The QualityStocks Daily Stock List
- BK Technologies (BKTI)
- Elite Pharmaceuticals (ELTP)
- Zacatecas Silver (ZCTSF)
- HPQ Silicon (HPQFF)
- Elevation Gold Mining (EVGDF)
- Guru Organic Energy (GUROF)
- Nuo Therapeutics (AURX)
- SRM Entertainment Inc. (SRM)
- RVL Pharmaceuticals plc (RVLP)
- Mind Medicine Inc. (MNMD)
- Southern Copper Corporation (SCCO)
- Tesla Inc. (TSLA)
BK Technologies (BKTI)
The Online Investor, MarketBeat, StockMarketWatch, QualityStocks and TradersPro reported earlier on BK Technologies (BKTI), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
BK Technologies Corp. (NYSE American: BKTI) is a holding firm that is focused on designing, manufacturing and marketing wireless communication products.
The firm has its headquarters in West Melbourne, Florida and was incorporated in 1997, on October 24th. It operates as part of the communications equipment manufacturing industry in the communications sector. The firm serves consumers around the globe and has four companies in its corporate family.
The company manufactures high-specification communications equipment that is valuable and reliable and can be used by government agencies and public safety professionals. Its products are manufactured and distributed globally under the RELM, BKR, BK Technologies and BK Radio brand names.
The enterprise provides base stations, repeaters, LMR (2-way land mobile radios) and related subsystems and components. Its RELM branded products offer 2-way communications for industrial and commercial concerns like transportation, schools, construction firms and hotels services. Its BK Radio, BKR and BK Technologies branded products include P-25 digital products, LMR equipment for professional radio users mainly in the military and other agencies of the local, state and federal governments. The enterprise also serves the commercial, industrial and business markets which include airlines, construction companies, educational institutions, railroads and disaster recovery; and public safety and government markets, which include emergency medical personnel, law enforcement, rescue, fire and homeland security.
The firm recently released its second quarter financial results for 2021 which show increases in revenue, reflecting the strong demand for its communications technology. It is focused on the development and launching of new products, including its multiband BKR 9000 product, which will position the firm to compete for bigger contracts in new verticals.
BK Technologies (BKTI), closed Thursday's trading session at $10.346, up 15.5978%, on 35,455 volume. The average volume for the last 3 months is 1.899M and the stock's 52-week low/high is $8.79/$19.90.
Elite Pharmaceuticals (ELTP)
QualityStocks, Stockpalooza, PennyTrader Publisher, PennyStocks24, OTCPicks, MarketBeat, Real Pennies, Capital Equity Report, MadPennyStocks, Marketbeat.com, PennyStock MarketBulls, SmarTrend Newsletters, First Penny Picks, Stock Analyzer, Top Stock Picks, Pennybuster, PennyInvest, TopPennyStockMovers, OTCBB Journal, Greenbackers, StockEgg, RagingStock Bull, CRWEPicks, FeedBlitz, Penny Pick Finders, HotOTC, HotStockChat, CoolPennyStocks, Wise Alerts, Buzz Stocks, BullRally, Beacon Equity Research, TheMicrocapNews, MicrocapVoice, GoldminePennyStocks, StockOnion, Stock Fortune Teller, SmallCapVoice, SmallCap Network, SecretStockPromo, Stock Preacher, Pumps and Dumps, StocksImpossible, Planet Penny Stocks, OTC Advisors, PennyStockVille, WiseAlerts, StockRich, Alternative Energy, Penny Stock Rumble, Penny Invest, PennyStockProphet and Promotion Stock Secrets reported earlier on Elite Pharmaceuticals (ELTP), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Elite Pharmaceuticals Inc. (OTCQB: ELTP) is a pharmaceutical firm that is focused on conducting research on, developing, manufacturing and licensing drug delivery products and systems.
The firm’s headquarters are in the state of New Jersey, United States and was incorporated in 1997, on October 1st. It operates as part of the healthcare industry, under the biotech and pharma sub-industry. The firm serves consumers around the globe.
The company is party to a strategic marketing alliance with Lannett Company Inc. and Glenmark Pharmaceuticals Inc. It is also party to a development and license agreement with SunGen Pharma LLC.; and a manufacturing and license agreement with Epic Pharma LLC. The company operates through the New Drug applications for branded and generic products.
The enterprise’s products include Isradipine capsules for cardiovascular ailments; Naltrexone tablets for pain management; Phendimetrazine Tartrate tablets for bariatrics; and HydromorphoneHCl tablets for pain. It also offers Methadone HCl tablets for pain; Trimipramine Maleate Immediate Release antidepressant capsules; Amphetamine sulfate immediate release, Dextroamphetamine sulfate and Dextroamphetamine saccharate tablets for central nervous system diseases. In addition to this, the enterprise manufactures controlled-release products on a contract basis for 3rd parties, in the areas of infection, bariatric, allergy and pain. The enterprise is also focused on the development of a range of abuse deterrent opioid products.
The firm recently released it latest financial results for the year 2021, which show increases in its revenues and profits. Currently, it is focused on diversifying its products in order to stabilize the firm and support its growth.
Elite Pharmaceuticals (ELTP), closed Thursday's trading session at $0.059, up 20.4082%, on 1,898,664 volume. The average volume for the last 3 months is 3,707 and the stock's 52-week low/high is $0.0269/$0.06.
Zacatecas Silver (ZCTSF)
We reported earlier on Zacatecas Silver (ZCTSF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Zacatecas Silver Corp (OTC: ZCTSF) (CVE: ZAC) (FRA: 7TV) is a mineral resource firm focused on acquiring, exploring for and evaluating mineral properties in Mexico.
The firm has its headquarters in Vancouver, Canada and was incorporated in 2020, on July 22nd. It operates as part of the silver industry, under the basic materials sector. The firm serves consumers around the globe.
The company primarily explores for silver and gold resources. It owns 100% interests in the Zacatecas project, which comprises of 149 mining concessions. The Zacatecas Silver property is in Zacatecas State, Mexico, within the prospective Fresnillo Silver Belt. The company holds approximately 7,826 hectares (19,338 acres) of ground that is for low and intermediate sulphidation silver-base metal mineralization and potentially low sulphidation gold-dominant mineralization. The company’s property shares common boundaries with Pan American Silver Corp. claims and El Orito which is owned by Endeavour Silver. There are four main high-grade silver target areas within the Zacatecas concessions: the Panuco Deposit, Muleros, El Cristo and San Manuel-San Gill. Its property also includes El Oro, El Orito, La Cantera, Monserrat, El Penon, San Judas and San Juan silver-base metal vein targets. It also holds 100% interests in the Esperanza project, which comprises of 7 mining concessions that cover an area of 4,649 hectares located in the Morelos State, Mexico.
The firm, which recently held its annual AGM, remains focused on advancing its exploration efforts at its properties. Their success will bring in additional revenues into the firm while also creating shareholder value.
Zacatecas Silver (ZCTSF), closed Thursday's trading session at $0.1853, off by 2.4737%, on 3,707 volume. The average volume for the last 3 months is 38,770 and the stock's 52-week low/high is $0.18/$0.785.
HPQ Silicon (HPQFF)
We reported earlier on HPQ Silicon (HPQFF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
HPQ Silicon Inc. (OTCQB: HPQFF) (CVE: HPQ) (FRA: O08) is an innovative silicon solutions firm that is engaged in the provision of silica (SiO2) and silicon-based (Si) solutions.
The firm has its headquarters in Montreal, Canada and was incorporated in 1996, on December 20th. Prior to its name change in July 2022, the firm was known as HPQ-Silicon Resources Inc. It operates as part of the other industrial metals and mining industry, under the basic materials sector. The firm serves consumers around the globe, with a focus on those in Canada.
The company is developing a portfolio of high-value-added silicon (Si) products sought after by battery and electric vehicle manufacturers, among other industries. It is engaged in developing the PUREVAPTM Quartz Reduction Reactors, which can permit the one-step transformation of quartz (SiO2) into high-purity silicon (Si). The company is developing the PUREVAPTM Nano Silicon Reactor (NSiR) through its subsidiary, HPQ NANO Silicon Powders Inc. NSiR is a process that can use material produced by the QRR as feedstock, to make a wide range of nano/micro spherical powders and nanowires of different sizes. Through its subsidiary, HPQ Silica POLVERE Inc., the company is developing a plasma-based process that allows direct Quartz to Fumed Silica transformation, removing the use of hazardous chemicals in the making of Fumed silica and eliminating the hydrogen chloride gas associated with its manufacturing.
The firm, which recently provided its shareholders with an update on the PUREVAPTM Gen3 Quartz Reduction Reactor project, is well-positioned for significant growth given that the demand for silicon is projected to surpass 3.8 million tons by 2025. This will generate significant value for its shareholders while also opening it up to new investment opportunities.
HPQ Silicon (HPQFF), closed Thursday's trading session at $0.265, up 2.8328%, on 38,770 volume. The average volume for the last 3 months is 24,645 and the stock's 52-week low/high is $0.1375/$0.3074.
Elevation Gold Mining (EVGDF)
We reported earlier on Elevation Gold Mining (EVGDF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Elevation Gold Mining Corp (OTCQX: EVGDF) (CVE: ELVT) (FRA: 54NA) is a precious metals exploration and mining firm that is focused on acquiring, exploring for, developing and operating precious metal properties.
The firm has its headquarters in Vancouver, Canada and was incorporated in 2007, on June 7th. Prior to its name change in September 2021, the firm was known as Northern Vertex Mining Corp. It operates as part of the gold industry, under the basic materials sector. The firm primarily serves consumers in the United States.
The company primarily explores for gold and silver deposits. Its principal operation is the 100% owned Moss Mine in Mohave County, Arizona. The company also holds the title to the Hercules exploration property, located in Lyon County, Nevada, which is a prospective gold exploration project. The Moss Gold Mine is located in Mohave County in northwestern Arizona, approximately an hour and a half’s drive south of Las Vegas, Nevada. The Moss Mine is mining the Moss vein system, which consists of fault-hosted epithermal quartz-calcite veins with associated vein stockwork. The Moss mine extracts gold and silver from ore via heap leaching. The Hercules Gold Project covers approximately 1,323 unpatented and approximately four patented lode mining claims, covering an area of approximately 10,000 hectares. The company generates revenue from the sale of gold and silver.
The enterprise recently announced its latest financial results, which show increases in its revenues. It remains committed to increasing gold production, which will bring in additional revenues into the firm.
Elevation Gold Mining (EVGDF), closed Thursday's trading session at $0.07516, even for the day. The average volume for the last 3 months is 1,370 and the stock's 52-week low/high is $0.0679/$0.1869.
Guru Organic Energy (GUROF)
Trades Of The Day, MarketBeat and Daily Trade Alert reported earlier on Guru Organic Energy (GUROF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Guru Organic Energy Corp. (OTC: GUROF) (TSE: GURU) (FRA: GUU) is a dynamic, fast-growing beverage firm that is engaged in the provision of natural, organic and plant-based energy drinks.
The firm has its headquarters in Montreal, Canada and was incorporated in 1999 by Joseph Zakher. It operates as part of the beverages-non-alcoholic industry, under the consumer defensive sector. The firm primarily serves consumers inCanada and the United States.
The company was the first to launch a natural, plant-based energy drink globally, in1999. It is committed to achieving its mission of cleaning the energy drink industry in Canada and the United States and hasbuilt an inspiring brand with a clean list of organic ingredients, including natural caffeine, with zero sucralose and zero aspartame, which offer consumers Feel Good Energy that never comes at the expense of their health. The company’s subsidiaries include GURU Beverage Inc. and GURU Beverage Co.
The enterprise’s products include GURU Organic Energy - Guayusa Tropical Punch, GURU Organic Energy - Lite, GURU Organic Energy - Yerba Mate and GURU Organic Energy - Matcha. It manufactures its products using ingredients such as green tea, matcha, guarana, ginseng, echinacea, monk fruit and stevia. The enterprise markets organic energy drinks in Canada and the United States through a distribution network of over 25,000 points of sale, and through guruenergy.com and Amazon.
The firm, which recently released its latest financial results, remains committed to growing its market share and generating sustainable long-term profitable growth for its shareholders.
Guru Organic Energy (GUROF), closed Thursday's trading session at $1.58, off by 0.132735%, on 1,370 volume. The average volume for the last 3 months is 40 and the stock's 52-week low/high is $1.3794/$6.93.
Nuo Therapeutics (AURX)
We reported earlier on Nuo Therapeutics (AURX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Nuo Therapeutics Inc. (OTCQB: AURX) is a regenerative therapies firm that is focused on developing, commercializing and marketing cell-based technologies that harness the regenerative capacity of the human body to trigger natural healing.
The firm has its headquarters in Houston, Texas and was incorporated in 1998, on April 29th. Prior to its name change in November 2014, the firm was known as Cytomedix Inc. It operates as part of the biotechnology industry, under the healthcare sector. The firm mainly serves consumers in the United States.
The enterprise’s flagship product, Aurix, is a biodynamic hematogel that uses a patient’s own platelets and plasma as a catalyst for healing. Its commercial offering comprises of point of care technology for the safe and effective separation of autologous blood to produce a platelet-based therapy for the chronic wound care market. This offering is known as Aurix (the Aurix System), which produces a platelet-rich plasma (PRP) gel at the point of care using the patient’s own platelets and plasma sourced from a small draw of peripheral blood. Aurix comprises a natural, endogenous complement of protein and non-protein signal molecules that contribute to effective healing. It is the only therapy of its kind that is FDA-cleared for use on ulcers and wounds of all types. The enterprise derives revenues from the sale of the Aurix product to customers consisting of primarily hospital outpatient wound care clinics and other private practice physicians treating chronic wounds.
The firm was recently selected to exhibit the Aurix System at the Vizient Innovative Technology Exchange, an offer that will allow it to demonstrate their product to supply chain and clinical leaders from all around and help extend its consumer reach greatly.
Nuo Therapeutics (AURX), closed Thursday's trading session at $1.25, even for the day. The average volume for the last 3 months is 331,502 and the stock's 52-week low/high is $0.8502/$4.50.
SRM Entertainment Inc. (SRM)
Stockhouse reported earlier on SRM Entertainment Inc. (SRM), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
SRM Entertainment (NASDAQ: SRM), a company that designs, develops and manufactures custom toys and souvenirs for the world's largest theme parks and entertainment venues, has closed on its initial public offering. The offering was comprised of 1,250,000 shares of its common stock that were offered to the public at an initial price of $5 per share. According to the announcement, SRM shares began trading on the NASDAQ Capital Market on Aug. 15, 2023. The offering resulting in gross proceeds of $6.25 million, before standard underwriting discounts and commissions and other related expenses are deducted. The announcement also noted that the company has granted the underwriters a 45-day option to purchase up to an additional 187,500 shares of common stock at the same public offering price, minus underwriting discounts and commissions.
To view the full press release, visit https://ibn.fm/BBTNo
About SRM Entertainment Inc.
SRM Entertainment designs, develops and manufactures custom toys and souvenirs for the world's largest theme parks and entertainment venues. The company provides exclusive custom products that are available worldwide at venues such as Walt Disney Parks and Resorts, Universal Studios, SeaWorld and other attractions. The company also recently launched its retail product line, which includes patented SMURFS Sip with Me cups. For additional information about this company, please visit www.SRMEntertainment.com.
SRM Entertainment Inc. (SRM), closed Thursday's trading session at $3.42, off by 5%, on 333,731 volume. The average volume for the last 3 months is 1.191M and the stock's 52-week low/high is $3.35/$6.01.
RVL Pharmaceuticals plc (RVLP)
QualityStocks and MarketBeat reported earlier on RVL Pharmaceuticals plc (RVLP), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
RVL Pharmaceuticals (NASDAQ: RVLP), a specialty pharmaceutical company focused on the commercialization of UPNEEQ(R) (oxymetazoline hydrochloride ophthalmic solution) (“UPNEEQ”), 0.1%, for the treatment of acquired blepharoptosis, or low-lying eyelid, in adults, is featured in a recent Cantor Fitzgerald research brief. Noting that the stock “looks even more interesting post solid quarter and ahead of potential strategic acquisition,” the research brief reiterated its previous Overweight (“OW”) rating and 12-month price target of $6 for RVL Pharmaceutical shares, stating that RVLP’s commercial opportunities continue to be underappreciated and that upward earnings estimate revisions should drive the stock higher. The brief noted that RVLP’s prescription eye drop, UPNEEQ 0.1%, had been selected the winner in the professional treatment category as “Best Eye Drop for Drooping Lids” in the NewBeauty annual beauty awards, and that the company is in discussions with strategic targets that could accelerate UPNEEQ sales, broaden RVLP’s portfolio and leverage the field force investment.
“RVLP said it is in advanced discussions with certain companies that it could potentially partner with or acquire to support growth and integrate into its infrastructure with meaningful synergies,” stated the Cantor Fitzgerald report. “RVLP [also] reported second quarter 2023 UPNEEQ net product sales of $8.3 million and operating expenses of $14.4 million, down 2% and 32%, year over year, respectively. RVLP highlighted its lower baseline of operating expenses. It streamlined its operating expenses to extend its cash runway, optimize its marketing mix and support strategic business development. This streamlining had a modest unfavorable impact on sales during the quarter. And at 6/30/23, the company had cash and cash equivalents of $19.2 million and senior secured indebtedness with aggregate principal maturities of $70.7 million.”
To view the full press release, visit https://ibn.fm/B1Eph
About RVL Pharmaceuticals plc
RVL Pharmaceuticals plc is a specialty pharmaceutical company focused on the commercialization of UPNEEQ (oxymetazoline hydrochloride ophthalmic solution), 0.1%, for the treatment of acquired blepharoptosis, or low-lying eyelid, in adults. UPNEEQ is the first nonsurgical treatment option approved by the FDA for acquired blepharoptosis. For additional information about this company, please visit www.RVLPharma.com.
RVL Pharmaceuticals plc (RVLP), closed Thursday's trading session at $0.18, off by 13.0435%, on 1,202,843 volume. The average volume for the last 3 months is 459,595 and the stock's 52-week low/high is $0.18/$2.99.
Mind Medicine Inc. (MNMD)
QualityStocks, InvestorPlace, Schaeffer's, The Wealth Report, The Street, MarketBeat, The Stock Dork, MarketClub Analysis, Daily Trade Alert and Trades Of The Day reported earlier on Mind Medicine Inc. (MNMD), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
A new study published in the journal “Frontiers in Psychology” suggests that psychedelic use and mindfulness meditation may trigger leadership improvements and result in positive leadership outcomes. The preliminary study joins a growing body of scientific literature on the impact of psychedelic use on various facets of mental health.
Prior research has found that psychedelics may be effective at treating a myriad of severe mental health conditions at minimal doses and with barely any adverse side effects. Researchers have been particularly interested in how psychedelics affect the brain and whether they could leverage these effects to treat anything from anxiety and depression to post-traumatic stress disorder (PTSD).
The team behind the recent study was spurred by anecdotal reports by individuals who claimed that they used psychedelic states and mindfulness to improve their leadership and achieve peak growth.
Past studies had shown that mindfulness meditation can have a broad range of effects including helping to reduce stress and improve emotional regulation, sleep, and creativity. As such, the recent study focused on the effects of psychedelics such as LSD and mindfulness on growth and leadership skills with the aim of determining if this combination had the potential to improve leadership development.
Corresponding study author and Karolinska Institute’s Department of Clinical Neuroscience postdoctoral researcher Otto Simonsson noted that although there is some research on how psychedelic and mindfulness medication can affect mental health, there is hardly any scientific literature on whether these two altered states can impact leadership development. Simonsson and his colleagues used two samples of participants with 4,867 U.S. adults and 4,865 adults from the United Kingdom from the Prolific Academic platform.
Researchers asked the participants to provide responses on how psychedelic experiences or mindful meditation affected their leadership. The researchers then broke the written responses down into classes such as “no negative impact on leadership,” “no impact” and “positive impact.”
The study reported that 43.6% of the 3,150 study participants with management positions reported trying mindfulness meditation, only 17.7% said they had experience with psychedelics, and around 10% had tried both psychedelics and mindfulness meditation. Slightly more than 70% reported a positive impact on their leadership practices due to mindfulness meditation, 28.8% said mindfulness meditation had no impact, and just 0.2% reported a negative impact.
Furthermore, 40.6% indicated a positive impact after psychedelic use, 58.0% said psychedelic use had no impact, and 1.4% said psychedelic use had a negative impact on leadership practices.
As many more entities such as Mind Medicine Inc. (NASDAQ: MNMD) (NEO: MMED) (DE: MMQ) publish their findings about the therapeutic and wellness benefits of psychedelics, these substances could see widespread popularity among members of the public.
Mind Medicine Inc. (MNMD), closed Thursday's trading session at $3.94, off by 4.6005%, on 462,426 volume. The average volume for the last 3 months is 879,045 and the stock's 52-week low/high is $2.12/$19.95.
Southern Copper Corporation (SCCO)
MarketBeat, SmarTrend Newsletters, InvestorPlace, The Street, QualityStocks, Louis Navellier, The Online Investor, Daily Wealth, The Wealth Report, Daily Trade Alert, TopStockAnalysts, Trades Of The Day, StreetAuthority Daily, Zacks, Marketbeat.com, Barchart, Early Bird, TheStockAdvisor, Money Morning, DividendStocks, Kiplinger Today, Market Intelligence Center Alert, Schaeffer's, Market Authority, Investopedia, Uncommon Wisdom, MarketClub Analysis, Top Pros' Top Picks, The Growth Stock Wire, INO.com Market Report, Investment House, Cabot Wealth, ChartAdvisor, MiningNewsWire, The Stock Enthusiast, StreetInsider, TheStockAdvisors, InvestmentHouse, AllPennyStocks, BestOtc, CRWEFinance, DrStockPick, Greenbackers, CRWEPicks, Investiv, Investing Futures, Dividend Opportunities, Forbes, CRWEWallStreet, StockLockandLoad, Wealth Insider Alert, Wealth Daily, Vantage Wire, TradingMarkets, TradingAuthority Daily, TradersPro, The Tycoon Report, The Trading Report, The Motley Fool, MarketDNA, StockRockandRoll, Investor Update, StockHotTips, Profit Confidential, PennyToBuck, PennyOmega, Navellier Growth, Money and Markets, 24/7 Trader, InvestorsObserver Team, InvestorIntel, InvestorGuide and Streetwise Reports reported earlier on Southern Copper Corporation (SCCO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Recent data from the Consumer Price Index (CPI) has reduced investor interest in gold and copper, causing prices for the two metals to falter amid mixed cues about inflation in the United States. Prices for the precious metal steadied last week after a month of relative lows while copper prices went down partly thanks to growing concerns over reduced imports by China, arguably the largest copper consumer on the globe.
CPI data showed that consumer inflation levels in the U.S. went up from June to July and were well over the U.S. Federal Reserve’s annual inflation target range. The data inspired reduced optimism in the capital markets and contributed to an increase in volatility levels in stocks and risk-driven assets, pushing more investors away from gold as a safe haven to the dollar.
Gold has traditionally seen an increase in demand and prices during times of economic upheaval because it can allow investors to retain the value of their holdings. However, even though the global economy has been wracked by issue after issue for several years, actions by the U.S. Fed and other central banks have hampered investor interest in the precious metal.
As the energy crisis worsened and most countries around the world faced increasing living costs, the Fed increased benchmark interest rates to fight against inflation. Consecutive interest rate hikes in 2022 and 2023 increased investor interest in assets that would pay them interest rates and reduced demand for gold even though conditions were ripe for gold to flourish as a safe-haven asset.
All indications show that the Fed will keep benchmark interest rates steady next month, which will likely have a dampening effect on gold demand and prices.
Copper, on the other hand, saw its prices fall to under $8,000 a ton in May due to poor economic data from major copper consumer China. While most of the world loosened and ultimately removed coronavirus lockdown policies, China kept large swathes of territory locked down well into late 2023, crippling the nation’s economic growth and hampering the global economy as well.
Although copper gained when China finally ended lockdowns and began ramping up manufacturing to pre-COVID levels, the red metal has lost most of these gains. Amalgamated Metal Trading’s head of research Dan Smith said that May was an especially slow month for industrial metals in terms of demand. He noted that disappointing economic data from China has also added pressure to metals markets and contributed to their poor performance in recent months.
Major copper producers such as Southern Copper Corporation (NYSE: SCCO) are likely to keep tabs on how developments in China unfold so that they get an idea about how market conditions could evolve over the coming months and years.
Southern Copper Corporation (SCCO), closed Thursday's trading session at $79.95, off by 0.435865%, on 891,789 volume. The average volume for the last 3 months is 119.548M and the stock's 52-week low/high is $42.42/$87.59.
Tesla Inc. (TSLA)
The Street, InvestorPlace, Green Car Stocks, StreetInsider, Schaeffer's, Kiplinger Today, Investopedia, MarketClub Analysis, Zacks, MarketBeat, The Online Investor, Daily Trade Alert, Money Morning, Options Elite, StreetAuthority Daily, Trades Of The Day, Market Intelligence Center Alert, Energy and Capital, All about trends, Cabot Wealth, StocksEarning, TopStockAnalysts, Wealth Daily, CNBC Breaking News, Uncommon Wisdom, InvestorGuide, Barchart, The Motley Fool, Early Bird, Street Insider, MarketWatch, Daily Profit, Profit Confidential, Marketbeat.com, Louis Navellier, Money and Markets, ProfitableTrading, AllPennyStocks, Investors Alley, InvestorIntel, Alternative Energy, SmarTrend Newsletters, Wyatt Investment Research, Wealth Insider Alert, StrategicTechInvestor, Trading Tips, Investor Guide, Top Pros' Top Picks, Wall Street Daily, Investing Daily, Greenbackers, TipRanks, CustomerService, Market Intelligence Center, StockEarnings, Investment U, The Wealth Report, smartmoneytrading, INO Market Report, FreeRealTime, Stock Up Featured, The Street Report, MarketTamer, INO.com Market Report, Daily Wealth, Trading Concepts, Wall Street Elite, wyatt research newsletter, Hit and Run Candle Sticks, The Growth Stock Wire, National Inflation Association, Investiv, SureMoney, Money Wealth Matters, Jon Markman’s Pivotal Point, Eagle Financial Publications, StockMarketWatch, Short Term Wealth, GorillaTrades, Wall Street Profit Search and Investing Futures reported earlier on Tesla Inc. (TSLA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
A recent report from Japanese publication Nikkei has revealed that 4 out of every 10 batteries in Tesla Inc. (NASDAQ: TSLA) electric vehicles are sourced from China. The report brings attention to China’s stranglehold over the electric vehicle battery metals segment and America’s reliance on the eastern Asian nation for critical mineral supplies.
Arguably the largest electric vehicle maker on the globe, Tesla pioneered the modern electric vehicle and encouraged both new and established automakers to begin developing alternate energy cars such as battery electric cars. The key component in each EV is the massive rechargeable battery pack, usually installed along the bottom of the vehicle, that powers the car and supplies all of its energy needs.
These lithium-ion battery packs use metals such as lithium, cobalt and nickel, whose supply is significantly reliant on either Chinese mining or processing. Nikkei leveraged machine-learning algorithms to analyze Tesla’s global complex supply chain network and found that the American EV maker has more than 13,000 suppliers globally. The publication estimates that at least 39% of Tesla’s battery materials come from China.
With China dominating key segments in the mining industry, including accounting for 36% of worldwide global reserves and handling 60% of global lithium refining, it’s not surprising that Tesla has some Chinese companies in its supply web. The Nikkei report also stated that China holds the largest amount of companies that specialize in “inorganic chemistry” in the world, further extending its monopoly in specialized segments such as nonferrous metal smelting.
China’s monopoly over the rare-earth metals segment grants it unprecedented leverage on the global stage and allows the country to exert its influence with the threat of cutting rare-earth metal supplies. Japan has been at the receiving end of such a supply cut, as China recently announced that it would halt exports of two rare earth metals to protect its internal supply chains.
Consequently, the United States and other nations, along with Western automakers are working to decouple their supply chains from China amid increasing tension between Beijing and Washington. Tesla, for instance, has plans to expand its battery manufacturing capabilities and has partnered with Panasonic to increase EV battery production by 10% at Gigafactory Nevada.
The Biden administration is also providing incentives for electric vehicles with as many locally made raw materials as possible to make them more affordable for consumers. This will most likely incentivize battery manufacturers to pitch tent in the U.S. market while pushing EV makers into partnerships with domestic battery makers.
Tesla Inc. (TSLA), closed Thursday's trading session at $219.22, off by 2.828%, on 120,718,417 volume. The average volume for the last 3 months is 34,697 and the stock's 52-week low/high is $101.81/$313.80.
The QualityStocks Company Corner
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
- Renovaro BioSciences Inc. (NASDAQ: RENB)
- Knightscope Inc. (NASDAQ: KSCP)
- IGC Pharma Inc. (NYSE American: IGC)
- Advanced Container Technologies Inc. (OTC: ACTX)
- GolfLync Inc.
- Mullen Automotive Inc. (NASDAQ: MULN)
- SuperCom Ltd. (NASDAQ: SPCB)
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF)
- Data443 Risk Mitigation Inc. (OTC: ATDS)
- FingerMotion Inc. (NASDAQ: FNGR)
- InMed Pharmaceuticals Inc. (NASDAQ: INM)
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
Uppsala University researchers have created a novel method for detecting brain-cancer mutations in children, granting scientists an unprecedented glimpse into the inner workings of pediatric brain tumors. The novel technique allowed the researchers to determine that the mutations they saw changed how tumor cells interact with cancer drugs, potentially opening the door for the development of better means of diagnosing pediatric brain cancer and building personalized treatment plans. Medulloblastoma, a type of brain tumor that originates in the cerebellum, is the most common type of brain cancer in children. It has a 70% survival rate after five years if treatment begins before the cancer has spread to the spinal column and a 60% success rate if the tumor has spread. Current medulloblastoma treatment protocols involve surgery to remove as much of the tumor as possible while obtaining a tumor sample to inform further treatment plans (chemotherapy, radiation and clinical trials). In particularly aggressive cases, survivors often have to deal with adverse side effects of treatment, such as diminished learning abilities and balance problems. As it becomes easier to detect and track the mutations that happen in different cancers, drug manufacturers such as CNS Pharmaceuticals Inc. (NASDAQ: CNSP) may find it easier to develop formulations that are better equipped to keep in check or even reverse the progress of some of the hardest cancers to treat.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Thursday's trading session at $1.88, up 3.2967%, on 35,120 volume. The average volume for the last 3 months is 502,510 and the stock's 52-week low/high is $0.6105/$8.64.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Researchers Develop Novel Way to Detect Pediatric Brain-Cancer Mutations
- InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Announces Q2 2023 Results, Presents Updated Findings from Potentially Pivotal Study of Berubicin
- Israeli Company Seeks to Develop Micro-Robots to Boost Precision Medicine
Renovaro BioSciences Inc. (NASDAQ: RENB)
The QualityStocks Daily Newsletter would like to spotlight Renovaro BioSciences Inc. (NASDAQ: RENB) .
Renovaro BioSciences (NASDAQ: RENB), (formerly NASDAQ: ENOB), in March of 2023 announced that Dr. Anahid Jewett, a renowned cancer researcher in the field of immunotherapy for cancer, presented proof-of-concept data from two sets of humanized mouse studies conducted independently by her laboratory at the University of California, Los Angeles ("UCLA"). According to the announcement, the experiments demonstrated highly significant reductions in the weight and volume of pancreatic tumors following therapy with the company's proprietary technology combining cell-, gene- and immunotherapy. The results correlated with strong immune responses, indicating proof-of-concept of the way the technology was intended to act. "I have been working in this field for three decades and designed the mouse model that we have used in my laboratory to study the company's potential cancer therapies. The results are some of the most impressive I have ever seen. The fact that we saw very similar highly statistically significant results across two independently conducted studies is particularly encouraging, indicating reproducibility of the findings. I look forward to continuing our work together towards clinical trials, and potentially offering hope to many who are suffering with diseases with few good treatment options," Dr. Jewett said.
To view the full press release, visit https://ibn.fm/QVpTy
Renovaro BioSciences Inc. (NASDAQ: RENB), formerly Enochian BioSciences Inc., is an advanced, pre-clinical biotechnology firm in cell, gene and immunotherapy focused on solid tumors with short life expectancy. The company aims to unlock potentially long-term or life-long cancer remission in some of the deadliest cancers, and to potentially treat or cure serious infectious diseases such as Human Immunodeficiency Virus (HIV) and Hepatitis B Virus (HBV) infection.
The oncology platform is now at the forefront of Renovaro’s development activities. While Renovaro’s current efforts focus primarily on pancreatic cancer, it plans to include other solid tumors with short life-expectancy in the first in human Phase I/IIa studies that are on track to start by mid-2024. The company’s Pre-Investigational New Drug (pre-IND) submission included a human study plan covering pancreatic cancer, as well as other cancers that are difficult to treat, potentially including triple-negative breast cancer, head and neck cancers and mesothelioma.
Renovaro’s proprietary, novel technology uses cell- and gene-therapy to promote a renewed immune response against solid tumors. Important confirmatory results from two humanized mouse models using the company’s novel dendritic cell-based therapy, independently conducted by Dr. Anahid Jewett, a renowned cancer researcher in the field of immunotherapy at UCLA, were presented previously at two scientific conferences and were the foundation supporting a pre-IND submission to the U.S. Food and Drug Administration. Notably, Dr. Jewett’s findings from these studies consistently demonstrated 80% to 90% pancreatic tumor reduction in size and weight that was correlated with significant enhancement of key aspects of the immune response.
Renovaro is headquartered in Los Angeles, California.
RENB-DC11
Renovaro’s product development strategy is anchored in the use of “non-self” or allogeneic cells that enhance targeted immune response. Its lead candidate, RENB-DC11, is an innovative therapeutic vaccination platform that could potentially be used to induce life-long remissions from some of the deadliest solid tumors.
Treatment with RENB-DC11 has now been shown to significantly reduce the size of human pancreatic tumors in humanized mice in three independent studies. The reduction in tumor size correlated with statistically significant increases in key components of an immune response.
Pre-IND was completed in June 2023, with IND filing forecast for first half of 2024. First in-human Phase I/IIa trials are predicted shortly after in H1 of 2024, including pancreatic and other solid tumors with poor treatment options and life-expectancy.
Renovaro believes that RENB-DC11 could represent the most promising and effective strategy to achieve life-long remission for a number of common and deadly tumors.
Other Development Candidates
In addition to its lead oncology platform, Renovaro’s development pipeline includes a platform targeting infectious diseases, including:
- RENB-HV12 – An engineered allogeneic T-Cell vaccine, this therapeutic HIV vaccine candidate enhances immune infiltration, immune killing and immune surveillance. Potential pre-IND submission is planned for first half of 2024, with IND-submission expected in second half of 2024.
- RENB-HV21 – Leveraging allogeneic NK plus Gamma Delta T (GDT) cells as potential therapy for HIV, ENOB-HV21 shows promising preliminary results without confounding factors. Renovaro owns an exclusive license and has completed the Pre-IND submission, with a potential IND submission and human trials expected in 2024.
- RENB-HV01 – Caring Cross, a non-profit corporation, has shown that its proprietary CAR-T cells cure HIV in a mouse model. Studies in humans have begun. Renovaro has entered into a profit-sharing sublicense with Caring Cross and would share in profits if the product is commercialized.
- RENB-HB01 – This therapeutic approach aims to eliminate all HBV rapidly (“seek and kill”) with a two to three dose treatment regimen. It is expected to be applicable for early disease to maximize impact with low risk of toxicity. Pre-IND comments have been received from the FDA for its AAV-delivery system.
LOI to Merge with GEDi Cube International Ltd.
On August 9, 2023, Renovaro announced its execution of a binding, exclusive letter of intent to merge a subsidiary with cutting-edge health AI company GEDi Cube International Ltd. The combined company is expected to create a potential multiplier effect to accelerate earlier diagnosis, more effective therapy, and precision in silico drug discovery.
GEDi Cube’s innovative technology, developed over nearly a decade, has already validated earlier diagnoses of lung cancer in humans at a leading university hospital. GEDi Cube has likewise created the early diagnosis technology for 12 additional cancers, including pancreatic and breast cancer.
“I believe joining forces with GEDi Cube could enhance the efficacy of our upcoming trials and speed up the discovery of novel treatment approaches, thereby extending our life-saving technology to more cancer patients and renewing hope for them and their families,” Dr. Mark Dybul, CEO of Renovaro, stated in the news release.
GEDi Cube is led by CEO Craig Rhodes, who brings to that company tremendous industry experience leading life sciences groups at industry leaders Intel, Oracle and NVIDIA.
Market Opportunity
Pancreatic cancer alone is diagnosed globally in approximately 495,000 people each year, including roughly 64,000 in the U.S. Nearly 466,000 of those patients die annually, including approximately 51,000 in the U.S. Because of limited treatment options, life expectancy is very poor – with an approximately 10% patient survival rate at five years after diagnosis.
The global pancreatic cancer treatment market was valued at $2.15 billion in 2021 and is projected to grow from $2.48 billion in 2022 to $6.85 billion by 2029, according to Fortune Business Insights. That growth represents a CAGR of 15.7% for the forecast period.
A separate report from Fortune Business Insights projects that the global HIV drug market will grow from $30.46 billion in 2021 to $45.58 billion in 2028, recording a CAGR of 5.9% over the forecast period.
According to GlobalData, the value of the market for hepatitis B treatment is forecast to experience a significant increase in the coming years, with revenues expected to grow from $1.6 billion in 2022 to $10.5 billion in 2029. That represents a very rapid CAGR of 30% over the period. An estimated 296 million people suffer from the condition worldwide.
Management Team
Dr. Mark Dybul is the CEO of Renovaro. He has served as a tenured professor in the Department of Medicine at Georgetown University Medical Center since June 2017. He also served as Faculty Co-Director of the Center for Global Health and Quality from 2017-2021. Dr. Dybul has worked on HIV and public health for nearly 30 years as a clinician, scientist, teacher and administrator, including as an architect and eventually the Global Ambassador of the U.S. President’s Emergency Plan for AIDS Relief and the Executive Director of the Global Fund to Fight AIDS, Tuberculosis and Malaria from 2013 through May of 2017, and as the co-director of the Global Health Law Program at the O’Neill Institute for National and Global Health Law from 2009 through 2012. He is a member of the U.S. National Academy of Medicine.
Luisa Puche is the company’s CFO. She has served as a senior accounting and financial advisor and president of Puche Group LLC from 2015-2019. She served in various key executive roles, including Interim Chief Accounting Officer, at Brightstar Corp., a $10 billion global wireless device services provider. Ms. Puche began her career at Ernst & Young, where she served for approximately 10 years. Leveraging her broad global audit, advisory and corporate expertise, she has provided strong cross-functional leadership experience managing small and large projects for both publicly traded and privately held companies in various industries, including a global implementation of the latest revenue recognition accounting standard for Del Monte, as well as the global implementation of their SOX-404 program.
Francois Binette, Ph.D., is the Chief Operating Officer and Executive Vice President of Research & Development at Renovaro. He has over 25 years of product development expertise in Advanced Therapies and Regenerative Medicine. His broad industry experience spans a wide range of serious medical conditions, from orthopedics to ophthalmology, CNS and immuno-oncology. His career includes positions at Genzyme, Biosyntech, the DePuy Franchise of Johnson and Johnson, Medtronic and Lineage Cell Therapeutics. He received his Ph.D. from Laval University in Québec, followed by post-doctoral training at the Sanford-Burnham Institute in La Jolla and Harvard Medical School in Boston.
Renovaro BioSciences Inc. (NASDAQ: RENB), closed Thursday's trading session at $1.62, up 4.5161%, on 503,454 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $8.64/$.
Recent News
- Renovaro BioSciences Inc. (NASDAQ: RENB) - InvestorNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB) Announces Presentation of 'Impressive' Results from Proof-of-Concept Studies
- InvestorNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB) Eyes Bright Future in Advancing Potentially Curative Therapies
- InvestorNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB) Committed to Rapid Advancement of Its Proprietary Oncology Platform
Knightscope, Inc. (NASDAQ: KSCP)
The QualityStocks Daily Newsletter would like to spotlight Knightscope, Inc. (NASDAQ: KSCP).
Knightscope, Inc. [Nasdaq: KSCP] ("Knightscope" or the "Company"), a leading developer of autonomous security robots and blue light emergency communication systems, today announced a new contract with a self storage company for an initial order of five K5 Autonomous Security Robots ("ASRs"). The client is one of the fastest-growing self-storage platforms in the United States with assets located primarily in high-density, urban markets. Knightscope's K5 ASRs will patrol five storage locations – two in the Portland area and three others in California – to help mitigate risk and elevate the protection offered to tenants and their belongings. In addition to their security duties, the robots will broadcast helpful messages to passersby and act as brand ambassadors. Based on the success of these initial deployments, the client will be considering deployments at many other store locations.
Knightscope, Inc. (NASDAQ: KSCP), founded in 2013 and based in Mountain View, California, is a leader in the development of autonomous security capabilities targeting to disrupt the $500 billion security industry. Knightscope’s technology uniquely combines self-driving technology, robotics, artificial intelligence and electric vehicles.
Knightscope designs and builds Autonomous Security Robots (ASRs) that provide 24/7/365 security to the places you live, work, visit and study. The company’s client list covers public institutions and commercial business operations, including multiple Fortune 1000 companies to date. These ASRs have been proven to enhance safety at hospitals, logistics facilities, manufacturing plants, schools and corporations. ASRs act as highly cost-effective complementary systems to traditional security and law enforcement officials, providing an additional advantage by continuing to offer uninterrupted patrolling capabilities across the country.
The company’s ASRs have assisted in the arrest of suspects involved in crimes ranging from armed robbery to hit-and-runs. Their machine-embedded thermal scanning capability even aided in preventing the breakout of a major fire. You can learn more about the crime fighting wins at www.knightscope.com/crime
The company has achieved several milestones since its creation in 2013, including:
- Establishing itself in a 15,000-square-foot facility located in Mountain View, California, in the heart of Silicon Valley, where Knightscope designs, engineers and builds its technology (Made in the USA)
- Operating for more than 1 million hours in the field and securing contracts across five time zones, from Hawaii to Rhode Island
- Raising over $100 million since inception to build its technology from scratch and generating over $13 million in lifetime revenue, validating both the market opportunity and the technology
Growth Capital & Proposed Nasdaq Listing
With backing from more than 28,000 investors and four major corporations and over $100 million raised since inception, Knightscope is poised to be an industry leader in the future of public safety and security.
On December 1, 2021, Knightscope announced the commencement of an offering of up to $40 million of its Class A common stock, with shares to be listed immediately following closing on the Nasdaq Global Market under the ticker symbol ‘KSCP’. The offering is for up to 4 million shares priced at $10 per share. Learn more at www.knightscope.com/investors
Company Mission – Reimagining Public Safety
Knightscope’s long-term vision has an eye on the greater good. The company’s mission is to make the United States of America the safest nation in the world while supporting the 2+ million law enforcement and security professionals across the country.
Crime has an estimated negative economic impact in excess of $2 trillion annually. As crime is reduced, positive impacts will likely be realized across several aspects of society, including housing, financial markets, insurance, municipal budgets, local business and safety in general.
Knightscope CEO William Santana Li was interviewed by Kevin O’Leary, more commonly known as Shark Tank’s Mr. Wonderful. When asked to explain how the benefits provided by the ASRs outrank a human doing the same job, Li said, “First, just the simple presence of a physical deterrent causes criminal behavior to change. Second, the machines are self-driving cars that patrol all around and recharge themselves. They also generate 90 terabytes of data per year. No human would ever be able to process that. The robots are intended to be eyes and ears for the humans, not a one-to-one replacement.”
The Knightscope solution to reduce crime combines the physical presence of ASRs, sometimes referred to as proprietary Autonomous Data Machines, with real-time onsite data collection and analysis. The ASRs are fitted with eye-level 360° cameras, thermal scanning, public address announcements and various other features that work in tandem with humans to provide law enforcement officers and security guards unprecedented situational awareness.

Those 90 terabytes of data are then formatted in a useable way, so law enforcement can leverage that information and execute their responsibilities more effectively.
Public Safety Innovation
The company’s recurring revenue business model is set up to mimic the recurring societal problem of crime, and it takes into consideration the fact that innovation in the security and public safety industry has been stagnant for decades. Because the traditional practices of the sector have remained unchanged for years, automation has potential to drive substantial cost savings – and significant improvement in capabilities.
Human security guards are one of both the largest expenses and the largest liabilities for companies. Knightscope’s robots are offered at an effective price of $3 to $9 per hour, compared with approximately $85 for an armed off-duty law enforcement officer and $15 to $35 for an unarmed security guard.
This innovation has the potential to drive considerable cost savings. Based on these estimates, manufacturing costs can be recovered as soon as the first year of operation.
Product Offerings
The company has nine patents and a framework of unique intellectual property. Knightscope currently offers a K1 stationary machine, a K3 indoor machine and a K5 outdoor machine. A K7 multi-terrain four-wheel version is in development.

The ASRs autonomously patrol client sites without the need for remote control, providing a visible, force multiplying, physical security presence to help protect assets, monitor changes in the area and deter crime. The data is accessible through the Knightscope Security Operations Center (KSOC), an intuitive, browser-based interface that enables security professionals to review events generated by the ASRs providing effectively ‘mobile smart eyes and ears’. Learn more at www.knightscope.com/ksoc
The ASRs and the related technologies were developed ground up by the company and are Made in the USA.
The Robot Roadshow

Knightscope has created the ultimate hybrid physical and virtual event, bringing its Autonomous Security Robot technologies to cities across the country for interactive and in-person demonstrations.
Each roadshow landing is hosted virtually by a Knightscope expert, and visitors can interact directly with each of the company’s ASRs and see the Knightscope Security Operations Center (KSOC) user interface in action. Learn more at www.knightscope.com/roadshow
Management Team
Chief Executive Officer William Santana Li is a veteran entrepreneur, a former executive at Ford Motor Company and the founder of GreenLeaf, a company that grew to be the world’s second-largest automotive recycler and is now part of LKQ Corporation (NASDAQ: LKQ).
Chief Client Officer Stacy Dean Stephens brings his experience as a former Dallas law enforcement officer, as well as his skills as a seasoned entrepreneur, to assist on the client acquisition side.
Chief Intelligence Officer Mercedes Soria is an award-winning technologist and former Deloitte software engineer.
Chief Design Officer Aaron Lehnhardt brings over two decades of two- and three-dimensional product and industrial design in modeling and VR to the table, on top of his experience as a senior designer at Ford Motor Company.
Chief Financial Officer Mallorie Burke is a seasoned financial executive and strategic advisor for both private and publicly traded technology companies with a successful track record of mergers & acquisitions, corporate growth and exit strategies, including public listings.
General Counsel Peter Weinberg leverages 30 years of diverse corporate counsel experience, spanning from startups to well-established companies, private and public. He has significant experience training personnel at all levels in critical areas to improve corporate compliance and productivity.
Knightscope, Inc. (NASDAQ: KSCP), closed Thursday's trading session at $1.29, up 8.4034%, on 3,071,374 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.36/$3.65.
Recent News
- Knightscope (NASDAQ: KSCP) - Knightscope Inks Deal with Major Player in Self Storage
- InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Provides Q2 Update Video, Announces New ASR Deployment, Adds Roadshow Date to Calendar
- InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Continues Sales Momentum with Three New Contracts, Takes Roadshow to Pittsburg
IGC Pharma Inc. (NYSE American: IGC)
The QualityStocks Daily Newsletter would like to spotlight IGC Pharma Inc. (NYSE American: IGC).
A recent examination of survey data from Gallup shows that Republican backing for cannabis legalization has reached an unprecedented peak on a nationwide scale. However, a deepening partisan schism has emerged over the last couple of decades, with Democrats having swiftly and more openly embraced this cause. Most Democrats and Independents leaning toward the Democratic Party (amounting to 83%) and also Republicans and Independents with an inclination toward the GOP (accounting for 55%) are in favor of dismantling the ban on cannabis.
These insights, extracted from Gallup's 2022 survey data, illustrate a disparity of 28% in opinion on the matter of cannabis reform between these political factions. The divergence is now broader compared to the scenario two decades ago when backing from both sides was less than 50%.
According to the most recent research, Democrats have demonstrated a more positive attitude toward marijuana legalization than Republicans since 2003. The data continues by revealing that between 2013 and 2022, there was a synchronized increase in the number of each political party's supporters of legalizing marijuana. However, Democratic group growth has exceeded Republican faction growth. This has caused the political gap on this matter to be slightly more apparent in the most recent measuring year compared to two decades ago. This lag between public sentiment and actions in Congress suggest that it may take a while for federal marijuana policy to be overhauled. Despite this gloomy possibility, companies such as IGC Pharma Inc. (NYSE American: IGC) are on course to thrive as sellers of federally approved cannabis-based treatments if their R&D programs yield successful outcomes.
IGC Pharma Inc. (NYSE American: IGC), through subsidiary IGC Pharma, develops, patents, and markets advanced THC-based drug formulations for the treatment of symptoms related to various diseases including but not limited to Alzheimer’s disease, Tourette syndrome, chronic pain, and pet seizures.
IGC’s leading drug candidate, IGC-AD1, has completed Phase 1 of a safety and tolerability trial and entered Phase 2 trials for treating agitation in patients with Alzheimer’s dementia, the first study in humans of a natural tetrahydrocannabinol (THC) compound plus another molecule (www.clinicaltrials.gov). As of September 2022, the IGC trial is the only ongoing Phase 2 trial of a natural THC-based formulation on Alzheimer’s patients.
The company’s other drug candidate, TGR-63, is an enzyme inhibitor that has shown in preclinical trials the potential to reduce neurotoxicity in Alzheimer’s cell lines. Both drug candidates have shown their ability to ameliorate beta amyloid plaques in Alzheimer’s cell lines and improve memory in Alzheimer’s mouse models. Beta amyloid plaques are a key hallmark of Alzheimer’s and an important target of Alzheimer’s pharmaceutical drug development.
Neuro Psychiatric Symptoms (NPS) are not only debilitating for Alzheimer’s patients; they also place an immense emotional burden on their caregivers. Beyond reducing symptoms, IGC-AD1’s active molecules and TGR-63 have also shown promise in preclinical trials to reduce important hallmarks of Alzheimer’s including plaques and tangles, as well as improving the treatment of memory loss.
Over the past eight years, the IGC team has amassed a deep knowledge of cannabinoid science, including extraction, isolation, purification, and development. The company’s strategy is to leverage its unique end-to-end capabilities, platform, and expertise to develop a class-leading program and bring it to market quickly and cost efficiently to treat neurodegenerative diseases such as Alzheimer’s.
The company also has a family of cannabidiol (CBD)-based consumer products (www.Holief.com) such as pain relief creams, pain relief gels, purpose gummies, tinctures, and capsules targeting women’s wellness, with a particular focus on premenstrual syndrome (PMS) and dysmenorrhea (period cramps). In addition, the company targets individuals that need sleep-aids with its specially formulated low melatonin cannabinoid gummies.
IGC has also introduced a low-calorie CBD- and caffeine-infused energy beverage brand (www.SundaySeltzer.com) that is currently available for purchase. The company’s brands are founded on the belief that effective natural solutions should be affordable and accessible to everyone. As the demand for natural products targeting women’s wellness and energy drinks continue to grow, these products are seeing strong traction in the market.
The company operates three facilities – a large GMP (Good Manufacturing Production Standards) certified facility that includes extraction, distillation, and manufacturing, in Washington State; a GMP-211 (pharmaceutical) grade facility in Maryland; and a facility licensed for controlled substances including cannabis in Bogota, Colombia, with complete access to legal licensed cannabis where the company conducts its testing.
In addition, the company’s development under Magistral Formulations is approved by INVIMA (Colombia National Food and Drug Surveillance Institute) to treat neurological disorders, non-oncological chronic pain, and mental disorders.
IGC’s intellectual property (IP) portfolio comprises of eight patents that it controls and seven patent applications. The portfolio includes #11,446,276, a patent for extreme low dose THC treatment of Alzheimer’s that was granted in September 2022.
The company is headquartered in Potomac, Maryland.
IGC-AD1
IGC-AD1 is the company’s leading drug candidate for the treatment and relief of Alzheimer’s symptoms. A significant amount of research on Alzheimer’s cell lines has shown that the active agents in IGC-AD1 reduce plaques and neurofibrillary tangles that are the hallmarks of Alzheimer’s. Further, micro-dosing of THC, as shown in cell lines, could increase the functioning of mitochondria and potentially promote the growth of new neural pathways (neurogenesis). The research shows that micro-dosing of THC affects the brain radically differently from the normal higher dosing of THC.
While there is a significant body of research showing that THC is neuro-toxic at normal levels of dosing, micro-dosing of THC has been shown to be non-toxic to neurons. With the results of these preclinical studies, the company developed an oral formulation, IGC-AD1. The company recently completed a safety and tolerability Phase 1 trial on Alzheimer’s patients and has initiated a Phase 2, multi-site, double-blind, randomized, placebo-controlled trial of the safety and efficacy of IGC-AD1 on agitation in participants with dementia due to Alzheimer’s disease at sites in the U.S. and Canada. IGC expects the Phase 2 trial to take between 9 and 12 months to complete, barring unknown factors such as, for example, a resurgence of COVID and the enforcement of lockdowns and travel restrictions.
With further successful trials and FDA approvals, IGC hopes to bring a drug based on natural THC as an effective treatment for agitation in Alzheimer’s to market.
TGR-63
The company’s other molecule, TGR-63, has been shown to reduce the neurotoxicity that impacts memory loss in preclinical trials with mice. On a dose dependent manner, transgenic Alzheimer’s mice treated with TGR-63 showed improvement in memory relative to control.
Both drug candidates, IGC-AD1 and TGR-63, have shown their ability to reduce the brain plaques associated with memory loss in Alzheimer’s in mice.
With further successful trials and FDA approvals, IGC hopes to bring TGR-63 as a treatment for Alzheimer’s disease to market.
Market Opportunity
Alzheimer’s disease impacts over 55 million people worldwide and about 5.5 million individuals in the U.S. Over 70% of these patients face debilitating symptoms, including anxiety, depression, and agitation (Mendez, 2021). Agitation in dementia patients can include excessive physical movement and verbal activity, restlessness, pacing, belligerence, aggression, screaming, crying, and wandering.
In 2020, the estimated healthcare costs for Alzheimer’s disease in the U.S. were $305 billion. Medicare and Medicaid covered about 70% of those costs, leaving considerable burden on patients and families. At the current rate of growth of Alzheimer’s and other dementia diagnoses, those costs are estimated to reach over $1 trillion by 2050.
Currently, there are no FDA-approved medications to alleviate the symptoms of dementia due to Alzheimer’s disease, providing a tremendous opportunity for formulations that can have an impact on quality of life and disease progression.
Management Team
Richard Prins has been chairman at IGC since 2012 and served as an independent director since 2007. From March 1996 to 2008, he was the Director of Investment Banking at Ferris, Baker Watts, Incorporated. Prins served in a consulting role to RBC until January 2009. He currently volunteers full time with a non-profit organization, Advancing Native Missions, and is a private investor. Since February 2003, he has been on the board of Amphastar Pharmaceuticals Inc. He holds a bachelor’s degree from Colgate University and an MBA from Oral Roberts University.
Ram Mukunda is CEO and President of IGC. He has been the chief inventor and architect of most of the company’s patent filings and is responsible for the company’s strategic positioning. Prior to IGC, he was founder and CEO of Startec Global Communications, which he took public in 1997. He served as Strategic Planning Advisor at Intelsat, a communications satellite services provider. From 2001 to 2003, he was a Council Member at Harvard’s Kennedy School of Government, Belfer Center of Science and International Affairs. He was named the 1998 Ernst & Young Entrepreneur of the Year. He holds bachelor’s degrees in electrical engineering and mathematics, and a master’s degree in engineering from the University of Maryland.
Dr. Jagadeesh Rao is the company’s Principal Scientist. His career spans two decades in the public sector and product R&D for Johnson & Johnson. He leads IGC’s scientists in the development of pharmaceutical and OTC products. He worked for the federal National Institutes of Health, and for the National Institute on Drug Abuse. His Ph.D. in Neurochemistry is from the National Institute of Mental Health & Neurosciences in India. He did postdoctoral training at the University of Illinois-Chicago.
Claudia Grimaldi is a Director, Vice President, Principal Financial Officer, and Chief Compliance Officer for IGC. She also serves as a Director/Manager Director for some of the company’s subsidiaries. She graduated with highest honors from Javeriana University in Colombia with a bachelor’s degree in psychology. She holds an MBA, graduating with highest honors, from Meredith College in North Carolina. In addition, she has attended the Darden School of Business Financial Management Executives program and the Corporate Governance Program at Columbia Business School. She is currently pursuing her Directorship Certification with the National Association of Corporate Directors. She is fluent in both English and Spanish.
IGC Pharma Inc. (NYSE American: IGC), closed Thursday's trading session at $0.34, even for the day, on 67,472 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.2785/$0.74.
Recent News
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Gallup Poll Finds Highest Ever Support for Cannabis Legalization Among Republicans
- 420 with CNW - Cannabis Industry Lobbying Increases as Looming Government Shutdown Leaves SAFE Banking in Balance
- 420 with CNW - CNN's Dr. Gupta Discusses Marijuana Use by Seniors
Advanced Container Technologies Inc. (OTC: ACTX)
The QualityStocks Daily Newsletter would like to spotlight Advanced Container Technologies Inc. (OTC: ACTX).
Cannabis retailers in the northeast sold a record number of products in July, achieving new sales records and generating millions of dollars. State data shows that cannabis sales in Rhode Island, Maine and Massachusetts are booming, with Maine selling $20.8 million worth of cannabis products in July alone and breaking cannabis sales records for the third month in a row. Marijuana businesses in Maine saw more than $118 million in cannabis sales over the first half of 2022 and are on track to end the year at a record $159 million in sales. The Cannabis Control Commission reported that recreational cannabis sales in Massachusetts reached $136 million in March while medical marijuana purchases were at $18.7 million, adding up to a total of $154.7 million in cannabis sales. July was the third consecutive record-setting month of cannabis sales in Massachusetts and surpassed the $152.9 million worth of cannabis sold in June. Rhode Island, on the other hand, set a sales record at $6.8 million worth of recreational cannabis products in July. The state has consistently set recreational marijuana sales records since launching the adult-use market last December. Consumers in Rhode Island also spent $2.7 million on medical cannabis, bringing the state's total cannabis sales to a record $9.5 million. These glowing sales figures show that many more companies, such as Advanced Container Technologies Inc. (OTC: ACTX), are benefiting from the boom in cannabis sales since there is an entire ecosystem of interconnected industries making it possible for marijuana companies to serve their customers.
Advanced Container Technologies Inc. (OTC: ACTX) is in the business of selling and distributing self-contained, automated, indoor “micro-farms” called Grow Pods, along with related equipment and supplies. Additionally, the company designs and sells patented proprietary medical-grade plastic containers, known as the Medtainer®, that store and grind pharmaceuticals, herbs, teas and other solids or liquids.
ACTX is the leading distributor of Grow Pods. With a controlled environment, food and herbs can be grown without pesticides, harmful chemicals or risk of pathogen contamination, and with low energy consumption. Restaurants, grocery stores, non-profits, MSOs and entrepreneurs can use Grow Pods to ensure a fresh supply of ultra-clean produce year-round.
The company entered the Grow Pod business in October 2020 with its acquisition of all shares of Advanced Container Technologies Inc., a California corporation. As of February 28, 2022, ACTX is exploring the acquisition of the assets and the assumption of some or all of the liabilities of GP Solutions Inc., the developer and manufacturer of Grow Pods, for which ACTX is currently the sole U.S. distributor.
Because Grow Pods can be located almost anywhere, produce can be grown closer to the point of consumption and harvested at its peak, providing nutritious fruits and vegetables where needed. Indoor micro-farms, utilizing a practice known as vertical farming, have attracted the attention of governments and universities, which are now promoting vertical farming as a way to combat food insecurity and inequities.
The United States Department of Agriculture (USDA) has stated that vertical farming “is no longer a futuristic concept.” The department is enthusiastic about vertical farming, particularly those utilizing repurposed shipping containers, such as Grow Pods. Arizona State University reports that vertical farming reduces water use by 90 percent compared to conventional farming but produces 10 times the crop yield.
Products
Grow Pods
One of the company’s main business units is focused on selling advanced, self-contained hydroponic containers called Grow Pods. These unique and innovative automated systems are essentially micro-farms that can be placed virtually anywhere and, with their controlled and specially filtered environment, allow cultivation of a wide variety of crops, 365 days a year. The Grow Pod controlled environment offers major advantages for the production of high-value crops. The ability to grow year-round and the ability to cultivate in a smaller footprint using less water and power are some of the primary advantages of the system. Grow Pods offer constant temperature, humidity and airflow control, as well as automated watering and lighting schedules for optimal growth and minimal labor requirements, regardless of crop.
Containers
ACTX meets the needs of the pharmaceutical and medical markets, including the cannabis and hemp industries, with patented packaging systems. The company designs, customizes, brands and sells proprietary medical grade plastic containers that can store pharmaceuticals, herbs, teas and other solids or liquids, with a special built-in feature that can grind solids and shred herbs. The company’s flagship container product is the patented Medtainer®, a child resistant, medical-grade herb container and grinder that is water-tight, air-tight and smell proof. Packaging in the cannabis industry is critical, with numerous stringent regulations about how cannabis products must be packaged and labeled. ACTX also offers custom-branded, compliant vacuum seal bags and other retail container solutions.
Equipment and Supplies
ACTX markets and sells two principal products: Grow Pods, which are specially modified insulated shipping containers manufactured by GP Solutions Inc., in which plants, herbs and spices may be grown hydroponically in a controlled environment, and Medtainers®, which may be used to store pharmaceuticals, herbs, teas and other solids or liquids and can grind solids and shred herbs. The company also markets and sells various products related to Grow Pods and the Medtainer®, as well as providing private labeling and branding services for purchasers of Medtainers® and certain related products.
GP Solutions manufactures and sells other products, such as humidity controllers and LED lighting systems for vertical farming. The company’s specially designed lighting panels are programmed to emit the exact wavelength of light that each crop requires. The system has a daybreak-to-nightfall feature that gives plants the proper chromatic signals to grow rapidly and fruitfully. High efficiency LED light strips supply the crops with a red and blue light spectrum required for photosynthesis in the spectrum that plants need most.
Market Overview
The global vertical farming market is expected to reach $33.02 billion by 2030, according to a new report by Grand View Research. The market is forecast to expand at a CAGR of 25.5 percent from 2022 to 2030, according to Grand View. Escalating production of biopharmaceutical products, including cannabis, is anticipated to drive the market. The building-based segment of the market is expected to register a significant CAGR of 27.8 percent over the projected period. In addition, the climate control segment is expected to see high growth.
The global cannabis packaging market is expected to reach $14.34 billion by 2028, according to analysis by Reports and Data. The analysis forecasts 1,700 percent growth in cannabis users by the end of 2026, with packaging likely observing a whopping 26.42 percent growth in the forecast period. There are significant barriers to entry in the cannabis packaging market, giving an advantage to companies already established in the sector. These barriers include developing a thorough knowledge of the myriad regulations that govern cannabis packaging (which differ in each state), and child-resistance requirements.
Management Team
Douglas P. Heldoorn is the Founder and Chairman of Advanced Container Technologies Inc. He also holds the positions of President, CEO and COO at the company. Mr. Heldoorn has served on the Board of Directors since its inception in 2013. He has also previously held the position of Executive General Manager at Nissan Motor Corp.
Jeffory A. Carlson is CFO and Treasurer of ACTX. Mr. Carlson has also served as the company’s Corporate Controller since 2014.
Advanced Container Technologies Inc. (OTC: ACTX), closed Thursday's trading session at $0.21, even for the day. The average volume for the last 3 months is and the stock's 52-week low/high is $0.0141/$0.65.
Recent News
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Three States in the Northeast Set July Cannabis Sales Records
- 420 with CNW - Court Order Halts NY Marijuana Dispensary Openings
- 420 with CNW - Minnesota Marijuana Drinks Makers Enjoying Great Market Conditions
GolfLync Inc.
The QualityStocks Daily Newsletter would like to spotlight GolfLync Inc.
GolfLync, the premier social media platform for golf enthusiasts, is proud to announce a significant milestone, surpassing 50,000 members across the United States. The exponential growth is a testament to the platform's ability to bring golfers together, fostering vibrant golfing communities and connections that continue to flourish.
From local golf clubs to nationwide networks, GolfLync's Virtual Golf Clubs(TM) ("VGC") have transformed the way golfers connect, share, and experience the game they love. Members are embracing the platform's innovative features, with private and public VGCs serving as hubs for planning tee times, sharing insights, and building lasting golfing friendships.
GolfLync Inc. matches golfers looking for a game through the company’s smartphone app, GolfLync. The company bills GolfLync as “the social network for golfers,” matching golf games and players similar to the way a dating app matches those looking for romance.
The app allows like-minded golfers to connect for a game simply by logging in. GolfLync helps golfers who are looking to grow their golf network find other players with similar interests and on course preferences. Whether you have recently moved to a new area and are looking for new golfing buddies, travel frequently and would like to play a round of golf while on the road, or just want to meet new golfers in your area, GolfLync is your answer. Spouses who enjoy golfing together can find other golfing couples to tee it up with. For a regular group that finds itself unexpectedly down a player, GolfLync can help find that last-minute addition to complete the foursome.
The company is based in Scottsdale, Arizona.

GolfLync App
GolfLync was created for golfers of all skill levels and preferences to connect with compatible players of similar skill. Golfers can find a tee time through GolfLync, join existing tee times and create new leagues. The app allows golfers to meet fellow players before committing to spend four hours on the course with them. GolfLync allows users to find new golf friends based on their preferences, such as walking or riding a cart, listening to music, friendly wagering, imbibing a favorite beverage at the 19th Hole and more. GolfLync is available for both Android and iOS as a free download.
Market Opportunity
According to a report by Statista, a leading provider of market and consumer data, in 2022, the number of people participating in golf in the United States reached 25.6 million, with 15.5 million additional players participating in off-course activities like driving ranges. In 2020, over 502 million rounds of golf were played in the U.S. alone. The game, traditionally dominated by male players, is changing, with increased interest from women golfers driven by social media influencers around the game.
Lumen Sports puts the total number of golf courses in the U.S. at more than 16,700. According to Lumen, about 75% of those are public courses open to all golfers, with the rest considered private golf clubs that require a membership.

Management Team
Noah DiPasquale is a co-founder and CEO of GolfLync Inc., leading the marketing and operations of the platform. He is also the founder and CEO of Epic Golf Club, a premier national membership and private golf society which partners with hundreds of top tier private golf clubs allowing Epic members access to their courses and recently founded the Epic Foundation, a Scottsdale-based 501c3. He holds a B.S. in Business Administration, Management and Operations from the W.A. Franke College of Business at Northern Arizona University and an MBA in Marketing from the University of Phoenix.
Michael Quiel is a co-founder of GolfLync Inc. and the President of the organization. He leads the application development and research teams. Michael understands how to build successful companies. His deep knowledge of investment banking, finance and building successful business partnerships is unparalleled. He’s an expert at capital formation and growth hacking companies. He has raised over $250 million in capital and taken multiple companies public.
Recent News
- GolfLync Inc. - GolfLync Inc. Surpasses 50,000 Members Nationwide, Creating Thriving Golfing Communities
- GolfLync Inc. is Now Seen as one of the Hottest New Apps in Social Media
- InvestorNewsBreaks – GolfLync Inc. Announces Astounding 15x Membership Surge

Mullen Automotive Inc. (NASDAQ: MULN)
The QualityStocks Daily Newsletter would like to spotlight Mullen Automotive Inc. (MULN).
Mullen Automotive (NASDAQ: MULN), an emerging electric vehicle ("EV") manufacturer, today announced the start of the company's $25 million stock buyback program with the repurchase of 3.7 million shares of common stock for an aggregate of $3,626,000. The buyback program started on Aug. 16, 2023, and can continue through Dec. 31, 2023. Mullen's chairman and CEO David Michery also purchased 102,040 shares in the open market on Aug. 16, 2023, at a price of $0.9842 per share. The company is committed to taking all available measures to regain compliance with the NASDAQ minimum $1.00 bid price requirement. "We believe that our stock is undervalued," Michery was quoted in the press release. "The company has begun production of our Class 3 EV with deliveries pending to customers and a strong balance sheet allowing us to execute on our business plan."
To view the full press release, visit https://ibn.fm/W8AFp
Mullen Automotive Inc. (NASDAQ: MULN) is a Southern California-based automotive company that owns and partners with several synergistic businesses working toward the unified goal of creating clean and scalable energy solutions. Mullen has evolved over the past decade in sync with consumers and technology trends. Today, the company is working diligently to provide exciting EV options built entirely in the United States and made to fit perfectly into the American consumer’s life. Mullen strives to make EVs more accessible than ever by building an end-to-end ecosystem that takes care of all aspects of EV ownership.
Commencement of Trading on Nasdaq
On November 5, 2021, Mullen announced its commencement of trading on the Nasdaq Capital Market.
“Today is a monumental day for Mullen Automotive. I am especially proud of our team, investors and all who have believed in Mullen and taken us to this point as a publicly traded company on the Nasdaq Capital Market,” David Michery, CEO and Chairman of Mullen Automotive, stated in the news release. “Trading on Nasdaq now opens us up to new investors, both institutional and retail shareholders, and broadens our awareness and company profile, while increasing awareness of Mullen and our technology platform and opening new opportunities in EV and beyond. The road ahead has never been brighter for Mullen, and I am proud to lead us into the future.”
The milestone came in the wake of the company’s stock-for-stock merger with Net Element Inc.
The Mullen FIVE
The Mullen FIVE EV Crossover, debuting at the Los Angeles International Auto Show (LAIAS) on November 17, 2021, embodies Mullen’s Southern California roots with an inspired design focused on two complementary Golden State themes – California landscape and California urban.
The FIVE is built on an EV Crossover skateboard platform that offers multiple powertrain configurations and trim levels in a svelte design that is Strikingly Different™ and exciting to experience in person.
Prior to the start of LAIAS, the Mullen FIVE was selected as a finalist by the LA Auto Show for Top EV SUV in the ZEVA “People’s Choice” Awards.
LAIAS provides Mullen an opportunity to display multiple variants of the FIVE model while also showcasing its powertrain, battery and charging technology. The company intends to bring the FIVE to market in 2024, and reservations are currently open here.
Mullen’s development portfolio also includes EV Fleet Vans, which it intends to bring to market in Q2 2022, and the pure electric, high performance Mullen DragonFLY.
Expansion of Manufacturing Capacity
On November 2, 2021, Mullen announced plans to expand its facility in Robinsonville, Mississippi.
Mullen’s Advanced Manufacturing and Engineering Facility (AMEC) currently occupies 124,000 square feet of manufacturing space. The total available land on the property is over 100 acres, and Mullen is moving ahead with plans to build out another 1.2 million square feet of manufacturing space to support class 1 and class 2 EV cargo vans and the Mullen FIVE EV Crossover.
On the expanded site, Mullen plans to build a body shop, a fully automated paint shop and a general assembly shop.
EV Market Outlook
The global EV market was reported to consist of 3,269,671 units in 2019, a figure that is expected to grow at a CAGR of 21.1% through 2030 to a total of 26,951,318 units worldwide. This market’s monetary value was estimated at $162.34 billion in 2019 and is expected to grow at a CAGR of 22.6%, resulting in an approximate value of $802.81 billion by 2027. The primary driver for this exponential growth is a worldwide increase in vehicle emissions regulations.
Management Team
David Michery is the CEO and Founder of Mullen and has been leading the company and its divisions since inception in 2014. With over 25 years of executive management, marketing, distressed assets, and business restructuring experience, Mr. Michery brings a wealth of relevant knowledge and expertise to the Mullen brand. He has notably created 12 trademarks so far to develop the company brand and vision.
Mr. Michery is working toward a sustainable future accessible to all by creating a suite of clean-energy electric vehicles at varied price points. With entirely U.S.-based manufacturing and operations, he is also determined to have Mullen Technologies play a role in shaping a self-sustaining local economy by creating more jobs in America.
Mr. Michery manages risks and company expectations as a pathway to success and has personally overseen several businesses that totaled over $1 billion in transactions. His key strength is the ability to be fiscally responsible and lead teams to complete projects on time and within budget. As a seasoned professional in this space, Mr. Michery has demonstrated skill in building businesses from the ground up and into successful entities that subsequently sold for hundreds of millions of dollars.
Mullen Automotive Inc. (MULN), closed Thursday's trading session at $0.79, off by 19.3878%, on 84,344,639 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.7793/$196.0425.
Recent News
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Announces Commencement of Stock Buyback Program
- InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Unveils Ultra-High-Performance FIVE RS Crossover
- Hyundai Sets Sights on Indian EV Market with Five New Models
SuperCom Ltd. (NASDAQ: SPCB)
The QualityStocks Daily Newsletter would like to spotlight SuperCom Ltd. (NASDAQ: SPCB) .
SuperCom (NASDAQ: SPCB), a global provider of secured solutions for the e-Government, Internet of Things ("IoT") and cybersecurity sectors, has secured a new contract to provide alcohol monitoring technologies and services in California. The contract, which is for an initial three-year term and is valued at up to $3 million, was secured through SPCB's wholly owned subsidiary Leaders in Community Alternatives ("LCA"). Noting that the contract further solidifies the company's strong position in the electronic monitoring industry, SuperCom called the contract win another milestone for the company's expansion into the U.S. market. According to the announcement, the contract is already in effect, running through 2026 with potential for extensions and recurring revenue.
"Our reputation for providing reliable and high-quality electronic monitoring services was pivotal in securing the contract," said SuperCom president and CEO Ordan Trabelsi in the press release. "This contract marks another strong achievement, building on our track record of securing numerous new electronic monitoring contracts in the United States and Europe. We are proud to have been chosen once again to support California's criminal justice system. Our long-standing relationships with county customers and law enforcement underscores their trust in our technology and services. As we move forward, we remain committed to fostering strong partnerships, driving technological advancements, and delivering top-tier solutions that address the unique challenges faced by the criminal justice system. With a focus on technology innovation and global expansion, SuperCom is well-positioned to help lead the electronic monitoring industry into the future."
To view the full press release, visit https://ibn.fm/66gYJ
SuperCom Ltd. (NASDAQ: SPCB) provides secured solutions for the e-government, IoT and cybersecurity sectors. Since 1988, the company has been a trusted global provider of traditional and digital identity offerings, providing cutting-edge electronic and digital security solutions to governments and organizations, both private and public, around the world.
SuperCom’s mission is to revolutionize the public safety sector worldwide through proprietary electronic monitoring technology, data intelligence, and complementary services.
The company is headquartered in Tel Aviv, Israel, with offices in California and other regions in the U.S.
Business Units
IoT and Connectivity
SuperCom IoT products and solutions provide advanced electronic monitoring solutions and services to criminal justice agencies, enabling customers to detect unauthorized movement of people, vehicles, and other monitored objects. The company provides an all-in-one, field-proven PureSecurity offender monitoring suite, accompanied by services such as GPS monitoring, home detention, domestic violence prevention, and more. The company’s services are specifically tailored to meet each client’s needs.

SuperCom’s proprietary Puresecurity suite of hardware, connectivity, and software components is the foundation for its criminal justice services and offerings. SuperCom is leveraging its extensive technology expertise to implement groundbreaking artificial intelligence (AI) technologies into various parts of its core offerings. By leveraging the power of AI, SuperCom’s PureSecurity platform can offer new abilities, such as amplified data analysis, predictive modeling, and streamlined automation – all geared toward optimizing decision-making and operational efficiency.
Competitive advantages of SuperCom’s technology include:
- Long Battery Life (No Tag Charging Required)
- Ultra Lightweight Form Factor
- Next-Gen Location Tech
- Protection of Domestic Violence Victims
- And More
Cybersecurity
In 2015, SuperCom identified the cybersecurity market as a fast-growing space with significant advantages due to synergistic technologies and a shared customer base with its e-Gov and IoT business units. Consequently, SuperCom strategically acquired Prevision Ltd., a company with a strong presence in the market and a broad range of competitive cybersecurity services.
During the first quarter of 2016, SuperCom acquired Safend Ltd., an international provider of cutting-edge endpoint data protection guarding against corporate data loss and theft through content discovery and inspection, encryption methodologies, and comprehensive device and port control.
Both acquisitions significantly expanded the breadth of the company’s global cybersecurity capabilities.
e-Gov
Through proprietary e-government platforms and innovative solutions for traditional and biometrics enrollment, personalization, issuance, and border control services, SuperCom has helped governments, and national agencies design and issue secured multi-identification, or Multi-ID, documents and robust digital identity solutions to their citizens, visitors, and lands.
The company has focused on expanding its activities in the traditional identification, or ID, and electronic identification, or e-Gov, markets, including the design, development, and marketing of identification technologies and solutions to governments in Europe, Asia, America, and Africa using SuperCom’s e-Government platforms.
Market Opportunity
Data from Berg Insight estimates the market for electronic monitoring solutions will grow from $1.2 billion in 2021 to $2.1 billion in 2026, marking a CAGR of 10.8% for the forecast period.
High recidivism rates, prison overcrowding, and soaring incarceration costs are some factors that are driving the electronic monitoring of offenders’ market growth.
An analysis by ReportLinker forecasts that the global cybersecurity market will grow from an estimated value of $173.5 billion in 2022 to $266.2 billion by 2027, achieving a CAGR of 8.9% for the period.
The increased number of data breaches worldwide, the ability of malicious actors to operate from anywhere in the world, the links between cyberspace and physical systems, and the difficulty of reducing vulnerabilities and consequences in complex cyber networks are some factors driving the cybersecurity market growth.
Management Team
Ordan Trabelsi is President and CEO of SuperCom. He has over 15 years of experience as CEO, growing high-tech companies globally. He also has experience in research and development and product innovation, as well as hands-on experience in cybersecurity, encryption, advanced mathematics, and mobile and internet network technologies. Prior to joining SuperCom, he served as co-founder and CEO of Klikot Inc., a global social networking company. He holds an MBA from Columbia University and a B.Sc. in Computer Engineering from The Technion: Israel Institute of Technology.
Barak Trabelsi is COO of SuperCom. He has expertise in big data, cyber, mobile, and internet network technologies, as well as extensive experience in product development and strategies. Prior to joining SuperCom, he served as Senior Product Manager at Equinox Ltd. Before that, he served for four years as VP of R&D at Sigma Wave, a wireless, security, and internet-focused company. He holds a B.Sc. in Computer Science and Business, as well as an MBA from Tel Aviv University.
Gil Alfi is VP of Sales at Safend Ltd., SuperCom’s cybersecurity subsidiary. He joined SuperCom in 2016 as VP of Business Development for Safend. He has more than 18 years of experience in technology companies. He served as an R&D team technology lead for more than seven years and as Director of Product Management for various telecom and wireless companies for more than 10 years. Prior to joining SuperCom, he served as Regional Sales Director at Safend, managing sales regions in Europe and Africa. He holds a B.Sc. in Computer Science and Mathematics and an M.Sc. in Computer Science from Bar-Ilan University.
SuperCom Ltd. (NASDAQ: SPCB), closed Thursday's trading session at $0.596, off by 2.1346%, on 77,441 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.55/$4.595.
Recent News
- SuperCom Ltd. (NASDAQ: SPCB) - InvestorNewsBreaks - SuperCom Ltd. (NASDAQ: SPCB) Announces 'Milestone' Multimillion-Dollar Electronic Monitoring Contract
- SuperCom Secures New Electronic Monitoring Contract in California Valued at up to $3 Million
- SuperCom Ltd. (NASDAQ: SPCB) Is 'One to Watch'
Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF)
The QualityStocks Daily Newsletter would like to spotlight Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF).
Reflex Advanced Materials (CSE: RFLX) (OTCQB: RFLXF) (FSE: HF2), a mineral exploration company, has issued 100,000 common shares to fulfill its obligations to date outlined in a mineral property option agreement. According to the announcement, the issue of shares is in connection with the Sept. 1, 2021, agreement, which is between the company and Alex Pleson and which gives Reflex Advanced Materials the sole and exclusive right to acquire up to a 100% interest in the Zigzag lithium property. The ZigZag lithium property includes eight mining claims comprising an estimated 2,710 hectares near Crescent Lake, Ontario, a region known for its highly prospective lithium deposits. The company noted that, under the terms of the agreement, the company must pay the optionor a total of $250,000, issue a total of 500,000 common shares and incur a minimum of $310,000 in aggregate exploration expenditures on or before August 3, 2024, in order to exercise the option. When these steps have been completed, Reflex Advanced Materials will acquire a 100% interest in the property, subject to a 3% net smelter royalty.
The announcement provided an update of what the company has already completed in order to obtain the interest in the property. "The company has the right to carry over excess expenditures incurred in one period to a subsequent period, and may accelerate cash and expenditure obligations in order to acquire its interest in the property in a shorter period of time than as set out in the property agreement and may at any time accelerate the exercise of the option by paying to the optionor an amount of funds equal to the remaining amount of expenditures and cash to exercise the option at the time of such payments," the announcement stated. "The 2023 shares, having recently been issued to the optionor, are subject to a statutory hold period expiring Dec. 17, 2023, being the date that is four months and one day from the date of issuance."
To view the full press release, visit https://ibn.fm/acQMr
Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) is a strategic minerals company focused on locating and developing economic properties in the strategic metals and advanced materials space. The company aims to improve domestic specialty mineral infrastructure efficiencies to meet surging national demand from North American manufacturers, effectively positioning itself as one of the only North American suppliers of high purity natural graphite for hi-tech applications.
Reflex Advanced Materials is based in Vancouver, British Columbia. Its project portfolio includes the Ruby Graphite Deposit in Montana and the ZigZag Lithium Property in Ontario.
Projects
Ruby Graphite Project
Located in a mining-friendly jurisdiction in southwest Montana, the Ruby Graphite Deposit is a low cost, rapid re-entry opportunity that produced roughly 2,400 tons of graphite from 1902 to 1948. Reflex Advanced Materials holds mining rights for 755 hectares at the Ruby Graphite Project, with 96 federal lode mining claims. Recent samples assay at 95.8% to 98.4% total carbon.
The site is notable as the only combined U.S. graphite flake and vein graphite source. Vein graphite is ideal for energy storage applications, because it requires fewer steps to achieve purity than synthetic alternatives and is therefore far less environmentally damaging. This is expected to play a key role in the project’s development as demand for electric vehicles continues to surge.
In March 2023, the company announced its submittal of permit applications to the Bureau of Land Management in respect of its exploration of the Ruby Graphite Project. Its initial drill program, expected to take place in the summer of 2023, includes plans for 3,500 total meters of drilling, cored to an average depth of 130 meters. The targets for this drill program have been identified using historical data from original mine operations and data gathered for the initial 43-101 technical report on the project, dated January 31, 2023.
ZigZag Lithium Property
Located in the Thunder Bay Mining Division of Ontario, the ZigZag Lithium Property consists of eight mining claims spanning roughly 2,710 hectares. Mineralization at the property, most notably lithium, is based in pegmatite dikes and concentrated in spodumene crystals, which are consistent throughout the entire unit.
Spodumene is readily observable in outcrops and in drill cores, with crystal sizes ranging from 3-15cm, on average.
Reflex Advanced Materials and American Energy Technologies Company Metallurgical Partnership
Reflex Advanced Materials has entered into a material processing agreement with American Energy Technologies Co., which is based in Arlington Heights, Illinois, to conduct metallurgical testwork with the goal of creating a technical support data package for Reflex’s target customer base, U.S. Federal agencies and qualification programs with hi-tech customers in the battery and battery storage business.
The resulting coated, spherionized, purified graphite (CSPG) material that is expected to be created from the aforementioned tests will be used to provide potential customers of CSPG with samples so that they can begin the material qualification process.
Market Opportunity
Graphite is an ideal battery anode and has dominated the market since the proliferation of lithium-ion batteries. Despite this demand, there is currently no significant production of lithium-ion battery anode material in North America.
Instead, most graphite sold in North America today is sourced from Chinese producers. U.S. President Joe Biden highlighted this sourcing disparity in a 2022 address:
“The United Stated depends on unreliable foreign sources for many of the strategic and critical materials necessary for the clean energy transition – such as lithium, nickel, cobalt, graphite and manganese for large-capacity batteries,” he said. “Demand for such materials is projected to increase exponentially as the world transitions to a clean energy economy.”
The U.S. Department of Energy is in the process of awarding $2.8 billion to expand domestic manufacturing of batteries for electric vehicles and combat this foreign dependency. Reflex Advanced Materials has identified its Ruby Graphite Project as a prime candidate for U.S.-sponsored initiatives due to the rarity and scarcity of natural graphite deposits in the country.
Processing graphite domestically in the U.S. is expected to provide Reflex Advanced Materials a competitive advantage as manufacturers begin to seek out American supply in the face of increased diplomatic tension. This is critical, as a rise in anode demand is expected to fuel a shortage of 8 million tonnes of graphite by 2040. World Bank Group projects 494% growth in total graphite demand by 2050.
Leadership Team
Paul Gorman is the CEO and a Director of Reflex Advanced Materials. He brings to the company over 25 years of experience in junior mining finance, public listings, viability assessment and operational rationalization. For 18 years, Mr. Gorman served as president and managing partner of Riverbank Capital, where he played an instrumental role in raising more than $85 million for small-cap companies. In 2008, he funded Industrial Minerals Inc. (later Northern Graphite) and served in an advisory role for four other graphite companies, contributing significantly to the revitalization of the junior graphite space in North America. Mr. Gorman founded Mega Graphite Inc. in 2009 and has served as chief executive for three other companies.
Tasheel Jeerh, CPA, is the company’s CFO. He is a finance and accounting professional with over a decade of experience spanning both public and private sectors. Prior to joining Reflex Advanced Materials, Mr. Jeerh played a pivotal role in the growth of a private upstream oil and gas firm, dealing with over $2 billion in M&A activity and $1 billion in financing activities. He gained his designation at PricewaterhouseCoopers, where he worked as a manager in the assurance practice.
Greg Bell is Project Manager for Reflex Advanced Materials. He is a multi-disciplined engineering management professional with more than 40 years of experience in the natural resources sector. Mr. Bell has successfully built and managed several start-up operations in various capacities. He has been active in graphite and lithium exploration for the past seven years.
Christopher W. Hill leads the company’s Corporate Development initiatives. He is an investor and entrepreneur with over a decade of experience in the capital markets. Mr. Hill began his career as an investment advisor and then began to consult and advise private companies on their paths to becoming publicly traded. He specializes in corporate development and strategic financing utilizing his large network in the capital markets.
Reflex Advanced Materials Corp. (RFLXF), closed Thursday's trading session at $0.18, off by 4.6105%, on 43,938 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.15/$0.765.
Recent News
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - InvestorNewsBreaks - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) (FSE: HF2) Issues 100K Shares to Fulfill Mineral Property Option Agreement
- InvestorNewsBreaks - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) (FSE: HF2) Mobilizes Drill Rigs, Advances Exploration Efforts at Ruby Graphite Project
- Exxon Mobil Looks to Join Lithium Mining
Data443 Risk Mitigation Inc. (OTC: ATDS)
The QualityStocks Daily Newsletter would like to spotlight Data443 Risk Mitigation Inc. (OTC: ATDS).
Data443 (OTC: ATDS), a data security and privacy software company for "All Things Data Security," today announced operating results for the second quarter and first half of 2023. Among the highlights, the company reported that it was identified as a sample vendor in the "Data Discovery" category in the 2023 Data Security, Privacy, and Storage and Data Protection Technologies Gartner(R) Hype Cycle(TM) Reports, and that its Ransomware Recovery Manager was named "Best of Inexpensive Protection" and "Best for Kiosks" in "The Best Ransomware Protection for 2023." "We continue to win new contracts and deepen our relationships with existing clients, including those within the fintech sector who entrust us with trillions of dollars' worth of transactions and tens of millions of sensitive files annually," said Data443's CEO and Founder Jason Remillard. "We believe these partnerships indicate the market's confidence in our ability to safeguard the most critical digital assets. Moreover, we believe our constant push to improve our products keeps us at the forefront of cybersecurity. We recently added antivirus and ransomware protection to our Data Placement Manager and launched an updated Ransomware Recovery Manager, reinforcing our reputation as a provider of premium security solutions."
To view the full press release, visit https://ibn.fm/3e6ky
Data443 Risk Mitigation Inc. (OTC: ATDS) is a data security and privacy software company for ALL THINGS DATA SECURITY™. The company is committed to organizing the world’s information by identifying and protecting all sensitive data regardless of location, platform or format.
Data443 provides software and services to enable secure data across devices and databases – at rest and in transit – locally, on a network, or in the cloud. With over 10,000 customers in more than 100 countries, Data443 provides a modern approach to data governance and security. The company’s framework helps customers prioritize risk, identify security gaps, and implement effective data protection and privacy management strategies.
Data443 derives revenue primarily from contracts for subscriptions to access its SaaS platforms, and ancillary services provided in connection with its subscription services. In today’s ever-changing environment with unique and complex requirements for data privacy, governance and hybrid workforces, every organization needs to know where all their data is, who has access to it and how sensitive it is. Data443 provides the tools needed to give companies control over their data processing activities, with capabilities for identifying, reporting and migrating or deleting sensitive data.
The company is headquartered in Research Triangle Park, North Carolina.
Products
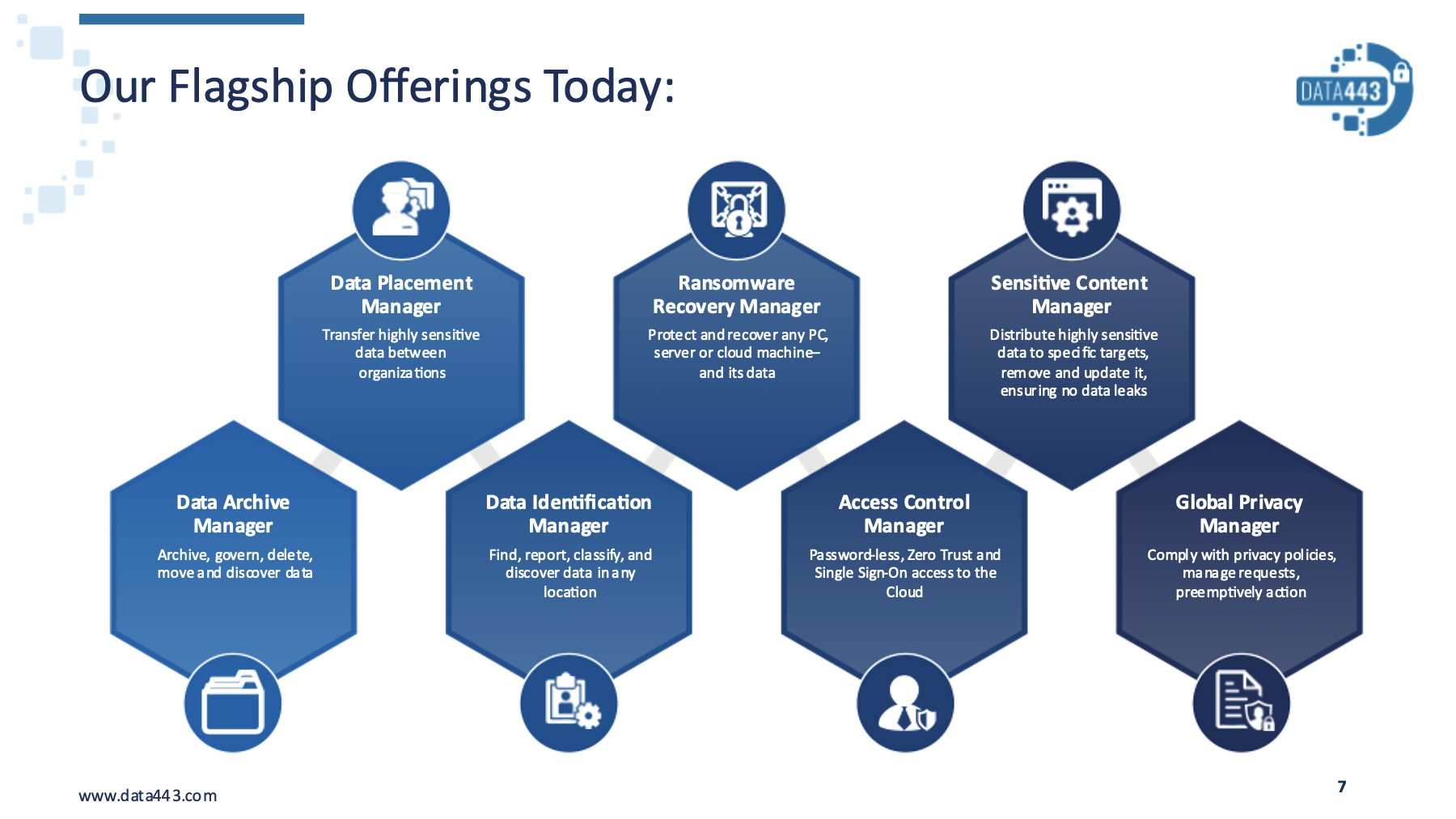
Focused on data security with a privacy-forward methodology, the Data443 product suite delivers solutions designed to securely manage data and data privacy needs on-premises, in the cloud and in hybrid environments. Offerings include:
- Data Identification Manager reduces risk by shining a light on dark data across cloud, on-premises and hybrid environments. From a centralized dashboard, Data Identification Manager provides the ability to automatically inventory all data repositories, classify and tag all data, and enable global search and discovery – all through an agentless deployment.
- Data Placement Manager quickly and securely transfers sensitive data over any public or private network. Available as an HP Nonstop server-based application and for Windows, Linux or any public cloud provider, Data Placement Manager enables the scheduling, routing, formatting and transfer of business-critical data.
- Data Archive Manager is an “all information, anywhere” archiving solution designed to handle and manage all types of privacy requests across cloud, on-premises and hybrid environments. With over 15 years operational history and hundreds of clients managing millions of mailboxes, the platform is purpose-built for information archiving, retention and privacy request management.
- Data Hound™ is a data discovery, classification and capture toolset that enables organizations to perform quick scans, detailed reporting and subsequent data actions based on policy.
- Ransomware Recovery Manager is the only industry solution that actively recovers the device, operating system and data with a simple reboot. Using patented, proven technology, the product produces 100% effectiveness for the whole device and datasets.
- Access Control Manager provides user ID and passwordless access to quickly enable trust across an organization’s entire ecosystem. Its unique architecture allows it to leverage multiple distributed authoritative sources to understand and resolve a typical access request – with the ability to enable or deny the action on the fly.
- Global Privacy Manager provides organizations one comprehensive view, for all privacy requirements, across all enterprise data, all at once. This unmatched visibility into an organization’s data assets ensures that all private and sensitive data can be identified and protected and that enterprises can obey all relevant privacy laws in any jurisdiction.
- Sensitive Content Manager is a security-centric collaboration service designed to give organizations the tools needed for successful content sharing, collaboration and safe distribution with full enterprise management in mind. With a continuous sync feature, encrypted data is automatically downloaded and updated in real time – regardless of location – ensuring that users have the most accurate data available.
Market Outlook
A report from Allied Market Research estimates that the global data security market was worth about $19 billion in 2021 and is projected to reach a value of $54.23 billion by 2027. That represents a CAGR of more than 18% for the forecast period, making data security one of the hottest areas within IT.
Separately, Fortune Business Insights estimates the global data privacy software market is valued at $2.36 billion in 2022 and projects it will grow to $25.85 billion by 2029. That represents a CAGR of 40.8% over the forecast period.
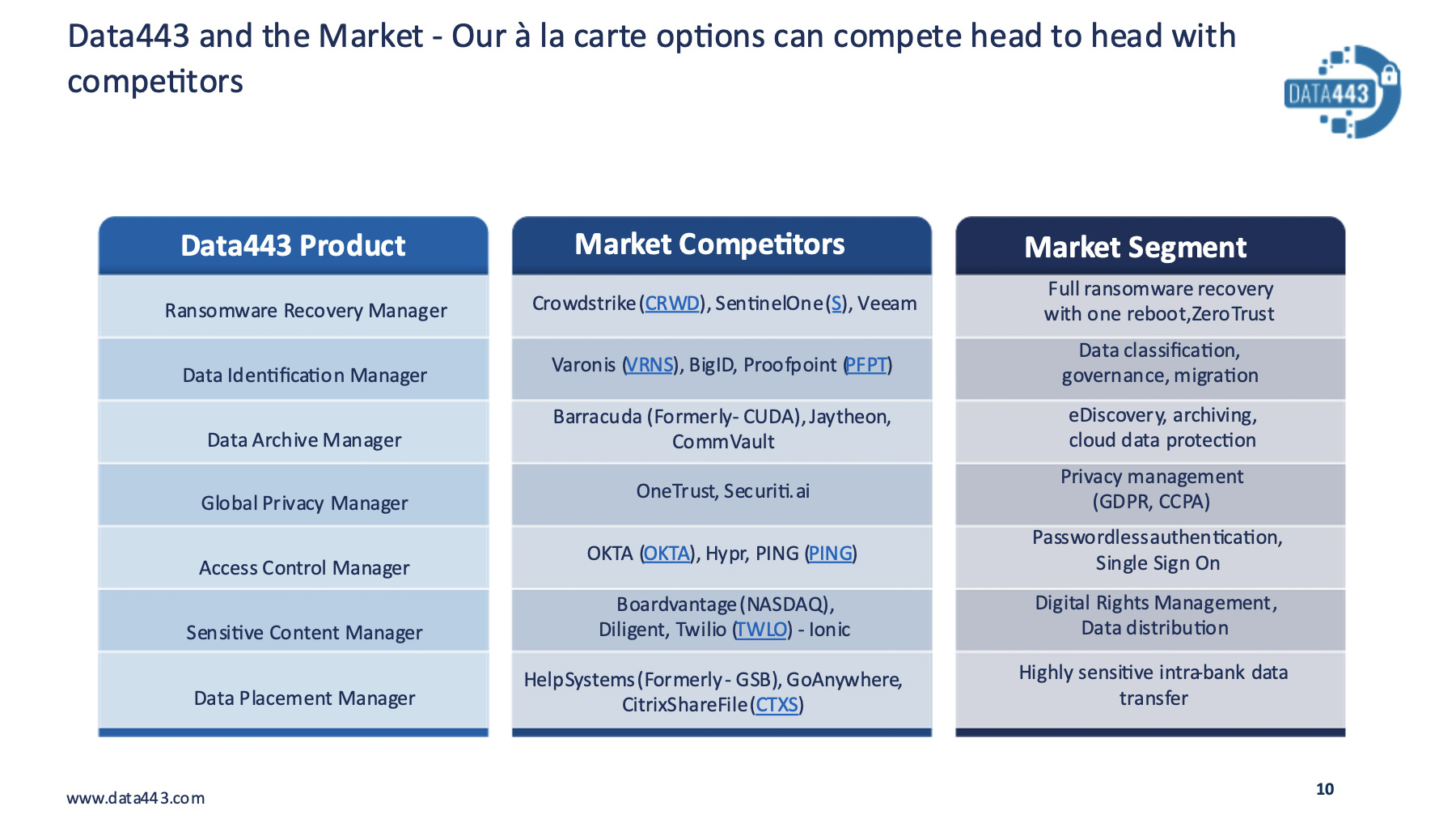
Management Team
Jason Remillard is President, CEO and Founder of Data443. He is responsible for overseeing global expansion, management, execution and corporate development. With over 25 years in global enterprise and B2C software sales and marketing, he brings deep leadership and technical experience, having spent previous time at Fortune 500 companies such as Deutsche Bank, TD Bank, IBM & Merrill Lynch.
Greg McCraw is CFO at Data443. He has over 25 years of experience helping businesses strengthen their accounting and finance operations. He previously served as Vice President of Finance for a dental services organization active in acquisitions, and, prior to that, he was managing director of a boutique accounting and finance consulting firm advising Fortune 500 clients in pharmaceutical, financial services, and private equity sectors on how to execute on regulatory and compliance solutions.
Bennett Pursell is Data443’s Chief Technology Officer. He has over 20 years of experience in IT architecture, security governance and systems integration. Prior to his role at Data443, he served as Head of Technology Architecture at Moody’s Investor Services and was Vice President and Technical Architect of Cloud Computing at Deutsche Bank, along with a host of technical and project management roles dating back to 2006, after starting his career as a web developer with a few startups and running research labs.
Kirill Kashigin is Chief Software Architect at Data443. He leads the development and quality teams, and serves as technical adviser and subject matter expert, bringing vast technical knowledge on privacy management and data security. Formerly the CTO of FileFacets, he has nearly 20 years in development of high-performance systems and deployment.
Data443 Risk Mitigation Inc. (OTC: ATDS), closed Thursday's trading session at $0.0226, off by 3.8298%, on 17,601 volume. The average volume for the last 3 months is 17,601 and the stock's 52-week low/high is $0.0175/$5.50.
Recent News
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Named as Sample Vendor in 2023 Gartner Hype Cycle Reports
- TechMediaBreaks - Data443 Risk Mitigation Inc.'s (ATDS) Comprehensive Suite Helping Businesses Stay Ahead of Cyber Criminals Worldwide
FingerMotion Inc. (NASDAQ: FNGR)
The QualityStocks Daily Newsletter would like to spotlight FingerMotion Inc. (NASDAQ: FNGR) .
FingerMotion Inc. (NASDAQ: FNGR) is an evolving technological company with core competencies in mobile payment and recharge platform solutions in China. FingerMotion is in the process of developing additional value-added technologies to market to users.
Founded in 2016, FingerMotion’s goal is to serve over a billion users in the Chinese market and expand its model to other regional markets. The company has offices in Hong Kong, Shanghai and New York City.
Current Offerings
FingerMotion is analyzing and transforming mobile data to improve the lifestyle of the public through technology and innovation. The company’s current offerings include:
- Telecommunications Products and Services – FingerMotion’s proprietary universal exchange platform, ‘PigeonHole Integration System (PIS)’, offers seamless integration between telecom operators and online stores. The service platform’s offerings include top up and recharge, data plan, mobile phone, loyalty points redemption and subscription plans. The platform offers reliable and secure transactions, real-time reconciliation, simple integration for partners and efficient settlements.
- SMS and MMS Services – The integrated platform is registered as FingerMotion’s IP in China and provides a robust back-end control panel for corporate partners to manage their own messaging settings. FingerMotion’s clients range from insurance to financial industries, ecommerce firms, airlines and more. The platform offers competitive pricing for partners and provides quick and efficient review to meet timely marketing initiatives.
- Big Data Insights – FingerMotion brings Big Data-enabled insurance solutions through its Big Data Insights arm, Sapientus. The company’s strategic partnerships with the largest Chinese telecommunications giants allow access to uncover behavior insights through geolocation and mobile data usage. Its Big Data offerings include risk scoring, precise marketing, simplified underwriting and customized products.
- Rich Communication Services (RCS) – FingerMotion’s RCS platform will be a proprietary business messaging solution that enables businesses and brands to communicate their services to customers via 5G infrastructure. The company expects its RCS platform to offer a better user experience, more efficiency and cost-effectiveness when compared to other solutions.
Telecommunications and Insurtech Markets
The global telecommunications market was valued at $1.74 trillion in 2019 and is expected to grow at a CAGR of 5% from 2020 to 2027. The steady increase is expected to be driven by the adoption of 5G and the increased popularity of Internet of Things (IoT) applications.
The Chinese telecom market was valued at $254.1 billion in 2017 and is also constantly expanding. The current Chinese telecom market is dominated by three mobile operators – China Mobile, China Unicom and China Telecom, which together are responsible for around 1.6 billion active subscribers (https://ibn.fm/zfwy9).
In addition, the insurtech (insurance technology) market was valued at $2.72 billion globally in 2020 and is expected to grow at a CAGR of 48.8% from 2021 to 2028. The large increase is attributed to the rising use of technology solutions for everyday activities like acquiring insurance coverage (https://ibn.fm/TGo7D).
Through its proprietary platforms and technologies, FingerMotion is uniquely positioned to capitalize on the telecom and insurtech markets’ growth and opportunities.
Management Team
Martin J. Shen is the Chief Executive Officer of FingerMotion Inc. He has over 15 years of experience in senior management roles within entrepreneurial startups and large multinational corporations. He has acquired a wide range of corporate management, financial oversight and operation administration expertise through these roles. In his most recent role, he founded Imperial Distributors (formerly known as AP Martin Pharmaceutical Supplies Ltd.), establishing the company as the preferred choice for distributional support to regional pharmacies throughout Western Canada. Before founding Imperial, Mr. Shen served as the Chief Operating Officer and Chief Financial Officer at Wales and Son Industrial (formerly Weir Minerals), a firm specializing in global delivery and support for mining slurry equipment. He began his career at PricewaterhouseCoopers in Vancouver, with work tours in the tax department in Singapore and the tax audit and advisory group in Hong Kong. Mr. Shen is a U.S. Certified Public Accountant and holds a Bachelor of Science from the University of British Columbia.
Lee Yew Hon is the company’s Chief Financial Officer. From 2006 until November 2020, he was the Chief Financial Officer of Cubinet Interactive Group of Companies, and he also took on the Chief Operating Officer role in 2011. During his tenure, he was instrumental in leading Cubinet and building teams across the Southeast Asia region, setting up financial processes within a short time. Mr. Lee spearheaded the growth of Cubinet to other regions, including Europe, the Middle East and Russia. He received his diploma from Tunku Abdul Rahman College in 1996. He is a Chartered Accountant, a member of the Malaysia Institute of Accountants (MIA) and an Associate Member of the Chartered Institute of Management Accountants, UK (ACMA).
Li Li is the Senior Vice President of FingerMotion. She recently served as Advisor to Shenzhen WuYiKa Technology Co. Ltd., a comprehensive service platform dedicated to online service distribution and payment. The company has become a fast and efficient provider of new media marketing solutions for the mobile internet. She has held high-level management positions with multiple industry names, including Hangzhou JiuYue Information Technology Co. Ltd. and Hangzhou LingXuan Information Technology. Ms. Li started her career in 2004, founding Shanghai ChuangYeZZ Network Technology Co. Ltd. and serving as its Vice President. With the close cooperation of local operators, the company launched SMS, MMS, WAP, mobile JAVA games, Hunan Satellite TV e-magazine and other wireless internet services to meet the rapid development of wireless internet and application requirements. She received her degree from Nanjing Academy of Engineering.
FingerMotion Inc. (FNGR), closed Thursday's trading session at $4.75, off by 1.4523%, on 229,449 volume. The average volume for the last 3 months is 222,159 and the stock's 52-week low/high is $0.62/$9.795.
Recent News
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) CEO Featured in C-Level Interview with BestGrowthStocks.Com
- InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Releases Financial Report for Q1 2024
- InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) to Present at LD Micro Invitational XIII
InMed Pharmaceuticals Inc. (NASDAQ: INM)
The QualityStocks Daily Newsletter would like to spotlight InMed Pharmaceuticals Inc. (NASDAQ: INM).
InMed Pharmaceuticals Inc. (NASDAQ: INM) is a global leader in the manufacturing and clinical development of rare cannabinoids. InMed is a clinical stage company developing cannabinoid-based pharmaceutical drug candidates, as well as manufacturing technologies for pharmaceutical-grade rare cannabinoids.
The company is dedicated to delivering new therapeutic alternatives to treat conditions with high unmet medical needs. The company is also developing a proprietary manufacturing technology to produce pharmaceutical-grade rare cannabinoids in the lab and has recently announced an LOI to acquire a leading rare cannabinoid manufacturer.
Research and Technology
There are more than 100 rare cannabinoids found in only trace amounts in the cannabis plant, together making up less than 1% of the plant’s biomass. InMed is initially focused on the therapeutic benefits of cannabinol (CBN) in diseases with high unmet medical need. Preclinical studies of CBN demonstrated an excellent safety profile and showed CBN has potential for therapeutic benefit over other cannabinoids such as tetrahydrocannabinol (THC) and cannabidiol (CBD).
Evidence suggests there may be great therapeutic potential in rare cannabinoids. Each has a specific chemical structure, and different cannabinoids have been observed to have distinct physiological properties in humans, including therapeutic potential for specific diseases as well as unique safety profiles. CBN is the active pharmaceutical ingredient (API) in InMed’s two lead programs for dermatological and ocular diseases.
InMed’s most advanced compound, INM-755, is a CBN topical cream under clinical development for the treatment of epidermolysis bullosa, a severe genetic skin disorder. To date, INM-755 has been evaluated in two Phase 1 clinical trials in healthy volunteers. InMed has filed Clinical Trial Applications in several countries as part of a global Phase 2 clinical trial of INM-755 (cannabinol) cream in epidermolysis bullosa. Responses from the National Competent Authorities and Ethics Committees are expected throughout the summer of 2021.
InMed is also involved in developing INM-088, an ocular CBN formulation being researched for the treatment of glaucoma, the second leading cause of blindness in the developed world. InMed is currently evaluating several formulations to deliver CBN into the eye to address issues of dosing frequency, side effects and treatment penetration. INM-088 is being designed for topical delivery to the eye. This localized delivery results in very little drug being absorbed or migrating into the bloodstream, thus minimizing potential adverse side effects. INM-088 shows promise to reduce intraocular pressure and provide neuroprotection of the eye.
Manufacturing
The limited availability of rare cannabinoids like CBN makes them economically impractical to extract directly from the plant for pharmaceutical use. InMed is developing IntegraSyn, a cannabinoid synthesis manufacturing system to create rare cannabinoids in the lab that are bioidentical to the compounds derived from the cannabis plant. IntegraSyn uses multiple standard pharmaceutical processes and has achieved a cannabinoid yield of 5 grams per liter, surpassing commercial viability and significantly exceeding currently reported industry yields. InMed is now focusing on manufacturing scale-up to larger batch sizes while continuing process optimization, targeting increased cannabinoid yield and further reducing overall cost of goods.
BayMedica Inc. Acquisition
On June 29, 2021, InMed announced it had entered into a non-binding letter of intent to acquire BayMedica Inc., a private company based in Nevada and California that specializes in the manufacture and commercialization of rare cannabinoids.
As noted in the news release, BayMedica is a revenue-stage biotechnology company leveraging its significant expertise in synthetic biology and pharmaceutical chemistry to develop efficient, scalable and proprietary manufacturing approaches to produce high quality, regulatory-compliant rare cannabinoids for consumer applications. BayMedica is currently commercializing the rare cannabinoid CBC (cannabichromene) as a B2B supplier to distributors and manufacturers marketing products in the health and wellness sector. BayMedica is planning additional rare cannabinoid launches for the coming year.
Pursuant to the indicative terms of the LOI, InMed and BayMedica intend to negotiate and enter into a definitive agreement under which InMed would acquire 100% of BayMedica in exchange for 1.6 million InMed common shares to be issued to BayMedica’s equity and convertible debt holders, with any such issued InMed common shares being subject to a six-month contractual hold period.
Market Outlook
There is a rapidly growing demand for rare cannabinoids. However, their low natural concentration makes traditional harvesting of these compounds cost prohibitive. Biosynthesis allows production of rare cannabinoids in the lab that are bioidentical to compounds found in nature, with significantly higher yields which reduce costs. Biosynthesis can produce pharmaceutical-grade, bioidentical, THC-free compounds at a cost that’s 70 to 90 percent less than wholesale prices of naturally harvested rare cannabinoids.
Cannabinoid-based pharmaceuticals are expected to overtake the market as rare cannabinoids become less expensive and more available. According to Statista, the value of the consumer market for cannabinoid-based pharmaceuticals in the United States is forecast to grow to $25 billion by 2025 and to $50 billion by 2029, with cannabinoid-based pharmaceuticals used to treat health conditions including pain, respiratory conditions, autoimmune conditions and more.
Management Team
Eric A. Adams has been CEO and president of InMed since June 2016. He has more than 25 years of experience in establishing corporate entities, capital formation, global market development, mergers and acquisitions, licensing and corporate governance. He previously served as CEO at enGene Inc. Prior to enGene, he held senior positions in global market development with QLT Inc. (Vancouver), Advanced Tissue Sciences Inc. (La Jolla, CA), Abbott Laboratories (Chicago, IL) and Fresenius AG (Germany).
Bruce S. Colwill is InMed’s CFO. He has more than 25 years of financial leadership experience in public and private companies. Prior to InMed, he served as CFO of General Fusion Inc., a private clean energy company. He was also CFO at Entrée Resources Inc., a mineral exploration company, from 2011 to 2016. He has held CFO roles at Neuromed Pharmaceuticals Ltd., Response Biomedical Corp, Forbes Medi-Tech Inc. and Euronet Worldwide Inc.
Alexandra D.J. Mancini is Senior Vice President, Clinical and Regulatory Affairs at InMed. She has more than 30 years of global biopharmaceutical research and development experience. She has been an executive with numerous biotech companies, including senior vice president of Clinical and Regulatory Affairs at Sirius Genomics; senior vice president of Clinical and Regulatory Affairs at INEX Pharmaceuticals; and vice president of Regulatory Affairs at QLT Inc.
Eric C. Hsu is Senior Vice President, Pre-Clinical Research and Development at InMed. He joined InMed with more than 18 years of scientific leadership experience in the field of gene therapy. He has held various positions within enGene Inc., including vice president of Research and vice president of Scientific Affairs and Operations. He received his Doctorate from the Department of Medical Biophysics at the University of Toronto.
Michael Woudenberg is Vice President, Chemistry, Manufacturing and Controls at InMed. He has more than 20 years of successful drug development, process engineering, GMP manufacturing and leadership experience. He has held positions with 3M, Cardiome Pharma, Arbutus Biopharma and, most recently, was Managing Director of Phyton Biotech LLC.
InMed Pharmaceuticals Inc. (INM), closed Thursday's trading session at $0.88, off by 0.452489%, on 8,436 volume. The average volume for the last 3 months is 8,420 and the stock's 52-week low/high is $0.875/$19.50.
Recent News
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Participation in Water Tower Research Fireside Chat Series
- InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Issues Update on BayMedica Rare Cannabinoid Business
- InvestorNewsBreaks – InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Key Milestones, 2023 Business Outlook
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Three States in the Northeast Set July Cannabis Sales Records
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) to Roll Out New LMS Integration Capability
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Codelco Slashes 2023 Copper Output Predictions
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Delivers 47% Revenue Growth and Expanded Gross Margins for Second Quarter of 2023
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Announces Corporate Update, $2M Debt Financing
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Releases Q2 Financial Report, Corporate Update
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: XPHYF) (FSE: 4XT) - BioNxt Solutions Announces Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Completes $29.8M Issue of Ordinary Shares
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Leading Out in Sustainably Producing Nickel for Growing Steel Space
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Argo Edge Cloud Security Platform Tested at DEF Con with 125,000+ Cyberattacks
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Researchers Develop Novel Way to Detect Pediatric Brain-Cancer Mutations
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Announces Strong Q2 Results, Eyes Growing Momentum
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Begins Preparing for CYB003 Capsule Manufacturing for Potential Phase 3 Trial
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Q2 2023 Results
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) and Davidson Technologies, Inc. Collaborate on Applications to Advance National Defense Efforts
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Study Evaluates Whether Neuropathic Pain, Depression Can Be Simultaneously Treated by Psilocybin
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - TechMediaBreaks - Electronic Servitor Publication Network Inc.'s (XESP) 'Growth-as-a-Service' Platform Helping Companies Enhance User Engagement
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd. (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) May Become Vital Cog in Tin's Tenuous Supply Chain
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Schedules Release of Q2 2023 Results, Conference Call
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Reports on Q2 2023 Financial Results, Corporate Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) CEO Featured in C-Level Interview with BestGrowthStocks.Com
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Pushing AI Conversation Forward
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - InvestorNewsBreaks - First Tellurium Corp. (CSE: FTEL) (OTC: FSTTF) Confirms Crucial Structural Connection at Deer Horn
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) Completes New Drone-Borne Magnetic Survey at Augustus
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Inks POC Agreement with One of World's Largest Industrial Equipment Manufacturers
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Another Quarter of Strong Growth, Improved Profitability
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - GEMXX Corp. (GEMZ) Expanding Brand Awareness As It Strengthens Mine Project Financing
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Eyes 2024 Delivery of FP300 Commercial Systems, Answering Larger-Scale Need with FP1500
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Surpasses 50,000 Members Nationwide, Creating Thriving Golfing Communities
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Granted FDA Orphan Drug Designation for REQORSA(R) for Patients with SCLC
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces New coyni Mobile Point-of-Sale App
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Releases Q2 2023 Financial Report, Corporate Update
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Completes Design Concept for Next-Generation EV Inverter
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Announces Addition of New to the Street to Strategic Media Community
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Files 2022 Annual Report with SEC
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Gallup Poll Finds Highest Ever Support for Cannabis Legalization Among Republicans
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Moves Operations to Hong Kong in Strategic Move to Expand Global Presence
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Participation in Water Tower Research Fireside Chat Series
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Moves Forward in Production of World's First Rapid Blood Alcohol Detox Drink
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - Knightscope Inks Deal with Major Player in Self Storage
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces Intent for Human Clinical Study for Diabetes and Weight Loss Following Positive DehydraTECH(TM)-CBD Results in Animals
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Releases Q2 2023 Financial Report, Corporate Update
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - Mydecine Innovations Group Provides Corporate Update
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures SFDA Approval for sugarBEAT(R)
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - InvestorNewsBreaks - NextPlat Corp. (NASDAQ: NXPL) Announces Q2 2023 Results
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Announces Commencement of Stock Buyback Program
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Adds Leading Plant-Based TiNDLE Chicken to Growing Online Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR), NeoGames' Pariplay Complete Pariplay Systems Integration
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Amended, Restated Q3 2023 MD&A
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Hosts Webinar Showcasing Revolutionary UltraShear Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Shares Encouraging Results on Potential Cancer Treatment Approach
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - High Potential, Low Price: Top Marijuana Penny Stocks Under $1 for August 2023
- Renovaro BioSciences Inc. (NASDAQ: RENB) - InvestorNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB) Announces Presentation of 'Impressive' Results from Proof-of-Concept Studies
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - InvestorNewsBreaks - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) (FSE: HF2) Issues 100K Shares to Fulfill Mineral Property Option Agreement
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Files Technical Report, Unveils New Logo
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - RJD Green Inc. (RJDG) Is 'One to Watch'
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Commences First Large-Scale Commercial Pilot with Congeladora Nino
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Continues Looking for Opportunities to Innovate, Offer 'neXt-Level' Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Releases Q1 2023 Financial Report, Corporate Update
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Strategic Engagement to Step up IP and Patent Expansion Efforts
- SuperCom Ltd. (NASDAQ: SPCB) - InvestorNewsBreaks - SuperCom Ltd. (NASDAQ: SPCB) Announces 'Milestone' Multimillion-Dollar Electronic Monitoring Contract
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Q2 2023 Financial Report Captures Industry's Attention with 180% Revenue Increase
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Participation in Upcoming Sidoti Microcap Conference
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Positive PFS Results, 'Incredible Potential' for SWA Project
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Exceeds Three Million Whipshots® Cans Sold, Announces Largest Expansion Yet
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces its Six Corporate Guests TV Interviews; Broadcastings on Newsmax and The FOX Business Network, Airings Start on Saturday, August 12, 2023, at 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Shares Status Update on Key Corporate Efforts
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Reports Record Revenue of $63.8 Million in Fiscal Year 2023, Posts 16% Growth and Makes Significant Strides Toward Profitability
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Updates Plans to Release Q2 2023 Results, Rescheduled Earnings Call/Webcast
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Appoints Life Sciences Exec as New CEO
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - Newly merged Entity Superlatus, Inc with Trxade Health, Inc. Appoints Leading CPG Management Team
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Moving to Become the Leading Metal Separation Products and Services Provider, One Investment at a Time
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Completes First Phase of Resource Expansion Campaign as Part of Restart Program
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Releases Q1 2024 Financial Report, Corporate Update
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Releases Q2, YTD 2023 Financial, Operational Report
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Expands Virtual Reality Law Enforcement Training with New Immersive Scenarios
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Three States in the Northeast Set July Cannabis Sales Records
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) to Roll Out New LMS Integration Capability
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Codelco Slashes 2023 Copper Output Predictions
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Delivers 47% Revenue Growth and Expanded Gross Margins for Second Quarter of 2023
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Announces Corporate Update, $2M Debt Financing
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Releases Q2 Financial Report, Corporate Update
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: XPHYF) (FSE: 4XT) - BioNxt Solutions Announces Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Completes $29.8M Issue of Ordinary Shares
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Leading Out in Sustainably Producing Nickel for Growing Steel Space
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Argo Edge Cloud Security Platform Tested at DEF Con with 125,000+ Cyberattacks
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Researchers Develop Novel Way to Detect Pediatric Brain-Cancer Mutations
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Announces Strong Q2 Results, Eyes Growing Momentum
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Begins Preparing for CYB003 Capsule Manufacturing for Potential Phase 3 Trial
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Q2 2023 Results
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) and Davidson Technologies, Inc. Collaborate on Applications to Advance National Defense Efforts
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Study Evaluates Whether Neuropathic Pain, Depression Can Be Simultaneously Treated by Psilocybin
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - TechMediaBreaks - Electronic Servitor Publication Network Inc.'s (XESP) 'Growth-as-a-Service' Platform Helping Companies Enhance User Engagement
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd. (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) May Become Vital Cog in Tin's Tenuous Supply Chain
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Schedules Release of Q2 2023 Results, Conference Call
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Reports on Q2 2023 Financial Results, Corporate Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) CEO Featured in C-Level Interview with BestGrowthStocks.Com
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Pushing AI Conversation Forward
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - InvestorNewsBreaks - First Tellurium Corp. (CSE: FTEL) (OTC: FSTTF) Confirms Crucial Structural Connection at Deer Horn
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) Completes New Drone-Borne Magnetic Survey at Augustus
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Inks POC Agreement with One of World's Largest Industrial Equipment Manufacturers
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Another Quarter of Strong Growth, Improved Profitability
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - GEMXX Corp. (GEMZ) Expanding Brand Awareness As It Strengthens Mine Project Financing
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Eyes 2024 Delivery of FP300 Commercial Systems, Answering Larger-Scale Need with FP1500
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Surpasses 50,000 Members Nationwide, Creating Thriving Golfing Communities
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Granted FDA Orphan Drug Designation for REQORSA(R) for Patients with SCLC
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces New coyni Mobile Point-of-Sale App
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Releases Q2 2023 Financial Report, Corporate Update
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Completes Design Concept for Next-Generation EV Inverter
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Announces Addition of New to the Street to Strategic Media Community
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Files 2022 Annual Report with SEC
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Gallup Poll Finds Highest Ever Support for Cannabis Legalization Among Republicans
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Moves Operations to Hong Kong in Strategic Move to Expand Global Presence
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Participation in Water Tower Research Fireside Chat Series
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Moves Forward in Production of World's First Rapid Blood Alcohol Detox Drink
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - Knightscope Inks Deal with Major Player in Self Storage
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces Intent for Human Clinical Study for Diabetes and Weight Loss Following Positive DehydraTECH(TM)-CBD Results in Animals
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Releases Q2 2023 Financial Report, Corporate Update
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - Mydecine Innovations Group Provides Corporate Update
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures SFDA Approval for sugarBEAT(R)
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - InvestorNewsBreaks - NextPlat Corp. (NASDAQ: NXPL) Announces Q2 2023 Results
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Announces Commencement of Stock Buyback Program
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Adds Leading Plant-Based TiNDLE Chicken to Growing Online Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR), NeoGames' Pariplay Complete Pariplay Systems Integration
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Amended, Restated Q3 2023 MD&A
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Hosts Webinar Showcasing Revolutionary UltraShear Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Shares Encouraging Results on Potential Cancer Treatment Approach
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - High Potential, Low Price: Top Marijuana Penny Stocks Under $1 for August 2023
- Renovaro BioSciences Inc. (NASDAQ: RENB) - InvestorNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB) Announces Presentation of 'Impressive' Results from Proof-of-Concept Studies
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - InvestorNewsBreaks - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) (FSE: HF2) Issues 100K Shares to Fulfill Mineral Property Option Agreement
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Files Technical Report, Unveils New Logo
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - RJD Green Inc. (RJDG) Is 'One to Watch'
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Commences First Large-Scale Commercial Pilot with Congeladora Nino
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Continues Looking for Opportunities to Innovate, Offer 'neXt-Level' Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Releases Q1 2023 Financial Report, Corporate Update
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Strategic Engagement to Step up IP and Patent Expansion Efforts
- SuperCom Ltd. (NASDAQ: SPCB) - InvestorNewsBreaks - SuperCom Ltd. (NASDAQ: SPCB) Announces 'Milestone' Multimillion-Dollar Electronic Monitoring Contract
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Q2 2023 Financial Report Captures Industry's Attention with 180% Revenue Increase
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Participation in Upcoming Sidoti Microcap Conference
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Positive PFS Results, 'Incredible Potential' for SWA Project
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Exceeds Three Million Whipshots® Cans Sold, Announces Largest Expansion Yet
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces its Six Corporate Guests TV Interviews; Broadcastings on Newsmax and The FOX Business Network, Airings Start on Saturday, August 12, 2023, at 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Shares Status Update on Key Corporate Efforts
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Reports Record Revenue of $63.8 Million in Fiscal Year 2023, Posts 16% Growth and Makes Significant Strides Toward Profitability
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Updates Plans to Release Q2 2023 Results, Rescheduled Earnings Call/Webcast
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Appoints Life Sciences Exec as New CEO
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - Newly merged Entity Superlatus, Inc with Trxade Health, Inc. Appoints Leading CPG Management Team
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Moving to Become the Leading Metal Separation Products and Services Provider, One Investment at a Time
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Completes First Phase of Resource Expansion Campaign as Part of Restart Program
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Releases Q1 2024 Financial Report, Corporate Update
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Releases Q2, YTD 2023 Financial, Operational Report
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Expands Virtual Reality Law Enforcement Training with New Immersive Scenarios
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Three States in the Northeast Set July Cannabis Sales Records
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) to Roll Out New LMS Integration Capability
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Codelco Slashes 2023 Copper Output Predictions
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Delivers 47% Revenue Growth and Expanded Gross Margins for Second Quarter of 2023
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Announces Corporate Update, $2M Debt Financing
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Releases Q2 Financial Report, Corporate Update
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: XPHYF) (FSE: 4XT) - BioNxt Solutions Announces Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Completes $29.8M Issue of Ordinary Shares
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Leading Out in Sustainably Producing Nickel for Growing Steel Space
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Argo Edge Cloud Security Platform Tested at DEF Con with 125,000+ Cyberattacks
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Researchers Develop Novel Way to Detect Pediatric Brain-Cancer Mutations
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Announces Strong Q2 Results, Eyes Growing Momentum
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Begins Preparing for CYB003 Capsule Manufacturing for Potential Phase 3 Trial
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Q2 2023 Results
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) and Davidson Technologies, Inc. Collaborate on Applications to Advance National Defense Efforts
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Study Evaluates Whether Neuropathic Pain, Depression Can Be Simultaneously Treated by Psilocybin
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - TechMediaBreaks - Electronic Servitor Publication Network Inc.'s (XESP) 'Growth-as-a-Service' Platform Helping Companies Enhance User Engagement
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd. (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) May Become Vital Cog in Tin's Tenuous Supply Chain
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Schedules Release of Q2 2023 Results, Conference Call
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Reports on Q2 2023 Financial Results, Corporate Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) CEO Featured in C-Level Interview with BestGrowthStocks.Com
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Pushing AI Conversation Forward
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - InvestorNewsBreaks - First Tellurium Corp. (CSE: FTEL) (OTC: FSTTF) Confirms Crucial Structural Connection at Deer Horn
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) Completes New Drone-Borne Magnetic Survey at Augustus
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Inks POC Agreement with One of World's Largest Industrial Equipment Manufacturers
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Another Quarter of Strong Growth, Improved Profitability
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - GEMXX Corp. (GEMZ) Expanding Brand Awareness As It Strengthens Mine Project Financing
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Eyes 2024 Delivery of FP300 Commercial Systems, Answering Larger-Scale Need with FP1500
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Surpasses 50,000 Members Nationwide, Creating Thriving Golfing Communities
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Granted FDA Orphan Drug Designation for REQORSA(R) for Patients with SCLC
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces New coyni Mobile Point-of-Sale App
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Releases Q2 2023 Financial Report, Corporate Update
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Completes Design Concept for Next-Generation EV Inverter
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Announces Addition of New to the Street to Strategic Media Community
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Files 2022 Annual Report with SEC
- Ideanomics Inc. (NASDAQ: IDEX) - Ideanomics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Gallup Poll Finds Highest Ever Support for Cannabis Legalization Among Republicans
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Moves Operations to Hong Kong in Strategic Move to Expand Global Presence
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Participation in Water Tower Research Fireside Chat Series
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Moves Forward in Production of World's First Rapid Blood Alcohol Detox Drink
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - Knightscope Inks Deal with Major Player in Self Storage
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces Intent for Human Clinical Study for Diabetes and Weight Loss Following Positive DehydraTECH(TM)-CBD Results in Animals
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Releases Q2 2023 Financial Report, Corporate Update
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - Mydecine Innovations Group Provides Corporate Update
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures SFDA Approval for sugarBEAT(R)
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - InvestorNewsBreaks - NextPlat Corp. (NASDAQ: NXPL) Announces Q2 2023 Results
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Announces Commencement of Stock Buyback Program
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Adds Leading Plant-Based TiNDLE Chicken to Growing Online Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR), NeoGames' Pariplay Complete Pariplay Systems Integration
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Amended, Restated Q3 2023 MD&A
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Hosts Webinar Showcasing Revolutionary UltraShear Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Shares Encouraging Results on Potential Cancer Treatment Approach
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - High Potential, Low Price: Top Marijuana Penny Stocks Under $1 for August 2023
- Renovaro BioSciences Inc. (NASDAQ: RENB) - InvestorNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB) Announces Presentation of 'Impressive' Results from Proof-of-Concept Studies
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - InvestorNewsBreaks - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) (FSE: HF2) Issues 100K Shares to Fulfill Mineral Property Option Agreement
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Files Technical Report, Unveils New Logo
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - RJD Green Inc. (RJDG) Is 'One to Watch'
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Commences First Large-Scale Commercial Pilot with Congeladora Nino
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Continues Looking for Opportunities to Innovate, Offer 'neXt-Level' Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Releases Q1 2023 Financial Report, Corporate Update
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Strategic Engagement to Step up IP and Patent Expansion Efforts
- SuperCom Ltd. (NASDAQ: SPCB) - InvestorNewsBreaks - SuperCom Ltd. (NASDAQ: SPCB) Announces 'Milestone' Multimillion-Dollar Electronic Monitoring Contract
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Q2 2023 Financial Report Captures Industry's Attention with 180% Revenue Increase
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Participation in Upcoming Sidoti Microcap Conference
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Positive PFS Results, 'Incredible Potential' for SWA Project
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Exceeds Three Million Whipshots® Cans Sold, Announces Largest Expansion Yet
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces its Six Corporate Guests TV Interviews; Broadcastings on Newsmax and The FOX Business Network, Airings Start on Saturday, August 12, 2023, at 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Shares Status Update on Key Corporate Efforts
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Reports Record Revenue of $63.8 Million in Fiscal Year 2023, Posts 16% Growth and Makes Significant Strides Toward Profitability
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Updates Plans to Release Q2 2023 Results, Rescheduled Earnings Call/Webcast
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Appoints Life Sciences Exec as New CEO
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - Newly merged Entity Superlatus, Inc with Trxade Health, Inc. Appoints Leading CPG Management Team
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Moving to Become the Leading Metal Separation Products and Services Provider, One Investment at a Time
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Completes First Phase of Resource Expansion Campaign as Part of Restart Program
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Releases Q1 2024 Financial Report, Corporate Update
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Releases Q2, YTD 2023 Financial, Operational Report
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. Expands Virtual Reality Law Enforcement Training with New Immersive Scenarios
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.









































