The QualityStocks Daily Stock List
- Biocept Inc. (BIOC)
- New World Development (NWWDF)
- Grown Rogue (GRUSF)
- Cipher Pharmaceuticals (CPHRF)
- Fathom Nickel (FNICF)
- Ayr Wellness (AYRWF)
- Riley Exploration Permian Inc. (REPX)
- TerrAscend Corp. (TSNDF)
- Seelos Therapeutics Inc. (SEEL)
- Fisker Inc. (FSR)
- Freeport-McMoRan Inc. (FCX)
- Tilray Brands Inc. (TLRY)
Biocept Inc. (BIOC)
plrinvest, RedChip, BUYINS.NET, StockMarketWatch, MarketBeat, INO.com Market Report, MarketClub Analysis, Marketbeat.com, TraderPower, QualityStocks, Jason Bond, StreetInsider, SmallCapVoice, PoliticsAndMyPortfolio, Promotion Stock Secrets, SmallCapNetwork, StocksEarning, The Street, PennyPro, Trading Concepts, TradersPro, InvestorPlace, Top Pros' Top Picks, Joseph Green, Schaeffer's, The Online Investor, Weekly Wizards, StreetAuthority Daily, StockOodles, Stock News Now, Red Bull Stocks, AwesomeStocks, smartOTC and The Stock Dork reported earlier on Biocept Inc. (BIOC), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Biocept Inc. (NASDAQ: BIOC) (FRA: 8003) is an early stage molecular oncology diagnostics firm that is focused on the development and commercialization of circulating tumor nucleic acid and tumor cell assays, using a standard blood sample.
The firm has its headquarters in San Diego, California and was incorporated in 1997, on May 12th. It operates in the health care sector, under the biotech and pharma sub-industry and serves consumers and patients in California.
The company is party to a partnership agreement with CLEARED4 which entails the development of a system for managing and tracking coronavirus test results and testing requirements for its consumers. It markets its research and clinical trial services to clinical research organizations, biopharmaceutical firms and pharmaceutical firms, and sells its cancer diagnostic assays to oncologists and other physicians directly, at cancer centers, labs, hospitals and group and private practices in the U.S.
The enterprise’s cancer assays offer information to healthcare providers to identify alterations that qualify cancer patients for targeted therapy. Its assays for solid tumor indications include ovarian cancer, pancreatic cancer, melanoma, prostate cancer, colorectal cancer, gastric cancer, non-small cell lung cancer and breast cancer. The enterprise also provides lab services to pathologists, pulmonologists, urologists, surgical oncologists, neuro-oncologists and medical oncologists, as well as other physicians.
The firm was recently awarded a patent by South Korea for its Primer-switch technology, which identifies rare mutations in circulating tumor DNA through the use of real-time PCR. The product’s methodology adds to the company’s Target Selector assays, which help in physician decision-making. This move strengthens confidence in the company as well as its portfolio, which may trigger investor interest and bring more funds into the firm.
Biocept Inc. (BIOC), closed Friday's trading session at $1.34, up 59.4479%, on 103,239,842 volume. The average volume for the last 3 months is 9,433 and the stock's 52-week low/high is $0.80/$29.70.
New World Development (NWWDF)
We reported earlier on New World Development (NWWDF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
New World Development Company Limited (OTC: NWWDF) (OTC: NDVLY) (HKG: 0017) (BMV: 17N) (FRA: NWDA) is an investment holding firm that is focused on developing and investing in properties, providing contracting, property management and transportation services.
The firm has its headquarters in Central, Hong Kong and was incorporated in 1970, on May 29th. It operates as part of the real estate-diversified industry, under the real estate sector. The firm serves consumers in Hong Kong and Mainland China.
The enterprise’s property portfolio includes retail, residential, office and industrial properties. It also offers commercial aircraft leasing services; and constructs residential and commercial projects, as well as offers protection and savings-related life and medical insurance products. In addition to this, it is involved in the duty-free operation and general trading, civil engineering, loyalty program, marketing, promotion, fashion retailing and trading, and piling and ground investigation businesses; and development and operation of sports parks. Further, the enterprise manages Hong Kong convention and exhibition center; operates department stores; operates hotels, golf and tennis academies, and shopping malls; and offers information technology, healthcare, training courses, ticketing, financial, project management, management, advertising, business and investment consultancy, estate agency, wellness and rehabilitation, endoscopic, carpark management, supply chain management, and catering services, as well as property agency, management, and consultancy services.
The company was recently recognized with the "ESG Leading Enterprise Award 2023" from The Hong Kong General Chamber of Small and Medium Business, a move that may open it up to new growth and investment opportunities.
New World Development (NWWDF), closed Friday's trading session at $2.086, even for the day. The average volume for the last 3 months is 136,217 and the stock's 52-week low/high is $1.830529/$3.27.
Grown Rogue (GRUSF)
StocksEarning and Wealth Insider Alert reported earlier on Grown Rogue (GRUSF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Grown Rogue International Inc. (OTC: GRUSF) (CNSX: GRIN) is a craft marijuana firm that is focused on premium flower and flower-derived products.
The firm has its headquarters in Medford, Oregon and was incorporated in 2009, on November 30th. It operates as part of the drug manufacturers-specialty and generic industry, under the healthcare sector. The firm serves consumers around the globe.
The company’s mission is to bring low cost, high quality, craft cannabis from the amazing terroir and legacy of Oregon’s Rogue Valley to consumers nationwide. It operates a vertically integrated cannabis brand that produces and distributes cannabis products. The company’s subsidiaries include Grown Rogue Gardens LLC;Grown Rogue Unlimited LLC; GRIP LLC; GRU Properties LLC; GR Michigan LLC; Grown Rogue Distribution LLC; and others.
The enterprise operates approximately 40,000 square feet of flowering canopy in its three indoor facilities in Michigan and Oregon. It produces a range of cultivars for consumers, which are classified as sativas, indicas and hybrids. Its products include sungrown and indoor premium flower along with nitro-sealed indoor and sungrown pre-rolls and jars as well as cartridges and extracts, including shatter, wax, oil and sugar, which are categorized according to desired user experiences: relax, optimize, groove, uplift and energize.
The firm remains committed to accelerating their growth and entering into new growth and investment opportunities that will bring its quality flower to limited license markets. This may in turn bring in additional revenues and help create value for its shareholders.
Grown Rogue (GRUSF), closed Friday's trading session at $0.277, up 30.9693%, on 440,476 volume. The average volume for the last 3 months is 3,214 and the stock's 52-week low/high is $0.04/$0.277.
Cipher Pharmaceuticals (CPHRF)
We reported earlier on Cipher Pharmaceuticals (CPHRF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Cipher Pharmaceuticals Inc. (OTC: CPHRF) (TSE: CPH) (FRA: PHE) is a specialty pharmaceutical firm that is focused on the development of drugs for the skin.
The firm has its headquarters in Mississauga, Canada and was incorporated in 2000. It operates as part of the drug manufacturers-specialty and generic industry, under the healthcare sector. The firm primarily serves consumers in Canada.
The enterprise’s offerings include Actikerall, which is indicated for the treatment of palpable or moderately thick hyperkeratotic actinic keratosis; Epuris (isotretinoin), an oral retinoid indicated for the treatment of severe nodular and/or inflammatory acne, acne conglobate, and recalcitrant acne; Vaniqa, a topical cream for the slowing of the growth of unwanted facial hair in women; Ozanex for the topical treatment of impetigo; Brinavess for the rapid conversion of onset atrial fibriallation to sinus rhythm in adults; Aggrastat, a reversible GP IIb/IIIa inhibitor for use in patients with Acute Coronary Syndrome; and Durela, an opioid analgesic for the management of moderate to moderately severe pain in adults. The enterprise's licensed products include Lipofe indicated as adjunctive therapy to diet to reduce triglycerides in adult patients with severe hypertriglyceridemia; Absorica, an oral retinoid indicated for the treatment of severe recalcitrant nodular acne; and Conzip, an opioid agonist indicated for the management of moderate to moderately severe chronic pain in adults.
The company, which recently announced its latest financial results, remains committed to furthering its growth and generating additional value for its shareholders.
Cipher Pharmaceuticals (CPHRF), closed Friday's trading session at $3.1991, off by 3.213%, on 1,600 volume. The average volume for the last 3 months is 89,418 and the stock's 52-week low/high is $1.31/$5.375.
Fathom Nickel (FNICF)
InvestorIntel and AllPennyStocks reported earlier on Fathom Nickel (FNICF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Fathom Nickel Inc. (OTCQB: FNICF) (CNSX: FNI) (FRA: 6Q5) is a resource exploration and development firm that is focused on the identification, acquisition and exploration of base and precious metals for use in electric vehicle and battery markets.
The firm has its headquarters in Calgary, Canada and was incorporated in 2018, on November 23rd. It operates as part of the other industrial metals and mining industry, under the basic materials sector. The firm serves consumers in Canada.
The company primarily explores for nickel deposits. It targets magmatic nickel sulfide discoveries to support the rapidly growing global electric vehicle market. The company has a portfolio of two exploration projects located in the prolific Trans Hudson Corridor in Saskatchewan: Albert Lake Project and Gochager Lake Project. Its flagship project is the Albert Lake Project, which comprises of 31 mineral claims that cover an area of 90,460 hectares located in the La Ronge Mining District of Saskatchewan, Canada. Other properties in the Trans Hudson Corridor include Thompson Nickel Belt, Lynn Lake and Raglan Nickel Belt. The company also holds a 100% interest in Gochager Lake Property, which comprises of approximately nine mineral claims that cover approximately 4,696 hectares located in north-central Saskatchewan. The property is approximately 60km southwest of its Albert Lake Property.
The firm, which recently announced another drilling round for its Gochager Lake Project, remains committed to advancing its exploration efforts and creating additional value for its shareholders.
Fathom Nickel (FNICF), closed Friday's trading session at $0.222, up 5.7143%, on 120,630 volume. The average volume for the last 3 months is 204,720 and the stock's 52-week low/high is $0.0242/$0.2634.
Ayr Wellness (AYRWF)
MarketBeat, InvestorPlace, Daily Trade Alert, wyatt research newsletter and The Online Investor reported earlier on Ayr Wellness (AYRWF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Ayr Wellness Inc. (OTCQX: AYRWF) (CNSX: AYR.A) (FRA: 3KJB) is a vertically integrated cannabis multi-state operator which cultivates, manufactures and retails cannabis products and branded cannabis packaged goods.
The firm has its headquarters in Miami, Florida and was incorporated in 2017, on July 31st. Prior to its name change in February 2021, the firm was known as Ayr Strategies Inc. It operates as part of the drug manufacturers-specialty and generic industry, under the healthcare sector. The firm serves consumers across the globe.
The company’s mission is to cultivate the finest quality cannabis at scale and deliver remarkable experiences to its customers every day.
The enterprise operates through the cannabis sales segment. Its products include flower, edibles, vapes, beverages, concentrates and others. Its flower brands include TrailStix, Kynd and Road Tripper. Its vape brands include Origyn, Entourage and Secret Orchard. Its beverage brands include Levia and CannaPunch. Wicked and Secret Orchard are its edible brands. Haze is its concentrate brand. It delivers cannabis-infused drinks with zero calories and sugar. The enterprise’s retail dispensaries are located in Florida, Arizona, New Jersey, Illinois, Massachusetts, Nevada and Pennsylvania. It owns and operates a chain of cannabis retail stores under AYR, Liberty Health Sciences and The Dispensary brand names.
The firm recently released its latest financial results, with its CEO noting that they remained focused on positioning itself for sustainable long-term growth and profitability across all its markets, growing its footprint in Florida and improving its operations, a move that will in turn generate value for its shareholders.
Ayr Wellness (AYRWF), closed Friday's trading session at $2.79, up 9.4118%, on 739,241 volume. The average volume for the last 3 months is 63,825 and the stock's 52-week low/high is $0.5661/$4.41.
Riley Exploration Permian Inc. (REPX)
MarketBeat, InsiderTrades, The Wealth Report, StocksEarning, QualityStocks and FreeRealTime reported earlier on Riley Exploration Permian Inc. (REPX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Riley Exploration Permian (NASDAQ: REPX), an independent oil and natural gas company focused on steadily growing its reserves, production and cash flow, announced that it has entered into an Equity Distribution Agreement. According to the announcement, the company has inked agreements with Truist Securities Inc., Janney Montgomery Scott LLC and Roth Capital Partners LLC as sales agents. According to the agreement, the company may offer and sell shares of the company stock from time to time through the agents. The company may offer up to $50 million shares of stock, par value $0.001 per share. The company noted that the offer and sale of the shares has been registered according to regulations and was declared effective by the Securities and Exchange. ROTH MKM acted as lead agent for the offering.
To view the full press release, visit https://ibn.fm/ubItg
About Riley Exploration Permian Inc.
Riley Permian is an independent oil and natural gas company focused on steadily growing its reserves, production and cash flow per share through the acquisition, exploration, development and production of oil, natural gas, and natural gas liquids in the Permian Basin. For more information, visit the company website at www.RileyPermian.com.
Riley Exploration Permian Inc. (REPX), closed Friday's trading session at $33.03, off by 0.929814%, on 43,456 volume. The average volume for the last 3 months is 443,712 and the stock's 52-week low/high is $15.91/$47.7902.
TerrAscend Corp. (TSNDF)
We reported earlier on TerrAscend Corp. (TSNDF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Officials in California have announced the launch of a program to crack down on illegal cannabis operations in the state and boost the legal market. The program will connect Attorney General Rob Tonta’s office with local authorities and support them in efforts to combat the illicit marijuana market by targeting the finances of illegal cannabis operators. Tonta said that the anti-illicit cannabis program will deal with illegal marijuana as a land use and public nuisance issue.
Even though California is now home to the largest legal cannabis market on the entire globe, the state’s cannabis program has struggled to compete with the illegal market since its inception. Bonta noted that illicit marijuana is “problematic for many reasons” and said that constituents expect their leaders to team up and address the escalating issue of illegal cannabis.
Illegal operators regularly outsell California’s legal cannabis market, depriving the state of tax revenue and funneling funds into illegal operations. Furthermore, the illicit cannabis market has been implicated in illicit land use, significant water waste and environmental pollution. Cheaper cannabis from the black market has also made it incredibly difficult for licensed cannabis retailers to remain competitive as a myriad of fees and taxes makes legal cannabis significantly more expensive than black-market marijuana.
In addition, because illegal sellers aren’t subject to safety and quality controls, black-market cannabis can contain contaminants that endanger the health of unsuspecting cannabis users. In some cases, the criminal enterprises behind the illicit drug trade exploit and traffic their workers.
The new anti-illicit cannabis program will provide education and resources for enforcement programs and help collect evidence that could be instrumental in prosecuting large statewide cases. California’s Department of Justice will fund the program through administrative orders, fines, enforcement actions, lien, and settlements, allowing the program to be almost wholly self-funded. Fresno is the first city to join the program and will work with the justice department to help provide running costs.
A news release revealed that the statewide illicit cannabis enforcement efforts will supplement civil and criminal action by California Governor Gavin Newsom’s Unified Cannabis Enforcement Task Force and the state Department of Cannabis Control. According to Fresno City Attorney Andrew Janz, the city’s partnership with the attorney general’s office is meant to help boost Fresno’s legal cannabis industry and expand the city’s tax base. He hopes that other large cities in California will emulate Fresno’s joint venture and accelerate efforts to target the state’s massive cannabis black market.
It isn’t only California that is battling black-market operators. As multistate operators such as TerrAscend Corp. (TSX: TSND) (OTCQX: TSNDF) are all too aware, the challenge of illicit actors plagues all markets where marijuana is legal.
TerrAscend Corp. (TSNDF), closed Friday's trading session at $2.18, off by 0.909091%, on 908,321 volume. The average volume for the last 3 months is 918,556 and the stock's 52-week low/high is $1.00/$2.50.
Seelos Therapeutics Inc. (SEEL)
QualityStocks, MarketBeat, StockMarketWatch, MarketClub Analysis, TradersPro, Schaeffer's, StockEarnings, BUYINS.NET, Trades Of The Day and INO Market Report reported earlier on Seelos Therapeutics Inc. (SEEL), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
With Europe embarking on psychedelic reform and the potential legalization of therapeutic psychedelics, a lobby group comprised of industry players and developers says regulators should leverage knowledge from experienced psychedelic users to inform their decisions. In a document sent to the European Medicines Agency (EMA), the Psychedelic Access Research and European Alliance (PAREA) said the regional bloc needs a standard voice of authority for therapeutic psychedelic use, which includes experienced psychedelic users.
The lobby group called for authorities to create a multidisciplinary advisory body to provide professionals and regulators with guidance on best practices for psychedelic-assisted therapies amid the ongoing surge of psychedelic research. Several studies and clinical trials involving psychedelics such as LSD, MDMA and psilocybin (magic mushrooms) have revealed that psychedelics have the potential to offer sustained relief against several mental-health conditions with minimal adverse side effects.
For instance, psilocybin and ecstasy pills have shown positive results against conditions such as post-traumatic stress disorder (PTSD) and hard-to-treat depression, which don’t always respond to conventional mental health-treatments, including talk therapy and antidepressants. Scientists are also researching the potential of psilocybin, the main hallucinogenic agent in magic mushrooms, in the treatment of the eating disorder anorexia.
Although European regulators are just considering the possibility of therapeutic psychedelics, regulators in Australia and some parts of the United States have already legalized the prescription of psychedelic-based treatments to treat specific mental-health disorders. With some regulators calling for faster regulatory action on psychedelics in the European Union, PAREA asked the drug regulator to push for centralized leadership on the clinical and practical use of psychedelic-assisted therapies.
Psychedelics are expected to revolutionize the psychiatric industry over the next few decades, thanks to their therapeutic potential against typically hard-to-treat mental disorders such as PTSD and treatment-resistant depression. However, unlike traditional mental-health treatments, psychedelic-assisted therapy requires plenty of preparation, pre- and postcare, and guidance during the psychedelic trip. The lobby group wants regulators to ensure all this complementary care is standardized and has approval from multiple relevant groups.
According to PAREA’s briefing document to the European Medicine Agency, an advisory body would benefit from using knowledge from a wide variety of stakeholders. This includes psychedelic organizations, national competent authorities, physicians, drug developers, patient organizations and professional organizations such as Germany’s Mind Foundation the Netherlands’ Beckley Academy and the Open Foundation. Such groups use up-to-date research from institutions such as Johns Hopkins University in the U.S. and the Imperial College London to provide much-needed training to psychedelic therapists.
As efforts to bring about psychedelic policy reform ramp up around the world, industry actors such as Seelos Therapeutics Inc. (NASDAQ: SEEL) could see their operating environment become increasingly supportive when compared to the current regulatory climate.
Seelos Therapeutics Inc. (SEEL), closed Friday's trading session at $1.02, up 5.4264%, on 642,306 volume. The average volume for the last 3 months is 8.693M and the stock's 52-week low/high is $0.58/$1.66.
Fisker Inc. (FSR)
Schaeffer's, InvestorPlace, QualityStocks, StocksEarning, MarketBeat, MarketClub Analysis, Kiplinger Today, The Street, StockEarnings, The Online Investor, Early Bird, Daily Trade Alert, Trades Of The Day, Money Wealth Matters, TradersPro, Investopedia, CNBC Breaking News, INO Market Report, InsiderTrades, Louis Navellier, The Night Owl, Cabot Wealth, wyatt research newsletter, TipRanks, StreetInsider and InvestorsUnderground reported earlier on Fisker Inc. (FSR), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Argonne National Laboratory scientists have found a fluoride electrolyte that could potentially improve the range and performance of next-generation electric vehicle batteries by enhancing EV battery energy density and extending battery lifespan. Sodium fluoride is a fluorine compound that acts as a raw ingredient in many toothpaste brands, thanks to its ability to protect teeth from decay. A recent study involving the fluorine compound has uncovered previously unknown applications for fluorine-containing compounds that could revolutionize the nascent electric vehicle sector.
Electric vehicles represent the next era of vehicular transportation characterized by low to zero-emission transportation largely powered by clean energy. Most automakers have pledged to replace their vehicle lines with electric cars, and the U.S. government is investing billions of dollars to accelerate the growth of the country’s EV industry.
However, range anxiety is a major barrier to EV adoption for most drivers. Investing in widespread and reliable networks of charging infrastructure and developing efficient and energy-dense electric vehicle batteries can ultimately allow the EV industry to overcome this barrier. According to Argonne’s Chemical Sciences and Engineering division group leader, Zhengcheng (John) Zhang, a new era of EV batteries that surpass conventional lithium-ion designs is “on the horizon.”
The research team achieved this feat by replacing the electrolyte in conventional battery designs with a fluoride solvent that provides protection for hundreds of charging and discharging cycles. This new fluoride-based electrolyte allowed test battery cells to maintain their performance over hundreds of cycles. Nonlithium-ion batteries offer more than two times the energy density in relation to weight compared to lithium-ion batteries. Furthermore, they are much more efficient and can power electric cars for longer travel distances, opening the possibility of developing batteries powerful enough to run long-haul trucks and even aircraft.
Current electric vehicle batteries become inefficient after a certain weight as the additional weight cancels out any extra energy. Larger batteries are also more expensive to produce and would ultimately increase the cost of EVs. As such, automakers can’t add larger battery packs to their EV models to increase their range; they ideally need more energy-dense battery designs that can store more energy at the same or even less weight and at low costs.
Developing batteries for long-haul trucks and aircraft would significantly accelerate U.S. efforts to cut emissions from transportation. On top of higher energy density, nonlithium-ion battery designs are more resistant to the degradation caused by repeated cycles of charging and discharging. This allows the batteries to retain more of their range over time, preserving their performance for longer and giving them a longer lifespan.
Additionally, the fluoride electrolyte is cheaper to produce, more ecofriendly and resistant to fires compared to conventional lithium-ion batteries. Zhang says the battery design’s usefulness will “extend to other types of battery systems outside of lithium-ion.”
Given that the electric vehicle industry is still in its infancy, it is plausible to expect that as competition becomes tougher, startups such as Fisker Inc. (NYSE: FSR) will have to come up with significant innovations in battery technology and other aspects in order to stand a chance of surviving and thriving in an industry that is already crowded in some major markets.
Fisker Inc. (FSR), closed Friday's trading session at $6.24, up 0.160514%, on 5,587,852 volume. The average volume for the last 3 months is 10.943M and the stock's 52-week low/high is $4.265/$10.025.
Freeport-McMoRan Inc. (FCX)
MarketClub Analysis, The Street, InvestorPlace, Schaeffer's, Kiplinger Today, SmarTrend Newsletters, StocksEarning, MarketBeat, Barchart, StreetAuthority Daily, Trades Of The Day, StreetInsider, Investopedia, Money Morning, TopStockAnalysts, Daily Trade Alert, Zacks, QualityStocks, Louis Navellier, VectorVest, Trading Markets, Marketbeat.com, ProfitableTrading, The Growth Stock Wire, Daily Wealth, The Wealth Report, Top Pros' Top Picks, The Online Investor, TheStockAdvisors, StreetAlerts, Trading Tips, Wall Street Elite, All about trends, Streetwise Reports, TradingMarkets, Leeb's Market Forecast, StockEarnings, Market FN, DividendStocks, Early Bird, Dividend Opportunities, INO.com Market Report, TheStockAdvisor, Investors Alley, StockTwits, InvestmentHouse, Investor Guide, Investment House, Wealth Insider Alert, Trading Concepts, INO Market Report, InvestorGuide, Cabot Wealth, Money and Markets, Options Trader Elite, The Best Newsletters, Market Intelligence Center Alert, Energy and Capital, The Motley Fool, Darwin Investing Network, Bourbon and Bayonets, Trade of the Week, AnotherWinningTrade, Wealth Daily, Investing Signal, Wyatt Investment Research, Buttonwood Research, MarketWatch, Market Authority, Investor Update, Super Stock Investor, Wealthpire Inc., Wall Street Daily, TradingAuthority Daily, Stock Research Newsletter, Profit Confidential, Street Insider, WStreet Market Commentary, Weekly Wizards, Uncommon Wisdom, TradersPro, AllPennyStocks, InvestorsUnderground, CNBC Breaking News, Daily Markets, MiningNewsWire, FeedBlitz, FNNO Newsletters, Rick Saddler, 24/7 Trader, Dynamic Wealth Report, Normandy Investment Research, Inside Investing Daily, Oakshire Financial, SmallCapVoice, Investment U, The Tycoon Report, Investiv, Profits Run, Investing Daily, Inside Trading, Weekly Market Strategies, StreetAuthority Investor Update, Navellier Growth, Pennybuster, BUYINS.NET, ChartAdvisor, Stock Barometer, Insider Wealth Alert, Market Intelligence Center, Daily Profit, Stockhouse, Greenbackers, Hit and Run Candle Sticks, Forbes, Average Joe Options, Stock Beast, TradingPub, Traders.com, Trader Prep, The Street Report, The Stock Dork, The Night Owl, The Daily Market Alert, Terry's Tips, Taipan Daily, Super Stock Picker, StreetAuthority Financial, Stocks To Watch, StockRockandRoll, Penny Sleuth, Money Wealth Matters, InvestorIntel, Investors Daily Edge, Investors Underground, Jason Bond, MarketTamer, StockMarketWatch, Milestone Capital Growth Portfolio, StockLockandLoad, Penny Stock Buzz, PowerRatings Stocks, Schaeffer’s, Shah's Insights & Indictments, SmallCap Network, InsiderTrades and MicroCap Gems reported earlier on Freeport-McMoRan Inc. (FCX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The past couple of years have been a rollercoaster for gold. Inflationary pressures should have caused gold prices to surge, but consistent interest rate hikes by the U.S. Federal Reserve kept investor attention on other interest-paying assets. And then, in mid-2022, a record gold purchase run by central banks around the world caused gold prices to spike and hit record highs. As central banks reigned in their gold purchases, demand for the precious metal reduced significantly and prices dropped to a five-month low in mid-August.
Despite its relatively volatile performance in recent years, adding gold to your investment portfolio in 2023 may still be a good move. Investors and experts remain unsure of the economy’s direction even as the year draws to a close, adding a degree of uncertainty regarding the market’s performance in the short term.
Although inflation in the United States and other Western countries has cooled significantly from the highs of 2022, an unexpected increase in July is forcing consumers to spend more. Investing in gold will add some stability to your investment portfolio and protect your finances from an uncertain and evolving economy. Gold is especially great at stabilizing portfolios against shifting economic winds during times of uncertainty, such as the one we are currently experiencing.
The yellow metal has long been a safe-haven asset capable of protecting finances from depreciation during recessions and inflation. Although gold may have lost some of its safe-haven appeal in recent years, it is still a great way for investors to stabilize their portfolios and somewhat fortify them against inflationary pressures. Gold is one of the few assets capable of retaining its value through economic upheavals, making it a good hedge against inflation that can preserve investors’ purchasing power over the long-term despite shifts in the greenback’s value.
Battle Bank president Frank Trotter said high inflation periods are an “excellent time” to increase gold allocations. He notes that long-term analysis has consistently found gold to be a decent hedge against inflation. Gold will also allow you to diversify your portfolio, reduce your exposure to risky assets and reduce any losses you may sustain as your other investments lose value, he said. Trotter explains that with a possible recession and even stagflation in the offing, investors can reduce their stock and bond exposure by reallocating some of their resources to gold.
Finally, the precious metal is incredibly liquid compared to assets such as real estate, collectibles and stocks, which can be great investments but are difficult to quickly convert into usable cash. Investors with a quick need for cash will find it easy to quickly offload their gold.
If you would like to diversify your portfolio, add stability, and retain liquidity, you should consider investing in gold.
If you are more inclined to buy gold company shares, then you should do extensive due diligence on extraction companies such as Freeport-McMoRan Inc. (NYSE: FCX) and invest in those that satisfy your requirements.
Freeport-McMoRan Inc. (FCX), closed Friday's trading session at $39.29, off by 1.1821%, on 6,337,304 volume. The average volume for the last 3 months is 27.769M and the stock's 52-week low/high is $26.033/$46.73.
Tilray Brands Inc. (TLRY)
Schaeffer's, InvestorPlace, The Street, StocksEarning, MarketClub Analysis, MarketBeat, StockEarnings, Trades Of The Day, QualityStocks, Daily Trade Alert, StockMarketWatch, Kiplinger Today, StreetInsider, The Online Investor, Wealth Insider Alert, Market Intelligence Center Alert, BUYINS.NET, Investopedia, Zacks, CFN Media Group, CNBC Breaking News, The Street Report, Daily Profit, INO Market Report, StreetAuthority Daily, Early Bird, Inside Trading, The Rich Investor, Tip.us, Top Pros' Top Picks, InvestmentHouse, Trading For Keeps, Trading Concepts, FreeRealTime, Eagle Financial Publications, InvestorsObserver Team, Investors Alley, Investment House, Outsider Club, wyatt research newsletter, Wealth Daily, VectorVest, TheTradingReport, The Night Owl, StrategicTechInvestor, Money Morning, Rick Saddler, InvestorsUnderground, AllPennyStocks, MarketClub, Marketbeat.com, Louis Navellier, Jim Cramer, Jason Bond and Stock Up Featured reported earlier on Tilray Brands Inc. (TLRY), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The rollout of recreational cannabis in New York has faced yet another hurdle as a judge from the state’s Supreme Court has put a halt on all applications for business licenses to establish adult-use marijuana dispensaries. The judge cited inconsistent and unclear information from regulating officials. This recent setback adds to the challenges that have characterized the launch of legalized marijuana in New York.
The issue revolves around a program called the Conditional Adult Use Retail Dispensary (CAURD), managed by the Office of Cannabis Management (OCM). When the state legalized marijuana in March 2021, lawmakers and cannabis regulators initially announced that a portion of retail licenses would be reserved for small nonprofit organizations and individuals who had been adversely affected by the war on drugs.
This exclusion left out several potential marijuana businesses, including those with existing medical cannabis licenses, often owned by larger multistate operators. Some of these operators took legal action against the state earlier this year. Additionally, a group of service-disabled military veterans filed a lawsuit against the state, arguing that the CAURD program violates their rights and is unconstitutional because they were not eligible for permits.
As a response to this lawsuit, Judge Kevin Bryant, presiding over the State Supreme Court, issued an injunction on Aug. 7, 2023, which temporarily suspended all CAURD applications. This injunction was subsequently upheld on Aug. 18. While the OCM has granted licenses to a few hundred emerging CAURD businesses, records from the agency indicate that only 23 legal marijuana retail stores are operational in the state. Interestingly, more than 1,500 unlicensed cannabis retailers are currently operating exclusively in New York City.
Last week, a glimmer of hope emerged for certain applicants when the state released a list of 30 CAURD applicants that had made significant progress in the application process and were thus exempt from Judge Bryant’s order. However, in a subsequent ruling last week, Bryant noted that information from the OCM indicated that not all 30 applicants had fulfilled the necessary licensing prerequisites. As a result, he directed OCM to provide an updated list of potential injunction-exempt applicants under oath, planning a comprehensive, individualized order.
This series of events has taken many by surprise and left both applicants and observers feeling perplexed. Osbert Orduña, CEO of the Cannabis Place, which is in the process of applying for a CAURD license to operate in Queens, expressed disappointment in the recent developments. He emphasized that this setback has negative repercussions for small businesses, potential employees and cannabis consumers in New York — all affected by a lawsuit initiated by a small group of individuals.
These regulatory hiccups can be massively disruptive, and even established companies such as Tilray Brands Inc. (NASDAQ: TLRY) (TSX: TLRY) may possibly reconsider staying in a market that doesn’t have a reliable regulatory regime guiding operators, creating uncertainty about the stability of the rules under which they operate.
Tilray Brands Inc. (TLRY), closed Friday's trading session at $3.02, up 2.027%, on 39,800,847 volume. The average volume for the last 3 months is 122,624 and the stock's 52-week low/high is $1.50/$5.12.
The QualityStocks Company Corner
- Longeveron Inc. (NASDAQ: LGVN)
- NextPlat Corp. (NASDAQ: NXPLW) (NASDAQ: NXPL)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI)
- GolfLync Inc.
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
- Knightscope Inc. (NASDAQ: KSCP)
- GEMXX Corp. (OTC: GEMZ)
- RVL Pharmaceuticals plc (NASDAQ: RVLP)
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF)
- Renovaro BioSciences Inc. (NASDAQ: RENB)
Longeveron Inc. (NASDAQ: LGVN)
The QualityStocks Daily Newsletter would like to spotlight Longeveron Inc. (NASDAQ: LGVN) .
In June 2023, Longeveron filed with the SEC to offer subscription rights worth up to $30 million of shares of Class A common stock to stockholders and holders of warrants. The rights offering, which became effective on August 17, 2023, is being made through a distribution of five tradable subscription rights (listed on The NASDAQ Capital Market under the ticker symbol LGVNR) to purchase shares of Class A common stock for each share of common stock and warrant to purchase common stock owned as of the record date of August 18, 2023, at a $3.00 subscription price per share. The subscription period opened on August 22, 2023, and will continue until September 21, 2023, unless extended at the company's sole discretion.
During Q2 2023, the company announced children in its ELPIS I Trial of Lomecel-B(TM) for HLHS had 100% survival up to five years of age after receiving the drug, as compared with an approximate 80% survival rate in historical trials. Lomecel-B(TM) for HLHS has previously received Fast Track Designation, Rare Pediatric Disease Designation, and Orphan Drug Designation from the U.S. Food and Drug Administration ("FDA").
In August 2023, the company announced the Phase 2 ELPIS II trial, funded by the National Heart, Lung, and Blood Institute ("NHLBI") and using the company's Lomecel-B(TM) asset in Hypoplastic Left Heart Syndrome ("HLHS"), has surpassed the 50% enrollment threshold. Additionally, the trial has activated its eighth clinical site location – one additional site beyond the seven originally planned – in order to try to expedite the completion of the trial enrollment.
The company expects to share top-line results from its Phase 2a CLEAR MIND trial of Lomecel-B(TM) for the treatment of Alzheimer's disease in early October 2023.
Longeveron is conducting a Phase 2 study of Lomecel-B in patients with Aging-related Frailty in conjunction with Japan's National Center for Geriatrics & Gerontology and Tokyo's Juntendo University Hospital.
Longeveron Inc. (NASDAQ: LGVN) is a clinical-stage biotechnology company developing regenerative medicines to address unmet medical needs for specific aging-related and life-threatening conditions. The Company’s research and therapies are aimed at improving the outcome of infants born with a life-threatening heart condition, as well as improving the healthspan for the aging population – the number of years a person is expected to live in relatively good health, free of chronic disease and disabilities of aging, with function and ability to perform activities of daily living.
Longeveron is involved in clinical trials in the following indications: Hypoplastic left heart syndrome (HLHS), Alzheimer’s disease, and Aging-related Frailty.
The Company’s philosophy revolves around the idea that regenerative medicine may hold the potential to improve certain rare medical conditions and contribute to healthy aging. While there has been a remarkable rise in life expectancy over the last century due to medical and public health advancements, this increase in longevity has not been paralleled by the number of years a person is expected to live in relatively good health, free of chronic disease and disabilities of aging.
Longeveron’s lead investigational product is Lomecel-B™, an allogeneic Medicinal Signaling Cell therapy product isolated from the bone marrow of young, healthy adult donors. As humans age, they experience a decrease in immune system function, a decline in blood vessel functioning, chronic inflammation, and other issues. Clinical data has suggested that Lomecel-B™ may address these conditions through multiple mechanisms of action (MOA) that simultaneously target key aging-related processes.
The Company is headquartered in Miami, Florida.
Lomecel-B™
Lomecel-B™ is being evaluated in multiple clinical trials for aging-related chronic diseases and other life-threatening conditions under U.S. FDA-approved Investigational New Drug applications. Lomecel-B™ has multiple potential mechanisms of action encompassing pro-vascular, pro-regenerative, anti-inflammatory, and tissue repair and healing effects with broad potential applications across a spectrum of disease areas.
The drug is made from special living cells called Medicinal Signaling Cells (MSCs) that are isolated from fresh bone marrow tissue that has been donated by adult donors aged 18 to 45. Once the MSCs have been isolated from the fresh bone marrow through a careful selection process, the cells are culture-expanded (allowed to replicate under controlled laboratory conditions) into the billions using specialized techniques and processes. After a specific number of expansion cycles, called “passages,” the cells are harvested, separated into specific doses (e.g., 50 million cells), and cryopreserved until future use.
These cells have been shown to have characteristics that allow them to be transplanted from a donor to host without triggering a harmful immune response in the recipient, and they can be administered on an outpatient basis in as little as 40 minutes after thawing. Because of these characteristics, Lomecel-B™ is considered an “off-the-shelf” product.
In some trials, such as for Alzheimer’s disease and Aging-related Frailty, Lomecel-B™ is administered via peripheral intravenous infusion, while, in the Company’s HLHS trial, Lomecel-B™ is administered via direct injection into the heart tissue.
Market Opportunity
Longeveron estimates the potential market size for Lomecel-B™ in the treatment of HLHS to be up to $1 billion annually, globally.
U.S. patients suffering from Aging-related Frailty are estimated using U.S. Census Bureau statistics to be approximately 8.1 million. That population potentially represents a market for Lomecel-B™ of between $4 billion and $8 billion globally per year, according to Company estimates.
Additionally, the Alzheimer’s Association puts the number of Americans with that disease at 5.1 million, highlighting another potentially addressable market for Lomecel-B™, that’s worth $5 billion to $10 billion annually.
Management Team
Wa’el Hashad is CEO of Longeveron. He has more than 35 years of experience in the pharmaceutical and biotech industries. He has launched several successful brands in the U.S. and worldwide markets. Prior to joining Longeveron, he was president and CEO of Avanir Pharmaceuticals. Before Avanir, he was the chief commercial officer of Seres Therapeutics. He also has held senior leadership positions at Amgen, Boehringer Ingelheim, and Eli Lilly and Company. He holds a bachelor’s degree in pharmacy from Cairo University and an MBA from the University of Akron.
Joshua M. Hare, M.D., FACC, FAHA, is Co-Founder, Chief Science Officer and Chairman of Longeveron. He is a double board-certified cardiologist and is the founding director of the Interdisciplinary Stem Cell Institute at the University of Miami’s Miller School of Medicine. He is a recipient of the Paul Beeson Physician Faculty Scholar in Aging Research Award and is an elected member of the American Association of Physicians and The American Society for Clinical Investigation. He is also an elected Fellow of the American Heart Association. He received a bachelor’s degree from the University of Pennsylvania and his M.D. from The Johns Hopkins University School of Medicine.
Lisa Locklear is CFO at Longeveron. She previously served as the senior vice president and CFO for Avanir Pharmaceuticals. Prior to Avanir, she held senior financial roles at GSN Games, CoreLogic, Ingram Micro, the Walt Disney Company, and Price Waterhouse, with assignments in Paris and London. She holds a bachelor’s degree in plant science from the University of California, Davis, and an MBA from the University of California, Irvine. She is a licensed CPA (inactive) and is a member of the American Institute of Certified Public Accountants, the California Society of CPAs, and Financial Executives International.
Dr. Nataliya Agafonova, M.D., is the Chief Medical Officer at Longeveron. She previously served as clinical development lead, senior medical director, and product development chair at Otsuka Pharmaceuticals. Before that, she was the clinical development lead and senior medical director at Bristol-Myers Squibb. She previously held senior leadership positions at Ardea Bioscience, Biogen, Amgen, and Genzyme Corporation. She earned an M.D. from the Ukrainian National Medical University and completed her internal medicine residency at Kharkov State University Hospital in Ukraine.
Certain statements in this corporate profile that are not historical facts are forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, which reflect management’s current expectations, assumptions, and estimates of future operations, performance and economic conditions, and involve risks and uncertainties that could cause actual results to differ materially from those anticipated by the statements made herein. Forward-looking statements are generally identifiable by the use of forward-looking terminology such as “believe,” “expects,” “may,” “looks to,” “will,” “should,” “plan,” “intend,” “on condition,” “target,” “see,” “potential,” “estimates,” “preliminary,” or “anticipates” or the negative thereof or comparable terminology, or by discussion of strategy or goals or other future events, circumstances, or effects. Factors that could cause actual results to differ materially from those expressed or implied in any forward-looking statements in this release include, but are not limited to, statements regarding the offer and sale of securities, the terms of the offering, about the ability of Longeveron’s clinical trials to demonstrate safety and efficacy of the Company’s product candidates, and other positive results; the timing and focus of the Company’s ongoing and future preclinical studies and clinical trials and the reporting of data from those studies and trials; the size of the market opportunity for the Company’s product candidates, including its estimates of the number of patients who suffer from the diseases being targeted; the success of competing therapies that are or may become available; the beneficial characteristics, safety, efficacy and therapeutic effects of the Company’s product candidates; the Company’s ability to obtain and maintain regulatory approval of its product candidates in the U.S., Japan and other jurisdictions; the Company’s plans relating to the further development of its product candidates, including additional disease states or indications it may pursue; the Company’s plans and ability to obtain or protect intellectual property rights, including extensions of existing patent terms where available and its ability to avoid infringing the intellectual property rights of others; the need to hire additional personnel and the Company’s ability to attract and retain such personnel; the Company’s estimates regarding expenses, future revenue, capital requirements and needs for additional financing; the Company’s need to raise additional capital, and the difficulties it may face in obtaining access to capital, and the dilutive impact it may have on its investors; the Company’s financial performance and ability to continue as a going concern, and the period over which it estimates its existing cash and cash equivalents will be sufficient to fund its future operating expenses and capital expenditure requirements. Additionally, Longeveron makes no assurance that any public offering of its securities as described herein will occur on the timelines, in the manner or on the terms anticipated due to numerous factors. Further information relating to factors that may impact the Company’s results and forward-looking statements are disclosed in the Company’s filings with the Securities and Exchange Commission, including Longeveron’s Annual Report on Form 10-K for the year ended December 31, 2022, filed with the Securities and Exchange Commission on March 14, 2023 and its Quarterly Report on Form 10-Q for the second quarter of 2023 filed with the SEC on August 11, 2023. The forward-looking statements contained in this corporate profile are made as of the date of this corporate profile, and the Company disclaims any intention or obligation, other than imposed by law, to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise.
Investor Contact
Mike Moyer
LifeSci Advisors
Tel: 617-308-4306
Email: [email protected]
Date prepared: August 31, 2023
Longeveron Inc. (NASDAQ: LGVN), closed Friday's trading session at $2.41, up 0.416667%, on 95,711 volume. The average volume for the last 3 months is 19,616 and the stock's 52-week low/high is $2.2302/$5.22.
Recent News
- Longeveron Inc. (NASDAQ: LGVN) - Longeveron Inc. (NASDAQ: LGVN) Is 'One to Watch'
- InvestorNewsBreaks - Longeveron Inc. (NASDAQ: LGVN; LGVNR) Announces ELPIS 1 Trial Data Accepted for AHA Presentation, IBN Partnership
- InvestorNewsBreaks - Longeveron Inc. (NASDAQ: LGVN) to Present at Upcoming H.C. Wainwright Global Investment Conference
NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW)
The QualityStocks Daily Newsletter would like to spotlight NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW).
E-commerce companies have a greater chance of succeeding in an increasingly competitive and cutthroat market when they are clear on value proposition. Consumer retail surged during the coronavirus pandemic as nearly a hundred countries instituted self-isolation orders and locked down one-half the global population, forcing countless consumers to rely solely on online stores for their purchases. Convenience was the key drawing factor for most consumers and remains a big factor in the attractiveness of online commerce.
According to a Catalyst/Kantar study, 66% of consumers used convenience as their main criteria for choosing online stores while 57% valued shoppability, 52% used product assortment as their criteria and 47% cited price. However, with consumers around the world facing significant increases in the cost of living, this year's Australian E-Commerce Report found that consumers are focusing more and more on loyalty programs and discounts and looking for price comparisons to minimize their online shopping costs.
Years of the coronavirus pandemic, an ongoing energy crisis and the reverberating effects of the Russia-Ukraine war have left the global economy battered, increasing costs almost across the board and forcing consumers to dig deeper into their pockets to survive. Online retailers can attract these cost-conscious online buyers by increasingly investing in value proposition and tailoring the online shopping experience to match current economic conditions. As more players such as NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) enter specialized segments of e-commerce such as healthcare e-commerce, we could see more targeted forms of value proposition fronted by companies intent on growing market share.
NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW), a next generation e-commerce platform, was created with vision and purpose to capitalize on high growth sectors and global markets. The company collaborates with businesses – large and small – to simplify and accelerate online commerce and uniquely enables customers and partners to optimize their e-commerce reach, presence and revenue. NextPlat recently launched a new e-commerce development program to provide American businesses with easy access to the massive Chinese consumer market.
Current Initiatives
NextPlat provides cutting edge technology in an advanced e-commerce ecosystem. The company is actively expanding its global network of online storefronts serving thousands of consumers, enterprises and governments. The company also has developed a next generation platform built for Web3 that enables the creation and sale of digital assets, as well as optimizing e-commerce transactions and business building activities. The company’s current initiatives include:
- E-Commerce Development Program – In April 2023, NextPlat announced it had entered into a merchant sourcing agreement with Alibaba.com Singapore E-Commerce Private Limited (“Alibaba”) and its Tmall Global e-commerce platform whereby the two companies will collaborate to increase the sale of products produced and sold by American companies to the multi-trillion-dollar Chinese consumer market. Alibaba’s Tmall Global e-commerce platform will provide NextPlat customers a turn-key solution through which products can be sold to the Chinese consumer market. The launch of the Florida E-Commerce Development Program is the first in a series of new NextPlat programs designed to assist U.S. businesses in expanding their online sales capabilities to reach new international customers in the Chinese market. NextPlat intends to rapidly expand this unique e-commerce development opportunity to businesses throughout the United States and all of North America, as well as Central and South America. The new development program features NextPlat’s turnkey global e-commerce solution for customers and leverages NextPlat’s relationships with key partners, including Tmall Global, China’s largest cross-border B2C online marketplace.
- Progressive Care Inc. – In August 2022, NextPlat completed a strategic $7 million investment in Progressive Care Inc. (OTCQB: RXMD), a personalized health care services and technology company. In a news release announcing the investment, NextPlat CEO Charles M. Fernandez noted that the company is “committed to harnessing the power of digital technologies to capitalize on the ongoing digital transformation of Progressive Care and the entire health care industry.” NextPlat intends to accelerate Progressive Care’s digital health care transformation with the launch of a new e-commerce platform for health care products later this year.
- NextPlat NFT Platform – Building on its existing e-commerce initiatives, NextPlat is working to bridge the gap between tangible and digital e-commerce marketplaces by incorporating burgeoning Web3 technologies. The company intends to launch a fully integrated NFT platform in the coming months that will enable brands to create, manage and authenticate digital assets while serving as a new source of revenue for NextPlat. Through this model, the company will receive a portion of the revenue generated from branded NFT drops, as well as subsequent secondary market transactions.
- Global Telesat Communications and Orbital SatCom Corp. – Targeting both domestic and international markets, NextPlat’s subsidiaries leverage partnerships with major e-commerce platforms such as Amazon, Alibaba, eBay and Walmart to serve a growing base that includes more than 50,000 corporate, governmental and individual customers. In total, the brands market more than 10,000 individual products, with a focus on satellite-based connectivity solutions. In addition to exploring accretive M&A opportunities, NextPlat aims to diversify its range of products and broaden its geographic footprint moving forward in an effort to better capitalize on the tremendous growth potential in the United States, Europe and Asia.
“Our goal for 2023 and beyond is to leverage our improved operational capabilities and enhanced leadership team as we expand our offerings in communications and connectivity into the high-growth health care market where we intend to launch an array of innovative new offerings,” Fernandez said in a March 2023 news release detailing the company’s record top-line performance. “Although there remain supply chain headwinds and the challenge of global inflation, we are confident that we have the right combination of market-tested expertise, technology and partnerships that will enable us to bring the power of e-commerce to more customers, brands and industries in the United States and abroad.”
Market Opportunity
The rapid growth of e-commerce over the last decade is expected to continue for the foreseeable future. According to data published by Forbes, roughly 20.8% of all retail purchases are expected to take place online in 2023, accounting for total sales of $6.31 trillion worldwide. It total, e-commerce sales are expected to grow by 10.4% YoY in 2023, accounting for a whopping 24% of all retail purchases by 2026.
For NextPlat, existing partnerships in the industry could be key to capitalizing on this growth. The Forbes report indicates that Amazon accounts for roughly 38.7% of e-commerce sales, while sites like Walmart, eBay and Alibaba round out the list of most visited e-commerce websites. Alibaba is especially interesting due to NextPlat’s recent strategic merchant sourcing agreement with Tmall Global. The Chinese market is “mammoth,” as a recent Alizila report noted. The country’s annual online retail sales of physical goods have nearly doubled in the last five years, reaching approximately 13.8 trillion yuan in 2022, which is nearly $2 trillion USD.
The health care portion of the e-commerce market is generating particularly bullish forecasts, bolstered by the continued adoption of the 340B Drug Pricing Program in the U.S., which requires most drug manufacturers to provide outpatient drugs to covered entities at significantly reduced prices. Industry reports suggest that the global health care e-commerce market will expand at a compound annual growth rate of 16.8% from 2022 to 2030, climbing to a value of more than $1.37 trillion by the end of the forecast period.
Management Team
Charles M. Fernandez, CEO, Executive Chairman and Director of NextPlat, has over three decades of experience in identifying profitable start-up and dislocation opportunities, building significant value and executing exit strategies as an entrepreneur and global investor. Successful across multiple sectors, Fortune Magazine actually labeled Fernandez ‘a restructuring whiz’. As President of Fairholme Capital Management, which he joined in 2008, Mr. Fernandez co-managed all three Fairholme funds and brought in a $2 billion gain for shareholders. Throughout his impressive career, he has participated in more than 100 significant mergers, acquisitions and product development projects across multiple industries. Mr. Fernandez was the founder, Chairman and CEO of eApeiron Solutions LLC, a brand protection and e-commerce company in partnership with Alibaba (NYSE: BABA) and Eastman Kodak (NYSE: KODK), which was successfully sold to Smartrac, a unit of Avery Dennison Corp. (NYSE: AVY).
Rodney Barreto is Chairman and CEO of the Barreto Group and Director of Nextplat. Mr. Barreto’s business career spans over 35 years, including his role at the Barreto Group and, earlier, as the founding partner of Floridian Partners LLC, a corporate and public affairs consulting firm recognized by policy makers as one of the top in its industry in Florida. He chaired the Super Bowl Host Committee in 2007, 2010 and 2020, helping to raise more than $100 million for the success of Miami Super Bowls. As a philanthropist and conservationist, Mr. Barreto is also a three-time appointee to the Florida Fish and Wildlife Conservation Commission, where he has served for over 10 years including holding the title of Chairman eight times. He has twice chaired the Annual U.S. Conference of Mayors, was Chairman of the 1999 Breeder’s Cup Championship held in South Florida and was the Chairman of the 1999 Sister Cities International Convention in Miami. Currently, Mr. Barreto is the Membership Chairman of the Florida Council of 100, and a member of the Boards of Fairchild Tropical Botanic Garden, the Baptist Health South Florida Giving Society, the Bonefish and Tarpon Trust, the Guy Harvey Ocean Foundation, and a member of Miami Dade County Schools Superintendent Carvalho’s Business Advisory Council. Prior to his career in public affairs and real estate, Mr. Barreto was a City of Miami police officer and is a member of the Florida Highway Patrol Advisory Council.
NextPlat Corp. (NXPL), closed Friday's trading session at $2.16, up 1.4085%, on 13,831 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $5.22/$.
Recent News
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Being Clear on Value Proposition Key to eCommerce Success
- Bloomreach Unveils Conversational AI Tool for Online Shoppers
- Blockchain Could Help Improve Security, Transparency in Healthcare eCommerce
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
Lexaria Bioscience Corp. (NASDAQ: LEXX), a global innovator in drug delivery platforms, recently celebrated the receipt of U.S. patent #11,700,875, titled "Compositions and Methods For Sublingual Delivery of Nicotine." The patent covers various forms and sources of nicotine, including nicotine benzoate, nicotine polacrilex, nicotine citrate, nicotine detartrate, and many others, for use in sublingual delivery formats like oral pouches. "The new U.S. patent is remarkable and ‘strategically important' in many ways. First, it is the company's 35th overall granted patent worldwide and the 13th granted in the United States. Second, a total of 30 claims were awarded within this patent, the highest number of claims accepted by the USPTO in the company's existing portfolio. And with researchers agreeing that patents with a higher number of claims are better protected and can be an important measure of value, the claims increase the value of the new patent and perhaps even the company's entire IP portfolio," a recent article reads. "Third… the new U.S. patent could lead to the award of patents within this family in other countries… Moreover, the new patent grants IP protection within the United States, one of the world's leading oral nicotine markets, according to an analysis by the Foundation for a Smoke-Free World. The U.S. nicotine pouches market is projected to grow at a 33% CAGR between 2023 and 2030."
To view the full article, visit https://cnw.fm/ymT0t
Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing.
DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future.
Lexaria has also collaborated with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has been granted patent protection for specific delivery of nicotine, vitamins, NSAIDs, antiviral drugs, cannabinoids and more.
Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats.
The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products.
DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions.
DehydraTECH Technology
Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following:
- Speeds up delivery – the effects of the product are felt by the subject in just minutes.
- Increases bioavailability – the technology is much more effective at delivering a drug or product into the bloodstream.
- Increases brain absorption – animal testing suggests significant improvement in the quantity of drug delivered across the blood-brain barrier.
- Improves drug potency – more of the ingested product is made available to the body, so lower doses are required to achieve the desired effect.
- Reduces drug administration cost – lower doses mean lower overall drug costs.
- Masks unwanted taste – the technology eliminates or reduces the need for sweeteners.
Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery.
Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine.
Market Outlook
Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments:
- Oral Cannabinoids – a market estimated to be worth $18.4 billion in 2021 and expected to reach $46.2 billion by 2025.
- Antivirals – an estimated $52.1 billion market in 2021 that’s expected to grow to $66.7 billion by 2025.
- Oral Mucosal Nicotine – smokeless tobacco products, a $13.6 billion market in 2018, is forecast to grow at 7.2 percent annually through 2025.
- Human Hormones – estrogen and testosterone replacement therapies represented a $21.9 billion market in 2019, with a forecast CAGR of 7.7 percent through 2027.
- Ibuprofen and Naproxen – NSAID sales totaled $15.6 billion globally in 2019 and are projected to reach $24.4 billion by 2027.
- Vitamin D3 – the global market size was $1.1 billion in 2021, growing at 7 percent per year and expected to reach $1.7 billion in 2026.
Management Team
Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations.
John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents.
Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations.
Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch.
Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia.
Lexaria Bioscience Corp. (LEXX), closed Friday's trading session at $0.8901, up 3.1402%, on 16,309 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.6488/$3.5953.
Recent News
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - CBDNewsBreaks - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) 'Strategically Important' Patent Grants Protection in One of World's Leading Oral Nicotine Markets
- Lexaria Bioscience Corp. (NASDAQ: LEXX) Eyes What Is Considered One Of America's Most Serious Health Problems
- InvestorNewsBreaks - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces Update on IND Application Progress
Lucy Scientific Discovery Inc. (NASDAQ: LSDI)
The QualityStocks Daily Newsletter would like to spotlight Lucy Scientific Discovery Inc. (NASDAQ: LSDI).
In July 2023, LSDI launched Twilight, a new sleep aid product
The company in July 2023 announced its acquisition of SANA-013 from Wesana Health; SANA-013 is being developed for the treatment of several mental health and central nervous system conditions
LSDI in May 2023 launched its new Mindful product line designed to enhance well-being and promote a mindful approach to life
Also in May 2023, the company announced a partnership with non-profit TheraPsil to advance medical psilocybin access and research
LSDI announced in April 2023 that its board had approved the repurchase of up to 500,000 shares of its Class A common stock
Lucy Scientific Discovery (NASDAQ: LSDI) is featured in a recent FinancialNewsMedia ("FNM") Editorial. The editorial provides an overview of LSDI's position in a global cannabis market projected to hit $51 billion this year. According to the press release, the global cannabis market has seen consistent growth year after year, with increasing legalization of cannabis fueling that growth. The release notes that a recent Statista report forecast the global cannabis market to reach $51.27 billion in 2023 with revenue expected to show an annual growth rate ("CAGR") of 14.95%. For the purposes of the report, the cannabis market was categorized into three different "application perspectives": spiritual purpose, health-condition management and wellness management. "The recreational cannabis market includes the consumption of cannabis for nonmedicinal reasons, such as boosting physical performance, pursuing creative activities, and experiencing spirituality," the press release stated. "Recreational cannabis can be consumed in the form of extracts, dried cannabis, edibles and beverages. Active cannabis companies in the markets this week include: Lucy Scientific Discovery Inc."
To view the full press release, visit https://ibn.fm/FpK0Q
Lucy Scientific Discovery Inc. (NASDAQ: LSDI) is an early-stage psychotropics manufacturing company focused on becoming the premier contract research, development and manufacturing organization for the emerging psychotropics-based medicines industry.
The company holds a Controlled Drugs and Substances Dealer’s License granted by Health Canada’s Office of Controlled Substances. This specialized license authorizes LSDI to develop, sell, deliver and manufacture pharmaceutical-grade active pharmaceutical ingredients (APIs) used in controlled substances and their raw material precursors. Lucy Scientific Discovery and its wholly owned subsidiary, LSDI Manufacturing Inc., operate under Part J of the Food and Drug Regulations promulgated under the Food and Drugs Act (Canada).
The company’s mission is to make its products and research services available for the development of medicines and experimental therapies to address certain psychiatric health disorders and other medical needs, including various mental health and addiction disorders. LSDI targets customers that include an increasing number of the leading universities, hospitals and other public, private and government institutions throughout the world that have launched research programs aimed at understanding the therapeutic potential of a range of psychedelic substances.
The company is headquartered in Victoria, British Columbia, Canada.
Products
LSDI produces a variety of high-quality natural, synthetic and biosynthetic products to meet the needs of the rapidly growing psychotropics-based medicines market. The company believes the emerging psychotropics industry will pave the way to a brighter future in mental health and overall wellness. LSDI is dedicated to advancing the frontiers of mind science and facilitating the development of psychotropic and psychedelic treatment therapies.
In Canada, the psychedelic compounds that LSDI is approved to produce under its Dealer’s License are regulated under the Controlled Drugs and Substances Act, or CDSA. Those compounds include:
- Psilocybin
- Psilocin
- Lysergic acid diethylamide, or LSD
- N,N-Dimethyltryptamine, or N,N-DMT
- 3,4-Methylenedioxymethamphetamine, or MDMA
- 4-Bromo-2,5-Dimethoxybenzeneethanamine, or 2C-B
The company also sells its consumer psychotropic products directly online and through retailers. Those products, described as microdose mushroom formulations, include a sleep aid, Twilight, and a mindfulness enhancer, Mindful.
Market Opportunity
According to a report from Global Market Insights, the psychotropics drug market had an estimated value of $20.2 billion in 2022 and is projected to reach a value of nearly $37.6 billion by 2032. That represents a CAGR of 6.4% for the forecast period. Factors driving market growth include the increasing prevalence of mental disorders, technological advancements in drug development, a rising geriatric population and increasing healthcare expenditures, the report states.
A growing awareness of mental health issues and an effort to reduce the stigma surrounding psychiatric disorders have encouraged more individuals to seek help, which in turn boosts the market. In addition, advancements in neuroscience, pharmacology and drug development have led to the discovery of new and more effective central nervous system therapeutics.
Innovative treatments offering better outcomes with fewer side effects attract patients and healthcare providers, also driving market growth.
Management Team
LSDI’s executive team brings deep experience in the development and commercialization of products featuring controlled substances, as well as the navigation of regulatory structures applicable to these products.
Richard Nanula is Chairman and CEO of LSDI. He has more than 35 years of leadership experience at several of the largest companies in the world, having been a senior executive at The Walt Disney Company, Amgen, Colony Capital and Starwood Hotels and Resorts. He has also served as a board member for Boeing Corporation and Starwood Capital, where he provided corporate guidance and oversight. He holds an MBA from Harvard Business School.
Assad J. Kazeminy, Ph.D., is Chief Scientific Officer at LSDI. He previously served as CEO of Irvine Pharmaceutical Services Inc. and Avrio Biopharmaceutical LLC, and he has founded several drug development companies. He has over 30 years of research and development experience in the biopharmaceutical industry. He received his Ph.D. in Pharmaceutical Science and Biochemistry from Esfahan University in Iran. He completed a Post Doctorate course of study at the University of Southern California Medical School, Department of Pharmacology.
Brian Zasitko, CPA, CA, is the company’s CFO. He is a Director of Invictus Accounting Group LLP, a professional services firm providing finance, advisory and accounting services. He also serves as CFO of Lobe Sciences Ltd., a company developing psychedelic compounds as therapeutics for the treatment of mild traumatic brain injuries and post-traumatic stress disorder. He has an undergraduate degree from Simon Fraser University and a CPA (CA) from Certified Professional Accountants, British Columbia.
Lucy Scientific Discovery Inc. (NASDAQ: LSDI), closed Friday's trading session at $0.720101, up 22.051%, on 646,000 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.55/$4.00.
Recent News
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Lucy Scientific Discovery Inc. (NASDAQ: LSDI) Is 'One to Watch'
- InvestorNewsBreaks - Lucy Scientific Discovery Inc. (NASDAQ: LSDI) Strengthening Position in Projected $51B Global Cannabis Market
- InvestorNewsBreaks - Lucy Scientific Discovery Inc. (NASDAQ: LSDI) Acquires High Times Intellectual Property, Featured in Financial News Media Article
GolfLync Inc.
The QualityStocks Daily Newsletter would like to spotlight GolfLync Inc.
GolfLync, the company behind the GolfLync app – a social network for matching golf games and players, is soaring due to its unique and feature-strong ability to bring together people who love golf, a sport that is experiencing a post-pandemic revival. "The app (available as a free download on the Apple App Store and the Google Play Store) is powered by an intelligent algorithm that brings golfers together based upon a balanced range of factors, including handicap, preferred playing style, and location, along with various related interests and specifications. The app readily supports everything from matching individual partners to building large and diverse groups called ‘Virtual Golf Clubs'(TM). These GolfLync VGCs are becoming especially popular, and now spread around the country," a recent article reads. "GolfLync CEO Noah DiPasquale says ‘It basically operates like Tinder for golfers,' while Michael Quiel, co-founder, summed it up: ‘Our commitment to providing a user-friendly and feature-rich platform has driven the exceptional growth and engagement we are witnessing.' According to the company, GolfLync has seen a 15x growth in membership in just the past 4 months."
To view the full article, visit https://ibn.fm/JYaJg
GolfLync Inc. matches golfers looking for a game through the company’s smartphone app, GolfLync. The company bills GolfLync as “the social network for golfers,” matching golf games and players similar to the way a dating app matches those looking for romance.
The app allows like-minded golfers to connect for a game simply by logging in. GolfLync helps golfers who are looking to grow their golf network find other players with similar interests and on course preferences. Whether you have recently moved to a new area and are looking for new golfing buddies, travel frequently and would like to play a round of golf while on the road, or just want to meet new golfers in your area, GolfLync is your answer. Spouses who enjoy golfing together can find other golfing couples to tee it up with. For a regular group that finds itself unexpectedly down a player, GolfLync can help find that last-minute addition to complete the foursome.
The company is based in Scottsdale, Arizona.

GolfLync App
GolfLync was created for golfers of all skill levels and preferences to connect with compatible players of similar skill. Golfers can find a tee time through GolfLync, join existing tee times and create new leagues. The app allows golfers to meet fellow players before committing to spend four hours on the course with them. GolfLync allows users to find new golf friends based on their preferences, such as walking or riding a cart, listening to music, friendly wagering, imbibing a favorite beverage at the 19th Hole and more. GolfLync is available for both Android and iOS as a free download.
Market Opportunity
According to a report by Statista, a leading provider of market and consumer data, in 2022, the number of people participating in golf in the United States reached 25.6 million, with 15.5 million additional players participating in off-course activities like driving ranges. In 2020, over 502 million rounds of golf were played in the U.S. alone. The game, traditionally dominated by male players, is changing, with increased interest from women golfers driven by social media influencers around the game.
Lumen Sports puts the total number of golf courses in the U.S. at more than 16,700. According to Lumen, about 75% of those are public courses open to all golfers, with the rest considered private golf clubs that require a membership.

Management Team
Noah DiPasquale is a co-founder and CEO of GolfLync Inc., leading the marketing and operations of the platform. He is also the founder and CEO of Epic Golf Club, a premier national membership and private golf society which partners with hundreds of top tier private golf clubs allowing Epic members access to their courses and recently founded the Epic Foundation, a Scottsdale-based 501c3. He holds a B.S. in Business Administration, Management and Operations from the W.A. Franke College of Business at Northern Arizona University and an MBA in Marketing from the University of Phoenix.
Michael Quiel is a co-founder of GolfLync Inc. and the President of the organization. He leads the application development and research teams. Michael understands how to build successful companies. His deep knowledge of investment banking, finance and building successful business partnerships is unparalleled. He’s an expert at capital formation and growth hacking companies. He has raised over $250 million in capital and taken multiple companies public.
Recent News
- GolfLync Inc. - InvestorNewsBreaks – GolfLync Inc.’s User-Friendly and Feature-Rich Platform Driving Exceptional Growth, Engagement
- InvestorNewsBreaks – GolfLync Inc.’s Pebble Beach Sweepstakes Provides Once-in-a-Lifetime Opportunity for Golf Enthusiasts
- GolfLync Inc., Looking Remarkably Like an Early-Stage Unicorn

Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF)
The QualityStocks Daily Newsletter would like to spotlight Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF).
Eloro Resources (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM), an exploration and mining company, is now embarked on the process of finalizing the release of its initial mineral resource estimate ("MRE") report, which will seek to provide the market with further granularity as to the Iska Iska property's viability as a commercial mining operation going forward. "Previously exploited by artisanal miners in the early 1960s, repeated surveys of Eloro's Iska Iska and Mina Casiterita properties – the latter situated to Iska Iska's immediate southwest, have outlined the appearance of an extensive, near surface, magnetic intrusive body spread across the two sites. These findings have been further complemented by an extensive program of exploratory drill work across the sites- dating back to early 2021 and amounting to a cumulative 85,000 meters of diamond drilling across 122 individual holes," a recent article reads. "Recent conclusions drawn from this drill work have driven the mining company to vastly increase their initial projections related to the potential scale of the deposit at hand; Eloro's most recent model now suggests that the company could potentially have access to a high-grade deposit (defined as having potential deposits of > 90g AG eq/t) measuring 800 meters wide and over 1,100 meters in depth."
To view the full article, visit https://ibn.fm/xLxVs
Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) is a publicly traded exploration and mine development company with a portfolio of gold and base-metal properties in Bolivia, Peru and Quebec.
The company has an option to acquire a 99% interest in the highly prospective Iska Iska Property, classified as a silver-tin polymetallic epithermal-porphyry complex, a significant mineral deposit type in the Potosi Department of southern Bolivia. Iska Iska is a road-accessible, royalty-free property.
Eloro also owns an 82% interest in the La Victoria Gold/Silver Project, located in the North-Central Mineral Belt of Peru, some 50 kilometers south of Barrick’s Lagunas Norte Gold Mine and Pan American Silver’s La Arena Gold Mine. La Victoria consists of eight mining concessions and eight mining claims encompassing approximately 89 square kilometers. La Victoria has good infrastructure, with access to road, water and electricity, and is located at an altitude that ranges from 3,150 meters to 4,400 meters above sea level.
The company has a strong management and technical team working diligently to uncover the value of both Iska Iska and La Victoria. Eloro is based in Toronto, Canada.
Projects
Iska Iska – Potosi, Bolivia
Iska Iska is associated with a Miocene possibly collapsed/resurgent caldera, emplaced on Ordovician age rocks with major breccia pipes, dacitic domes and hydrothermal breccias. The property is wholly controlled by the title holder, Empresa Minera Villegas S.R.L. It is located 48 kilometers north of Tupiza city, in the Sud Chichas Province of the Department of Potosi. This is an important mineral deposit type in the prolific South Mineral Belt of Bolivia. Eloro commissioned a NI 43-101 Technical Report on Iska Iska, which was completed by Micon International Limited and is available on Eloro’s website and under its filings on SEDAR.
A fully financed drill program is currently underway on the property, situated near world-class deposits including Silver Sand, San Bartolomé, Pulacayo, San Cristobal, San Vicente, Chorolque, Tasna, Choroma and Siete Suyos. Iska Iska is in the southwest part of the Eastern Cordillera, which hosts a number of major polymetallic mines and mineral deposits. Drilling and continuous channel sampling results have demonstrated some very high metal values, especially silver and tin, within an immense system, where mineralization has been encountered in every drill hole to date. The company believes there is excellent potential for world-class bulk mineable deposits.
La Victoria – Ancash, Peru
The La Victoria project, targeting gold and silver production, is situated near world-class, low-cost gold producers Pan American Silver and Barrick Gold Corporation. Located in Ancash Department, La Victoria sits on the western slopes of the Peruvian Andes. The property is located 12 hours from Lima, with a travel distance of 600 kilometers. The nearest road accessible population centers from La Victoria are Huandoval, Pallasca and Cabana. The project includes four principal mineralized zones in Peru’s prolific North-Central Mineral Belt – San Markito, Victoria, Victoria South and Ccori Orcco – with excellent potential for gold discovery. Operations at La Victoria are planned to proceed with a 2,000-meter diamond drilling program to test targets to outline potential resources at San Markito. Trenching and sampling confirmed high silver values and veins at San Markito in 2020.
Market Outlook
According to industry association The Silver Institute, the outlook for silver demand is exceptionally promising, with global demand forecast to rise to a record high of 1.112 billion ounces in 2022. The increase will be driven by record silver industrial fabrication, which is forecast to improve by 5%, as silver’s use expands primarily in solar energy and electric vehicle (EV) manufacturing. The institute states that government commitments to carbon neutrality have resulted in a rapid expansion of green energy projects, driving record photovoltaic panel installations which are expected to lift silver demand in this segment to an all-time high in 2022.
Rising demand in the electronics industry is also boosting the demand for tin, which is primarily used in solder. The electronics and electrical industries use solders containing 40-70% tin, which provide strong and reliable joints under a variety of environmental conditions. At present, the majority of the assemblers are using patented tin-and-copper-based solders. Mordor Intelligence estimated tin demand at 387 kilotons in 2021 and forecasts demand growth of 2.5% annually through 2027. Over the medium term, surging demand from the EV market and increasing applications in the electrical and electronics industry is expected to drive the market.
Management Team
Thomas G. Larsen is CEO of Eloro. He has more than 40 years of experience in the investment industry, specializing in corporate finance and management of junior resource companies, raising in excess of C$200 million. He previously held the position of President and Chief Executive Officer of Champion Iron Limited. Prior to that, he was President and Chief Executive Officer of Champion Iron Mines Limited.
Dr. Bill Pearson is Executive VP of Exploration for Eloro. He has more than 40 years of direct experience in the exploration and production of minerals worldwide. He played an integral role in the acquisitions of Desert Sun Mining Corp. by Yamana Gold in 2006 and Central Sun Mining by B2 Gold in 2009. He was formerly VP Exploration at Desert Sun Mining and Senior VP at Central Sun Mining.
Miles Nagamatsu, CPA, is CFO at Eloro. He has over 30 years of experience in accounting, management, lending, restructurings and turnarounds. Since 1993, he has acted as a CFO of public and private companies primarily in the mineral exploration and investment management sectors. He holds a Bachelor of Commerce degree from McMaster University.
Osvaldo Arce Burgoa is General Manager at Eloro. He is a geological and mineral processing engineer with 26 years of experience in Bolivia. He is a former President of the Bolivian Geological Society, Main Technical Advisor of the National Mining Corporation (COMIBOL) and has served as exploration manager and chief geologist at various mining and exploration companies. He has authored two books on Bolivian geology and holds a doctorate in mining engineering from Tohoku University in Sendai, Japan.
Eloro Resources Ltd. (OTCQX: ELRRF), closed Friday's trading session at $1.56, off by 0.636943%, on 47,580 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.50/$3.135.
Recent News
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - MiningNewsBreaks - Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Recent Models Suggest Potential Access to High-Grade Deposit
- Taliban Reveals It Has Penned $6.5B Worth of Mining Deals
- InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Exec Featured in Recent The Market Herald Release
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
CNS Pharmaceuticals recently announced updated results from its pivotal clinical study on GBM treatment through its lead drug candidate, Berubicin
The novel anthracycline demonstrated its capability to be an innovative potential treatment option for GBM
151 patients have been enrolled in the study, marking the data cutoff point and a significant milestone toward reporting topline results before the end of the year
Based on the results seen preclinically, the company remains optimistic that Berubicin may provide a much-needed clinical benefit for GBM patients
CNS Pharmaceuticals (NASDAQ: CNSP), a clinical-stage biotechnology company specializing in the development of novel treatments with a focus on brain cancer, glioblastoma ("GBM"), and neuro-oncology, recently announced the presentation of updated safety results from its potentially pivotal clinical study on GBM treatment through its lead drug candidate, Berubicin. Most notably, this novel anthracycline demonstrated its capability to be a safe and innovative potential treatment option for GBM, having shown aspects integral to effective therapy for the disease (https://ibn.fm/yp5AC).
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Friday's trading session at $1.48, off by 3.8961%, on 28,796 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.6105/$8.64.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Looking Forward to Upcoming Interim Results for Potentially Pivotal GBM Study
- InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Announces 200-Patient Enrollment Milestone in Potentially Pivotal Berubicin Study
- Policies Encouraging High-Volume Facilities Disadvantage Oncology Patients in Rural Areas
Knightscope, Inc. (NASDAQ: KSCP)
The QualityStocks Daily Newsletter would like to spotlight Knightscope, Inc. (NASDAQ: KSCP).
Knightscope (NASDAQ: KSCP), a leading developer of autonomous security robots ("ASRs") and blue light emergency communication systems, has announced another new deployment and contract renewal for K5 ASRs.
The announcement reads, "Knightscope's commitment to making children safe extends beyond the classroom and into extracurricular and recreational activities. With over 3.5 billion followers worldwide, soccer is the number one sport on planet Earth, and Knightscope has begun protecting a new generation of players and fans in New York. A soccer academy and sports complex in Long Island now has a K5 ASR securing its entrance against trespassers and patrolling its parking lot to deter vehicle and catalytic converter thefts….
"A Fortune 1000 hotel and resort chain client renewed its K5 ASR contract in the Seattle area for the second year. Hospitality is a growing segment among Knightscope's clientele. The company has already secured numerous deployments and multi-year renewals in hotels, casinos and resorts, and this client boasts 6,500 properties globally, over 5,400 locations of which are in the United States."
To view the full press release, visit https://ibn.fm/4RAr8
Knightscope, Inc. (NASDAQ: KSCP), founded in 2013 and based in Mountain View, California, is a leader in the development of autonomous security capabilities targeting to disrupt the $500 billion security industry. Knightscope’s technology uniquely combines self-driving technology, robotics, artificial intelligence and electric vehicles.
Knightscope designs and builds Autonomous Security Robots (ASRs) that provide 24/7/365 security to the places you live, work, visit and study. The company’s client list covers public institutions and commercial business operations, including multiple Fortune 1000 companies to date. These ASRs have been proven to enhance safety at hospitals, logistics facilities, manufacturing plants, schools and corporations. ASRs act as highly cost-effective complementary systems to traditional security and law enforcement officials, providing an additional advantage by continuing to offer uninterrupted patrolling capabilities across the country.
The company’s ASRs have assisted in the arrest of suspects involved in crimes ranging from armed robbery to hit-and-runs. Their machine-embedded thermal scanning capability even aided in preventing the breakout of a major fire. You can learn more about the crime fighting wins at www.knightscope.com/crime
The company has achieved several milestones since its creation in 2013, including:
- Establishing itself in a 15,000-square-foot facility located in Mountain View, California, in the heart of Silicon Valley, where Knightscope designs, engineers and builds its technology (Made in the USA)
- Operating for more than 1 million hours in the field and securing contracts across five time zones, from Hawaii to Rhode Island
- Raising over $100 million since inception to build its technology from scratch and generating over $13 million in lifetime revenue, validating both the market opportunity and the technology
Growth Capital & Proposed Nasdaq Listing
With backing from more than 28,000 investors and four major corporations and over $100 million raised since inception, Knightscope is poised to be an industry leader in the future of public safety and security.
On December 1, 2021, Knightscope announced the commencement of an offering of up to $40 million of its Class A common stock, with shares to be listed immediately following closing on the Nasdaq Global Market under the ticker symbol ‘KSCP’. The offering is for up to 4 million shares priced at $10 per share. Learn more at www.knightscope.com/investors
Company Mission – Reimagining Public Safety
Knightscope’s long-term vision has an eye on the greater good. The company’s mission is to make the United States of America the safest nation in the world while supporting the 2+ million law enforcement and security professionals across the country.
Crime has an estimated negative economic impact in excess of $2 trillion annually. As crime is reduced, positive impacts will likely be realized across several aspects of society, including housing, financial markets, insurance, municipal budgets, local business and safety in general.
Knightscope CEO William Santana Li was interviewed by Kevin O’Leary, more commonly known as Shark Tank’s Mr. Wonderful. When asked to explain how the benefits provided by the ASRs outrank a human doing the same job, Li said, “First, just the simple presence of a physical deterrent causes criminal behavior to change. Second, the machines are self-driving cars that patrol all around and recharge themselves. They also generate 90 terabytes of data per year. No human would ever be able to process that. The robots are intended to be eyes and ears for the humans, not a one-to-one replacement.”
The Knightscope solution to reduce crime combines the physical presence of ASRs, sometimes referred to as proprietary Autonomous Data Machines, with real-time onsite data collection and analysis. The ASRs are fitted with eye-level 360° cameras, thermal scanning, public address announcements and various other features that work in tandem with humans to provide law enforcement officers and security guards unprecedented situational awareness.

Those 90 terabytes of data are then formatted in a useable way, so law enforcement can leverage that information and execute their responsibilities more effectively.
Public Safety Innovation
The company’s recurring revenue business model is set up to mimic the recurring societal problem of crime, and it takes into consideration the fact that innovation in the security and public safety industry has been stagnant for decades. Because the traditional practices of the sector have remained unchanged for years, automation has potential to drive substantial cost savings – and significant improvement in capabilities.
Human security guards are one of both the largest expenses and the largest liabilities for companies. Knightscope’s robots are offered at an effective price of $3 to $9 per hour, compared with approximately $85 for an armed off-duty law enforcement officer and $15 to $35 for an unarmed security guard.
This innovation has the potential to drive considerable cost savings. Based on these estimates, manufacturing costs can be recovered as soon as the first year of operation.
Product Offerings
The company has nine patents and a framework of unique intellectual property. Knightscope currently offers a K1 stationary machine, a K3 indoor machine and a K5 outdoor machine. A K7 multi-terrain four-wheel version is in development.

The ASRs autonomously patrol client sites without the need for remote control, providing a visible, force multiplying, physical security presence to help protect assets, monitor changes in the area and deter crime. The data is accessible through the Knightscope Security Operations Center (KSOC), an intuitive, browser-based interface that enables security professionals to review events generated by the ASRs providing effectively ‘mobile smart eyes and ears’. Learn more at www.knightscope.com/ksoc
The ASRs and the related technologies were developed ground up by the company and are Made in the USA.
The Robot Roadshow

Knightscope has created the ultimate hybrid physical and virtual event, bringing its Autonomous Security Robot technologies to cities across the country for interactive and in-person demonstrations.
Each roadshow landing is hosted virtually by a Knightscope expert, and visitors can interact directly with each of the company’s ASRs and see the Knightscope Security Operations Center (KSOC) user interface in action. Learn more at www.knightscope.com/roadshow
Management Team
Chief Executive Officer William Santana Li is a veteran entrepreneur, a former executive at Ford Motor Company and the founder of GreenLeaf, a company that grew to be the world’s second-largest automotive recycler and is now part of LKQ Corporation (NASDAQ: LKQ).
Chief Client Officer Stacy Dean Stephens brings his experience as a former Dallas law enforcement officer, as well as his skills as a seasoned entrepreneur, to assist on the client acquisition side.
Chief Intelligence Officer Mercedes Soria is an award-winning technologist and former Deloitte software engineer.
Chief Design Officer Aaron Lehnhardt brings over two decades of two- and three-dimensional product and industrial design in modeling and VR to the table, on top of his experience as a senior designer at Ford Motor Company.
Chief Financial Officer Mallorie Burke is a seasoned financial executive and strategic advisor for both private and publicly traded technology companies with a successful track record of mergers & acquisitions, corporate growth and exit strategies, including public listings.
General Counsel Peter Weinberg leverages 30 years of diverse corporate counsel experience, spanning from startups to well-established companies, private and public. He has significant experience training personnel at all levels in critical areas to improve corporate compliance and productivity.
Knightscope, Inc. (NASDAQ: KSCP), closed Friday's trading session at $0.882, off by 7.9044%, on 1,363,999 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.36/$3.65.
Recent News
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Deploys K5 ASRs for New Client, 2nd-Year Contract Renewal
- InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) K5 ASRs Deployed to Oregon Hospital and Texas-Based Industrial Facility
- InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP), RippleLink Partner to Provide Private LTE, Enhanced Connectivity to ASR Customers
GEMXX Corp. (OTC: GEMZ)
The QualityStocks Daily Newsletter would like to spotlight GEMXX Corp. (OTC: GEMZ) .
GEMXX (OTC: GEMZ), a mine-to-market global jewelry producer, is ramping up its capacity for gold ore processing at one of its Canadian assets, enthusiastic over the better-than-expected amount of ore produced from stockpiled surface material. "GEMXX's Snow Creek Mine property in British Columbia completed site preparation for gold processing last year and mobilized equipment in May to begin bringing the property into full production. GEMXX reported the first production of gold from the site on Aug. 15 as ore collected from a 4,500-cubic-yard sample during construction of the facility was ‘cleaned out.' The operation yielded 38.73 ounces of gold, amounting to 0.86 ounces per 100 cubic yards, according to the company's statement," a recent article reads. According to the company's CEO Jay Maull, GEMXX is anticipating that gold ore from the 498-acre site, together with ammolite from its Alberta resources, will bolster the company's long-term plans to expand its jewelry production and "de-risk revenues." "These results are a promising start to what is anticipated to be a highly lucrative first year of gold mining," Ian McClintock, the president of GEMXX's exploration partner Crazy Horse Mining Inc., is quoted as saying.
To view the full article, visit https://ibn.fm/lWneg
GEMXX Corp. (OTC: GEMZ) is a mine-to-market enterprise specializing in gold, gemstone, and jewelry production. With ownership of mining resources, production facilities, and operational assets, the company maintains control over every aspect of its production process, from gold mining and gemstone extraction to jewelry manufacturing and global distribution.
 As a prominent player in the industry, GEMXX stands out as a leading producer of high-quality finished Ammolite jewelry. Notably, it holds the distinction of being the sole public company engaged in Ammolite mining worldwide. In addition to its Ammolite operations, the company is actively involved in gold mining and prides itself on its ability to design and manufacture exquisite jewelry pieces and exceptionally rare, natural fossil decor items for clientele around the globe.
As a prominent player in the industry, GEMXX stands out as a leading producer of high-quality finished Ammolite jewelry. Notably, it holds the distinction of being the sole public company engaged in Ammolite mining worldwide. In addition to its Ammolite operations, the company is actively involved in gold mining and prides itself on its ability to design and manufacture exquisite jewelry pieces and exceptionally rare, natural fossil decor items for clientele around the globe.
One of GEMXX’s key advantages lies in mining its own gold reserves to be utilized in its jewelry production. This strategic approach provides the company with a cost-saving edge over other producers in the market.
Ammolite is similar to black opal and is a biogenic gem like amber and pearl. It is derived from the fossilized shells of ammonites, a group of extinct marine nautiluses.
GEMXX’s world class gemstone cutters and jewelry designers are continuously leading the Ammolite industry. Its team believes in the company’s philosophy, vision and goals, and works every day to continue to drive the Ammolite industry to the forefront of the gem world.
The company has offices in Las Vegas and Hong Kong.
Projects and Operations
GEMXX has formulated an ambitious growth plan that, while challenging, is deemed attainable. The company’s strategy revolves around bolstering its market share through several key initiatives. Firstly, GEMXX aims to strengthen its position in current markets by nurturing and expanding existing relationships with customers and partners.
Secondly, the company plans to venture into untapped markets strategically. By identifying and targeting new areas, GEMXX seeks to establish a presence in regions that present promising opportunities for growth.
Additionally, GEMXX envisions growth through acquisitions. By considering and integrating key services, distribution networks and retail outlets into its fold, the company aims to consolidate its market position and capitalize on synergies for enhanced success.
To cater to the rising demand for its products, GEMXX has placed a primary focus on increasing gemstone production. The company’s southern properties, situated in Alberta, Canada, hold valuable deposits of rough Ammolite gemstone. By tapping into these resources, GEMXX is poised to meet the demand for its exquisite gemstone products and further fuel its expansion plans.
GEMXX possesses significant mineral assets in the form of a Mineral Work Permit covering an 800-acre area and two Ammonite Shell Mineral agreements encompassing 217 acres within the same region. The company’s management effectively operated mines in close proximity to these properties. Moreover, core sampling, along with fossil outcroppings on the riverbanks, confirms a substantial Ammolite resource present in these designated areas.
Both the Mineral Work Permit and the Ammonite Shell Mineral agreements grant GEMXX unrestricted access to all Ammolite resources within their respective demarcations. Notably, the company is not obligated to pay any royalties to third parties, thereby enabling GEMXX to fully capitalize on the potential of these valuable resources.
Furthermore, there are no stringent regulatory conditions that GEMXX must fulfill to gain or retain access to the Ammolite deposits. This freedom of access allows the company to proceed with its mining and production operations unimpeded, providing an advantageous position for future growth and success.
In March 2023, GEMXX made a significant announcement, revealing its acquisition of a 50% ownership stake in Crazy Horse Mining Inc., a Canadian gold mining company with assets situated in the province of British Columbia. As part of this deal, Crazy Horse’s assets, which encompass a 100% interest in two gold projects, called Snow Creek and Rosella Creek, spread across a substantial area exceeding 700 acres, now become part of GEMXX’s portfolio.
 Under the terms of this strategic partnership, GEMXX and Crazy Horse will jointly share the expenses related to mining operations on these projects. Additionally, the two companies will share the gold produced from these ventures, leading to a collaborative and mutually beneficial arrangement.
Under the terms of this strategic partnership, GEMXX and Crazy Horse will jointly share the expenses related to mining operations on these projects. Additionally, the two companies will share the gold produced from these ventures, leading to a collaborative and mutually beneficial arrangement.
Initial tests conducted on the property, combined with gold already recovered this season, confirm all expectations for the claims and substantiate the company’s estimated extraction target of over 100,000 ounces of easily recoverable gold. To validate and provide a more comprehensive assessment of this estimate, an S-K 1300-compliant Resource Report is scheduled to be conducted during the summer of 2023.
By acquiring this stake in Crazy Horse Mining Inc., GEMXX has positioned itself for further growth in the gold mining sector and is poised to capitalize on cost of goods savings in its jewelry business.
Market Opportunity
Leading independent market research companies such as Data Monitor and GIA estimate the worldwide market for luxury or premium lifestyle products, which include gems and jewelry, at over $90 billion annually and growing. Ammolite sales around the world have seen unprecedented growth over the past 20 years. Worldwide retail sales are now estimated to be over $100 million.

Ammolite jewelry and fossils are featured aboard cruise ships and can be found in specialty shops in almost every cruise port in North America. Asian markets have grown since feng shui master Edward Li called Ammolite the most influential stone of the new millennium, referring to it as the “Seven Color Prosperity Stone.” Home shopping channels in Japan, Australia, France, Germany, the UK, Canada and the U.S. have all featured Ammolite jewelry.
Ammolite and ammonites can also be found on many ecommerce sales platforms, including Amazon, eBay and Etsy. Ammolite is sold around the world in tourist and traditional jewelry markets. The company has established customers in home shopping channels, cruise tourism, jewelry retailers, Asian feng shui markets, Asian retail markets and ecommerce platforms.
Management Team
With over 160 years in Ammolite management, operations, and sales, GEMXX possesses an unparalleled wealth of knowledge and expertise. Its team members have extensive backgrounds in every facet of the Ammolite business, allowing the company to excel in product development, maintain rigorous quality control measures, and maximize profitability. The breadth and depth of the GEMXX team’s experience enable the company to navigate the industry with precision, ensuring that GEMXX remains at the forefront of the Ammolite market. GEMXX leverages its collective wisdom to drive innovation, deliver exceptional products, and optimize business strategies to achieve long-term success.
Jay Maull is Founder, CEO and Chairman of GEMXX. With a career spanning more than three decades, he has been deeply involved in the Ammolite industry, from mining and production to marketing. He has owned and operated the world’s largest Ammolite mine and has delivered exceptional Ammolite products to customers across all continents. He has also established the world’s largest Ammolite ecommerce platform.
Richard Clowater is President of GEMXX. He is a skilled sales and marketing professional with a focus on research, data analysis and strategic planning. He has successfully implemented initiatives to expand markets, boost profits and foster customer loyalty. He has an impressive track record of negotiating sales and contracts worth over $250 million with influential stakeholders, including key purchasing personnel, C-suite executives and government entities at all levels.
Tom Dryden is a Vice President of GEMXX and brings a wealth of experience and expertise to the production and marketing of Ammolite, spanning over 30 years. His extensive involvement in the industry has granted him unparalleled knowledge of the Bearpaw Ammonite bearing formations. As a recognized authority in the field, Mr. Dryden’s research and papers on Canadian Ammonites have garnered global recognition, being published worldwide. In his role at GEMXX, Mr. Dryden assumes the responsibility of overseeing the company’s Canadian-based production facilities.
P. K. Chung is Business Manager Asia at GEMXX. With a track record of over 25 years in Ammolite business management, production and marketing in Asia, she is a recognized authority in the industry. Based in the Hong Kong gem district, she possesses an intricate understanding of the Asian gem and jewelry markets, including market dynamics, consumer preferences and industry trends specific to the region. Her strategic insights and deep connections enable GEMXX to thrive in this influential market.
GEMXX Corp. (OTC: GEMZ), closed Friday's trading session at $0.04805, off by 3.9%, on 11,009 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.0365/$0.998.
Recent News
- GEMXX Corp. (OTC: GEMZ) - TinyGemsBreaks - GEMXX Corp. (GEMZ) Eyes Promising Start to Expected 'Highly Lucrative' First Year of Gold Mining
- TinyGemsBreaks - Why GEMXX Corp. (GEMZ) Is 'One to Watch'
- GEMXX Corp.'s (GEMZ) Strategic Investments Pay Off, as Company Positions itself as a World Class Producer and Distributor of Gemstones and Jewelry
RVL Pharmaceuticals plc (NASDAQ: RVLP)
The QualityStocks Daily Newsletter would like to spotlight RVL Pharmaceuticals plc (NASDAQ: RVLP).
RVL Pharmaceuticals plc (NASDAQ: RVLP) is a specialty pharmaceutical company focused on the commercialization of UPNEEQ® (oxymetazoline hydrochloride ophthalmic solution), 0.1%, which is available by prescription for the treatment of acquired blepharoptosis, or low-lying eyelid(s), in adults.
UPNEEQ® (RVL-1201) is the first non-surgical treatment option approved by the U.S. Food and Drug Administration (FDA) for acquired blepharoptosis. The company received FDA approval in July 2020 and launched UPNEEQ® in September 2020 to a limited number of eye care professionals, with commercial operations expanded in 2021 among ophthalmology, optometry, and oculoplastic specialties.
In February 2022, UPNEEQ® was launched into the medical aesthetics market in the United States. Patients can purchase UPNEEQ® from eye care or medical aesthetic professionals, or through RVL Pharmacy LLC, the company’s wholly owned pharmacy. The company plans to promote UPNEEQ® to people with acquired ptosis and those who are bothered by low-lying lids. RVL Pharmaceuticals believes there is a significant commercial opportunity for UPNEEQ®, given the meaningful unmet need for a non-invasive treatment across millions of acquired-ptosis patients in the United States. The company’s near-term focus is to continue the rollout of UPNEEQ® into the medical aesthetics market through its dedicated aesthetics sales force while continuing to support ongoing utilization and expanded penetration of UPNEEQ® in ocular medicine markets.
RVL Pharmaceuticals continues to raise patient and physician awareness of acquired ptosis and UPNEEQ® through medical conferences, HCP and DTC advertising, social media (e.g., Facebook and Instagram), and marketing partnerships.
The company is incorporated in Ireland and headquartered in Bridgewater, New Jersey.
UPNEEQ®
UPNEEQ® is an oxymetazoline hydrochloride ophthalmic solution for the treatment of acquired blepharoptosis, or low-lying eyelid(s), in adults. It is the first and only FDA-approved ophthalmic solution for this indication.
The once-daily UPNEEQ® eye drop has been shown in clinical trials to result in an average one-millimeter lift of the upper eyelid, and to improve superior visual field in patients with a functional deficit. Patients’ eyelids demonstrate lift in as little as five minutes post dose, with the lift effect lasting as long as eight hours. The preservative-free solution is safe and well-tolerated. Trials demonstrated side effects similar to those of placebo.
The active ingredient in UPNEEQ® is oxymetazoline 0.1%, a direct-acting α-adrenergic receptor agonist that targets receptors in the Müller’s muscle, which causes the muscle to contract and lift the upper eyelid. UPNEEQ® delivers eye-opening results for patients along the entire spectrum of age and condition severity.
UPNEEQ’s health care provider customers include optometrists, ophthalmologists, oculoplastic surgeons, facial plastic surgeons, dermatologists and a broad range of practitioners qualified to diagnose and treat acquired blepharoptosis in adults.
The target patient population comprises adults with droopy or low-lying eyelids, the majority of whom are female. While the exact prevalence of acquired ptosis is unknown, RVL Pharmaceuticals believes it to be a common age-related condition.
Market Opportunity
A survey of eye care providers and medical aesthetics specialists revealed that they believe that approximately half of adult patients visiting their practices are affected by droopy or low-lying eyelids. Further, the company estimates that approximately 60% of adult women self-identify as having some degree of droopy or low-lying eyelids, and a majority of those women indicate that they are bothered by the position of their eyelids.
The global medical aesthetics market is expected to reach a value of $18 billion in 2027, rising at a compound annual growth rate of over 10%, with North America representing the largest share of the global market. Similarly, the global eye care market is expected to reach a value of $86 billion by 2026, rising at a compound annual growth rate of over 6%. An estimated 100 million adults visit an eye care provider each year in the United States alone.
RVL Pharmaceuticals believes the growth in medical aesthetics and eye care markets will be driven by an aging population and increasing life expectancy, which is resulting in more consumers with a desire for improved appearance and well-being over a longer period of time. Other contributing factors include rising disposable income globally and in the U.S.; growing awareness, utilization, and acceptance of elective or minimally invasive and non-invasive interventions; and continued innovation and improved accessibility to treatments due to an increase in the number of physicians who offer eye care and medical aesthetics services.
Management Team
Brian Markison is Chairman of the Board and Chief Executive Officer of RVL Pharmaceuticals. He has more than 30 years of operational, marketing, commercial development, and sales experience with international pharmaceutical companies. He previously served as the President and CEO of Fougera Pharmaceuticals Inc., a specialty pharmaceutical company. Before that he was Chairman and CEO of King Pharmaceuticals Inc. He also held various senior leadership positions at Bristol-Myers Squibb. He received a bachelor’s degree from Iona College.
James Schaub is Executive Vice President and Chief Operating Officer of RVL Pharmaceuticals. Prior to that he served as COO of Trigen Laboratories. He previously was Vice President, M&A of Fougera Pharmaceuticals. Before that he spent five years with King Pharmaceuticals. Mr. Schaub holds a bachelor’s degree in economics from Middlebury College and an M.B.A. from Rutgers Business School.
RVL Pharmaceuticals plc (NASDAQ: RVLP), closed Friday's trading session at $0.1075, up 1.6068%, on 3,333,795 volume. The average volume for the last 3 months is 1.72M and the stock's 52-week low/high is $0.0929/$2.99.
Recent News
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - BioMedNewsBreaks - Why RVL Pharmaceuticals plc (NASDAQ: RVLP) Is 'One to Watch'
- InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Announces Participation at Upcoming H.C. Wainwright Global Investment Conference
- RVL Pharmaceuticals plc (NASDAQ: RVLP) Is 'One to Watch'
Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF)
The QualityStocks Daily Newsletter would like to spotlight Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) .
Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) is a public oil and gas exploration, exploitation and development company focusing on conventional oil and gas reservoirs in Western Canada. The company uses its experience to develop, acquire and drill assets with potential for primary and secondary recovery.
Prospera is primarily focused on optimizing hydrocarbon recovery from legacy fields through environmentally safe and efficient reservoir development methods and production practices. It is in the midst of a three-stage restructuring process aimed at prioritizing cost effective operations while appreciating production capacity and reducing liabilities.
The company is based in Calgary, Alberta, Canada.
Operations
Prospera’s core properties include more than 42,000 cumulative acres across Cuthbert, Luseland and Heart Hills in Saskatchewan and Red Earth and Pouce Coupe in Alberta. In total, the company estimates that there are half a billion barrels of oil in place at these sites accounting for 20+ years of forward project lifespan, with as little as 8% of total reserves having been recovered via legacy vertical well technology.
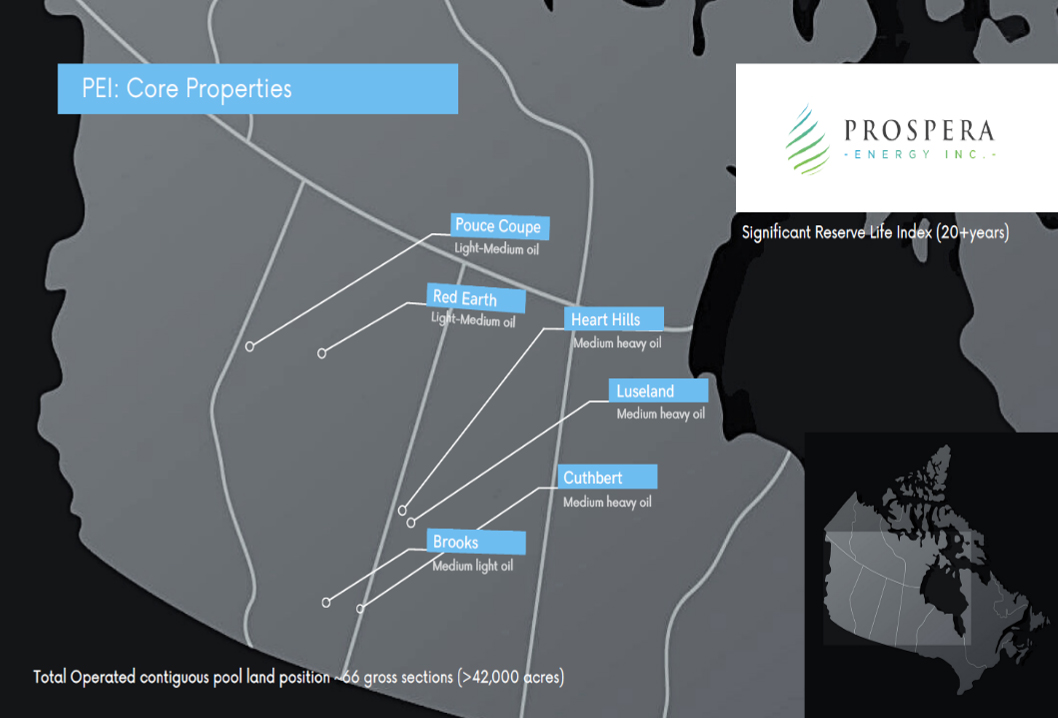
Restructuring Initiative
In 2021, Prospera enacted a top-down reorganization. The early results of these efforts were on display in May 2023, when the company reported a three-fold year-over-year increase in annual revenue for 2022 alongside drastically reduced operating costs and record-high cash flow from operations.
Prospera noted in the news release that it has positioned itself in 2023 to execute the second phase of its development plan aimed at increasing production through medium-oil development in Alberta and leveraging horizontal wells to capture the significant remaining reserves in Saskatchewan.
During the company’s investor summit in August 2023, Prospera CEO Samuel David provided more information regarding this three-phase strategy:
Phase I
Prospera completed the first phase of its restructuring by optimizing operations at its existing assets and addressing legacy arrears and non-compliance issues.
At the beginning of this transformation, the company was producing just 80 barrels of oil equivalent (BOE) per day. In Q4 of last year, Prospera peaked at nearly 1,200 BOE per day. Its breakeven is around 500 barrels per day, illustrating the opportunity for free cash flow. This prospect has driven Prospera’s capital development and optimization in recent quarters.
After a temporary slowdown in production due to harsh winter conditions, Prospera is currently producing about 800 BOE per day and anticipates an additional 300-500 barrels of daily production following the completion of ongoing site maintenance work.
This sustained increase has pushed the company’s NPV from roughly $3 million prior to the restructuring efforts to approximately $72 million today.
In an effort to build on this progress and maximize its available resources, Prospera piloted two horizontal reentries to assess a potential horizontal well transformation at its properties.
Phase II
Following up on the optimization efforts of Phase I, Prospera aims to commence a horizontal well transformation at its properties in the coming months. Based on its pilot wells from Phase I, the company has proposed 10 horizontal well locations at its Cuthbert and Heart Hills properties.
Prospera has likewise proposed eight medium light oil direction wells at its Alberta property, and it is exploring strategic acquisitions aimed at expanding its core area and diversifying its product mix.
Other facets of Phase II include piloting an enhanced oil recovery (EOR) application and continuing to execute its liability management goals and ESG initiatives. Prospera has already abandoned 60 vertical wells as part of its three-year LMR plan to reclaim surface land and reduce the environmental footprint of its operations.
Phase III
Beginning next year, Phase III of Prospera’s corporate redevelopment strategy will focus on continuing the company’s horizontal modular development to appreciate production and optimize recovery of remaining reserves. Prospera intends to implement full-scale EOR applications based on the results of its Phase II pilot program, which is forecast to optimize recovery by greater than 10%.
Prospera also intends to continue its acquisition strategy to diversify its product mix. Its goal, as detailed by in August 2023 investor summit, is to attain 50% light oil, 40% heavy oil and 10% gas – all while continuing to eliminate carbon emissions as part of its existing ESG initiatives.
Poised for Growth
Following its transformational efforts in 2022, Prospera is poised to achieve record growth in 2023. The company has forecast significant reductions in production costs through 2024, alongside sizable increases in daily production.
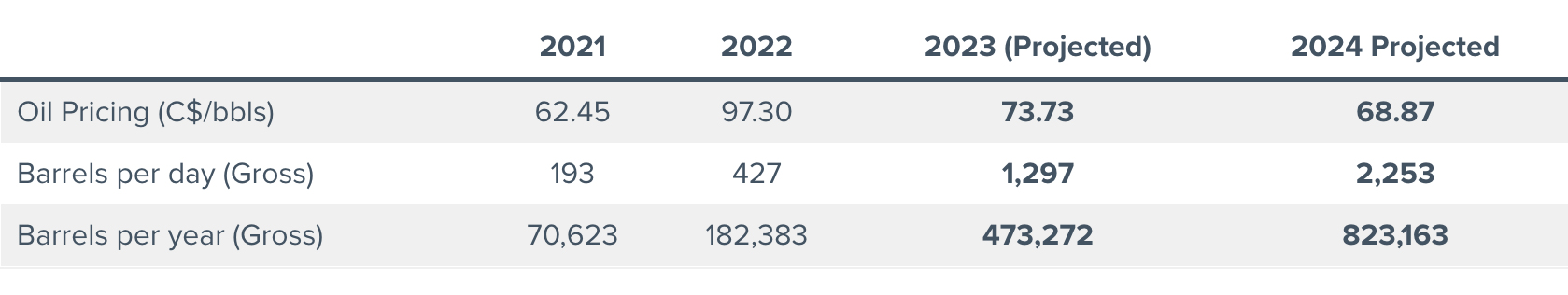
Prospera is currently exploring strategic acquisition targets to potentially increase its production beyond 5,000 BPD while expanding its reserve base to a billion barrels.
Market Opportunity
While the oil and gas industry faces long-term geopolitical and macroeconomic uncertainty, there is a clear trend to secure supply in the short term. According to Deloitte, the global upstream industry ended 2022 with some of the highest free cash flows on record, driving reinvestment in hydrocarbons and overall investment in clean energy.
The Energy Information Administration recently forecast a dip in global oil inventories over each of the next five quarters, placing upward pressure on oil prices. The agency further forecasts a YoY increase in fuel consumption, exacerbating the effects of OPEC+ production cuts that are set to remain in place through 2024.
For Prospera, these forecasts are promising. The company aims to build on its recent financial growth in the coming months (Prospera reported a three-fold YoY increase in revenue to $13.9 million in 2022), hitting a projected $57 million in total revenue by the end of 2024 while working to expand its core area holdings through accretive M&A transactions.
Leadership Team
Prospera is led by a team with extensive, diverse petroleum industry experience spanning both reservoir management and operations of oil and gas assets. The team boasts a proven track record of reorganizing companies, structuring financing arrangements and positioning for growth.
Samuel David is the company’s President and CEO. He brings to Prospera over 32 years of experience in operation, development and management of oil and gas assets and companies. Mr. David holds a B.Sc. in Mechanical Engineering and a B.A. in Economics from the University of Calgary. His background consists of both engineering and executive management experience with majors Petro-Canada, AEC Oil & Gas (now EnCana / Cenovus) and Husky Energy, as well as founding and operating juniors Ventura Energy and First West Petroleum. Mr. David has proven expertise in corporate planning, production, reservoir engineering, depletion strategies, EOR, property evaluations, acquisitions and divestitures.
George Magarian is VP Subsurface for Prospera. He is a professional petroleum geologist (APEGA) with over 36 years’ experience in the Western Canada Sedimentary Basin. After graduating with an Honors B.Sc. degree in Earth Science from the University of Waterloo, Mr. Magarian spearheaded many successful exploration programs, conducted evaluations for improved recovery schemes and assessed/exploited unconventional oil reservoir opportunities. He has held roles of increasing responsibility, from exploration geologist at oil industry major Petro-Canada and intermediates Anderson Exploration and Jordan Petroleum, to geoscience manager and VP exploration at junior companies Ionic Energy, Gentry Resources and Westfire Energy.
Chris Ludtke is the company’s VP Finance & Accounting. He is a high functioning finance leader with extensive expertise in finance, budgets and planning, accounting, economic evaluation, management, governance and sound decision making. Mr. Ludtke has 20 years of experience within the oil and gas, clean energy and renewables industries, including 12+ years working for Husky Energy before moving into an executive role in the junior oil and gas and hydrogen space. He graduated from the University of Lethbridge (Bachelor of Management) and is a Chartered Professional Accountant in the Province of Alberta.
Matthew Kenna is the CFO of Prospera. He has over 30 years’ experience leading organizations and helping them expand, drive efficiencies and grow profitability. Mr. Kenna is a professional accountant (CPA, CMA) and spent 15 years heading up the financial and operating departments at KUDU Industries, where he fostered financing arrangements, client relationships and manufacturing teams to take the organization from $35M to $150M in revenue. He has extensive experience turning companies around, growing them and building efficient organizations.
Prospera Energy Inc. (OTC: GXRFF), closed Friday's trading session at $0.096, up 2.5641%, on 15,500 volume. The average volume for the last 3 months is 195,282 and the stock's 52-week low/high is $0.0352/$0.134.
Recent News
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) On Track for Expansion in 2023 Amid Growing Oil and Gas Market in Canada
- MiningNewsBreaks - Why Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Is 'One to Watch'
- InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Initiates Coverage through IBN
Renovaro BioSciences Inc. (NASDAQ: RENB)
The QualityStocks Daily Newsletter would like to spotlight Renovaro BioSciences Inc. (NASDAQ: RENB) .
Renovaro BioSciences Inc. (NASDAQ: RENB), formerly Enochian BioSciences Inc., is an advanced, pre-clinical biotechnology firm in cell, gene and immunotherapy focused on solid tumors with short life expectancy. The company aims to unlock potentially long-term or life-long cancer remission in some of the deadliest cancers, and to potentially treat or cure serious infectious diseases such as Human Immunodeficiency Virus (HIV) and Hepatitis B Virus (HBV) infection.
The oncology platform is now at the forefront of Renovaro’s development activities. While Renovaro’s current efforts focus primarily on pancreatic cancer, it plans to include other solid tumors with short life-expectancy in the first in human Phase I/IIa studies that are on track to start by mid-2024. The company’s Pre-Investigational New Drug (pre-IND) submission included a human study plan covering pancreatic cancer, as well as other cancers that are difficult to treat, potentially including triple-negative breast cancer, head and neck cancers and mesothelioma.
Renovaro’s proprietary, novel technology uses cell- and gene-therapy to promote a renewed immune response against solid tumors. Important confirmatory results from two humanized mouse models using the company’s novel dendritic cell-based therapy, independently conducted by Dr. Anahid Jewett, a renowned cancer researcher in the field of immunotherapy at UCLA, were presented previously at two scientific conferences and were the foundation supporting a pre-IND submission to the U.S. Food and Drug Administration. Notably, Dr. Jewett’s findings from these studies consistently demonstrated 80% to 90% pancreatic tumor reduction in size and weight that was correlated with significant enhancement of key aspects of the immune response.
Renovaro is headquartered in Los Angeles, California.
RENB-DC11
Renovaro’s product development strategy is anchored in the use of “non-self” or allogeneic cells that enhance targeted immune response. Its lead candidate, RENB-DC11, is an innovative therapeutic vaccination platform that could potentially be used to induce life-long remissions from some of the deadliest solid tumors.
Treatment with RENB-DC11 has now been shown to significantly reduce the size of human pancreatic tumors in humanized mice in three independent studies. The reduction in tumor size correlated with statistically significant increases in key components of an immune response.
Pre-IND was completed in June 2023, with IND filing forecast for first half of 2024. First in-human Phase I/IIa trials are predicted shortly after in H1 of 2024, including pancreatic and other solid tumors with poor treatment options and life-expectancy.
Renovaro believes that RENB-DC11 could represent the most promising and effective strategy to achieve life-long remission for a number of common and deadly tumors.
Other Development Candidates
In addition to its lead oncology platform, Renovaro’s development pipeline includes a platform targeting infectious diseases, including:
- RENB-HV12 – An engineered allogeneic T-Cell vaccine, this therapeutic HIV vaccine candidate enhances immune infiltration, immune killing and immune surveillance. Potential pre-IND submission is planned for first half of 2024, with IND-submission expected in second half of 2024.
- RENB-HV21 – Leveraging allogeneic NK plus Gamma Delta T (GDT) cells as potential therapy for HIV, ENOB-HV21 shows promising preliminary results without confounding factors. Renovaro owns an exclusive license and has completed the Pre-IND submission, with a potential IND submission and human trials expected in 2024.
- RENB-HV01 – Caring Cross, a non-profit corporation, has shown that its proprietary CAR-T cells cure HIV in a mouse model. Studies in humans have begun. Renovaro has entered into a profit-sharing sublicense with Caring Cross and would share in profits if the product is commercialized.
- RENB-HB01 – This therapeutic approach aims to eliminate all HBV rapidly (“seek and kill”) with a two to three dose treatment regimen. It is expected to be applicable for early disease to maximize impact with low risk of toxicity. Pre-IND comments have been received from the FDA for its AAV-delivery system.
LOI to Merge with GEDi Cube International Ltd.
On August 9, 2023, Renovaro announced its execution of a binding, exclusive letter of intent to merge a subsidiary with cutting-edge health AI company GEDi Cube International Ltd. The combined company is expected to create a potential multiplier effect to accelerate earlier diagnosis, more effective therapy, and precision in silico drug discovery.
GEDi Cube’s innovative technology, developed over nearly a decade, has already validated earlier diagnoses of lung cancer in humans at a leading university hospital. GEDi Cube has likewise created the early diagnosis technology for 12 additional cancers, including pancreatic and breast cancer.
“I believe joining forces with GEDi Cube could enhance the efficacy of our upcoming trials and speed up the discovery of novel treatment approaches, thereby extending our life-saving technology to more cancer patients and renewing hope for them and their families,” Dr. Mark Dybul, CEO of Renovaro, stated in the news release.
GEDi Cube is led by CEO Craig Rhodes, who brings to that company tremendous industry experience leading life sciences groups at industry leaders Intel, Oracle and NVIDIA.
Market Opportunity
Pancreatic cancer alone is diagnosed globally in approximately 495,000 people each year, including roughly 64,000 in the U.S. Nearly 466,000 of those patients die annually, including approximately 51,000 in the U.S. Because of limited treatment options, life expectancy is very poor – with an approximately 10% patient survival rate at five years after diagnosis.
The global pancreatic cancer treatment market was valued at $2.15 billion in 2021 and is projected to grow from $2.48 billion in 2022 to $6.85 billion by 2029, according to Fortune Business Insights. That growth represents a CAGR of 15.7% for the forecast period.
A separate report from Fortune Business Insights projects that the global HIV drug market will grow from $30.46 billion in 2021 to $45.58 billion in 2028, recording a CAGR of 5.9% over the forecast period.
According to GlobalData, the value of the market for hepatitis B treatment is forecast to experience a significant increase in the coming years, with revenues expected to grow from $1.6 billion in 2022 to $10.5 billion in 2029. That represents a very rapid CAGR of 30% over the period. An estimated 296 million people suffer from the condition worldwide.
Management Team
Dr. Mark Dybul is the CEO of Renovaro. He has served as a tenured professor in the Department of Medicine at Georgetown University Medical Center since June 2017. He also served as Faculty Co-Director of the Center for Global Health and Quality from 2017-2021. Dr. Dybul has worked on HIV and public health for nearly 30 years as a clinician, scientist, teacher and administrator, including as an architect and eventually the Global Ambassador of the U.S. President’s Emergency Plan for AIDS Relief and the Executive Director of the Global Fund to Fight AIDS, Tuberculosis and Malaria from 2013 through May of 2017, and as the co-director of the Global Health Law Program at the O’Neill Institute for National and Global Health Law from 2009 through 2012. He is a member of the U.S. National Academy of Medicine.
Luisa Puche is the company’s CFO. She has served as a senior accounting and financial advisor and president of Puche Group LLC from 2015-2019. She served in various key executive roles, including Interim Chief Accounting Officer, at Brightstar Corp., a $10 billion global wireless device services provider. Ms. Puche began her career at Ernst & Young, where she served for approximately 10 years. Leveraging her broad global audit, advisory and corporate expertise, she has provided strong cross-functional leadership experience managing small and large projects for both publicly traded and privately held companies in various industries, including a global implementation of the latest revenue recognition accounting standard for Del Monte, as well as the global implementation of their SOX-404 program.
Francois Binette, Ph.D., is the Chief Operating Officer and Executive Vice President of Research & Development at Renovaro. He has over 25 years of product development expertise in Advanced Therapies and Regenerative Medicine. His broad industry experience spans a wide range of serious medical conditions, from orthopedics to ophthalmology, CNS and immuno-oncology. His career includes positions at Genzyme, Biosyntech, the DePuy Franchise of Johnson and Johnson, Medtronic and Lineage Cell Therapeutics. He received his Ph.D. from Laval University in Québec, followed by post-doctoral training at the Sanford-Burnham Institute in La Jolla and Harvard Medical School in Boston.
Renovaro BioSciences Inc. (NASDAQ: RENB), closed Friday's trading session at $3.2, off by 0.928793%, on 313,419 volume. The average volume for the last 3 months is 975,036 and the stock's 52-week low/high is $0.3928/$3.59.
Recent News
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Tracking Off-Label Cancer Treatment Could Ease Future Trials, Remedies
- InvestorNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB) Congratulates GEDiCube on New Joint Effort Clinical Trials
- Renovaro BioSciences Inc. (NASDAQ: RENB) Aims to Unlock Potential Treatments and Cures for Serious Infectious Diseases and Cancer
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) to Roll Out New LMS Integration Capability
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) Is 'One to Watch'
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp to Acquire Additional Private Lands at its Kay Mine Project
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Announces Expanding Coverage in Behavioral Health Field for Acute and Ambulatory Care Settings
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Announces Corporate Update, $2M Debt Financing
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Rebrands as Scinai Immunotherapeutics Ltd.
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: XPHYF) (FSE: 4XT) - BioNxt Solutions Announces Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Completes $29.8M Issue of Ordinary Shares
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Releases First ESG Report
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Looking Forward to Upcoming Interim Results for Potentially Pivotal GBM Study
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Names New Member of Board of Directors
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Receives Notification of Approval for U.S. Patent Covering Deuterated Tryptamines
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Publishes New Research Backing Its Approach to Outcome-Based Engagement
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Announces Update to Constrained Quadratic Model Hybrid Solver
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - TechMediaBreaks - Electronic Servitor Publication Network Inc.'s (XESP) Unique Digital Engagement Engine(TM) Creates CX Edge
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - MiningNewsBreaks - Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Recent Models Suggest Potential Access to High-Grade Deposit
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Releases Q2 2023 Financial, Milestone Report
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces SOP of Next-Gen Coil Driver(TM) Technology
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) CEO Featured in C-Level Interview with BestGrowthStocks.Com
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Helping Implement Solutions Across Changing Financial Landscape
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) CEO Discusses Metal-Rich Properties, Potential in Recent Podcast
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Intersects 1.29% Li2O over 23 Meters at Flagship Augustus Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - Foresight: Eye-Net and SoftBank Corp. Collaborate to Introduce Cross-Collision Prevention V2X Solution Utilizing 5G MEC
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Record Capacity Levels for Fr8Fleet
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - TinyGemsBreaks - GEMXX Corp. (GEMZ) Eyes Promising Start to Expected 'Highly Lucrative' First Year of Gold Mining
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Seeks Approval for Warrant Repricing
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - InvestorNewsBreaks – GolfLync Inc.’s User-Friendly and Feature-Rich Platform Driving Exceptional Growth, Engagement
- Genprex Inc. (NASDAQ: GNPX) - Genprex Inc. (NASDAQ: GNPX) Obtains New License for Novel Diabetes Gene Therapy Pipeline
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) 1-for-10 Reverse Stock Split Trading to Begin Today
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Releases Q2 2023 Financial Report, Corporate Update
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Completes Design Concept for Next-Generation EV Inverter
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Blog Points Out Benefits of Adding NFTs to More Traditional Customer/Fan Rewards Programs
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Study Suggests Marijuana Users Are Less Predisposed to Type 2 Diabetes
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Moves Operations to Hong Kong in Strategic Move to Expand Global Presence
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) to Present at H.C. Wainwright 25th Annual Global Investment Conference
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Retains Litigation Firm to Investigate Frivolous Claims, Unauthorized Tagging of Its Stock
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Deploys K5 ASRs for New Client, 2nd-Year Contract Renewal
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Longeveron Inc. (NASDAQ: LGVN) Is 'One to Watch'
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - CBDNewsBreaks - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) 'Strategically Important' Patent Grants Protection in One of World's Leading Oral Nicotine Markets
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Lucy Scientific Discovery Inc. (NASDAQ: LSDI) Is 'One to Watch'
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Adds New Member to Board of Directors
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Receives CSE Listing Conditional Approval
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures SFDA Approval for sugarBEAT(R)
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Being Clear on Value Proposition Key to eCommerce Success
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Receives Delisting Notification from NASDAQ, Files an Appeal
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTC: PLTXF) (Frankfurt: WNT1) Adds Chicago's First Vegan Omakase by Bloom Sushi to XMarket Food Hall Lineup
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Releases Q2 2023 Financial, Operational Report
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Amended, Restated Q3 2023 MD&A
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces 'Key Transition' Focused on Sale, Installation, Qualification of BaroFold Manufacturing System
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Participate at Upcoming H.C. Wainwright Global Investment Conference
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) On Track for Expansion in 2023 Amid Growing Oil and Gas Market in Canada
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTC: RWBYF) Announces Results for Three and Six Months Ended June 30, 2023
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Tracking Off-Label Cancer Treatment Could Ease Future Trials, Remedies
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - BMI Predicts Lithium Shortfall in 2025
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) Poised to Grow in Country with Exceptional Economic Forecast
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - RJD Green Inc.'s (RJDG) Subsidiary IOSoft Systems Enables Digital Evolution for Healthcare Payers and Providers
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - BioMedNewsBreaks - Why RVL Pharmaceuticals plc (NASDAQ: RVLP) Is 'One to Watch'
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Creates New Subsidiary to Combat N2O Greenhouse Gas Emissions
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters LOIs to Participate in Leading Travel Brand, Sell Select IP Assets
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Announces LOI to Acquire NUGL/Kaya in a Transaction Valued at CAD $43,289,624.60; CSE to Halt Trading Until Shareholder Approval
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Helping Governments Improve Public Safety, Reduce Recidivism Rates through Suite of Proven Products Focused on Effectiveness of Home Confinement with Electronic Monitoring
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Receives SD& Handset, VK7 Vehicle Kit PO from Leading Healthcare Customer
- SOHM Inc. (OTC: SHMN) - InvestorNewsBreaks - SOHM, Inc. (SHMN) Announces Strategic Board Appointment of Dr. Krishna Bhat, MD, Ph.D., FACC
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Credit Facility to Fund Strategic Acquisitions
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Reports on Definitive Feasibility Study Positive Results
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Announces Whipshots® New Limited-Edition Pumpkin Spice Flavor
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Proudly Announces its 500th Special Edition 1-hour Broadcast with Guest Appearances, Airs on Newsmax, Saturday, September 9, 2023, 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Shares Status Update on Key Corporate Efforts
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Receives Nasdaq Notice on Late Filing of Its Form 10-Q
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Issues Response to Short-Seller's Statement
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Appoints Life Sciences Exec as New CEO
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - Superlatus Announces Planned Expansion into Pet Food Market
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Demonstrates Power of REE Processing During US Embassy Staff Visit to Kingston Plant
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Closes on Acquisition of Portfolio of Canadian Uranium Exploration Projects
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN), Fuji Pharma Co. Ltd. (TSE: 4554) Announce Exclusive Negotiation Agreement
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Releases Q2, YTD 2023 Financial, Operational Report
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies Unveils Critical Incident Management Virtual Reality Training Scenario for High-Stress Law Enforcement Environments
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) to Roll Out New LMS Integration Capability
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) Is 'One to Watch'
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp to Acquire Additional Private Lands at its Kay Mine Project
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Announces Expanding Coverage in Behavioral Health Field for Acute and Ambulatory Care Settings
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Announces Corporate Update, $2M Debt Financing
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Rebrands as Scinai Immunotherapeutics Ltd.
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: XPHYF) (FSE: 4XT) - BioNxt Solutions Announces Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Completes $29.8M Issue of Ordinary Shares
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Releases First ESG Report
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Looking Forward to Upcoming Interim Results for Potentially Pivotal GBM Study
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Names New Member of Board of Directors
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Receives Notification of Approval for U.S. Patent Covering Deuterated Tryptamines
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Publishes New Research Backing Its Approach to Outcome-Based Engagement
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Announces Update to Constrained Quadratic Model Hybrid Solver
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - TechMediaBreaks - Electronic Servitor Publication Network Inc.'s (XESP) Unique Digital Engagement Engine(TM) Creates CX Edge
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - MiningNewsBreaks - Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Recent Models Suggest Potential Access to High-Grade Deposit
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Releases Q2 2023 Financial, Milestone Report
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces SOP of Next-Gen Coil Driver(TM) Technology
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) CEO Featured in C-Level Interview with BestGrowthStocks.Com
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Helping Implement Solutions Across Changing Financial Landscape
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) CEO Discusses Metal-Rich Properties, Potential in Recent Podcast
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Intersects 1.29% Li2O over 23 Meters at Flagship Augustus Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - Foresight: Eye-Net and SoftBank Corp. Collaborate to Introduce Cross-Collision Prevention V2X Solution Utilizing 5G MEC
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Record Capacity Levels for Fr8Fleet
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - TinyGemsBreaks - GEMXX Corp. (GEMZ) Eyes Promising Start to Expected 'Highly Lucrative' First Year of Gold Mining
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Seeks Approval for Warrant Repricing
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - InvestorNewsBreaks – GolfLync Inc.’s User-Friendly and Feature-Rich Platform Driving Exceptional Growth, Engagement
- Genprex Inc. (NASDAQ: GNPX) - Genprex Inc. (NASDAQ: GNPX) Obtains New License for Novel Diabetes Gene Therapy Pipeline
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) 1-for-10 Reverse Stock Split Trading to Begin Today
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Releases Q2 2023 Financial Report, Corporate Update
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Completes Design Concept for Next-Generation EV Inverter
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Blog Points Out Benefits of Adding NFTs to More Traditional Customer/Fan Rewards Programs
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Study Suggests Marijuana Users Are Less Predisposed to Type 2 Diabetes
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Moves Operations to Hong Kong in Strategic Move to Expand Global Presence
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) to Present at H.C. Wainwright 25th Annual Global Investment Conference
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Retains Litigation Firm to Investigate Frivolous Claims, Unauthorized Tagging of Its Stock
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Deploys K5 ASRs for New Client, 2nd-Year Contract Renewal
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Longeveron Inc. (NASDAQ: LGVN) Is 'One to Watch'
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - CBDNewsBreaks - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) 'Strategically Important' Patent Grants Protection in One of World's Leading Oral Nicotine Markets
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Lucy Scientific Discovery Inc. (NASDAQ: LSDI) Is 'One to Watch'
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Adds New Member to Board of Directors
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Receives CSE Listing Conditional Approval
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures SFDA Approval for sugarBEAT(R)
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Being Clear on Value Proposition Key to eCommerce Success
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Receives Delisting Notification from NASDAQ, Files an Appeal
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTC: PLTXF) (Frankfurt: WNT1) Adds Chicago's First Vegan Omakase by Bloom Sushi to XMarket Food Hall Lineup
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Releases Q2 2023 Financial, Operational Report
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Amended, Restated Q3 2023 MD&A
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces 'Key Transition' Focused on Sale, Installation, Qualification of BaroFold Manufacturing System
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Participate at Upcoming H.C. Wainwright Global Investment Conference
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) On Track for Expansion in 2023 Amid Growing Oil and Gas Market in Canada
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTC: RWBYF) Announces Results for Three and Six Months Ended June 30, 2023
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Tracking Off-Label Cancer Treatment Could Ease Future Trials, Remedies
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - BMI Predicts Lithium Shortfall in 2025
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) Poised to Grow in Country with Exceptional Economic Forecast
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - RJD Green Inc.'s (RJDG) Subsidiary IOSoft Systems Enables Digital Evolution for Healthcare Payers and Providers
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - BioMedNewsBreaks - Why RVL Pharmaceuticals plc (NASDAQ: RVLP) Is 'One to Watch'
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Creates New Subsidiary to Combat N2O Greenhouse Gas Emissions
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters LOIs to Participate in Leading Travel Brand, Sell Select IP Assets
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Announces LOI to Acquire NUGL/Kaya in a Transaction Valued at CAD $43,289,624.60; CSE to Halt Trading Until Shareholder Approval
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Helping Governments Improve Public Safety, Reduce Recidivism Rates through Suite of Proven Products Focused on Effectiveness of Home Confinement with Electronic Monitoring
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Receives SD& Handset, VK7 Vehicle Kit PO from Leading Healthcare Customer
- SOHM Inc. (OTC: SHMN) - InvestorNewsBreaks - SOHM, Inc. (SHMN) Announces Strategic Board Appointment of Dr. Krishna Bhat, MD, Ph.D., FACC
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Credit Facility to Fund Strategic Acquisitions
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Reports on Definitive Feasibility Study Positive Results
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Announces Whipshots® New Limited-Edition Pumpkin Spice Flavor
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Proudly Announces its 500th Special Edition 1-hour Broadcast with Guest Appearances, Airs on Newsmax, Saturday, September 9, 2023, 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Shares Status Update on Key Corporate Efforts
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Receives Nasdaq Notice on Late Filing of Its Form 10-Q
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Issues Response to Short-Seller's Statement
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Appoints Life Sciences Exec as New CEO
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - Superlatus Announces Planned Expansion into Pet Food Market
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Demonstrates Power of REE Processing During US Embassy Staff Visit to Kingston Plant
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Closes on Acquisition of Portfolio of Canadian Uranium Exploration Projects
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN), Fuji Pharma Co. Ltd. (TSE: 4554) Announce Exclusive Negotiation Agreement
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Releases Q2, YTD 2023 Financial, Operational Report
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies Unveils Critical Incident Management Virtual Reality Training Scenario for High-Stress Law Enforcement Environments
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) to Roll Out New LMS Integration Capability
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) Is 'One to Watch'
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp to Acquire Additional Private Lands at its Kay Mine Project
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Announces Expanding Coverage in Behavioral Health Field for Acute and Ambulatory Care Settings
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Announces Corporate Update, $2M Debt Financing
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Rebrands as Scinai Immunotherapeutics Ltd.
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: XPHYF) (FSE: 4XT) - BioNxt Solutions Announces Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Completes $29.8M Issue of Ordinary Shares
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Releases First ESG Report
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Looking Forward to Upcoming Interim Results for Potentially Pivotal GBM Study
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Names New Member of Board of Directors
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Receives Notification of Approval for U.S. Patent Covering Deuterated Tryptamines
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Publishes New Research Backing Its Approach to Outcome-Based Engagement
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Announces Update to Constrained Quadratic Model Hybrid Solver
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - TechMediaBreaks - Electronic Servitor Publication Network Inc.'s (XESP) Unique Digital Engagement Engine(TM) Creates CX Edge
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - MiningNewsBreaks - Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Recent Models Suggest Potential Access to High-Grade Deposit
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Releases Q2 2023 Financial, Milestone Report
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces SOP of Next-Gen Coil Driver(TM) Technology
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) CEO Featured in C-Level Interview with BestGrowthStocks.Com
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Helping Implement Solutions Across Changing Financial Landscape
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) CEO Discusses Metal-Rich Properties, Potential in Recent Podcast
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Intersects 1.29% Li2O over 23 Meters at Flagship Augustus Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - Foresight: Eye-Net and SoftBank Corp. Collaborate to Introduce Cross-Collision Prevention V2X Solution Utilizing 5G MEC
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Record Capacity Levels for Fr8Fleet
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - TinyGemsBreaks - GEMXX Corp. (GEMZ) Eyes Promising Start to Expected 'Highly Lucrative' First Year of Gold Mining
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Seeks Approval for Warrant Repricing
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - InvestorNewsBreaks – GolfLync Inc.’s User-Friendly and Feature-Rich Platform Driving Exceptional Growth, Engagement
- Genprex Inc. (NASDAQ: GNPX) - Genprex Inc. (NASDAQ: GNPX) Obtains New License for Novel Diabetes Gene Therapy Pipeline
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) 1-for-10 Reverse Stock Split Trading to Begin Today
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Releases Q2 2023 Financial Report, Corporate Update
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Completes Design Concept for Next-Generation EV Inverter
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Blog Points Out Benefits of Adding NFTs to More Traditional Customer/Fan Rewards Programs
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - Study Suggests Marijuana Users Are Less Predisposed to Type 2 Diabetes
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Moves Operations to Hong Kong in Strategic Move to Expand Global Presence
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) to Present at H.C. Wainwright 25th Annual Global Investment Conference
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Retains Litigation Firm to Investigate Frivolous Claims, Unauthorized Tagging of Its Stock
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Deploys K5 ASRs for New Client, 2nd-Year Contract Renewal
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Longeveron Inc. (NASDAQ: LGVN) Is 'One to Watch'
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - CBDNewsBreaks - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) 'Strategically Important' Patent Grants Protection in One of World's Leading Oral Nicotine Markets
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Lucy Scientific Discovery Inc. (NASDAQ: LSDI) Is 'One to Watch'
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Adds New Member to Board of Directors
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Receives CSE Listing Conditional Approval
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures SFDA Approval for sugarBEAT(R)
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Being Clear on Value Proposition Key to eCommerce Success
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Receives Delisting Notification from NASDAQ, Files an Appeal
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (OTC: PLTXF) (Frankfurt: WNT1) Adds Chicago's First Vegan Omakase by Bloom Sushi to XMarket Food Hall Lineup
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Releases Q2 2023 Financial, Operational Report
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) Releases Amended, Restated Q3 2023 MD&A
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces 'Key Transition' Focused on Sale, Installation, Qualification of BaroFold Manufacturing System
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Participate at Upcoming H.C. Wainwright Global Investment Conference
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) On Track for Expansion in 2023 Amid Growing Oil and Gas Market in Canada
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTC: RWBYF) Announces Results for Three and Six Months Ended June 30, 2023
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Tracking Off-Label Cancer Treatment Could Ease Future Trials, Remedies
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - BMI Predicts Lithium Shortfall in 2025
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) Poised to Grow in Country with Exceptional Economic Forecast
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - RJD Green Inc.'s (RJDG) Subsidiary IOSoft Systems Enables Digital Evolution for Healthcare Payers and Providers
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - BioMedNewsBreaks - Why RVL Pharmaceuticals plc (NASDAQ: RVLP) Is 'One to Watch'
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Creates New Subsidiary to Combat N2O Greenhouse Gas Emissions
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters LOIs to Participate in Leading Travel Brand, Sell Select IP Assets
- Silo Pharma Inc. (OTCQB: SILO) - Silo Wellness Announces LOI to Acquire NUGL/Kaya in a Transaction Valued at CAD $43,289,624.60; CSE to Halt Trading Until Shareholder Approval
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Helping Governments Improve Public Safety, Reduce Recidivism Rates through Suite of Proven Products Focused on Effectiveness of Home Confinement with Electronic Monitoring
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Receives SD& Handset, VK7 Vehicle Kit PO from Leading Healthcare Customer
- SOHM Inc. (OTC: SHMN) - InvestorNewsBreaks - SOHM, Inc. (SHMN) Announces Strategic Board Appointment of Dr. Krishna Bhat, MD, Ph.D., FACC
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Credit Facility to Fund Strategic Acquisitions
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Reports on Definitive Feasibility Study Positive Results
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands Announces Whipshots® New Limited-Edition Pumpkin Spice Flavor
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Proudly Announces its 500th Special Edition 1-hour Broadcast with Guest Appearances, Airs on Newsmax, Saturday, September 9, 2023, 3:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Shares Status Update on Key Corporate Efforts
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - The Alkaline Water Company Receives Nasdaq Notice on Late Filing of Its Form 10-Q
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Issues Response to Short-Seller's Statement
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Appoints Life Sciences Exec as New CEO
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - Superlatus Announces Planned Expansion into Pet Food Market
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Demonstrates Power of REE Processing During US Embassy Staff Visit to Kingston Plant
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Closes on Acquisition of Portfolio of Canadian Uranium Exploration Projects
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN), Fuji Pharma Co. Ltd. (TSE: 4554) Announce Exclusive Negotiation Agreement
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Releases Q2, YTD 2023 Financial, Operational Report
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies Unveils Critical Incident Management Virtual Reality Training Scenario for High-Stress Law Enforcement Environments
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.









































