions. The company aims to facilitate the future of capital markets by leveraging the regulatory experience of Wall Street bankers and the proven track record of technology veterans to bring focus to compliance, efficiency, security and transparency.
Horizon’s flagship product is the revolutionary trading app ‘Upstream’, a MERJ Exchange Market, and the first regulated market powered by a blockchain to offer both digital securities and NFT trading. Upstream traders experience T+0 settlement, best bids and offers displayed on a transparent public orderbook that prevents predatory market practices – all from a user-friendly trading app.
Products
Horizon Fintex offers a full suite of end-to-end blockchain-enhanced software solutions to create a seamless experience for both issuers and investors. Its product suite includes:
- Securitization & Issuance – ETSware is an end-to-end Electronic Trading System streamlining capital raising from primary issuance through compliant secondary trading.
- KYC Compliance Onboarding – KYCware is a white label Know Your Customer (KYC) and Anti-Money Laundering (AML) compliance software solution offering best-in-class cryptographic security to compliantly onboard and verify user identity through a smartphone application.
- AML Screening Software – AMLCop offers advanced Anti-Money Laundering (AML) software to streamline the verification of user details against a proprietary database of global sanctions, politically exposed persons (PEPs) and watchlists.
- Cap. Table Management Tools – CustodyWare equips registered U.S. transfer agents with next-generation cap. table management software to manage securities on behalf of their clients pursuant to an SEC-registered or exempt securities offering.
- Exchange & Trading App Technology – Open Order Book offers Ethereum blockchain securities exchange software to power the next generation of trading venues for digital assets.
Upstream – The Horizon-Powered Trading App
Upstream is a joint venture with MERJ Exchange (merj.exchange), an affiliate of the World Federation of Exchanges.
Upstream aims to be the premiere global trading hub offering issuers around the world exposure to a digital-first investor base that can trade using USDC digital currency along with credit, debit, PayPal, and USD (fiat) to increase liquidity and enhance price discovery; while also offering investors access to dual-listed companies, IPOs, crowdfunded companies, U.S. & Int’l. equities, digital coupons and NFTs directly from a user-friendly trading app.
Upstream aims to unlock liquidity for investors of all levels while offering industry-leading levels of transparency, accessibility and investor protections enforced using Ethereum blockchain technology.
Management Team
Brian Collins is the CEO of Horizon Fintex. He founded the company in 2010. From 1999-2010, Mr. Collins was CEO of Abbey Technology in Switzerland, specializing in the design of trading software for Swiss banks. Prior to this, he worked for Credit Suisse in Zürich, designing and building proprietary equity trading solutions. Mr. Collins graduated in 1990 with a BS in Computer Systems from the University of Limerick, Ireland.
Mark Elenowitz is the company’s President. He is a Wall Street veteran with over 29 years of experience. Mr. Elenowitz was the co-founder of a U.S. broker dealer and is Managing Director of two U.S. broker dealers, responsible for advising clients on compliance, capital structure and capital market navigation. He was responsible for leading the first successful Reg A+ IPO of a company to list on the NYSE and others which listed directly onto Nasdaq. He is a noted speaker at Small Cap and Reg A events, including the SEC Small Business Forum, and has been profiled in BusinessWeek and CNBC, as well as several other publications. Mr. Elenowitz is a graduate of the University of Maryland School of Business and Management with a BS in Finance and holds Series 24, 62, 63, 79, 82 and 99 licenses.
Dr. Andrew Le Gear is the CTO of Horizon Fintex. Prior to joining the company in 2013, he worked as a software engineer with Dell Inc. (2012-2013) and Lehman Brothers and Nomura Plc. (2007-2012). Dr. Le Gear was a co-founder of Juneberi Ltd., a research-driven software tech start-up (2004-2007). He graduated in 2006 with a Ph.D. in Computer Science from the University of Limerick, Ireland.
Peter Hall is the company’s CIO. Prior to joining Horizon Fintex in 2011, he worked at Microsoft (2008-2011), Atos Origin (2004-2008) and AIT Group Plc. (1998-2002). Mr. Hall has held CISSP certification since 2010. He graduated from the University of Sheffield, UK in 1995 and earned an MS from the University College London in 2006.
Mike Boswell is the CFO of Horizon Fintex. A Wall Street veteran, he co-founded a U.S. broker dealer and served as Chief Compliance Officer. Mr. Boswell was also Managing Director of TriPoint Capital Advisors, a merchant banking and financial consulting company, and CFO of Mission Solutions Group, a privately held defense sector firm. He earned an MBA from John Hopkins University and a BS in Mechanical Engineering from the University of Maryland. Mr. Boswell holds Series 24, 62, 63, 79, 82 and 99 licenses.
Recent News
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Delves into Key Dual Listing Considerations for Issues
- Google Cloud Launches Web3 Portal, Receives Mixed Reactions from Crypto Industry
- InvestorNewsBreaks — Upstream Offers Companies Global Investor Base Through Dual Listing

Nutriband Inc. (NASDAQ: NTRB)
The QualityStocks Daily Newsletter would like to spotlightFathom Nutriband Inc. (NASDAQ: NTRB) .
150+ Americans die every day from synthetic opioid (e.g. fentanyl) overdose
Nutriband's AVERSA(TM) Fentanyl technology is being developed as an innovative solution to abuse and misuse rates in prescription opioid patch medications
Nutriband raised $8.4 million in a private placement to fund AVERSA Fentanyl through FDA Approval Process
Fentanyl addiction continues to devastate communities, with current treatment options facing limitations. However, as often happens in healthcare, innovation breeds hope with a new option on the horizon planning to seek FDA marketing approval. The challenges related to the powerful painkiller (up to 50x stronger than heroin and 100x stronger than morphine) are well documented. According to the Centers for Disease Control and Prevention, more than 150 Americans die every day from overdoses related to synthetic opioids like fentanyl. Nutriband (NASDAQ: NTRB) is looking to provide a much-needed new option for managing severe and chronic pain that requires the use of an opioid (https://ibn.fm/7AYt6). The Orlando-based company stated last month that it raised gross proceeds of $8.4 million towards the remaining clinical development of AVERSA(TM) Fentanyl and submission of its New Drug Application ("NDA") with the U.S. Food and Drug Administration.
Nutriband Inc. (NASDAQ: NTRB) is engaged in the development of a portfolio of transdermal pharmaceutical products. The company’s AVERSA™ technology can be incorporated into any transdermal patch and includes aversive agents to prevent abuse, diversion, misuse and accidental exposure to drugs with abuse potential, specifically opioids.
AVERSA technology has the potential to improve the safety profile of transdermal drugs susceptible to abuse, such as fentanyl, while making sure that these drugs remain accessible to patients who need them. The technology is covered by a broad intellectual property portfolio with patents granted in the United States, Europe, Japan, Korea, Russia, Canada, Mexico and Australia.
The company’s business model is to apply its transdermal technology to existing FDA-approved drugs with a goal of improving safety, efficacy and patient comfort while qualifying for a limited-development regulatory pathway that reduces the number of clinical trials required for approval of new drugs.
Nutriband has three subsidiaries, including 4P Therapeutics, its clinical and regulatory subsidiary; Pocono Pharmaceutical, a contract manufacturer for a wide range of clients; and Active Intelligence, a developer of sports recovery products. This ownership of manufacturing and clinical development capabilities drastically reduces costs for AVERSA and other technologies.
In April 2024, Nutriband announced that the company had been engaged by and received a first order from Fit For Life Group, a major brand license holder. A fully executed supplier agreement is expected to follow. Nutriband’s wholly owned Active Intelligence subsidiary will act as manufacturer.
The company is headquartered in Orlando, Florida.
Products
Nutriband’s lead product candidate is AVERSA Fentanyl, an abuse-deterrent fentanyl transdermal patch. The company announced in March 2024 that it will submit a New Drug Application to the U.S. Food and Drug Administration seeking approval to market AVERSA Fentanyl.
Nutriband has partnered with Kindeva Drug Delivery, a leading global contract development and manufacturing organization, to incorporate Nutriband’s AVERSA abuse-deterrent transdermal technology into Kindeva’s FDA-approved transdermal fentanyl patch system. Because Nutriband’s abuse-deterrent technology is incorporated into the fentanyl patch but is physically separate from and does not come in contact with the drug layer, the clinical trials typically needed to demonstrate safety and efficacy for a new drug formulation would not be required.
AVERSA Fentanyl has the potential to be the first and only abuse deterrent patch approved anywhere in the world. The company plans to seek an expedited review by the FDA, as has been granted for certain abuse-deterrent oral opioid products, which shortens the regulatory review period to six months from the conventional 10-month FDA review cycle for NDAs.
Nutriband’s AVERSA product development pipeline also includes abuse deterrent versions of currently approved and marketed transdermal patches containing buprenorphine, an opioid used to treat opioid use disorder, and methylphenidate, a central nervous system stimulant used in the treatment of attention deficit hyperactivity disorder (ADHD). Both are labeled with FDA-required warnings for the risk of abuse and misuse, as well as warnings against accidental exposure.
Market Opportunity
Nutriband cites a market analysis report from Boston-based Health Advances, a healthcare and life sciences consulting firm. According to the report, upon FDA approval, AVERSA Fentanyl has the potential to reach peak annual sales of $200 million in the U.S.
The company further states that, should non-abuse-deterrent transdermal fentanyl products lose FDA marketing approval, AVERSA Fentanyl would have greater pricing flexibility and would have the potential to generate more than $500 million in annual revenue.
Management Team
Gareth Sheridan is Co-Founder and CEO of Nutriband. He was Ireland’s ‘Young Entrepreneur of the Year’ in 2014 for establishing Nutriband. He has worked as a Business Mentor with 100 Minds, a social enterprise that brings together some of Ireland’s top college students and connects them with a cause to achieve large charitable goals. He received a B.Sc. in Business and Management from Dublin Institute of Technology.
Serguei Melnik is Co-Founder and President of Nutriband. He has been involved in general business consulting for companies in the U.S. financial markets and setting up legal and financial frameworks for operations of foreign companies in the U.S. He previously was the COO of Florida-based Asconi Corporation. He also was a lawyer in the Department of Foreign Affairs, JSC Bank “Inteprinzbanca,” in Chisinau, Moldova, and prior to that practiced law in Moldova. He is fluent in four languages.
Jeff Patrick, Pharm.D., is Chief Scientific Officer of Nutriband. He currently serves as Director of the Drug Development Institute at the Ohio State University Comprehensive Cancer Center. His prior roles included Global Vice President at Mallinckrodt Pharmaceuticals Inc.; and roles at Dyax, Myogen/Gilead, Actelion and Sanofi-Synthelabo Inc. He was a clinical pharmacist at the University of Tennessee Medical Center and a clinical assistant professor of pharmacy at the University of Tennessee College of Pharmacy.
Gerald Goodman is CFO of Nutriband. He is a certified public accountant with his own firm, Gerald Goodman CPA. He also practiced with Madsen & Associates, CPAs, and was a partner in the accounting firm of Wiener, Goodman & Company. He is also a director of Lifestyle Medical Network Inc., which provides management services to healthcare providers. He is a graduate of Pennsylvania State University, where he received a bachelor’s degree in accounting.
Nutriband Inc. (NASDAQ: NTRB), closed Monday's trading session at $3.4, off by 2.5788%, on 11,928 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.53/$5.9274.
Recent News
- Nutriband Inc. (NASDAQ: NTRB) - Fentanyl Scourge: Nutriband Inc.'s (NASDAQ: NTRB) New Transdermal Patch with Abuse-Deterrent Tech Funded to Seek FDA Approval
- InvestorNewsBreaks - Nutriband Inc. (NASDAQ: NTRB) Engages IBN for Corporate Communications Expertise
- InvestorNewsBreaks - Nutriband Inc. (NASDAQ: NTRB) Closes $8.4M Private Placement
Cepton Inc. (NASDAQ: CPTN)
The QualityStocks Daily Newsletter would like to spotlight Cepton Inc. (NASDAQ: CPTN).
Cepton (NASDAQ: CPTN), a Silicon Valley innovator and leader in high-performance lidar solutions for automotive and smart infrastructure applications, has unveiling StudioViz(TM), its proprietary lidar simulation platform. The powerful 3D-point cloud simulation platform is designed to expedite ADAS and AV development while also reducing implementation costs for automotive customers. StudioViz was designed by in-house Cepton software teams and features strategic functionalities such as virtual environment creation, high-fidelity lidar data visualization, placement optimization, sensor fusion assessment, perception solution development, and manufacturing validation and scaling."StudioViz accelerates the testing, development and deployment of Cepton's lidar technology by offering an efficient and cost-effective solution," said Cepton product owner Christian Nickolaou. "The deployment of lidar at scale is such a complicated process that many things need to be tested and validated before a solution can be even considered for use case-specific volume adoption, such as sensor performance, system compatibility and mounting locations. Upfront time, labor and monetary investments in those early stages of the deployment can be monumental, if OEMs heavily rely on physical implementation and benchmarking."
To view the full press release, visit https://ibn.fm/FjHlv
Cepton Inc. (NASDAQ: CPTN) is a provider of state-of-the-art, intelligent, lidar-based solutions serving a range of markets, including automotive (ADAS/AV), smart cities, smart spaces and smart industrial applications. General Motors (NYSE:GM) has granted a series production award for Cepton’s lidar, the biggest such award to date in the automotive space. Cepton’s is the lidar component of GM’s Ultra Cruise autonomous driving platform. By leveraging its patented Micro Motion Technology (MMT®) lidar platform, the company develops reliable, scalable and cost-effective solutions that deliver long-range, high-resolution 3D perception for smart applications.
Cepton was established in 2016 by co-founders Dr. Jun Pei and Dr. Mark McCord. The company is headquartered in San Jose, California, and serves a fast-growing customer base through an international presence spanning North America, Germany, Japan, India and China.
Micro Motion Technology (MMT®)
Cepton was built from the ground up to meet key lidar industry challenges for mass market adoption. This company’s portfolio of proprietary technology is uniquely aimed at facilitating this industry growth through a combination of performance, reliability, affordability and design integration.
Key among its innovations is MMT®, a mirrorless, frictionless, rotation-free 3D imaging platform designed specifically for lidars. Its benefits for OEMs and system integrators include:
- Reliability – The durable design uses common, easily attainable materials.
- Versatility – The platform is capable of achieving near- to ultra-long range with a wide field of view.
- Efficiency – MMT® features a compact form factor, low power usage and inexpensive components.
- Scalability – Its simple design means that scale-up to high manufacturing volumes is easily attainable.
Because of their compact form factor, Cepton lidars are embeddable and ideally suited for advanced driver-assistance system (ADAS) integration, whether behind windshield, in headlamp or in fascia.
Agreement with KOITO
KOITO Manufacturing Co. Ltd., the world’s premier Tier 1 auto lighting supplier, originally started an evaluation of Cepton’s MMT® based lidars in 2018. In 2020, KOITO made an investment in Cepton aimed at accelerating the company’s development and enabling KOITO’s industrialization of high-performance and high reliability lidar sensors for ADAS and autonomous vehicle (AV) applications.
Through this collaboration, Cepton was able to secure the largest ADAS lidar series production award[1] with General Motors as a sole source in the automotive space. The award covers GM vehicles for the initial period of 2023-2027.
On August 5, 2021, the two companies deepened their relationship when KOITO committed to invest a further $50 million in Cepton’s business through its participation in a Private Investment in Public Equity (PIPE) offering of shares of common stock of Growth Capital Acquisition Corp. in connection with Cepton’s recent merger.
Collaboration with GM
On July 13, 2021, Cepton announced that it had secured an ADAS lidar series production award from a leading, Detroit-based global automotive OEM – the biggest lidar production award by any OEM to any lidar company. It was later clarified that the OEM was General Motors, and Cepton’s lidar is part of GM’s ADAS Ultra Cruise system.
GM is “expected to deploy Cepton lidars in its next generation of advanced driver assistance systems (ADAS) across multiple vehicle classes and models – not just luxury cars.” As such, the agreement marks the potential for “an industry-first, mass-market adoption of lidar technology for automotive ADAS, with an anticipated deployment in consumer vehicles starting in 2023.”
On July 28, 2021, Ford Motor Company (NYSE: F) distributed an article on Medium noting, “Ford has been engaged with Cepton almost since their inception in 2016, both for R&D collaboration and small-scale deployments. Cepton LiDAR are deployed in some of [Ford’s] smart city projects. Based on Ford’s guidance, Cepton delivered a custom version of their LiDAR to enable R&D on advanced ADAS features.”
Market Outlook
Driven by increasing development and adoption in automobile safety applications, environmental mapping and 3D-modeling, the global lidar market is forecast to experience considerable growth over the coming years. A research report published by MarketsAndMarkets suggests that the sector will grow to an estimated $3.4 billion by 2026, achieving a CAGR of 21.6% over the next five years.
The report further highlights increasing investments in lidar startups by automotive giants as a driver of growth opportunities in the sector, particularly in North America.
In 2020, ground-based lidar accounted for the lion’s share of the overall lidar market, and this trend is expected to continue as the automotive sector continues to rapidly advance adoption across the full spectrum of vehicle classes. One factor not to be underestimated is the high barrier of entry and the exceptionally long time required for automotive OEMs to vet and award a production win to a lidar company. It is a commonly held view that the over 50 lidar companies will inevitably coalesce into a handful serving all OEMs.
Cepton, having a head start through its established partnership with leading global OEM GM, is uniquely positioned to capitalize on this market growth in the years to come.
Management Team
Cepton’s founder-led team is made up of lidar industry pioneers with decades of collective experience across advanced lidar and imaging technologies.
Jun Pei, Ph.D., is the company’s CEO and Co-Founder. He is a technology specialist with a focus in optics and electronics. Prior to founding Cepton, Dr. Pei founded AEP Technology, a firm focused on developing advanced 3D optical instruments. He received his Ph.D. in electrical engineering from Stanford University.
Mark McCord, Ph.D., is Cepton’s CTO and Co-Founder. Prior to founding Cepton, he led advanced development at KLA-Tencor. Dr. McCord also formerly served as an associate professor at Stanford University, where he earned his Ph.D. in electrical engineering.
Winston Fu, Ph.D., is the company’s CFO. Dr. Fu is the founder of Silicon Valley venture capital firm LDV Partners. Prior to joining Cepton, he served as CFO and Chairman of Active-Semi before its acquisition. Dr. Fu has also helped to build many technology companies as an entrepreneur and/or board member. He received his Ph.D. in applied physics from Stanford University, as well as an MBA from the Kellogg School of Management at Northwestern University.
[1] Largest known ADAS lidar series production award based on number of vehicle models awarded
Cepton Inc. (NASDAQ: CPTN), closed Monday's trading session at $3.03, off by 0.6557377%, on 6,968 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.38/$8.90.
Recent News
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Launches Proprietary StudioViz(TM) End-to-End Lidar Simulator
- InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Schedules Q1 2024 Earnings Release, Conference Call
- InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Releases Q4, FY 2023 Financial Report, Business Update
Turbo Energy S.A. (NASDAQ: TURB)
The QualityStocks Daily Newsletter would like to spotlight Turbo Energy S.A. (NASDAQ: TURB).
World-leading solar energy company Lightsource BP is poised to begin working on a solar park in central Greece with the financial backing of Eurobank and the National Recovery and Resilience Plan Greece 2.0. Construction on the 560 MW photovoltaic park, Enipeas Solar Park, will end sometime in 2026, and the completed park will be the second largest in all of Europe, after Germany's 605 MW Witznitz solar farm. United Kingdom-based solar-energy developer Lightsource BP has finished more than 8.4 GW of solar energy projects across 19 markets globally, but Enipeas will be its largest-ever photovoltaic investment. Solar energy will be a critical component of the global transition to renewables alongside wind energy and other green-energy sources, including geothermal and hydroelectric. Companies such as Lightsource will be integral in the race to replace fossil fuels with zero-emissions renewables such as solar, cut greenhouse-gas emissions and mitigate climate change. Lightsource BP was formed as Lightsource Renewable Energy in 2010 before partnering with BP (formerly British Petroleum) and rebranding as Lightsource BP. Now one of the largest solar developers on the globe, Lightsource BP jumped into the Greek market and has deployed more than 1GW of solar and storage projects in Greece so far. Other players in the solar energy space such as Turbo Energy S.A. (NASDAQ: TURB) are also doing their part to accelerate the rate at which photovoltaic systems are deployed at scale. All these undertakings point to a clean-energy future coming sooner than critics may have thought possible.
Turbo Energy S.A. (NASDAQ: TURB), a leader in the field of photovoltaic energy storage, has filed a patent application with the Spanish Patent and Trademark Office ("OEPM"); the patent covers Turbo's Sunbox Commercial & Industrial ("Sunbox C&I") product, which is already available on the market. The Sunbox product is designed to provide the most efficient, adaptable solution for both isolated and grid-connected projects. The Sunbox C&I software allows Turbo Energy's proprietary technology to be combined with any previously operational facility, allowing for standardization of the system as well as being more cost effective. The Sunbox system is the most efficient electrical management system on the market today. "We are pleased to announce this patent application submission," said Turbo Energy CEO Mariano Soria in the press release. "We are optimistic that the OEPM will approve our presentation after having thoroughly studied the technological base of the market and having already successfully tested our solution with our clients. Following Spanish approval, we plan to continue our expansion strategy for Sunbox by seeking an international patent that will provide Turbo Energy with substantial global revenue-growth opportunities."
To view the full press release, visit https://ibn.fm/MZ5XY
Turbo Energy S.A. (NASDAQ: TURB) designs, develops and distributes equipment for the generation, management and storage of photovoltaic energy in Spain, Europe and internationally.
Turbo Energy’s products include lithium-ion batteries and inverters. Additionally, the company recently launched its flagship product, the Sunbox, an all-in-one device that integrates most of the equipment required for a residential photovoltaic installation. The Sunbox is powered by AI and features a software system that monitors the generation, use and management of photovoltaic energy by analyzing large amounts of data related to energy generation, consumption, market prices and weather forecasts. This AI system optimizes battery usage, reducing electricity bills and providing peak-use reduction and uninterruptible power supply functions.
Turbo Energy currently sells its photovoltaic energy equipment primarily through distributors for residential consumers in Spain, but it possesses the expertise and international perspective to expand its product portfolio into industrial and commercial scale and markets, as well as advancing the internationalization process it has already started. The company plans to expand into the industrial photovoltaic sector with its new Sunbox, launched in 2023, in higher power and capacity variants. Its goal is to become a significant player in this sector and contribute to the growth of renewable energy solutions.
The company was incorporated in 2013 and is based in Valencia, Spain. It operates as a subsidiary of Umbrella Solar Investment S.A.
Products
Lithium-Ion Batteries
Turbo Energy is one of the leading companies that introduced lithium-ion batteries for photovoltaic energy storage in Spain. Primarily for the home energy storage market, the company’s batteries have capacities from 2.24 kWh to 5.1 kWh in 24 and 48 volts. In addition, its 48V / 5.1 kWh units are available in a dual battery system.
Inverters
The inverter converts the direct current produced by the photovoltaic panels into alternating current that can be used by household appliances. It also regulates battery charging and discharging based on energy needs and optimizes utilization of generated renewable energy. Turbo Energy currently offers multiple models that cover most household installations.
All-in-One Sunbox
This product incorporates inverters, batteries and the rest of the components necessary to operate and protect the photovoltaic installation. This saves installation cost and assembly and configuration time while preventing errors. Notably, the latest Sunbox models also offer an EV charging option.
Software System
In communication with the inverter, the company’s software monitors energy flows between the photovoltaic panels, household consumption, storage and an optional electric vehicle charging station. The software allows users to customize an automatic backup mode based on weather forecasts, or manually select which part of the battery will be reserved for possible power outages. It also allows the battery to be used in a peak shaving mode, which leverages AI to trigger battery power when grid energy is most expensive, effectively reducing the amount of high-cost power drawn from the grid.
Market Opportunity
According to a report by Fortune Business Insights, a global research and reporting firm, the solar energy storage battery market was estimated to be worth $3.33 billion in 2022 and is projected to reach a value of more than $20 billion by 2030, marking a CAGR of 24.2% over the forecast period.
These batteries are crucial components of renewable energy systems, allowing for the storage of excess electricity generated by solar panels, so it can be used during times of no or low sunlight. By storing energy and supplying it when needed, these batteries reduce reliance on the power grid and maximize self-consumption while helping users avoid peak electricity rates. They also contribute to the transition toward a cleaner and more sustainable energy future by enabling residential consumers and businesses to use solar power even when the sun is not shining.
Management Team
Enrique Selva Bellvís is the CEO and founder of the Umbrella Group. In addition, he serves as vice-president of the Valencian Association of Energy Sector Companies industry group. Before his career in the solar energy sector, he was the founder and CEO of Innova Ingenieros Consultores. He holds a degree in industrial engineering with a specialization in energy from the Polytechnic University of Valencia and completed the Management Development Programme at the IESE Business School.
Mariano Soria is the Chief Innovation Officer for the Umbrella Group and serves as General Manager of Turbo Energy. He was CEO of Punt Moble XXI S.L. and continues to serve on that company’s board. Before that, he was the General Manager of REJMAR S.A., a land development company. He received his degree in industrial engineering and industrial organization from the Polytechnic University of Valencia, and his MBA from the European University of Madrid.
Alejandro Moragues is CFO of Turbo Energy. Previously, he held the position of Senior Corporate Auditor for U.S. company Euronet Worldwide Inc. and was an external auditor for PricewaterhouseCoopers. He holds a bachelor’s degree in business administration and management from the Polytechnic University of Valencia.
Manuel Cercos is Chief Commercial Officer at Turbo Energy. Previously, he held positions at Técnicas Aplicadas en Baterías S.L., where he served as Sales Director and Sales Manager. Before that, he worked as a Sales Technician at DAISA.
Turbo Energy S.A. (NASDAQ: TURB), closed Monday's trading session at $1.12, off by 6.6667%, on 8,618 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.855/$7.90.
Recent News
- Turbo Energy S.A. (NASDAQ: TURB) - InvestorNewsBreaks - Turbo Energy S.A. (NASDAQ: TURB) Announces Filing of Patent Application in Spain for Sunbox Commercial, Industrial Product
- Lightsource to Set Up Europe's Second-Largest Solar Facility
- Turbo Energy S.A. (NASDAQ: TURB) Signs Distribution Agreement with El Corte Ingles
Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF)
The QualityStocks Daily Newsletter would like to spotlight Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF).
At the start of last month, the U.S. Securities and Exchange Commission ("SEC") revealed that it would take more time in the implementation of new reporting rules for public companies as they dealt with the courts. This comes after the U.S. Court of Appeals for the Fifth Circuit halted the change, within days of adoption. The rules, which touch on environment, social and government ("ESG") issues, are being challenged in other federal courts and by different groups. For instance, workers supported by right-wing groups in New York have filed a lawsuit accusing the state's pension managers of breaking their fiduciary duty for moving from investing in fossil-fuel companies. In California, the U.S. Chamber of Commerce is suing to block the new carbon disclosure regulations from implementation. The disclosures focus on carbon emissions along a company's supply chain, and while companies are already reporting their emissions, some are worried that the data may not be accurate, which will make it harder to validate. The same cannot be said for the United Kingdom and European Union. These regions have committed to strong ESG disclosure standards. Certa CEO and founder, Jagmeet Lamba, revealed that Sustainable Finance Disclosure Regulations in the United Kingdom were driving business greatly. He added that companies that weren't prepping for climate-related disclosures were getting left behind. Lamba advised companies to start strengthening their reporting infrastructure while the industry was still young, with a focus on ensuring that ESG was handled by an individual passionate about impact and ESG and using flexible software that can grow and adapt with one's growth and success. The companies that are already implementing ESG, such as Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF), can provide pointers to those that are just starting out on this journey of integrating ESG principles within all operations.
Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) is a strategic minerals company focused on locating and developing economic properties in the strategic metals and advanced materials space. The company aims to improve domestic specialty mineral infrastructure efficiencies to meet surging national demand from North American manufacturers, effectively positioning itself as one of the only North American suppliers of high purity natural graphite for hi-tech applications.
Reflex Advanced Materials is based in Vancouver, British Columbia. Its project portfolio includes the Ruby Graphite Deposit in Montana and the ZigZag Lithium Property in Ontario.
Projects
Ruby Graphite Project
Located in a mining-friendly jurisdiction in southwest Montana, the Ruby Graphite Deposit is a low cost, rapid re-entry opportunity that produced roughly 2,400 tons of graphite from 1902 to 1948. Reflex Advanced Materials holds mining rights for 755 hectares at the Ruby Graphite Project, with 96 federal lode mining claims. Recent samples assay at 95.8% to 98.4% total carbon.
The site is notable as the only combined U.S. graphite flake and vein graphite source. Vein graphite is ideal for energy storage applications, because it requires fewer steps to achieve purity than synthetic alternatives and is therefore far less environmentally damaging. This is expected to play a key role in the project’s development as demand for electric vehicles continues to surge.
In March 2023, the company announced its submittal of permit applications to the Bureau of Land Management in respect of its exploration of the Ruby Graphite Project. Its initial drill program, expected to take place in the summer of 2023, includes plans for 3,500 total meters of drilling, cored to an average depth of 130 meters. The targets for this drill program have been identified using historical data from original mine operations and data gathered for the initial 43-101 technical report on the project, dated January 31, 2023.
ZigZag Lithium Property
Located in the Thunder Bay Mining Division of Ontario, the ZigZag Lithium Property consists of eight mining claims spanning roughly 2,710 hectares. Mineralization at the property, most notably lithium, is based in pegmatite dikes and concentrated in spodumene crystals, which are consistent throughout the entire unit.
Spodumene is readily observable in outcrops and in drill cores, with crystal sizes ranging from 3-15cm, on average.
Reflex Advanced Materials and American Energy Technologies Company Metallurgical Partnership
Reflex Advanced Materials has entered into a material processing agreement with American Energy Technologies Co., which is based in Arlington Heights, Illinois, to conduct metallurgical testwork with the goal of creating a technical support data package for Reflex’s target customer base, U.S. Federal agencies and qualification programs with hi-tech customers in the battery and battery storage business.
The resulting coated, spherionized, purified graphite (CSPG) material that is expected to be created from the aforementioned tests will be used to provide potential customers of CSPG with samples so that they can begin the material qualification process.
Market Opportunity
Graphite is an ideal battery anode and has dominated the market since the proliferation of lithium-ion batteries. Despite this demand, there is currently no significant production of lithium-ion battery anode material in North America.
Instead, most graphite sold in North America today is sourced from Chinese producers. U.S. President Joe Biden highlighted this sourcing disparity in a 2022 address:
“The United Stated depends on unreliable foreign sources for many of the strategic and critical materials necessary for the clean energy transition – such as lithium, nickel, cobalt, graphite and manganese for large-capacity batteries,” he said. “Demand for such materials is projected to increase exponentially as the world transitions to a clean energy economy.”
The U.S. Department of Energy is in the process of awarding $2.8 billion to expand domestic manufacturing of batteries for electric vehicles and combat this foreign dependency. Reflex Advanced Materials has identified its Ruby Graphite Project as a prime candidate for U.S.-sponsored initiatives due to the rarity and scarcity of natural graphite deposits in the country.
Processing graphite domestically in the U.S. is expected to provide Reflex Advanced Materials a competitive advantage as manufacturers begin to seek out American supply in the face of increased diplomatic tension. This is critical, as a rise in anode demand is expected to fuel a shortage of 8 million tonnes of graphite by 2040. World Bank Group projects 494% growth in total graphite demand by 2050.
Leadership Team
Paul Gorman is the CEO and a Director of Reflex Advanced Materials. He brings to the company over 25 years of experience in junior mining finance, public listings, viability assessment and operational rationalization. For 18 years, Mr. Gorman served as president and managing partner of Riverbank Capital, where he played an instrumental role in raising more than $85 million for small-cap companies. In 2008, he funded Industrial Minerals Inc. (later Northern Graphite) and served in an advisory role for four other graphite companies, contributing significantly to the revitalization of the junior graphite space in North America. Mr. Gorman founded Mega Graphite Inc. in 2009 and has served as chief executive for three other companies.
Tasheel Jeerh, CPA, is the company’s CFO. He is a finance and accounting professional with over a decade of experience spanning both public and private sectors. Prior to joining Reflex Advanced Materials, Mr. Jeerh played a pivotal role in the growth of a private upstream oil and gas firm, dealing with over $2 billion in M&A activity and $1 billion in financing activities. He gained his designation at PricewaterhouseCoopers, where he worked as a manager in the assurance practice.
Greg Bell is Project Manager for Reflex Advanced Materials. He is a multi-disciplined engineering management professional with more than 40 years of experience in the natural resources sector. Mr. Bell has successfully built and managed several start-up operations in various capacities. He has been active in graphite and lithium exploration for the past seven years.
Christopher W. Hill leads the company’s Corporate Development initiatives. He is an investor and entrepreneur with over a decade of experience in the capital markets. Mr. Hill began his career as an investment advisor and then began to consult and advise private companies on their paths to becoming publicly traded. He specializes in corporate development and strategic financing utilizing his large network in the capital markets.
Reflex Advanced Materials Corp. (RFLXF), closed Monday's trading session at $0.072, off by 15.4433%, on 24,060 volume. The average volume for the last 3 months is 17,658 and the stock's 52-week low/high is $0.063/$0.45.
Recent News
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - US ESG Battle Now Shifts to Courts
- Investment Advisor Says Investors Expect More Personalization on ESG
- Headhunter Says Top ESG Professionals Becoming Disillusioned by Lack of Commitment
Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI)
The QualityStocks Daily Newsletter would like to spotlight Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI).
Scinai Immunotherapeutics (NASDAQ: SCNI) is a biopharmaceutical company focused on developing inflammation and immunology ("I&I") biological products and providing CDMO services through its Scinai Bioservices business unit. The company today announced that it has received a staff determination letter on April 30, 2024, from the Listing Qualifications Department of the Nasdaq Stock Market LLC. In the letter, Nasdaq notified the company that, due to its non-compliance with the minimum $1.00 bid price requirement set forth in Nasdaq Listing Rule 5550(a)(2), the company's American Depositary Shares ("ADSs") would be scheduled for delisting from the Nasdaq Capital Market and would be suspended for trading at the opening of business on May 7, 2024, unless the company timely requests a hearing before an independent Nasdaq Hearings Panel. Scinai appealed the delisting determination and requested a hearing before the panel. In addition, the company also announced that its board of directors has approved a ratio change of the ADSs to its non-traded ordinary shares, increasing the number of ordinary shares represented by each ADS from 400 to 4,000, which is equivalent to a reverse split of 1 for 10. No action is required by the holders of the ADSs to affect the ratio change, and no fractional new ADSs will be issued in connection with the change in ADS ratio.
To view the full press release, visit https://ibn.fm/plmL5
Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) is a biotechnology company focused on developing, manufacturing, and commercializing innovative immunotherapeutic products primarily for the treatment of infectious diseases and autoimmune diseases.
In collaboration with the prestigious Max Planck Institute for Multidisciplinary Sciences (MPG) and the University Medical Center Göttingen (UMG), both in Germany, Scinai is developing a pipeline of innovative nanosized antibody (NanoAb) therapies addressing diseases underserved by current treatments and with large and growing markets, such as COVID-19, asthma and psoriasis.
NanoAbs, also known as VHH-antibodies or Nanobodies, are alpaca-derived nanosized antibodies that exhibit multiple significant competitive advantages over existing antibody therapies, including stability at high temperatures, superior binding affinity, more effective and convenient routes of administration and efficient production. Scinai is uniquely positioned to advance nanosized antibody innovation from R&D through commercialization.
The company’s highly experienced and successful pharmaceutical industry leadership team includes former senior executives from Novartis, GSK and Bristol-Myers Squibb.
Since its founding, Scinai has executed eight clinical trials, including a seven-country, 12,400-participant Phase 3 trial of a prior influenza vaccine candidate, and it built, owns and operates a 20,000 sq. ft. state-of-the-art GMP biologics manufacturing facility housing its laboratories, production facilities and offices.
Lead Candidate: Inhaled COVID-19 NanoAb
In December 2021, Scinai signed definitive agreements with the Max Planck Society – parent organization of the Max Planck Institute for Multidisciplinary Sciences– and the UMG to enter a strategic collaboration for the development and commercialization of innovative COVID-19 NanoAbs.
The company is planning a rapid development path that leverages its expertise and capabilities in biological drug development and manufacturing. Scinai anticipates preclinical proof-of-concept results for an inhaled COVID-19 NanoAb by the end of 2022, with initial Phase 1/2a human clinical trial results expected in 2023.
The intended inhaled mechanism of delivery of Scinai’s COVID-19 NanoAb formulation may serve as a significant differentiator when compared to approved monoclonal antibodies, which are injected. Inhaled delivery has shown to be cheaper, more convenient and likely safer for patients and providers.
NanoAb Pipeline: Psoriasis, Asthma and More
The COVID-19 NanoAb development agreement is part of a broader five-year research collaboration agreement signed in March 2022 covering discovery, development and commercialization of NanoAbs for several other disease indications with large market medical needs, including asthma, psoriasis, macular degeneration and psoriatic arthritis.
Scinai has an exclusive worldwide license for development and commercialization of COVID-19 NanoAbs and exclusive options for similar worldwide licenses for NanoAbs for the above mentioned additional large market disorders currently underserved by approved therapeutic antibodies.
Academic research teams from MPG and UMG have verified strong affinity by the new NanoAbs to their biological target molecules and high thermostability. They have also demonstrated strong neutralization by several NanoAb candidates of their respective target molecules. Neutralization studies of the other NanoAbs are expected to begin later in 2022.
Based on the promising results, Scinai will focus development efforts beginning with the following NanoAbs:
- NanoAbs targeting IL-17 as drug candidates for the potential treatment of psoriasis and psoriatic arthritis
- NanoAbs targeting IL-13 and NanoAbs targeting TSLP as drug candidates for the potential treatment of asthma
These are conditions for which the antibody target is validated by existing treatments and the mechanism of action is well understood. Both represent large medical needs and growing markets. Scinai anticipates preclinical proof-of-concept for at least one of these NanoAbs in 2023. This is in addition to the aforementioned human clinical Phase 1/2a for the inhaled COVID-19 NanoAb therapy, which is also anticipated in 2023.
CDMO Services
While NanoAb pipeline development is Scinai’s core focus, the company also offers its cGMP manufacturing facility, aseptic fill and finish suite, laboratories and experienced professionals for contract development and manufacturing organization (CDMO) services. This offering is designed to keep the Scinai team abreast of the latest industry developments and trends while building experience and generating revenue to support the company’s NanoAb pipeline development.
Market Opportunity
COVID-19 treatment, target of the company’s lead NanoAb therapy candidate, had an estimated market size of $22 billion in 2021.
Future Scinai drug candidates will target conditions with large markets growing at attractive CAGRs.
The global asthma treatment market was valued at $18.08 billion in 2019 and is projected to reach $26.01 billion by 2027, exhibiting a CAGR of 4.5% during the forecast period, according to Fortune Business Insights. The research firm predicts that the global psoriasis treatment market will grow from $26.37 billion in 2022 to $47.24 billion by 2029, exhibiting a CAGR of 8.7% over the forecast period.
Management Team
Amir Reichman is Scinai’s CEO. He previously was Head of Global Vaccines Engineering Core Technologies at GSK Vaccines in Belgium. Prior to that, he held leadership roles at Novartis Vaccines’ Global Vaccines Supply Chain Management organization. He was the first employee of NeuroDerm Ltd., a company focused on transdermal drug delivery, and served as Senior Scientist until his departure in 2009. He earned a M.Sc. in Biotechnology Engineering from Ben-Gurion University and an MBA in Finance and Health Care Management from the University of Pennsylvania’s Wharton School.
Tamar Ben-Yedidia, Ph.D., is Chief Science Officer at Scinai. She has more than 30 years of experience in immunology, with specific expertise in the development of vaccines. She began her career with Biotechnology General Ltd., working on development of a recombinant Hepatitis-B vaccine. She later joined the Weizmann Institute of Science, working on the design of a peptide-based vaccine against several pathogens. She is widely published, with numerous refereed articles and invited reviews in various scientific journals. She received her Ph.D. from the Weizmann Institute.
Elad Mark is COO at Scinai. He has over 15 years of biotechnology industry experience encompassing diverse project stages including feasibility studies, conceptual and detailed design, commissioning, qualification and process validation. Prior to joining Scinai, he led Novartis’s $800 million investment in a biologics facility in Singapore. With Biopharmax and Antero, both global pharmaceutical engineering companies, he successfully led projects in Israel, China and Singapore. He holds a BSc. in Engineering from the Afeka Tel Aviv Academic College of Engineering and an MBA from the Open University of Israel.
Uri Ben-Or is CFO at Scinai. He has served as CFO with public life science companies traded on the TASE, OTC and Nasdaq. Ben-Or provides his services to Scinai through CFO Direct, a company he founded and for which he serves as CEO. He served as the VP of Finance of Glycominds, a leading biotechnology company, and as CFO of a spin-off from Telrad Networks. He also served as a Corporate Controller at Menorah Capital Markets and as an Auditor at PWC. He holds a B.A. in Business from the College of Administration, an MBA from Bar-Ilan University, and is a CPA.
Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI), closed Monday's trading session at $0.449, off by 3.6687%, on 24,514 volume. The average volume for the last 3 months is 27,128 and the stock's 52-week low/high is $0.4311/$2.27.
Recent News
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Announces Nasdaq Delisting Notification and Appeal, ADS Ratio Change
- Vitamin D Could Balance Gut Microbiota, Boost Cancer Immunotherapy
- InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Chief Science Officer to Present at This Month's Europe Dermatology Drug Development Summit
Btab Ecommerce Group Inc. (OTC: BBTT)
The QualityStocks Daily Newsletter would like to spotlightFathom Btab Ecommerce Group Inc. (OTC: BBTT) .
Btab Ecommerce and Integrated Wellness Acquisition Corp recently signed an LOI, to initiate a business combination, aiming for a process completion during the second half of 2024
Completion of the transaction is contingent upon various factors including due diligence, regulatory approvals, and approval by both companies' boards and stockholders
Btab provides online technology affordable to the small and medium size online businesses that resell Btab's proprietary products and third party products, including product supply, marketing platforms, physical showrooms, order and product return fulfillment, and after-sales support
The global e-commerce market, estimated at $8.8 trillion in 2024, is projected to reach $18.81 trillion by 2029
Btab Ecommerce Group (OTC: BBTT), an ecommerce company, and Integrated Wellness Acquisition Corp (NYSE: WEL), announced in February 2024 the signing of a Letter of Intent ("LOI") for a proposed business combination. This agreement will result in Btab acquiring control of WEL, with Btab valued at an initial enterprise value of $250 million (https://ibn.fm/1Lqwi).
Btab Ecommerce Group Inc. (OTC: BBTT) is a next-generation e-commerce company with significant social impact. The company believes that every business deserves an equal opportunity to succeed in the modern retail market, so it provides e-commerce and social commerce solutions to help small businesses excel in both online and offline environments.
The company’s long-term plan is to become the world’s largest product supplier for small businesses using e-commerce technology as a distribution tool. Btab operates through its network in Australia, Asia, the United States and the United Kingdom.
Btab offers comprehensive solutions including product supply, commerce platforms for selling and marketing, physical showrooms that allow customers to touch and feel products, goods storage, marketing management, delivery and pick-up direction and after-sales support including arranging exchanges and returns. The company takes all of these concerns off of its clients’ plates, allowing them to focus on running successful retail businesses.
Btab supplies products to resellers, either from its own manufacturing facility or from third-party manufacturers and wholesalers. The company also connects resellers with manufacturers and wholesalers around the world, allowing them to access better deals and a greater product range by leveraging Btab’s buying power.
In February 2024, Btab and Integrated Wellness Acquisition Corp (NYSE: WEL), a special purpose acquisition company, announced their entry into a letter of intent providing for a proposed business combination that will result in Btab acquiring control of WEL. The transaction would value Btab at $250 million.
Btab is headquartered in Sydney and Perth, Australia, and the company is expanding its headquarters into the U.S.
Platforms
Btab provides affordable ecommerce services and supplies technology and products to small businesses to allow them to compete in an underserved market segment. The company seeks to expand its reach into Europe and the Americas, where it intends to provide small businesses with products and services not currently commercially available to them.
Btab believes growth of the e-commerce segment in Asia alone will be significant well into the next decade and beyond as rising numbers of internet users take advantage of online shopping and increasing spending power. The company’s vision is to provide all small and medium businesses with an equal opportunity to improve using the same online technology that’s utilized by large multinationals.
Btab’s mission is to make online technology affordable to all small- and medium-sized businesses and use the Btab Network to assist as many businesses as possible to succeed. Some of its platform offerings include:
- Btab Commerce provides ecommerce management services to manufacturers, wholesalers and retailers in the Btab Network.
- Social3 is a next generation platform for all things social and commerce.
- Marketplace Australia is a social commerce site for all products and services in Australia. It is a combination of a social platform, a products marketplace platform and an online stores platform.
- Aussie Markets is an online marketplace focusing on Australian-made products.
- Marketplace Deals is a social commerce site for products and services around the world. It is a combination of a social platform, a products marketplace platform and an online stores platform.
- Chemist Deals is a social commerce site for health and beauty products. It is a combination of a social platform, a products marketplace and an online stores platform.
- Global Manufacturers Network is a social commerce platform for manufacturers around the world.
- InterestPin is a social commerce platform for all products and services around the world. It is also a tool to help users collect, organize and share all the beautiful things they find on the web.
- Btab Domains offers domain name registration, hosting, email, SSL certificates, a website builder and related services.
Market Opportunity
A report from Mordor Intelligence, a global research and intelligence firm, estimates the worldwide e-commerce market at $8.8 trillion in 2024 and projects growth to $18.81 trillion by 2029, expanding at a CAGR of 15.8% during the forecast period.
Increasing global internet penetration and the continued growth of smartphone usage around the world are projected to positively impact market growth, according to the report. Other growth drivers include a trend toward established businesses and corporations moving retail operations online or upgrading online operations, the ease for retailers of using online marketing tools such as Google advertisements and Facebook ads and the ease of access for small- and medium-sized businesses to start up or expand online businesses, the report states.
Key Management Team
Binson Lau is, CEO and Director at Btab Ecommerce Group.
Ronald A. Woessner is the company’s Seniorr Vice President and General Counsel.
Btab Ecommerce Group Inc. (OTC: BBTT), closed Monday's trading session at $0.179, off by 0.5555556%, on 23,400 volume. The average volume for the last 3 months is 16,106 and the stock's 52-week low/high is $0.031/$0.745.
Recent News
- Btab Ecommerce Group Inc. (OTC: BBTT) - Btab Ecommerce Group Inc. (BBTT) Signs Letter of Intent with Integrated Wellness Acquisition Corp. (NYSE: WEL), Accelerating Company Growth
- Btab Ecommerce Group Inc. (BBTT) Is 'One to Watch'
- Btab Ecommerce Group Signs LOI to Merge with Integrated Wellness Acquisition Corp
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Regains Compliance with Nasdaq Minimum Bid Price Requirement
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Announces Securities Purchase Agreements, Anticipates Receiving Approximately $4.2M from Placement
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Advanced Gold Exploration Inc. (CSE: AUEX) (FSE: 4TG) (OTC: AUHIF) - MiningNewsBreaks - Why Advanced Gold Exploration Inc. (CSE: AUEX) (OTC: AUHIF) (FSE: 4TG) Is 'One to Watch'
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Partners with Volunteer State Community College to Launch AI, Technical Training Programs
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) (MUN: A0I0) (BER: A0I0) Announces Promising Results at PHC Project in Brazil
- Fresh2 Group Ltd. (NASDAQ: FRES) - Appointment of New Independent Director
- Astiva Health - Astiva Health Inc. Innovative Holistic Approach Underscores Understanding That Effective Primary Care Is Foundational
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Goldman Sachs Predicts Surge in Commodity Prices as Central Banks Slash Rates
- Astrotech Corp. (NASDAQ: ASTC) - USDA Says Hemp Harvests, Prices are Rising
- Aston Bay Holdings Ltd. (TSX.V: BAY) (OTCQB: ATBHF) - InvestorNewsBreaks - Aston Bay Holdings Ltd. (TSX.V: BAY) (OTCQB: ATBHF), American West Metals Commence Drilling, Geophysics at Storm Copper Project
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Google Cloud to Bring Medically-Tuned AI Technology to Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Releases Year-End Review, Outlook for 2024
- Bebuzee Inc. (OTC: BBUZ) - InvestorNewsBreaks - Bebuzee Inc. (BBUZ) Announces Launch of Transformative Super App
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Announces Nasdaq Delisting Notification and Appeal, ADS Ratio Change
- Bravo Multinational Inc. (OTC: BRVO) - TechMediaBreaks - Bravo Multinational Inc. (BRVO) Positioning for Opportunity in Video Streaming Sector
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - InvestorNewsBreaks - BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: BXT) Announces Highly Promising Results from ODF Cladribine PK Study
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Notes 'Surge' in Revenue in FY 2023 Financial Report
- Btab Ecommerce Group Inc. (OTC: BBTT) - Btab Ecommerce Group Inc. (BBTT) Signs Letter of Intent with Integrated Wellness Acquisition Corp. (NYSE: WEL), Accelerating Company Growth
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Releases Update Report on Reid Drill Results
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Launches Proprietary StudioViz(TM) End-to-End Lidar Simulator
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Partners with Fortegra to Offer Protection Plans for Cybersecurity Clients
- Clene Inc. (NASDAQ: CLNN) - Study Suggests Metabolism Issues After Spinal-Cord Injury Result from Disturbed Neuronal Activity
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Researchers Seek to Develop Genetically Engineered Brain Cancer Treatment
- Correlate Energy Corp. (OTCQB: CIPI) - UN Secretary-General Says Climate Action Shouldn't Adversely Affect Poor
- Coyuchi Inc. - Louisiana Lawmakers Want to Restrict State Universities from Using ESG
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (Cboe CA: CYBN) Issues Corporate Update, Eyes Runway for Upcoming Clinical Milestones
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Two Contracts to Provide Integrated Cardiometabolic Solutions
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks - Diamond Lake Minerals Inc. (DLMI), BOXABL Announce Partnership to Bring Affordable Housing to the Public, Digital Tokens to Investors
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces 112% Revenue Increase, Significant Milestones for Fiscal Year 2023
- D-Wave Quantum Inc. (NYSE: QBTS) - Quantum Computing Could Make Genomic Analysis Hyper-Fast
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - Electronic Servitor Publication Network Inc. (XESP) Combines Powerful Digital Engagement Engine(TM) with Simple Pricing Model to Drive Growth of Client Companies
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Unconventional Market Factors Cause Gold Market to Rise
- Energy and Water Development Corp. (OTCQB: EAWD) - InvestorNewsBreaks — Energy and Water Development Corp. (EAWD) Selects IBN as Corporate Communications Partner
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Reports Financial Results, Operational Highlights for Q1 2024
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Releases Q4, Year-End 2023 Results; Schedules Earnings Call
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Receives PACEpilot Innovation to Watch Award
- FingerMotion Inc. (NASDAQ: FNGR) - US FCC Orders Halt to Broadband Services by Chinese Companies
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - Climate Change Puts Mining Companies in Catch-22
- Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) - Securing a Domestic Supply Chain: Why Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) Matters for North America
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Exploration Update on Quebec Lithium Projects, Private Placement
- Flora Growth Corp. (NASDAQ: FLGC) - InvestorNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Responds to Reported Plans for DEA to Reschedule Cannabis
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Paving Way for Safer, More Efficient Autonomous Vehicles in Japan
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Reports 30% YOY Revenue Growth for Q1 2024; Projects $25-$30m Annual Revenue for 2024
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Names New CEO, Outlines Key Components of Strategic Expansion Plans
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - York Trial Program Graduates Second Cohort of Green-Job Students
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- GivBux Inc. (OTC: GBUX) - GivBux Inc. (GBUX) Is 'One to Watch'
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc.'s (NASDAQ: GMGI) Meridianbet Setting New Standard for Customer Engagement, Community Impact
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) CEO Featured Guest in Most Recent Bell2Bell Podcast
- GolfLync Inc. - InvestorNewsBreaks - GolfLync Inc. Reaches 100K Membership Milestone, Sees 3,033% Growth Rate in 2023
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation at Upcoming Investor, Industry Events
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Releases Record 2023 Financial Report Showing 100% Increase in Revenue
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Presents Key Data in First-Ever Scientific Presentation of Proprietary HeartBeam AI
- HealthLynked Corp. (OTCQB: HLYK) - HealthLynked Corp. (HLYK) Q4, FY2023 Financial Results; Show Bottom-line Improvements, Core Network Application Ramp-Up Expected in 2024
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Closes Final Tranche of Nonbrokered Private Placement
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Delves into Key Dual Listing Considerations for Issues
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Termination of Merger Deal
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc.'s (NASDAQ: IDEX) Solectrac Announces Strategic Reorganization
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Releases Interim Results from Phase 2 Study Evaluating Treatment to Reduce Alzheimer's Agitation
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces Delayed Effective Date of Reverse Split
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Welcomes Leading Authority in Alzheimer's Research to Scientific Advisory Board
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Releases Shareholder Update, Outlines Key Milestones for Company Progress
- BlockQuarry Corp. (OTC: BLQC) - InvestorNewsBreaks - BlockQuarry Corp (BLQC) Makes Executive Team Announcement, Notes Operational and Financial Outlook Update
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Buy Rating Increased in Ascendiant Capital Markets Latest Report
- Laredo Oil Inc. (OTC: LRDC) - InvestorNewsBreaks - Laredo Oil Inc.'s (LRDC) Subsidiary Secures Permit for Midfork Well
- Longeveron Inc. (NASDAQ: LGVN) - InvestorNewsBreaks - Longeveron Inc. (NASDAQ: LGVN) Schedules Release of Q1 2024 Financial Results, Earnings Conference Call/Webcast
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - InvestorNewsBreaks - Lexaria Bioscience Corp. (NASDAQ: LEXX) Launches New Research Program to Evaluate DehydraTECH-GLP-1 Drugs
- Life Electric Vehicles Holdings Inc. (OTC: LFEV) - InvestorNewsBreaks - Life Electric Vehicles Holdings Inc. (LFEV) Engages IBN for Corporate Communications Expertise
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com Inc. (NASDAQ: LTRY) Announces Soft Launch of Sweepstakes Operations to Benefit Street Child United
- LQwD Technologies Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - InvestorNewsBreaks - LQWD Technologies Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Releases Updated Info Regarding Bitcoin Lightning Network Operations
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Shines in the Basic Materials Sector
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (FSE: 0NFA) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Board Approves Issuance of Shares for Debt Settlement
- Mountain Top Properties Inc. (OTC: MTPP) - InvestorNewsBreaks - Mountain Top Properties Inc. (MTPP) to Tap into Resilient Hamptons' Real Estate Market
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Positive Nasdaq Listing Decision
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Web3 Summit in Dubai Ends Successfully
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Nutriband Inc. (NASDAQ: NTRB) - Fentanyl Scourge: Nutriband Inc.'s (NASDAQ: NTRB) New Transdermal Patch with Abuse-Deterrent Tech Funded to Seek FDA Approval
- OK Stone Engineering Inc. - InvestorNewsBreaks — OK Stone Engineering Partners with Oren Klaff at Special Investor Event
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc.'s (NASDAQ: MULN) Inks Agreement with NAFG, Board Adopts Limited Duration Stockholder Rights Plan
- PaxMedica Inc. (NASDAQ: PXMD) - PaxMedica Inc. (NASDAQ: PXMD) Announces PAX-101 Batch Completion for NDA Application to FDA
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Releases Third-Quarter 2024 Financial Report
- Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Financial, Operational Report for FY 2023
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Biweekly Status Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Partners with FluGen to Develop First-of-Its-Kind Intranasal Flu Vaccine for Global Distribution
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces New UB Product Line, Plans Investor Teleconference
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Announces Participation at Upcoming EF Hutton Global Conference
- Progressive Care Inc. (OTCQB: RXMD) - BioMedNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Developing Robust Product Lineup to Address ASD, Associated Neurodevelopmental Conditions
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Signs Definitive Agreement, Plan of Merger with Telix Pharmaceuticals
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - Deloitte Survey Finds 8 in 10 Investors Have ESG Guidelines
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTC: RWBYF) Files Documents Reporting Financial Results for Q4, FY 2023
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - US ESG Battle Now Shifts to Courts
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF), G Mining Ventures Announce Combination to Set the Stage for a Leading Intermediate Gold Producer in the Americas
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc.'s (RJDG) ioSoft Systems Division Engaged to 'Create Significant Enhancements' for Rail Pro Services
- N2OFF Inc. (NASDAQ: NITO) - InvestorNewsBreaks - N2OFF Inc. (NASDAQ: NITO) (FSE: 80W) to Target Hemp Flower Market in California
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Software Effective Solutions Corp. (OTC: SFWJ) - InvestorNewsBreaks - Software Effective Solutions Corp. (d/b/a MedCana) (SFWJ) Responds to Announcement Rescheduling of Marijuana by DEA
- Sigyn Therapeutics Inc. (OTCQB: SIGY) - BioMedNewsBreaks - Sigyn Therapeutics Inc. (SIGY) Leveraging Portfolio to Overcome Current Limitations in Healthcare
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Shares Positive Results for PTSD Treatment
- SUIC Worldwide Holdings Ltd. (OTC: SUIC) - SUIC Worldwide Holdings Ltd.'s (SUIC) I.Hart Group Expands Franchise Development Efforts with New Authorizations in China and Japan
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Reports Record Revenues, EBITDA and Gross Margin Growth, in Annual Fiscal Conference Call
- Siyata Mobile Inc. (NASDAQ: SYTA) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Releases Q4, Full-Year 2023 Financial Results
- SOBRsafe Inc. (NASDAQ: SOBR) - InvestorNewsBreaks - SOBR Safe Inc. (NASDAQ: SOBR) Sees Continued Growth, Signs Two New Contracts
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) CEO Talks Distribution in Recent Proactive Investors Interview
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Installs, Validates First Commercial-Scale DLE Column in North America
- Starco Brands Inc. (OTCQB: STCB) - Whipshots® by Starco Brands Shatters Expectations with Explosive Growth and Nationwide Expansion
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - InvestorNewsBreaks - Vision Marine Technologies Inc. (NASDAQ: VMAR) Announces Filing of Patent for Cooling System Control in Electric Marine Vessel
- SenesTech Inc. (NASDAQ: SNES) - InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Schedules Release of Q1 2024 Financial Results, Earnings Call
- Sekur Private Data Ltd. (FRA: GDT0) (CSE: SKUR) (OTCQB: SWISF) - InvestorNewsBreaks - Sekur Private Data Ltd. (CSE: SKUR) (OTCQX: SWISF) (FSE: GDT0) Featured in New to the Street Episode Airing Tonight
- TAAT Global Alternatives Inc. (CSE: TAAT) (FRANKFURT: 2TP) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- Tartisan Nickel Corp. (CSE: TN) (OTCQB: TTSRF) - Tartisan Nickel Corp. (CSE: TN) (OTCQB: TTSRF) (FSE: 8TA) Moves Toward Feasibility Study, Exploration at Kenbridge Nickel Project on Strength of PEA Report
- SOBRsafe Inc. (NASDAQ: SOBR) - InvestorNewsBreaks - SOBR Safe Inc. (NASDAQ: SOBR) Sees Continued Growth, Signs Two New Contracts
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Board Declares Special Cash Dividend
- Torr Metals Inc. (TSX.V: TMET) - Unlocking the Riches: Torr Metals Inc. (TSX.V: TMET) Aims to Test Untapped Copper and Gold Potential in British Columbia and Ontario, Canada
- Turbo Energy S.A. (NASDAQ: TURB) - InvestorNewsBreaks - Turbo Energy S.A. (NASDAQ: TURB) Announces Filing of Patent Application in Spain for Sunbox Commercial, Industrial Product
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Responds to Senate Passing of Bill Banning Russian Uranium Imports
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - InvestorNewsBreaks - Vision Marine Technologies Inc. (NASDAQ: VMAR) Announces Filing of Patent for Cooling System Control in Electric Marine Vessel
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Announces Participation at Upcoming Anxiety and Depression Association Conference
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) CEO Discusses Medicare Reimbursement Approval for CARE Oral Medical Devices in Proactive Interview
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Bolsters BolaWrap® 150 Deployments, Promoting Safer Communities Nationwide
- Zoned Properties Inc. (ZDPY) - Zoned Properties Inc. (ZDPY) Advancing Initiatives Through the Cannabis Sector
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Regains Compliance with Nasdaq Minimum Bid Price Requirement
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Announces Securities Purchase Agreements, Anticipates Receiving Approximately $4.2M from Placement
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Advanced Gold Exploration Inc. (CSE: AUEX) (FSE: 4TG) (OTC: AUHIF) - MiningNewsBreaks - Why Advanced Gold Exploration Inc. (CSE: AUEX) (OTC: AUHIF) (FSE: 4TG) Is 'One to Watch'
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Partners with Volunteer State Community College to Launch AI, Technical Training Programs
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) (MUN: A0I0) (BER: A0I0) Announces Promising Results at PHC Project in Brazil
- Fresh2 Group Ltd. (NASDAQ: FRES) - Appointment of New Independent Director
- Astiva Health - Astiva Health Inc. Innovative Holistic Approach Underscores Understanding That Effective Primary Care Is Foundational
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Goldman Sachs Predicts Surge in Commodity Prices as Central Banks Slash Rates
- Astrotech Corp. (NASDAQ: ASTC) - USDA Says Hemp Harvests, Prices are Rising
- Aston Bay Holdings Ltd. (TSX.V: BAY) (OTCQB: ATBHF) - InvestorNewsBreaks - Aston Bay Holdings Ltd. (TSX.V: BAY) (OTCQB: ATBHF), American West Metals Commence Drilling, Geophysics at Storm Copper Project
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Google Cloud to Bring Medically-Tuned AI Technology to Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Releases Year-End Review, Outlook for 2024
- Bebuzee Inc. (OTC: BBUZ) - InvestorNewsBreaks - Bebuzee Inc. (BBUZ) Announces Launch of Transformative Super App
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Announces Nasdaq Delisting Notification and Appeal, ADS Ratio Change
- Bravo Multinational Inc. (OTC: BRVO) - TechMediaBreaks - Bravo Multinational Inc. (BRVO) Positioning for Opportunity in Video Streaming Sector
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - InvestorNewsBreaks - BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: BXT) Announces Highly Promising Results from ODF Cladribine PK Study
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Notes 'Surge' in Revenue in FY 2023 Financial Report
- Btab Ecommerce Group Inc. (OTC: BBTT) - Btab Ecommerce Group Inc. (BBTT) Signs Letter of Intent with Integrated Wellness Acquisition Corp. (NYSE: WEL), Accelerating Company Growth
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Releases Update Report on Reid Drill Results
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Launches Proprietary StudioViz(TM) End-to-End Lidar Simulator
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Partners with Fortegra to Offer Protection Plans for Cybersecurity Clients
- Clene Inc. (NASDAQ: CLNN) - Study Suggests Metabolism Issues After Spinal-Cord Injury Result from Disturbed Neuronal Activity
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Researchers Seek to Develop Genetically Engineered Brain Cancer Treatment
- Correlate Energy Corp. (OTCQB: CIPI) - UN Secretary-General Says Climate Action Shouldn't Adversely Affect Poor
- Coyuchi Inc. - Louisiana Lawmakers Want to Restrict State Universities from Using ESG
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (Cboe CA: CYBN) Issues Corporate Update, Eyes Runway for Upcoming Clinical Milestones
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Two Contracts to Provide Integrated Cardiometabolic Solutions
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks - Diamond Lake Minerals Inc. (DLMI), BOXABL Announce Partnership to Bring Affordable Housing to the Public, Digital Tokens to Investors
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces 112% Revenue Increase, Significant Milestones for Fiscal Year 2023
- D-Wave Quantum Inc. (NYSE: QBTS) - Quantum Computing Could Make Genomic Analysis Hyper-Fast
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - Electronic Servitor Publication Network Inc. (XESP) Combines Powerful Digital Engagement Engine(TM) with Simple Pricing Model to Drive Growth of Client Companies
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Unconventional Market Factors Cause Gold Market to Rise
- Energy and Water Development Corp. (OTCQB: EAWD) - InvestorNewsBreaks — Energy and Water Development Corp. (EAWD) Selects IBN as Corporate Communications Partner
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Reports Financial Results, Operational Highlights for Q1 2024
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Releases Q4, Year-End 2023 Results; Schedules Earnings Call
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Receives PACEpilot Innovation to Watch Award
- FingerMotion Inc. (NASDAQ: FNGR) - US FCC Orders Halt to Broadband Services by Chinese Companies
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - Climate Change Puts Mining Companies in Catch-22
- Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) - Securing a Domestic Supply Chain: Why Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) Matters for North America
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Exploration Update on Quebec Lithium Projects, Private Placement
- Flora Growth Corp. (NASDAQ: FLGC) - InvestorNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Responds to Reported Plans for DEA to Reschedule Cannabis
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Paving Way for Safer, More Efficient Autonomous Vehicles in Japan
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Reports 30% YOY Revenue Growth for Q1 2024; Projects $25-$30m Annual Revenue for 2024
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Names New CEO, Outlines Key Components of Strategic Expansion Plans
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - York Trial Program Graduates Second Cohort of Green-Job Students
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- GivBux Inc. (OTC: GBUX) - GivBux Inc. (GBUX) Is 'One to Watch'
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc.'s (NASDAQ: GMGI) Meridianbet Setting New Standard for Customer Engagement, Community Impact
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) CEO Featured Guest in Most Recent Bell2Bell Podcast
- GolfLync Inc. - InvestorNewsBreaks - GolfLync Inc. Reaches 100K Membership Milestone, Sees 3,033% Growth Rate in 2023
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation at Upcoming Investor, Industry Events
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Releases Record 2023 Financial Report Showing 100% Increase in Revenue
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Presents Key Data in First-Ever Scientific Presentation of Proprietary HeartBeam AI
- HealthLynked Corp. (OTCQB: HLYK) - HealthLynked Corp. (HLYK) Q4, FY2023 Financial Results; Show Bottom-line Improvements, Core Network Application Ramp-Up Expected in 2024
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Closes Final Tranche of Nonbrokered Private Placement
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Delves into Key Dual Listing Considerations for Issues
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Termination of Merger Deal
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc.'s (NASDAQ: IDEX) Solectrac Announces Strategic Reorganization
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Releases Interim Results from Phase 2 Study Evaluating Treatment to Reduce Alzheimer's Agitation
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces Delayed Effective Date of Reverse Split
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Welcomes Leading Authority in Alzheimer's Research to Scientific Advisory Board
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Releases Shareholder Update, Outlines Key Milestones for Company Progress
- BlockQuarry Corp. (OTC: BLQC) - InvestorNewsBreaks - BlockQuarry Corp (BLQC) Makes Executive Team Announcement, Notes Operational and Financial Outlook Update
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Buy Rating Increased in Ascendiant Capital Markets Latest Report
- Laredo Oil Inc. (OTC: LRDC) - InvestorNewsBreaks - Laredo Oil Inc.'s (LRDC) Subsidiary Secures Permit for Midfork Well
- Longeveron Inc. (NASDAQ: LGVN) - InvestorNewsBreaks - Longeveron Inc. (NASDAQ: LGVN) Schedules Release of Q1 2024 Financial Results, Earnings Conference Call/Webcast
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - InvestorNewsBreaks - Lexaria Bioscience Corp. (NASDAQ: LEXX) Launches New Research Program to Evaluate DehydraTECH-GLP-1 Drugs
- Life Electric Vehicles Holdings Inc. (OTC: LFEV) - InvestorNewsBreaks - Life Electric Vehicles Holdings Inc. (LFEV) Engages IBN for Corporate Communications Expertise
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com Inc. (NASDAQ: LTRY) Announces Soft Launch of Sweepstakes Operations to Benefit Street Child United
- LQwD Technologies Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - InvestorNewsBreaks - LQWD Technologies Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Releases Updated Info Regarding Bitcoin Lightning Network Operations
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Shines in the Basic Materials Sector
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (FSE: 0NFA) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Board Approves Issuance of Shares for Debt Settlement
- Mountain Top Properties Inc. (OTC: MTPP) - InvestorNewsBreaks - Mountain Top Properties Inc. (MTPP) to Tap into Resilient Hamptons' Real Estate Market
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Positive Nasdaq Listing Decision
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Web3 Summit in Dubai Ends Successfully
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Nutriband Inc. (NASDAQ: NTRB) - Fentanyl Scourge: Nutriband Inc.'s (NASDAQ: NTRB) New Transdermal Patch with Abuse-Deterrent Tech Funded to Seek FDA Approval
- OK Stone Engineering Inc. - InvestorNewsBreaks — OK Stone Engineering Partners with Oren Klaff at Special Investor Event
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc.'s (NASDAQ: MULN) Inks Agreement with NAFG, Board Adopts Limited Duration Stockholder Rights Plan
- PaxMedica Inc. (NASDAQ: PXMD) - PaxMedica Inc. (NASDAQ: PXMD) Announces PAX-101 Batch Completion for NDA Application to FDA
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Releases Third-Quarter 2024 Financial Report
- Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Financial, Operational Report for FY 2023
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Biweekly Status Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Partners with FluGen to Develop First-of-Its-Kind Intranasal Flu Vaccine for Global Distribution
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces New UB Product Line, Plans Investor Teleconference
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Announces Participation at Upcoming EF Hutton Global Conference
- Progressive Care Inc. (OTCQB: RXMD) - BioMedNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Developing Robust Product Lineup to Address ASD, Associated Neurodevelopmental Conditions
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Signs Definitive Agreement, Plan of Merger with Telix Pharmaceuticals
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - Deloitte Survey Finds 8 in 10 Investors Have ESG Guidelines
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTC: RWBYF) Files Documents Reporting Financial Results for Q4, FY 2023
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - US ESG Battle Now Shifts to Courts
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF), G Mining Ventures Announce Combination to Set the Stage for a Leading Intermediate Gold Producer in the Americas
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc.'s (RJDG) ioSoft Systems Division Engaged to 'Create Significant Enhancements' for Rail Pro Services
- N2OFF Inc. (NASDAQ: NITO) - InvestorNewsBreaks - N2OFF Inc. (NASDAQ: NITO) (FSE: 80W) to Target Hemp Flower Market in California
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Software Effective Solutions Corp. (OTC: SFWJ) - InvestorNewsBreaks - Software Effective Solutions Corp. (d/b/a MedCana) (SFWJ) Responds to Announcement Rescheduling of Marijuana by DEA
- Sigyn Therapeutics Inc. (OTCQB: SIGY) - BioMedNewsBreaks - Sigyn Therapeutics Inc. (SIGY) Leveraging Portfolio to Overcome Current Limitations in Healthcare
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Shares Positive Results for PTSD Treatment
- SUIC Worldwide Holdings Ltd. (OTC: SUIC) - SUIC Worldwide Holdings Ltd.'s (SUIC) I.Hart Group Expands Franchise Development Efforts with New Authorizations in China and Japan
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Reports Record Revenues, EBITDA and Gross Margin Growth, in Annual Fiscal Conference Call
- Siyata Mobile Inc. (NASDAQ: SYTA) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Releases Q4, Full-Year 2023 Financial Results
- SOBRsafe Inc. (NASDAQ: SOBR) - InvestorNewsBreaks - SOBR Safe Inc. (NASDAQ: SOBR) Sees Continued Growth, Signs Two New Contracts
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) CEO Talks Distribution in Recent Proactive Investors Interview
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Installs, Validates First Commercial-Scale DLE Column in North America
- Starco Brands Inc. (OTCQB: STCB) - Whipshots® by Starco Brands Shatters Expectations with Explosive Growth and Nationwide Expansion
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - InvestorNewsBreaks - Vision Marine Technologies Inc. (NASDAQ: VMAR) Announces Filing of Patent for Cooling System Control in Electric Marine Vessel
- SenesTech Inc. (NASDAQ: SNES) - InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Schedules Release of Q1 2024 Financial Results, Earnings Call
- Sekur Private Data Ltd. (FRA: GDT0) (CSE: SKUR) (OTCQB: SWISF) - InvestorNewsBreaks - Sekur Private Data Ltd. (CSE: SKUR) (OTCQX: SWISF) (FSE: GDT0) Featured in New to the Street Episode Airing Tonight
- TAAT Global Alternatives Inc. (CSE: TAAT) (FRANKFURT: 2TP) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- Tartisan Nickel Corp. (CSE: TN) (OTCQB: TTSRF) - Tartisan Nickel Corp. (CSE: TN) (OTCQB: TTSRF) (FSE: 8TA) Moves Toward Feasibility Study, Exploration at Kenbridge Nickel Project on Strength of PEA Report
- SOBRsafe Inc. (NASDAQ: SOBR) - InvestorNewsBreaks - SOBR Safe Inc. (NASDAQ: SOBR) Sees Continued Growth, Signs Two New Contracts
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Board Declares Special Cash Dividend
- Torr Metals Inc. (TSX.V: TMET) - Unlocking the Riches: Torr Metals Inc. (TSX.V: TMET) Aims to Test Untapped Copper and Gold Potential in British Columbia and Ontario, Canada
- Turbo Energy S.A. (NASDAQ: TURB) - InvestorNewsBreaks - Turbo Energy S.A. (NASDAQ: TURB) Announces Filing of Patent Application in Spain for Sunbox Commercial, Industrial Product
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Responds to Senate Passing of Bill Banning Russian Uranium Imports
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - InvestorNewsBreaks - Vision Marine Technologies Inc. (NASDAQ: VMAR) Announces Filing of Patent for Cooling System Control in Electric Marine Vessel
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Announces Participation at Upcoming Anxiety and Depression Association Conference
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) CEO Discusses Medicare Reimbursement Approval for CARE Oral Medical Devices in Proactive Interview
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Bolsters BolaWrap® 150 Deployments, Promoting Safer Communities Nationwide
- Zoned Properties Inc. (ZDPY) - Zoned Properties Inc. (ZDPY) Advancing Initiatives Through the Cannabis Sector
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Regains Compliance with Nasdaq Minimum Bid Price Requirement
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Announces Securities Purchase Agreements, Anticipates Receiving Approximately $4.2M from Placement
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Advanced Gold Exploration Inc. (CSE: AUEX) (FSE: 4TG) (OTC: AUHIF) - MiningNewsBreaks - Why Advanced Gold Exploration Inc. (CSE: AUEX) (OTC: AUHIF) (FSE: 4TG) Is 'One to Watch'
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Partners with Volunteer State Community College to Launch AI, Technical Training Programs
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) (MUN: A0I0) (BER: A0I0) Announces Promising Results at PHC Project in Brazil
- Fresh2 Group Ltd. (NASDAQ: FRES) - Appointment of New Independent Director
- Astiva Health - Astiva Health Inc. Innovative Holistic Approach Underscores Understanding That Effective Primary Care Is Foundational
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Goldman Sachs Predicts Surge in Commodity Prices as Central Banks Slash Rates
- Astrotech Corp. (NASDAQ: ASTC) - USDA Says Hemp Harvests, Prices are Rising
- Aston Bay Holdings Ltd. (TSX.V: BAY) (OTCQB: ATBHF) - InvestorNewsBreaks - Aston Bay Holdings Ltd. (TSX.V: BAY) (OTCQB: ATBHF), American West Metals Commence Drilling, Geophysics at Storm Copper Project
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Google Cloud to Bring Medically-Tuned AI Technology to Ambient Documentation
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Releases Year-End Review, Outlook for 2024
- Bebuzee Inc. (OTC: BBUZ) - InvestorNewsBreaks - Bebuzee Inc. (BBUZ) Announces Launch of Transformative Super App
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Announces Nasdaq Delisting Notification and Appeal, ADS Ratio Change
- Bravo Multinational Inc. (OTC: BRVO) - TechMediaBreaks - Bravo Multinational Inc. (BRVO) Positioning for Opportunity in Video Streaming Sector
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - InvestorNewsBreaks - BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: BXT) Announces Highly Promising Results from ODF Cladribine PK Study
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Notes 'Surge' in Revenue in FY 2023 Financial Report
- Btab Ecommerce Group Inc. (OTC: BBTT) - Btab Ecommerce Group Inc. (BBTT) Signs Letter of Intent with Integrated Wellness Acquisition Corp. (NYSE: WEL), Accelerating Company Growth
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Releases Update Report on Reid Drill Results
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Launches Proprietary StudioViz(TM) End-to-End Lidar Simulator
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc. (NASDAQ: CISO) Partners with Fortegra to Offer Protection Plans for Cybersecurity Clients
- Clene Inc. (NASDAQ: CLNN) - Study Suggests Metabolism Issues After Spinal-Cord Injury Result from Disturbed Neuronal Activity
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Researchers Seek to Develop Genetically Engineered Brain Cancer Treatment
- Correlate Energy Corp. (OTCQB: CIPI) - UN Secretary-General Says Climate Action Shouldn't Adversely Affect Poor
- Coyuchi Inc. - Louisiana Lawmakers Want to Restrict State Universities from Using ESG
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (Cboe CA: CYBN) Issues Corporate Update, Eyes Runway for Upcoming Clinical Milestones
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Two Contracts to Provide Integrated Cardiometabolic Solutions
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks - Diamond Lake Minerals Inc. (DLMI), BOXABL Announce Partnership to Bring Affordable Housing to the Public, Digital Tokens to Investors
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces 112% Revenue Increase, Significant Milestones for Fiscal Year 2023
- D-Wave Quantum Inc. (NYSE: QBTS) - Quantum Computing Could Make Genomic Analysis Hyper-Fast
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - Electronic Servitor Publication Network Inc. (XESP) Combines Powerful Digital Engagement Engine(TM) with Simple Pricing Model to Drive Growth of Client Companies
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Unconventional Market Factors Cause Gold Market to Rise
- Energy and Water Development Corp. (OTCQB: EAWD) - InvestorNewsBreaks — Energy and Water Development Corp. (EAWD) Selects IBN as Corporate Communications Partner
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Reports Financial Results, Operational Highlights for Q1 2024
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Releases Q4, Year-End 2023 Results; Schedules Earnings Call
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Receives PACEpilot Innovation to Watch Award
- FingerMotion Inc. (NASDAQ: FNGR) - US FCC Orders Halt to Broadband Services by Chinese Companies
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - Climate Change Puts Mining Companies in Catch-22
- Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) - Securing a Domestic Supply Chain: Why Fathom Nickel Inc. (CSE: FNI) (FSE: 6Q5) (OTCQB: FNICF) Matters for North America
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Exploration Update on Quebec Lithium Projects, Private Placement
- Flora Growth Corp. (NASDAQ: FLGC) - InvestorNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Responds to Reported Plans for DEA to Reschedule Cannabis
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Paving Way for Safer, More Efficient Autonomous Vehicles in Japan
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Reports 30% YOY Revenue Growth for Q1 2024; Projects $25-$30m Annual Revenue for 2024
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Names New CEO, Outlines Key Components of Strategic Expansion Plans
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - York Trial Program Graduates Second Cohort of Green-Job Students
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- GivBux Inc. (OTC: GBUX) - GivBux Inc. (GBUX) Is 'One to Watch'
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc.'s (NASDAQ: GMGI) Meridianbet Setting New Standard for Customer Engagement, Community Impact
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) CEO Featured Guest in Most Recent Bell2Bell Podcast
- GolfLync Inc. - InvestorNewsBreaks - GolfLync Inc. Reaches 100K Membership Milestone, Sees 3,033% Growth Rate in 2023
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation at Upcoming Investor, Industry Events
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Releases Record 2023 Financial Report Showing 100% Increase in Revenue
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Presents Key Data in First-Ever Scientific Presentation of Proprietary HeartBeam AI
- HealthLynked Corp. (OTCQB: HLYK) - HealthLynked Corp. (HLYK) Q4, FY2023 Financial Results; Show Bottom-line Improvements, Core Network Application Ramp-Up Expected in 2024
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Closes Final Tranche of Nonbrokered Private Placement
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Delves into Key Dual Listing Considerations for Issues
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Termination of Merger Deal
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc.'s (NASDAQ: IDEX) Solectrac Announces Strategic Reorganization
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Releases Interim Results from Phase 2 Study Evaluating Treatment to Reduce Alzheimer's Agitation
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces Delayed Effective Date of Reverse Split
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Welcomes Leading Authority in Alzheimer's Research to Scientific Advisory Board
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Releases Shareholder Update, Outlines Key Milestones for Company Progress
- BlockQuarry Corp. (OTC: BLQC) - InvestorNewsBreaks - BlockQuarry Corp (BLQC) Makes Executive Team Announcement, Notes Operational and Financial Outlook Update
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Buy Rating Increased in Ascendiant Capital Markets Latest Report
- Laredo Oil Inc. (OTC: LRDC) - InvestorNewsBreaks - Laredo Oil Inc.'s (LRDC) Subsidiary Secures Permit for Midfork Well
- Longeveron Inc. (NASDAQ: LGVN) - InvestorNewsBreaks - Longeveron Inc. (NASDAQ: LGVN) Schedules Release of Q1 2024 Financial Results, Earnings Conference Call/Webcast
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - InvestorNewsBreaks - Lexaria Bioscience Corp. (NASDAQ: LEXX) Launches New Research Program to Evaluate DehydraTECH-GLP-1 Drugs
- Life Electric Vehicles Holdings Inc. (OTC: LFEV) - InvestorNewsBreaks - Life Electric Vehicles Holdings Inc. (LFEV) Engages IBN for Corporate Communications Expertise
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com Inc. (NASDAQ: LTRY) Announces Soft Launch of Sweepstakes Operations to Benefit Street Child United
- LQwD Technologies Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - InvestorNewsBreaks - LQWD Technologies Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Releases Updated Info Regarding Bitcoin Lightning Network Operations
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Shines in the Basic Materials Sector
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (FSE: 0NFA) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Board Approves Issuance of Shares for Debt Settlement
- Mountain Top Properties Inc. (OTC: MTPP) - InvestorNewsBreaks - Mountain Top Properties Inc. (MTPP) to Tap into Resilient Hamptons' Real Estate Market
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Positive Nasdaq Listing Decision
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Web3 Summit in Dubai Ends Successfully
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Nutriband Inc. (NASDAQ: NTRB) - Fentanyl Scourge: Nutriband Inc.'s (NASDAQ: NTRB) New Transdermal Patch with Abuse-Deterrent Tech Funded to Seek FDA Approval
- OK Stone Engineering Inc. - InvestorNewsBreaks — OK Stone Engineering Partners with Oren Klaff at Special Investor Event
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc.'s (NASDAQ: MULN) Inks Agreement with NAFG, Board Adopts Limited Duration Stockholder Rights Plan
- PaxMedica Inc. (NASDAQ: PXMD) - PaxMedica Inc. (NASDAQ: PXMD) Announces PAX-101 Batch Completion for NDA Application to FDA
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Releases Third-Quarter 2024 Financial Report
- Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Financial, Operational Report for FY 2023
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Biweekly Status Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Partners with FluGen to Develop First-of-Its-Kind Intranasal Flu Vaccine for Global Distribution
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces New UB Product Line, Plans Investor Teleconference
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Announces Participation at Upcoming EF Hutton Global Conference
- Progressive Care Inc. (OTCQB: RXMD) - BioMedNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Developing Robust Product Lineup to Address ASD, Associated Neurodevelopmental Conditions
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Signs Definitive Agreement, Plan of Merger with Telix Pharmaceuticals
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - Deloitte Survey Finds 8 in 10 Investors Have ESG Guidelines
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTC: RWBYF) Files Documents Reporting Financial Results for Q4, FY 2023
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - US ESG Battle Now Shifts to Courts
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF), G Mining Ventures Announce Combination to Set the Stage for a Leading Intermediate Gold Producer in the Americas
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc.'s (RJDG) ioSoft Systems Division Engaged to 'Create Significant Enhancements' for Rail Pro Services
- N2OFF Inc. (NASDAQ: NITO) - InvestorNewsBreaks - N2OFF Inc. (NASDAQ: NITO) (FSE: 80W) to Target Hemp Flower Market in California
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Software Effective Solutions Corp. (OTC: SFWJ) - InvestorNewsBreaks - Software Effective Solutions Corp. (d/b/a MedCana) (SFWJ) Responds to Announcement Rescheduling of Marijuana by DEA
- Sigyn Therapeutics Inc. (OTCQB: SIGY) - BioMedNewsBreaks - Sigyn Therapeutics Inc. (SIGY) Leveraging Portfolio to Overcome Current Limitations in Healthcare
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Shares Positive Results for PTSD Treatment
- SUIC Worldwide Holdings Ltd. (OTC: SUIC) - SUIC Worldwide Holdings Ltd.'s (SUIC) I.Hart Group Expands Franchise Development Efforts with New Authorizations in China and Japan
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Reports Record Revenues, EBITDA and Gross Margin Growth, in Annual Fiscal Conference Call
- Siyata Mobile Inc. (NASDAQ: SYTA) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Releases Q4, Full-Year 2023 Financial Results
- SOBRsafe Inc. (NASDAQ: SOBR) - InvestorNewsBreaks - SOBR Safe Inc. (NASDAQ: SOBR) Sees Continued Growth, Signs Two New Contracts
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) CEO Talks Distribution in Recent Proactive Investors Interview
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Installs, Validates First Commercial-Scale DLE Column in North America
- Starco Brands Inc. (OTCQB: STCB) - Whipshots® by Starco Brands Shatters Expectations with Explosive Growth and Nationwide Expansion
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - InvestorNewsBreaks - Vision Marine Technologies Inc. (NASDAQ: VMAR) Announces Filing of Patent for Cooling System Control in Electric Marine Vessel
- SenesTech Inc. (NASDAQ: SNES) - InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Schedules Release of Q1 2024 Financial Results, Earnings Call
- Sekur Private Data Ltd. (FRA: GDT0) (CSE: SKUR) (OTCQB: SWISF) - InvestorNewsBreaks - Sekur Private Data Ltd. (CSE: SKUR) (OTCQX: SWISF) (FSE: GDT0) Featured in New to the Street Episode Airing Tonight
- TAAT Global Alternatives Inc. (CSE: TAAT) (FRANKFURT: 2TP) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- Tartisan Nickel Corp. (CSE: TN) (OTCQB: TTSRF) - Tartisan Nickel Corp. (CSE: TN) (OTCQB: TTSRF) (FSE: 8TA) Moves Toward Feasibility Study, Exploration at Kenbridge Nickel Project on Strength of PEA Report
- SOBRsafe Inc. (NASDAQ: SOBR) - InvestorNewsBreaks - SOBR Safe Inc. (NASDAQ: SOBR) Sees Continued Growth, Signs Two New Contracts
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Board Declares Special Cash Dividend
- Torr Metals Inc. (TSX.V: TMET) - Unlocking the Riches: Torr Metals Inc. (TSX.V: TMET) Aims to Test Untapped Copper and Gold Potential in British Columbia and Ontario, Canada
- Turbo Energy S.A. (NASDAQ: TURB) - InvestorNewsBreaks - Turbo Energy S.A. (NASDAQ: TURB) Announces Filing of Patent Application in Spain for Sunbox Commercial, Industrial Product
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Responds to Senate Passing of Bill Banning Russian Uranium Imports
- Vision Marine Technologies Inc. (NASDAQ: VMAR) - InvestorNewsBreaks - Vision Marine Technologies Inc. (NASDAQ: VMAR) Announces Filing of Patent for Cooling System Control in Electric Marine Vessel
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Announces Participation at Upcoming Anxiety and Depression Association Conference
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) CEO Discusses Medicare Reimbursement Approval for CARE Oral Medical Devices in Proactive Interview
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Bolsters BolaWrap® 150 Deployments, Promoting Safer Communities Nationwide
- Zoned Properties Inc. (ZDPY) - Zoned Properties Inc. (ZDPY) Advancing Initiatives Through the Cannabis Sector
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































