The QualityStocks Daily Stock List
- Sensus Healthcare (SRTS)
- Phunware (PHUN)
- Oncolytics Biotech (ONCY)
- Dragon Victory International (LYL)
- Aptinyx (APTX)
- comScore Inc. (SCOR)
- Monopar Therapeutics (MNPR)
- TuSimple Holdings (TSP)
- PolyNovo (CALZF)
- John Wood Group (WDGJF)
- Austal Ltd (AUTLF)
- World Wireless Communications (WWRL)
Sensus Healthcare (SRTS)
MarketBeat, TradersPro, TraderPower, Zacks, StockMarketWatch, Daily Trade Alert, Trades Of The Day, StreetInsider, QualityStocks, MarketClub Analysis, InvestorPlace and INO Market Report reported earlier on Sensus Healthcare (SRTS), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Sensus Healthcare Inc. (NASDAQ: SRTS) (FRA: 5TX) is a medical device firm that develops cost-effective, minimally invasive as well as non-invasive treatments for both non-oncological and oncological skin conditions, which have been proven to be highly effective.
Sensus Healthcare Inc. was founded by Stephen Cohen, Richard Golin, Kalman Gishman and Joseph C. Sardano in May 2010 and is based in Boca Raton, Florida. The company is a part of the Medical Equipment and Supplies Manufacturing Industry and serves consumers from all over the world. Sensus Healthcare Inc.’s revenue is generated primarily in the United States.
The company uses a low-energy X-ray technology termed as SRT (superficial radiation therapy), which it incorporated into its treatment device portfolio. The devices include the SRT-100 Vision, the SRT-100+ and the SRT-100. Its superficial radiotherapy system is used by dermatologists and oncologists to offer patients who opt out of surgery an alternative way to treat squamous cell and basal cell skin cancer, as well as other skin conditions, like keloids. Sensus Healthcare Inc. also provides a professional skin care line, known as Sensus Skin Solutions.
Sensus Healthcare Inc. recently introduced its ScultpturaTM robotic radiation oncology system which offers brachytherapy and triple-modulated intraoperative radiotherapy to treat patients who are going through treatment for cancer in surgery or at the site of the tumor in an efficient and fast way.
The firm recently received a patent for its Sculptura system, which will allow for more ways to deliver radiation to treat all types of solid tumors. Sensus Healthcare Inc. enhances the quality of life of patients across the globe and with its latest design, may make it easier to treat cancerous tumors, which makes the potential demand of the product pretty high.
Sensus Healthcare (SRTS), closed Monday's trading session at $7.33, up 15.6151%, on 1,649,027 volume. The average volume for the last 3 months is 50.328M and the stock's 52-week low/high is $4.05/$15.25.
Phunware (PHUN)
StockMarketWatch, MarketClub Analysis, QualityStocks, StocksEarning, Schaeffer's, TradersPro, InvestorPlace, MarketBeat, TaglichBrothers, InvestorsUnderground and BUYINS.NET reported earlier on Phunware (PHUN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Phunware Inc. (NASDAQ: PHUN) (FRA: 2RJ) is focused on the development of an MaaS (Multiscreen-as-a-Service) integrated enterprise platform for mobile that offers companies the data, solutions and products needed to monetize, manage and engage their audiences and mobile application portfolios on a worldwide scale.
Phunware Inc. is based in Austin, Texas and was established in 2009 by Luan Dang and Alan S. Knitowski. The firm is party to a partnership agreement with GAIN Innovation for the Texas government’s contract bids.
Phunware Inc. is part of the information technology services industry and helps highly respected brands across the globe create category-defining mobile experiences, with over a billion active devices using its platform every month.
Phunware Inc.’s product portfolio is made of enterprise mobile software which includes crypto networking, messaging and notifications, alerts, business analytics and intelligence, marketing automation, location-based services, as well as content management. The firm’s software development kits include analytics and loyalty, advertising, messaging, content management and mobile engagement. Phunware Inc. also provides audience monetization which is inclusive of one-time and recurring transactional media purchases using insertion orders, audience engagement, user acquisition application discovery and application transactions for mobile audience building. This is in addition to providing virtual and physical beacons and low and high density Wi-Fi.
Phunware Inc. recently announced its partnership with Vizzia which will provide a digital front door solution to healthcare organizations on mobile. This solution will offer visitors, patients and staff a more integrated and cohesive healthcare experience which will increase efficiency and in turn, output.
Phunware (PHUN), closed Monday's trading session at $1.66, up 38.3333%, on 50,327,864 volume. The average volume for the last 3 months is 539,630 and the stock's 52-week low/high is $1.00/$4.60.
Oncolytics Biotech (ONCY)
IRGnews Alert, Streetwise Reports, Greenbackers, MarketBeat, SmallCapVoice, BUYINS.NET, Wall Street Mover, StreetInsider, QualityStocks, MarketClub Analysis, The Street, DrStockPick, BestOtc, PennyToBuck, CRWEWallStreet, CRWEFinance, PennyOmega, StockHotTips, StockMarketWatch, TopPennyStockMovers, TradersPro, CRWEPicks, OTCPicks, Clutch Investments, Penny Stock Rumble, Zacks, PinnacleDigest, Wall Street Resources, Street Insider, CoolPennyStocks, StockEgg, Stock Rich, SmallCapReview, Pennybuster, FeedBlitz, Penny Invest, HotOTC, Penny Stock Buzz, Momentum Traders, OTC Markets Group and Real Pennies reported earlier on Oncolytics Biotech (ONCY), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Oncolytics Biotech Inc. (NASDAQ: ONCY) (TSE: ONC) (OTC: ONCWF) (FRA: ONYN) is a development stage biopharmaceutical firm which is engaged in the discovery and development of pharmaceutical products for the treatment of cancers that are resistant to conventional therapies.
Oncolytics Biotech is based in Calgary, Canada and was established on April 2, 1998 by Bradley George Thompson and Matthew C. Coffey. The firm’s technologies are based off of discoveries that were made at the University of Calgary.
Oncolytics Biotech Inc. is party to a collaboration agreement with Pfizer Inc. and Merck KGaA, which entails the co-development of pelareorep, in conjunction with avelumab and paclixatel, an anti-PD-L1 antibody for the treatment of hormone-receptor positive metastatic breast cancer.
Oncolytics Biotech Inc.’s pipeline includes REOLYSIN, also known as reovirus, which is a proprietary isolate and non-pathogen candidate as a possible cancer therapy. Other product candidates include Tecentriq, Retfanlimab, Opdivo, Keytruda, Bavencio and pelareorep. Pelareorep is an immuno-oncology viral agent for the treatment of hematological malignancies and solid tumors. Pelareorep targets cancer via a unique mechanism of action with 2 components.
Oncolytics Biotech Inc. published data on its pelareorep candidate, which found that the drug candidate delivered a significant boost that was known to increase the efficacy of checkpoint inhibitors in December, 2020. In a joint study with SOLTI that focused on early stage breast cancer, the companies found that the candidate had the potential to overcome the immunosuppressive nature of the tumor environment, which means that the candidate could possibly become an approved therapy in the future.
Oncolytics Biotech (ONCY), closed Monday's trading session at $1.5, up 19.0476%, on 539,893 volume. The average volume for the last 3 months is 1.212M and the stock's 52-week low/high is $0.8021/$2.11.
Dragon Victory International (LYL)
StockMarketWatch, QualityStocks, MarketClub Analysis and BUYINS.NET reported earlier on Dragon Victory International (LYL), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Dragon Victory International Ltd (NASDAQ: LYL) is a holding firm which is focused on the operation of an online crowdfunding platform that’s reward based in China.
The company has its headquarters in Hangzhou, in the People’s Republic of China and was incorporated in 2015, on June 19 by Yu Han. It serves consumers across the globe as well as idea generators, start-up ventures, medium and small sized companies.
The firm operates through these segments: Taikexi and Longyun. The Taikexi segment under which Finder’s fees services and Auto-Parts procurement services operate, is engaged in the provision of logistics services and auto parts sourcing while the Longyun segment under which the Crowdfunding platform service and the Incubation Services operate, is involved in the incubation and crowdfunding business.
The company also offers business incubation services related to strategic planning, sales and marketing and general and guidance resources in ancillary services, such as assisting with feasibility studies, operations, accounting, legal, human resources and other types of services. It also provides procurement services to auto-repair businesses that need auto parts from suppliers. This is in addition to operating 5etou, which allows projects looking for finances to connect with sources of finance.
The enterprise uses blockchain and smart contract technology in most of its dealings and may soon get into the cryptocurrency space, which is a burgeoning field at the moment. This move would not only help the firm expand its reach into new markets but also allow it to invest in new ventures, which would be beneficial to its stocks and its shareholders.
Dragon Victory International (LYL), closed Monday's trading session at $0.5601, up 24.3285%, on 1,213,170 volume. The average volume for the last 3 months is 518,250 and the stock's 52-week low/high is $0.4505/$1.89.
Aptinyx (APTX)
MarketBeat, StockMarketWatch, TradersPro, The Online Investor, StreetInsider, MarketClub Analysis, Top Pros' Top Picks, Schaeffer's, InvestorPlace, Zacks and QualityStocks reported earlier on Aptinyx (APTX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Aptinyx Inc. (NASDAQ: APTX) is a clinical-stage biopharmaceutical firm that is engaged in discovering, developing and commercializing new, synthetic and proprietary small molecules for treating nervous system and brain disorders.
The firm has its headquarters in Evanston, Illinois and was incorporated in June 2015 by Joseph R. Moskal and Norbert G. Riedel. It operates as part of the biotechnology research services industry, under the health care sector, in the biotech and pharma sub-industry. The firm serves consumers in the U.S. and has two companies in its corporate family.
The enterprise has a platform used for the discovery of proprietary compounds that work via NMDA receptor modulation, which improves pathways involved in nerve cell communication. It is party to a research collaboration agreement with Allergan Plc.
The company’s product pipeline is made up of an NMDA receptor modulator dubbed NYX-458, which is currently undergoing phase 1 clinical trials to evaluate its effectiveness in treating Parkinson’s disease cognitive impairment; and an NMDA receptor modulator known as NYX-783, which has been indicated for the treatment of post-traumatic stress disorder. In addition to this, it also develops NYX-2925, which has been indicated for treating painful diabetic peripheral neuropathy. The company also develops the AGN-241751 program.
The firm’s NYX-2925 was recently granted Fast Track Designation by the FDA for the treatment of pain related to diabetic peripheral neuropathy. This move will not only benefit patients who suffer from neuropathic pain linked to diabetic peripheral neuropathy if the treatment is approved by the FDA but also encourage more investments into the firm, which will in turn boost growth.
Aptinyx (APTX), closed Monday's trading session at $0.3266, up 23.9939%, on 523,447 volume. The average volume for the last 3 months is 902,717 and the stock's 52-week low/high is $0.20/$3.7599.
comScore Inc. (SCOR)
StreetInsider, MarketBeat, The Street, Street Insider, InvestorPlace, QualityStocks, Zacks, Daily Trade Alert, Barchart, The Motley Fool, MarketClub Analysis, Louis Navellier, internetnews, internet, BUYINS.NET, BestChartNow, Hit and Run Candle Sticks, AllPennyStocks, Money Morning, Penny Stock Buzz, Schaeffer's, SmarTrend Newsletters, StockMarketWatch, StreetAuthority Daily, TradersPro and Marketbeat.com reported earlier on comScore Inc. (SCOR), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
comScore, Inc. (NASDAQ: SCOR) is an information and analytics firm that measures audiences, consumer behavior and advertising across media platforms.
The firm has its headquarters in Reston, Virginia and was incorporated in August 1999 by Gian Mark Flugoni and Magid M. Abraham. It serves consumers around the globe.
The company helps measure what individuals do as they interact with the digital world across various technology platforms and devices, such as desktop computers, televisions, tablets and smartphones. Its objective is to help clients monetize audiences globally. The company serves technology providers, agencies, brand advertisers, content owners, movie studios, television networks and digital publishers.
The enterprise provides ratings and planning services and products that include the Plan Metrix, which provides understanding of consumer lifestyle; the Video Metrix, which measures digital video consumption; and the Mobile and Media Multi-Platform Metrix, which measures apps and websites on tablets, smartphones and computers. Other ratings and planning products include the Total Home Panel Suite, which captures connected TV, OTT and IOT content consumption and device usage; and the comScore Campaign Ratings for verification of desktop and mobile video campaigns. In addition to this, the enterprise also provides analytics and optimization services and products that offer solutions for evaluation, optimization and planning of advertising campaigns.
The company recently entered into a new agreement with Cornerstone Media Group. Cornerstone intends to expand its offering to the local market space through the exclusive use of comScore’s TV ratings currency data. This move will bring in additional revenue into the company and help grow its consumer reach, which will be good for the company’s growth.
comScore Inc. (SCOR), closed Monday's trading session at $1.2, up 17.6471%, on 902,717 volume. The average volume for the last 3 months is 386,104 and the stock's 52-week low/high is $1.00/$3.94.
Monopar Therapeutics (MNPR)
StreetInsider, The Stock Dork, StockMarketWatch, QualityStocks, MarketClub Analysis and MarketBeat reported earlier on Monopar Therapeutics (MNPR), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Monopar Therapeutics Inc. (NASDAQ: MNPR) (FRA: 1IY) is a clinical-stage biopharmaceutical firm that is focused on the development of proprietary therapeutics to improve clinical outcomes for cancer patients.
The firm has its headquarters in Wilmette, Illinois and was incorporated in December 2014, by Andrew P. Mazar, Christopher M. Starr and Chandler D. Robinson. It operates as part of the pharmaceutical and medicine manufacturing industry, under the healthcare sector. The firm has two companies in its corporate family and serves consumers in the United States.
The company is party to a collaboration agreement with the Cancer Science Institute of Singapore, which entails evaluating MNPR-202 activity and related analogs in various types of cancer. It is also party to a collaboration with NorthStar Medical Radioisotopes LLC, for the development of radio-immuno-therapeutics which target severe coronavirus infections; and another with Grupo Español de Investigaciónen Sarcomas, to develop camsirubicin for patients with advanced soft tissue sarcoma.
The enterprise’s product portfolio is made up of a topoisomerase II-alpha targeted doxorubicin analog known as Camsirubicin, which has been designed to retain anti-cancer activity and minimize the toxic effects of therapies on patients’ hearts; and a clonidine mucobuccal tablet dubbed Validive, which is undergoing a phase 2 trial testing its efficacy in treating radiation induced severe oral mucositis in patients with oropharyngeal cancer. It also develops a humanized monoclonal antibody dubbed MNPR-101, to treat advanced cancers and preclinical stage severe coronavirus infections.
The success of the company’s camsirubicin drug, which has shown potential signs of anti-tumor activity, will benefit patients with cancer while bringing in more investors into the company, which will boost its growth significantly.
Monopar Therapeutics (MNPR), closed Monday's trading session at $3.95, up 32.107%, on 389,193 volume. The average volume for the last 3 months is 7.177M and the stock's 52-week low/high is $1.391/$5.24.
TuSimple Holdings (TSP)
InvestorPlace, Trades Of The Day, MarketBeat, Daily Trade Alert, The Street, Schaeffer's, Investopedia and Daily Markets reported earlier on TuSimple Holdings (TSP), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
TuSimple Holdings Inc. (NASDAQ: TSP) is an autonomous technology firm that is focused on the development of autonomous technology specifically designed for semi-trucks.
The firm has its headquarters in San Diego, California and was incorporated in 2015 by Xiao Di Hou and Mo Chen. It operates as part of the trucking industry, under the industrials sector. The firm serves consumers around the world, with a focus on those in the United States.
The company’s mission is to improve the efficiency and safety of the trucking industry through advanced autonomous driving technology. Its world class autonomous driving system has been specifically designed to meet the unique demands of heavy-duty trucks. The company hopes to address the truck freight industry’s most pressing challenges. It generates revenues primarily from providing freight capacity services.
The enterprise has developed a nationwide transportation network dubbed the Autonomous Freight Network, which allows freight to be moved throughout the country using Level 4 (L4) autonomous trucks. The AFN is an ecosystem comprised of L4 autonomous semi-trucks, high definition digital mapped routes, terminals, and a cloud-based autonomous operations oversight system known as TuSimple Connect. It also designs original deep learning detection algorithms, visual positioning, and multi-sensor integration technology.
The firm, which recently announced its latest financial results, remains focused on transforming the global truck freight industry. This will not only make it possible for trucks to drive safely and autonomously but also bring in additional revenues and investments into the firm, which will be good for the firm’s growth.
TuSimple Holdings (TSP), closed Monday's trading session at $2.48, off by 3.876%, on 7,192,919 volume. The average volume for the last 3 months is 13,351 and the stock's 52-week low/high is $2.44/$43.79.
PolyNovo (CALZF)
We reported earlier on PolyNovo (CALZF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
PolyNovo Ltd (OTC: CALZF) (ASX: PNV) (FRA: MFJ) is a medical devices firm that is focused on the development of biodegradable medical devices.
The firm has its headquarters in Port Melbourne, Australia and was incorporated in 1998, on August 12th. Prior to its name change in November 2014, the firm was known as Calzada Ltd. It operates as part of the medical devices industry, under the healthcare sector. The firm serves consumers around the globe.
The company is focused on improving the outcomes of patients by offering the best surgical solutions available. It operates through the New Zealand, Ireland, United Kingdom, Singapore, Australia and the United States geographical segments. The company generates the majority of its revenues from America.
The enterprise develops innovative medical devices using the patented biodegradable polymer technology, NovoSorb. Its products include the NovoSorb Biodegradable Temporizing Matrix (BTM), which is indicated for deep or full partial thickness burns, surgical and reconstructive wounds and traumatic wounds. It is used by physicians to physiologically treat and close fully debrided clean surgical wounds. It also develops NovoSorb drug elution depots; NovoSorb Dermal Beta Cell Implant to host pancreatic islets in the skin; and hernia devices for hernia repair and tissue reinforcement products.
The firm remains focused on expanding treatment options using its NovoSorb technology as well as expanding its manufacturing facility and preparing for growth. This will not only improve the outcomes of patients around the globe and encourage more investments into the firm but also help create significant value for its shareholders.
PolyNovo (CALZF), closed Monday's trading session at $1.31, even for the day. The average volume for the last 3 months is 200 and the stock's 52-week low/high is $0.2501/$1.50.
John Wood Group (WDGJF)
MarketBeat, Marketbeat.com, Trades Of The Day, Daily Trade Alert, StreetInsider and The Street reported earlier on John Wood Group (WDGJF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
John Wood Group PLC (OTC: WDGJF) (LON: WG) (FRA: JWG1) is an energy service firm that is engaged in the provision of consulting, engineering and project management solutions to the energy and built environment globally.
The firm has its headquarters in Aberdeen, the United Kingdom and was incorporated in 1961 by Ian Wood. It operates as part of the oil and gas integrated industry, under the energy sector. The firm serves consumers across the globe.
The company operates its business through the Asset Solutions Americas, Asset Solutions EAAA, Technical Consulting Solutions and Investment Services segments. It generates most of its revenues from the Asset Solutions Americas and Asset Solutions EAAA segments combined. Geographically, most of the company's revenue is derived from the United States.
The enterprise provides renewable energy, future fuels, and low carbon solutions; and engineering solutions, including decarbonization and optimization in energy and industry. It also offers environment and infrastructure solutions, which include environmental studies and compliance, public infrastructure, environmental remediation, and geotechnical and materials services; mine planning and design, mineral processing and metallurgy, mineral resources and project assessment services; clean energy solutions; and automation and control solutions, such as asset protection and facility automation solutions, as well as virtual, simulation and learning systems.
The firm, which recently completed the sale of its Built Environment consulting business, has been awarded a multi-region engineering services contract by BP. This will not only support safe and efficient energy production but also help generate value for the firm’s shareholders and bolster its overall growth.
John Wood Group (WDGJF), closed Monday's trading session at $1.93, up 14.881%, on 200 volume. The average volume for the last 3 months is 2,150 and the stock's 52-week low/high is $1.185/$3.40.
Austal Ltd (AUTLF)
MarketBeat and InvestorPlace reported earlier on Austal Ltd (AUTLF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Austal Ltd (OTC: AUTLF) (ASX: ASB) (FRA: LX6) is a shipbuilding firm focused on designing, manufacturing and supporting customized aluminum commercial and defense vessels.
The firm has its headquarters in Henderson, Australia and was incorporated in 1988. It operates as part of the aerospace and defense industry, under the industrials sector. The firm serves consumers around the globe.
The company operates through the Australasia shipbuilding, Australasia support, USA shipbuilding and USA support segments. The Australasia shipbuilding segment offers aluminum commercial vessels for international markets excluding the USA while the Australasia support segment offers training and on-going support and maintenance for vessels. On the other hand, the USA shipbuilding segment manufactures aluminum defense vessels for the U.S. Navy while the USA support segment offers training and on-going support and maintenance of these performance vessels for the U.S. Navy.
The enterprise designs, constructs, and supports offshore and wind-farm vessels, passenger vehicle ferries and passenger ferries; patrol boats for border protection agencies and government law enforcement; and naval and other defense vessels. It also develops and integrates advanced vessel control and information management systems, including an integrated monitoring and control system known as Marinelink; as well as motion control systems and interceptors.
The company, which recently launched its new steel shipbuilding facility, has secured a billion-dollar contract from the U.S. Coast Guard to design and build offshore patrol cutters. This move will increase its operating income and help extend its reach globally, which will in turn positively influence the company’s overall growth.
Austal Ltd (AUTLF), closed Monday's trading session at $1.55, even for the day. The average volume for the last 3 months is 100 and the stock's 52-week low/high is $1.23/$1.96.
World Wireless Communications (WWRL)
We reported earlier on World Wireless Communications (WWRL), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
World Wireless Communications, Inc. (OTC: WWRL) is a telecom service provider that is focused on designing and developing wired and wireless telemetry and remote control systems.
The firm has its headquarters in Greenwood Village, Colorado and was incorporated in 1996. It operates as part of the telecom services industry, under the communication services sector. The firm serves consumers around the globe.
The enterprise operates through the Radio product and X-traWeb products segments. Its products and services allow data from remote devices to be accessed through an encrypted and secure internet connection, using a standard web browser in automatic meter reading and facilities management fields. It also provides a micro-controller known as X-Node, which collects, processes, and transmits data to wireless or wired internet access; and the X-Gate product, which serves as a data collecting and transmitting system on a wireless or wired basis. The enterprise also offers a line of 900 MHz spread spectrum radios, which include a frequency hopping spread spectrum radio dubbed the 900 SS Hopper, which provides communication in different environments; and 900 SS MicroHopper, which offers a smaller form-factor and lower power-consumption for short-range applications. This is in addition to manufacturing and selling antennas. The enterprise also maintains about eighty specialty antennas for use in marine, law enforcement and custom applications.
The wireless RAN market is expected to grow significantly between the 2022-2028 period, which positions the company well to occupy a larger market share and boost its growth. This will also bring in additional investors and generate value for its shareholders.
World Wireless Communications (WWRL), closed Monday's trading session at $0.0189, even for the day. The average volume for the last 3 months is 2.06M and the stock's 52-week low/high is $0.0001/$0.05.
The QualityStocks Company Corner
- Energy Fuels Inc. (TSX: EFR) (NYSE American: UUUU)
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF)
- GeoSolar Technologies Inc.
- Advanced Container Technologies Inc. (OTC: ACTX)
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF)
- Silo Pharma Inc. (OTCQB: SILO)
- Cepton Inc. (NASDAQ: CPTN)
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
- Flora Growth Corp. (NASDAQ: FLGC)
- Mullen Automotive Inc. (NASDAQ: MULN)
- Odyssey Group International Inc. (OTC: ODYY)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR)
The QualityStocks Daily Newsletter would like to spotlight Energy Fuels Inc. (UUUU).
Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR),based in Lakewood, Colorado, is the country’s largest producer of uranium and the leading conventional producer of vanadium, both designated by the U.S. government as critical minerals.
As the leading U.S. diversified uranium miner, Energy Fuels’ uranium production portfolio stands apart in the world. Energy Fuels has more uranium production facilities, more production capacity, and more in-ground resources than any other company in the United States. In fact, the company’s assets have produced over one-third of all U.S. uranium over the past 15 years and is uniquely positioned to increase production to meet new demand.
Energy Fuels utilizes both conventional and in-situ recovery (“ISR”) technology to produce uranium from three strategic facilities:
- White Mesa Mill in Utah (conventional) has a licensed capacity of over 8 million pounds of U3O8 per year. The highly strategic White Mesa Mill is the only conventional uranium mill in the country and is proximate to some of the largest and highest-grade uranium mines and projects in the U.S., including the Company’s Canyon mine, La Sal Complex, Henry Mountains Complex and Roca Honda Project. White Mesa Mill provides Energy Fuels with significant production scalability as uranium demand increases. The White Mesa Mill also has other diverse businesses, including vanadium, rare earth elements (REE’s), alternate feed materials recycling and land cleanup, all described below.
- Nichols Ranch Plant (ISR) is located in the productive Powder River Basin district of Wyoming and has a total licensed capacity of 2 million pounds of U3O8 per year. Nichols Ranch has produced 1.2 million pounds of U3O8 since commissioning in 2014, and it has significant future expansion potential from 34 fully licensed wellfields containing significant in-ground uranium resources.
- Alta Mesa Plant (ISR) is located on over 200,000 acres of private land in Texas. The fully licensed and constructed ISR project has a total operating capacity of 1.5 million pounds of uranium per year and produced nearly 5 million pounds of U3O8 between 2005 and 2013. This low-cost production facility is currently on standby, maintained in a state of readiness to respond to expected increases in demand.

In addition to being the largest uranium miner in the U.S., Energy Fuels’ overall portfolio also includes a pipeline of high-quality, large-scale exploration and development projects that are permitted or are in advanced stages of permitting, as well as an industry-leading U.S. NI 43-101 Mineral Resource portfolio.
FACTOID: Energy Fuels has led industry efforts over the past two-plus years to get the U.S. government to recognize the importance of domestically produced uranium, including the 2018 – 2019 Uranium Section 232, the ongoing Nuclear Fuel Working Group and the recently announced creation of the U.S. strategic uranium reserve. The U.S. is by far the largest consumer of uranium in the world, yet we import almost all of our requirements; Energy Fuels aims to change that.
Nuclear Market Potential
Multiple studies in top scientific journals have shown that nuclear power is cleanest and most economical way to produce reliable electricity as worldwide demand continues to soar. Nuclear power is presently the only available and affordable low-carbon power source that can meet both current and future baseload electricity demands while simultaneously reducing air pollution and mitigating climate change. U.S. nuclear power plants currently generate nearly 20% of the nation’s electricity overall and 55% of its carbon‐free electricity and even a modest increase in electricity demand would require significant new nuclear capacity by 2025. According to the World Nuclear Association (WNA), there are currently 441 operable reactors, with another 54 units under construction and 439 in various stages of planning; in addition, the WNA has identified a potentially massive supply/demand gap through 2040 of 1 billion pounds. These factors among others are expected to significantly drive increased demand for uranium.
Reasons Nuclear is Gaining Traction
- Nuclear reactors emit no greenhouse gases during operation. Over their full lifetimes, they result in comparable emissions to renewable forms of energy such as wind and solar.
- Unlike any other form of energy, the waste from nuclear energy is contained and managed securely. Used fuel is currently being safely stored for ultimate disposal or future reprocessing, and 96% of this waste can potentially be recycled.
- Greater demand for clean electricity to power everything from homes to automobiles, reducing dependence on fossil fuels.
No. 1 U.S. Producer of Vanadium in 2019

Energy Fuels also produces vanadium as a byproduct of uranium production. Vanadium is designated a critical mineral, essential to the economic and national security of the United States. Energy Fuels was the largest producer of vanadium in the U.S. in 2019, and has significant high-grade, in-ground vanadium resources, as well as a separate high-purity vanadium production circuit at their White Mesa Mill, which is also the only conventional vanadium mill in the country. Crucial for use in the steel, aerospace, and chemical industries, vanadium plays a critical role in the production of high-strength and light-weight metallic alloys and demand is expected to increase across the globe.
Energy Fuels has several fully permitted and developed standby mines containing large quantities of high-grade vanadium, along with uranium, including:
- La Sal Complex (Utah)
- Whirlwind Mine (Colorado/Utah)
- Rim Mine (Colorado)
Vanadium has also gained increased attention as a catalyst in next-generation high-capacity, “community-scale” batteries used for energy storage generated from renewable sources. Demand is only expected to grow as this market expands. With recent upgrades in its vanadium production operations, in 2019 Energy Fuels produced commercial levels of the highest purity (99.7%) vanadium in the mill’s history and can rapidly adjust production to meet volatile market conditions. Energy Fuels is one of the very few known avenues that provides investors access the vanadium market.
Rare Earth Element (REE) Production, Alternate Feed Material Recycling, and Land Cleanup
The White Mesa Mill also provides the company with diverse cashflow generating opportunities. Security of supply for Rare Earth Elements (REEs) supporting U.S. military and defense requirements is a major issue today. Energy Fuels has been approached by a number of entities, including the U.S. government, inquiring about the potential to process certain REEs at the mill. The White Mesa Mill is currently licensed to process certain REEs, including tantalum and niobium. And, early indications are that the mill can be utilized to produce several other REEs. The White Mesa Mill is also the only facility in North America licensed and capable of recycling alternate feed materials (AFMs). AFMs are essentially low-level waste materials that contain recoverable quantities of natural (or unenriched) uranium. The Company typically generates between $5 and $15 million per year from AFM recycling. Finally, Energy Fuels is seeking to become involved in the cleanup of legacy Cold War era uranium mines in the Four Corners region of the U.S., including on the Navajo Nation. The U.S. Environmental Protection Agency (EPA) has access to over $1.5 billion for the cleanup of just a fraction of the sites on the Navajo Nation. The White Mesa Mill is fully licensed to receive much of this material, we are one of the government’s lowest cost options, and we have the ability to recycle the material and produce usable uranium from it.
Management Team
Mark S. Chalmers, President and CEO
Mark S. Chalmers is the president and chief executive officer of Energy Fuels, a position he has held since Feb. 1, 2018, following his role as chief operating officer of Energy Fuels from July 1, 2016 – Jan. 31, 2018. From 2011 to 2015, Chalmers served as executive general manager of Production for Paladin Energy Ltd., a uranium producer with assets in Australia and Africa, including the Langer Heinrich and Kayelekera mines where, as head of operations, he oversaw sustained, significant increases in production while reducing operating costs. He also possesses extensive experience in in situ recovery (“ISR”) uranium production, including management of the Beverley Uranium Mine owned by General Atomics (Australia), and the Highland mine owned by Cameco Corporation (USA). Chalmers has also consulted to several of the largest players in the uranium supply sector, including BHP Billiton, Rio Tinto, and Marubeni, and until recently served as the chair of the Australian Uranium Council, a position he held for 10 years. Chalmers is a registered professional engineer and holds a Bachelor of Science in Mining Engineering from the University of Arizona.
W. Paul Goranson, COO
W. Paul Goranson is the chief operating officer for Energy Fuels. Goranson has 30 years of mining, processing and regulatory experience in the uranium extraction industry that includes both conventional and in-situ recovery (“ISR”) mining, and he is a registered professional engineer. Prior to the acquisition by Energy Fuels of Uranerz Energy Corporation, Goranson served as president, chief operating officer and director for Uranerz, where he was responsible for operations of the Nichols Ranch ISR Uranium Project. In addition to those duties, he also managed uranium marketing, regulatory and government affairs, exploration and land. Prior to joining Uranerz, Goranson served as president of Cameco Resources, where he led the operations at the Smith Ranch-Highland, Crow Butte and North Butte ISR uranium recovery facilities. Goranson also served as vice president of Mesteña Uranium LLC, and he has served in senior positions with Rio Algom Mining, (a subsidiary of BHP Billiton), and Uranium Resource Inc. Goranson has a Bachelor of Science in Natural Gas Engineering from Texas A&I University, and a Master of Science in Environmental Engineering from Texas A&M University-Kingsville.
David C. Frydenlund, CFO, General Counsel, Corporate Secretary
David C. Frydenlund is chief financial officer, general counsel, and corporate secretary of Energy Fuels. His responsibilities include oversight of all legal matters relating to the company’s activities. His expertise extends to NRC, EPA, state and federal regulatory and environmental laws and regulations. From 1997 to 2012, Frydenlund was vice president of regulatory affairs, general counsel and corporate secretary of Denison Mines Corp., and its predecessor International Uranium Corporation (“IUC”). He also served as a director of IUC from 1997 to 2006 and CFO of IUC from 2000 to 2005. From 1996 to 1997, Frydenlund was vice president of the Lundin Group of international public mining and oil and gas companies, and prior thereto was a partner with the Vancouver law firm of Ladner Downs (now Borden Ladner Gervais) where his practice focused on corporate, securities and international mining transactions law. Frydenlund holds a bachelor’s degree in business and economics from Simon Fraser University, a master’s degree in economics and finance from the University of Chicago and a law degree from the University of Toronto.
Curtis H. Moore, Vice President of Marketing and Corporate Development
Curtis H. Moore is the vice president of Marketing and Corporate Development for Energy Fuels. He oversees product marketing for Energy Fuels, and is closely involved in mergers & acquisitions, investor relations, public relations, and corporate legal. He has been with Energy Fuels for over 12 years, holding various roles of increasing responsibility. Prior to joining Energy Fuels, Moore worked in multi-family real estate development, government relations and public affairs, production homebuilding, and private law practice. Moore is a licensed attorney in the State of Colorado. He holds Juris Doctor and MBA degrees from the University of Colorado at Boulder, and a Bachelor of Arts dual degree in Economics-Government from Claremont McKenna College in Claremont, California.
Energy Fuels Inc. (UUUU), closed Monday's trading session at $7.2, up 4.1968%, on 2,060,447 volume. The average volume for the last 3 months is 65,506 and the stock's 52-week low/high is $4.69/$11.39.
Recent News
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Kazatomprom Plans to Increase Uranium Quota on Trans-Caspian Route
- InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Announces Q3-2022 Results, Continued Strengthening of Leading Position in Uranium, Rare Earth Elements
- Energy Fuels Announces Q3-2022 Results, Including Continued Robust Balance Sheet and Market-Leading U.S. Uranium & Rare Earth Positions
FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF)
The QualityStocks Daily Newsletter would like to spotlight FuelPositive Corp. (NHHHF).
FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) is a growth stage company focused on licensing, partnership and acquisition opportunities building upon various technological achievements. The company is committed to providing commercially viable and sustainable clean energy solutions, including carbon-free ammonia (NH3), for use across a broad spectrum of industries and applications.
FuelPositive is headquartered in Toronto, Canada.
Hydrogen Economy Problems and FuelPositive’s Carbon-Free Technology
The hydrogen economy is currently facing many challenges. Traditional NH3 manufacturing exists on a massive scale, but centralized facilities result in some of the world’s most concentrated CO2 emissions. In total, an estimated 200 million metric tonnes of NH3 are consumed each year, with greater than 80% utilized by the agricultural sector. NH3 is also being positioned as a viable alternative to fossil fuels.
FuelPositive’s flagship carbon-free ammonia technology provides an innovative solution to these environmental concerns. Developed by Dr. Ibrahim Dincer and his team, the company’s platform allows for the in-situ production of NH3 in an entirely sustainable manner, using only water, air and sustainable electricity.
The production of hydrogen is energy intensive, but it is just one variable hindering the growth of the hydrogen economy. Other hurdles include:
- Storage – The storage of hydrogen by compression or liquification are both cost prohibitive and unsustainable.
- Distribution – The distribution network for effective hydrogen deployment has yet to be developed, as the extreme high-pressure distribution requirements to transport hydrogen would result in enormous infrastructure costs.
- End Use – R&D on the transportation-related end use applications for hydrogen is in its infancy, but almost any vehicle on the road today can be easily converted to run on NH3 at a considerably lower cost per mile traveled when compared to traditional fossil fuels.
A key benefit of FuelPositive’s patent-pending, first-of-its-kind carbon-free NH3 technology is its flexibility. The process allows for small, medium or large-scale production of NH3 on location, minimizing or even eliminating the challenges and volatility associated with storage and transportation to end use. As such, with an appropriately sized FuelPositive system and access to renewable energy, the end use applications for the company’s platform are nearly infinite.
Manufacturing Partnership
On May 19, 2021, FuelPositive announced its selection of National Compressed Air Canada Ltd. (“NCA”) to undertake manufacturing of the company’s Phase 2 hydrogen-ammonia synthesizer commercial prototype systems for carbon-free ammonia production.
In a news release detailing the partnership, FuelPositive CEO Ian Clifford noted, “This critical milestone for FuelPositive will confirm the broad application potential for our technology and is the backbone of our Carbon-Free Hydrogen-NH3 offering. Partnering with the knowledgeable and experienced team at NCA on this commercialization project will bring our development-stage program to life.”
Global Ammonia Market Outlook
The global ammonia market was valued at $52.71 billion in 2017 and is forecast to reach $81.42 billion by 2025, growing at a CAGR of 5.59%, according to data from Fior Markets (https://ibn.fm/1OfOB).
The agricultural industry consumes more than 80% of global NH3. Smaller percentages can be attributed to the waste, water treatment, refrigerants, antiseptic, textile, mining and pharmaceutical industries.
One of the most polluting industries on the planet consists of conventional agribusinesses. These polluters are responsible for more greenhouse emissions per year than transportation. This is where FuelPositive’s technology is expected to be extremely beneficial.
Management Team
Ian Clifford is Director, CEO and Founder of FuelPositive Corp. He has over 25 years of experience in the fields of technology and marketing and has successfully led the company to global brand recognition through its unique energy solutions. Since 2006, Mr. Clifford has raised over $50 million in equity financing for FuelPositive. He also co-founded digIT Interactive, a full-service internet marketing company serving Fortune 500 clients, which he sold at the peak of the market in 2000.
Greg Gooch serves as a Director and President of FuelPositive. His multifaceted career in the electronics and finance industries has positioned him as a key advisor and funding partner to start-ups and new technology companies for over 40 years. Mr. Gooch has been involved with FuelPositive since its early days and has remained a significant supporter and consultant to the company over the years. He has a bachelor’s from McGill University and an MBA from the University of Western Ontario.
Dr. Ibrahim Dincer is a scientific advisor to FuelPositive and is recognized as a pioneer and international leader in the area of sustainable energy technologies. Along with his team, Dr. Dincer invented the modular carbon-free ammonia (NH3) production technology that FuelPositive is commercializing. His area of specialty covers various topics including ammonia, hydrogen energy and fuel cells; renewable energy systems; energy storage systems and applications; carbon capturing technologies, and integrated and hybrid energy systems He is currently managing an exemplary team of researchers in this commercialization project.
Marek Warunkiewicz is the company’s Communications & Branding Specialist. He brings more than 40 years of entrepreneurial expertise to the FuelPositive team, having held marketing, branding, advertising, project management and graphic design positions with various companies. Mr. Warunkiewicz has successfully created business-to-business marketing and advertising campaigns for a diverse group of clients ranging from high-tech to agriculture. He co-founded digIT Interactive and ZENN Motor Company alongside Ian Clifford.
Luna Clifford is the Director of Communications for FuelPositive. She has over 10 years of experience as a business owner and advisor, helping build and operate several successful start-up enterprises while managing complex stakeholder relationships. Ms. Clifford excels in strategic planning and team building, and she has completed extensive studies in the fields of communications and health care.
FuelPositive Corp. (NHHHF), closed Monday's trading session at $0.10695, up 4.3415%, on 65,506 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $11.39/$.
Recent News
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Enters Final Phase of Assembling and Validating First Farm-Ready Demonstration System - Milestone Update: Hydrogen Purity Exceeds Expectations
- GreenEnergyBreaks - FuelPositive Corp. (TSX.V: NHHH) (OTCQB: NHHHF) Empowering Farmers to Reduce Over-Reliance on Global Supply Chains
- GreenEnergyBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Supporting Best Farming Practices in Cradle-to-Cradle Commitment to Clean Solutions
GeoSolar Technologies Inc.
The QualityStocks Daily Newsletter would like to spotlight GeoSolar Technologies Inc.
- 2021 represented the first time in half a century that largeurban counties experienced negative growth
- Migration to suburbs and exurban counties contribute more tocarbon emissions, highlighting the need for a transition torenewable energy to combat climate change
- GeoSolar Technologies is an emerging leader with its whole-homesystem using solar, geothermal, and proprietary air filtrationtechnologies
Americans in 2021 exited urban counties in droves, heading forsuburban and exurban counties at a record pace. From anenvironmental perspective, the transition can have unintendednegative consequences, as people generally have more propertyoutside the city, drive more, and use more energy. It doesn’thaven’t to be that way, though, with net-zero pioneers like NorbertKlebl, Co-Founder, Development Director of
GeoSolar Technologies Inc. (“GST”) is a Colorado-based climate technology company and the creator of the Smart Green Home® system for newly built and existing residences and commercial buildings. The company is focused on revolutionizing the way we heat, cool and power homes with 100% natural energy sources. Its patent-pending integrated system harnesses energy from the earth and sun to power and purify homes and automobiles without the use of fossil fuels.
In a GST home, the sun’s energy is captured on the roof to generate all of the electricity required. Additionally, the consistent climate of the earth is used to keep the home at a perfect temperature year-round, and the company’s proprietary air purifying unit ensures that the air inside the home is safe and healthy.
GST’s home technology has been installed in multiple test homes in Colorado and achieved exceptional results, including some of the most impressive energy efficiency ratings (HERS) in the industry.
GeoSolar Technologies is currently accepting investment as part of a Regulation A+ offering. Everyone* can invest now for as little as $300. For more information, visit the company’s profile on Manhattan Street Capital and review its Offering Circular.
GeoSolar Technologies Inc. (“GST”) has been qualified by the U.S. Securities and Exchange Commission (SEC) to conduct a Regulation A+ capital raise. GST is already a publicly traded company who makes quarterly and annual filings with the SEC and is subject to quarterly PCAOB audits. This is the first time shares of GeoSolar Technologies are being made available for public purchase. Upon completion of this Regulation A+ offering, the company intends to seek a listing of its stock.
The Decarbonization Movement
 Soaring and unstable energy/fuel costs continue to highlight the importance of rethinking the traditional approach to powering homes, from top to bottom. While most everyone is well aware of the remarkable, multi-trillion-dollar opportunity the electric vehicle transformation offers to investors (in addition to the benefits to the climate problem), few recognize that the all-electric home market is as large as electric vehicles and equally important to reducing carbon emissions.
Soaring and unstable energy/fuel costs continue to highlight the importance of rethinking the traditional approach to powering homes, from top to bottom. While most everyone is well aware of the remarkable, multi-trillion-dollar opportunity the electric vehicle transformation offers to investors (in addition to the benefits to the climate problem), few recognize that the all-electric home market is as large as electric vehicles and equally important to reducing carbon emissions.
U.S. energy expenditures clocked in at $3,891 per person in 2018, leading to estimated spending of $1.3 trillion on energy that year alone. Despite this, fewer than 3% of U.S. homes are currently powered by solar. This number is poised to increase exponentially as both new and existing residences transition to zero carbon models.
GST estimates that if all the homes in America were powered by its technology, carbon pollution could be reduced by an estimated 1.9 trillion pounds per year, greatly reducing the negative impacts on our climate.
GeoSolarPlus®
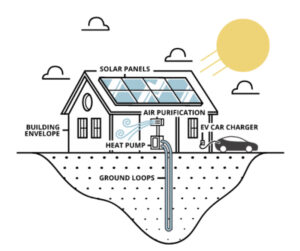 The GeoSolarPlus (“GSP”) system combines solar power, geothermal ground-sourced energy and other clean energy technologies into one fully integrated system.
The GeoSolarPlus (“GSP”) system combines solar power, geothermal ground-sourced energy and other clean energy technologies into one fully integrated system.
Key benefits of the GSP system include:
- Making a real planet-changing difference in reducing air pollution
- Eliminating or significantly reducing homeowners’ future utility bills
- Enjoying lifetime energy independence and protection from price escalation and energy shortages
- Eliminating greenhouse gas emissions from operation of home and daily life
- Increasing home value
- An integrated design for seamless operation of renewable energy systems
- Maintaining a significantly healthier living environment
- Leveraging existing renewable energy tax credits and electrification incentives
- Creating stable jobs capable of supporting families in the decarbonized future
Click here to learn more about how GeoSolarPlus works.
Management Team
The GST leadership and management team includes some of the world’s most experienced and respected leaders in the fields of decarbonization and sustainable homes.
Stone Douglass is the Chairman and CEO of GST. He is a seasoned, 30-year public company executive and former Chairman and CEO of the Piper Aircraft Company.
Brent Mosbarger is the company’s Co-Founder and leads its commercial operations. He is a highly respected solar engineer whose experience includes roles with Chevron Energy’s green operations and serving as project manager and executive for a $400 million solar/geothermal innovation project.
Peter Romenesko is a Senior Strategic Advisor with GST. He brings to the company considerable experience as an engineer and large-scale project manager for Johnson Controls and Siemens.
Dr. Norbert Klebl is the company’s Co-Founder and Development Director. Recognized as one of the world’s leading experts in the field of zero-carbon innovation, he is a former McKinsey partner of 16 years with an MBA from Columbia.
Dar-Lon Chang is GST’s Director of New Product Development. Prior to joining GST, he had a 16-year career with ExxonMobil Energy Research. He received his PhD in engineering from the University of Illinois.
* Must be over 18, certain states are not currently available and will be added soon.
Recent News
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Reimagining the Suburbs as Environmentally Green
- QualityStocksNewsBreaks — GeoSolar Technologies Inc.’s Renewable Energy and All-Home Solution Providing Resilience to Extreme Weather Events
- GeoSolar Technologies Inc. Eases Green Transition for Building Owners with Total Home Renewable Energy Systems

Advanced Container Technologies Inc. (OTC: ACTX)
The QualityStocks Daily Newsletter would like to spotlight Advanced Container Technologies Inc. (OTC: ACTX).
The government of Germany recently published a proposal detailingits
Advanced Container Technologies Inc. (OTC: ACTX) is in the business of selling and distributing self-contained, automated, indoor “micro-farms” called Grow Pods, along with related equipment and supplies. Additionally, the company designs and sells patented proprietary medical-grade plastic containers, known as the Medtainer®, that store and grind pharmaceuticals, herbs, teas and other solids or liquids.
ACTX is the leading distributor of Grow Pods. With a controlled environment, food and herbs can be grown without pesticides, harmful chemicals or risk of pathogen contamination, and with low energy consumption. Restaurants, grocery stores, non-profits, MSOs and entrepreneurs can use Grow Pods to ensure a fresh supply of ultra-clean produce year-round.
The company entered the Grow Pod business in November 2020 with its acquisition of all shares of Advanced Container Technologies Inc., a California corporation. As of February 28, 2022, ACTX is exploring the acquisition of the assets and the assumption of some or all of the liabilities of GP Solutions Inc., the developer and manufacturer of Grow Pods, for which ACTX is currently the sole U.S. distributor.
Because Grow Pods can be located almost anywhere, produce can be grown closer to the point of consumption and harvested at its peak, providing nutritious fruits and vegetables where needed. Indoor micro-farms, utilizing a practice known as vertical farming, have attracted the attention of governments and universities, which are now promoting vertical farming as a way to combat food insecurity and inequities.
The United States Department of Agriculture (USDA) has stated that vertical farming “is no longer a futuristic concept.” The department is enthusiastic about vertical farming, particularly those utilizing repurposed shipping containers, such as Grow Pods. Arizona State University reports that vertical farming reduces water use by 90 percent compared to conventional farming but produces 10 times the crop yield.
Products
Grow Pods
One of the company’s main business units is focused on selling advanced, self-contained hydroponic containers called Grow Pods. These unique and innovative automated systems are essentially micro-farms that can be placed virtually anywhere and, with their controlled and specially filtered environment, allow cultivation of a wide variety of crops, 365 days a year. The Grow Pod controlled environment offers major advantages for the production of high-value crops. The ability to grow year-round and the ability to cultivate in a smaller footprint using less water and power are some of the primary advantages of the system. Grow Pods offer constant temperature, humidity and airflow control, as well as automated watering and lighting schedules for optimal growth and minimal labor requirements, regardless of crop.
Containers
ACTX meets the needs of the pharmaceutical and medical markets, including the cannabis and hemp industries, with patented packaging systems. The company designs, customizes, brands and sells proprietary medical grade plastic containers that can store pharmaceuticals, herbs, teas and other solids or liquids, with a special built-in feature that can grind solids and shred herbs. The company’s flagship container product is the patented Medtainer®, a child resistant, medical-grade herb container and grinder that is water-tight, air-tight and smell proof. Packaging in the cannabis industry is critical, with numerous stringent regulations about how cannabis products must be packaged and labeled. ACTX also offers custom-branded, compliant vacuum seal bags and other retail container solutions.
Equipment and Supplies
ACTX markets and sells two principal products: Grow Pods, which are specially modified insulated shipping containers manufactured by GP Solutions Inc., in which plants, herbs and spices may be grown hydroponically in a controlled environment, and Medtainers®, which may be used to store pharmaceuticals, herbs, teas and other solids or liquids and can grind solids and shred herbs. The company also markets and sells various products related to Grow Pods and the Medtainer®, as well as providing private labeling and branding services for purchasers of Medtainers® and certain related products.
GP Solutions manufactures and sells other products, such as humidity controllers and LED lighting systems for vertical farming. The company’s specially designed lighting panels are programmed to emit the exact wavelength of light that each crop requires. The system has a daybreak-to-nightfall feature that gives plants the proper chromatic signals to grow rapidly and fruitfully. High efficiency LED light strips supply the crops with a red and blue light spectrum required for photosynthesis in the spectrum that plants need most.
Market Overview
The global vertical farming market is expected to reach $33.02 billion by 2030, according to a new report by Grand View Research. The market is forecast to expand at a CAGR of 25.5 percent from 2022 to 2030, according to Grand View. Escalating production of biopharmaceutical products, including cannabis, is anticipated to drive the market. The building-based segment of the market is expected to register a significant CAGR of 27.8 percent over the projected period. In addition, the climate control segment is expected to see high growth.
The global cannabis packaging market is expected to reach $14.34 billion by 2028, according to analysis by Reports and Data. The analysis forecasts 1,700 percent growth in cannabis users by the end of 2026, with packaging likely observing a whopping 26.42 percent growth in the forecast period. There are significant barriers to entry in the cannabis packaging market, giving an advantage to companies already established in the sector. These barriers include developing a thorough knowledge of the myriad regulations that govern cannabis packaging (which differ in each state), and child-resistance requirements.
Management Team
Douglas P. Heldoorn is the Founder and Chairman of Advanced Container Technologies Inc. He also holds the positions of President, CEO and COO at the company. Mr. Heldoorn has served on the Board of Directors since its inception in 2013. He has also previously held the position of Executive General Manager at Nissan Motor Corp.
Jeffory A. Carlson is CFO and Treasurer of ACTX. Mr. Carlson has also served as the company’s Corporate Controller since 2014.
Advanced Container Technologies Inc. (OTC: ACTX), closed Monday's trading session at $0.45, even for the day. The average volume for the last 3 months is and the stock's 52-week low/high is $0.2005/$1.87.
Recent News
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - German Plan to Legalize Cannabis Won't Permit Imports
- 420 with CNW - Federal Commission Considers Amending Sentencing Guidelines on Past Cannabis Use
- 420 with CNW - AOC Calls Out Biden for Excluding Undocumented Immigrants from Cannabis Pardons
Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F)
The QualityStocks Daily Newsletter would like to spotlight Hillcrest Energy Technologies Ltd. (OTCQB: HLRTF).
•The manufacture of lower cost electric vehicles is conditional onthe decrease of component costs, with battery packs forming one ofthe largest parts of their cost structure
•Hillcrest Energy Technologies’ revolutionary inverter technology,which forms an integral part of an electrical vehicle’s powertrain,allows EV’s to improve thermal management and reduce switchinglosses
•Improved power quality resulting from more efficient powerinverters can enable automakers to obtain higher powertrainefficiency and consequently, reduce their battery packs
•Hillcrest Energy Technologies separately seized the opportunity toannounce the appointment of Samuel Yik as their new CFO
One of the world’s most inexpensive electric vehicles, the ChangliFreeman, currently has a listing price of approximately $1,000. Itweighs 323 kilograms, packs up to a 1.2 kW motor, can reach 35km/hand has a claimed range of 40-100 kilometers (https://ibn.fm/ay2wL). By contrast and as of 2018, a Tesla Model 3’s inverter alone wasestimated to cost the manufacturer upwards of $1,100 per unit.Hardware costs have come down dramatically in ensuing years; as of2020, a Model Y inverter was reported to cost approximately $700per unit, a $400 reduction relative to the 2018 Model 3 version.Nevertheless, component costs remain a key constraint to thefurther reduction of automotive manufacturing costs, a challengewhich
Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) is a clean technology company based in Vancouver, British Columbia, engaged in developing high-value, high-performance power conversion technologies and digital control systems for next-generation powertrains and grid-connected renewable energy systems.
From concept to commercialization, Hillcrest invests in the development of energy solutions that power a more sustainable and electrified future. Hillcrest power inverter technology helps produce efficiencies in electrification and maximize the performance of electric systems, including electric vehicles (EV), motors and generators.
The company offers a flexible, single-inverter architecture that can be applied at nearly every stage of the electrification ecosystem, from renewable energy generation through the charging and operation of an EV, to provide full-cycle efficiency and performance improvements.
As momentum to electrify and decarbonize energy systems accelerates, Hillcrest believes the power inverter is increasingly emerging as a key component. While system cohorts such as battery packs, PV panels and electric motors are often in the spotlight, the inverter holds the key to unlocking efficiency and performance improvements.
Hillcrest power inverter technology is:
- REVOLUTIONARY: high-efficiency inverter technology has the potential to revolutionize how motors respond and how efficiency is gained.
- AGILE: able to deliver and deploy high-efficiency inverter solutions purpose-designed to meet specific customer needs.
- INNOVATIVE: technology-forward, clean-energy experts who are focused on advancing and optimizing efficient alternative energy use across all electric vehicle and charging platforms.
- A MARKET LEADER: a next-generation technology provider to the automotive industry’s top suppliers and manufacturers.
Technology & Applications
Hillcrest’s first application for its inverter technology – a 250 kW|800V Hillcrest SiC high efficiency traction inverter – is focused on the growing EV market. Hillcrest technology eliminates traditional design trade-offs faced across the power industry – deploying higher switching frequencies has historically meant a greater increase in losses, lower system efficiency and higher heat. Through a combination of hardware and software expertise, Hillcrest enables power applications to leverage higher switching frequencies AND
- Realize improved power system performance and reliability
- Operate at higher power levels without compromising efficiency
The expected benefits of Hillcrest’s traction inverter have been confirmed via testing and shared in a technical white paper, published in April 2022, that confirmed the following results:
- Significant efficiency gains – 99%-plus inverter efficiency
- Increased power density targeting 50kW/L+
- Significantly increased motor efficiency
- Lower stress on mechanical and electrical parts, enhancing reliability
- Improved thermal management
Hillcrest has also filed a patent for an enhanced powertrain solution that offers the potential to simplify EV charging and redefine how the industry envisions charging infrastructure. The company believes the most exciting benefit of the enhanced powertrain solution is the ability to eliminate the onboard charger and booster from an EV, as well as faster, anywhere charging including direct DC, wireless, and bidirectional charging across current and future power levels. Hillcrest sees this as a true EV charging game changer.
The company’s technology applies to nearly every clean energy industry segment:
- Wind power – an inverter is deployed at a wind turbine generator to convert the AC output, with at least one additional inverter used to deliver the power to the grid/battery.
- Solar power – an inverter is used to convert the DC output from the photovoltaic panels into the AC power that flows to the grid/battery/home.
- Energy storage – an inverter is deployed to convert the DC output from the storage system or batteries to the AC power that flows to the grid/home/EV.
- EV fast chargers – an inverter converts the AC input from the grid/storage system to the DC output needed to charge an EV’s battery.
Market Outlook
According to an April 2022 market analysis by Vantage Market Research (VMR), the global power inverter market is expected to reach a value of $95 billion by 2028, driven by increasing demand for EVs, energy generating wind turbines and solar-powered photovoltaic systems. That jump is forecast from an estimated $70.5 billion market value in 2021 and represents a compound annual growth rate of more than 5%.
According to the VMR report, many governments in countries around the world are supporting alternative options for efficient and nonpolluting energy generation. This has boosted demand for wind energy and solar energy systems. Hillcrest is aiming to capture a share of this future market growth across nearly every segment of the clean energy industry.
Management Team
Don Currie is the founding CEO of Hillcrest Energy Technologies. He has led the company’s successful transition from fossil fuels into clean energy technologies. Earlier in his career, he held various senior level positions, including director, officer and vice president of corporate communications with Enhanced Oil Resources Inc., an oil and gas exploration and production company based in Houston. Prior to that, he worked in other private and public ventures spanning the mining, gaming and technology sectors.
Jamie L. Hogue is the COO of Hillcrest. She brings more than two decades of progressive policy leadership, economic analysis and organizational development experience to Hillcrest. She builds collaborative processes and solutions that drive growing organizations toward a more resilient future. She previously served as the director of operations for Arizona State University’s Ten Across initiative – a compelling observatory positioned on the front lines of economic, social and climate change. She earned a master’s degree in public administration and a bachelor’s degree in economics from Arizona State University.
Ari Berger is Chief Technology Officer at Hillcrest. He brings over a decade of commercial experience with a track record of deploying new electrification technologies and go-to-market strategies. In 2015, he founded NIG Systems Ltd. in Israel, which specializes in custom high performance control systems design. Prior to this, he previously worked for Bental Industries, a leading motor manufacturer. He holds a master’s degree in system control engineering from the Technion – Israel Institute of Technology.
Raj Clair is CFO at Hillcrest. She is a CPA who began her career at Deloitte and has served in advanced finance positions in the energy and resources sector. She has been responsible for reporting, audits and internal controls, as well as working on budgeting and forecasting. She has worked with various publicly listed companies, including SEC registrants, and has both Canadian and U.S. experience. She holds a bachelor’s degree in accounting from Simon Fraser University.
Hillcrest Energy Technologies Ltd. (NASDAQ: HLRTF), closed Monday's trading session at $0.0879, even for the day. The average volume for the last 3 months is and the stock's 52-week low/high is $0.072/$0.1724.
Recent News
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd.'s (CSE: HEAT) (OTCQB: HLRTF) Enhanced Solution Holds Potential to Simplify, Redefine EV Charging
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Assisting EV Manufacturers in Reducing Costs; Company Announces Appointment of Samuel Yik as CFO
- InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Engages IMS for Investor Communications Expertise
Silo Pharma Inc. (OTCQB: SILO)
The QualityStocks Daily Newsletter would like to spotlight Silo Pharma Inc. (OTCQB: SILO).
The psychedelic medicine field is growing rapidly, as centers
Silo Pharma Inc. (OTCQB: SILO), a developmental stage biopharmaceutical company, is focused on merging traditional therapeutics with psychedelic research for people suffering from indications such as post-traumatic stress disorder (PTSD), fibromyalgia, Alzheimer’s disease, Parkinson’s disease, and other rare neurological disorders. Silo’s mission is to identify assets to license and fund research that the company believes will be transformative to the wellbeing of patients and the health care industry.
Silo is committed to developing innovative solutions to address a variety of underserved conditions. Combining Silo’s resources with world-class medical research partners, the company looks to make significant advances in the medical and psychedelic space.
Silo works to identify and partner with leading medical universities, providing the needed financial resources to develop safe therapeutic treatments while moving cutting-edge research through the clinical stage and into commercialization. The company is well-capitalized with access to additional funds as opportunities present themselves.
Silo recently engaged Donohoe Advisory Associates LLC for consulting and advisory services in connection with the potential uplisting of Silo’s common shares to the Nasdaq Stock Market.
Research
Silo has entered into research agreements and partnerships with multiple leading medical universities.
The company is involved in a sponsored study with Maastricht University utilizing repeated low doses of ketamine and psilocybin to examine the effects on cognitive and emotional dysfunctions in Parkinson’s disease and to understand its mechanism of action. The investigator in the Netherlands is acquiring the substances for the study and will then finalize the documentation to submit to the ethics committee.
Additionally, in June 2021, Silo announced its entry into a scientific research agreement with the University of California San Francisco (UCSF). The agreement will leverage four other clinical trials being planned by the university to determine the effects of psilocybin on inflammation. The study will take place at The Translational Psychedelic Research (TrPR) Program at UCSF.
Silo also recently extended its exclusive option agreement with the University of Maryland, Baltimore (UMB) to explore a novel invention generally known as joint-homing peptides. These peptides are being developed for use in the investigation and treatment of arthritogenic processes and can be used for enhanced targeting of therapeutic agents.
This agreement includes the study of two separate peptides. The first is an option and study for the treatment of arthritis. The second is a patented licensed peptide for the central nervous system, with an initial study for MS autoimmune diseases, in addition to rheumatoid arthritis. Animal studies are underway for both initial indications relating to the UMB agreement, with the potential for studies evaluating additional indications in the future.
Finally, Silo signed an agreement with Columbia University granting it an option to license certain assets currently under development, including an Alzheimer’s disease formulation targeting NDMARs and 5-HT4Rs, as well as a prophylactic treatment for stress-induced disorders and PTSD. Both candidates are currently being tested in mice and have already provided early data.
In addition to its university partnerships, Silo entered a joint venture agreement with Zylo Therapeutics Inc. (“ZTI”) focused on the development of ketamine and psilocybin using ZTI’s Z-Pod™ technology for the transdermal time released delivery of therapeutics. In November 2021, the company announced ZTI’s reception of its first ketamine shipment and initiation of loading ketamine into its Z-Pod technology. In a news release, Eric Weisblum, CEO of Silo, called the development an “important milestone” that will help the company “study the benefits of slow-release transdermal release of Ketamine.”
Market Overview
According to Coherent Market Insights, the fibromyalgia treatment market was valued at $2.78 billion in 2018 and has a projected CAGR of 3.3% over the forecast period 2018 to 2026. Fibromyalgia is a condition that causes pain all over the body, sleep problems, fatigue, and emotional and mental distress.
The global PTSD therapeutics market is expected to reach $10.68 billion by 2026 with a CAGR of 4.5% during the forecast period from 2018 to 2026, according to a report by Credence Research. Growing prevalence of PTSD is the chief factor driving the global treatment market. Increases in events such as wars, combat, and interpersonal violence has been a major contributing factor. Other factors like growing emphasis on rehabilitation initiatives by governments for treating their war veterans has also been facilitating the increase in demand for PTSD therapeutics.
Fortune Business Insights reports the global Parkinson’s disease treatment market is predicted to grow to $8.38 billion by 2026, with a CAGR of 8.1% during the forecast period. Parkinson’s is a neurodegenerative disease of the central nervous system which primarily affects the brain, causing uncontrollable shaking and tremors, difficulties in balance and restricted body movement making it difficult for the person to function or perform a daily routine.
Management Team
Eric Weisblum is CEO and founder of Silo Pharma. He has over 25 years of Wall Street experience, most recently in the biotechnology sector. He has served on the board of Aikido Pharma and was the president of Sableridge Capital. He has a proven track record in licensing therapeutic assets and assisting in their development. He brings to the company nearly 20 years of expertise in structuring and trading financial instruments. He holds a bachelor’s degree from the University of Hartford’s Barney School of Business.
Dr. Kevin Muñoz was appointed to the Silo board of directors in November 2020. He teaches biomedical sciences and medical intervention for the Passaic County Technical Institute. He previously served as Director of Operations at Physical Medicine and Rehabilitation. He began his career with Harlem Health Promotion Center in New York City as a research assistant. He earned a bachelor’s degree from the University of Michigan and a Doctor of Medicine from Xavier University School of Medicine.
Josh Woolley, M.D., Ph.D., is a Scientific Advisor for Silo. He is an associate professor in the Department of Psychiatry and Behavioral Sciences at the University of California, San Francisco. He is also a psychiatrist on staff at the San Francisco Veterans Affairs Medical Center. He is the director and founder of the Bonding and Attunement in Neuropsychiatric Disorders Laboratory. He received both his M.D. and his Ph.D. in Neuroscience from UCSF, where he completed his psychiatry residency training.
Charles Nemeroff, M.D., Ph.D., is a Scientific Advisor for Silo Pharma. He directs the Institute for Early Life Adversity Research within the Department of Psychiatry and Behavioral Sciences as part of the Mulva Clinic for the Neurosciences. He was chair of the Department of Psychiatry and Behavioral Sciences and clinical director of the Center on Aging at the University of Miami Miller School of Medicine. He received his M.D. and Ph.D. in neurobiology from the University of North Carolina School of Medicine.
Silo Pharma Inc. (OTCQB: SILO), closed Monday's trading session at $3.6, off by 4%, on 6,880 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $3.52/$12.445.
Recent News
- Silo Pharma Inc. (OTCQB: SILO) - New Psychedelic Journal to Focus on Publishing Psychedelic Research
- Silo Pharma Inc. (NASDAQ: SILO) Extends Columbia University Agreement to Continue Research for Therapeutics to Treat Alzheimer's Disease
- PsychedelicNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Confident in Preclinical Work on Its Novel Ketamine Formulation
Cepton Inc. (NASDAQ: CPTN)
The QualityStocks Daily Newsletter would like to spotlight Cepton Inc. (NASDAQ: CPTN).
Cepton Inc. (NASDAQ: CPTN) is a provider of state-of-the-art, intelligent, lidar-based solutions serving a range of markets, including automotive (ADAS/AV), smart cities, smart spaces and smart industrial applications. General Motors (NYSE:GM) has granted a series production award for Cepton’s lidar, the biggest such award to date in the automotive space. Cepton’s is the lidar component of GM’s Ultra Cruise autonomous driving platform. By leveraging its patented Micro Motion Technology (MMT®) lidar platform, the company develops reliable, scalable and cost-effective solutions that deliver long-range, high-resolution 3D perception for smart applications.
Cepton was established in 2016 by co-founders Dr. Jun Pei and Dr. Mark McCord. The company is headquartered in San Jose, California, and serves a fast-growing customer base through an international presence spanning North America, Germany, Japan, India and China.
Micro Motion Technology (MMT®)
Cepton was built from the ground up to meet key lidar industry challenges for mass market adoption. This company’s portfolio of proprietary technology is uniquely aimed at facilitating this industry growth through a combination of performance, reliability, affordability and design integration.
Key among its innovations is MMT®, a mirrorless, frictionless, rotation-free 3D imaging platform designed specifically for lidars. Its benefits for OEMs and system integrators include:
- Reliability – The durable design uses common, easily attainable materials.
- Versatility – The platform is capable of achieving near- to ultra-long range with a wide field of view.
- Efficiency – MMT® features a compact form factor, low power usage and inexpensive components.
- Scalability – Its simple design means that scale-up to high manufacturing volumes is easily attainable.
Because of their compact form factor, Cepton lidars are embeddable and ideally suited for advanced driver-assistance system (ADAS) integration, whether behind windshield, in headlamp or in fascia.
Agreement with KOITO
KOITO Manufacturing Co. Ltd., the world’s premier Tier 1 auto lighting supplier, originally started an evaluation of Cepton’s MMT® based lidars in 2018. In 2020, KOITO made an investment in Cepton aimed at accelerating the company’s development and enabling KOITO’s industrialization of high-performance and high reliability lidar sensors for ADAS and autonomous vehicle (AV) applications.
Through this collaboration, Cepton was able to secure the largest ADAS lidar series production award[1] with General Motors as a sole source in the automotive space. The award covers GM vehicles for the initial period of 2023-2027.
On August 5, 2021, the two companies deepened their relationship when KOITO committed to invest a further $50 million in Cepton’s business through its participation in a Private Investment in Public Equity (PIPE) offering of shares of common stock of Growth Capital Acquisition Corp. in connection with Cepton’s recent merger.
Collaboration with GM
On July 13, 2021, Cepton announced that it had secured an ADAS lidar series production award from a leading, Detroit-based global automotive OEM – the biggest lidar production award by any OEM to any lidar company. It was later clarified that the OEM was General Motors, and Cepton’s lidar is part of GM’s ADAS Ultra Cruise system.
GM is “expected to deploy Cepton lidars in its next generation of advanced driver assistance systems (ADAS) across multiple vehicle classes and models – not just luxury cars.” As such, the agreement marks the potential for “an industry-first, mass-market adoption of lidar technology for automotive ADAS, with an anticipated deployment in consumer vehicles starting in 2023.”
On July 28, 2021, Ford Motor Company (NYSE: F) distributed an article on Medium noting, “Ford has been engaged with Cepton almost since their inception in 2016, both for R&D collaboration and small-scale deployments. Cepton LiDAR are deployed in some of [Ford’s] smart city projects. Based on Ford’s guidance, Cepton delivered a custom version of their LiDAR to enable R&D on advanced ADAS features.”
Market Outlook
Driven by increasing development and adoption in automobile safety applications, environmental mapping and 3D-modeling, the global lidar market is forecast to experience considerable growth over the coming years. A research report published by MarketsAndMarkets suggests that the sector will grow to an estimated $3.4 billion by 2026, achieving a CAGR of 21.6% over the next five years.
The report further highlights increasing investments in lidar startups by automotive giants as a driver of growth opportunities in the sector, particularly in North America.
In 2020, ground-based lidar accounted for the lion’s share of the overall lidar market, and this trend is expected to continue as the automotive sector continues to rapidly advance adoption across the full spectrum of vehicle classes. One factor not to be underestimated is the high barrier of entry and the exceptionally long time required for automotive OEMs to vet and award a production win to a lidar company. It is a commonly held view that the over 50 lidar companies will inevitably coalesce into a handful serving all OEMs.
Cepton, having a head start through its established partnership with leading global OEM GM, is uniquely positioned to capitalize on this market growth in the years to come.
Management Team
Cepton’s founder-led team is made up of lidar industry pioneers with decades of collective experience across advanced lidar and imaging technologies.
Jun Pei, Ph.D., is the company’s CEO and Co-Founder. He is a technology specialist with a focus in optics and electronics. Prior to founding Cepton, Dr. Pei founded AEP Technology, a firm focused on developing advanced 3D optical instruments. He received his Ph.D. in electrical engineering from Stanford University.
Mark McCord, Ph.D., is Cepton’s CTO and Co-Founder. Prior to founding Cepton, he led advanced development at KLA-Tencor. Dr. McCord also formerly served as an associate professor at Stanford University, where he earned his Ph.D. in electrical engineering.
Winston Fu, Ph.D., is the company’s CFO. Dr. Fu is the founder of Silicon Valley venture capital firm LDV Partners. Prior to joining Cepton, he served as CFO and Chairman of Active-Semi before its acquisition. Dr. Fu has also helped to build many technology companies as an entrepreneur and/or board member. He received his Ph.D. in applied physics from Stanford University, as well as an MBA from the Kellogg School of Management at Northwestern University.
[1] Largest known ADAS lidar series production award based on number of vehicle models awarded
Cepton Inc. (NASDAQ: CPTN), closed Monday's trading session at $1.94, off by 13.0045%, on 386,887 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.01/$80.16.
Recent News
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Technologies Inc. (NASDAQ: CPTN) Partner with Leading Entities to Complete Project Resulting in Enhanced Road Safety
- InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) to Participate in 13th Craig-Hallum Alpha Select Conference
- TinyGemsBreaks - Cepton Inc. (NASDAQ: CPTN) Secures LOI for up to $100M to Help Fund next Stage of Growth
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
The QualityStocks Daily Newsletter would like to spotlight BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV).
•BiondVax has an exclusive worldwide license to commercializeNanoAbs for the treatment of COVID-19
•The company plans to initiate a Phase 1/2a clinical trial of itsinhaled COVID-19 therapeutic NanoAb in 2023
•BiondVax has exclusive options for worldwide licenses for NanoAbsto treat other disorders with large, underserved needs
•The company has filed a Registration Statement with the SEC for anunderwritten offering of shares
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) is a biopharmaceutical company focused on developing, manufacturing and commercializing innovative products for the prevention and treatment of infectious diseases and other illnesses.
In collaboration with the prestigious Max Planck Institute for Multidisciplinary Sciences (MPG) and the University Medical Center Göttingen (UMG), both in Germany, BiondVax is developing a pipeline of innovative nanosized antibody (NanoAb) therapies addressing diseases underserved by current treatments and with large and growing markets, such as COVID-19, asthma and psoriasis.
NanoAbs, also known as VHH-antibodies or Nanobodies, are alpaca-derived nanosized antibodies that exhibit multiple significant competitive advantages over existing antibody therapies, including stability at high temperatures, superior binding affinity, more effective and convenient routes of administration and efficient production. BiondVax is uniquely positioned to advance nanosized antibody innovation from R&D through commercialization.
The company’s highly experienced and successful pharmaceutical industry leadership team includes former senior executives from Novartis, GSK and Bristol-Myers Squibb.
Since its founding, BiondVax has executed eight clinical trials, including a seven-country, 12,400-participant Phase 3 trial of a prior influenza vaccine candidate, and it built, owns and operates a 20,000 sq. ft. state-of-the-art GMP biologics manufacturing facility housing its laboratories, production facilities and offices.
Lead Candidate: Inhaled COVID-19 NanoAb
In December 2021, BiondVax signed definitive agreements with the Max Planck Society – parent organization of the Max Planck Institute for Multidisciplinary Sciences– and the UMG to enter a strategic collaboration for the development and commercialization of innovative COVID-19 NanoAbs.
The company is planning a rapid development path that leverages its expertise and capabilities in biological drug development and manufacturing. BiondVax anticipates preclinical proof-of-concept results for an inhaled COVID-19 NanoAb by the end of 2022, with initial Phase 1/2a human clinical trial results expected in 2023.
The intended inhaled mechanism of delivery of BiondVax’s COVID-19 NanoAb formulation may serve as a significant differentiator when compared to approved monoclonal antibodies, which are injected. Inhaled delivery has shown to be cheaper, more convenient and likely safer for patients and providers.
NanoAb Pipeline: Psoriasis, Asthma and More
The COVID-19 NanoAb development agreement is part of a broader five-year research collaboration agreement signed in March 2022 covering discovery, development and commercialization of NanoAbs for several other disease indications with large market medical needs, including asthma, psoriasis, macular degeneration and psoriatic arthritis.
BiondVax has an exclusive worldwide license for development and commercialization of COVID-19 NanoAbs and exclusive options for similar worldwide licenses for NanoAbs for the above mentioned additional large market disorders currently underserved by approved therapeutic antibodies.
Academic research teams from MPG and UMG have verified strong affinity by the new NanoAbs to their biological target molecules and high thermostability. They have also demonstrated strong neutralization by several NanoAb candidates of their respective target molecules. Neutralization studies of the other NanoAbs are expected to begin later in 2022.
Based on the promising results, BiondVax will focus development efforts beginning with the following NanoAbs:
- NanoAbs targeting IL-17 as drug candidates for the potential treatment of psoriasis and psoriatic arthritis
- NanoAbs targeting IL-13 and NanoAbs targeting TSLP as drug candidates for the potential treatment of asthma
These are conditions for which the antibody target is validated by existing treatments and the mechanism of action is well understood. Both represent large medical needs and growing markets. BiondVax anticipates preclinical proof-of-concept for at least one of these NanoAbs in 2023. This is in addition to the aforementioned human clinical Phase 1/2a for the inhaled COVID-19 NanoAb therapy, which is also anticipated in 2023.
Market Opportunity
COVID-19 treatment, target of the company’s lead NanoAb therapy candidate, had an estimated market size of $22 billion in 2021.
Future BiondVax drug candidates will target conditions with large markets growing at attractive CAGRs.
The global asthma treatment market was valued at $18.08 billion in 2019 and is projected to reach $26.01 billion by 2027, exhibiting a CAGR of 4.5% during the forecast period, according to Fortune Business Insights. The research firm predicts that the global psoriasis treatment market will grow from $26.37 billion in 2022 to $47.24 billion by 2029, exhibiting a CAGR of 8.7% over the forecast period.
Management Team
Amir Reichman is BiondVax’s CEO. He previously was Head of Global Vaccines Engineering Core Technologies at GSK Vaccines in Belgium. Prior to that, he held leadership roles at Novartis Vaccines’ Global Vaccines Supply Chain Management organization. He was the first employee of NeuroDerm Ltd., a company focused on transdermal drug delivery, and served as Chief Engineer and Senior Scientist until his departure in 2009. He earned a M.Sc. in Biotechnology Engineering from Ben-Gurion University and an MBA in Finance and Health Care Management from the University of Pennsylvania’s Wharton School.
Tamar Ben-Yedidia, Ph.D., is Chief Science Officer at BiondVax. She has more than 30 years of experience in immunology, with specific expertise in the development of vaccines. She began her career with Biotechnology General Ltd., working on development of a recombinant Hepatitis-B vaccine. She later joined the Weizmann Institute of Science, working on the design of a peptide-based vaccine against several pathogens. She is widely published, with numerous refereed articles and invited reviews in various scientific journals. She received her Ph.D. from the Weizmann Institute.
Elad Mark is COO at BiondVax. He has over 15 years of biotechnology industry experience encompassing diverse project stages including feasibility studies, conceptual and detailed design, commissioning, qualification and process validation. Prior to joining BiondVax, he led Novartis’s $800 million investment in a biologics facility in Singapore. With Biopharmax and Antero, both global pharmaceutical engineering companies, he successfully led projects in Israel, China and Singapore. He holds a BSc. in Engineering from the Afeka Tel Aviv Academic College of Engineering and an MBA from the Open University of Israel.
Uri Ben-Or is CFO at BiondVax. He has served as CFO with public life science companies traded on the TASE, OTC and Nasdaq. Ben-Or provides his services to BiondVax through CFO Direct, a company he founded and for which he serves as CEO. He served as the VP of Finance of Glycominds, a leading biotechnology company, and as CFO of a spin-off from Telrad Networks. He also served as a Corporate Controller at Menorah Capital Markets and as an Auditor at PWC. He holds a B.A. in Business from the College of Administration, an MBA from Bar-Ilan University, and is a CPA.
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV), closed Monday's trading session at $0.8291, off by 1.2859%, on 46,994 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.5335/$3.49.
Recent News
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Is 'One to Watch'
- InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Featured in Investment Research Report
- InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) to Present at RAFT 14 Conference
Flora Growth Corp. (NASDAQ: FLGC)
The QualityStocks Daily Newsletter would like to spotlight Flora Growth Corp. (NASDAQ: FLGC).
•Flora Growth is a cannabis cultivator and international branddeveloper with a strong base of operations at its licensed facilityin northern Colombia
•Flora recently announced its plans to acquire multi-nationaloperator Franchise Global Health (“FGH”), the latest of itsstrategic acquisitions designed to boost revenues and expandFlora’s international footprint
•FGH is focused on the German market, where its subsidiary was thefirst company to sell cannabis
•Germany is in turn expected to provide the companies a launchingpad for success in Europe, where opportunity is defined by the factthat currently Germany provides 75 to 80 percent of Europe’scannabis sales
•Flora has already reported cooperative agreements to increase itsproduct distribution pipeline to Switzerland and the Czech Republic
Cannabis cultivator, brand manufacturer and global distributor
Flora Growth Corp. (NASDAQ: FLGC) is an internationally focused cannabis brand builder that leverages natural, cost-effective cultivation practices to supply cannabis derivatives to its diverse business divisions, including cosmetics, hemp textiles, and food and beverage. Flora Growth operates one of the largest outdoor cultivation facilities in the world with an aim of marketing a higher-quality premium product at below-market prices. By prioritizing natural ingredients and value-chain sustainability across its portfolio, the company creates premium products that help consumers restore and thrive.
Flora Growth completed the first traditional cannabis IPO on Nasdaq in May 2021. Although currently headquartered in Toronto, Ontario, with plans to relocate its head office to Miami, Florida, the company’s base of operations is in Colombia, where it has built an extensive distribution network that includes Colombia’s largest distributors.
Currently, Flora Growth is organically growing market share for its existing brand portfolio (pharmaceuticals, textiles, cosmetics, and food & beverage) while seeking revenue-generating acquisitions that offer an accretive distribution network to amplify revenue growth.
Existing Brand & Product Portfolio
Flora Growth’s portfolio spans a number of verticals – each with a thoughtful brand designed to resonate with its intended end consumer. In line with the company’s mission, each brand prioritizes natural ingredients and value-chain sustainability.
Flora Lab S.A.S
Flora Lab is the company’s GMP certified manufacturing and R&D center focused on producing pharmaceuticals, cosmetics, and nutraceuticals for domestic and international markets. Its offerings include product lines that are private label, white-label, and custom formulas.
Through Flora Lab, Flora Growth has relationships with 1,500+ distribution channels, manufactures 63+ OTC products registered with INVIMA (Colombia National Food and Drug Surveillance Institute), and holds multiple GMP certifications enabling international export in an effort to leverage Flora Lab’s capacity to produce a wide range of CBD-infused products.
Flora Beauty
Flora Beauty is the company’s CBD beauty and cosmetics division founded by fashion and beauty industry icon Paulina Vega. Its current offerings include two CBD skincare brands targeting the U.S. and Latin American markets – MIND NATURALS and AWE. These lines exemplify Flora Growth’s socially conscious approach to business.
Currently, Flora Beauty products are offered globally through e-commerce, as well as through Falabella’s 111 retail locations across Latin America. The company is in negotiations with major department stores to launch the line in the U.S. and is also exploring opportunities in the U.K. and other European markets.

KASA Wholefoods
KASA Wholefoods is a Colombian manufacturer of food and beverages leveraging responsibly sourced exotic fruits from the Amazon. KASA has a $10 million+ distribution agreement with Tropi, Colombia’s largest food distributor, which has 130,000+ distribution points across the country.
Mambe, KASA’s leading brand, is already offered through over 980 distribution points across Colombia. Flora Growth expects this network to grow to over 1,200 distribution points in 2021, including one of Colombia’s largest coffee chains, Tostao Café & Pan.

Hemp Textiles & Co.
Through its Hemp Textiles division, Flora Growth intends to utilize its large land package and cultivation infrastructure to capture market share in the rapidly growing hemp industrials segment.
The company’s first brand through this division, Stardog Loungewear, offers a line of comfortable loungewear made from natural, organic materials. Stardog has been distributing globally through e-commerce and brick and mortar channels in Bogota since fall 2020, and the company intends to open U.S. brick and mortar locations in 2021.

Accretive M&A
Flora Growth is targeting transactions to complete the supply chain via key infrastructure to enhance its global distribution with the aim to compete on low-cost, high-quality inputs paired with premium brands that create business lines with robust margins.
To date, Flora has announced two major transactions.
Koch & Gsell (Acquisition)
- Amplify CPG portfolio’s revenue growth through leading brand, Heimat, currently with TTM revenues of $7.6 million.
- Leverage Koch &Gsell’s distribution network of 2,500+ stores to introduce Flora to the Swiss, European and Asian markets.
- Bring patented hemp cigarette manufacturing technology into new markets utilizing Flora’s high-quality cannabis.
Hoshi International (Investment)
- Equity Investment of €2 million into Hoshi to establish Flora as a preferred supplier to two EU processing facilities.
- Opens gateway for Flora Growth’s cannabis through international distribution agreements in the EU and U.K.
- Hoshi’s experienced team and increased access to the EU cannabis market to serve as a catalyst for revenue growth.
Cultivation
Key to Flora Growth’s expansion efforts is its cultivation strategy. The company’s Cosechemos farm, located in Bucaramanga, Colombia, is currently licensed to cultivate 247 acres of cannabis. Through three successful pilot crop plantings, the location has demonstrated a production cost of just $0.06/gram. For comparison, the average cost of North American cannabis (based on 2019 figures from Aphria, Tilray, Sundial, and Aurora) equates to roughly $1.89/gram.
Flora Growth is uniquely positioned to capitalize on Colombia’s favorable growing conditions, low-cost infrastructure, and affordable local workforce as it looks to ramp up its cultivation efforts moving forward.

Leadership Team
Bernard Wilson is the Chairman of Flora Growth. A senior financial professional, Dr. Wilson is the former Vice-Chairman of PricewaterhouseCoopers LLP and is the Chairman of the Founders Board of the Institute of Corporate Directors. He has also served as Chairman of the Canadian Chamber of Commerce; Chairman of the International Chamber of Commerce – Canada; and Member of the Canada/U.S. Trade Committee. Dr. Wilson draws on this experience to ensure Flora Growth adheres to effective corporate governance practices.
Luis Merchan is the company’s President and CEO. He is a proven executive with over a decade of experience in enterprise sales management, corporate strategy, merchandising and expense management, and customer experience. Mr. Merchan previously served as Macy’s Inc.’s Vice President of Workforce Strategy and Operations, where he managed the enterprise’s multi-billion-dollar P&L expense line for the entire 540 store portfolio. Throughout his tenure at Macy’s, he led various sales and marketing initiatives, including the B2B corporate sales team that was responsible for $160 million in annual revenue. Mr. Merchan obtained his Bachelor of Industrial Engineering from Pontifical Xaverian University in Bogota, Colombia, and his MBA from McNeese State University. He also holds a Graduate Certificate in Marketing Management from Harvard.
Juan Manuel Galan is a Strategic Advisor to the Flora Growth management team. Mr. Galan currently serves as a senior consultant to The World Bank. He is a politician and former senator of Colombia, serving three terms from 2006 to 2018 as a member of the Colombian Liberal Party. He is also a former professor at the University of Rosario and holds more than 20 years of journalistic, academic, governmental and parliamentary experience. During his time as a senator, Mr. Galan was a key leader, with 29 bills and 27 debates on political control, and 17 laws to his name. The most relevant of those laws was authoring the medical cannabis law that resulted in the legalization of medical cannabis in Colombia.
Stan Bharti is a Director of Flora Growth. Mr. Bharti currently serves as Executive Chairman of Forbes & Manhattan. He has more than 30 years of professional experience in business, finance, markets, operations and more, with a focus on the resource and technology sectors. To date, Mr. Bharti has amassed over $3 billion worth of investment capital for the companies with which he has worked and their shareholders. He is a Professional Mining Engineer and holds a master’s degree in engineering from Moscow, Russia, and University of London, England.
Javier Franco is the company’s VP of Agriculture. Mr. Franco is a master horticulturist with more than 25 years of experience in the design, implementation, and management of cultivation and propagation facilities of more than 30 species of cut flowers in Latin America. He completed his agricultural studies at Zamorano University in Honduras and later at an International Exchange Program at Ohio State University. Mr. Franco has directed technical, commercial, and research groups in the cut flower, fruit and vegetable markets in Latin America and has participated in the commercial development of new technologies applied in agribusiness. He has also led the agri-management of organic crops and certifications of Good Agricultural Practices.
Flora Growth Corp. (FLGC), closed Monday's trading session at $0.5092, off by 2.0769%, on 649,812 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.50/$5.6092.
Recent News
- Flora Growth Corp. (NASDAQ: FLGC) - Flora Growth Corp. (NASDAQ: FLGC) Agreement to Acquire Multi-national Cannabis Operator Positions Company for New Strategic Growth in Europe
- CannabisNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Increasing Access to Safe, Legal CBD and THC to Global Consumers
- Cannabis Regulation in the Spotlight During Midterm Elections at the Heels of Presidential Pardon; Flora Growth Corp. (NASDAQ: FLGC) Leveraging Colombian Stance to Manufacture Products at Lower Costs
Mullen Automotive Inc. (NASDAQ: MULN)
The QualityStocks Daily Newsletter would like to spotlight Mullen Automotive Inc. (MULN).
Mullen Automotive Inc. (NASDAQ: MULN) is a Southern California-based automotive company that owns and partners with several synergistic businesses working toward the unified goal of creating clean and scalable energy solutions. Mullen has evolved over the past decade in sync with consumers and technology trends. Today, the company is working diligently to provide exciting EV options built entirely in the United States and made to fit perfectly into the American consumer’s life. Mullen strives to make EVs more accessible than ever by building an end-to-end ecosystem that takes care of all aspects of EV ownership.
Commencement of Trading on Nasdaq
On November 5, 2021, Mullen announced its commencement of trading on the Nasdaq Capital Market.
“Today is a monumental day for Mullen Automotive. I am especially proud of our team, investors and all who have believed in Mullen and taken us to this point as a publicly traded company on the Nasdaq Capital Market,” David Michery, CEO and Chairman of Mullen Automotive, stated in the news release. “Trading on Nasdaq now opens us up to new investors, both institutional and retail shareholders, and broadens our awareness and company profile, while increasing awareness of Mullen and our technology platform and opening new opportunities in EV and beyond. The road ahead has never been brighter for Mullen, and I am proud to lead us into the future.”
The milestone came in the wake of the company’s stock-for-stock merger with Net Element Inc.
The Mullen FIVE
The Mullen FIVE EV Crossover, debuting at the Los Angeles International Auto Show (LAIAS) on November 17, 2021, embodies Mullen’s Southern California roots with an inspired design focused on two complementary Golden State themes – California landscape and California urban.
The FIVE is built on an EV Crossover skateboard platform that offers multiple powertrain configurations and trim levels in a svelte design that is Strikingly Different™ and exciting to experience in person.
Prior to the start of LAIAS, the Mullen FIVE was selected as a finalist by the LA Auto Show for Top EV SUV in the ZEVA “People’s Choice” Awards.
LAIAS provides Mullen an opportunity to display multiple variants of the FIVE model while also showcasing its powertrain, battery and charging technology. The company intends to bring the FIVE to market in 2024, and reservations are currently open here.
Mullen’s development portfolio also includes EV Fleet Vans, which it intends to bring to market in Q2 2022, and the pure electric, high performance Mullen DragonFLY.
Expansion of Manufacturing Capacity
On November 2, 2021, Mullen announced plans to expand its facility in Robinsonville, Mississippi.
Mullen’s Advanced Manufacturing and Engineering Facility (AMEC) currently occupies 124,000 square feet of manufacturing space. The total available land on the property is over 100 acres, and Mullen is moving ahead with plans to build out another 1.2 million square feet of manufacturing space to support class 1 and class 2 EV cargo vans and the Mullen FIVE EV Crossover.
On the expanded site, Mullen plans to build a body shop, a fully automated paint shop and a general assembly shop.
EV Market Outlook
The global EV market was reported to consist of 3,269,671 units in 2019, a figure that is expected to grow at a CAGR of 21.1% through 2030 to a total of 26,951,318 units worldwide. This market’s monetary value was estimated at $162.34 billion in 2019 and is expected to grow at a CAGR of 22.6%, resulting in an approximate value of $802.81 billion by 2027. The primary driver for this exponential growth is a worldwide increase in vehicle emissions regulations.
Management Team
David Michery is the CEO and Founder of Mullen and has been leading the company and its divisions since inception in 2014. With over 25 years of executive management, marketing, distressed assets, and business restructuring experience, Mr. Michery brings a wealth of relevant knowledge and expertise to the Mullen brand. He has notably created 12 trademarks so far to develop the company brand and vision.
Mr. Michery is working toward a sustainable future accessible to all by creating a suite of clean-energy electric vehicles at varied price points. With entirely U.S.-based manufacturing and operations, he is also determined to have Mullen Technologies play a role in shaping a self-sustaining local economy by creating more jobs in America.
Mr. Michery manages risks and company expectations as a pathway to success and has personally overseen several businesses that totaled over $1 billion in transactions. His key strength is the ability to be fiscally responsible and lead teams to complete projects on time and within budget. As a seasoned professional in this space, Mr. Michery has demonstrated skill in building businesses from the ground up and into successful entities that subsequently sold for hundreds of millions of dollars.
Mullen Automotive Inc. (MULN), closed Monday's trading session at $0.265, off by 5.4922%, on 170,746,710 volume. The average volume for the last 3 months is 165.784M and the stock's 52-week low/high is $0.2101/$15.90.
Recent News
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Eliminates Some $13M in Debt
- Mullen Automotive Inc. (NASDAQ: MULN) 'Strikingly Different' Test Drive Tour Shines Spotlight on Mullen FIVE
- InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Shares Responses from Participants of Its Strikingly Different FIVE EV Crossover Tour
Odyssey Group International Inc. (OTC: ODYY)
The QualityStocks Daily Newsletter would like to spotlight Odyssey Group International Inc. (OTC: ODYY).
•Odyssey Health Inc. is a medical product developer with a missionof filling unmet needs when it comes to acute emergencies involvingpatients’ central nervous systems
•The company recently announced the formation of a wholly ownedsubsidiary to help Odyssey develop its products as they movethrough the regulatory process
•Odyssey’s solutions include a breath-powered intranasal deliverymechanism and synthetic neurosteroids designed to providerapid-response treatment for patients with concussions and a highlyfatal pediatric neurological disease
•Odyssey Health also is developing solutions for heart diseasedetection, obstructed-airway choking and nerve agent exposure
•The concussion therapeutic solution recently completed Phase Itesting successfully and the company is preparing for Phase IIefficacy testing in cooperation with military service personnel
Medical technology solutions innovator
Odyssey Group International Inc. (OTC: ODYY) is a medical technology company focused on developing lifesaving medical products that offer technological and clinical advantages over current standards of care.
The company’s portfolio of product technologies is diverse, featuring four unique medical products in development. Odyssey’s goal is to deliver superior products with enhanced clinical utility and market potential, thereby yielding a high rate of return for its shareholders and partners. It is guided by a senior management team with significant experience relating to refining technologies, building commercial systems and forging strategic partnerships.
Product Portfolio
Pharmaceuticals
Odyssey has two pharmaceutical products in development:
- PRV-002 is a novel compound for the treatment of concussion, which currently has no FDA-approved drug. In pre-clinical studies, PRV-002 has been shown to significantly improve both neuroscore and memory score following injury in rats subjected to concussion models. Importantly, the first-in-class novel neurosteroid demonstrated no drug-related toxicity in these trials.
- PRV-002 is currently being evaluated in a phase I clinical trial for the treatment of concussion, with phase II trials planned for launch in Fall 2022. Odyssey has also highlighted the potential of PRV-002 for additional indications such as Alzheimer’s disease, Parkinson’s disease, ALS and chromic traumatic encephalopathy (CTE).
- PRV-001 is a novel compound intended to treat Niemann-Pick disease, a rare neurodegenerative-lysosomal storage disorder that affects an estimated 1 in 150,000 individuals in the U.S., demonstrating a 5x higher incidence in Middle Eastern populations.
- Odyssey expects to receive Orphan Drug designation from the FDA for PRV-001, which would accelerate its pathway to FDA approval and provide seven years of market exclusivity.
Medical Devices
Odyssey is also developing two medical device candidates:
- CardioMap® is intended to provide early, non-invasive testing for heart disease. The system offers a number of potential advantages over traditional EKGs, including requiring less training to operate, offering heightened sensitivity and coming in a small and portable form factor. CardioMap is being developed for a 510(k) regulatory pathway, which requires a study to demonstrate equivalence to legacy EKG offerings.
- When approved, CardioMap is expected to be the only device in its class that has a predictive value, illustrating ‘grey’ areas where deterioration has begun but not yet led to pathology. Odyssey expects this feature to provide a powerful incentive for doctors to use the CardioMap device in end markets such as hospitals, doctors’ offices, rehabilitation centers and sports medicine practices.
- Save-A-Life (SAL) is a patented, single-action, instantaneous, handheld, mechanical anti-choking device that creates a vacuum chamber in the mouth to dislodge throat obstructions in a matter of seconds, all without harm to the victim. The device is currently in development, with a proof of concept established.
- Odyssey believes that, once FDA-approved, its anti-choking device will quickly become the “accepted” standard and leader in the treatment of choking incidents globally. Its low-cost manufacturing and convenient portable design give SAL a competitive edge over competing devices utilizing cumbersome masks.
Market Opportunities
Odyssey’s varied development pipeline positions it to address a number of sizable market opportunities with significant unmet medical need. Concussions alone currently account for medical costs of roughly $10-15 billion annually in the U.S., despite the lack of a currently approved FDA drug treatment. This need is particularly apparent in the military and sports industry, where the likelihood of athlete head-injury recurrence is estimated at 75%.
It is for this reason that, in March 2021, Odyssey announced the formation of a sports advisory board featuring well-known athletes supporting the company’s efforts to enhance public awareness of traumatic brain injuries and concussions, as well as the need for an FDA-approved therapy. Members of Odyssey’s sports advisory board include NFL Hall of Famers Kurt Warner & Brett Favre and two-time Olympic gold medalist Abby Wambach.
With its CardioMap platform, Odyssey is targeting the global cardiac monitoring market, which was valued at $28 billion in 2021 by Insight Partners and forecast to reach $43 billion by 2028.
Save-A-Life targets a similarly underserved market. Choking is the fourth-leading cause of death in children, and approximately 5,000 choking deaths occur each year in the U.S. While 95% of these deaths result from in-home incidents, current choking rescue devices fail to address in-home applications.
Management Team
Joseph Michael Redmond is the President, CEO and Chairman of Odyssey. He has over 30 years of commercial experience in medical device companies, previously serving as CEO of Parallax Health Sciences Inc., V.P. of Business Development for DxTech Inc. and V.P. of Sales and Marketing for Bioject Medical Technologies Inc. While at Bioject, Mr. Redmond helped raise over $15 million in capital, entered into several licensing and distribution deals with major biotech and pharmaceutical companies and grew the market cap of the company from under $10 million to over $400 million. He started his career at Abbott Labs and holds a B.A. from Denison University.
Christine M. Farrell is the company’s CFO and Secretary. Prior to joining Odyssey, Ms. Farrell was Vice President of Finance for Bioject Medical Technologies Inc. She also held accounting and financial management positions with Spar-Tek Industries, a manufacturer of high quality and cutting-edge technology for the plywood industry, and Action Machinery, a seller of new and used robotic machine tools and equipment. Ms. Farrell holds a B.A. in Accounting from the University of Washington and an M.B.A. from Willamette University.
Dr. Jacob W. Vanlandingham is Odyssey’s Head of Drug Development. Dr. Vanlandingham holds a Ph.D. in neuroscience with a molecular biology focus. He is a member of the Society for Neuroscience, American Society for Nutritional Sciences, National Neurotrauma Society, Faculty for Undergraduate Research in Neuroscience and the International Association of Medical Science Educators.
Odyssey Group International Inc. (OTC: ODYY), closed Monday's trading session at $0.21, off by 6.6667%, on 248,921 volume. The average volume for the last 3 months is 248,921 and the stock's 52-week low/high is $0.11/$0.64.
Recent News
- Odyssey Group International Inc. (OTC: ODYY) - New Pharmaceutical Subsidiary Geared to Aid Odyssey Health, Inc. (ODYY) Development of Novel Solutions for Brain Related Medical Treatments
- Experts Concerned That Concussion Protocols Don't Factor in Women
- InvestorNewsBreaks - Odyssey Health Inc. (ODYY) Announces Expansion of Executive Team
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Monday's trading session at $0.1901, off by 0.209974%, on 115,854 volume. The average volume for the last 3 months is 112,394 and the stock's 52-week low/high is $0.16/$1.48.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - New Brain Cancer Clinical Trial Platform Launched in Australia
- BioMedNewsBreaks - CNS Pharmaceuticals Inc.'s (NASDAQ: CNSP) Targeted 'Finish Line Is Within Sight' as It Pursues Potential GBM Treatment
- BioMedNewsBreaks - CNS Pharmaceuticals Inc.'s (NASDAQ: CNSP) Berubicin Shows Superior Performance to Other Anthracyclines, Standards of Care for GBM
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Issues Letter to Stockholders
- Aditxt Inc. (NASDAQ: ADTX) - Research Finds That Stem Cells Reduce Lung Transplant Rejection Rates
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - German Plan to Legalize Cannabis Won't Permit Imports
- American Cannabis Partners - 420 with CNW — New Jersey Legislators Approve Bill Offering Cannabis Businesses Limited 280E Tax Workaround
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc.'s (NASDAQ: AMST) AI-Driven Platform Assisting Customers to Thrive and Scale
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Announces Continued Listing Status Granted by NASDAQ Hearing Panel
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Kirby Medical Center to Significantly Reduce Documentation Burden for Physicians and Improve Patient Experiences
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Is 'One to Watch'
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - Blue Hat (BHAT. US) announces the signing of the memorandum of understanding on strategic cooperation with a Singapore Partner
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Appoints Veteran Sales Executive as CRO
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Ideally Positioned as Cybersecurity Industry Surges
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - New Brain Cancer Clinical Trial Platform Launched in Australia
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Buildings are Proving Integral to the Energy Transition, and Correlate Infrastructure Partners Inc. (CIPI) is Looking to solve the Challenge of Retrofitting Them to Meet Net Zero Carbon Goals
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. CEO Spotlighted in Latest Bell2Bell Podcast Release
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. Accelerates Manufacturing on High Demand Despite Supply Chain Challenges
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Announces Participation Schedule for Upcoming Investor Events
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Signs New Employer Contracts to Provide Full Suite of Digital Health Solutions
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Extends Business Value of Industry-First Quantum Hybrid Solver with New Features Supporting Weighted Constraints and Presolve Techniques
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - InvestorNewsBreaks - Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) (FRA: 6X0) Announces Delic Labs' Participation at 11th Annual MJBizCon
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Appoints Veteran Auto Industry Exec to Lead Liteborne Subsidiary
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Research Shows Need to Consider Consequences of Mercury Mitigation Strategies in Artisanal Mining
- MiningNewsBreaks - Eloro Resources Ltd.'s (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Drill Program Unearths Significant Proportions of Precious Metals
- Researchers Find New Way to Use Gold to Measure Whisky's Maturity
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Kazatomprom Plans to Increase Uranium Quota on Trans-Caspian Route
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Named Engine Tech Innovation of Year Winner by AutoTech Breakthrough Awards
- FingerMotion Inc. (NASDAQ: FNGR) - FingerMotion Inc. (NASDAQ: FNGR) Benefitting From Recurring Revenue Streams from the Top-Up Business, Expands its Product Line
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Corporate Restructuring
- Flora Growth Corp. (NASDAQ: FLGC) - Flora Growth Corp. (NASDAQ: FLGC) Agreement to Acquire Multi-national Cannabis Operator Positions Company for New Strategic Growth in Europe
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) to Present at LD Micro Main Event XV
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Multi-Year Commitment with Kimberly Clark de Mexico, S.A.B. de C.V.
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Enters Final Phase of Assembling and Validating First Farm-Ready Demonstration System - Milestone Update: Hydrogen Purity Exceeds Expectations
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Reimagining the Suburbs as Environmentally Green
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Full Acquisition of Interest in RKings Competitions
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Appoints New Global Health Services CEO
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation in Upcoming Investor, Industry Conferences
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces Plans to Report Q3 2022 Financial Results
- HeartBeam Inc. (NASDAQ: BEAT) - Study Finds Smart Watches Can Accurately Detect Atrial Fibrillation
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Technologies Inc. (NASDAQ: CPTN) Partner with Leading Entities to Complete Project Resulting in Enhanced Road Safety
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) CEO Talks Future of 'Recession-Resistant' Cannabis Market
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd.'s (CSE: HEAT) (OTCQB: HLRTF) Enhanced Solution Holds Potential to Simplify, Redefine EV Charging
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Partners with 'Chopped' Champion and Celebrity Chef Katsuji Tanabe
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Change in ADS Ratio
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Q3 2022 Earnings Call Details, Strategic Focus on the EV Market
- India Globalization Capital Inc. (NYSE American: IGC) - India Globalization Capital Inc. (NYSE American: IGC) Is 'One to Watch'
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Receives Notification of Minimum-Bid Price Requirement Compliance
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Support of EB Awareness Week
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces New OTC Hearing Aids to Expand iHEAR Brand
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope Inc. (NASDAQ: KSCP) Announces New Contracts, Indicative of Company's Continued Reach
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces Zero Adverse Effects from Successful HYPER-H21-4 Study of DehydraTECH(TM)-CBD for High Blood Pressure
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Names New Auditing Company
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Committed to Enhancing Blockchain Micropayment Transactions on Lightning Network
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Committed to Facilitating BTC Transactions Worldwide via Lightning Network
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the First Quarter of Fiscal 2023
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Closing Under Share Subscription Agreement, Appointment of New CFO
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) to Be Featured on the RedChip Money Report(R)
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Eliminates Some $13M in Debt
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) (FSE: N29), CEO Acquire Arway Shares
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - New Pharmaceutical Subsidiary Geared to Aid Odyssey Health, Inc. (ODYY) Development of Novel Solutions for Brain Related Medical Treatments
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Finalizes Acquisition of IP, Assets and Online Domain
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Announces Launch of VegasLounge with Markor Technology
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Update on Status of MCTO
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Appoints Key Life Sciences Thought Leader to Its Board
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO), Dr. Denese SkinScience to Introduce UST Nanoemulsion Hair Regrowth Product in 2023
- Prime Harvest Inc. - 420 with CNW — U.S. Federal Court Rules Against Border Patrol Attempts to Prevent Cannabis Equipment Imports
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Host Conference Call to Discuss Q3 2022 Results, Provide Pipeline Update
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Epigenetics May Revolutionize Treatment of Cancer
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Issues Expansion Update
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - ESGBreaks - Reklaim Ltd.'s (TSX.V: MYID) (OTCQB: MYIDF) Revolutionary System Enables Consumers to Take Control of Their Data
- REZYFi, Inc. - 420 with CNW — Debt Financing Outstrips Equity in the US Cannabis Industry
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Partners with Sapphire Records and Multi-Platinum Artist David Veslocki
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Hosts Delegation from Leading Multinational Food and Agricultural Companies
- Sharing Services Global Corporation (SHRG) - Sharing Services Global Corp. (SHRG) Guided by Vision, Mission, Values as It Reshapes How Entrepreneurs Succeed
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Launching New Solution to Help Drive Standards-Based Approach to Additive Manufacturing
- Silo Pharma Inc. (OTCQB: SILO) - New Psychedelic Journal to Focus on Publishing Psychedelic Research
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Expands Sales Team to Add Telecom Industry Veteran
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Participation in Upcoming Sidoti Microcap Conference
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - VERSES to Participate at the LD Micro Main Event XV
- Standard Lithium Ltd. (NYSE American: SLI) - Standard Lithium to Participate in PI Financial's The Future is Lithium Conference
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street / Newsmax TV Announces Episode #403, Eight Interviews, Airing November 6, 2022, 10-11 AM ET
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Signs Distribution Deal, Now Available in Austin
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaline88(R) 2 Liter, Individual or Shaq Paq, Eyes Growing Success
- Tingo Inc. (OTC: TMNA) - Binance Announces $500m in Loans to Cryptocurrency Miners
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Notes WIPO Publication of International PCT Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc.'s (NASDAQ: MEDS) Bonum Health(TM) to Work with Insurance Brokers to Offer Telemedicine Services for SMB Employer Groups
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Announces Acquisition of Rio Tinto's Roughrider Uranium Project
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) to Host Q2 2023 Financial Results Call, Webcast
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. to Report Third Quarter 2022 Financial Results on Wednesday, November 9, 2022 at 5:00 p.m. ET
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - InvestorNewsBreaks - XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) Announces Major Milestone on Path to Commercialization of Lead Product
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Issues Letter to Stockholders
- Aditxt Inc. (NASDAQ: ADTX) - Research Finds That Stem Cells Reduce Lung Transplant Rejection Rates
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - German Plan to Legalize Cannabis Won't Permit Imports
- American Cannabis Partners - 420 with CNW — New Jersey Legislators Approve Bill Offering Cannabis Businesses Limited 280E Tax Workaround
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc.'s (NASDAQ: AMST) AI-Driven Platform Assisting Customers to Thrive and Scale
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Announces Continued Listing Status Granted by NASDAQ Hearing Panel
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Kirby Medical Center to Significantly Reduce Documentation Burden for Physicians and Improve Patient Experiences
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Is 'One to Watch'
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - Blue Hat (BHAT. US) announces the signing of the memorandum of understanding on strategic cooperation with a Singapore Partner
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Appoints Veteran Sales Executive as CRO
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Ideally Positioned as Cybersecurity Industry Surges
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - New Brain Cancer Clinical Trial Platform Launched in Australia
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Buildings are Proving Integral to the Energy Transition, and Correlate Infrastructure Partners Inc. (CIPI) is Looking to solve the Challenge of Retrofitting Them to Meet Net Zero Carbon Goals
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. CEO Spotlighted in Latest Bell2Bell Podcast Release
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. Accelerates Manufacturing on High Demand Despite Supply Chain Challenges
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Announces Participation Schedule for Upcoming Investor Events
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Signs New Employer Contracts to Provide Full Suite of Digital Health Solutions
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Extends Business Value of Industry-First Quantum Hybrid Solver with New Features Supporting Weighted Constraints and Presolve Techniques
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - InvestorNewsBreaks - Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) (FRA: 6X0) Announces Delic Labs' Participation at 11th Annual MJBizCon
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Appoints Veteran Auto Industry Exec to Lead Liteborne Subsidiary
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Research Shows Need to Consider Consequences of Mercury Mitigation Strategies in Artisanal Mining
- MiningNewsBreaks - Eloro Resources Ltd.'s (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Drill Program Unearths Significant Proportions of Precious Metals
- Researchers Find New Way to Use Gold to Measure Whisky's Maturity
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Kazatomprom Plans to Increase Uranium Quota on Trans-Caspian Route
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Named Engine Tech Innovation of Year Winner by AutoTech Breakthrough Awards
- FingerMotion Inc. (NASDAQ: FNGR) - FingerMotion Inc. (NASDAQ: FNGR) Benefitting From Recurring Revenue Streams from the Top-Up Business, Expands its Product Line
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Corporate Restructuring
- Flora Growth Corp. (NASDAQ: FLGC) - Flora Growth Corp. (NASDAQ: FLGC) Agreement to Acquire Multi-national Cannabis Operator Positions Company for New Strategic Growth in Europe
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) to Present at LD Micro Main Event XV
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Multi-Year Commitment with Kimberly Clark de Mexico, S.A.B. de C.V.
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Enters Final Phase of Assembling and Validating First Farm-Ready Demonstration System - Milestone Update: Hydrogen Purity Exceeds Expectations
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Reimagining the Suburbs as Environmentally Green
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Full Acquisition of Interest in RKings Competitions
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Appoints New Global Health Services CEO
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation in Upcoming Investor, Industry Conferences
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces Plans to Report Q3 2022 Financial Results
- HeartBeam Inc. (NASDAQ: BEAT) - Study Finds Smart Watches Can Accurately Detect Atrial Fibrillation
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Technologies Inc. (NASDAQ: CPTN) Partner with Leading Entities to Complete Project Resulting in Enhanced Road Safety
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) CEO Talks Future of 'Recession-Resistant' Cannabis Market
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd.'s (CSE: HEAT) (OTCQB: HLRTF) Enhanced Solution Holds Potential to Simplify, Redefine EV Charging
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Partners with 'Chopped' Champion and Celebrity Chef Katsuji Tanabe
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Change in ADS Ratio
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Q3 2022 Earnings Call Details, Strategic Focus on the EV Market
- India Globalization Capital Inc. (NYSE American: IGC) - India Globalization Capital Inc. (NYSE American: IGC) Is 'One to Watch'
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Receives Notification of Minimum-Bid Price Requirement Compliance
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Support of EB Awareness Week
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces New OTC Hearing Aids to Expand iHEAR Brand
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope Inc. (NASDAQ: KSCP) Announces New Contracts, Indicative of Company's Continued Reach
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces Zero Adverse Effects from Successful HYPER-H21-4 Study of DehydraTECH(TM)-CBD for High Blood Pressure
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Names New Auditing Company
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Committed to Enhancing Blockchain Micropayment Transactions on Lightning Network
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Committed to Facilitating BTC Transactions Worldwide via Lightning Network
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the First Quarter of Fiscal 2023
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Closing Under Share Subscription Agreement, Appointment of New CFO
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) to Be Featured on the RedChip Money Report(R)
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Eliminates Some $13M in Debt
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) (FSE: N29), CEO Acquire Arway Shares
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - New Pharmaceutical Subsidiary Geared to Aid Odyssey Health, Inc. (ODYY) Development of Novel Solutions for Brain Related Medical Treatments
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Finalizes Acquisition of IP, Assets and Online Domain
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Announces Launch of VegasLounge with Markor Technology
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Update on Status of MCTO
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Appoints Key Life Sciences Thought Leader to Its Board
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO), Dr. Denese SkinScience to Introduce UST Nanoemulsion Hair Regrowth Product in 2023
- Prime Harvest Inc. - 420 with CNW — U.S. Federal Court Rules Against Border Patrol Attempts to Prevent Cannabis Equipment Imports
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Host Conference Call to Discuss Q3 2022 Results, Provide Pipeline Update
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Epigenetics May Revolutionize Treatment of Cancer
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Issues Expansion Update
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - ESGBreaks - Reklaim Ltd.'s (TSX.V: MYID) (OTCQB: MYIDF) Revolutionary System Enables Consumers to Take Control of Their Data
- REZYFi, Inc. - 420 with CNW — Debt Financing Outstrips Equity in the US Cannabis Industry
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Partners with Sapphire Records and Multi-Platinum Artist David Veslocki
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Hosts Delegation from Leading Multinational Food and Agricultural Companies
- Sharing Services Global Corporation (SHRG) - Sharing Services Global Corp. (SHRG) Guided by Vision, Mission, Values as It Reshapes How Entrepreneurs Succeed
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Launching New Solution to Help Drive Standards-Based Approach to Additive Manufacturing
- Silo Pharma Inc. (OTCQB: SILO) - New Psychedelic Journal to Focus on Publishing Psychedelic Research
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Expands Sales Team to Add Telecom Industry Veteran
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Participation in Upcoming Sidoti Microcap Conference
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - VERSES to Participate at the LD Micro Main Event XV
- Standard Lithium Ltd. (NYSE American: SLI) - Standard Lithium to Participate in PI Financial's The Future is Lithium Conference
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street / Newsmax TV Announces Episode #403, Eight Interviews, Airing November 6, 2022, 10-11 AM ET
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Signs Distribution Deal, Now Available in Austin
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaline88(R) 2 Liter, Individual or Shaq Paq, Eyes Growing Success
- Tingo Inc. (OTC: TMNA) - Binance Announces $500m in Loans to Cryptocurrency Miners
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Notes WIPO Publication of International PCT Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc.'s (NASDAQ: MEDS) Bonum Health(TM) to Work with Insurance Brokers to Offer Telemedicine Services for SMB Employer Groups
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Announces Acquisition of Rio Tinto's Roughrider Uranium Project
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) to Host Q2 2023 Financial Results Call, Webcast
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. to Report Third Quarter 2022 Financial Results on Wednesday, November 9, 2022 at 5:00 p.m. ET
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - InvestorNewsBreaks - XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) Announces Major Milestone on Path to Commercialization of Lead Product
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Issues Letter to Stockholders
- Aditxt Inc. (NASDAQ: ADTX) - Research Finds That Stem Cells Reduce Lung Transplant Rejection Rates
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - German Plan to Legalize Cannabis Won't Permit Imports
- American Cannabis Partners - 420 with CNW — New Jersey Legislators Approve Bill Offering Cannabis Businesses Limited 280E Tax Workaround
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc.'s (NASDAQ: AMST) AI-Driven Platform Assisting Customers to Thrive and Scale
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Announces Continued Listing Status Granted by NASDAQ Hearing Panel
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Partners with Kirby Medical Center to Significantly Reduce Documentation Burden for Physicians and Improve Patient Experiences
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Is 'One to Watch'
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - Blue Hat (BHAT. US) announces the signing of the memorandum of understanding on strategic cooperation with a Singapore Partner
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Appoints Veteran Sales Executive as CRO
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Ideally Positioned as Cybersecurity Industry Surges
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - New Brain Cancer Clinical Trial Platform Launched in Australia
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Buildings are Proving Integral to the Energy Transition, and Correlate Infrastructure Partners Inc. (CIPI) is Looking to solve the Challenge of Retrofitting Them to Meet Net Zero Carbon Goals
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. CEO Spotlighted in Latest Bell2Bell Podcast Release
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. Accelerates Manufacturing on High Demand Despite Supply Chain Challenges
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Announces Participation Schedule for Upcoming Investor Events
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Signs New Employer Contracts to Provide Full Suite of Digital Health Solutions
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Extends Business Value of Industry-First Quantum Hybrid Solver with New Features Supporting Weighted Constraints and Presolve Techniques
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - InvestorNewsBreaks - Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) (FRA: 6X0) Announces Delic Labs' Participation at 11th Annual MJBizCon
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Appoints Veteran Auto Industry Exec to Lead Liteborne Subsidiary
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Research Shows Need to Consider Consequences of Mercury Mitigation Strategies in Artisanal Mining
- MiningNewsBreaks - Eloro Resources Ltd.'s (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Drill Program Unearths Significant Proportions of Precious Metals
- Researchers Find New Way to Use Gold to Measure Whisky's Maturity
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Kazatomprom Plans to Increase Uranium Quota on Trans-Caspian Route
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Named Engine Tech Innovation of Year Winner by AutoTech Breakthrough Awards
- FingerMotion Inc. (NASDAQ: FNGR) - FingerMotion Inc. (NASDAQ: FNGR) Benefitting From Recurring Revenue Streams from the Top-Up Business, Expands its Product Line
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Announces Corporate Restructuring
- Flora Growth Corp. (NASDAQ: FLGC) - Flora Growth Corp. (NASDAQ: FLGC) Agreement to Acquire Multi-national Cannabis Operator Positions Company for New Strategic Growth in Europe
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) to Present at LD Micro Main Event XV
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Announces Multi-Year Commitment with Kimberly Clark de Mexico, S.A.B. de C.V.
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Enters Final Phase of Assembling and Validating First Farm-Ready Demonstration System - Milestone Update: Hydrogen Purity Exceeds Expectations
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Reimagining the Suburbs as Environmentally Green
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Full Acquisition of Interest in RKings Competitions
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Appoints New Global Health Services CEO
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Announces Participation in Upcoming Investor, Industry Conferences
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Announces Plans to Report Q3 2022 Financial Results
- HeartBeam Inc. (NASDAQ: BEAT) - Study Finds Smart Watches Can Accurately Detect Atrial Fibrillation
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Technologies Inc. (NASDAQ: CPTN) Partner with Leading Entities to Complete Project Resulting in Enhanced Road Safety
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) CEO Talks Future of 'Recession-Resistant' Cannabis Market
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd.'s (CSE: HEAT) (OTCQB: HLRTF) Enhanced Solution Holds Potential to Simplify, Redefine EV Charging
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Partners with 'Chopped' Champion and Celebrity Chef Katsuji Tanabe
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Change in ADS Ratio
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Q3 2022 Earnings Call Details, Strategic Focus on the EV Market
- India Globalization Capital Inc. (NYSE American: IGC) - India Globalization Capital Inc. (NYSE American: IGC) Is 'One to Watch'
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Receives Notification of Minimum-Bid Price Requirement Compliance
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Support of EB Awareness Week
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces New OTC Hearing Aids to Expand iHEAR Brand
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope Inc. (NASDAQ: KSCP) Announces New Contracts, Indicative of Company's Continued Reach
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces Zero Adverse Effects from Successful HYPER-H21-4 Study of DehydraTECH(TM)-CBD for High Blood Pressure
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Names New Auditing Company
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Committed to Enhancing Blockchain Micropayment Transactions on Lightning Network
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Committed to Facilitating BTC Transactions Worldwide via Lightning Network
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the First Quarter of Fiscal 2023
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Closing Under Share Subscription Agreement, Appointment of New CFO
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) to Be Featured on the RedChip Money Report(R)
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Eliminates Some $13M in Debt
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) (FSE: N29), CEO Acquire Arway Shares
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - New Pharmaceutical Subsidiary Geared to Aid Odyssey Health, Inc. (ODYY) Development of Novel Solutions for Brain Related Medical Treatments
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Finalizes Acquisition of IP, Assets and Online Domain
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Announces Launch of VegasLounge with Markor Technology
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Update on Status of MCTO
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Appoints Key Life Sciences Thought Leader to Its Board
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO), Dr. Denese SkinScience to Introduce UST Nanoemulsion Hair Regrowth Product in 2023
- Prime Harvest Inc. - 420 with CNW — U.S. Federal Court Rules Against Border Patrol Attempts to Prevent Cannabis Equipment Imports
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Host Conference Call to Discuss Q3 2022 Results, Provide Pipeline Update
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Epigenetics May Revolutionize Treatment of Cancer
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Issues Expansion Update
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - ESGBreaks - Reklaim Ltd.'s (TSX.V: MYID) (OTCQB: MYIDF) Revolutionary System Enables Consumers to Take Control of Their Data
- REZYFi, Inc. - 420 with CNW — Debt Financing Outstrips Equity in the US Cannabis Industry
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Partners with Sapphire Records and Multi-Platinum Artist David Veslocki
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Hosts Delegation from Leading Multinational Food and Agricultural Companies
- Sharing Services Global Corporation (SHRG) - Sharing Services Global Corp. (SHRG) Guided by Vision, Mission, Values as It Reshapes How Entrepreneurs Succeed
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Launching New Solution to Help Drive Standards-Based Approach to Additive Manufacturing
- Silo Pharma Inc. (OTCQB: SILO) - New Psychedelic Journal to Focus on Publishing Psychedelic Research
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Expands Sales Team to Add Telecom Industry Veteran
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Announces Participation in Upcoming Sidoti Microcap Conference
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - VERSES to Participate at the LD Micro Main Event XV
- Standard Lithium Ltd. (NYSE American: SLI) - Standard Lithium to Participate in PI Financial's The Future is Lithium Conference
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street / Newsmax TV Announces Episode #403, Eight Interviews, Airing November 6, 2022, 10-11 AM ET
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Signs Distribution Deal, Now Available in Austin
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaline88(R) 2 Liter, Individual or Shaq Paq, Eyes Growing Success
- Tingo Inc. (OTC: TMNA) - Binance Announces $500m in Loans to Cryptocurrency Miners
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Notes WIPO Publication of International PCT Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc.'s (NASDAQ: MEDS) Bonum Health(TM) to Work with Insurance Brokers to Offer Telemedicine Services for SMB Employer Groups
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Announces Acquisition of Rio Tinto's Roughrider Uranium Project
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) to Host Q2 2023 Financial Results Call, Webcast
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies, Inc. to Report Third Quarter 2022 Financial Results on Wednesday, November 9, 2022 at 5:00 p.m. ET
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - InvestorNewsBreaks - XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) Announces Major Milestone on Path to Commercialization of Lead Product
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































