The QualityStocks Daily Stock List
- Semler Scientific, Inc. (SMLR)
- Adamis Pharmaceuticals (ADMP)
- Avenue Therapeutics (ATXI)
- Bright Health Group (BHG)
- Urban-Gro Inc. (UGRO)
- Mizuho Financial Group (MFG)
- Impact Fusion International (IFUS)
- Medivolve Inc. (COPRF)
- Elevate Credit (ELVT)
- Opendoor Technologies (OPEN)
- QuantumScape Corp. (QS)
- Hecla Mining Company (HL)
Semler Scientific, Inc. (SMLR)
QualityStocks, Wall Street Resources, MarketBeat, StreetInsider, Money Morning, MarketClub Analysis, Marketbeat.com, Daily Trade Alert and Barchart reported earlier on Semler Scientific, Inc. (SMLR), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Semler Scientific, Inc. is a medical risk-assessment Company headquartered in San Jose, California. Its mission is to develop, manufacture, and market patented products that identify the risk profile of medical patients to allow healthcare providers to capture full reimbursement potential for their services. Semler Scientific provides technology and software solutions to improve the clinical effectiveness of healthcare insurers and physician groups. Semler Scientific lists on the OTC Markets Group’s OTCQB.
In essence, Semler Scientific provides diagnostic and testing services to the U.S.’s leading health plans and providers. The Company’s goal is to develop, manufacture, and market innovative proprietary products and services, which assist its customers in evaluating and treating chronic diseases.
Semler Scientific manufactures the QuantaFlo™ system for Vascular Disease testing. The QuantaFlo™ system is very suitable for use in primary care offices, specialty practices, health fairs, or during home assessments.
The QuantaFlo™ PAD test delivers quick, accurate results in approximately five minutes at the point of care. In March 2015, The Company received Food and Drug Administration (FDA) 510 (k) clearance for the next generation version of QuantaFlo™, which commercially launched in August 2015. The QuantaFlo software can be installed on any Windows based PC, laptop or tablet.
Semler Scientific's first patented and FDA cleared product, introduced commercially in 2011, measured arterial blood flow in the extremities to aid in the diagnosis of peripheral arterial disease.
In addition, Semler Scientific has its WellChec™ service. WellChec™ provides turn-key assessment testing across the U.S. for an array of conditions. It provides turnkey solutions for administering vital clinical tests that can impact HCC classifications, CPT coding, HEDIS and Quality Measures. In April 2015, Semler launched its multi-test service platform, WellChec™ .
Semler Scientific, Inc. (SMLR), closed Tuesday's trading session at $41.72, up 7.5258%, on 29,823 volume. The average volume for the last 3 months is 2.917M and the stock's 52-week low/high is $27.02/$153.21.
Adamis Pharmaceuticals (ADMP)
MarketBeat, StockMarketWatch, MarketClub Analysis, AllPennyStocks, QualityStocks, BUYINS.NET, TraderPower, The Street, Barchart, Investors Insights, Microcap MarketPlace, Money Morning, StreetInsider, The Campaign for a Sound Dollar, Schaeffer's, Marketbeat.com, Wall Street Resources, Triple Crown Stocks, Daily Trade Alert, Dynamic Wealth Report, FeedBlitz, Investing Futures, Investors Alley, TradersPro, SmallCapVoice, The Online Investor, Microcap Money, StocksEarning, Wealth Insider Alert, SeriousTraders and Trades Of The Day reported earlier on Adamis Pharmaceuticals (ADMP), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Adamis Pharmaceuticals Corporation (NASDAQ: ADMP) (FRA: CY3B) is a specialty biopharmaceutical firm which is engaged in the development, commercialization and distribution of pharmaceutical products in the therapeutic areas of respiratory, allergy, infectious, immunology and oncology diseases.
Adamis Pharmaceuticals Corp. serves consumers in the United States and is based in San Diego, California. The firm was established in June 2006 and has a licensed vaccine technology known as somatic transgene immunization technology.
Adamis Pharmaceuticals Corporation’s product portfolio includes the epinephrine injection pre-filled syringe, which is used in the emergency treatment of acute allergic reactions, include anaphylaxis. The firm also provides dry powder products made up of naloxone injection product candidates indicated for the treatment of an opioid overdose, APC-6000; APC-8000 also known as Tadafil which is indicated for the treatment of erectile dysfunction; APC-1000 or beclomethasone, a metered dose inhaler product indicated for asthma and APC-4000 or fluticasone, indicated for the treatment of asthma. Additionally, the firm offers women’s and men’s health products, topical compounds for pain, urological preparations, injectables, hospital outsourcing product, hormone replacement therapies, corticosteroids and some animal veterinary pharmaceutical products.
Adamis Pharmaceuticals Corporation recently publicized its submission of a new investigational drug to the FDA developed as a coronavirus treatment. The drug; Tempol, has shown both potent antioxidant, anticoagulant and anti-inflammatory activity, which shows that the drug could play a crucial role in not only treating the coronavirus but also preventing hospitalization. With new variants of the virus coming up, new therapies will be needed. Tempol may help fill this role, especially if the new vaccines aren’t effective in protecting against these new mutations.
Adamis Pharmaceuticals (ADMP), closed Tuesday's trading session at $0.1464, up 16.3752%, on 5,955,324 volume. The average volume for the last 3 months is 3.155M and the stock's 52-week low/high is $0.12/$1.48.
Avenue Therapeutics (ATXI)
QualityStocks, MarketClub Analysis, StocksEarning, MarketBeat, Zacks, StreetInsider, StockMarketWatch, Schaeffer's, TradersPro, Marketbeat.com, INO Market Report, TradersPro Morning, The Online Investor, SmallCapVoice and PoliticsAndMyPortfolio reported earlier on Avenue Therapeutics (ATXI), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Avenue Therapeutics Inc. (NASDAQ: ATXI) (FRA: 49Y) is a specialty pharmaceutical firm that is engaged in the acquisition, licensing, development and commercialization of products that are mainly used in the intensive/acute care hospital setting.
The firm has its headquarters in New York and was incorporated in 2015, on February 9th. It operates in the healthcare sector, under the biotech and pharma sub-industry. The company is a majority-controlled subsidiary firm of Fortress Biotech Inc., which is a biopharmaceutical firm that is also engaged in the acquisition, development and commercialization of new biotechnology and pharmaceutical products.
The company develops intravenous Tramadol, which is currently undergoing phase 3 clinical trials for the treatment of moderate to severe post-surgical pain. The synthetic dual-acting opioid works by inhibiting noradrenaline and serotonin re-uptake and blocking spinal level nociceptive impulses and could be used as an alternative for conventional opioids. The oral formulation of this candidate is marketed in the U.S. for moderate to severe pain in grown-ups. Its parenteral formulations include subcutaneous and intramuscular formulations. The drug itself is marketed in the U.S. under the Ultram trade name. Ultracet, which is a combination product of acetaminophen and tramadol, is also sold in the U.S.
The firm is pursuing regulatory approval for its IV tramadol drug formulation, after the FDA cited in their Complete Response Letter that they did not find any controls, manufacturing or chemistry issues with its candidate. If successful, the firm will introduce the drug to the market, which will increase its revenue and boost share prices as well encourage more investments into the firm.
Avenue Therapeutics (ATXI), closed Tuesday's trading session at $2.84, up 10.0775%, on 3,381,837 volume. The average volume for the last 3 months is 10.182M and the stock's 52-week low/high is $2.2901/$27.75.
Bright Health Group (BHG)
QualityStocks, MarketBeat, Trades Of The Day, Daily Trade Alert and Schaeffer's reported earlier on Bright Health Group (BHG), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Bright Health Group Inc. (NYSE: BHG) is an integrated care delivery firm that is focused on the delivery and financing of health insurance plans.
The firm has its headquarters in Minneapolis, Minnesota and was incorporated in 2015. Prior to its name change in February 2021, the firm was known as Bright Health Inc. It operates as part of the insurance carriers industry. The firm has seven firms in its corporate family and serves consumers in the United States.
The company aligns the best local resources in healthcare delivery with the financing of care, which helps lower costs, decrease systemic waste, optimize clinical outcomes and drive superior consumer experience. It generates revenue from income from investments as well as premiums, which include fee-for-service provider revenue received from payers and consumers, and value-based provider revenue.
The enterprise operates through the Bright Health Care and NeueHealth segments. Through the former segment, it provides commercial health plan products and Medicare products to roughly 620,000 consumers in 199 markets and fourteen states. The enterprise, through its latter segment, delivers high-quality in-person and virtual clinical care to about 75,000 patients via its 61 affiliated risk-bearing primary care clinics. In addition to this, it offers services through Care Partners, a network of hospitals, clinics and doctors.
The company has a strategic investment with Cigna Ventures, which brings deep healthcare expertise that complements the company’s mission of expanding access to affordable and quality healthcare. This move will also bring in more investors into the company as well as extend their consumer reach, which will be good for its growth.
Bright Health Group (BHG), closed Tuesday's trading session at $1.17, even for the day, on 24,899 volume. The average volume for the last 3 months is 209,463 and the stock's 52-week low/high is $0.80/$9.58.
Urban-Gro Inc. (UGRO)
Schaeffer's, QualityStocks, MarketBeat and The Street reported earlier on Urban-Gro Inc. (UGRO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Urban-Gro Inc. (NASDAQ: UGRO) is involved in the integration of environmental equipment systems for the creation of indoor cultivation facilities for the horticulture market.
The engineering design services firm has its headquarters in Lafayette, Colorado and was incorporated in 2014, on March 20th by Octavio Gutierrez and Bradley J. Nattrass. It operates as part of the farm and heavy construction machinery industry, under the industrials sector. The firm serves consumers around the globe, with a focus on the commercial horticulture market in Europe, Canada and the U.S.
The enterprise provides design and engineering services, which include cultivation design, cultivation space programming and full-facility plumbing, electrical and mechanical engineering, equipment and facility commissioning services, training services which range from staff training sessions to standard operating procedures, program pricing and overview, and related-party software and hardware platforms. It also provides an integrated suite of crop management products and cultivation equipment systems, which include purpose-built heating, ventilation and air conditioning equipment systems, fertigation and irrigation distribution, wastewater reclamation and water treatment systems, and environmental controls, microbial mitigation and odor reduction systems, specialty fans, rolling and automated container benching systems, and commercial horticulture lighting solutions.
The company, which recently completed its acquisition of Emerald C.M., announced its latest financial results, which show significant increases in its revenues. It remains focused on meeting its consumer needs following the addition of construction management services to its portfolio, and enhancing shareholder value and driving long-term growth, which will positively influence its investments.
Urban-Gro Inc. (UGRO), closed Tuesday's trading session at $3.85, up 13.2353%, on 209,512 volume. The average volume for the last 3 months is 2.275M and the stock's 52-week low/high is $2.74/$14.77.
Mizuho Financial Group (MFG)
Louis Navellier, MarketBeat, The Street, Wall Street Resources, StreetInsider, InvestorPlace, TopStockAnalysts, StockMarketWatch, Daily Trade Alert, Marketbeat.com, SmarTrend Newsletters, StreetAuthority Daily, TheStockAdvisors, Trades Of The Day, Wyatt Investment Research, The Online Investor, AllPennyStocks, Penny Invest, Navellier Growth, MarketClub Analysis, Uncommon Wisdom, Wallstreetbuzz, Investor Update, INO.com Market Report, WallstreetsHotteststocks and StockEgg reported earlier on Mizuho Financial Group (MFG), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Mizuho Financial Group, Inc. (NYSE: MFG) (OTC: MZHOF) (TYO: 8411) (FRA: MZ8) (BCBA: MFG) is a holding firm that is engaged in the provision of financial services, including trust banking, banking, securities and other businesses related to financial services.
The firm has its headquarters in Tokyo, Japan and was incorporated in 1864. It operates as part of the banks-regional industry, under the financial services sector. The firm serves consumers around the globe, with a focus on those in Europe, the Americas, Asia/Oceania and Japan.
The enterprise operates through the Asset Management Company, Global Markets Company, Global Corporate Company, Corporate & Institutional Company and Retail & Business Banking Company segments. It offers syndicated, housing, and card loans; deposit products; business matching services; and advisory services related to mergers and acquisitions and overseas expansions. It also provides consulting services, which includes payroll services; asset succession and asset management; and sells lottery tickets issued by prefectures and ordinance-designated cities. In addition to this, the enterprise provides financial solutions, such as bonds, mergers and acquisitions advisory, risk hedging products, etc. for corporate clients to meet their needs in investment management, fund-raising and financial strategies; financial services that include funding support and public bonds underwriting; advisory services and solutions, such as advice on proposals on different investment products to financial institutions; real estate agency services; and solutions based on their business strategy, capital management and financial strategy.
The company plans to acquire Rakuten Securities’ fintech business. This move may help speed up Mizuho’s shift to digital banking, which will generate additional investments while facilitating its entrance into the burgeoning field.
Mizuho Financial Group (MFG), closed Tuesday's trading session at $2.25, even for the day. The average volume for the last 3 months is 46,663 and the stock's 52-week low/high is $2.15/$2.94.
Impact Fusion International (IFUS)
Penny Stock Chaser, Nebula Stocks, NYC Marketing Inc, Wallstreetlivechat, The Dean, Beacon Equity Research, Super Hot Penny Stocks, Stock Exploder, Liquid Tycoon, FeedBlitz, NanoCap Gems, MicroStockProfit, MicrocapVoice, GusherStocks, Bull in Advantage, Light Speed Stocks, AlphaTrade, Penny Stock MoneyTrain, WallstreetsHotteststocks, Penny Stock Pick Alert, Penny Stock Pick Report, PennyStockRumors.net, PennyTrader, SpeculatingStocks, Stock Source, The Cervelle Group and Penny Stock Finder reported earlier on Impact Fusion International (IFUS), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Impact Fusion International Inc. (OTC: IFUS) is a firm that is involved in the business of marketing products in the health and wellness sector.
The firm has its headquarters in Margate, Florida and was incorporated in 2002. Prior to its name change in April 2009, the firm was known as Red Reef Laboratories International Inc. It operates as part of the advertising agencies industry, under the communication services sector. The firm serves consumers around the globe.
The company’s mission is to market products to all international markets through research-validation-commercialization, for the well-being and health of humans and animals.
The enterprise invents, develops and markets a range of products, which include its Nutri-Mastic proprietary blend that has been designed to support digestive health and function, vital organ health, the immune system, cardiac muscle and heart function and healthy blood cells. It also offers Intact Endurance, which provides nutrients to increase optimal energy levels in the body; and Instant Nutrition herbal health support products. In addition to this, the enterprise also offers Equine Intact products and Pet Intact nutritional supplements. Further, it sells alternative proprietary formulations for dairy and beef cattle under the Supreme Gold Plus brand; and herbal health support beverages under the Mastic Blast Beverage brand.
The firm recently announced that it had received authorization from the USDA to submit a grant application for its Supreme Gold brand technology. The funds will increase commercialization of the firm’s Supreme Gold plus brands domestically and in the future, facilitate its expansion into the global marketplace. This will drive investments into the firm and help create shareholder value.
Impact Fusion International (IFUS), closed Tuesday's trading session at $0.018, off by 2.7027%, on 46,663 volume. The average volume for the last 3 months is 6,553 and the stock's 52-week low/high is $0.005/$0.03815.
Medivolve Inc. (COPRF)
QualityStocks and Emerging Markets reported earlier on Medivolve Inc. (COPRF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Medivolve Inc. (OTC: COPRF) (FRA: 4NC) is a health care firm that is focused on the commercialization of technologies to help combat the coronavirus.
The firm has its headquarters in Toronto, Canada and was incorporated in 2005, on November 8th. Prior to its name change in October 2009, the firm was known as Continent Resources Inc. It operates as part of the diagnostics and research industry, under the healthcare sector. The firm serves consumers in North America, with a focus on Canada.
The company is seeking ground-breaking innovations, technologies and exclusive partnerships to help fight Covid-19 and generate risk-adjusted returns for investors. It hopes to transform human health management.
The enterprise offers a clinical diagnostic network, a telehealth platform and a data driven AI framework to improve patient care. It also provides a network of retail collection sites which offer a range of point-of-care services, including clinical services in diagnostic testing and vaccinations. This is in addition to being involved in the development of technologies to prevent, identify and treat medical issues. The enterprise offers solutions in Covid-19 testing and immunity scoring. Its testing services include the antigen test, rapid antibody blood test and the reverse transcription- polymerase chain reaction test (RT-PCR test). It uses its channel partner Aditxt Therapeutics Inc. to provide Covid-19 immunity status monitoring via its mobile testing centers.
The firm recently announced its latest financial results, which show significant increases in revenue growth and gross profits. It plans to expand its testing services beyond the coronavirus, which will facilitate its entrance into new markets and help generate value for its shareholders.
Medivolve Inc. (COPRF), closed Tuesday's trading session at $0.0111, off by 1.7699%, on 6,553 volume. The average volume for the last 3 months is 61,919 and the stock's 52-week low/high is $0.0073/$0.20.
Elevate Credit (ELVT)
Marketbeat, Zacks, StreetInsider, StockMarketWatch, InvestorPlace, Trades Of The Day, MarketClub Analysis and BUYINS.NET reported earlier on Elevate Credit (ELVT), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Elevate Credit Inc. (NYSE: ELVT) (LON: 0XWD) (FRA: 0EV) is a company engaged in the provision of online credit solutions.
The firm has its headquarters in Fort Worth, Texas and was incorporated in 2014, on January 31st. It operates as part of the credit services industry, under the financial services sector. The firm serves consumers in the United Kingdom and the United States.
The company is committed to rewarding the good financial behavior of borrowers with features like rates of interest that can reduce over time, free credit monitoring and free financial training. It has a single reportable segment, which offers online financial services for subprime credit consumers.
The enterprise operates its Blueprint platform to provide unsecured online credit solutions to consumers who are not well-served by traditional bank products and are looking for alternative options to storefront installment loans, pawn, title loans and payday loans. Its products include a credit card product known as Today Card; a line of credit product dubbed Elastic; and an installment loan product known as Rise. The enterprise’s products use segment-optimized analytics and provide flexible rates and terms, free credit monitoring, credit bureau reporting, online financial literacy videos and tools and other related features. To date, the enterprise has originated over $10 billion in non-prime credit to over 2.7 million non-prime consumers.
The company’s recently released financial results show increases in its revenues. It remains focused on better meeting its consumers’ needs, which will bring in additional revenues into the company and bolster its overall growth.
Elevate Credit (ELVT), closed Tuesday's trading session at $1.05, even for the day. The average volume for the last 3 months is 16.677M and the stock's 52-week low/high is $1.04/$4.25.
Opendoor Technologies (OPEN)
All about trends, Schaeffer's, The Street, StreetInsider, MarketBeat, InvestorPlace, MarketClub Analysis, IT News Daily, SmarTrend Newsletters, StocksEarning, Street Insider, Forbes, The Online Investor, Trades Of The Day, Daily Trade Alert, StreetAuthority Daily, Zacks, internetnews, TheStockAdvisors, Hit and Run Candle Sticks, Wyatt Investment Research, Investopedia, Market Wrap Daily, Greenbackers, Eagle Financial Publications, CRWEWallStreet, Coattail Investor, Barchart, Investment House, QualityStocks, StockTwits, StreetAuthority Financial, Taipan Daily, The Best Newsletters, Wealth Daily, FNNO Newsletters, Tiny Gems, Dynamic Wealth Report, Today's Financial News, TheStockAdvisor, Trade of the Week, Inside Investing Daily, ChartPoppers, Wealth Insider Alliance, Daily Stocks, The Tycoon Report, Profit Confidential, Howard Erickson, Integrity Solution IR, The Stock Psycho, Investing Futures, The Stock Enthusiast, The Motley Fool, InvestorGuide, SmallCap Network, Money and Markets and The Trading Report reported earlier on Opendoor Technologies (OPEN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Opendoor Technologies Inc. (NASDAQ: OPEN) (FRA: 25M) (BMV: OPEN1) is a digital platform that is involved in the purchase, sale and trade-in of residential properties.
The firm has its headquarters in Tempe, Arizona and was incorporated in 2013, on December 30th by Eric Wu. It operates as part of the real estate services industry, under the real estate sector. The firm serves consumers in the United States.
The company’s mission is to empower every individual with the freedom to move and is led by a team made up of innovators, operators and problem solvers. It is focused on reinventing the most important transaction in life with a radically simple and new way to sell and buy one’s home. It has done so by rebuilding the whole consumer real estate experience, making selling and buying possible on a mobile device.
The enterprise allows sellers to use opendoor.com, where they can receive an offer, sign and pick their closing date. On the other hand, those who’d like to purchase a house can use the Opendoor app, which allows them to visit and tour both Opendoor and non-Opendoor homes and make an offer. The enterprise operates in a growing number of neighborhoods and cities across the U.S.
The company recently entered into a partnership with Zillow. This move will not only open the company up to new growth opportunities and help extend the company’s consumer reach but may also bring in additional revenues and investments into the company.
Opendoor Technologies (OPEN), closed Tuesday's trading session at $2.7, off by 8.1633%, on 16,703,762 volume. The average volume for the last 3 months is 6.18M and the stock's 52-week low/high is $2.66/$25.325.
QuantumScape Corp. (QS)
InvestorPlace, Schaeffer's, StocksEarning, MarketClub Analysis, The Street, MarketBeat, The Online Investor, Daily Trade Alert, Top Pros' Top Picks, wyatt research newsletter, Trades Of The Day, TipRanks, CNBC Breaking News, BUYINS.NET and Atomic Trades reported earlier on QuantumScape Corp. (QS), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
With several countries across the world looking to reduce carbon emissions from transportation, zero-emission electric cars may become exceedingly common on roads in the next few decades. But even though electric cars produce zero emissions at the tailpipe, some critics have argued that they are not 100% green because they are powered by coal-generated electricity.
To be completely free of emissions, electric vehicles would have to run on electricity that was generated via renewable sources. The U.S. government has been instrumental in encouraging electric vehicle adoption through subsidies that have made EVs somewhat affordable. Sources say that authorities are now looking to make electric cars eligible for biofuel credits.
A proposal by the U.S. Environmental Protection Agency (EPA) to add electric vehicles to the federal Renewable Fuel Standard (RFS) would be one of the most significant changes to the program since it was launched more than a decade ago. According to sources familiar with the issue, the EPA expects to send the proposals to the White House by the end of this week for approval.
Although the EPA hasn’t confirmed the details of the proposal, spokesman Tim Carroll said that the agency is keen on meeting deadlines to implement the Renewable Fuel Standard program. The program would require that oil refiners either blend biofuels into America’s fuel pool or buy RINS (tradable) credits from refiners that choose the first option. If electric vehicles are included in the program, automakers will have access to a new kind of credit called electric RINS or e-RINS. Industry players also say that EV charging companies and landfills that sell biogas to power plants could benefit from the new credit.
With America looking to replace the gas-powered vehicles on its roads with zero-emission electric cars, e-RINS could help accelerate EV adoption. Electric vehicles are much more expensive compared to traditional cars, making it impossible for a significant chunk of drivers to go electric. Even with federal and state subsidies, electric cars remain out of the reach of many Americans.
Reuters reported in June that the Biden administration had turned to Tesla for guidance on how to include electric cars in the country’s renewable fuel subsidy program. Emails seen by Reuters revealed that the administration reached out to the mammoth electric car maker in its first days and has held a series of meetings with vehicle makers in the EV space since as it worked on expanding the RFS program to electric cars.
As the uptake of EVs grows, companies such as QuantumScape Corp. (NYSE: QS) are likely to see demand grow for their solid-state batteries in order to address the stationary power storage needs as well as to serve the expanding electric vehicle market.
QuantumScape Corp. (QS), closed Tuesday's trading session at $8.07, even for the day, on 21,814 volume. The average volume for the last 3 months is 9.682M and the stock's 52-week low/high is $7.72/$43.08.
Hecla Mining Company (HL)
MarketClub Analysis, SmarTrend Newsletters, InvestorPlace, Schaeffer's, Wyatt Investment Research, Lebed.biz, StocksEarning, MarketBeat, Top Pros' Top Picks, TopStockAnalysts, StreetAuthority Daily, INO.com Market Report, The Street, Money Morning, QualityStocks, Zacks, Marketbeat.com, Jason Bond, Kiplinger Today, Daily Trade Alert, StreetInsider, Today's Financial News, Wall Street Grand, Trades Of The Day, TheStockAdvisors, Streetwise Reports, StockOodles, The Wealth Report, TradersPro, Gryphon Digest, Penny Detectives, National Inflation Association, SureMoney, TradingAuthority Daily, Stockhouse, Darwin Investing Network, ChartAdvisor, PennyStockLive, Options Elite, INO Market Report, Wall Street Daily, Penny Sleuth, Profit Confidential, ProfitableTrading, TraderPower, The Growth Stock Wire, Daily Markets, Greenbackers, Forbes, DrStockPick, Wealth Insider Alert, TradingMarkets, WealthMakers, Investopedia, CustomerService, CRWEWallStreet, CRWEPicks, CRWEFinance, Weiss Research, BestOtc, Barchart, Daily Wealth, MarketArmor.com, AllPennyStocks, PennyToBuck, MonsterStocksPicks, Residual Income Report, Money and Markets, Rockwell Trading, Investing Futures, Stock Stars, Traders For Cash Flow, StockHotTips, Trade of the Week, InvestorGuide, Investor Update, Investor Guide, Investing Lab, PennyOmega and SmallCapVoice reported earlier on Hecla Mining Company (HL), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Earlier last week, gold continued its rally surpassing its $1,700 an ounce price. This comes as weak data drives a shift in sentiment in the precious metals market. The price of spot gold gained a little over 1% to reach $1,719.04 an ounce. This is its highest price in three weeks.
Meanwhile, benchmark 10-year treasury yields in the United States declined to a new low as the dollar extended its decline, which in turn, made gold cheaper for other currency holders.
Experts believe that bullion is coming nearer to its 50-day moving average after its surge last Monday when a U.S. manufacturing gauge plunged more than expected. Since April, the price has been trading below the technical market, which is a sign of poor sentiment, which saw the metal decline into a bear market.
Currently, the safe-haven metal is at a critical juncture as traders wait for nonfarm payrolls for more insights on the path the central bank will take on monetary policy in the future. Experts argue that a weaker-than-expected print may cause a further decline in rate hike expectations, which will in turn strengthen bullion at a technical turning point.
In a Bloomberg note, ABN Ambro Bank NV analyst Georgette Boele stated that gold had surpassed the $1,700 per ounce mark again, which meant that the break below $1,650–$1,700 per ounce was a false break.
Other U.S. employment data, which includes figures from the ADP Research Institute and job openings, will also be crucial this week. In an interview with Reuters, RJO Futures market strategist Bob Haberkorn stated that if data released on the jobs came out much weaker than expected, gold would rally.
Haberkorn added that if it came out much stronger than expected, the market could interpret that as well, which meant that the Federal Reserve could keep going higher with rates.
Thus far, the Fed has struggled with weakening the labor market using a tighter central bank policy in an effort to fight inflation. Tighter policy affects gold negatively by pushing up bond yields, which in turn reduces the attractiveness of the nonyielding asset.
Expectations for even more aggressive hike in rates in the United States and safe haven flows have also driven a dollar rally, which is negatively correlated with this particular precious metal. In her note, Boele added that investors weren’t as interested in the dollar as a safe haven, noting that risk aversion had also eased.
Precious metals companies such as Hecla Mining Company (NYSE: HL) will be watching the market movements closely to see in which directions the coming weeks and months are likely to go in terms of commodity prices.
Hecla Mining Company (HL), closed Tuesday's trading session at $4.38, even for the day, on 12,354 volume. The average volume for the last 3 months is 48,823 and the stock's 52-week low/high is $3.41/$7.66.
The QualityStocks Company Corner
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF)
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF)
- Delic Holdings Inc. (CSE: DELC) (OTCQB: DELCF)
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN)
- QSAM Biosciences Inc. (OTCQB: QSAM)
- Odyssey Group International Inc. (OTC: ODYY)
- Knightscope Inc. (NASDAQ: KSCP)
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI)
- Flora Growth Corp. (NASDAQ: FLGC)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- GeoSolar Technologies Inc.
- HeartBeam Inc. (NASDAQ: BEAT)
Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F)
The QualityStocks Daily Newsletter would like to spotlight Hillcrest Energy Technologies Ltd. (OTCQB: HLRTF).
Hillcrest Energy Technologies (CSE: HEAT) (OTCQB: HLRTF) , a clean-technology company developing transformative power conversion technologies and control system solutions for next-generation EVs and electrical systems, has named a new chief of commercialization. The company has appointed James Bolen, an advisor to the company, to the position. Bolen has decades of experience developing and commercializing products designed for automotive and industrial power spaces. Bolen noted that HLRTF’s development program is focused on a high level of customer intimacy, which ensures consistent customer requirements and product specifications, followed by application-specific customization. The company has dedicated itself to assembling an experienced, capable leadership team that is committed to moving Hillcrest technologies to market. “James’ extensive knowledge, experience and network will continue to be instrumental in the successful commercialization of our technologies,” said Hillcrest Energy Technologies CEO Don Currie in the press release. “In James’ expanded role, his leadership within Hillcrest will continue to provide the company with valuable access and insights into customer needs and expectations.” To view the full press release, visit https://ibn.fm/zRlqu
Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) is a clean technology company based in Vancouver, British Columbia, engaged in developing high-value, high-performance power conversion technologies and digital control systems for next-generation powertrains and grid-connected renewable energy systems.
From concept to commercialization, Hillcrest invests in the development of energy solutions that power a more sustainable and electrified future. Hillcrest power inverter technology helps produce efficiencies in electrification and maximize the performance of electric systems, including electric vehicles (EV), motors and generators.
The company offers a flexible, single-inverter architecture that can be applied at nearly every stage of the electrification ecosystem, from renewable energy generation through the charging and operation of an EV, to provide full-cycle efficiency and performance improvements.
As momentum to electrify and decarbonize energy systems accelerates, Hillcrest believes the power inverter is increasingly emerging as a key component. While system cohorts such as battery packs, PV panels and electric motors are often in the spotlight, the inverter holds the key to unlocking efficiency and performance improvements.
Hillcrest power inverter technology is:
- REVOLUTIONARY: high-efficiency inverter technology has the potential to revolutionize how motors respond and how efficiency is gained.
- AGILE: able to deliver and deploy high-efficiency inverter solutions purpose-designed to meet specific customer needs.
- INNOVATIVE: technology-forward, clean-energy experts who are focused on advancing and optimizing efficient alternative energy use across all electric vehicle and charging platforms.
- A MARKET LEADER: a next-generation technology provider to the automotive industry’s top suppliers and manufacturers.
Technology & Applications
Hillcrest’s first application for its inverter technology – a 250 kW|800V Hillcrest SiC high efficiency traction inverter – is focused on the growing EV market. Hillcrest technology eliminates traditional design trade-offs faced across the power industry – deploying higher switching frequencies has historically meant a greater increase in losses, lower system efficiency and higher heat. Through a combination of hardware and software expertise, Hillcrest enables power applications to leverage higher switching frequencies AND
- Realize improved power system performance and reliability
- Operate at higher power levels without compromising efficiency
The expected benefits of Hillcrest’s traction inverter have been confirmed via testing and shared in a technical white paper, published in April 2022, that confirmed the following results:
- Significant efficiency gains – 99%-plus inverter efficiency
- Increased power density targeting 50kW/L+
- Significantly increased motor efficiency
- Lower stress on mechanical and electrical parts, enhancing reliability
- Improved thermal management
Hillcrest has also filed a patent for an enhanced powertrain solution that offers the potential to simplify EV charging and redefine how the industry envisions charging infrastructure. The company believes the most exciting benefit of the enhanced powertrain solution is the ability to eliminate the onboard charger and booster from an EV, as well as faster, anywhere charging including direct DC, wireless, and bidirectional charging across current and future power levels. Hillcrest sees this as a true EV charging game changer.
The company’s technology applies to nearly every clean energy industry segment:
- Wind power – an inverter is deployed at a wind turbine generator to convert the AC output, with at least one additional inverter used to deliver the power to the grid/battery.
- Solar power – an inverter is used to convert the DC output from the photovoltaic panels into the AC power that flows to the grid/battery/home.
- Energy storage – an inverter is deployed to convert the DC output from the storage system or batteries to the AC power that flows to the grid/home/EV.
- EV fast chargers – an inverter converts the AC input from the grid/storage system to the DC output needed to charge an EV’s battery.
Market Outlook
According to an April 2022 market analysis by Vantage Market Research (VMR), the global power inverter market is expected to reach a value of $95 billion by 2028, driven by increasing demand for EVs, energy generating wind turbines and solar-powered photovoltaic systems. That jump is forecast from an estimated $70.5 billion market value in 2021 and represents a compound annual growth rate of more than 5%.
According to the VMR report, many governments in countries around the world are supporting alternative options for efficient and nonpolluting energy generation. This has boosted demand for wind energy and solar energy systems. Hillcrest is aiming to capture a share of this future market growth across nearly every segment of the clean energy industry.
Management Team
Don Currie is the founding CEO of Hillcrest Energy Technologies. He has led the company’s successful transition from fossil fuels into clean energy technologies. Earlier in his career, he held various senior level positions, including director, officer and vice president of corporate communications with Enhanced Oil Resources Inc., an oil and gas exploration and production company based in Houston. Prior to that, he worked in other private and public ventures spanning the mining, gaming and technology sectors.
Jamie L. Hogue is the COO of Hillcrest. She brings more than two decades of progressive policy leadership, economic analysis and organizational development experience to Hillcrest. She builds collaborative processes and solutions that drive growing organizations toward a more resilient future. She previously served as the director of operations for Arizona State University’s Ten Across initiative – a compelling observatory positioned on the front lines of economic, social and climate change. She earned a master’s degree in public administration and a bachelor’s degree in economics from Arizona State University.
Ari Berger is Chief Technology Officer at Hillcrest. He brings over a decade of commercial experience with a track record of deploying new electrification technologies and go-to-market strategies. In 2015, he founded NIG Systems Ltd. in Israel, which specializes in custom high performance control systems design. Prior to this, he previously worked for Bental Industries, a leading motor manufacturer. He holds a master’s degree in system control engineering from the Technion – Israel Institute of Technology.
Raj Clair is CFO at Hillcrest. She is a CPA who began her career at Deloitte and has served in advanced finance positions in the energy and resources sector. She has been responsible for reporting, audits and internal controls, as well as working on budgeting and forecasting. She has worked with various publicly listed companies, including SEC registrants, and has both Canadian and U.S. experience. She holds a bachelor’s degree in accounting from Simon Fraser University.
Hillcrest Energy Technologies Ltd. (NASDAQ: HLRTF), closed Tuesday's trading session at $0.089, up 0.168824%, on 48,823 volume. The average volume for the last 3 months is 203,390 and the stock's 52-week low/high is $0.072/$0.1789.
Recent News
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Names New Executive
- GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Poised for Opportunity with Flexible Inverter Architecture
- Electric Vehicles: The Greatest Comeback of All Time
FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF)
The QualityStocks Daily Newsletter would like to spotlight FuelPositive Corp. (NHHHF).
• The UN has announced that the world is facing a global emergency, with up to 354 million people facing famine
• FuelPositive’s Green Ammonia Production System brings zero-carbon options to farms worldwide, offering an alternative means of supporting crops and farm equipment with lower energy costs and emissions
• The green ammonia market is expected to reach $5.48 billion by 2030 – driven by the need to adapt to lower emissions and the necessity to make agriculture more affordable
An announcement from the UN in mid-September issued the warning that the world is facing a global emergency of unprecedented magnitude – up to 345 million people are facing starvation, and 70 million are closer to starvation as a result of Russia’s war in Ukraine. Executive director of the UN’s World Food Program, David Beasley, told the Security Council that the number of people facing food insecurity in the 82 counties where the agency operates is two-and-a-half times the number of acutely food insecure people before the COVID-19 pandemic ( https://ibn.fm/Vg5eU ). Beasley further expressed concern that 50 million people in 45 countries are suffering from acute malnutrition and are “knocking on famine’s door.” Russia’s blockade of Ukrainian ports to bring Russian fertilizer back to global markets has exacerbated issues – driving up the cost of food and fuel worldwide. FuelPositive (TSX.V: NHHH) (OTCQB: NHHHF) , a company focused on licensing, partnership, and acquisition opportunities related to energy-efficient technologies and sustainability, is committed to providing commercially viable and sustainable clean energy solutions, including green ammonia, for use across a vast number of industries and applications. The opportunities for green ammonia are significant, with 80 percent of the world’s ammonia being used in the agricultural sector. FuelPositive’s on-farm alternative has the potential to significantly reduce the carbon dioxide emissions impacting the world due to the use of fertilizer in agricultural settings.
FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) is a growth stage company focused on licensing, partnership and acquisition opportunities building upon various technological achievements. The company is committed to providing commercially viable and sustainable clean energy solutions, including carbon-free ammonia (NH3), for use across a broad spectrum of industries and applications.
FuelPositive is headquartered in Toronto, Canada.
Hydrogen Economy Problems and FuelPositive’s Carbon-Free Technology
The hydrogen economy is currently facing many challenges. Traditional NH3 manufacturing exists on a massive scale, but centralized facilities result in some of the world’s most concentrated CO2 emissions. In total, an estimated 200 million metric tonnes of NH3 are consumed each year, with greater than 80% utilized by the agricultural sector. NH3 is also being positioned as a viable alternative to fossil fuels.
FuelPositive’s flagship carbon-free ammonia technology provides an innovative solution to these environmental concerns. Developed by Dr. Ibrahim Dincer and his team, the company’s platform allows for the in-situ production of NH3 in an entirely sustainable manner, using only water, air and sustainable electricity.
The production of hydrogen is energy intensive, but it is just one variable hindering the growth of the hydrogen economy. Other hurdles include:
- Storage – The storage of hydrogen by compression or liquification are both cost prohibitive and unsustainable.
- Distribution – The distribution network for effective hydrogen deployment has yet to be developed, as the extreme high-pressure distribution requirements to transport hydrogen would result in enormous infrastructure costs.
- End Use – R&D on the transportation-related end use applications for hydrogen is in its infancy, but almost any vehicle on the road today can be easily converted to run on NH3 at a considerably lower cost per mile traveled when compared to traditional fossil fuels.
A key benefit of FuelPositive’s patent-pending, first-of-its-kind carbon-free NH3 technology is its flexibility. The process allows for small, medium or large-scale production of NH3 on location, minimizing or even eliminating the challenges and volatility associated with storage and transportation to end use. As such, with an appropriately sized FuelPositive system and access to renewable energy, the end use applications for the company’s platform are nearly infinite.
Manufacturing Partnership
On May 19, 2021, FuelPositive announced its selection of National Compressed Air Canada Ltd. (“NCA”) to undertake manufacturing of the company’s Phase 2 hydrogen-ammonia synthesizer commercial prototype systems for carbon-free ammonia production.
In a news release detailing the partnership, FuelPositive CEO Ian Clifford noted, “This critical milestone for FuelPositive will confirm the broad application potential for our technology and is the backbone of our Carbon-Free Hydrogen-NH3 offering. Partnering with the knowledgeable and experienced team at NCA on this commercialization project will bring our development-stage program to life.”
Global Ammonia Market Outlook
The global ammonia market was valued at $52.71 billion in 2017 and is forecast to reach $81.42 billion by 2025, growing at a CAGR of 5.59%, according to data from Fior Markets (https://ibn.fm/1OfOB).
The agricultural industry consumes more than 80% of global NH3. Smaller percentages can be attributed to the waste, water treatment, refrigerants, antiseptic, textile, mining and pharmaceutical industries.
One of the most polluting industries on the planet consists of conventional agribusinesses. These polluters are responsible for more greenhouse emissions per year than transportation. This is where FuelPositive’s technology is expected to be extremely beneficial.
Management Team
Ian Clifford is Director, CEO and Founder of FuelPositive Corp. He has over 25 years of experience in the fields of technology and marketing and has successfully led the company to global brand recognition through its unique energy solutions. Since 2006, Mr. Clifford has raised over $50 million in equity financing for FuelPositive. He also co-founded digIT Interactive, a full-service internet marketing company serving Fortune 500 clients, which he sold at the peak of the market in 2000.
Greg Gooch serves as a Director and President of FuelPositive. His multifaceted career in the electronics and finance industries has positioned him as a key advisor and funding partner to start-ups and new technology companies for over 40 years. Mr. Gooch has been involved with FuelPositive since its early days and has remained a significant supporter and consultant to the company over the years. He has a bachelor’s from McGill University and an MBA from the University of Western Ontario.
Dr. Ibrahim Dincer is a scientific advisor to FuelPositive and is recognized as a pioneer and international leader in the area of sustainable energy technologies. Along with his team, Dr. Dincer invented the modular carbon-free ammonia (NH3) production technology that FuelPositive is commercializing. His area of specialty covers various topics including ammonia, hydrogen energy and fuel cells; renewable energy systems; energy storage systems and applications; carbon capturing technologies, and integrated and hybrid energy systems He is currently managing an exemplary team of researchers in this commercialization project.
Marek Warunkiewicz is the company’s Communications & Branding Specialist. He brings more than 40 years of entrepreneurial expertise to the FuelPositive team, having held marketing, branding, advertising, project management and graphic design positions with various companies. Mr. Warunkiewicz has successfully created business-to-business marketing and advertising campaigns for a diverse group of clients ranging from high-tech to agriculture. He co-founded digIT Interactive and ZENN Motor Company alongside Ian Clifford.
Luna Clifford is the Director of Communications for FuelPositive. She has over 10 years of experience as a business owner and advisor, helping build and operate several successful start-up enterprises while managing complex stakeholder relationships. Ms. Clifford excels in strategic planning and team building, and she has completed extensive studies in the fields of communications and health care.
FuelPositive Corp. (NHHHF), closed Tuesday's trading session at $0.0894, off by 1.4333%, on 203,390 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.1789/$.
Recent News
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Green Ammonia Systems Offer Relief from Rising Fertilizer Costs and Helps Reduce Emissions
- FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Working to Eliminate Emissions Linked to Grey Ammonia Production, Manure Use
- GreenEnergyBreaks - FuelPositive Corporation's (TSX.V: NHHH) (OTCQB: NHHHF) Green Ammonia System Poised to Offer Supply and Price Stability
Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF)
The QualityStocks Daily Newsletter would like to spotlight Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF).
A stroke is a form of brain injury caused by blood supply to the brain being interrupted. This medical emergency requires prompt treatment ; responding too late increases the likelihood of brain damage. Figures show that strokes are the third leading cause of death and disability around the globe, with billions in funds being allocated for long-term healthcare services, support and rehabilitation for stroke survivors. Long-term rehabilitation is usually exhaustive and lengthy, highlighting the need for a new treatment approach, especially since traditional stroke treatments are not 100% effective. This may be realized sooner than we thought. Algernon Pharmaceuticals announced last month that it plans to conduct a phase I study looking into the use of DMT infusions for stroke. A consultant of the company, Dr. David Nutt, stated that Algernon’s approach with DMT strengthened the natural recovery of the brain by improving neuroplasticity, which in turn facilitated the development of new neural networks. Nutt, who is also a neuropsychopharmacology professor at Imperial College London , added that this new approach was different from anything that had been tried previously. Currently, a number of jurisdictions allow ketamine infusions, and companies such as Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) have several centers that are operational in this regard; the centers also conduct development programs involving other psychedelic compounds with known medicinal potential.
Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) is the leading psychedelic wellness platform, committed to bringing science-backed benefits to all and reframing the psychedelic conversation. The company owns and operates an umbrella of related businesses, including trusted media and e-commerce platforms like Reality Sandwich and Delic Radio; Delic Labs, the only licensed entity by Health Canada to exclusively focus on research and development of psilocybin vaporization technology; Meet Delic, the premiere psychedelic wellness event; and Ketamine Infusion Centers, one of the largest ketamine clinics in the country.
Delic is backed by a team of industry and cannabis veterans and a diverse network, whose mission is to provide education, research, high-quality products, and treatment options to the masses. Its founders helped build the multi-billion-dollar cannabis industry and aim to do the same in psychedelics as it follows a similar path toward legalization. In its quest to advance the new psychedelic renaissance upon us, Delic has become the pioneer in its field, creating an ecosystem of opportunities by investing in cutting-edge ideas.
The Vancouver-based company was formed in 2019 to address the growing interest in psychedelic wellness backed by science. Delic was the first psychedelic umbrella platform. It is currently a trusted source for those interested in psychedelic culture, education, treatments, and more.
While other emerging companies focus on patent medicine and big pharma for substances limited by government regulation, Delic is blazing a unique trail. It identifies ancillary and fully legal opportunities like IP, new media, live events, ketamine clinics (with the ability to offer additional psychedelic treatments once legalized, and large-scale production and brings them under its big tent of resources and reach.
The Big Problems Delic Is Addressing
- Fifty percent of Americans will meet the criteria for a mental health condition sometime in their lifetime. The FDA has approved psilocybin therapy as a breakthrough therapy for depression.
- Every 40 seconds, someone in the world commits suicide. Ketamine has been shown to decrease thoughts of suicide significantly. In 2019, the FDA approved esketamine as a fast-acting antidepressant.
- Traditional palliative care methods do not eradicate end-of-life (EOL) anxiety. LSD and psilocybin have been shown to reduce EOL anxiety for terminally ill patients. Eighty percent of terminally ill patients with psilocybin sessions experienced significant reductions in depression and anxiety.
- Approximately 50 million people in the U.S. are addicted to some tobacco product. Research shows that psilocybin is helping people quit smoking.
The Delic Ecosystem
The Delic Ecosystem covers three main areas: media, health, and science. The media focus is educating and motivating the masses through a variety of digital platforms, like Delic’s Reality Sandwich digital magazine, a free public education platform providing psychedelic guides, news and culture (1.4+ million page views in 2020 and 54k social media followers across all platforms); Meet Delic, the first-ever psychedelic wellness summit and the premier psychedelic wellness event based in Las Vegas (over 2,000 live attendees and 5,000+ email subscribers); and Delic Radio (over 43 episodes and 100k total streams). Delic has also been featured in numerous media outlets like Forbes, NBC News, The Joe Rogan Experience, Daily Beast, High Times, and The Dr. Drew Podcast.
The focus of Delic’s health operations is the most accessible psychedelic treatments that can help billions of people live happier lives. Delic does this through one of the largest ketamine clinic chains in the country, Ketamine Infusion Centers (KICs), a limited liability corporation formed under the laws of Arizona that runs three ketamine clinics located in Bakersfield, California, and Phoenix, Arizona. Its management team has over 15 years of experience in the clinic and medical space, scaling and operating over 20 clinics, with a plan to open 10 more clinics in the next 18 months. Together, these clinics have overseen 4,000+ treatments delivered to date.
The focus of Delic’s science operations is developing IP and advanced extraction and testing facilities that are the backbone of the legal market. Delic carries this out through Delic Labs, a licensed cannabis and psilocybin research laboratory based in Vancouver. It’s the only entity licensed by Health Canada to exclusively focus on research and development of psilocybin vaporization technology.
Founded by award-winning chemists, Delic Labs focuses on extraction optimization, analytical testing, and chemical process development to advance the cannabis and psilocybin industries. Health Canada gave it a Section 56 Exemption to work with psilocybin compounds, allowing the company to possess and research these products for development and quality control before they hit the market.
Latest Acquisition – Homestead Book Company
On March 4, 2021, Delic announced its acquisition of Seattle-based Homestead Book Company. Homestead is a legacy counterculture distributor of psychedelic media. It’s also the creator of one of the first self-contained psilocybin mushroom grow kits.
The acquisition of Homestead is an exciting one, as it shows how Delic is increasing accessibility to this nascent industry within regulated jurisdictions. Homestead has sold tens of thousands of mushroom kits globally and was one of the earliest distributors for High Times and many other counterculture publications.
The Homestead acquisition allows Delic to increase its product offerings on its website, Reality Sandwich, which recently hit a record for average monthly traffic of over 200,000 unique visitors and over 2.6 million active readers in 2020.
Market Outlook
The psychedelic renaissance is here. Just in time to help address the global mental health crises, plant medicines have the potential to help billions of people live happier lives. Thanks to university-led and FDA-approved studies, North America is leading the way in advancing an industry as psychedelics are becoming accepted globally for therapeutic, medical, and recreational use. Here are some statistics:
- 32 million people in the U.S. have used psychedelics at least once
- 17% of all American adults between 21 and 64 have used psychedelics at least once
- $500 billion is spent in the U.S. every year on prescription drugs
- $238 billion is spent in the U.S. every year on mental health treatments and ancillary services
- The anxiety disorder and depression treatment market is estimated at $16 billion
- $187.8 billion was spent in 2013 on mental health and substance abuse disorders
Management Team
Delic Co-Founder and CCO Jackee Stang was an executive at High Times, a leading counterculture publication that became the voice for the cannabis industry. The monthly magazine had a circulation of over 500,000 copies per issue. Its website attracted 500,000 to five million users each month by 2014.
Likewise, company Co-Founder and CEO Matt Stang was a previous owner and operator of High Times, a position from which he played an instrumental in legalizing cannabis in multiple states and launched the Cannabis Cup in America. After interacting with the cannabis community for two decades, he helped found Delic in 2019 as one of the first psychedelic corporations. He shapes the company’s vision and path using his expertise in branding, marketing, business development, and product viability.
Delic’s VP of Business Development, John Coleman, Ph.D., is a former president of Anandia Labs, a biotech company focused on genetics and analytics. Having experience in both science and business, Dr. Coleman is well-equipped to lead Delic’s business development efforts as it strives to enter new vertical markets.
Zak Garcia is the company’s Chief Marketing Officer. He was the former CMO of Bulletproof Inc., maker of the well-known Bulletproof Coffee brand. Mr. Garcia is a marketing and leadership strategist who helped grow Bulletproof Coffee to over $250 million in revenue.
Delic Holdings Corp. (DELCF), closed Tuesday's trading session at $0.0316, off by 16.7325%, on 2,906 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.03/$0.2509.
Recent News
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Slow-Release DMT Infusions May Help Develop New Therapy for Stroke Patients
- VA Closely Watching Developments in Psychedelic Space
- Could Psychedelics Help Veterans Suffering from Chronic Pain?
Cybin Inc. (NEO: CYBN) (OTC: CYBN)
The QualityStocks Daily Newsletter would like to spotlight Cybin Inc. (NEO: CYBN) (NYSE American: CYBN).
Cybin (NEO: CYBN) (NYSE American: CYBN ), a biopharmaceutical company focused on progressing Psychedelics to Therapeutics(TM), joined partner The Chopra Foundation in recognizing World Mental Health Day, which was Monday, Oct. 11, 2022. The two organizations are working together to research the potential of psychedelic-based therapies for the treatment of mental health and to provide education about the use of psychedelics. Founded by Dr. Deepak Chopra, The Chopra Foundation is a not-for-profit organization dedicated to improving health and well-being. World Mental Health Day was designed to create an opportunity for those with mental health issues, along with advocates, governments, employers, employees and other stakeholders, to recognize progress and identify ways that mental health and well-being can become a global priority. “World Mental Health Day sends a reminder that our mental health is just as important as our physical health,” said Cybin CEO Doug Drysdale in the press release. “At Cybin, mental health and well-being is at the center of everything that we do. It's this value that drives us to invest our energy and resources to discover new and innovative treatment options for mental health conditions. The commitment from organizations like The Chopra Foundation plays a critical role in this goal, and we are proud to partner with the Foundation to continue to educate and support what is possible for the treatment of mental health from psychedelics.” To view the full press release, visit https://ibn.fm/WXPdb
Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) is a Canada-based life sciences company focused on the pharmaceutical development of psychedelic products, as well as the functional mushroom market.
The early-stage company boasts an experienced management team featuring industry veterans from pharmaceutical and consumer product backgrounds who have run multiple clinical trials and collectively helped facilitate billions of dollars in product revenues. The team is dedicated to the development of products and protocols within the psychedelic, pharmaceutical and nutraceutical industries.
In particular, Cybin aims to further build upon and expand its intellectual property (IP) portfolio, which is structured around unique psilocybin delivery mechanisms that target a number of different therapeutic indications. In addition, the company has dedicated itself toward furthering its research and IP within the fields of synthetic compounds, extraction methods, the isolation of chemical compounds, new drug formulations and protocol regimes.
Serenity Life Sciences & Natures Journey Inc.
The company’s business model is centered around its two core subsidiaries, Serenity Life Sciences and Natures Journey Inc., which comprise Cybin’s two-pronged approach toward delivering fungi-derived psychedelic and medicinal products.
Serenity Life Sciences is focused on furthering research and development of psilocybin-based medications. Psilocybin is found in certain species of mushrooms and is a non-habit forming, naturally occurring psychedelic compound. Research into psilocybin has shown positive results for the treatment of depression, anxiety, PTSD, addiction, eating disorders, ADHD and other indications.
Natures Journey Inc. operates the Journey brand, which specializes in developing proprietary medicinal mushroom products that target and promote mental wellness, immune boosting detoxification and overall general health and wellbeing.
Partnership with the Toronto Centre for Psychedelic Science (TCPS)
Staying true to its axiom of being a research-first medicinal mushroom life sciences company, Cybin recently announced its entry into a strategic partnership with the Toronto Centre for Psychedelic Science (TCPS), with the goal of furthering its ongoing psilocybin research efforts and expanding Cybin’s psilocybin IP portfolio (http://nnw.fm/9EUkI).
“While there is evidence to support psilocybin as a treatment for certain indications, the Toronto Centre for Psychedelic Science is taking a clinical approach to prove or disprove the safety and efficacy of psilocybin-based microdosing through an open science approach,” Paul Glavine, CEO of Cybin, stated in a news release.
“We are excited to join forces with Cybin and to offer our expertise. A number of firms had approached TCPS, but Cybin demonstrated a superior commitment to high-quality research and integrity in product development. Our high standards for scientific rigor and transparency will find a fitting home within the culture Cybin is cultivating in Canada and abroad,” Thomas Anderson, co-founder of the Toronto Centre for Psychedelic Science, added.
Journey’s Product Monetization & Market Potential for Nutraceutical Supplements
Although Cybin is at the forefront of companies seeking to conduct clinical trials aimed at gaining regulatory approval for psilocybin and other psychedelic products, the company has also placed a great deal of emphasis on generating meaningful revenue from its very outset.
Cybin’s Journey brand has is launching a range of supplements comprised of popular fungi-derived ingredients such as Reishi, Lion’s Mane and Cordyceps. Purported to aid focus and concentration while promoting neurogenesis, Journey’s range of nutraceutical products provides Cybin with a crucial foothold within the non-psychedelic legal supplement market, which is valued at over $25 billion globally and growing at a 9% year-over-year rate.
Pharmaceutical Psychedelics
In addition to the company’s range of non-psychedelic supplements, Cybin has plans to carry out a clinical trial with a new delivery system for its psilocybin-based medications later this year. Ultimately, the company aims to enter into technology transfer agreements with global pharmaceutical companies after phase 1 & phase 2 clinical trials are complete in order to accelerate regulatory approvals in major indications in global markets with entire lifecycle product management.
With products such as psilocybin truffles already legal in nations such as the Netherlands, Jamaica and Bulgaria, Cybin has positioned itself to capitalize on an eventual legalization of psychedelic mushroom-derived products in the future. Working within a regulatory environment with strong similarities to that which dealt with cannabis prior to the industry’s eventual legalization by the Canadian government in 2018, Cybin is laying the groundwork for the moment pharmaceutical psychedelics gain acceptance in North America and abroad.
Amalgamation Agreement and Financing
Cybin recently announced its entry into an amalgamation agreement dated June 26, 2020, with Clarmin Explorations Inc. (TSX.V: CX) and 2762898 Ontario Inc., a wholly owned subsidiary of Clarmin (http://nnw.fm/w04LH). Completion of the transactions contemplated in the amalgamation agreement will result in the reverse takeover of Clarmin by Cybin.
In connection with the proposed transaction, Cybin plans to complete a “best-efforts” brokered private placement of subscription receipts of Cybin, with a syndicate of agents co-led by Stifel Nicolaus Canada Inc. (Stifel GMP) and Eight Capital, to raise a minimum of C$14 million ($10 million) and a maximum of C$21 million ($15 million), with a 15% agents’ option.
To date, Cybin has raised approximately C$10,400,000 through an initial financing round and its series A financing round.
Cybin Inc. (NEO: CYBN) (NYSE American: CYBN), closed Tuesday's trading session at $0.6, even for the day, on 3,070 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.3903/$2.355.
Recent News
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN), The Chopra Foundation Note World Mental Health Day
- Cybin & The Chopra Foundation Recognize World Mental Health Day
- InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) 'Focused on Adding Targeted IP'
QSAM Biosciences Inc. (OTCQB: QSAM)
The QualityStocks Daily Newsletter would like to spotlight QSAM Biosciences Inc. (OTCQB: QSAM).
A new report has highlighted that the rate of death due to cancer in America has steadily declined in the last few decades. As of January 2022, more than 18 million individuals who have survived cancer live in America. This is a new record, especially if one compares the figures against the 3 million survivors recorded in the U.S. in 1971. Researchers predict that by 2040, this number will have reached 24 million. The report, which was released late last month by the American Association for Cancer Research, notes that the number of individuals who survived after receiving their diagnosis had been consistently rising. This, experts believe, is because of the significant advances made in the diagnosis and treatment of cancer. The report states that remarkable advancements have been made in the field these last 10 years, leading to great improvements in the detection, diagnosis, treatment and prevention of cancer. Improved effectiveness of treatments against the most common types of cancer, including prostate, colorectal, breast and lung, have also helped improve the five-year rate of survival for all cancers, moving to about 70% in the period between 2011 and 2017 from less than 50% in the 70s. With entities such as QSAM Biosciences Inc. (OTCQB: QSAM) also focused on delivering cutting-edge cancer therapies, the loss of lives to different cancers is likely to keep reducing as the years go by.
QSAM Biosciences Inc. (OTCQB: QSAM) is a clinical stage biotechnology company focused on bringing to market targeted therapeutic radiopharmaceuticals. The company is committed to advancing the fight against cancer through the discovery, development and delivery of effective treatment options for adult and pediatric patients.
QSAM Biosciences was founded in 2020 by Executive Chairman Dr. C. Richard Piazza and CEO Douglas Baum. It is headquartered in Austin, Texas.
CycloSam®
CycloSam®, QSAM Biosciences’ initial technology, is a clinical-stage bone targeting radiopharmaceutical invented by world-renowned scientists at IsoTherapeutics Group LLC. By leveraging a patented, low specific activity form of Samarium-153 (resulting in far less undesirable europium impurity) and what management believes to be a superior chelating agent in DOTMP, CycloSam is designed to selectively target sites of high bone mineral turnover to deliver a prescribed tumor-killing dose of radiation to the bone tumor sites while minimizing radiation exposure to nearby healthy tissue. These parameters are currently being tested in an FDA-cleared clinical trial.
CycloSam® has been shown in laboratory testing to cause significantly less (30x less) buildup of long-lived radionuclidic impurities than prior FDA-approved drugs, which management believes will enable the ability to safely administer therapeutic doses via higher and multiple-dose regimens and effectively expand its potential clinical utility to therapeutic uses in areas of high unmet medical needs.
The indications for CycloSam® currently being evaluated by QSAM Biosciences include:
- Metastatic Bone Cancers – On April 28, 2022, QSAM Biosciences announced that the first patient had commenced treatment in its clinical trial evaluating CycloSam in patients with metastatic bone cancer. As noted in the release, the study is a Phase 1 open-label, dose-escalation trial to evaluate the safety, tolerability, dosimetry, and preliminary efficacy of CycloSam®.
- Pediatric Osteosarcoma/Ewing’s Sarcoma – On February 2, 2022, the company announced that the U.S. FDA has granted Rare Pediatric Disease Designation to CycloSam for the treatment of osteosarcoma. Combined with a previously granted orphan drug designation for osteosarcoma received in 2021, this milestone “may allow QSAM to potentially bring CycloSam® to market more rapidly through additional incentives and eligibilities,” according to CEO Douglas Baum.
- Bone Marrow Ablation – In a 2020 single patient Investigational New Drug (IND) study, an investigator concluded that high-dose CycloSam® can be administered safely to ablate bone marrow in advance of a stem cell transplant with no apparent renal toxicity and no unexpected adverse events attributable to the drug.
QSAM Biosciences’ preclinical and clinical development pipeline is supported by a strong IP portfolio. The company has secured 14 patents across three distinct patent families spanning the U.S., Japan, Canada and the European Union.
Market Outlook
Through its ongoing development of CycloSam®, QSAM Biosciences is targeting multiple large and underserved market opportunities. According to the American Cancer Society, roughly 400,000 new cases of malignant bone metastasis are diagnosed annually in the U.S. alone. Additionally, QSAM will pursue indications for osteosarcoma and Ewing’s sarcoma that are the most common primary malignancies of bone tissues in children.
Despite this pressing need, the current standard of care for bone cancer is aggressive and suboptimal, leading to marginal success with significant side effects and poor long-term survival prognosis. As a result, QSAM Biosciences estimates a sizable market opportunity for its development pipeline.
- Bone Metastasis has an estimated total addressable market of $20 billion in the U.S. based on total new cases and comparable drug pricing.
- Osteosarcoma/Ewing’s Sarcoma have a total addressable market of roughly $125 million in the U.S. based on approximately 1,000 new cases in 2021.
- The total addressable market for Bone Marrow Ablation is projected at $1 billion, with an estimated 32,000 procedures completed annually.
The company anticipates that the ability to administer CycloSam® for higher and multiple-dose regimens may expand its clinical utility for therapeutic uses in additional areas of high unmet medical needs.
Management Team
QSAM Biosciences is led by an experienced management team and board with an extensive record of FDA approvals, big pharma partnerships and M&A transactions.
Dr. C. Richard Piazza is the Executive Chairman of QSAM Biosciences. Since 2017, he has also served as President and CEO of IGL Pharma Inc., the licensor of CycloSam®, and as a consultant to IsoTherapeutics Group LLC, the inventors of the technology. Dr. Piazza also currently serves on the board of directors of NovaScan LLC, a privately held cancer detection and diagnostics company. He has more than 48 years of health care experience in both medical devices and pharmaceutical/biotech and has led several technology companies to market success, including numerous FDA approvals in both sectors. Dr. Piazza obtained a BS in Economics and a BS in Speech Pathology from the State University of New York and an MA & PhD in Economics from the University of Buffalo and Leeds University.
Douglas R. Baum is the company’s CEO and Director. He brings to QSAM Biosciences over 30 years of experience in the bioscience and biotech industries, including development, FDA/EMA approval and commercialization of multiple drugs and medical devices. Mr. Baum has overseen 15 product approvals through the FDA and EMA and raised over $85 million in capital to fund breakthrough technologies. From 2017 to 2020, he consulted with multiple medical schools and biotech and pharmaceutical companies, and, from 2012 to 2017, he served as President, Chief Executive Officer and Director of Xeris Pharmaceuticals Inc. Mr. Baum holds a Master of Science in Technology Commercialization and a BBA in International Business and Marketing from the University of Texas.
Adam King is the CFO of QSAM Biosciences. He is also the Founder and CEO of King Consulting Group, where he provides a range of financial and reporting services for clients. Before founding King Consulting Group in January 2021, Mr. King was the CFO for Netsertive, a venture-backed digital marketing company. From 2016 to 2018, he was the Office Managing Audit Director for BDO’s Greenville, South Carolina, office, in addition to serving as Audit Director in Raleigh, North Carolina, and Boston, Massachusetts. While at BDO, Mr. King worked with various clients, from tech and life science start-ups to billion-dollar publicly traded companies. He holds a Bachelor of Science in Accounting from Elon University and is a CPA in Raleigh, North Carolina.
QSAM Biosciences Inc. (OTCQB: QSAM), closed Tuesday's trading session at $5.93, even for the day, on 100 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $3.50/$14.80.
Recent News
- QSAM Biosciences Inc. (OTCQB: QSAM) - New Report Praises Advances in Cancer Treatment for Preventing Millions of Deaths
- Study Finds Antibiotics May Boost Melanoma Growth by Depleting Gut Microbiome
- TinyGemsBreaks - QSAM Biosciences Inc. (QSAM) Working to Make a Difference in Underserved Patient Populations
Odyssey Group International Inc. (OTC: ODYY)
The QualityStocks Daily Newsletter would like to spotlight Odyssey Group International Inc. (OTC: ODYY).
Odyssey Health (OTC: ODYY) , a company focused on developing unique, life-saving medical products, today announced the formation of Odyssey Neuropharma Inc. According to the update, the wholly owned subsidiary will focus on developing drugs to treat neurological disorders. The blood-brain barrier (“BBB”) represents a significant obstacle for drug delivery to the brain, and many drugs have failed in neuroscience research due to the inability to jump the hurdle that is the BBB. Odyssey will utilize its unique intranasal delivery device coupled with its powdered formulations to allow drugs to be delivered more directly to the brain, providing for quick and efficient delivery. The targeted approach requires less drug to accomplish neuroprotection, further reducing drug levels in the plasma and minimizing side effects. The company has a portfolio of products planned for development, including drugs for mild traumatic brain injury aka concussion, Niemann-Pick type C disease, and an organophosphate nerve agent antidote. To view the full press release, visit https://ibn.fm/zk9se
Odyssey Group International Inc. (OTC: ODYY) is a medical technology company focused on developing lifesaving medical products that offer technological and clinical advantages over current standards of care.
The company’s portfolio of product technologies is diverse, featuring four unique medical products in development. Odyssey’s goal is to deliver superior products with enhanced clinical utility and market potential, thereby yielding a high rate of return for its shareholders and partners. It is guided by a senior management team with significant experience relating to refining technologies, building commercial systems and forging strategic partnerships.
Product Portfolio
Pharmaceuticals
Odyssey has two pharmaceutical products in development:
- PRV-002 is a novel compound for the treatment of concussion, which currently has no FDA-approved drug. In pre-clinical studies, PRV-002 has been shown to significantly improve both neuroscore and memory score following injury in rats subjected to concussion models. Importantly, the first-in-class novel neurosteroid demonstrated no drug-related toxicity in these trials.
- PRV-002 is currently being evaluated in a phase I clinical trial for the treatment of concussion, with phase II trials planned for launch in Fall 2022. Odyssey has also highlighted the potential of PRV-002 for additional indications such as Alzheimer’s disease, Parkinson’s disease, ALS and chromic traumatic encephalopathy (CTE).
- PRV-001 is a novel compound intended to treat Niemann-Pick disease, a rare neurodegenerative-lysosomal storage disorder that affects an estimated 1 in 150,000 individuals in the U.S., demonstrating a 5x higher incidence in Middle Eastern populations.
- Odyssey expects to receive Orphan Drug designation from the FDA for PRV-001, which would accelerate its pathway to FDA approval and provide seven years of market exclusivity.
Medical Devices
Odyssey is also developing two medical device candidates:
- CardioMap® is intended to provide early, non-invasive testing for heart disease. The system offers a number of potential advantages over traditional EKGs, including requiring less training to operate, offering heightened sensitivity and coming in a small and portable form factor. CardioMap is being developed for a 510(k) regulatory pathway, which requires a study to demonstrate equivalence to legacy EKG offerings.
- When approved, CardioMap is expected to be the only device in its class that has a predictive value, illustrating ‘grey’ areas where deterioration has begun but not yet led to pathology. Odyssey expects this feature to provide a powerful incentive for doctors to use the CardioMap device in end markets such as hospitals, doctors’ offices, rehabilitation centers and sports medicine practices.
- Save-A-Life (SAL) is a patented, single-action, instantaneous, handheld, mechanical anti-choking device that creates a vacuum chamber in the mouth to dislodge throat obstructions in a matter of seconds, all without harm to the victim. The device is currently in development, with a proof of concept established.
- Odyssey believes that, once FDA-approved, its anti-choking device will quickly become the “accepted” standard and leader in the treatment of choking incidents globally. Its low-cost manufacturing and convenient portable design give SAL a competitive edge over competing devices utilizing cumbersome masks.
Market Opportunities
Odyssey’s varied development pipeline positions it to address a number of sizable market opportunities with significant unmet medical need. Concussions alone currently account for medical costs of roughly $10-15 billion annually in the U.S., despite the lack of a currently approved FDA drug treatment. This need is particularly apparent in the military and sports industry, where the likelihood of athlete head-injury recurrence is estimated at 75%.
It is for this reason that, in March 2021, Odyssey announced the formation of a sports advisory board featuring well-known athletes supporting the company’s efforts to enhance public awareness of traumatic brain injuries and concussions, as well as the need for an FDA-approved therapy. Members of Odyssey’s sports advisory board include NFL Hall of Famers Kurt Warner & Brett Favre and two-time Olympic gold medalist Abby Wambach.
With its CardioMap platform, Odyssey is targeting the global cardiac monitoring market, which was valued at $28 billion in 2021 by Insight Partners and forecast to reach $43 billion by 2028.
Save-A-Life targets a similarly underserved market. Choking is the fourth-leading cause of death in children, and approximately 5,000 choking deaths occur each year in the U.S. While 95% of these deaths result from in-home incidents, current choking rescue devices fail to address in-home applications.
Management Team
Joseph Michael Redmond is the President, CEO and Chairman of Odyssey. He has over 30 years of commercial experience in medical device companies, previously serving as CEO of Parallax Health Sciences Inc., V.P. of Business Development for DxTech Inc. and V.P. of Sales and Marketing for Bioject Medical Technologies Inc. While at Bioject, Mr. Redmond helped raise over $15 million in capital, entered into several licensing and distribution deals with major biotech and pharmaceutical companies and grew the market cap of the company from under $10 million to over $400 million. He started his career at Abbott Labs and holds a B.A. from Denison University.
Christine M. Farrell is the company’s CFO and Secretary. Prior to joining Odyssey, Ms. Farrell was Vice President of Finance for Bioject Medical Technologies Inc. She also held accounting and financial management positions with Spar-Tek Industries, a manufacturer of high quality and cutting-edge technology for the plywood industry, and Action Machinery, a seller of new and used robotic machine tools and equipment. Ms. Farrell holds a B.A. in Accounting from the University of Washington and an M.B.A. from Willamette University.
Dr. Jacob W. Vanlandingham is Odyssey’s Head of Drug Development. Dr. Vanlandingham holds a Ph.D. in neuroscience with a molecular biology focus. He is a member of the Society for Neuroscience, American Society for Nutritional Sciences, National Neurotrauma Society, Faculty for Undergraduate Research in Neuroscience and the International Association of Medical Science Educators.
Odyssey Group International Inc. (OTC: ODYY), closed Tuesday's trading session at $0.3199, off by 0.03125%, on 89,226 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.11/$0.64.
Recent News
- Odyssey Group International Inc. (OTC: ODYY) - InvestorNewsBreaks - Odyssey Health Inc. (ODYY) Announces Formation of Subsidiary Focused on Treating Neurological Disorders
- Helmets Will Not Protect Individuals Against Concussions, Expert Says
- Odyssey Health, Inc. (ODYY) PRV-002 Compound Takes on Heightened Significance Following Recent Concussion of Dolphins Quarterback Tua Tagovailoa, and the NFL and NFLPA's Announcement on Changes to Concussion Protocol
Knightscope, Inc. (NASDAQ: KSCP)
The QualityStocks Daily Newsletter would like to spotlight Knightscope, Inc. (NASDAQ: KSCP).
Knightscope, Inc. [Nasdaq: KSCP], a leading developer of autonomous security robots, today announced its Robot Roadshow - an engaging experiential event used to grab attention fast, forge direct connections with potential clients, and strike up conversations in a compelling fashion – will land at Miami Valley Gaming, 6000 OH-63, Lebanon, OH 45036, on 12 October 2022, from 10:00am to 2:00pm ET. The Robot Roadshow has made 63 landings in 23 states and Washington, D.C. to date. Knightscope’s crime-fighting robots tour the U.S. in a space-age, NASA-like “pod” allowing attendees to experience all the technology that is enabling these Autonomous Security Robots (ASRs) to help make sites safer today from Hawaii to Texas to North Carolina. A short video of a past event hosted by the Los Angeles Police Department may be viewed here .The Ohio Roadshow landing will be guided by Knightscope experts both virtually and in person. Visitors will be able to interact directly with each of the ASRs and see the Knightscope Security Operations Center (KSOC) user interface in action. Clients, investors and the media are all welcome to attend to learn more about Knightscope.
Knightscope, Inc. (NASDAQ: KSCP), founded in 2013 and based in Mountain View, California, is a leader in the development of autonomous security capabilities targeting to disrupt the $500 billion security industry. Knightscope’s technology uniquely combines self-driving technology, robotics, artificial intelligence and electric vehicles.
Knightscope designs and builds Autonomous Security Robots (ASRs) that provide 24/7/365 security to the places you live, work, visit and study. The company’s client list covers public institutions and commercial business operations, including multiple Fortune 1000 companies to date. These ASRs have been proven to enhance safety at hospitals, logistics facilities, manufacturing plants, schools and corporations. ASRs act as highly cost-effective complementary systems to traditional security and law enforcement officials, providing an additional advantage by continuing to offer uninterrupted patrolling capabilities across the country.
The company’s ASRs have assisted in the arrest of suspects involved in crimes ranging from armed robbery to hit-and-runs. Their machine-embedded thermal scanning capability even aided in preventing the breakout of a major fire. You can learn more about the crime fighting wins at www.knightscope.com/crime
The company has achieved several milestones since its creation in 2013, including:
- Establishing itself in a 15,000-square-foot facility located in Mountain View, California, in the heart of Silicon Valley, where Knightscope designs, engineers and builds its technology (Made in the USA)
- Operating for more than 1 million hours in the field and securing contracts across five time zones, from Hawaii to Rhode Island
- Raising over $100 million since inception to build its technology from scratch and generating over $13 million in lifetime revenue, validating both the market opportunity and the technology
Growth Capital & Proposed Nasdaq Listing
With backing from more than 28,000 investors and four major corporations and over $100 million raised since inception, Knightscope is poised to be an industry leader in the future of public safety and security.
On December 1, 2021, Knightscope announced the commencement of an offering of up to $40 million of its Class A common stock, with shares to be listed immediately following closing on the Nasdaq Global Market under the ticker symbol ‘KSCP’. The offering is for up to 4 million shares priced at $10 per share. Learn more at www.knightscope.com/investors
Company Mission – Reimagining Public Safety
Knightscope’s long-term vision has an eye on the greater good. The company’s mission is to make the United States of America the safest nation in the world while supporting the 2+ million law enforcement and security professionals across the country.
Crime has an estimated negative economic impact in excess of $2 trillion annually. As crime is reduced, positive impacts will likely be realized across several aspects of society, including housing, financial markets, insurance, municipal budgets, local business and safety in general.
Knightscope CEO William Santana Li was interviewed by Kevin O’Leary, more commonly known as Shark Tank’s Mr. Wonderful. When asked to explain how the benefits provided by the ASRs outrank a human doing the same job, Li said, “First, just the simple presence of a physical deterrent causes criminal behavior to change. Second, the machines are self-driving cars that patrol all around and recharge themselves. They also generate 90 terabytes of data per year. No human would ever be able to process that. The robots are intended to be eyes and ears for the humans, not a one-to-one replacement.”
The Knightscope solution to reduce crime combines the physical presence of ASRs, sometimes referred to as proprietary Autonomous Data Machines, with real-time onsite data collection and analysis. The ASRs are fitted with eye-level 360° cameras, thermal scanning, public address announcements and various other features that work in tandem with humans to provide law enforcement officers and security guards unprecedented situational awareness.

Those 90 terabytes of data are then formatted in a useable way, so law enforcement can leverage that information and execute their responsibilities more effectively.
Public Safety Innovation
The company’s recurring revenue business model is set up to mimic the recurring societal problem of crime, and it takes into consideration the fact that innovation in the security and public safety industry has been stagnant for decades. Because the traditional practices of the sector have remained unchanged for years, automation has potential to drive substantial cost savings – and significant improvement in capabilities.
Human security guards are one of both the largest expenses and the largest liabilities for companies. Knightscope’s robots are offered at an effective price of $3 to $9 per hour, compared with approximately $85 for an armed off-duty law enforcement officer and $15 to $35 for an unarmed security guard.
This innovation has the potential to drive considerable cost savings. Based on these estimates, manufacturing costs can be recovered as soon as the first year of operation.
Product Offerings
The company has nine patents and a framework of unique intellectual property. Knightscope currently offers a K1 stationary machine, a K3 indoor machine and a K5 outdoor machine. A K7 multi-terrain four-wheel version is in development.

The ASRs autonomously patrol client sites without the need for remote control, providing a visible, force multiplying, physical security presence to help protect assets, monitor changes in the area and deter crime. The data is accessible through the Knightscope Security Operations Center (KSOC), an intuitive, browser-based interface that enables security professionals to review events generated by the ASRs providing effectively ‘mobile smart eyes and ears’. Learn more at www.knightscope.com/ksoc
The ASRs and the related technologies were developed ground up by the company and are Made in the USA.
The Robot Roadshow

Knightscope has created the ultimate hybrid physical and virtual event, bringing its Autonomous Security Robot technologies to cities across the country for interactive and in-person demonstrations.
Each roadshow landing is hosted virtually by a Knightscope expert, and visitors can interact directly with each of the company’s ASRs and see the Knightscope Security Operations Center (KSOC) user interface in action. Learn more at www.knightscope.com/roadshow
Management Team
Chief Executive Officer William Santana Li is a veteran entrepreneur, a former executive at Ford Motor Company and the founder of GreenLeaf, a company that grew to be the world’s second-largest automotive recycler and is now part of LKQ Corporation (NASDAQ: LKQ).
Chief Client Officer Stacy Dean Stephens brings his experience as a former Dallas law enforcement officer, as well as his skills as a seasoned entrepreneur, to assist on the client acquisition side.
Chief Intelligence Officer Mercedes Soria is an award-winning technologist and former Deloitte software engineer.
Chief Design Officer Aaron Lehnhardt brings over two decades of two- and three-dimensional product and industrial design in modeling and VR to the table, on top of his experience as a senior designer at Ford Motor Company.
Chief Financial Officer Mallorie Burke is a seasoned financial executive and strategic advisor for both private and publicly traded technology companies with a successful track record of mergers & acquisitions, corporate growth and exit strategies, including public listings.
General Counsel Peter Weinberg leverages 30 years of diverse corporate counsel experience, spanning from startups to well-established companies, private and public. He has significant experience training personnel at all levels in critical areas to improve corporate compliance and productivity.
Knightscope, Inc. (NASDAQ: KSCP), closed Tuesday's trading session at $2.44, off by 1.6129%, on 124,930 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.3001/$27.50.
Recent News
- Knightscope (NASDAQ: KSCP) - Knightscope Robot Roadshow Landing in Lebanon, Ohio
- InvestorNewsBreaks - Knightscope Inc. (NASDAQ: KSCP) Robot Show to Take the Spotlight in Chicago
- InvestorNewsBreaks - Knightscope Inc. (NASDAQ: KSCP) K5 ASR to Enhance Security at Orange County Convention Center
Correlate Infrastructure Partners Inc. (OTCQB: CIPI)
The QualityStocks Daily Newsletter would like to spotlight Correlate Infrastructure Partners Inc. (OTCQB: CIPI).
• Correlate Infrastructure Partners Inc. is a provider of environmentally friendly utilities use optimization solutions, utilizing proprietary data-driven analytics and industry expertise to improve outcomes
• CIPI recently formed a partnership with Ultra Yield Solutions (“UYS”) to support clients in the controlled environment agriculture (“CEA”) industry, such as vertical indoor growers
• Under the agreement, UYS will focus on cost-efficient LED lighting solutions while CIPI will deliver renewable energy options and financing opportunities to help make the indoor farming operation even more sustainable
• The global indoor farming market size is expected to reach $44.3 billion this year before growing to $122.3 billion by 2030 at a CAGR of 13.5 percent, with vertical farming comprising the fastest growing segment
Last month, the world’s largest indoor vertical farming operation opened in Pennsylvania with a 140,000-square-foot facility promising about 160 jobs and seven-day-a-week production ( https://ibn.fm/AKwG3 ). The facility is likely to be a harbinger for the growth of indoor vertical farming practices, a subset of the farming technology-driven controlled environment agriculture (“CEA”) industry ( https://ibn.fm/77Omd ). A competing California company is already planning a larger facility in Virginia ( https://ibn.fm/1SM6d ). Indoor vertical farming favors leafy green horticulture, one reason it is seen as the modus operandi of the home industry for cannabis where personal use cultivation is legal, as well as the majority of commercial operations ( https://ibn.fm/tjaQE ). Regardless of the type of agriculture being cultivated, indoor operations are dependent on efficient light strategies to ensure success in a contained environment. Correlate Infrastructure Partners (OTCQB: CIPI) is a Louisiana-based clean energy solutions innovator dedicated to helping commercial operations effectively manage their utilities usage, both for the sake of their financial sheets as well as the resulting benefit to the global environment amid rising concerns about climate change.
Correlate Infrastructure Partners Inc. (OTCQB: CIPI), formerly Triccar Inc., through its two subsidiaries, Correlate and Solar Site Design, offers a complete suite of proprietary clean energy assessment and fulfilment solutions for the commercial real estate industry. The company believes scaling distributed clean energy solutions is critical in mitigating the effects of climate change. CIPI is at the forefront in creating an industry-leading energy solution and financing platform for the commercial and industrial sector. The company sees tremendous market opportunity in reducing site-specific energy consumption and deploying clean energy generation and energy efficiency solutions at scale.
The opportunity exists to remove friction between today’s legacy finance process and the needed clean-energy upgrades developed within the company’s program technologies. For the U.S. to reach its 2050 carbon goals, 200,000 commercial buildings must be retrofitted every year until that date. That represents approximately a 5-10x increase over the 2022 industry process run rate.
CIPI announced completion of its acquisition of 100% of the equity of Correlate Inc. and Loyal Enterprises LLC dba Solar Site Design on December 28, 2021. The company notes these acquisitions occurred at a key inflection point of its growth. CIPI currently enjoys channel and sales partnerships with Fortune 250 companies and a strong, proven industry network.
The company’s transparent, leading-edge model changes value delivery for both facility owners and proven solution providers seeking scale. CIPI believes its rapid growth is due to industry demand for actionable, cashflow positive energy programs and the underlying carbon reduction mandates taking effect globally.
CIPI has filed with the SEC for a name change to Correlate Infrastructure Partners Inc., which will more closely reflect its new platform and growth focus. The company has been aggressively moving to rebrand, with efforts including a revised website, investor presentation materials and an investor relations awareness campaign. The company’s shares will continue to trade on the OTCQB Venture Market under the current ‘CIPI’ ticker symbol until changes are approved.
Subsidiaries
Correlate, founded in 2015, is a portfolio-scale development and finance platform offering commercial and industrial facilities access to clean electrification solutions focused on locally-sited solar, energy storage, EV infrastructure, and intelligent efficiency measures. Its unique data-driven approach is powered by proprietary analytics, concierge subscription services, and a highly scalable national fulfillment network to help building owners profit from fully funded, turnkey decarbonization and facility health programs. The platform is designed for commercial and industrial real estate owners seeking to significantly improve net operating income while meeting carbon reduction goals. The platform provides energy programs for commercial property portfolios and requires no upfront capital. Client organizations reduce their risk and generate more profits by leveraging Correlate’s unique payment programs to put more cash in the bank. Deploying Correlate’s strategic energy programs and energy management systems allows property-owning organizations to complete big energy changes across their portfolios.
Solar Site Design, founded in 2013, is a U.S. Department of Energy Sunshot Catalyst winner that provides customer acquisition and project development tools for the commercial solar industry. Its commercial marketplace platform connects highly qualified project opportunities to leading solar construction companies nationwide. The Solar Site Design platform gives commercial and industrial property owners access to the best price for a commercial solar system. Commercial solar analysts provide property owners a site assessment and working project proposal. Solar Site Design’s team of solar engineers finalize the design while approved financing providers help clients explore financing options for their projects. Then, approved contractors in Solar Site Design’s Marketplace bid on the projects, ensuring commercial and industrial property owners get the best estimates for their projects. Solar Site Design’s marketplace process promotes transparency and fair pricing. Its team of experts has nearly 20 years of experience in the solar industry. Only reputable, experienced, certified (NABCEP), licensed, bonded and insured contractors are accepted into the Solar Site Design Marketplace.
Market Outlook
CIPI is in a rapidly growing market with a unique offering to address a total market of more than 5.9 million commercial buildings in the United States, according to the U.S. Energy Information Administration. Currently, the company’s wholly owned subsidiaries, Correlate and the Solar Site Design, have an opportunity pipeline of over $100 million in commercial projects with more than $20 million in awarded backlog. According to the Rocky Mountain Institute, portfolio energy optimization is a $290 billion market in the United States driving deep financial savings and energy efficiency across the commercial sector.
Commercial buildings consume more than 35% of the generated electricity in the U.S. and are underperforming in energy efficiency at every level. These buildings waste energy, emit too much carbon, and are too costly for owners and occupants, but retrofits are not happening at the rate or scale needed.
In today’s real estate market, portfolio property owners own most commercial buildings. Yet most building efficiency work is focused on single buildings, thereby missing the distinct needs of this owner class which has very different needs than traditional owner-occupiers. The diverse nature of commercial buildings, combined with technology and performance uncertainty, make simple energy optimization initiatives – which could greatly reduce energy use and improve building value – financially unattractive, resulting in slow adoption rates. CIPI’s financial instruments and software breakdown this issue known as the “split incentive”, unlocking the majority of the addressable market.
Management Team
CIPI has in place a nationally recognized management team that has been active in the energy market since 2005.
Todd Michaels is President and CEO of CIPI and founder of Correlate. He formerly served as Vice President for Innovation at SunEdison and Senior Director Distributed Solar at NRG Energy. He founded Correlate in 2015 and has 16 years of experience in the energy industry. He graduated from Indiana University with a B.S. in Computer Information Systems.
Channing Chen is CFO at CIPI and Correlate Inc. and brings over 16 years of experience in the solar industry as a developer, financier, and business unit leader. He has held executive management roles at Solar Power Partners (acquired by NRG Energy), where he was a founding employee, SunEdison, and NRG Energy (NYSE: NRG). Most recently, Mr. Chen was founder and Managing Partner at Breakaway Energy Partners LLC – a distributed energy financing and market-making platform. To date, Mr. Chen and his teams have raised over $1.5 billion in financing across residential, commercial, and utility scale solar and energy storage projects representing over 400 MWs. He holds a B.A. in Environmental Chemistry from the University of California at San Diego and an MBA from the University of Southern California. He is also an advisor and early-stage investor to several startup companies in the renewable energy space.
David Bailey is Chief Revenue Officer of Correlate Inc. With over 15 years of executive sales, supply chain management, and energy efficiency experience, he is responsible for ensuring the success of the National Commercial Sales Unit across multiple regional project teams. Mr. Bailey created and launched the Transformation Services team while at Wesco for its multibillion-dollar Distributed Energy Resource division, formerly Westinghouse. His focus was on IoT-enabled efficiency and plant floor automation-based services. Before that, he spent several years in Global Account Sales Management, with GE Supply as a Program Manager, and is a Commercial Leadership Program graduate. Mr. Bailey received his B.S. in Mechanical Engineering from the University of Kentucky.
Jason Loyet is VP of Commercial Sales of Correlate Inc. He is a cleantech executive with over 20 years of experience leading high growth solar energy and software start-ups. Mr. Loyet is a U.S. Department of Energy SunShot Catalyst award winner for his work building the Solar Site Design technology platform. Before joining the solar energy industry in 2005, he founded and sold two software companies in the streaming media (GlobalStreams) and newspaper publishing (MyCapture) industries. Mr. Loyet currently serves as a Member of the Board of Directors for the Tennessee Solar Energy Industry Association (TenneSEIA).
Deke Welling is Head of Project Development and Fulfillment Services at Correlate Inc. He has over 19 years’ experience in the energy industry with an emphasis on renewables and energy efficiency over the past seven years. Prior to entering the renewables sector, Mr. Welling was the CEO of Welling Resources, an energy development company focused on the exploration of oil and natural gas reserves in the U.S. It was this experience that led him into the renewables sector and leading a charge for more sustainable resources. Additionally, Mr. Welling also served as the CEO of Circle L Solar Inc., a top 100 solar installer in the United States since 2016. Through his leadership, Circle L Solar experienced a growth rate of over 2,250% from 2016 to 2019, resulting in his company being listed on the Inc. 5000 list of the fastest growing private companies in the U.S. (Rank #176) and being named ‘Top Energy Company’ and ‘Entrepreneur of Year for the Energy Industry’ by the American Business Awards® in 2019 and again for ‘Entrepreneur of the Year’ in 2021.
Kevin Warren is Head of Construction and Development Engineering at Correlate Inc. He is a solar veteran with over 12 years of experience in the field. Prior to co-founding CLS, Mr. Warren was the owner of Beacon Consulting and has originated, consulted, designed and/or engineered over 122 MW of PV installations ranging from small commercial to utility scale projects throughout Texas, California, Colorado and North Carolina. He holds a Photovoltaic Technical Sales Professional Certification from the North American Board of Certified Energy Practitioners and certifications from Solar Energy International in PV Installation, PV Technical Sales, PV battery-based design, PV design and engineering, and PV operations and maintenance. Along with PV expertise, Mr. Warren is a LEED Green Building Associate, a certified building analyst from the Building Performance Institute, a Certified Renewable Energy Professional from the Association of Energy Engineers and holds a designation in High-Performance Sustainable Buildings from the BOMI Institute. He studied Electrical Engineering at the University of Texas at Arlington.
Tom Kunhardt is Director of Customer Success at Correlate. He previously held a similar position at Clean.Tech and was Corporate Trainer, Learning & Development, at NRG Energy. He has 15 years of experience in the solar and clean energy industries helping homeowners and businesses find solutions to their energy needs. He holds a bachelor’s degree from the University of Massachusetts.
Correlate Infrastructure Partners Inc. (OTCQB: CIPI), closed Tuesday's trading session at $1.69, even for the day. The average volume for the last 3 months is and the stock's 52-week low/high is $0.32/$3.25.
Recent News
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Rising Interest in Indoor Farming and ESG Factors Portend Market Opportunities for Correlate Infrastructure Partners Inc. (CIPI) Partnership with CEA Lighting Experts
- Companies and Property Owners Seeking Ways to Meet SEC Climate Risk Disclosure Requirements; Correlate Infrastructure Partners Inc. (CIPI) is Ready to Help
- Correlate Infrastructure Partners Inc. (CIPI) Boosts Clients ESG Profiles, Helps Eliminate 'Greenwashing' Criticisms
Flora Growth Corp. (NASDAQ: FLGC)
The QualityStocks Daily Newsletter would like to spotlight Flora Growth Corp. (NASDAQ: FLGC).
For the millions of Americans who grew up under the prohibitionist era, cannabis was presented as a dangerous gateway drug that significantly contributed to drug abuse and criminal activity. Anti-cannabis propaganda was rife during this period, with prohibitionists assigning a wide range of negative effects to cannabis use despite the fact that there was barely any scientific research into the matter. Decades later, tens of millions of Americans now have access to the drug through state-legal markets . Attitudes toward the drug have changed over the past two decades, especially among younger generations , and more people believe that cannabis has more positive effects than negative. According to a recent poll that adds to the growing body of research on cannabis, it seems that most Americans no longer believe prohibitionist claims about the purported dangers of using cannabis. Rasmussen Reports polled 1,000 Americans to find out their views on drug misuse in America and whether or not cannabis was a gateway drug that opened the door to more dangerous drugs. An estimated 59% said that they believed drug misuse in America was getting worse while 9% said the health issue is showing improvements. Additionally, 29% of the respondents said that cannabis was “not at all dangerous” and 28% said that cannabis is “not very dangerous.” Although some of the opinions were mixed, the poll generally showed that Americans are less likely to believe claims that cannabis is a dangerous drug. It echoes sentiments that have been recorded by previous surveys: public sentiments about cannabis are steadily improving . This improvement is creating an ever-growing market for sector players such as Flora Growth Corp. (NASDAQ: FLGC) .
Flora Growth Corp. (NASDAQ: FLGC) is an internationally focused cannabis brand builder that leverages natural, cost-effective cultivation practices to supply cannabis derivatives to its diverse business divisions, including cosmetics, hemp textiles, and food and beverage. Flora Growth operates one of the largest outdoor cultivation facilities in the world with an aim of marketing a higher-quality premium product at below-market prices. By prioritizing natural ingredients and value-chain sustainability across its portfolio, the company creates premium products that help consumers restore and thrive.
Flora Growth completed the first traditional cannabis IPO on Nasdaq in May 2021. Although currently headquartered in Toronto, Ontario, with plans to relocate its head office to Miami, Florida, the company’s base of operations is in Colombia, where it has built an extensive distribution network that includes Colombia’s largest distributors.
Currently, Flora Growth is organically growing market share for its existing brand portfolio (pharmaceuticals, textiles, cosmetics, and food & beverage) while seeking revenue-generating acquisitions that offer an accretive distribution network to amplify revenue growth.
Existing Brand & Product Portfolio
Flora Growth’s portfolio spans a number of verticals – each with a thoughtful brand designed to resonate with its intended end consumer. In line with the company’s mission, each brand prioritizes natural ingredients and value-chain sustainability.
Flora Lab S.A.S
Flora Lab is the company’s GMP certified manufacturing and R&D center focused on producing pharmaceuticals, cosmetics, and nutraceuticals for domestic and international markets. Its offerings include product lines that are private label, white-label, and custom formulas.
Through Flora Lab, Flora Growth has relationships with 1,500+ distribution channels, manufactures 63+ OTC products registered with INVIMA (Colombia National Food and Drug Surveillance Institute), and holds multiple GMP certifications enabling international export in an effort to leverage Flora Lab’s capacity to produce a wide range of CBD-infused products.
Flora Beauty
Flora Beauty is the company’s CBD beauty and cosmetics division founded by fashion and beauty industry icon Paulina Vega. Its current offerings include two CBD skincare brands targeting the U.S. and Latin American markets – MIND NATURALS and AWE. These lines exemplify Flora Growth’s socially conscious approach to business.
Currently, Flora Beauty products are offered globally through e-commerce, as well as through Falabella’s 111 retail locations across Latin America. The company is in negotiations with major department stores to launch the line in the U.S. and is also exploring opportunities in the U.K. and other European markets.

KASA Wholefoods
KASA Wholefoods is a Colombian manufacturer of food and beverages leveraging responsibly sourced exotic fruits from the Amazon. KASA has a $10 million+ distribution agreement with Tropi, Colombia’s largest food distributor, which has 130,000+ distribution points across the country.
Mambe, KASA’s leading brand, is already offered through over 980 distribution points across Colombia. Flora Growth expects this network to grow to over 1,200 distribution points in 2021, including one of Colombia’s largest coffee chains, Tostao Café & Pan.

Hemp Textiles & Co.
Through its Hemp Textiles division, Flora Growth intends to utilize its large land package and cultivation infrastructure to capture market share in the rapidly growing hemp industrials segment.
The company’s first brand through this division, Stardog Loungewear, offers a line of comfortable loungewear made from natural, organic materials. Stardog has been distributing globally through e-commerce and brick and mortar channels in Bogota since fall 2020, and the company intends to open U.S. brick and mortar locations in 2021.

Accretive M&A
Flora Growth is targeting transactions to complete the supply chain via key infrastructure to enhance its global distribution with the aim to compete on low-cost, high-quality inputs paired with premium brands that create business lines with robust margins.
To date, Flora has announced two major transactions.
Koch & Gsell (Acquisition)
- Amplify CPG portfolio’s revenue growth through leading brand, Heimat, currently with TTM revenues of $7.6 million.
- Leverage Koch &Gsell’s distribution network of 2,500+ stores to introduce Flora to the Swiss, European and Asian markets.
- Bring patented hemp cigarette manufacturing technology into new markets utilizing Flora’s high-quality cannabis.
Hoshi International (Investment)
- Equity Investment of €2 million into Hoshi to establish Flora as a preferred supplier to two EU processing facilities.
- Opens gateway for Flora Growth’s cannabis through international distribution agreements in the EU and U.K.
- Hoshi’s experienced team and increased access to the EU cannabis market to serve as a catalyst for revenue growth.
Cultivation
Key to Flora Growth’s expansion efforts is its cultivation strategy. The company’s Cosechemos farm, located in Bucaramanga, Colombia, is currently licensed to cultivate 247 acres of cannabis. Through three successful pilot crop plantings, the location has demonstrated a production cost of just $0.06/gram. For comparison, the average cost of North American cannabis (based on 2019 figures from Aphria, Tilray, Sundial, and Aurora) equates to roughly $1.89/gram.
Flora Growth is uniquely positioned to capitalize on Colombia’s favorable growing conditions, low-cost infrastructure, and affordable local workforce as it looks to ramp up its cultivation efforts moving forward.

Leadership Team
Bernard Wilson is the Chairman of Flora Growth. A senior financial professional, Dr. Wilson is the former Vice-Chairman of PricewaterhouseCoopers LLP and is the Chairman of the Founders Board of the Institute of Corporate Directors. He has also served as Chairman of the Canadian Chamber of Commerce; Chairman of the International Chamber of Commerce – Canada; and Member of the Canada/U.S. Trade Committee. Dr. Wilson draws on this experience to ensure Flora Growth adheres to effective corporate governance practices.
Luis Merchan is the company’s President and CEO. He is a proven executive with over a decade of experience in enterprise sales management, corporate strategy, merchandising and expense management, and customer experience. Mr. Merchan previously served as Macy’s Inc.’s Vice President of Workforce Strategy and Operations, where he managed the enterprise’s multi-billion-dollar P&L expense line for the entire 540 store portfolio. Throughout his tenure at Macy’s, he led various sales and marketing initiatives, including the B2B corporate sales team that was responsible for $160 million in annual revenue. Mr. Merchan obtained his Bachelor of Industrial Engineering from Pontifical Xaverian University in Bogota, Colombia, and his MBA from McNeese State University. He also holds a Graduate Certificate in Marketing Management from Harvard.
Juan Manuel Galan is a Strategic Advisor to the Flora Growth management team. Mr. Galan currently serves as a senior consultant to The World Bank. He is a politician and former senator of Colombia, serving three terms from 2006 to 2018 as a member of the Colombian Liberal Party. He is also a former professor at the University of Rosario and holds more than 20 years of journalistic, academic, governmental and parliamentary experience. During his time as a senator, Mr. Galan was a key leader, with 29 bills and 27 debates on political control, and 17 laws to his name. The most relevant of those laws was authoring the medical cannabis law that resulted in the legalization of medical cannabis in Colombia.
Stan Bharti is a Director of Flora Growth. Mr. Bharti currently serves as Executive Chairman of Forbes & Manhattan. He has more than 30 years of professional experience in business, finance, markets, operations and more, with a focus on the resource and technology sectors. To date, Mr. Bharti has amassed over $3 billion worth of investment capital for the companies with which he has worked and their shareholders. He is a Professional Mining Engineer and holds a master’s degree in engineering from Moscow, Russia, and University of London, England.
Javier Franco is the company’s VP of Agriculture. Mr. Franco is a master horticulturist with more than 25 years of experience in the design, implementation, and management of cultivation and propagation facilities of more than 30 species of cut flowers in Latin America. He completed his agricultural studies at Zamorano University in Honduras and later at an International Exchange Program at Ohio State University. Mr. Franco has directed technical, commercial, and research groups in the cut flower, fruit and vegetable markets in Latin America and has participated in the commercial development of new technologies applied in agribusiness. He has also led the agri-management of organic crops and certifications of Good Agricultural Practices.
Flora Growth Corp. (FLGC), closed Tuesday's trading session at $0.6044, off by 3.9873%, on 920,047 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.55/$5.61.
Recent News
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Most Americans Don't Believe Propaganda About Marijuana's Purported Dangers
- Rosy Outlook for Global Cannabis Market Boosts Hopes for Flora Growth Corp. (NASDAQ: FLGC) as Supply Chain Expands
- CannabisNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Elevating Performance with Expanded Team
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
• Hypertension affects nearly half of all adults in the United States – 47% or 116 million people, some of which are medication-resistant
• A recent study from the University of Toledo College of Medicine and Life Sciences concluded gut bacteria might be the cause of medication-resistant hypertension – as demonstrated in rats used to conduct the study
• Lexaria’s patented DehydraTECH(TM) technology offers a viable solution for hypertension drug administration and has been received favorably by the FDA during a pre-Investigational New Drug meeting
Hypertension, or high blood pressure, remains one of the world’s most deadly health problems, considered a silent killer because it presents with no symptoms. Nearly half of the adult population in the United States (47% or 116 million) have been diagnosed with hypertension, as defined by having systolic blood pressure greater than 130 mmHg or diastolic blood pressure greater than 80 mmHg or taking medication for the condition ( https://cnw.fm/JjXeS ). Within this category of adults, some fall into a medication-resistant form of hypertension. Lexaria Bioscience (NASDAQ: LEXX) , a global innovator in drug delivery platforms, is on a mission to transform the way hypertension is treated through its patented technology, DehydraTECH(TM). DehydraTECH improves the bioavailability of pharmaceuticals and therapeutics in part by bypassing first-pass-liver processing. DehydraTECH also provides the following benefits:
• Masks unwanted tastes by eliminating the need for sugar-filled edibles
• Improves the speed of onset, with effects felt in minutes
• Increases bioavailability by more effectively delivering the drug into the bloodstream
• Increases brain absorption, with testing suggesting up to 10x improvement
• Reduces drug administration costs with a higher ratio of drug delivery
Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing.
DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future.
Lexaria has a collaborative research agreement with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has filed for patent protection for specific delivery of nicotine, vitamins, NSAIDs, testosterone, estrogen, cannabinoids, terpenes, PDE5 inhibitors (with brand names like Viagra), tobacco and more.
Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats.
The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products.
DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions.
DehydraTECH Technology
Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following:
- Speeds up delivery – the effects of the product are felt by the subject in just minutes.
- Increases bioavailability – the technology is much more effective at delivering a drug or product into the bloodstream.
- Increases brain absorption – animal testing suggests significant improvement in the quantity of drug delivered across the blood-brain barrier.
- Improves drug potency – more of the ingested product is made available to the body, so lower doses are required to achieve the desired effect.
- Reduces drug administration cost – lower doses mean lower overall drug costs.
- Masks unwanted taste – the technology eliminates or reduces the need for sweeteners.
Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery.
Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine.
Market Outlook
Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments:
- Oral Cannabinoids – a market estimated to be worth $18.4 billion in 2021 and expected to reach $46.2 billion by 2025.
- Antivirals – an estimated $52.1 billion market in 2021 that’s expected to grow to $66.7 billion by 2025.
- Oral Mucosal Nicotine – smokeless tobacco products, a $13.6 billion market in 2018, is forecast to grow at 7.2 percent annually through 2025.
- Human Hormones – estrogen and testosterone replacement therapies represented a $21.9 billion market in 2019, with a forecast CAGR of 7.7 percent through 2027.
- Ibuprofen and Naproxen – NSAID sales totaled $15.6 billion globally in 2019 and are projected to reach $24.4 billion by 2027.
- Vitamin D3 – the global market size was $1.1 billion in 2021, growing at 7 percent per year and expected to reach $1.7 billion in 2026.
Management Team
Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations.
John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents.
Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations.
Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch.
Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia.
Lexaria Bioscience Corp. (LEXX), closed Tuesday's trading session at $1.95, off by 2.5%, on 20,729 volume. The average volume for the last 3 months is 20,729 and the stock's 52-week low/high is $1.85/$7.15.
Recent News
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Patented DehydraTECH(TM) Technology Offering Solutions to Growing Hypertension Problem Worldwide
- Lexaria Bioscience Corp. (NASDAQ: LEXX) Working to Address Declining Antihypertensive Intensification and High Prescription Abandonment Rates, with DehydraTECH-CBD
- TinyGemsBreaks - Lexaria Bioscience Corp. (NASDAQ: LEXX) Redefining Drug Delivery Platforms
GeoSolar Technologies Inc.
The QualityStocks Daily Newsletter would like to spotlight GeoSolar Technologies Inc.
• October declared National Clean Energy Action Month as government mobilizes toward a future powered by sustainable energy and a healthier environment for all
• Although clean energy momentum continues to build, not all green energy sources are created equal; some can still leave significant environmental impact
• GeoSolar seeks to demonstrate that living in carbon-powered homes is no longer necessary; offers homeowners renewable ways to generate electricity without greenhouse gas emissions
As the climate crisis threatens the safety and health of people worldwide, forcing countries to fight weather disasters such as floods, droughts and wildfires, governments around the world – the US included – recognize that the window to prevent devastating consequences is rapidly closing. But as the adage goes, there is opportunity in every crisis. Still, once a crisis is in full swing, turning it into an opportunity often calls for new ways of thinking and responding. That’s where companies like GeoSolar Technologies (“GST”) with its SmartGreen(TM) Home system can step in to help fight against one of the biggest existential challenges of our time by transforming how homeowners heat, cool, and power homes with 100% natural energy sources.
GeoSolar Technologies Inc. (“GST”) is a Colorado-based climate technology company and the creator of the Smart Green Home® system for newly built and existing residences and commercial buildings. The company is focused on revolutionizing the way we heat, cool and power homes with 100% natural energy sources. Its patent-pending integrated system harnesses energy from the earth and sun to power and purify homes and automobiles without the use of fossil fuels.
In a GST home, the sun’s energy is captured on the roof to generate all of the electricity required. Additionally, the consistent climate of the earth is used to keep the home at a perfect temperature year-round, and the company’s proprietary air purifying unit ensures that the air inside the home is safe and healthy.
GST’s home technology has been installed in multiple test homes in Colorado and achieved exceptional results, including some of the most impressive energy efficiency ratings (HERS) in the industry.
GeoSolar Technologies is currently accepting investment as part of a Regulation A+ offering. Everyone* can invest now for as little as $300. For more information, visit the company’s profile on Manhattan Street Capital and review its Offering Circular.
GeoSolar Technologies Inc. (“GST”) has been qualified by the U.S. Securities and Exchange Commission (SEC) to conduct a Regulation A+ capital raise. GST is already a publicly traded company who makes quarterly and annual filings with the SEC and is subject to quarterly PCAOB audits. This is the first time shares of GeoSolar Technologies are being made available for public purchase. Upon completion of this Regulation A+ offering, the company intends to seek a listing of its stock.
The Decarbonization Movement
 Soaring and unstable energy/fuel costs continue to highlight the importance of rethinking the traditional approach to powering homes, from top to bottom. While most everyone is well aware of the remarkable, multi-trillion-dollar opportunity the electric vehicle transformation offers to investors (in addition to the benefits to the climate problem), few recognize that the all-electric home market is as large as electric vehicles and equally important to reducing carbon emissions.
Soaring and unstable energy/fuel costs continue to highlight the importance of rethinking the traditional approach to powering homes, from top to bottom. While most everyone is well aware of the remarkable, multi-trillion-dollar opportunity the electric vehicle transformation offers to investors (in addition to the benefits to the climate problem), few recognize that the all-electric home market is as large as electric vehicles and equally important to reducing carbon emissions.
U.S. energy expenditures clocked in at $3,891 per person in 2018, leading to estimated spending of $1.3 trillion on energy that year alone. Despite this, fewer than 3% of U.S. homes are currently powered by solar. This number is poised to increase exponentially as both new and existing residences transition to zero carbon models.
GST estimates that if all the homes in America were powered by its technology, carbon pollution could be reduced by an estimated 1.9 trillion pounds per year, greatly reducing the negative impacts on our climate.
GeoSolarPlus®
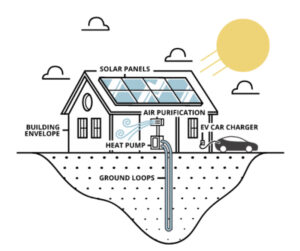 The GeoSolarPlus (“GSP”) system combines solar power, geothermal ground-sourced energy and other clean energy technologies into one fully integrated system.
The GeoSolarPlus (“GSP”) system combines solar power, geothermal ground-sourced energy and other clean energy technologies into one fully integrated system.
Key benefits of the GSP system include:
- Making a real planet-changing difference in reducing air pollution
- Eliminating or significantly reducing homeowners’ future utility bills
- Enjoying lifetime energy independence and protection from price escalation and energy shortages
- Eliminating greenhouse gas emissions from operation of home and daily life
- Increasing home value
- An integrated design for seamless operation of renewable energy systems
- Maintaining a significantly healthier living environment
- Leveraging existing renewable energy tax credits and electrification incentives
- Creating stable jobs capable of supporting families in the decarbonized future
Click here to learn more about how GeoSolarPlus works.
Management Team
The GST leadership and management team includes some of the world’s most experienced and respected leaders in the fields of decarbonization and sustainable homes.
Stone Douglass is the Chairman and CEO of GST. He is a seasoned, 30-year public company executive and former Chairman and CEO of the Piper Aircraft Company.
Brent Mosbarger is the company’s Co-Founder and leads its commercial operations. He is a highly respected solar engineer whose experience includes roles with Chevron Energy’s green operations and serving as project manager and executive for a $400 million solar/geothermal innovation project.
Peter Romenesko is a Senior Strategic Advisor with GST. He brings to the company considerable experience as an engineer and large-scale project manager for Johnson Controls and Siemens.
Dr. Norbert Klebl is the company’s Co-Founder and Development Director. Recognized as one of the world’s leading experts in the field of zero-carbon innovation, he is a former McKinsey partner of 16 years with an MBA from Columbia.
Dar-Lon Chang is GST’s Director of New Product Development. Prior to joining GST, he had a 16-year career with ExxonMobil Energy Research. He received his PhD in engineering from the University of Illinois.
* Must be over 18, certain states are not currently available and will be added soon.
Recent News
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Strives to Empower American Homeowners to Reach Net-Zero Emissions as Race Toward Green Future Accelerates
- Weathering the Storm: GeoSolar Technologies Inc. Electrifying Highly Efficient Homes
- QualityStocksNewsBreaks – GeoSolar Technologies Inc.’s SmartGreen(TM) Offering Homeowners Carbon-Free Living, Zero Utility Bills, Healthier Environment

HeartBeam Inc. (NASDAQ: BEAT)
The QualityStocks Daily Newsletter would like to spotlight HeartBeam Inc. (NASDAQ: BEAT) .
HeartBeam (NASDAQ: BEAT) , a cardiac technology company that has developed the first and only 3D-vector ECG platform for heart attack detection anytime, anywhere, has named a new chief medical officer. Renowned Stanford cardiologist Peter J. Fitzgerald, MD, PhD, will be joining the HeartBeam executive team and will support the planned advancement of the company’s product pipeline. Director of the Center for Cardiovascular Technology and Director of the Cardiovascular Core Analysis Laboratory at Stanford University Medical School, Fitzgerald is an interventional cardiologist and has led or participated in more than 175 clinical trials and has published some 650 manuscripts/chapters. He has lectured worldwide and has been principle and founder of 24 medical device companies, leading 18 of them to medium/large-cap life science companies. “We are thrilled to have Dr. Fitzgerald, one of the world’s preeminent opinion leaders in cardiology and digital health, join the HeartBeam leadership team and play a major role in defining best paths to adoption, clinical strategies and partnerships to advance our products in the market,” said HeartBeam CEO and founder Branislav Vajdic, PhD, in the press release. “In addition to his cardiovascular clinical and research expertise, Dr. Fitzgerald has a rich history of developing successful collaborations with partners in the industry and has developed deep connections across the investment and medical communities.” To view the full press release, visit https://ibn.fm/m5ju4
HeartBeam Inc. (NASDAQ: BEAT) is a cardiac technology company that has developed the first and only 3D-vector 12-lead electrocardiogram (ECG) platform for heart attack detection anytime, anywhere. The company’s proprietary ECG telehealth technology aims to redefine the way high risk cardiovascular patients are diagnosed in ambulatory and acute care settings. HeartBeam’s initial focus is on providing diagnostic data to help physicians with care management of patients with cardiovascular disease.
In August 2022, HeartBeam announced that it submitted its HeartBeam AIMI™ software for approval from the U.S. Food and Drug Administration (FDA). HeartBeam AIMI is a platform technology to improve the speed and accuracy of heart attack detection in acute care settings. The company expects FDA approval by the end of 2022, and a full commercial roll-out of HeartBeam AIMI is targeted for Q1 2023.
HeartBeam sees submission of its first product based on its platform technology as an important milestone toward commercialization, which underscores the company’s continued progress toward making the HeartBeam AIMI platform widely available to help emergency department physicians quickly and accurately identify a heart attack.
While the FDA conducts its regulatory review, HeartBeam will focus on executing key components of its commercialization plan and subscription revenue model. It will also continue to engage in discussions with strategic institutions, including academic centers, regional healthcare systems and regional community hospital systems that can utilize HeartBeam products.
The company is based in Santa Clara, California.
Products
HeartBeam’s development portfolio includes two products:
- HeartBeam AIMI is software that provides a 3D comparison of baseline and symptomatic 12-lead ECG to more accurately identify a heart attack in acute care settings and, as noted above, has been submitted for FDA approval; and
- HeartBeam AIMIGo™, the first and only credit card-sized 12-lead output ECG device coupled with a smartphone app and cloud-based diagnostic software system for remote heart attack detection.
HeartBeam is developing AIMIGo, a medical-grade detection and monitoring technology for use in remote heart attack detection, thereby allowing physicians to diagnose a patient’s heart attack as it occurs, even if the patient is not at a medical facility. The company’s system, once approved by the FDA, can be used by patients at home or almost anywhere and anytime to help their physicians assess whether chest pain is the result of a heart attack or another cause. While approximately 82% of chest pain ED visits are unnecessary, patients delay approximately 3 to 4 hours after symptoms begin, increasing mortality rates by 40%. The company’s goal is to shorten the time to treatment outside of the medical facility to improve patients’ well-being.
HeartBeam’s AIMIGo is a powerful, portable and easy-to-use prescription-based product. It comprises a smartphone app, a credit card-sized ECG device placed on a patient’s chest, the HeartBeam cloud platform, and a digital portal for the physician to view ECG results and direct patient action. For the first time outside of a medical setting, HeartBeam AIMIGo enables patients and their clinicians to determine if symptoms are due to a heart attack, quickly and easily, so care can be expedited, if needed.
Pending FDA clearance, AIMIGo is initially intended to be available by prescription, and is reimbursable under existing remote patient monitoring codes (RPM codes). This provides a new revenue stream to physicians who before did not have a way to monitor these high-risk patients. The RPM codes provide a monthly reoccurring revenue stream to the company, as well. On average, at current reimbursement rates, the practice will receive $1,300+ per year per patient they monitor, and the company will receive $600 per year per patient from this RPM reimbursement.
Market Overview
Adoption rates of telehealth services increased dramatically in recent years, with the COVID-19 pandemic serving as a major driver of growth. Among the areas seeing the greatest expansion are cardiology, radiology, behavioral health and online consultation.
Encouraging this growth, governments are actively developing new policies and reimbursement guidelines to promote the use of digital health platforms. The U.S. Centers for Medicare & Medicaid Services (CMS), for example, has recently expanded reimbursement for telehealth services. U.S. market growth is also being driven by the rising prevalence of chronic conditions and the growing geriatric population.
Remote heart attack detection is a previously unsolved problem with a massive and underserved market that is several times larger than the $2 billion total addressable market (TAM) in the U.S. for ECG cardiac arrhythmia monitoring.
Approximately 8 million Americans have suffered at least one heart attack, and a total of 18 million have been diagnosed with coronary artery disease (CAD). Based on these figures, HeartBeam projects a total addressable U.S. market TAM valued at $10 billion annually for its AIMIGo solution for remote heart attack monitoring of CAD.
Management Team
Branislav Vajdic, Ph.D., Chief Executive Officer and Founder of HeartBeam, Inc, combines over 30 years of experience in technology development and senior management positions. Dr. Vajdic has been deeply involved with the development of HeartBeam’s technology to fit his vision for the company. Prior to HeartBeam, from 2007 to 2010, Dr. Vajdic was CEO and Founder of NewCardio, a publicly traded company in the cardiovascular devices space. From 1984 to 2007, Dr. Vajdic was at Intel, where he held various senior management position. At Intel, Dr. Vajdic was the designer of first Flash memory and two key inventions that enabled Flash as a product and led engineering groups responsible for Pentium 1 through Pentium 4 designs. Dr. Vajdic was awarded two Intel Achievement Awards, the highest level of award for outstanding contributions to Intel. Dr. Vajdic is author of numerous patents and publications in the fields of cardiovascular devices, as well as chip design. Dr. Vajdic holds a Ph.D. in Electrical Engineering from the University of Minnesota.
Jon Hunt, Ph.D., has over 35 years’ experience in the medical/medical device industry with extensive domestic and international experience in general management, clinical/regulatory, sales and marketing. He also has diverse experience in Fortune 500 companies, as well as start-up environments. Dr. Hunt was the Vice President of Clinical Science and Technology, Medical Device Innovation Consortium, from July 2019 to July 2021, and Vice President of Clinical and Regulatory Affairs, Cryterion Medical from January 2018 to June 2019 (acquired by Boston Scientific Corporation in July 2018 for $202M). Dr. Hunt was the Founding President and CEO of Bardy Diagnostics, Inc. from October 2013 to November 2017 (acquired by Hill-Rom Holdings, Inc.). Prior to joining Bardy Diagnostics, Dr. Hunt spent the previous 11 years as the Vice President of Clinical & Regulatory Affairs with Cameron Health, Inc. (acquired by Boston Scientific Corporation). Dr. Hunt spent the previous 10 years with Cardiac Pacemakers, Inc., St. Jude Medical and Cardiac Pathways Corporation. Dr. Hunt began his career with Cardiac Pacemakers, Inc. (now Boston Scientific Corporation) as the Director of Clinical Programs. He subsequently held positions at St. Jude Medical in Clinical Affairs and as the Business Unit Director for the Cardiac Rhythm Management division for Europe, the Middle East and Africa. At Cardiac Pathways Corporation, Dr. Hunt held various executive positions as Vice President of International Sales and Marketing and Vice President of Worldwide Sales and Marketing (acquired by Boston Scientific Corporation). Dr. Hunt received his Ph.D. in Motor Control from The Pennsylvania State University, his Master’s from California State University, Long Beach and his undergraduate degree from Keele University in the United Kingdom.
Rick Brounstein, HeartBeam’s Chief Financial Officer, combines over 30 years of experience in health technology senior management. Since 2017, Mr. Brounstein has been and is currently a partner of Hardesty, LLC, a financial services firm, and Mr. Brounstein is currently a managing director of CTRLCFO, LLC, a firm Mr. Brounstein founded in 2016 to support funded start-ups in life science and technology. From 2008 to 2011, Mr. Brounstein was Chief Financial Officer of NewCardio, Inc., a microcap public company in the cardiology space, and, over his career, he has been with nine other companies in life science or technology, holding positions including Chief Financial Officer, Chief Operating Officer, Treasurer and Accounting Manager. From June 2001 through November 2007, Mr. Brounstein held several positions at Calypte Biomedical Corporation, a publicly traded medical device company, including Chief Financial Officer and Executive Vice President. In January 2007, Mr. Brounstein was appointed as the National Member Representative for the 2007 COSO Monitoring Project, which published new guidelines for monitoring internal financial controls in February 2009; Mr. Brounstein subsequently was a member of the FEI task force that issued the updated COSO Internal Control Framework in 2013. In March 2005, Mr. Brounstein was appointed to the SEC Advisory Committee on Smaller Public Companies. Mr. Brounstein earned his Certified Public Accountant (CPA) certification while working at Arthur Andersen LLP, formerly a public accounting firm. Mr. Brounstein holds a B.A. in accounting and an M.B.A. in finance, both from Michigan State University.
Ken Persen, HeartBeam’s Chief Technology Officer, combines over 28 years of experience in the medical device and digital health industries in engineering and senior management positions. Mr. Persen has been involved in several companies in Cardiac Rhythm Management, holding positions including Chief Executive Officer, Chief Technology Officer, Executive Vice President and Director of Engineering. Since 2016 and prior to joining HeartBeam, Mr. Persen was the Chief Technology Officer at LIVMOR, Inc., a digital health company. In addition, from 2016 through November 2021, he was also Chief Executive Officer of LIVMOR. Prior roles included Director of Engineering at Cameron Health (acquired by Boston Scientific), a late-stage medical device start up, and engineering and management positions at Guidant Corp. (acquired by Boston Scientific), a large medical device manufacturer. He has an undergraduate degree from University of Minnesota, Duluth, with a BA in Computer Science.
HeartBeam Inc. (NASDAQ: BEAT), closed Tuesday's trading session at $5.19, up 11.6129%, on 1,093,935 volume. The average volume for the last 3 months is 1.09M and the stock's 52-week low/high is $1.12/$6.74.
Recent News
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Appoints Seasoned Veteran as New Chief Medical Officer
- BioMedNewsBreaks - Why HeartBeam Inc. (NASDAQ: BEAT) Is 'One to Watch'
- InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Expands Platform to Add 24/7 Heart Monitoring Capability
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Issues Letter to Stockholders
- Aditxt Inc. (NASDAQ: ADTX) - People with Autoimmune Disorders More Likely to Suffer Post-Heart Attack Complications, Study Finds
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Hurricane Ian Forces Closure of Cannabis Businesses in Florida
- American Cannabis Partners - 420 with CNW — Bill on Illicit Cannabis Grows, Banned Pesticides Filed in Congress
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Learning Platforms Potential Solution to Provide Key Education for Skill-Based Hiring
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Names New Board Members, Management Changes
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- AREV Life Sciences Global Corp. (CSE: AREV) (OTC: AREVF) - AREV Notes U.S. Governments $2.9 Billion Assistance to Address Food Insecurity
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Kirby Medical Center to Significantly Reduce Documentation Burden for Physicians and Improve Patient Experiences
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - Blue Hat (BHAT. US) announces the signing of the memorandum of understanding on strategic cooperation with a Singapore Partner
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Secures CE Mark Approval for NeuroCap(TM)
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - Oklahoma Supreme Court Shoots Down Two Challenges to Cannabis Legalization Measure
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Empowering Organizations to Securely Grow, Operate, Innovate
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) CEO to Participate in Breakthrough for Brain Tumors(R) 5K Run
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Companies and Property Owners Seeking Ways to Meet SEC Climate Risk Disclosure Requirements; Correlate Infrastructure Partners Inc. (CIPI) is Ready to Help
- Coyuchi Inc. - InvestorNewsBreaks – Why Coyuchi Is ‘One to Watch’
- Cub Crafters Inc. - CubCrafters Inc.’s Popularity is Displayed by Quick Response to its Pending Public Offering, while the Backcountry Aircraft Manufacturer Awaits SEC Qualification
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - Cybin & The Chopra Foundation Recognize World Mental Health Day
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Publishes Promising Clinical Research for Its Biofeedback Posture Device
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Offers Quantum Computing Solutions to Help Enterprises Solve Optimization Business Challenges, Today
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - VA Closely Watching Developments in Psychedelic Space
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Launches Much-Anticipated VANTAGE PRO Lithium Fleet Golf Cart
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd.'s (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Iska Iska Drilling Results Support Potential for High-Grade Silver and Tin
- Researchers Call for Use of Metals in Fighting Infections
- MiningNewsBreaks - Why Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Is 'One to Watch'
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Washington Will Struggle to Fully Wean the US Off Russian Nuclear Fuel
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Innovating Solutions That Bridge Performance-Cost Gap to Electrification
- FingerMotion Inc. (NASDAQ: FNGR) - FingerMotion Inc. (NASDAQ: FNGR) Benefitting From Recurring Revenue Streams from the Top-Up Business, Expands its Product Line
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) Announces New Targets Identified from Recent Magnetic Study
- Flora Growth Corp. (NASDAQ: FLGC) - Rosy Outlook for Global Cannabis Market Boosts Hopes for Flora Growth Corp. (NASDAQ: FLGC) as Supply Chain Expands
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd.'s (NASDAQ: FRSX) (TASE: FRSX) Eye-Net, Pango Collaborate to Change Road Safety Paradigm in Israel
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Positioned for Continued Growth Amid Prevailing Market Trends
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Working to Eliminate Emissions Linked to Grey Ammonia Production, Manure Use
- GeoSolar Technologies Inc. - Weathering the Storm: GeoSolar Technologies Inc. Electrifying Highly Efficient Homes
- Golden Matrix Group Inc. (NASDAQ: GMGI) - TinyGemsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Rolling out Expansion Strategy to Capitalize on 'Compelling Opportunities'
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Appoints New Global Health Services CEO
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation in Upcoming Investor, Industry Conferences
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- GreenBox POS (NASDAQ: GBOX) - InvestorNewsBreaks - GreenBox POS (NASDAQ: GBOX) Announces Launch of Its coyni Platform
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - Why HeartBeam Inc. (NASDAQ: BEAT) Is 'One to Watch'
- Cepton Inc. (NASDAQ: CPTN) - TinyGemsBreaks - Cepton Inc.'s (NASDAQ: CPTN) Short-Range Lidar Solution Aims to Bring Safety Benefits to Everyone on the Road
- Hemptown USA - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Common Stock Upgraded to Mid-Tier OTC Equity Market
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) Amends Registration Statement to Remove Offering of 80M Shares
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Poised for Opportunity with Flexible Inverter Architecture
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Partners with 'Chopped' Champion and Celebrity Chef Katsuji Tanabe
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Q2 2022 Unaudited Financial Results
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Subsidiary Announced new B2B contracts for Electric Tractors
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Receives Notification of Minimum-Bid Price Requirement Compliance
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces 2022 Financial Results, Business Update
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces Completion of 2019, 2020 Audited Financial Statements
- Innovative Payment Solutions Inc. (OTCQB: IPSI) - Innovative Payment Solutions, Inc. Appoints Richard Rosenblum as President and Chief Financial Officer
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope Inc. (NASDAQ: KSCP) Robot Show to Take the Spotlight in Chicago
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Working to Address Declining Antihypertensive Intensification and High Prescription Abandonment Rates, with DehydraTECH-CBD
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Releases Q1 Financial, Operational Results
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging LN to Enhance Micropayment Experience
- South Korea's Biggest Credit Card Issuer Unveils Blockchain System to Identify Counterfeit Goods
- CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging Lightning Network to Offer Better Solutions
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for Fiscal 2022
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Closing Under Share Subscription Agreement, Appointment of New CFO
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participation at Upcoming MicroCap Rodeo Windy City Roundup
- Mullen Automotive Inc. (NASDAQ: MULN) - The Automotive Industry Needs to Adjust Labor Force to Embrace EVs
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) (FSE: N29) CEO to Present at Emerging Growth Conference
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - Helmets Will Not Protect Individuals Against Concussions, Expert Says
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Announces Partnership to Leverage Ecommerce Fulfillment Capabilities
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Announces Launch of VegasLounge with Markor Technology
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Reports Status of Subsidiary LOI with Alkaline Fuel Cell Power Corp.
- Predictive Oncology (NASDAQ: POAI) - Predictive Oncology Strengthens Offerings with GMP Lab and Addition of Leading Expert in Formulation Development
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc.'s (PBIO) Ultra Shear Technology(TM) Platform Showcased at the Conference of Food Engineering 2022
- Prime Harvest Inc. - 420 with CNW — Kentucky Governor Releases Report from Committee Studying Medical Cannabis Legalization
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Shares Update on Clinical Programs Enrollment
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Antibiotics May Boost Melanoma Growth by Depleting Gut Microbiome
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Issues Expansion Update
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - InvestorNewsBreaks - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Eyeing 'Strike When the Iron Is Hot' Opportunity
- REZYFi, Inc. - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Poised for Growth Amid Changing Data-Privacy Environment; Seeks to Bridge the Gap Between Consumers and Their Data
- RYAH Group Inc. (CSE: RYAH) - InvestorNewsBreaks – RYAH Group Inc. (CSE: RYAH) Announces Industry Veteran as New VP Overseeing IP Commercialization
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Goes Global
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Hosts Delegation from Leading Multinational Food and Agricultural Companies
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Announces Official Launch Event for Hapi Travel
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Appoints Industry Veteran as GM European Operations
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Initiates Safety Evaluation Study of Its Novel Time-Released Ketamine Formulation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Secures 1st Purchase Order of Cellular Boosters for Military Use
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Teams up with NFL Great, Drew Brees, to Promote TapouT
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - Comstock to Present at LD Micro Main Event XV
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Moves Closer to Commercialization of First Arkansas-Based Lithium Plant
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Lays Foundation for Significantly Expanded Manufacturing, Distribution Infrastructure
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Secures Purchase Renewal Contract with Louisville Division of The Kroger Company (NYSE: KR)
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) to Participate, Exhibit at 2022 NACS Show
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaine88(R) Gaining Rapid Traction in the Club Channel
- Tingo Inc. (OTC: TMNA) - Dubai Police Announces 24-Hour Timeline to Recover Lost Passports Using Blockchain Tech
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Notes WIPO Publication of International PCT Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - Trxade Health Inc. (NASDAQ: MEDS) Enters into $1.8M Registered Direct Offering
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) to Participate at Upcoming TD Securities Virtual Uranium Roundtable
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Shares Shareholder Voting Recommendations from Leading Proxy Advisory Firms
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Officers Successfully Deploy BolaWrap to Help Restrain Subject Having a Mental Health Crisis in LaGrange, Georgia
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - XPhyto Completes Rotigotine Transdermal Patch Optimization and Comparative Skin Absorption Study
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Issues Letter to Stockholders
- Aditxt Inc. (NASDAQ: ADTX) - People with Autoimmune Disorders More Likely to Suffer Post-Heart Attack Complications, Study Finds
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Hurricane Ian Forces Closure of Cannabis Businesses in Florida
- American Cannabis Partners - 420 with CNW — Bill on Illicit Cannabis Grows, Banned Pesticides Filed in Congress
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Learning Platforms Potential Solution to Provide Key Education for Skill-Based Hiring
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Names New Board Members, Management Changes
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- AREV Life Sciences Global Corp. (CSE: AREV) (OTC: AREVF) - AREV Notes U.S. Governments $2.9 Billion Assistance to Address Food Insecurity
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Kirby Medical Center to Significantly Reduce Documentation Burden for Physicians and Improve Patient Experiences
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - Blue Hat (BHAT. US) announces the signing of the memorandum of understanding on strategic cooperation with a Singapore Partner
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Secures CE Mark Approval for NeuroCap(TM)
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - Oklahoma Supreme Court Shoots Down Two Challenges to Cannabis Legalization Measure
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Empowering Organizations to Securely Grow, Operate, Innovate
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) CEO to Participate in Breakthrough for Brain Tumors(R) 5K Run
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Companies and Property Owners Seeking Ways to Meet SEC Climate Risk Disclosure Requirements; Correlate Infrastructure Partners Inc. (CIPI) is Ready to Help
- Coyuchi Inc. - InvestorNewsBreaks – Why Coyuchi Is ‘One to Watch’
- Cub Crafters Inc. - CubCrafters Inc.’s Popularity is Displayed by Quick Response to its Pending Public Offering, while the Backcountry Aircraft Manufacturer Awaits SEC Qualification
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - Cybin & The Chopra Foundation Recognize World Mental Health Day
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Publishes Promising Clinical Research for Its Biofeedback Posture Device
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Offers Quantum Computing Solutions to Help Enterprises Solve Optimization Business Challenges, Today
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - VA Closely Watching Developments in Psychedelic Space
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Launches Much-Anticipated VANTAGE PRO Lithium Fleet Golf Cart
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd.'s (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Iska Iska Drilling Results Support Potential for High-Grade Silver and Tin
- Researchers Call for Use of Metals in Fighting Infections
- MiningNewsBreaks - Why Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Is 'One to Watch'
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Washington Will Struggle to Fully Wean the US Off Russian Nuclear Fuel
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Innovating Solutions That Bridge Performance-Cost Gap to Electrification
- FingerMotion Inc. (NASDAQ: FNGR) - FingerMotion Inc. (NASDAQ: FNGR) Benefitting From Recurring Revenue Streams from the Top-Up Business, Expands its Product Line
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) Announces New Targets Identified from Recent Magnetic Study
- Flora Growth Corp. (NASDAQ: FLGC) - Rosy Outlook for Global Cannabis Market Boosts Hopes for Flora Growth Corp. (NASDAQ: FLGC) as Supply Chain Expands
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd.'s (NASDAQ: FRSX) (TASE: FRSX) Eye-Net, Pango Collaborate to Change Road Safety Paradigm in Israel
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Positioned for Continued Growth Amid Prevailing Market Trends
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Working to Eliminate Emissions Linked to Grey Ammonia Production, Manure Use
- GeoSolar Technologies Inc. - Weathering the Storm: GeoSolar Technologies Inc. Electrifying Highly Efficient Homes
- Golden Matrix Group Inc. (NASDAQ: GMGI) - TinyGemsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Rolling out Expansion Strategy to Capitalize on 'Compelling Opportunities'
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Appoints New Global Health Services CEO
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation in Upcoming Investor, Industry Conferences
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- GreenBox POS (NASDAQ: GBOX) - InvestorNewsBreaks - GreenBox POS (NASDAQ: GBOX) Announces Launch of Its coyni Platform
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - Why HeartBeam Inc. (NASDAQ: BEAT) Is 'One to Watch'
- Cepton Inc. (NASDAQ: CPTN) - TinyGemsBreaks - Cepton Inc.'s (NASDAQ: CPTN) Short-Range Lidar Solution Aims to Bring Safety Benefits to Everyone on the Road
- Hemptown USA - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Common Stock Upgraded to Mid-Tier OTC Equity Market
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) Amends Registration Statement to Remove Offering of 80M Shares
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Poised for Opportunity with Flexible Inverter Architecture
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Partners with 'Chopped' Champion and Celebrity Chef Katsuji Tanabe
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Q2 2022 Unaudited Financial Results
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Subsidiary Announced new B2B contracts for Electric Tractors
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Receives Notification of Minimum-Bid Price Requirement Compliance
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces 2022 Financial Results, Business Update
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces Completion of 2019, 2020 Audited Financial Statements
- Innovative Payment Solutions Inc. (OTCQB: IPSI) - Innovative Payment Solutions, Inc. Appoints Richard Rosenblum as President and Chief Financial Officer
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope Inc. (NASDAQ: KSCP) Robot Show to Take the Spotlight in Chicago
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Working to Address Declining Antihypertensive Intensification and High Prescription Abandonment Rates, with DehydraTECH-CBD
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Releases Q1 Financial, Operational Results
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging LN to Enhance Micropayment Experience
- South Korea's Biggest Credit Card Issuer Unveils Blockchain System to Identify Counterfeit Goods
- CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging Lightning Network to Offer Better Solutions
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for Fiscal 2022
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Closing Under Share Subscription Agreement, Appointment of New CFO
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participation at Upcoming MicroCap Rodeo Windy City Roundup
- Mullen Automotive Inc. (NASDAQ: MULN) - The Automotive Industry Needs to Adjust Labor Force to Embrace EVs
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) (FSE: N29) CEO to Present at Emerging Growth Conference
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - Helmets Will Not Protect Individuals Against Concussions, Expert Says
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Announces Partnership to Leverage Ecommerce Fulfillment Capabilities
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Announces Launch of VegasLounge with Markor Technology
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Reports Status of Subsidiary LOI with Alkaline Fuel Cell Power Corp.
- Predictive Oncology (NASDAQ: POAI) - Predictive Oncology Strengthens Offerings with GMP Lab and Addition of Leading Expert in Formulation Development
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc.'s (PBIO) Ultra Shear Technology(TM) Platform Showcased at the Conference of Food Engineering 2022
- Prime Harvest Inc. - 420 with CNW — Kentucky Governor Releases Report from Committee Studying Medical Cannabis Legalization
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Shares Update on Clinical Programs Enrollment
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Antibiotics May Boost Melanoma Growth by Depleting Gut Microbiome
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Issues Expansion Update
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - InvestorNewsBreaks - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Eyeing 'Strike When the Iron Is Hot' Opportunity
- REZYFi, Inc. - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Poised for Growth Amid Changing Data-Privacy Environment; Seeks to Bridge the Gap Between Consumers and Their Data
- RYAH Group Inc. (CSE: RYAH) - InvestorNewsBreaks – RYAH Group Inc. (CSE: RYAH) Announces Industry Veteran as New VP Overseeing IP Commercialization
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Goes Global
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Hosts Delegation from Leading Multinational Food and Agricultural Companies
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Announces Official Launch Event for Hapi Travel
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Appoints Industry Veteran as GM European Operations
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Initiates Safety Evaluation Study of Its Novel Time-Released Ketamine Formulation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Secures 1st Purchase Order of Cellular Boosters for Military Use
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Teams up with NFL Great, Drew Brees, to Promote TapouT
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - Comstock to Present at LD Micro Main Event XV
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Moves Closer to Commercialization of First Arkansas-Based Lithium Plant
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Lays Foundation for Significantly Expanded Manufacturing, Distribution Infrastructure
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Secures Purchase Renewal Contract with Louisville Division of The Kroger Company (NYSE: KR)
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) to Participate, Exhibit at 2022 NACS Show
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaine88(R) Gaining Rapid Traction in the Club Channel
- Tingo Inc. (OTC: TMNA) - Dubai Police Announces 24-Hour Timeline to Recover Lost Passports Using Blockchain Tech
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Notes WIPO Publication of International PCT Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - Trxade Health Inc. (NASDAQ: MEDS) Enters into $1.8M Registered Direct Offering
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) to Participate at Upcoming TD Securities Virtual Uranium Roundtable
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Shares Shareholder Voting Recommendations from Leading Proxy Advisory Firms
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Officers Successfully Deploy BolaWrap to Help Restrain Subject Having a Mental Health Crisis in LaGrange, Georgia
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - XPhyto Completes Rotigotine Transdermal Patch Optimization and Comparative Skin Absorption Study
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Issues Letter to Stockholders
- Aditxt Inc. (NASDAQ: ADTX) - People with Autoimmune Disorders More Likely to Suffer Post-Heart Attack Complications, Study Finds
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Hurricane Ian Forces Closure of Cannabis Businesses in Florida
- American Cannabis Partners - 420 with CNW — Bill on Illicit Cannabis Grows, Banned Pesticides Filed in Congress
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Learning Platforms Potential Solution to Provide Key Education for Skill-Based Hiring
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Names New Board Members, Management Changes
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- AREV Life Sciences Global Corp. (CSE: AREV) (OTC: AREVF) - AREV Notes U.S. Governments $2.9 Billion Assistance to Address Food Insecurity
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Kirby Medical Center to Significantly Reduce Documentation Burden for Physicians and Improve Patient Experiences
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - Blue Hat (BHAT. US) announces the signing of the memorandum of understanding on strategic cooperation with a Singapore Partner
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Secures CE Mark Approval for NeuroCap(TM)
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - Oklahoma Supreme Court Shoots Down Two Challenges to Cannabis Legalization Measure
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Empowering Organizations to Securely Grow, Operate, Innovate
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) CEO to Participate in Breakthrough for Brain Tumors(R) 5K Run
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Companies and Property Owners Seeking Ways to Meet SEC Climate Risk Disclosure Requirements; Correlate Infrastructure Partners Inc. (CIPI) is Ready to Help
- Coyuchi Inc. - InvestorNewsBreaks – Why Coyuchi Is ‘One to Watch’
- Cub Crafters Inc. - CubCrafters Inc.’s Popularity is Displayed by Quick Response to its Pending Public Offering, while the Backcountry Aircraft Manufacturer Awaits SEC Qualification
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - Cybin & The Chopra Foundation Recognize World Mental Health Day
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Publishes Promising Clinical Research for Its Biofeedback Posture Device
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Quantum Inc. (NYSE: QBTS) Offers Quantum Computing Solutions to Help Enterprises Solve Optimization Business Challenges, Today
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - VA Closely Watching Developments in Psychedelic Space
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Launches Much-Anticipated VANTAGE PRO Lithium Fleet Golf Cart
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd.'s (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Iska Iska Drilling Results Support Potential for High-Grade Silver and Tin
- Researchers Call for Use of Metals in Fighting Infections
- MiningNewsBreaks - Why Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Is 'One to Watch'
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Washington Will Struggle to Fully Wean the US Off Russian Nuclear Fuel
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Innovating Solutions That Bridge Performance-Cost Gap to Electrification
- FingerMotion Inc. (NASDAQ: FNGR) - FingerMotion Inc. (NASDAQ: FNGR) Benefitting From Recurring Revenue Streams from the Top-Up Business, Expands its Product Line
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) Announces New Targets Identified from Recent Magnetic Study
- Flora Growth Corp. (NASDAQ: FLGC) - Rosy Outlook for Global Cannabis Market Boosts Hopes for Flora Growth Corp. (NASDAQ: FLGC) as Supply Chain Expands
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd.'s (NASDAQ: FRSX) (TASE: FRSX) Eye-Net, Pango Collaborate to Change Road Safety Paradigm in Israel
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Positioned for Continued Growth Amid Prevailing Market Trends
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Working to Eliminate Emissions Linked to Grey Ammonia Production, Manure Use
- GeoSolar Technologies Inc. - Weathering the Storm: GeoSolar Technologies Inc. Electrifying Highly Efficient Homes
- Golden Matrix Group Inc. (NASDAQ: GMGI) - TinyGemsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Rolling out Expansion Strategy to Capitalize on 'Compelling Opportunities'
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Appoints New Global Health Services CEO
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation in Upcoming Investor, Industry Conferences
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- GreenBox POS (NASDAQ: GBOX) - InvestorNewsBreaks - GreenBox POS (NASDAQ: GBOX) Announces Launch of Its coyni Platform
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - Why HeartBeam Inc. (NASDAQ: BEAT) Is 'One to Watch'
- Cepton Inc. (NASDAQ: CPTN) - TinyGemsBreaks - Cepton Inc.'s (NASDAQ: CPTN) Short-Range Lidar Solution Aims to Bring Safety Benefits to Everyone on the Road
- Hemptown USA - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Common Stock Upgraded to Mid-Tier OTC Equity Market
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) Amends Registration Statement to Remove Offering of 80M Shares
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Poised for Opportunity with Flexible Inverter Architecture
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Partners with 'Chopped' Champion and Celebrity Chef Katsuji Tanabe
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Q2 2022 Unaudited Financial Results
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Subsidiary Announced new B2B contracts for Electric Tractors
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Receives Notification of Minimum-Bid Price Requirement Compliance
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces 2022 Financial Results, Business Update
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces Completion of 2019, 2020 Audited Financial Statements
- Innovative Payment Solutions Inc. (OTCQB: IPSI) - Innovative Payment Solutions, Inc. Appoints Richard Rosenblum as President and Chief Financial Officer
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope Inc. (NASDAQ: KSCP) Robot Show to Take the Spotlight in Chicago
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Working to Address Declining Antihypertensive Intensification and High Prescription Abandonment Rates, with DehydraTECH-CBD
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Releases Q1 Financial, Operational Results
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging LN to Enhance Micropayment Experience
- South Korea's Biggest Credit Card Issuer Unveils Blockchain System to Identify Counterfeit Goods
- CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging Lightning Network to Offer Better Solutions
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for Fiscal 2022
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Closing Under Share Subscription Agreement, Appointment of New CFO
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participation at Upcoming MicroCap Rodeo Windy City Roundup
- Mullen Automotive Inc. (NASDAQ: MULN) - The Automotive Industry Needs to Adjust Labor Force to Embrace EVs
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) (FSE: N29) CEO to Present at Emerging Growth Conference
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - Helmets Will Not Protect Individuals Against Concussions, Expert Says
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Announces Partnership to Leverage Ecommerce Fulfillment Capabilities
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Announces Launch of VegasLounge with Markor Technology
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Reports Status of Subsidiary LOI with Alkaline Fuel Cell Power Corp.
- Predictive Oncology (NASDAQ: POAI) - Predictive Oncology Strengthens Offerings with GMP Lab and Addition of Leading Expert in Formulation Development
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc.'s (PBIO) Ultra Shear Technology(TM) Platform Showcased at the Conference of Food Engineering 2022
- Prime Harvest Inc. - 420 with CNW — Kentucky Governor Releases Report from Committee Studying Medical Cannabis Legalization
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Shares Update on Clinical Programs Enrollment
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Antibiotics May Boost Melanoma Growth by Depleting Gut Microbiome
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Issues Expansion Update
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - InvestorNewsBreaks - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Eyeing 'Strike When the Iron Is Hot' Opportunity
- REZYFi, Inc. - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Poised for Growth Amid Changing Data-Privacy Environment; Seeks to Bridge the Gap Between Consumers and Their Data
- RYAH Group Inc. (CSE: RYAH) - InvestorNewsBreaks – RYAH Group Inc. (CSE: RYAH) Announces Industry Veteran as New VP Overseeing IP Commercialization
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Goes Global
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Hosts Delegation from Leading Multinational Food and Agricultural Companies
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Announces Official Launch Event for Hapi Travel
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Appoints Industry Veteran as GM European Operations
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Initiates Safety Evaluation Study of Its Novel Time-Released Ketamine Formulation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Secures 1st Purchase Order of Cellular Boosters for Military Use
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Teams up with NFL Great, Drew Brees, to Promote TapouT
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - Comstock to Present at LD Micro Main Event XV
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Moves Closer to Commercialization of First Arkansas-Based Lithium Plant
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Lays Foundation for Significantly Expanded Manufacturing, Distribution Infrastructure
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Secures Purchase Renewal Contract with Louisville Division of The Kroger Company (NYSE: KR)
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) to Participate, Exhibit at 2022 NACS Show
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaine88(R) Gaining Rapid Traction in the Club Channel
- Tingo Inc. (OTC: TMNA) - Dubai Police Announces 24-Hour Timeline to Recover Lost Passports Using Blockchain Tech
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Notes WIPO Publication of International PCT Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - Trxade Health Inc. (NASDAQ: MEDS) Enters into $1.8M Registered Direct Offering
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) to Participate at Upcoming TD Securities Virtual Uranium Roundtable
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Shares Shareholder Voting Recommendations from Leading Proxy Advisory Firms
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Officers Successfully Deploy BolaWrap to Help Restrain Subject Having a Mental Health Crisis in LaGrange, Georgia
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - XPhyto Completes Rotigotine Transdermal Patch Optimization and Comparative Skin Absorption Study
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































