The QualityStocks Daily Stock List
- Aprea Therapeutics (APRE)
- Blackboxstocks Inc. (BLBX)
- Synaptogenix (SNPX)
- Chico's FAS (CHS)
- FaZe Holdings (FAZE)
- Li-Cycle Holdings (LICY)
- Prenetics Global (PRE)
- Nutriband Inc. (NTRB)
- Marathon Digital Holdings Inc. (MARA)
- Alliance Resource Partners L.P. (ARLP)
- Compass Pathways PLC. (CMPS)
- ElectraMeccanica Vehicles Corp. Ltd. (SOLO)
Aprea Therapeutics (APRE)
MyBestStockAlerts, PennyStocks24, MarketClub Analysis, Actual Gains, The Stock Psycho, MarketBeat, Top Gun, QualityStocks, Stock Roach, Hot Stock Profits, Mina Mar Marketing Group, PennyPickAlerts, Honest Abe, Trades Of The Day, Penny Picks, Penny Stock Newsletter, Penny Stocks Finder, InvestorSoup, PennyStockRumors.net, OtcShortReport, Fortune Stock Alerts, Fast Moving Stocks, Damn Good Penny Picks, Stock Preacher, BUYINS.NET, Beacon Equity Research, Greenbackers, FreeRealTime, FeedBlitz, Club Penny Stocks Network, Center Stage Stocks, HotStockProfits, Market Wrap Daily, MarketWireStocks, MicroStockProfit, OTC Stock Review, PennyStockProfessor, Pennystocktweeters.com, PREPUMP STOCKS, Real Pennies, RockingPennyStocks, Stock Legends, StockHideout, Stocktamer, TooNiceStocks, TopPennyStockMovers and Orbit Stocks reported earlier on Aprea Therapeutics (APRE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Aprea Therapeutics Inc. (NASDAQ: APRE) is a biopharmaceutical firm that is focused on the development and commercialization of cancer therapies that reactivate the mutant p53 tumor suppressor protein.
The firm has its headquarters in Boston, Massachusetts and was incorporated in 2006. It operates as part of the healthcare sector, in the biotech and pharma sub-industry and serves consumers in Sweden and the U.S.
The company’s primary shareholders include KCIF Co-Investment Fund KB, Rosetta Capital Ltd. and an investment vehicle owned by Karolinska Development AB called KDev Investments AB.
The enterprise’s p53 tumor suppressor protein responds to different forms of cellular stress and activates the transcription of genes which cause apoptosis or cell cycle arrest. Its product portfolio comprises of a p53 reactivator dubbed APR-548, which is undergoing a phase 1 dose-escalation trial for oral administration in patients suffering from myelodysplastic syndromes; and a p53 small molecule reactivator dubbed Eprenetapopt (APR-246), which is currently in late-stage clinical development for various hematologic malignancies as well as other indications. They include acute myeloid leukemia and myelodysplastic syndromes, as well as refractory/relapsed TP53 mutant chronic lymphoid leukemia, non-small cell lung cancer, bladder cancer and gastric cancer. In addition to this, the enterprise is also involved in the development of APR-017, which causes p53-mediated apoptosis by restoring p53 activity in mutant p53 cancer cell lines.
The company recently announced positive results from its trial which evaluated eprenetapopt with azacitidine in the treatment of patients with TP53 mutant acute myeloid leukemia and myelodysplastic syndromes. The success of this new treatment will not only improve patient outcomes for these diseases, which currently have limited treatment options, but also boost investments into the firm, which will have a positive impact on the company’s growth.
Aprea Therapeutics (APRE), closed Thursday's trading session at $0.5145, up 30.5506%, on 418,539 volume. The average volume for the last 3 months is 359,611 and the stock's 52-week low/high is $0.304/$3.0824.
Blackboxstocks Inc. (BLBX)
MarketClub Analysis, QualityStocks, StockOnion, Profitable Trader Authority, PennyStockScholar, PennyStockProphet, Penny Pick Finders, PCG Advisory, OTCtipReporter and MarketBeat reported earlier on Blackboxstocks Inc. (BLBX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Blackboxstocks Inc. (NASDAQ: BLBX) is a development-stage firm that is engaged in the development and marketing of its financial technology and social media hybrid platform.
The firm has its headquarters in Dallas, Texas and was incorporated in 2011, on October 4th. Prior to its name change, the firm was known as SMSA Ballinger Acquisition Corp. It mainly serves consumers in the United States.
The company is a software-as-a-service firm with a growing base of users that spans over forty countries. Its web-based software uses predictive technology enhanced by AI to find unusual market activity and volatility that may cause a change in the price of options or stocks.
The enterprise provides users with a fully interactive social media platform that’s integrated into its dashboard, allowing users to exchange ideas and information efficiently and quickly via a common network. It also offers a live video/audio feature which enables members to broadcast on their own channels to share market insight and trading strategies within the Blackbox community. It serves as a tool for swing traders and day traders on the OTC markets, AMEX, NASDAQ and NYSE markets. The enterprise’s system monitors and analyzes more than 13,000 stocks on these markets simultaneously as its servers receive live feeds from the aforementioned markets.
The firm recently announced its latest financial results which show significant increases in its revenues. In addition to this, it crossed the threshold of 6,000 active paid subscribers. It is now focused on fueling product development, aggressive advertising and marketing with its goal being to drive subscriber growth. This may help extend its consumer reach as well as bring in more investors into the firm.
Blackboxstocks Inc. (BLBX), closed Thursday's trading session at $0.450001, up 29.4967%, on 363,353 volume. The average volume for the last 3 months is 78,548 and the stock's 52-week low/high is $0.2605/$5.50.
Synaptogenix (SNPX)
TradersPro and MarketClub Analysis reported earlier on Synaptogenix (SNPX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Synaptogenix Inc. (NASDAQ: SNPX) is a biopharmaceutical firm that is focused on the development of therapeutics for patients with neurodegenerative illnesses and developmental disorders.
The firm has its headquarters in New York and was incorporated in 2012, on October 31st. Prior to its name change, the firm was known as Neurotrop BioScience Inc. It operates as part of the biotechnology industry, under the healthcare sector. The firm serves consumers around the globe.
The company’s mission is to discover restorative, novel therapies for life-altering illnesses. It is led by an experienced team with deep expertise in neurodegenerative disorders and successful track records in both drug discovery and development. The company is party to licensing agreements with Icahn School of Medicine at Mount Sinai; Stanford University; and The Board of Trustees of the Leland Stanford Junior University.
The company is primarily focused on the development of a product platform based on a drug candidate dubbed Bryostatin-1, which has been designed to treat Alzheimer's disease. It has observed that this candidate regenerates synaptic networks in the brain, which may improve cognitive performance. It is also evaluating therapeutic applications of bryostatin for other neurodegenerative or cognitive diseases and dysfunctions, such as Multiple sclerosis, Fragile X syndrome, Parkinson’s disease, Stroke, Autism spectrum disorders, Traumatic Brain injury and Niemann-pick type C disease.
The firm remains focused on developing a new treatment option for Alzheimer’s disease. The success and approval of its formulation will not only benefit the millions with Alzheimer’s but also bolster the firm’s overall growth.
Synaptogenix (SNPX), closed Thursday's trading session at $1.21, up 1.6807%, on 88,661 volume. The average volume for the last 3 months is 1.973M and the stock's 52-week low/high is $0.96/$10.65.
Chico's FAS (CHS)
The Online Investor, StocksEarning, MarketClub Analysis, StreetInsider, The Street, Zacks, Forbes, MarketBeat, InvestorPlace, Barchart, Marketbeat.com, Schaeffer's, Wall Street Greek, Hit and Run Candle Sticks, StockEarnings, The Wealth Report, InvestorsObserver Team, SmarTrend Newsletters, Top Pros' Top Picks, Street Insider, TradersPro, Trading Concepts, Louis Navellier, Cabot Wealth, Kiplinger Today, Money Morning, StockMarketWatch, StreetAuthority Daily, TopStockAnalysts, Trading Markets, Daily Trade Alert, Daily Markets, BUYINS.NET, Stockhouse, FNNO Newsletters, Dividend Opportunities, Profit Confidential, Investing Daily, Daily Wealth, Investment House, Investor Guide, InvestorGuide, Market FN, Market Wrap Daily, Navellier Growth, One Hot Stock, OTC Stock Pick, Shah's Insights & Indictments, Stock Gumshoe, The Growth Stock Wire, TradingMarkets, Wealth Insider Alert, WStreet Market Commentary, Wyatt Investment Research and Options Elite reported earlier on Chico's FAS (CHS), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Chico's FAS, Inc. (NYSE: CHS) (FRA: YCB) (BMV: CHS) is a company involved in the retail of women’s private branded casual-to-dressy clothing, intimates and complementary accessories.
The firm has its headquarters in Fort Myers, Florida and was incorporated in 1983 by Marvin L. Gralnick and Helene B. Gralnick. It operates as part of the apparel retail industry, under the consumer cyclical sector. The firm serves consumers in the United States.
The enterprise’s product portfolio consists of the following brands: Chico's, White House Black Market (WHBM) and Soma. The Chico's brand is primarily involved in the sale of private branded clothing that focuses on women with moderate to high household income levels. The WHBM brand is focused on the sale of everyday basics and denim, relaxed workwear, polished casual apparel, feminine all-occasion dresses, black and white pieces, shoes, scarves, belts, handbags, and jewelry for women. On the other hand, the Soma brand focuses on selling private branded lingerie, loungewear and sleepwear products. As of January 2022, the enterprise operated 1,266 stores in the United States, the United States Virgin Islands and Puerto Rico; and 59 international franchise locations in Mexico and 2 domestic airport locations. It sells its products through catalogs and retail stores, as well as through chicos.com, whbm.com, soma.com and chicosofftherack.com. This is in addition to selling them through third party channels.
The company recently announced its latest financial results, with its CEO noting that they remained focused on making progress on long-term goals and creating shareholder value.
Chico's FAS (CHS), closed Thursday's trading session at $4.88, up 0.618557%, on 1,990,722 volume. The average volume for the last 3 months is 15,612 and the stock's 52-week low/high is $3.80/$7.305.
FaZe Holdings (FAZE)
Schaeffer's and The Street reported earlier on FaZe Holdings (FAZE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
FaZe Holdings Inc. (NASDAQ: FAZE) is a holding firm that operates a lifestyle and media platform in gaming and youth culture.
The firm has its headquarters in New York, the United States and was incorporated in 2010. Prior to its name change, the firm was known as B Riley Principal 150 Merger Corp. It operates as part of the internet content and information industry, under the communication services sector. The firm serves consumers around the globe.
The company operates through its subsidiaries, providing a digital-native lifestyle and media platform rooted in gaming and youth culture, re-imagining traditional entertainment for the next generation.
The enterprise produces content, designs merchandise and consumer products. Its content development is fueled by creators, social media influencers, and businesses which are comprised of Esports professionals and celebrities, content creators, athletes, and musician collaborators. It also creates advertising and sponsorship programs for brands, reaching roughly half a billion fans across different social platforms. This is in addition to delivering entertainment that spans video blogs, lifestyle and branded content, gaming highlights, and live streams of competitive gaming tournaments. Its gaming division includes 11 competitive e-sports teams while its e-sports division includes 9 competitive teams in Fortnite, PUBG, federation internationale de football association (FIFA), PUBG Mobile, Rocket League, Rainbow Six, VALORANT, Call of Duty League (Atlanta FaZe) and CS:GO.
The firm, whose latest financial results show increases in its revenues, remains focused on entering into new business opportunities and solidifying its foundation for growth. This will not only generate shareholder value but also bolster the firm’s overall growth.
FaZe Holdings (FAZE), closed Thursday's trading session at $1.76, up 0.571429%, on 15,618 volume. The average volume for the last 3 months is 557,839 and the stock's 52-week low/high is $1.45/$24.69.
Li-Cycle Holdings (LICY)
MarketBeat, MarketClub Analysis, StocksEarning, The Street and Schaeffer's reported earlier on Li-Cycle Holdings (LICY), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Li-Cycle Holdings Corp (NYSE: LICY) (BMV: LICYN) is a holding firm that is engaged in the provision of lithium-ion battery recycling and resource recovery solutions.
The firm has its headquarters in Toronto, Ontario and was incorporated in 2016 by Timothy Johnston and Ajay Kochhar. It operates as part of the waste management industry, under the industrials sector. The firm serves consumers in North America.
The company is on a mission to provide sustainable and safe customer-centric solutions for end-of-lifecycle lithium-ion batteries while meeting the rapidly growing demand for critical batteries.
The enterprise offers customer-centric solutions for each of its customers’ battery recycling needs. It provides its customers a home for the secure destruction of materials containing Internet Protocol (IP)-sensitive design information, such as research and development (R&D) batteries and battery materials. Its Spoke & Hub recycling and resource recovery process is designed at its Spokes, to process battery manufacturing scrap and end-of-life batteries to produce black mass and other intermediate products, and at its Hubs, to process black mass to produce critical battery materials, including nickel sulfate, cobalt sulfate, and lithium carbonate. These technologies use a combination of mechanical safe size reduction and hydrometallurgical resource recovery specifically designed for Li-ion battery recycling.
The firm recently launched a new battery recycling facility in Alabama. This move not only extends the firm’s reach in the U.S. and supports the recycling needs of its growing battery supply consumer base but will also open it up to new growth and investment opportunities.
Li-Cycle Holdings (LICY), closed Thursday's trading session at $4.93, up 1.4403%, on 575,407 volume. The average volume for the last 3 months is 33,002 and the stock's 52-week low/high is $4.48/$10.15.
Prenetics Global (PRE)
StreetInsider, Zacks, Super Stock Picker, Barchart, SmarTrend Newsletters, Daily Markets, INO.com Market Report, AllPennyStocks, Money Morning, The Street, Street Insider, BUYINS.NET, Coattail Investor, Investopedia, InvestorPlace, Money and Markets, Stock Tips Network, The Online Investor, TopStockAnalysts and Marketbeat.com reported earlier on Prenetics Global (PRE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Prenetics Global Limited (NASDAQ: PRE) is an investment holding firm that operates as a diagnostics and genetic testing firm offering preventive and diagnostic health testing and services.
The firm has its headquarters in Quarry Bay, Hong Kong and was incorporated in 2014 by Chi Hung Tzang and Sheng Wu Yeung. It operates as part of the diagnostics and research industry, under the healthcare sector. The firm serves patients around the globe.
The company is focused on bringing health closer to millions of individuals globally and decentralizing healthcare by making the three pillars of Prevention, Diagnostics and Personalized Care, comprehensive and accessible to anyone, anytime and anywhere. It has operations across 9 locations, including the United Kingdom, India, Hong Kong, Asia and South Africa.
The enterprise offers a rapid molecular detection system for a range of infectious diseases. Its products include a consumer genetic testing product known as CircleDNA; a non-invasive FIT-DNA colorectal cancer screening test dubbed ColoClear; and a rapid detection health monitoring system known as Circle HealthPod, which allows users to take COVID-19 tests at point-of-care or at home utilizing the nucleic acid amplification test. It also offers Circle SnapShot, an off-the-shelf at-home blood test; Circle One; Circle Medical, a diagnostic testing product; and F1x/Fem.
The company recently acquired a genomics firm known as ACT Genomics that specializes in precision oncology. This move strengthens its market share in the space and allows the company to better meet patient needs while also generating value for its shareholders.
Prenetics Global (PRE), closed Thursday's trading session at $1.9, even for the day, on 33,100 volume. The average volume for the last 3 months is 1,037 and the stock's 52-week low/high is $1.24/$8.75.
Nutriband Inc. (NTRB)
QualityStocks, StocksEarning, RedChip, Schaeffer's and The Online Investor reported earlier on Nutriband Inc. (NTRB), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Nutriband (NASDAQ: NTRB), a pioneer in the science of using novel transdermal solutions and other innovative technologies, has announced that it has commenced trading on Upstream, a MERJ Exchange Market that is a fully regulated global stock exchange for digital securities and NFTs. The company, which began trading on Upstream today using the NTRB ticker symbol, is among the first issuers to dual list on the market. The company released a digital collectible NFT commemorating the dual listing, which is available for all Upstream participants to claim with a claim code: NTRB! “We are thrilled to be amongst the first companies to dual list on Upstream's next generation marketplace," said Nutriband CEO Gareth Sheridan in the press release. “We are adding new value to our shareholders while expanding our company and mission to a modern, global investor-base.”
To view the full press release, visit https://ibn.fm/9tMVN
About Nutriband Inc.
Nutriband is primarily engaged in the development of a portfolio of transdermal pharmaceutical products. The company’s lead product under development is an abuse deterrent fentanyl patch incorporating its AVERSA(TM) abuse deterrence technology. AVERSA technology can be incorporated into any transdermal patch to prevent the abuse, misuse, diversion and accidental exposure of drugs with abuse potential. For more information about the company, please visit www.Nutriband.com.
Nutriband Inc. (NTRB), closed Thursday's trading session at $3.615, up 1.5449%, on 1,037 volume. The average volume for the last 3 months is 24.692M and the stock's 52-week low/high is $2.408572/$7.542858.
Marathon Digital Holdings Inc. (MARA)
InvestorPlace, MarketClub Analysis, Schaeffer's, QualityStocks, StockMarketWatch, MarketBeat, TradersPro, StocksEarning, Lebed.biz, The Online Investor, BUYINS.NET, Trades Of The Day, The Street, Marketbeat.com, TraderPower, Daily Trade Alert, Wall Street Mover, PoliticsAndMyPortfolio, TopPennyStockMovers, INO Market Report, FeedBlitz, Kiplinger Today, Wealth Insider Alert, StreetAuthority Daily, Barchart, DreamTeamNetwork, InvestorsUnderground, RedChip, AllPennyStocks, Stock Analyzer, Stock Beast, StockOodles, Street Insider, StreetInsider and Promotion Stock Secrets reported earlier on Marathon Digital Holdings Inc. (MARA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Marathon Digital Holdings (NASDAQ: MARA), a leader in supporting and securing the bitcoin ecosystem, today released unaudited bitcoin (“BTC”) production and miner installation updates for December 2022. “In 2022, we made significant progress in increasing our hash rate while also transitioning to more sustainable power sources,” said Fred Thiel, Marathon’s chairman and CEO. “Despite challenges earlier in the year, we doubled our hash rate to 7.0 exahashes per second year-over-year, with a majority of it now located at the King Mountain wind farm in West Texas. We finished the year with one of our most productive quarters to date, producing 1,562 bitcoin in the fourth quarter… In December, we expanded our agreement with Applied Digital to deploy roughly 12,000 S19 XPs, which are approximately 30% more energy efficient than the prior generation of servers, at their Jamestown facility in North Dakota… We also took proactive measures to strengthen our liquidity position and enhance the performance of our mining fleet.”
To view the full press release, visit https://ibn.fm/an5Xz
About Marathon Digital Holdings Inc.
Marathon is a digital asset technology company that focuses on supporting and securing the bitcoin ecosystem. The company is currently in the process of becoming one of the largest and most sustainably powered bitcoin mining operations in North America, while remaining asset light. For more information about the company, visit www.MarathonDH.com.
Marathon Digital Holdings Inc. (MARA), closed Thursday's trading session at $3.91, off by 7.346%, on 24,923,844 volume. The average volume for the last 3 months is 589,503 and the stock's 52-week low/high is $3.11/$33.90.
Alliance Resource Partners L.P. (ARLP)
The Online Investor, Zacks, TradersPro, QualityStocks, The Street, MarketBeat, InvestorPlace, Marketbeat.com, MarketClub Analysis, TopStockAnalysts, Dividend Opportunities, The Wealth Report, TheStockAdvisor, The Motley Fool, Money Morning, StreetAuthority Daily, Market Intelligence Center Alert, BUYINS.NET, Investing Daily, Daily Wealth, Daily Trade Alert, Rick Saddler, TraderPower, SmarTrend Newsletters, The Growth Stock Wire, Wealth Insider Alert, TheOptionSpecialist, TheStockAdvisors, Trading Concepts, Daily Markets, Trades Of The Day, Eagle Financial Publications, Early Bird, FNNO Newsletters, Greenbackers, Insider Wealth Alert, PoliticsAndMyPortfolio.com, Investor Update, Short Term Wealth, Top Pros' Top Picks, Leeb's Market Forecast, TheTradingReport, TheStreet Offers, Money and Markets, StreetInsider and Investment U reported earlier on Alliance Resource Partners L.P. (ARLP), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Last year, the biggest coal-mining companies globally raked in more than $97 billion in profits. This is quite a feat, especially based on the expectation that the coal industry was projected to be experiencing loses.
Data from S&P Capital IQ shows that the demand for fuel increasing globally is responsible for the tripling of coal profits in the 12 months ending June 30, 2022. Figures from the International Energy Agency show that in 2022, the demand for coal around the globe increase by 1.2% to set a new record.
Despite most countries pledging to stop using coal in favor of cleaner energy sources, energy security concerns caused by Ukraine’s invasion by Russia saw many revert back to this fuel as a source of power and heat. The European energy crisis and high gas prices also prompted a significant increase in the demand for coal. Last year, the average benchmark price for thermal coal in Europe was $295. This is double the price of the fuel in 2021 and almost four times higher than the fuel’s average price in the period between 2010-2020.
Companies that mine coal which is exported and traded globally have recorded bigger gains in comparison to state-owned miners who sell to domestic markets. Companies that raked in the highest profits were Glencore, whose earnings stood at $13.2 billion. In the 12 months ending on June 30, 2021, Glencore’s earnings stood at $1.2 billion.
China Shenhua also saw an increase in its profits, bringing in roughly $12.2 billion. BHP, which is the biggest producer of metallurgical coal outside China globally, raked in $9.5 billion. This figure represents a 3,200% increase, from the $288 million in profits that the mining giant recorded in the previous period.
Anglo American’s coal division also shifted from making millions in losses in the 12 months ending on June 30, 2021, to raking in more than $2 billion in the same period one year later. Anglo American is the fifth largest producer of metallurgical coal outside China. This coal is usually used in the manufacture of steel.
UBS analyst Myles Allsop stated that seaborne coal producers gained the most from the coal boom, noting that the war in Ukraine caused extreme tightness in the coal market. Allsop added that sanctions imposed on coal from Russia prevented the fuel from reaching the European market.
The improvement in the fortunes of coal mining companies comes just a year after the COP26 summit, where many pledged to phase out the use of coal. However, the energy crunch threw a spanner in the works, and as a result, coal producers such as Alliance Resource Partners L.P. (NASDAQ: ARLP) are enjoying a good spell on the international energy market.
Alliance Resource Partners L.P. (ARLP), closed Thursday's trading session at $19.51, off by 0.459184%, on 591,491 volume. The average volume for the last 3 months is 252,877 and the stock's 52-week low/high is $12.60/$27.63.
Compass Pathways PLC. (CMPS)
QualityStocks, InvestorPlace, MarketBeat, Daily Trade Alert, StreetInsider, Schaeffer's, Trades Of The Day and The Street reported earlier on Compass Pathways PLC. (CMPS), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Last week, legislators in New York filed a measure to legalize psychedelics such as ibogaine and psilocybin for the 2023 legislative session. The bill, which will be introduced formally this week, is being sponsored by assembly members Linda Rosenthal, Karines Reyes and Jo Anne Simon. The bill would make the use, possession, cultivation, creation, production, gifting, analysis, sharing or exchange of natural fungus or plant-based hallucinogens legal for individuals aged 21 and above.
The psychedelics that will be legalized if the bill is approved include psilocybin, mescaline, psilocyn, ibogaine and DMT. The bill will also allow individuals to use the entheogenic substances in religious ceremonies as well as engage in various psychedelic services. Local and state law enforcement will also be barred from providing assistance to or cooperating with the federal government for the purpose of enforcing laws on controlled substances against activities that have been legalized in the state of New York.
The bill outlines some protections, including that individuals will not lose public assistance or professional licenses or be denied behavioral or mental health services for using these substances. Additionally, the use of psychedelics will not be used as a basis for a child welfare investigation. Furthermore, employers will be prohibited from taking action against workers who lawfully use the substances off duty.
In addition to this, localities in the state will not be permitted to enact laws that criminalize psychedelics. Finally, the bill will remove DMT, ibogaine, mescaline, psilocyn and psilocybin from the state of New York’s list of banned substances.
We have seen Rosenthal’s interest in psychedelic policy grow these last few years. In 2021, Rosenthal filed a measure that would have removed psilocyn and psilocybin from New York’s controlled substances list. She also introduced a separate measure that would establish a psychedelic research institute to study the therapeutic potential of psychedelic substances for a range of conditions and make policy recommendations.
Additionally, assembly member Pat Burke and Rosenthal took part in a psychedelics symposium in 2022. Burke has separately sponsored another bill to legalize the medical use of psilocybin for eligible patients in a clinical setting.
The introduction of this new psychedelic legalization bill is the latest sign that this year will be good for psychedelic reform in the country, following the success in states such as Colorado, which saw voters approve a bill to legalize the possession of psychedelics.
In California, Senator Scott Wiener also refiled a measure that would legalize the possession of various psychedelics.
All this legislative activity points to a favorable future for industry players that are heavily investing in developing therapeutics from a variety of psychedelic compounds, such as Compass Pathways PLC. (NASDAQ: CMPS).
Compass Pathways PLC. (CMPS), closed Thursday's trading session at $7.42, off by 4.7497%, on 256,792 volume. The average volume for the last 3 months is 369,170 and the stock's 52-week low/high is $6.54/$21.99.
ElectraMeccanica Vehicles Corp. Ltd. (SOLO)
Green Car Stocks, InvestorPlace, QualityStocks, StocksEarning, Kiplinger Today, Schaeffer's, MarketClub Analysis, StockMarketWatch, TradersPro, BUYINS.NET, Trades Of The Day, MarketBeat, The Street, Daily Trade Alert, TopPennyStockMovers, The Online Investor, VectorVest, PoliticsAndMyPortfolio, Small Cap Firm, SmallCapVoice, Eagle Financial Publications and Cabot Wealth reported earlier on ElectraMeccanica Vehicles Corp. Ltd. (SOLO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The San Diego International Show, an annual event that brings the latest automobiles, SUVs and trucks as well as alternative fuel vehicles together in one place, concluded on Jan. 2, 2023. After two years of being postponed due to COVID-19, the auto show returned, jam-packed with attendees and thousands of the newest vehicles in a non-selling setup, displaying a wide selection of the latest models that either aren’t released in showrooms or are currently needed by people but difficult to find.
This year, it was impossible to avoid seeing the presence of electric as well as hybrid vehicles, which was a notable improvement over previous editions. While welcoming the new change, attendee Steve Byer, who owns an EV, said it followed naturally that since they owned a solar system in their home, getting an electric vehicle was a natural next step for them, especially given that they’d plug the vehicle into the power and awaken the next day to a full battery.
On the other hand, Guillermo Barajon and his wife were in search of a new all-electric vehicle, claiming that the recent spike in gas prices influenced their switch decision.
Joe Quiroz, another attendee in search of a new vehicle, intends to stay with a conventional gas-fueled vehicle. Quiroz did not appear to be concerned about the rising cost of gas or the forthcoming state regulation requiring all new vehicles, including trucks as well as SUVs being sold throughout California to be electrified by 2035.
Others, such as John Saad, remained neutral and looked for plug-in hybrids, which he claimed was currently the best family choice because the EV charging infrastructure is still being developed across the nation. According to Saad, these vehicles can travel a maximum of 40 miles in range on electricity, after which it must be driven home for another charge at night. On long trips, however, there is no need to drive then wait anxiously at charging facilities because the vehicle has a standard gasoline-powered engine that will carry riders the remaining distance.
Because of the simplicity of having a vehicle charged while at home and the low maintenance costs associated with electric vehicles, Steve Byer, who owns an electric vehicle, declared that he would never switch back to conventional gas vehicles. Despite the numerous benefits of electric vehicles, Byer warns that not all EVs will travel the claimed 100% of the distance on a single charge.
As such annual shows spread awareness about electric vehicles, the sales of all EV manufacturers such as ElectraMeccanica Vehicles Corp. Ltd. (NASDAQ: SOLO) are likely to gradually take off and surge as the years go by.
ElectraMeccanica Vehicles Corp. Ltd. (SOLO), closed Thursday's trading session at $0.6645, off by 0.820895%, on 372,055 volume. The average volume for the last 3 months is 276,561 and the stock's 52-week low/high is $0.5715/$2.44.
The QualityStocks Company Corner
- Vision Energy Corp. (OTCQB: VENG)
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
- MetAlert Inc. (OTC: MLRT)
- Freight Technologies Inc. (NASDAQ: FRGT)
- HeartBeam Inc. (NASDAQ: BEAT)
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
- SideChannel Inc. (OTCQB: SDCH)
- Data443 Risk Mitigation Inc. (OTC: ATDS)
- India Globalization Capital Inc. (NYSE American: IGC)
- Flora Growth Corp. (NASDAQ: FLGC)
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF)
Vision Energy Corp. (OTCQB: VENG)
The QualityStocks Daily Newsletter would like to spotlight Vision Energy Corp. (OTCQB: VENG).
•Through their ETBV project in Europe, renewable energy developerVision Energy is continuing its work to realize project developmentobjectives and has increased its core team of commercial andengineering professionals for its Green Energy Hub project
•The company’s Project Management and Consultancy (“PMC”) team, withdecades of experience working with major industry operators, hascollectively developed over 8 million cubic meters of bulk storagecapacity worldwide
•The Green Energy Hub project is strategically located in the NorthSea port of Vlissingen at the mouth of the Westerschelde estuary inthe Netherlands, and is positioned to be the first terminal inEurope focused on green and low-carbon energy products
•The global renewable energy market was valued at $881.7 billion in2020 and is expected to reach $1.98 trillion by 2030
Vision Energy Corp. (OTCQB: VENG) (“Vision Energy”) is a forward-looking energy company developing carbon reduced solutions for the commercial, industrial and transportation sectors. Vision Energy is leveraging its team’s proven track-record in site and asset procurement, accelerating development and permitting processes, plant design, and grid integration to facilitate low-carbon energy production, supply and distribution. The company is pursuing reliable offtake relationships and operating partnerships with energy industry participants and end users seeking carbon abatements across feedstock and fuels. Vision Energy is committed to providing low carbon energy solutions with maximized yield, with projects designed to exploit existing gas and power infrastructure, to integrate and facilitate import and/or distribution of reduced-carbon energy to domestic and global supply chains.
The company believes that hydrogen and liquid carriers of hydrogen are the most reliable alternatives to fossil fuels. Hydrogen is anticipated by many energy analysts to become more widely competitive as an alternative mobile energy source as early as 2030, as economies of scale drive down costs.
According to the International Energy Agency report ‘Hydrogen in North-Western Europe (2021)’, the region is well placed to lead hydrogen adoption as a clean energy source. Today, this region comprises approximately 5% of global hydrogen demand and 60% of European demand. Moreover, the region is home to the largest industrial ports in Europe, where much of this hydrogen demand is located, and presents a well-developed natural gas infrastructure connecting these ports with other industrial hubs. This gas network could be partially repurposed to facilitate hydrogen delivery from production sites to demand centers. Governments in this region also have ambitious goals for greenhouse gas emissions reduction and there is strong political interest in hydrogen as a pathway to maintaining industrial activity in the region.
Vision Energy is based in Jersey City, New Jersey.
Projects
Through wholly owned subsidiary Evolution Terminals BV, Vision Energy is pioneering a Green Energy Hub development project for the import, storage and distribution of low-carbon renewable fuels and hydrogen carriers, strategically located in the North Sea port of Vlissingen at the mouth of the Westerschelde estuary in the Netherlands. This Green Energy Hub is positioned to be the first terminal in Europe focused on green and low-carbon energy products.
Vision Energy is at an advanced stage of planning for the construction of its Green Energy Hub and is on schedule to file for the remaining construction and environmental permits by December 2022. The Green Energy Hub design is capable of receiving seagoing vessels, barges and coasters, served by a dedicated deep-water jetty as well as rail and truck loading infrastructure that will enable direct access to purpose-built storage and handling facilities for low-carbon fuels and hydrogen carriers, including ammonia, methanol and liquid organics. Phase 1 capital expense is estimated at approximately €450 million, including jetty infrastructure, and will provide for up to 400,000 cubic meters (CBM) of storage capacity with land already secured for future expansion.
Market Opportunity
In Northwestern Europe, the market for green hydrogen, or hydrogen produced by renewable energy, is growing rapidly. The current hydrogen demand projections outstrip the scheduled production for the next five to 10 years.
The company believes that all producers will face high demand. Moving beyond its initial Green Energy Hub, Vision Energy is focused on countries where governments support a regulatory standard that promotes hydrogen production and consumption. Many governments have established various incentives and financial mechanisms to accelerate and promote the use of hydrogen as a renewable energy source.
The EU, through its European Green Deal, has set an objective to become climate-neutral by 2050, implying the near total phase-out of fossil fuels in the EU energy system, and many countries are working to put in place subsidy programs for the development of green hydrogen facilities in anticipation of this goal.
Vision Energy projects its total addressable market at €10 billion by 2050.
Management Team
Andrew Hromyk is CEO of Vision Energy. He has supported and operated chemical and energy operations in the Permian Basin, central and south Texas, Arkansas, Alberta and internationally. An active investor, he has been involved with companies developing a diverse range of technologies, from enhanced and conventional hydrocarbon recovery processes to wireless infrastructure. He has participated in numerous industrial and commercial real estate developments. He also has served as a director of several private companies that became publicly traded on Nasdaq, NYSE and TSX. He studied economics at Chaminade University and the University of British Columbia.
Arron Smyth is Executive Vice President of Corporate Development at Vision Energy. He has more than 18 years of experience in financial services, investment banking, business leadership and operations in both developed and emerging markets. Since 2018, he has been Managing Director Europe for the First Finance group of companies, developing and supporting the group’s private equity investments and projects, including Evolution Terminals, the Netherlands-based developer of tank terminal and port infrastructure for the bulk storage and handling of clean and sustainable energy products.
Matthew Hidalgo is CFO of Vision Energy. He has over 15 years of experience in accounting, operations, finance, corporate restructuring and integrating acquisitions. He is a Managing Partner at Turquino Equity LLC, a private equity investment firm. Formerly, he was the controller and operations manager for the largest subsidiary of WPCS International Incorporated. Prior roles included managing accounting functions for several Australian subsidiaries. After graduating from Penn State with a bachelor’s degree in accounting, he began his career at PricewaterhouseCoopers.
Vision Energy Corp. (OTCQB: VENG), closed Monday's trading session at $2.05, up 3.0151%, on 73,251 volume with 375 trades. The average volume for the last 3 months is 57,207 and the stock's 52-week low/high is $1.04999995/$5.63000011.
Recent News
- Vision Energy Corp. (OTCQB: VENG) - Vision Energy Corp. (VENG) Expands Project Management and Development Team, Advancing Green Energy Hub Project in North Sea Port of Vlissingen
- TinyGemsBreaks - Vision Energy Corp.'s (VENG) Pioneering Terminal Set to Accelerate Energy Transition
- Vision Energy Corp. (VENG) Update Underscores Acceleration of its Green Energy Hub Development Near European Hydrogen Backbone
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
The QualityStocks Daily Newsletter would like to spotlight BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV).
To view the full press release, visit https://ibn.fm/fzoXk
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) is a biopharmaceutical company focused on developing, manufacturing and commercializing innovative products for the prevention and treatment of infectious diseases and other illnesses.
In collaboration with the prestigious Max Planck Institute for Multidisciplinary Sciences (MPG) and the University Medical Center Göttingen (UMG), both in Germany, BiondVax is developing a pipeline of innovative nanosized antibody (NanoAb) therapies addressing diseases underserved by current treatments and with large and growing markets, such as COVID-19, asthma and psoriasis.
NanoAbs, also known as VHH-antibodies or Nanobodies, are alpaca-derived nanosized antibodies that exhibit multiple significant competitive advantages over existing antibody therapies, including stability at high temperatures, superior binding affinity, more effective and convenient routes of administration and efficient production. BiondVax is uniquely positioned to advance nanosized antibody innovation from R&D through commercialization.
The company’s highly experienced and successful pharmaceutical industry leadership team includes former senior executives from Novartis, GSK and Bristol-Myers Squibb.
Since its founding, BiondVax has executed eight clinical trials, including a seven-country, 12,400-participant Phase 3 trial of a prior influenza vaccine candidate, and it built, owns and operates a 20,000 sq. ft. state-of-the-art GMP biologics manufacturing facility housing its laboratories, production facilities and offices.
Lead Candidate: Inhaled COVID-19 NanoAb
In December 2021, BiondVax signed definitive agreements with the Max Planck Society – parent organization of the Max Planck Institute for Multidisciplinary Sciences– and the UMG to enter a strategic collaboration for the development and commercialization of innovative COVID-19 NanoAbs.
The company is planning a rapid development path that leverages its expertise and capabilities in biological drug development and manufacturing. BiondVax anticipates preclinical proof-of-concept results for an inhaled COVID-19 NanoAb by the end of 2022, with initial Phase 1/2a human clinical trial results expected in 2023.
The intended inhaled mechanism of delivery of BiondVax’s COVID-19 NanoAb formulation may serve as a significant differentiator when compared to approved monoclonal antibodies, which are injected. Inhaled delivery has shown to be cheaper, more convenient and likely safer for patients and providers.
NanoAb Pipeline: Psoriasis, Asthma and More
The COVID-19 NanoAb development agreement is part of a broader five-year research collaboration agreement signed in March 2022 covering discovery, development and commercialization of NanoAbs for several other disease indications with large market medical needs, including asthma, psoriasis, macular degeneration and psoriatic arthritis.
BiondVax has an exclusive worldwide license for development and commercialization of COVID-19 NanoAbs and exclusive options for similar worldwide licenses for NanoAbs for the above mentioned additional large market disorders currently underserved by approved therapeutic antibodies.
Academic research teams from MPG and UMG have verified strong affinity by the new NanoAbs to their biological target molecules and high thermostability. They have also demonstrated strong neutralization by several NanoAb candidates of their respective target molecules. Neutralization studies of the other NanoAbs are expected to begin later in 2022.
Based on the promising results, BiondVax will focus development efforts beginning with the following NanoAbs:
- NanoAbs targeting IL-17 as drug candidates for the potential treatment of psoriasis and psoriatic arthritis
- NanoAbs targeting IL-13 and NanoAbs targeting TSLP as drug candidates for the potential treatment of asthma
These are conditions for which the antibody target is validated by existing treatments and the mechanism of action is well understood. Both represent large medical needs and growing markets. BiondVax anticipates preclinical proof-of-concept for at least one of these NanoAbs in 2023. This is in addition to the aforementioned human clinical Phase 1/2a for the inhaled COVID-19 NanoAb therapy, which is also anticipated in 2023.
Market Opportunity
COVID-19 treatment, target of the company’s lead NanoAb therapy candidate, had an estimated market size of $22 billion in 2021.
Future BiondVax drug candidates will target conditions with large markets growing at attractive CAGRs.
The global asthma treatment market was valued at $18.08 billion in 2019 and is projected to reach $26.01 billion by 2027, exhibiting a CAGR of 4.5% during the forecast period, according to Fortune Business Insights. The research firm predicts that the global psoriasis treatment market will grow from $26.37 billion in 2022 to $47.24 billion by 2029, exhibiting a CAGR of 8.7% over the forecast period.
Management Team
Amir Reichman is BiondVax’s CEO. He previously was Head of Global Vaccines Engineering Core Technologies at GSK Vaccines in Belgium. Prior to that, he held leadership roles at Novartis Vaccines’ Global Vaccines Supply Chain Management organization. He was the first employee of NeuroDerm Ltd., a company focused on transdermal drug delivery, and served as Chief Engineer and Senior Scientist until his departure in 2009. He earned a M.Sc. in Biotechnology Engineering from Ben-Gurion University and an MBA in Finance and Health Care Management from the University of Pennsylvania’s Wharton School.
Tamar Ben-Yedidia, Ph.D., is Chief Science Officer at BiondVax. She has more than 30 years of experience in immunology, with specific expertise in the development of vaccines. She began her career with Biotechnology General Ltd., working on development of a recombinant Hepatitis-B vaccine. She later joined the Weizmann Institute of Science, working on the design of a peptide-based vaccine against several pathogens. She is widely published, with numerous refereed articles and invited reviews in various scientific journals. She received her Ph.D. from the Weizmann Institute.
Elad Mark is COO at BiondVax. He has over 15 years of biotechnology industry experience encompassing diverse project stages including feasibility studies, conceptual and detailed design, commissioning, qualification and process validation. Prior to joining BiondVax, he led Novartis’s $800 million investment in a biologics facility in Singapore. With Biopharmax and Antero, both global pharmaceutical engineering companies, he successfully led projects in Israel, China and Singapore. He holds a BSc. in Engineering from the Afeka Tel Aviv Academic College of Engineering and an MBA from the Open University of Israel.
Uri Ben-Or is CFO at BiondVax. He has served as CFO with public life science companies traded on the TASE, OTC and Nasdaq. Ben-Or provides his services to BiondVax through CFO Direct, a company he founded and for which he serves as CEO. He served as the VP of Finance of Glycominds, a leading biotechnology company, and as CFO of a spin-off from Telrad Networks. He also served as a Corporate Controller at Menorah Capital Markets and as an Auditor at PWC. He holds a B.A. in Business from the College of Administration, an MBA from Bar-Ilan University, and is a CPA.
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV), closed Thursday's trading session at $3.164, up 6.1745%, on 20,691 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $26.10/$.
Recent News
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Announces Participation in Upcoming Biotech Showcase
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Releases Business Updates and Announces CEO's Upcoming Speaking Engagements
- InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Releases Letter to Shareholders
MetAlert Inc. (OTC: MLRT)
The QualityStocks Daily Newsletter would like to spotlight MetAlert Inc. (OTC: MLRT).
To view the full press release, visit https://ibn.fm/dNS7P
MetAlert Inc. (OTC: MLRT) is a pioneer in location sensitive health monitoring devices (estimated $47 billion industry in 2021) and wearable technology products (industry forecast to reach $174 billion by 2030).
With over 20 years of experience and an extensive patent portfolio (30+), MetAlert is a leader for consumers/patients afflicted with Alzheimer’s, dementia, and autism (ADA). This market represents approximately 2.9% of the world’s population (approximately 34 million people in 24 developed countries). Due to specific behaviors (problems with memory, adversity to wearing unknown items, etc.) of consumers/patients in this market segment, traditional products, such as an iPhone or Fitbit, are not a practical solution. This has created a significant market with very few competitors for MetAlert.
MetAlert and its subsidiaries are engaged in designing, developing, manufacturing, distributing, and selling products and services in GPS/BLE wearable technology, personal location, wandering assistive technology, and health data collection and monitoring. The company offers a global end-to-end hardware, software, and connectivity solution, in addition to developing two-way tracking technologies, which seamlessly integrate with consumer products and enterprise applications.
Using its award-winning, patented GPS SmartSole® as a hub for collecting and transmitting data to the cloud in real-time, MetAlert is expanding its value proposition to consumers and increasing its revenue per user (RPU) while creating the largest database of health statistics for ADA consumers/patients. MetAlert generates revenue from product sales, recurring subscriptions, intellectual property (IP) licensing, and professional services. The company has international distributors servicing customers in over 35 countries and is an approved U.S. military government contractor. Its customers include public health authorities and municipalities, emergency and law enforcement, private schools, assisted living facilities, NGOs, small business enterprises, senior care homes and consumers.
The company is headquartered in Los Angeles, California, with a sales office in London, England, and distributors across the globe.
Products
- GPS SmartSoles® HUB (launched Q4 2022) is a GPS/BLE-equipped insole that allows remote monitoring, data collection, and encrypted data transmission to the cloud.
- Telehealth (available Q4 2022) allows access remotely to doctors and other health professionals on an as-needed basis. This service will also function as the prescribing doctor once Medicare reimbursement codes are established.
- Concierge (available Q4 2022) provides 24/7/365 enhanced emergency response that coordinates with all relevant parties to quickly detect false alarms and escalate response as needed.
- Bluetooth Enabled Devices (available Q1 2023) include third-party devices that collect vitals and other health data and connect with the GPS Smartsoles® HUB.
- Artificial Intelligence (available Q1 2023) software will evaluate the Teradata of health information identifying trends and respond to preestablished alert thresholds.
- Take-Along Tracker is a small GPS tracking device – less than three inches long – that works with 4G cellular service and will have the same “HUB” functionality as the GPS Smartsoles®. This versatile and affordable mini tracker boasts super long battery life, with up to 14 days of operation per charge.
- RoomMate™ is a wall-mounted alert system that detects and alerts caregivers about patient behavior that could lead to falls and injuries. The system features 3D infrared and wall-mounted sensors, eliminating the need for any other physical installation or wearables. RoomMate™ offers patient privacy by design. Images are not stored, but all actions are logged. It’s a unique solution for looking after patients without intruding on their personal space.
Market Outlook
According to Grand View Research (Patient Monitoring Devices Market Size & Share Report, 2030), the global patient monitoring devices market size was valued at $47.0 billion in 2021 and is expected to expand at a compound annual growth rate (CAGR) of 7.8% from 2022 to 2030. The expansion of the industry can be attributed to the rise in demand for monitoring devices used to measure, distribute, record, and display a variety of biometric data, including blood pressure, temperature, and blood oxygen saturation level.
The growing number of chronic disorders, such as diabetes, stroke, and kidney disease, are driving the demand for patient monitoring devices. For instance, according to the World Health Organization (WHO), about 422 million people globally have diabetes. Likewise, the number of asthma and chronic obstructive pulmonary disease patients (COPD) is increasing rapidly.
According to the WHO, around 235 million people suffer from asthma. As a result, peak flow meters, which are used to gauge respiration rate, are increasingly used. The market for patient monitoring devices is driven by the simplicity with which it is handled, transported, and remotely accessible. Major market players are engaging in a variety of tactics to expand the industry, including partnerships, cooperation, innovation, launches, and mergers.
During the COVID-19 outbreak, social segregation and quarantining procedures were put into place worldwide. Many people avoided regular hospital visits as a result. Many people now need routine home temperature and oxygen level monitoring to maintain track of their health, thereby demanding monitoring devices at home.
Various government programs are supporting the pandemic outbreak. The FDA has granted Emergency Use Authorizations (EUAs) for a few wearables and patient monitoring devices to improve access to medicines, monitor patients more closely, and lessen the risk of SARS-CoV-2 exposure to medical professionals during the COVID-19 pandemic.
The growing popularity of wearable and remote patient monitoring devices is another factor fueling the market’s expansion. By fusing clinical symptomology with vital indicators, wearable technology helps in the diagnosis of many chronic diseases. Thus, there has been a dramatic rise in the usage of wearable technology to combat COVID-19.
The wearable medical device market is anticipated to reach $174.48 Billion by 2030, expanding at a 27.1% CAGR during the forecast period (2022-2030), according to Market Research Future.
MetAlert identifies the total addressable market for its wearable patient monitoring tech for those with Alzheimer’s, dementia, and autism at more than 34 million potential patients in North America, Europe, South Africa, and Asia.
Management Team
Patrick E. Bertagna is Founder, CEO and Chairman at MetAlert. He began his career in apparel sales in 1983 and was promoted to national sales manager within two years. In 1986, he founded his first company importing apparel from Europe and selling to U.S. retailers from JCPenney to Neiman Marcus. He has founded several technology and apparel companies, including MetAlert in 2002, which he took public in 2008. He attended Cal State University Northridge with a business major and a psychology minor.
Louis Rosenbaum is COO of MetAlert. He co-founded Global Trek Xploration and was an initial investor in MetAlert. He has successfully started companies in multiple industries, including apparel, environmental services, and the music industry, achieving annual revenues in the multi-millions of dollars. He previously was president of Elements, a women’s apparel company, and of Advanced Environmental Services.
Alex McKean is CFO at MetAlert. He is also the CFO of Encore Brands Inc., a position he has held since 2009. He has held positions as Controller and VP of Finance at 24:7 Film and InternetStudios.com, Director of FP&A/SVP at Franchise Mortgage Acceptance Company, Corporate Accounting Manager/Treasurer of Polygram Filmed Entertainment and Assistant Treasurer/Controller for State Street Bank. He holds an International MBA from Thunderbird School of Global Management and undergraduate degrees in business and political science from Trinity University.
MetAlert Inc. (OTC: MLRT), closed Thursday's trading session at $0.26, up 20.9865%, on 29,671 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.06/$1.00.
Recent News
- MetAlert Inc. (OTC: MLRT) - InvestorNewsBreaks - MetAlert Inc. (MLRT) Issues Shareholder Update
- MetAlert, Inc. (MLRT) Recognizes Link Between Cognition in Aging Population and Use of Hearing Aids
- Study Finds Smoking Linked to Mid-Life Cognitive Decline, Confusion
Freight Technologies Inc. (NASDAQ: FRGT)
The QualityStocks Daily Newsletter would like to spotlight Freight Technologies Inc. (NASDAQ: FRGT).
To view the full article, visit https://ibn.fm/fpu58
Freight Technologies Inc. (NASDAQ: FRGT) (“Fr8Tech”) is a technology company developing solutions to optimize and automate the supply chain process, providing a platform for B2B cross-border shipping in the NAFTA region. The company’s mission is to revolutionize cross-border shipping by providing carriers with increased growth opportunities and shippers with flexibility, visibility and simplicity for the once-complex process of international over-the-road shipping.
Freight Technologies, formerly known as Hudson Capital Inc., assumed its current name and ticker symbol on May 27, 2022. Its primary operating subsidiary and its marketplace are known as Fr8App, and it conducts operations throughout North America under the names of Fr8App and/or Freight App. The company is headquartered in Houston, Texas, with multiple locations across the U.S. and Mexico.
The Fr8Tech Solutions Suite
Fr8Tech leverages artificial intelligence to provide cloud-based platforms aimed at automating the over-the-road transportation process, effectively reducing human touch points and expediting load booking times. The company’s suite of solutions includes:
- Fr8app – A B2B marketplace powered by AI and Machine Learning offering a real-time broker portal to connect shippers with qualified carriers
- Fr8Radar – A tracking solution providing shippers and carriers real-time locational data via Fr8app’s mobile solution or through integration with third-party GPS alternatives
- Fr8TMS – A transportation management system designed to help shippers manage their freight and all of the documents involved in shipping transactions, including invoices, customs documents, confirmation rates and proof of deliveries
- Fr8FMS – A fleet management system allowing transportation companies to better manage their fleets, reduce operational costs and provide better service to their customers
- Fr8Data – A data solution offering real-time dashboards and reports to shippers and carriers in an effort to increase visibility and control while supporting better business decisions
- Fr8Fleet – A platform that provides private fleet management, enabling large corporate shippers to purchase dedicated capacity secured by Fr8app in exchange for a fixed fee
Commitment to the Environment
Through its core focus on technology, Fr8Tech seeks to reduce the carbon footprint of the logistics industry. Its solutions aim to minimize empty miles for transportation firms and reduce overall paper consumption.
Fr8University
Fr8University is an educational program offering classroom and on-the-job training for Fr8Tech team members. Through the program, employees learn in-depth business fundamentals and applications along the truckload freight industry value chain.
Led by corporate educator Mario Mena, Fr8University is designed as an investment in the company’s human capital, providing an opportunity to communicate Fr8Tech’s corporate culture while accelerating operational growth.
Market Outlook
Fr8Tech’s established foothold in Mexico is key to its current efforts to promote sustainable growth in the cross-border shipping industry. Ongoing disruption in U.S.-Chinese trade relations have strengthened Mexico’s status as the largest trading partner of the U.S., with cross-border annual freight spending estimated at $385 billion according to data from the U.S. Department of Transportation. Annual domestic shipping in Mexico is estimated at $34 billion, while annual domestic shipping in the U.S. is estimated to total $732 billion.
Despite the size of this industry, fragmentation and inefficiencies prevail in the space. Thousands of legacy brokers, tens of thousands of shippers and hundreds of thousands of carriers still rely on outdated systems to arrange transport, spending hours on the phone negotiating pricing, waiting days to find trucks and drivers, preparing and printing forms, and operating without tracking or visibility. Add in cross-border complexity relating to customs and additional paperwork, and you have an industry ripe for technological disruption.
Fr8Tech’s recent revenue growth trends have highlighted the company’s efforts to capitalize on this opportunity. In 2021, Fr8Tech achieved revenues of $21.5 million, marking a year-over-year increase of 134%. The company issued revenue guidance for fiscal 2022 of $40 million in a February 9, 2022, press release, which would account for a further 86% year-over-year increase.
Management Team
Javier Selgas is CEO and a Director of Freight Technologies Inc. and Freight App Inc. He brings to the company over 15 years of experience developing technology and digital marketing strategies, including serving as Country Manager for Osigu, Spain, and as head of AJEgroup’s IT division for the Asia-Pacific region. Prior to joining Fr8Tech, Mr. Selgas founded digital marketing agency Lanzadera Online. He has also served as an IT consultant to major corporations, including Endesa and Ibermatica.
Mike Flinker is President of Fr8Tech. He has over four decades of experience in the transportation industry, with 30+ years focused on cross-border logistics. Prior to joining Fr8Tech, Mr. Flinker founded FLS Transportation, the largest cross-border logistics company in Canada. He also previously held positions with Clarke Transport Inc., Canadian Pacific and Reimer Express Inc. (a division of Roadway Express).
Paul Freudenthaler is the company’s CFO and Secretary to the company Board. He has over 30 years of financial expertise, having previously served as CFO for several leading companies across multiple countries, including Macquarie in Mexico, Old Mutual in Latin America and Ascentium Capital in the U.S. Mr. Freudenthaler’s experience include leadership roles from which he guided IPOs and M&A transactions.
Luisa Lopez is COO of Fr8Tech. She brings to the company 25+ years of management experience in logistics, supply chain, operations and customer service. Ms. Lopez previously served as a Director of Landstar, where she was responsible for commercial and client development strategies in the Mexican market. Additionally, she managed more than 2,000 transport units specialized in staff and school mobility while with Traxion in Mexico.
Freight Technologies Inc. (NASDAQ: FRGT), closed Thursday's trading session at $0.2398, up 0.376727%, on 2,169,844 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.1799/$7.766.
Recent News
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Stream of Steady Business on Fr8App Platform
- InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) to Support Global Industrial Electric Manufacturer with Logistics Management Solution
- Freight Technologies' Platform Fr8App Integrates with FourKites®, Significantly Increasing Capability to Provide Track and Trace to Key Enterprise Customers
HeartBeam Inc. (NASDAQ: BEAT)
The QualityStocks Daily Newsletter would like to spotlight HeartBeam Inc. (NASDAQ: BEAT) .
To view the full press release, visit https://ibn.fm/TyEWU
HeartBeam Inc. (NASDAQ: BEAT) is a cardiac technology company that has developed the first and only 3D-vector 12-lead electrocardiogram (ECG) platform for heart attack detection anytime, anywhere. The company’s proprietary ECG telehealth technology aims to redefine the way high risk cardiovascular patients are diagnosed in ambulatory and acute care settings. HeartBeam’s initial focus is on providing diagnostic data to help physicians with care management of patients with cardiovascular disease.
In August 2022, HeartBeam announced that it submitted its HeartBeam AIMI™ software for approval from the U.S. Food and Drug Administration (FDA). HeartBeam AIMI is a platform technology to improve the speed and accuracy of heart attack detection in acute care settings. The company expects FDA approval by the end of 2022, and a full commercial roll-out of HeartBeam AIMI is targeted for Q1 2023.
HeartBeam sees submission of its first product based on its platform technology as an important milestone toward commercialization, which underscores the company’s continued progress toward making the HeartBeam AIMI platform widely available to help emergency department physicians quickly and accurately identify a heart attack.
While the FDA conducts its regulatory review, HeartBeam will focus on executing key components of its commercialization plan and subscription revenue model. It will also continue to engage in discussions with strategic institutions, including academic centers, regional healthcare systems and regional community hospital systems that can utilize HeartBeam products.
The company is based in Santa Clara, California.
Products
HeartBeam’s development portfolio includes two products:
- HeartBeam AIMI is software that provides a 3D comparison of baseline and symptomatic 12-lead ECG to more accurately identify a heart attack in acute care settings and, as noted above, has been submitted for FDA approval; and
- HeartBeam AIMIGo™, the first and only credit card-sized 12-lead output ECG device coupled with a smartphone app and cloud-based diagnostic software system for remote heart attack detection.
HeartBeam is developing AIMIGo, a medical-grade detection and monitoring technology for use in remote heart attack detection, thereby allowing physicians to diagnose a patient’s heart attack as it occurs, even if the patient is not at a medical facility. The company’s system, once approved by the FDA, can be used by patients at home or almost anywhere and anytime to help their physicians assess whether chest pain is the result of a heart attack or another cause. While approximately 82% of chest pain ED visits are unnecessary, patients delay approximately 3 to 4 hours after symptoms begin, increasing mortality rates by 40%. The company’s goal is to shorten the time to treatment outside of the medical facility to improve patients’ well-being.
HeartBeam’s AIMIGo is a powerful, portable and easy-to-use prescription-based product. It comprises a smartphone app, a credit card-sized ECG device placed on a patient’s chest, the HeartBeam cloud platform, and a digital portal for the physician to view ECG results and direct patient action. For the first time outside of a medical setting, HeartBeam AIMIGo enables patients and their clinicians to determine if symptoms are due to a heart attack, quickly and easily, so care can be expedited, if needed.
Pending FDA clearance, AIMIGo is initially intended to be available by prescription, and is reimbursable under existing remote patient monitoring codes (RPM codes). This provides a new revenue stream to physicians who before did not have a way to monitor these high-risk patients. The RPM codes provide a monthly reoccurring revenue stream to the company, as well. On average, at current reimbursement rates, the practice will receive $1,300+ per year per patient they monitor, and the company will receive $600 per year per patient from this RPM reimbursement.
Market Overview
Adoption rates of telehealth services increased dramatically in recent years, with the COVID-19 pandemic serving as a major driver of growth. Among the areas seeing the greatest expansion are cardiology, radiology, behavioral health and online consultation.
Encouraging this growth, governments are actively developing new policies and reimbursement guidelines to promote the use of digital health platforms. The U.S. Centers for Medicare & Medicaid Services (CMS), for example, has recently expanded reimbursement for telehealth services. U.S. market growth is also being driven by the rising prevalence of chronic conditions and the growing geriatric population.
Remote heart attack detection is a previously unsolved problem with a massive and underserved market that is several times larger than the $2 billion total addressable market (TAM) in the U.S. for ECG cardiac arrhythmia monitoring.
Approximately 8 million Americans have suffered at least one heart attack, and a total of 18 million have been diagnosed with coronary artery disease (CAD). Based on these figures, HeartBeam projects a total addressable U.S. market TAM valued at $10 billion annually for its AIMIGo solution for remote heart attack monitoring of CAD.
Management Team
Branislav Vajdic, Ph.D., Chief Executive Officer and Founder of HeartBeam, Inc, combines over 30 years of experience in technology development and senior management positions. Dr. Vajdic has been deeply involved with the development of HeartBeam’s technology to fit his vision for the company. Prior to HeartBeam, from 2007 to 2010, Dr. Vajdic was CEO and Founder of NewCardio, a publicly traded company in the cardiovascular devices space. From 1984 to 2007, Dr. Vajdic was at Intel, where he held various senior management position. At Intel, Dr. Vajdic was the designer of first Flash memory and two key inventions that enabled Flash as a product and led engineering groups responsible for Pentium 1 through Pentium 4 designs. Dr. Vajdic was awarded two Intel Achievement Awards, the highest level of award for outstanding contributions to Intel. Dr. Vajdic is author of numerous patents and publications in the fields of cardiovascular devices, as well as chip design. Dr. Vajdic holds a Ph.D. in Electrical Engineering from the University of Minnesota.
Jon Hunt, Ph.D., has over 35 years’ experience in the medical/medical device industry with extensive domestic and international experience in general management, clinical/regulatory, sales and marketing. He also has diverse experience in Fortune 500 companies, as well as start-up environments. Dr. Hunt was the Vice President of Clinical Science and Technology, Medical Device Innovation Consortium, from July 2019 to July 2021, and Vice President of Clinical and Regulatory Affairs, Cryterion Medical from January 2018 to June 2019 (acquired by Boston Scientific Corporation in July 2018 for $202M). Dr. Hunt was the Founding President and CEO of Bardy Diagnostics, Inc. from October 2013 to November 2017 (acquired by Hill-Rom Holdings, Inc.). Prior to joining Bardy Diagnostics, Dr. Hunt spent the previous 11 years as the Vice President of Clinical & Regulatory Affairs with Cameron Health, Inc. (acquired by Boston Scientific Corporation). Dr. Hunt spent the previous 10 years with Cardiac Pacemakers, Inc., St. Jude Medical and Cardiac Pathways Corporation. Dr. Hunt began his career with Cardiac Pacemakers, Inc. (now Boston Scientific Corporation) as the Director of Clinical Programs. He subsequently held positions at St. Jude Medical in Clinical Affairs and as the Business Unit Director for the Cardiac Rhythm Management division for Europe, the Middle East and Africa. At Cardiac Pathways Corporation, Dr. Hunt held various executive positions as Vice President of International Sales and Marketing and Vice President of Worldwide Sales and Marketing (acquired by Boston Scientific Corporation). Dr. Hunt received his Ph.D. in Motor Control from The Pennsylvania State University, his Master’s from California State University, Long Beach and his undergraduate degree from Keele University in the United Kingdom.
Rick Brounstein, HeartBeam’s Chief Financial Officer, combines over 30 years of experience in health technology senior management. Since 2017, Mr. Brounstein has been and is currently a partner of Hardesty, LLC, a financial services firm, and Mr. Brounstein is currently a managing director of CTRLCFO, LLC, a firm Mr. Brounstein founded in 2016 to support funded start-ups in life science and technology. From 2008 to 2011, Mr. Brounstein was Chief Financial Officer of NewCardio, Inc., a microcap public company in the cardiology space, and, over his career, he has been with nine other companies in life science or technology, holding positions including Chief Financial Officer, Chief Operating Officer, Treasurer and Accounting Manager. From June 2001 through November 2007, Mr. Brounstein held several positions at Calypte Biomedical Corporation, a publicly traded medical device company, including Chief Financial Officer and Executive Vice President. In January 2007, Mr. Brounstein was appointed as the National Member Representative for the 2007 COSO Monitoring Project, which published new guidelines for monitoring internal financial controls in February 2009; Mr. Brounstein subsequently was a member of the FEI task force that issued the updated COSO Internal Control Framework in 2013. In March 2005, Mr. Brounstein was appointed to the SEC Advisory Committee on Smaller Public Companies. Mr. Brounstein earned his Certified Public Accountant (CPA) certification while working at Arthur Andersen LLP, formerly a public accounting firm. Mr. Brounstein holds a B.A. in accounting and an M.B.A. in finance, both from Michigan State University.
Ken Persen, HeartBeam’s Chief Technology Officer, combines over 28 years of experience in the medical device and digital health industries in engineering and senior management positions. Mr. Persen has been involved in several companies in Cardiac Rhythm Management, holding positions including Chief Executive Officer, Chief Technology Officer, Executive Vice President and Director of Engineering. Since 2016 and prior to joining HeartBeam, Mr. Persen was the Chief Technology Officer at LIVMOR, Inc., a digital health company. In addition, from 2016 through November 2021, he was also Chief Executive Officer of LIVMOR. Prior roles included Director of Engineering at Cameron Health (acquired by Boston Scientific), a late-stage medical device start up, and engineering and management positions at Guidant Corp. (acquired by Boston Scientific), a large medical device manufacturer. He has an undergraduate degree from University of Minnesota, Duluth, with a BA in Computer Science.
HeartBeam Inc. (NASDAQ: BEAT), closed Thursday's trading session at $4.33, even for the day, on 69,979 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.12/$6.74.
Recent News
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Receives Key Patent from European Patent Office
- InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Releases Shareholder Letter, Including Corporate Update
- HeartBeam Inc. (NASDAQ: BEAT) CEO Featured Guest on Big Biz Show, Talks 'Breakthrough' Heart-Attack Detection Technology
Correlate Infrastructure Partners Inc. (OTCQB: CIPI)
The QualityStocks Daily Newsletter would like to spotlight Correlate Infrastructure Partners Inc. (OTCQB: CIPI).
•Correlate, a distributed energy solutions company, sees continuedopportunities from rising government support for clean renewableenergy
•Governmental push for green energy alternatives has grown over thepast few years, evidenced by its support for multiple climatemitigation goals, such as achieving 100% wcarbon pollution-freweelectricity by 2035 and an NDC of 50-52% reductions below 2005,covering all sectors and all gases by 2030
•Achieving these goals calls for maximizing energy efficiency fornearly everything - from buildings to vehicles, with a mix ofmandates and incentives
•New York’s recently approved climate plan to enact a “cap andinvest” program for limiting emissions is a recent example of thecountry’s ongoing move toward clean renewable energy solutions
Correlate Infrastructure Partners Inc. (OTCQB: CIPI), formerly Triccar Inc., through its two subsidiaries, Correlate and Solar Site Design, offers a complete suite of proprietary clean energy assessment and fulfilment solutions for the commercial real estate industry. The company believes scaling distributed clean energy solutions is critical in mitigating the effects of climate change. CIPI is at the forefront in creating an industry-leading energy solution and financing platform for the commercial and industrial sector. The company sees tremendous market opportunity in reducing site-specific energy consumption and deploying clean energy generation and energy efficiency solutions at scale.
The opportunity exists to remove friction between today’s legacy finance process and the needed clean-energy upgrades developed within the company’s program technologies. For the U.S. to reach its 2050 carbon goals, 200,000 commercial buildings must be retrofitted every year until that date. That represents approximately a 5-10x increase over the 2022 industry process run rate.
CIPI announced completion of its acquisition of 100% of the equity of Correlate Inc. and Loyal Enterprises LLC dba Solar Site Design on December 28, 2021. The company notes these acquisitions occurred at a key inflection point of its growth. CIPI currently enjoys channel and sales partnerships with Fortune 250 companies and a strong, proven industry network.
The company’s transparent, leading-edge model changes value delivery for both facility owners and proven solution providers seeking scale. CIPI believes its rapid growth is due to industry demand for actionable, cashflow positive energy programs and the underlying carbon reduction mandates taking effect globally.
CIPI has filed with the SEC for a name change to Correlate Infrastructure Partners Inc., which will more closely reflect its new platform and growth focus. The company has been aggressively moving to rebrand, with efforts including a revised website, investor presentation materials and an investor relations awareness campaign. The company’s shares will continue to trade on the OTCQB Venture Market under the current ‘CIPI’ ticker symbol until changes are approved.
Subsidiaries
Correlate, founded in 2015, is a portfolio-scale development and finance platform offering commercial and industrial facilities access to clean electrification solutions focused on locally-sited solar, energy storage, EV infrastructure, and intelligent efficiency measures. Its unique data-driven approach is powered by proprietary analytics, concierge subscription services, and a highly scalable national fulfillment network to help building owners profit from fully funded, turnkey decarbonization and facility health programs. The platform is designed for commercial and industrial real estate owners seeking to significantly improve net operating income while meeting carbon reduction goals. The platform provides energy programs for commercial property portfolios and requires no upfront capital. Client organizations reduce their risk and generate more profits by leveraging Correlate’s unique payment programs to put more cash in the bank. Deploying Correlate’s strategic energy programs and energy management systems allows property-owning organizations to complete big energy changes across their portfolios.
Solar Site Design, founded in 2013, is a U.S. Department of Energy Sunshot Catalyst winner that provides customer acquisition and project development tools for the commercial solar industry. Its commercial marketplace platform connects highly qualified project opportunities to leading solar construction companies nationwide. The Solar Site Design platform gives commercial and industrial property owners access to the best price for a commercial solar system. Commercial solar analysts provide property owners a site assessment and working project proposal. Solar Site Design’s team of solar engineers finalize the design while approved financing providers help clients explore financing options for their projects. Then, approved contractors in Solar Site Design’s Marketplace bid on the projects, ensuring commercial and industrial property owners get the best estimates for their projects. Solar Site Design’s marketplace process promotes transparency and fair pricing. Its team of experts has nearly 20 years of experience in the solar industry. Only reputable, experienced, certified (NABCEP), licensed, bonded and insured contractors are accepted into the Solar Site Design Marketplace.
Market Outlook
CIPI is in a rapidly growing market with a unique offering to address a total market of more than 5.9 million commercial buildings in the United States, according to the U.S. Energy Information Administration. Currently, the company’s wholly owned subsidiaries, Correlate and the Solar Site Design, have an opportunity pipeline of over $100 million in commercial projects with more than $20 million in awarded backlog. According to the Rocky Mountain Institute, portfolio energy optimization is a $290 billion market in the United States driving deep financial savings and energy efficiency across the commercial sector.
Commercial buildings consume more than 35% of the generated electricity in the U.S. and are underperforming in energy efficiency at every level. These buildings waste energy, emit too much carbon, and are too costly for owners and occupants, but retrofits are not happening at the rate or scale needed.
In today’s real estate market, portfolio property owners own most commercial buildings. Yet most building efficiency work is focused on single buildings, thereby missing the distinct needs of this owner class which has very different needs than traditional owner-occupiers. The diverse nature of commercial buildings, combined with technology and performance uncertainty, make simple energy optimization initiatives – which could greatly reduce energy use and improve building value – financially unattractive, resulting in slow adoption rates. CIPI’s financial instruments and software breakdown this issue known as the “split incentive”, unlocking the majority of the addressable market.
Management Team
CIPI has in place a nationally recognized management team that has been active in the energy market since 2005.
Todd Michaels is President and CEO of CIPI and founder of Correlate. He formerly served as Vice President for Innovation at SunEdison and Senior Director Distributed Solar at NRG Energy. He founded Correlate in 2015 and has 16 years of experience in the energy industry. He graduated from Indiana University with a B.S. in Computer Information Systems.
Channing Chen is CFO at CIPI and Correlate Inc. and brings over 16 years of experience in the solar industry as a developer, financier, and business unit leader. He has held executive management roles at Solar Power Partners (acquired by NRG Energy), where he was a founding employee, SunEdison, and NRG Energy (NYSE: NRG). Most recently, Mr. Chen was founder and Managing Partner at Breakaway Energy Partners LLC – a distributed energy financing and market-making platform. To date, Mr. Chen and his teams have raised over $1.5 billion in financing across residential, commercial, and utility scale solar and energy storage projects representing over 400 MWs. He holds a B.A. in Environmental Chemistry from the University of California at San Diego and an MBA from the University of Southern California. He is also an advisor and early-stage investor to several startup companies in the renewable energy space.
David Bailey is Chief Revenue Officer of Correlate Inc. With over 15 years of executive sales, supply chain management, and energy efficiency experience, he is responsible for ensuring the success of the National Commercial Sales Unit across multiple regional project teams. Mr. Bailey created and launched the Transformation Services team while at Wesco for its multibillion-dollar Distributed Energy Resource division, formerly Westinghouse. His focus was on IoT-enabled efficiency and plant floor automation-based services. Before that, he spent several years in Global Account Sales Management, with GE Supply as a Program Manager, and is a Commercial Leadership Program graduate. Mr. Bailey received his B.S. in Mechanical Engineering from the University of Kentucky.
Jason Loyet is VP of Commercial Sales of Correlate Inc. He is a cleantech executive with over 20 years of experience leading high growth solar energy and software start-ups. Mr. Loyet is a U.S. Department of Energy SunShot Catalyst award winner for his work building the Solar Site Design technology platform. Before joining the solar energy industry in 2005, he founded and sold two software companies in the streaming media (GlobalStreams) and newspaper publishing (MyCapture) industries. Mr. Loyet currently serves as a Member of the Board of Directors for the Tennessee Solar Energy Industry Association (TenneSEIA).
Deke Welling is Head of Project Development and Fulfillment Services at Correlate Inc. He has over 19 years’ experience in the energy industry with an emphasis on renewables and energy efficiency over the past seven years. Prior to entering the renewables sector, Mr. Welling was the CEO of Welling Resources, an energy development company focused on the exploration of oil and natural gas reserves in the U.S. It was this experience that led him into the renewables sector and leading a charge for more sustainable resources. Additionally, Mr. Welling also served as the CEO of Circle L Solar Inc., a top 100 solar installer in the United States since 2016. Through his leadership, Circle L Solar experienced a growth rate of over 2,250% from 2016 to 2019, resulting in his company being listed on the Inc. 5000 list of the fastest growing private companies in the U.S. (Rank #176) and being named ‘Top Energy Company’ and ‘Entrepreneur of Year for the Energy Industry’ by the American Business Awards® in 2019 and again for ‘Entrepreneur of the Year’ in 2021.
Kevin Warren is Head of Construction and Development Engineering at Correlate Inc. He is a solar veteran with over 12 years of experience in the field. Prior to co-founding CLS, Mr. Warren was the owner of Beacon Consulting and has originated, consulted, designed and/or engineered over 122 MW of PV installations ranging from small commercial to utility scale projects throughout Texas, California, Colorado and North Carolina. He holds a Photovoltaic Technical Sales Professional Certification from the North American Board of Certified Energy Practitioners and certifications from Solar Energy International in PV Installation, PV Technical Sales, PV battery-based design, PV design and engineering, and PV operations and maintenance. Along with PV expertise, Mr. Warren is a LEED Green Building Associate, a certified building analyst from the Building Performance Institute, a Certified Renewable Energy Professional from the Association of Energy Engineers and holds a designation in High-Performance Sustainable Buildings from the BOMI Institute. He studied Electrical Engineering at the University of Texas at Arlington.
Tom Kunhardt is Director of Customer Success at Correlate. He previously held a similar position at Clean.Tech and was Corporate Trainer, Learning & Development, at NRG Energy. He has 15 years of experience in the solar and clean energy industries helping homeowners and businesses find solutions to their energy needs. He holds a bachelor’s degree from the University of Massachusetts.
Correlate Infrastructure Partners Inc. (OTCQB: CIPI), closed Thursday's trading session at $1.06, even for the day. The average volume for the last 3 months is and the stock's 52-week low/high is $0.63/$3.25.
Recent News
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Encouraged by Growing Government Moves to Reduce Emissions
- Government's Drive to Expand Clean Energy Access Confirms Greener Market for Correlate Infrastructure Partners Inc. (CIPI)
- Correlate Infrastructure Partners Inc. (CIPI) - Making Energy Transition Efficient, Transparent, and Cost-Effective
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
Cancer is a
CAR T-cell therapy proving to be revolutionary
The FDA first approved the first
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Thursday's trading session at $2.91, off by 5.5195%, on 37,161 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.05/$41.40.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Four Developments in Cancer Research That Occurred in 2022
- InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) CEO to Present at Virtual Investor 2023 'Companies to Watch'
- InvestorNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Announces Strategic Board Appointment of Faith L. Charles, JD
SideChannel Inc. (OTCQB: SDCH)
The QualityStocks Daily Newsletter would like to spotlight SideChannel Inc. (OTCQB: SDCH).
To view the full podcast, visit https://ibn.fm/vAbfs
SideChannel Inc. (OTCQB: SDCH) simplifies cybersecurity for mid-market companies by matching them with highly experienced information security officers at a cost lower than building an in-house information security team or hiring a full-time CISO.
SideChannel’s team of virtual Chief Information Security Officers (vCISOs) possesses a combined 400-plus years of experience in cybersecurity. They’ve honed their skills and abilities in places like Anthem, Dick’s Sporting Goods, Best Buy, TD Bank and the Pentagon. SideChannel lends this talent to clients, creating value in the form of a bespoke cybersecurity program perfectly sized for the growing enterprise.
SideChannel is committed to creating top-tier cybersecurity programs for SMBs to help them protect their data and assets. To date, SideChannel has created more than 50 multi-layered cybersecurity programs for its clients.
Reports show that cyberattacks on SMBs have increased in recent years, as organizations’ network attack surfaces have grown exponentially with remote and in-office workers increasingly relying on cloud environments, mobile devices, software applications and third-party suppliers to conduct business.
SideChannel continues expanding its service offerings, workforce and customer base, attracting over 20 virtual CISOs to serve across industries including fintech, biotech, healthcare, manufacturing, legal, defense and technology services. The company is based in Worcester, Massachusetts.
Market Opportunity
An analysis from ReportLinker states that the global cybersecurity market is expected to grow from an estimated value of $173.5 billion in 2022 to $266.2 billion by 2027, recording a CAGR of 8.9% for the period.
The increased number of data breaches worldwide, the ability of malicious actors to operate from anywhere in the world, the links between cyberspace and physical systems, and the difficulty of reducing vulnerabilities and consequences in complex cyber networks are some factors driving cyber security market growth, according to the report.
A lack of cybersecurity professionals and the budget constraints among SMBs and start-ups in developing economies are expected to hinder market growth. Cybercriminals are using automated techniques to attack SMBs’ networks to take advantage of their weak security infrastructures. To save money, time and resources, SMBs are seeking cybersecurity solutions.
Enclave
Enclave expands upon SideChannel’s cybersecurity service offerings by solving a pervasive network security problem with a simple tool.
A comprehensive cloud and network security solution, Enclave enables IT teams to contain breaches faster, reduce network outages, minimize latency and strengthen overall security defense.
Enclave creates the foundation for a Zero Trust network security model IT can build upon.
With Enclave, IT can easily segment their company’s network, organize personnel and computing devices at the employee workload level, and implement security controls across all network segments.
Enclave was designed and purpose built to serve the growing security needs of SMBs, a traditionally underserved market that is more prone to cyberattacks but has limited protection due to smaller budgets, inadequate IT security staffing and a lack of cybersecurity awareness among top executives.
Enclave is an affordable and effective network security solution that shrinks the attack surface area exposed to a cyber intruder and significantly reduces the amount of effort required to operate securely.
Management Team
Brian Haugli is CEO of SideChannel. He has led programs for the U.S. Department of Defense, the Pentagon, and Fortune 500 companies. He is an expert on National Institute of Standards and Technology guidance, threat intelligence implementations and strategic organizational initiatives. He is a professor at Boston College, Woods College of Advancing Studies Master’s Program in Cybersecurity. He is also a contributing author for the Wiley book ‘Cybersecurity Risk Management’.
Ryan Polk is CFO at SideChannel. He has been the principal of Perissos Partners, an executive consulting firm, since June 2017. He also served in executive roles in the portfolio companies owned by Lacy Diversified, with combined revenue approaching $2 billion. He served as the Vice President for Corporate Financial Planning and Analysis for Brightpoint, a publicly traded, Fortune 500 mobile device logistics company. He earned a bachelor’s degree in accounting and industrial management from Purdue University.
Nicholas Hnatiw is Chief Technology Officer at SideChannel. Prior to joining the company, he served as the technical director for network operations supporting U.S. Cyber Command, U.S. Intelligence Agencies and other Department of Defense research organizations. He was also the CEO of Loki Labs, a cyber security firm. He earned a bachelor’s degree in computer engineering and computer science at the University of Massachusetts, Amherst.
Bill Roberts is SideChannel’s CISO. He most recently served as the vice president, IS & CISO for Hologic Inc., a global medical device company, where he established cyber security and IT compliance programs. Prior to Hologic, he was vice president of information security for Cytyc Corporation, which was acquired by Hologic in 2007. At Cytyc, he managed global IT as the company grew from 140 employees to 1,500 and from $40 million in revenue to over $750 million.
SideChannel Inc. (OTCQB: SDCH), closed Thursday's trading session at $0.107811, even for the day. The average volume for the last 3 months is and the stock's 52-week low/high is $0.06/$0.18.
Recent News
- SideChannel Inc. (OTCQB: SDCH) - InvestorNewsBreaks - SideChannel Inc. (SDCH) Featured on 'Cyber for Hire' Podcast
- InvestorNewsBreaks - SideChannel Inc. (SDCH) CEO Shares Purposeful Approach to Security Strategy in Hacker Valley Podcast
- SideChannel Inc. (SDCH) YOY Revenue Growth Underscores Increasing Popularity of vCISO, Cybersecurity Software Services
Data443 Risk Mitigation Inc. (OTC: ATDS)
The QualityStocks Daily Newsletter would like to spotlight Data443 Risk Mitigation Inc. (OTC: ATDS).
•With over 500 million ransomware attacks in 1H2021, corporationshave been increasingly looking for ways to safeguard their internalIT systems
•The need for data protection has been further heightened byincreased regulatory scrutiny from foreign and domesticgovernmental agencies
•In the U.S., the recently passed Federal ADPPA privacy law willcoincide with California’s Consumer Privacy Act, with the latterset to come into effect on January 1, 2023
•With a broad toolkit focused on data security and management,Data443 have sought to capitalize on the expanding global need fordata protection services
Towards the beginning of May 2021, U.S. fuel prices started tounexpectedly spike. The Colonial Pipeline company, operator of asystemically crucial 5,500 mile long pipeline engaged intransporting fuel from the Gulf Coast to the New York metro areawas shut down, suspending the flow of over 100 million gallons offuel every day between Texas and New York (https://ibn.fm/YoGfq). In ensuing days, news would break that DarkSide, an emerginghacking consortium had held the company’s IT systems hostage –freezing its data and threatening to leak sensitive corporateinformation online. Colonial Pipeline would go on to pay a ransomof $2.3 million or 63.7 bitcoin at the time. Whilst dramatic innature, the Colonial Pipeline ransomware attack was just one ofnearly 500 million ransomware attacks taking place in the firsthalf of 2021, a clear illustration of the need for corporations toengage in more robust cyber security efforts going forward.Focusing in on data security and a privacy-forward methodology,
Data443 Risk Mitigation Inc. (OTC: ATDS) is a data security and privacy software company for ALL THINGS DATA SECURITY™. The company is committed to organizing the world’s information by identifying and protecting all sensitive data regardless of location, platform or format.
Data443 provides software and services to enable secure data across devices and databases – at rest and in transit – locally, on a network, or in the cloud. With over 10,000 customers in more than 100 countries, Data443 provides a modern approach to data governance and security. The company’s framework helps customers prioritize risk, identify security gaps, and implement effective data protection and privacy management strategies.
Data443 derives revenue primarily from contracts for subscriptions to access its SaaS platforms, and ancillary services provided in connection with its subscription services. In today’s ever-changing environment with unique and complex requirements for data privacy, governance and hybrid workforces, every organization needs to know where all their data is, who has access to it and how sensitive it is. Data443 provides the tools needed to give companies control over their data processing activities, with capabilities for identifying, reporting and migrating or deleting sensitive data.
The company is headquartered in Research Triangle Park, North Carolina.
Products
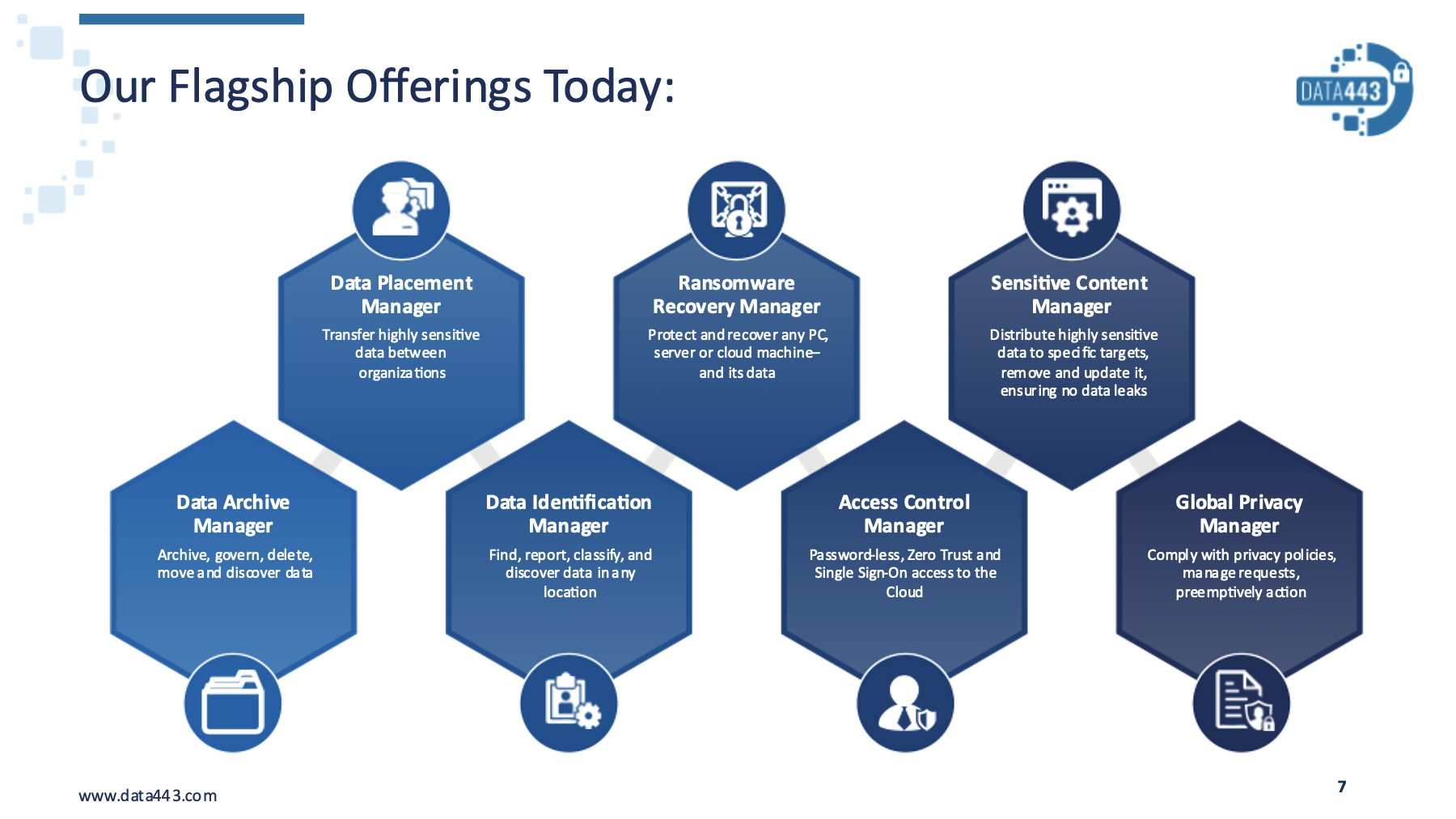
Focused on data security with a privacy-forward methodology, the Data443 product suite delivers solutions designed to securely manage data and data privacy needs on-premises, in the cloud and in hybrid environments. Offerings include:
- Data Identification Manager reduces risk by shining a light on dark data across cloud, on-premises and hybrid environments. From a centralized dashboard, Data Identification Manager provides the ability to automatically inventory all data repositories, classify and tag all data, and enable global search and discovery – all through an agentless deployment.
- Data Placement Manager quickly and securely transfers sensitive data over any public or private network. Available as an HP Nonstop server-based application and for Windows, Linux or any public cloud provider, Data Placement Manager enables the scheduling, routing, formatting and transfer of business-critical data.
- Data Archive Manager is an “all information, anywhere” archiving solution designed to handle and manage all types of privacy requests across cloud, on-premises and hybrid environments. With over 15 years operational history and hundreds of clients managing millions of mailboxes, the platform is purpose-built for information archiving, retention and privacy request management.
- Data Hound™ is a data discovery, classification and capture toolset that enables organizations to perform quick scans, detailed reporting and subsequent data actions based on policy.
- Ransomware Recovery Manager is the only industry solution that actively recovers the device, operating system and data with a simple reboot. Using patented, proven technology, the product produces 100% effectiveness for the whole device and datasets.
- Access Control Manager provides user ID and passwordless access to quickly enable trust across an organization’s entire ecosystem. Its unique architecture allows it to leverage multiple distributed authoritative sources to understand and resolve a typical access request – with the ability to enable or deny the action on the fly.
- Global Privacy Manager provides organizations one comprehensive view, for all privacy requirements, across all enterprise data, all at once. This unmatched visibility into an organization’s data assets ensures that all private and sensitive data can be identified and protected and that enterprises can obey all relevant privacy laws in any jurisdiction.
- Sensitive Content Manager is a security-centric collaboration service designed to give organizations the tools needed for successful content sharing, collaboration and safe distribution with full enterprise management in mind. With a continuous sync feature, encrypted data is automatically downloaded and updated in real time – regardless of location – ensuring that users have the most accurate data available.
Market Outlook
A report from Allied Market Research estimates that the global data security market was worth about $19 billion in 2021 and is projected to reach a value of $54.23 billion by 2027. That represents a CAGR of more than 18% for the forecast period, making data security one of the hottest areas within IT.
Separately, Fortune Business Insights estimates the global data privacy software market is valued at $2.36 billion in 2022 and projects it will grow to $25.85 billion by 2029. That represents a CAGR of 40.8% over the forecast period.
Management Team
Jason Remillard is President, CEO and Founder of Data443. He is responsible for overseeing global expansion, management, execution and corporate development. With over 25 years in global enterprise and B2C software sales and marketing, he brings deep leadership and technical experience, having spent previous time at Fortune 500 companies such as Deutsche Bank, TD Bank, IBM & Merrill Lynch.
Greg McCraw is CFO at Data443. He has over 25 years of experience helping businesses strengthen their accounting and finance operations. He previously served as Vice President of Finance for a dental services organization active in acquisitions, and, prior to that, he was managing director of a boutique accounting and finance consulting firm advising Fortune 500 clients in pharmaceutical, financial services, and private equity sectors on how to execute on regulatory and compliance solutions.
Bennett Pursell is Data443’s Chief Technology Officer. He has over 20 years of experience in IT architecture, security governance and systems integration. Prior to his role at Data443, he served as Head of Technology Architecture at Moody’s Investor Services and was Vice President and Technical Architect of Cloud Computing at Deutsche Bank, along with a host of technical and project management roles dating back to 2006, after starting his career as a web developer with a few startups and running research labs.
Kirill Kashigin is Chief Software Architect at Data443. He leads the development and quality teams, and serves as technical adviser and subject matter expert, bringing vast technical knowledge on privacy management and data security. Formerly the CTO of FileFacets, he has nearly 20 years in development of high-performance systems and deployment.
Data443 Risk Mitigation Inc. (OTC: ATDS), closed Thursday's trading session at $0.34, off by 15%, on 34,896 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.275/$6.99.
Recent News
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Seeks to Capitalize on Expanding Data Protection Market
- Data443 Risk Mitigation Inc. (ATDS) Lands $350,000 Add-On Contract, Closing 2022 on a High Note
- InvestorNewsBreaks - Why Data443 Risk Mitigation Inc. (ATDS) Is 'One to Watch'
India Globalization Capital Inc. (NYSE American: IGC)
The QualityStocks Daily Newsletter would like to spotlight India Globalization Capital Inc. (NYSE American: IGC).
After reformers encountered several defeats in December, SenateMajority Leader Chuck Schumer has pledged to continue workingtoward
India Globalization Capital Inc. (NYSE American: IGC), through subsidiary IGC Pharma, develops, patents, and markets advanced THC-based drug formulations for the treatment of symptoms related to various diseases including but not limited to Alzheimer’s disease, Tourette syndrome, chronic pain, and pet seizures.
IGC’s leading drug candidate, IGC-AD1, has completed Phase 1 of a safety and tolerability trial and entered Phase 2 trials for treating agitation in patients with Alzheimer’s dementia, the first study in humans of a natural tetrahydrocannabinol (THC) compound plus another molecule (www.clinicaltrials.gov). As of September 2022, the IGC trial is the only ongoing Phase 2 trial of a natural THC-based formulation on Alzheimer’s patients.
The company’s other drug candidate, TGR-63, is an enzyme inhibitor that has shown in preclinical trials the potential to reduce neurotoxicity in Alzheimer’s cell lines. Both drug candidates have shown their ability to ameliorate beta amyloid plaques in Alzheimer’s cell lines and improve memory in Alzheimer’s mouse models. Beta amyloid plaques are a key hallmark of Alzheimer’s and an important target of Alzheimer’s pharmaceutical drug development.
Neuro Psychiatric Symptoms (NPS) are not only debilitating for Alzheimer’s patients; they also place an immense emotional burden on their caregivers. Beyond reducing symptoms, IGC-AD1’s active molecules and TGR-63 have also shown promise in preclinical trials to reduce important hallmarks of Alzheimer’s including plaques and tangles, as well as improving the treatment of memory loss.
Over the past eight years, the IGC team has amassed a deep knowledge of cannabinoid science, including extraction, isolation, purification, and development. The company’s strategy is to leverage its unique end-to-end capabilities, platform, and expertise to develop a class-leading program and bring it to market quickly and cost efficiently to treat neurodegenerative diseases such as Alzheimer’s.
The company also has a family of cannabidiol (CBD)-based consumer products (www.Holief.com) such as pain relief creams, pain relief gels, purpose gummies, tinctures, and capsules targeting women’s wellness, with a particular focus on premenstrual syndrome (PMS) and dysmenorrhea (period cramps). In addition, the company targets individuals that need sleep-aids with its specially formulated low melatonin cannabinoid gummies.
IGC has also introduced a low-calorie CBD- and caffeine-infused energy beverage brand (www.SundaySeltzer.com) that is currently available for purchase. The company’s brands are founded on the belief that effective natural solutions should be affordable and accessible to everyone. As the demand for natural products targeting women’s wellness and energy drinks continue to grow, these products are seeing strong traction in the market.
The company operates three facilities – a large GMP (Good Manufacturing Production Standards) certified facility that includes extraction, distillation, and manufacturing, in Washington State; a GMP-211 (pharmaceutical) grade facility in Maryland; and a facility licensed for controlled substances including cannabis in Bogota, Colombia, with complete access to legal licensed cannabis where the company conducts its testing.
In addition, the company’s development under Magistral Formulations is approved by INVIMA (Colombia National Food and Drug Surveillance Institute) to treat neurological disorders, non-oncological chronic pain, and mental disorders.
IGC’s intellectual property (IP) portfolio comprises of eight patents that it controls and seven patent applications. The portfolio includes #11,446,276, a patent for extreme low dose THC treatment of Alzheimer’s that was granted in September 2022.
The company is headquartered in Potomac, Maryland.
IGC-AD1
IGC-AD1 is the company’s leading drug candidate for the treatment and relief of Alzheimer’s symptoms. A significant amount of research on Alzheimer’s cell lines has shown that the active agents in IGC-AD1 reduce plaques and neurofibrillary tangles that are the hallmarks of Alzheimer’s. Further, micro-dosing of THC, as shown in cell lines, could increase the functioning of mitochondria and potentially promote the growth of new neural pathways (neurogenesis). The research shows that micro-dosing of THC affects the brain radically differently from the normal higher dosing of THC.
While there is a significant body of research showing that THC is neuro-toxic at normal levels of dosing, micro-dosing of THC has been shown to be non-toxic to neurons. With the results of these preclinical studies, the company developed an oral formulation, IGC-AD1. The company recently completed a safety and tolerability Phase 1 trial on Alzheimer’s patients and has initiated a Phase 2, multi-site, double-blind, randomized, placebo-controlled trial of the safety and efficacy of IGC-AD1 on agitation in participants with dementia due to Alzheimer’s disease at sites in the U.S. and Canada. IGC expects the Phase 2 trial to take between 9 and 12 months to complete, barring unknown factors such as, for example, a resurgence of COVID and the enforcement of lockdowns and travel restrictions.
With further successful trials and FDA approvals, IGC hopes to bring a drug based on natural THC as an effective treatment for agitation in Alzheimer’s to market.
TGR-63
The company’s other molecule, TGR-63, has been shown to reduce the neurotoxicity that impacts memory loss in preclinical trials with mice. On a dose dependent manner, transgenic Alzheimer’s mice treated with TGR-63 showed improvement in memory relative to control.
Both drug candidates, IGC-AD1 and TGR-63, have shown their ability to reduce the brain plaques associated with memory loss in Alzheimer’s in mice.
With further successful trials and FDA approvals, IGC hopes to bring TGR-63 as a treatment for Alzheimer’s disease to market.
Market Opportunity
Alzheimer’s disease impacts over 55 million people worldwide and about 5.5 million individuals in the U.S. Over 70% of these patients face debilitating symptoms, including anxiety, depression, and agitation (Mendez, 2021). Agitation in dementia patients can include excessive physical movement and verbal activity, restlessness, pacing, belligerence, aggression, screaming, crying, and wandering.
In 2020, the estimated healthcare costs for Alzheimer’s disease in the U.S. were $305 billion. Medicare and Medicaid covered about 70% of those costs, leaving considerable burden on patients and families. At the current rate of growth of Alzheimer’s and other dementia diagnoses, those costs are estimated to reach over $1 trillion by 2050.
Currently, there are no FDA-approved medications to alleviate the symptoms of dementia due to Alzheimer’s disease, providing a tremendous opportunity for formulations that can have an impact on quality of life and disease progression.
Management Team
Richard Prins has been chairman at IGC since 2012 and served as an independent director since 2007. From March 1996 to 2008, he was the Director of Investment Banking at Ferris, Baker Watts, Incorporated. Prins served in a consulting role to RBC until January 2009. He currently volunteers full time with a non-profit organization, Advancing Native Missions, and is a private investor. Since February 2003, he has been on the board of Amphastar Pharmaceuticals Inc. He holds a bachelor’s degree from Colgate University and an MBA from Oral Roberts University.
Ram Mukunda is CEO and President of IGC. He has been the chief inventor and architect of most of the company’s patent filings and is responsible for the company’s strategic positioning. Prior to IGC, he was founder and CEO of Startec Global Communications, which he took public in 1997. He served as Strategic Planning Advisor at Intelsat, a communications satellite services provider. From 2001 to 2003, he was a Council Member at Harvard’s Kennedy School of Government, Belfer Center of Science and International Affairs. He was named the 1998 Ernst & Young Entrepreneur of the Year. He holds bachelor’s degrees in electrical engineering and mathematics, and a master’s degree in engineering from the University of Maryland.
Dr. Jagadeesh Rao is the company’s Principal Scientist. His career spans two decades in the public sector and product R&D for Johnson & Johnson. He leads IGC’s scientists in the development of pharmaceutical and OTC products. He worked for the federal National Institutes of Health, and for the National Institute on Drug Abuse. His Ph.D. in Neurochemistry is from the National Institute of Mental Health & Neurosciences in India. He did postdoctoral training at the University of Illinois-Chicago.
Claudia Grimaldi is a Director, Vice President, Principal Financial Officer, and Chief Compliance Officer for IGC. She also serves as a Director/Manager Director for some of the company’s subsidiaries. She graduated with highest honors from Javeriana University in Colombia with a bachelor’s degree in psychology. She holds an MBA, graduating with highest honors, from Meredith College in North Carolina. In addition, she has attended the Darden School of Business Financial Management Executives program and the Corporate Governance Program at Columbia Business School. She is currently pursuing her Directorship Certification with the National Association of Corporate Directors. She is fluent in both English and Spanish.
India Globalization Capital Inc. (NYSE American: IGC), closed Thursday's trading session at $0.3098, off by 10.6947%, on 577,236 volume. The average volume for the last 3 months is 577,236 and the stock's 52-week low/high is $0.2785/$1.16.
Recent News
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - Schumer Promises to Fight for Federal Cannabis Decriminalization in 2023
- InvestorNewsBreaks - India Globalization Capital Inc. (NYSE American: IGC) Set to Begin IGC-AD1 Phase 2 Trials at Sites Throughout Canada
- 420 with CNW - Kentucky Prepares to Implement Medical Cannabis Executive Order
Flora Growth Corp. (NASDAQ: FLGC)
The QualityStocks Daily Newsletter would like to spotlight Flora Growth Corp. (NASDAQ: FLGC).
To view the full press release, visit https://ibn.fm/QOlgi
Flora Growth Corp. (NASDAQ: FLGC) is an internationally focused cannabis brand builder that leverages natural, cost-effective cultivation practices to supply cannabis derivatives to its diverse business divisions, including cosmetics, hemp textiles, and food and beverage. Flora Growth operates one of the largest outdoor cultivation facilities in the world with an aim of marketing a higher-quality premium product at below-market prices. By prioritizing natural ingredients and value-chain sustainability across its portfolio, the company creates premium products that help consumers restore and thrive.
Flora Growth completed the first traditional cannabis IPO on Nasdaq in May 2021. Although currently headquartered in Toronto, Ontario, with plans to relocate its head office to Miami, Florida, the company’s base of operations is in Colombia, where it has built an extensive distribution network that includes Colombia’s largest distributors.
Currently, Flora Growth is organically growing market share for its existing brand portfolio (pharmaceuticals, textiles, cosmetics, and food & beverage) while seeking revenue-generating acquisitions that offer an accretive distribution network to amplify revenue growth.
Existing Brand & Product Portfolio
Flora Growth’s portfolio spans a number of verticals – each with a thoughtful brand designed to resonate with its intended end consumer. In line with the company’s mission, each brand prioritizes natural ingredients and value-chain sustainability.
Flora Lab S.A.S
Flora Lab is the company’s GMP certified manufacturing and R&D center focused on producing pharmaceuticals, cosmetics, and nutraceuticals for domestic and international markets. Its offerings include product lines that are private label, white-label, and custom formulas.
Through Flora Lab, Flora Growth has relationships with 1,500+ distribution channels, manufactures 63+ OTC products registered with INVIMA (Colombia National Food and Drug Surveillance Institute), and holds multiple GMP certifications enabling international export in an effort to leverage Flora Lab’s capacity to produce a wide range of CBD-infused products.
Flora Beauty
Flora Beauty is the company’s CBD beauty and cosmetics division founded by fashion and beauty industry icon Paulina Vega. Its current offerings include two CBD skincare brands targeting the U.S. and Latin American markets – MIND NATURALS and AWE. These lines exemplify Flora Growth’s socially conscious approach to business.
Currently, Flora Beauty products are offered globally through e-commerce, as well as through Falabella’s 111 retail locations across Latin America. The company is in negotiations with major department stores to launch the line in the U.S. and is also exploring opportunities in the U.K. and other European markets.

KASA Wholefoods
KASA Wholefoods is a Colombian manufacturer of food and beverages leveraging responsibly sourced exotic fruits from the Amazon. KASA has a $10 million+ distribution agreement with Tropi, Colombia’s largest food distributor, which has 130,000+ distribution points across the country.
Mambe, KASA’s leading brand, is already offered through over 980 distribution points across Colombia. Flora Growth expects this network to grow to over 1,200 distribution points in 2021, including one of Colombia’s largest coffee chains, Tostao Café & Pan.

Hemp Textiles & Co.
Through its Hemp Textiles division, Flora Growth intends to utilize its large land package and cultivation infrastructure to capture market share in the rapidly growing hemp industrials segment.
The company’s first brand through this division, Stardog Loungewear, offers a line of comfortable loungewear made from natural, organic materials. Stardog has been distributing globally through e-commerce and brick and mortar channels in Bogota since fall 2020, and the company intends to open U.S. brick and mortar locations in 2021.

Accretive M&A
Flora Growth is targeting transactions to complete the supply chain via key infrastructure to enhance its global distribution with the aim to compete on low-cost, high-quality inputs paired with premium brands that create business lines with robust margins.
To date, Flora has announced two major transactions.
Koch & Gsell (Acquisition)
- Amplify CPG portfolio’s revenue growth through leading brand, Heimat, currently with TTM revenues of $7.6 million.
- Leverage Koch &Gsell’s distribution network of 2,500+ stores to introduce Flora to the Swiss, European and Asian markets.
- Bring patented hemp cigarette manufacturing technology into new markets utilizing Flora’s high-quality cannabis.
Hoshi International (Investment)
- Equity Investment of €2 million into Hoshi to establish Flora as a preferred supplier to two EU processing facilities.
- Opens gateway for Flora Growth’s cannabis through international distribution agreements in the EU and U.K.
- Hoshi’s experienced team and increased access to the EU cannabis market to serve as a catalyst for revenue growth.
Cultivation
Key to Flora Growth’s expansion efforts is its cultivation strategy. The company’s Cosechemos farm, located in Bucaramanga, Colombia, is currently licensed to cultivate 247 acres of cannabis. Through three successful pilot crop plantings, the location has demonstrated a production cost of just $0.06/gram. For comparison, the average cost of North American cannabis (based on 2019 figures from Aphria, Tilray, Sundial, and Aurora) equates to roughly $1.89/gram.
Flora Growth is uniquely positioned to capitalize on Colombia’s favorable growing conditions, low-cost infrastructure, and affordable local workforce as it looks to ramp up its cultivation efforts moving forward.

Leadership Team
Bernard Wilson is the Chairman of Flora Growth. A senior financial professional, Dr. Wilson is the former Vice-Chairman of PricewaterhouseCoopers LLP and is the Chairman of the Founders Board of the Institute of Corporate Directors. He has also served as Chairman of the Canadian Chamber of Commerce; Chairman of the International Chamber of Commerce – Canada; and Member of the Canada/U.S. Trade Committee. Dr. Wilson draws on this experience to ensure Flora Growth adheres to effective corporate governance practices.
Luis Merchan is the company’s President and CEO. He is a proven executive with over a decade of experience in enterprise sales management, corporate strategy, merchandising and expense management, and customer experience. Mr. Merchan previously served as Macy’s Inc.’s Vice President of Workforce Strategy and Operations, where he managed the enterprise’s multi-billion-dollar P&L expense line for the entire 540 store portfolio. Throughout his tenure at Macy’s, he led various sales and marketing initiatives, including the B2B corporate sales team that was responsible for $160 million in annual revenue. Mr. Merchan obtained his Bachelor of Industrial Engineering from Pontifical Xaverian University in Bogota, Colombia, and his MBA from McNeese State University. He also holds a Graduate Certificate in Marketing Management from Harvard.
Juan Manuel Galan is a Strategic Advisor to the Flora Growth management team. Mr. Galan currently serves as a senior consultant to The World Bank. He is a politician and former senator of Colombia, serving three terms from 2006 to 2018 as a member of the Colombian Liberal Party. He is also a former professor at the University of Rosario and holds more than 20 years of journalistic, academic, governmental and parliamentary experience. During his time as a senator, Mr. Galan was a key leader, with 29 bills and 27 debates on political control, and 17 laws to his name. The most relevant of those laws was authoring the medical cannabis law that resulted in the legalization of medical cannabis in Colombia.
Stan Bharti is a Director of Flora Growth. Mr. Bharti currently serves as Executive Chairman of Forbes & Manhattan. He has more than 30 years of professional experience in business, finance, markets, operations and more, with a focus on the resource and technology sectors. To date, Mr. Bharti has amassed over $3 billion worth of investment capital for the companies with which he has worked and their shareholders. He is a Professional Mining Engineer and holds a master’s degree in engineering from Moscow, Russia, and University of London, England.
Javier Franco is the company’s VP of Agriculture. Mr. Franco is a master horticulturist with more than 25 years of experience in the design, implementation, and management of cultivation and propagation facilities of more than 30 species of cut flowers in Latin America. He completed his agricultural studies at Zamorano University in Honduras and later at an International Exchange Program at Ohio State University. Mr. Franco has directed technical, commercial, and research groups in the cut flower, fruit and vegetable markets in Latin America and has participated in the commercial development of new technologies applied in agribusiness. He has also led the agri-management of organic crops and certifications of Good Agricultural Practices.
Flora Growth Corp. (FLGC), closed Thursday's trading session at $0.2484, off by 0.64%, on 547,227 volume. The average volume for the last 3 months is 545,297 and the stock's 52-week low/high is $0.21/$2.38.
Recent News
- Flora Growth Corp. (NASDAQ: FLGC) - InvestorNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Releases Status Update on Commercial Cannabis Export Business
- 420 with CNW - Study Finds Marijuana Legalization Has Greater Impact on Arrests Than Decriminalization
- 420 with CNW - Survey Finds Prohibitionist States Have Higher Cases of Underage Marijuana Use
Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F)
The QualityStocks Daily Newsletter would like to spotlight Hillcrest Energy Technologies Ltd. (OTCQB: HLRTF).
•Hillcrest Energy began 2022 with proof-of-concept of its innovativetraction inverter technology that overcomes shortfalls in today’stech and improves EV motor efficiency
•The company ended the year with its first 800-volt 250-kilowattcommercial prototype that has attracted the attention of leading EVmakers and Tier 1 automotive suppliers
•Hillcrest announced two co-development partners and is now workingwith them both to integrate the ZVS traction inverter into theirspecific commercial applications
As the new year arrives, it is a commonplace for companies andinvestors to reflect upon the last 12 months and hypothesize aboutwhere the next year will lead. In the EV space, 2022 will likely beremembered as the start of a steep adoption trajectory as moremakes and models hit the roads. In that lane, the year was full ofimportant milestones for
Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) is a clean technology company based in Vancouver, British Columbia, engaged in developing high-value, high-performance power conversion technologies and digital control systems for next-generation powertrains and grid-connected renewable energy systems.
From concept to commercialization, Hillcrest invests in the development of energy solutions that power a more sustainable and electrified future. Hillcrest power inverter technology helps produce efficiencies in electrification and maximize the performance of electric systems, including electric vehicles (EV), motors and generators.
The company offers a flexible, single-inverter architecture that can be applied at nearly every stage of the electrification ecosystem, from renewable energy generation through the charging and operation of an EV, to provide full-cycle efficiency and performance improvements.
As momentum to electrify and decarbonize energy systems accelerates, Hillcrest believes the power inverter is increasingly emerging as a key component. While system cohorts such as battery packs, PV panels and electric motors are often in the spotlight, the inverter holds the key to unlocking efficiency and performance improvements.
Hillcrest power inverter technology is:
- REVOLUTIONARY: high-efficiency inverter technology has the potential to revolutionize how motors respond and how efficiency is gained.
- AGILE: able to deliver and deploy high-efficiency inverter solutions purpose-designed to meet specific customer needs.
- INNOVATIVE: technology-forward, clean-energy experts who are focused on advancing and optimizing efficient alternative energy use across all electric vehicle and charging platforms.
- A MARKET LEADER: a next-generation technology provider to the automotive industry’s top suppliers and manufacturers.
Technology & Applications
Hillcrest’s first application for its inverter technology – a 250 kW|800V Hillcrest SiC high efficiency traction inverter – is focused on the growing EV market. Hillcrest technology eliminates traditional design trade-offs faced across the power industry – deploying higher switching frequencies has historically meant a greater increase in losses, lower system efficiency and higher heat. Through a combination of hardware and software expertise, Hillcrest enables power applications to leverage higher switching frequencies AND
- Realize improved power system performance and reliability
- Operate at higher power levels without compromising efficiency
The expected benefits of Hillcrest’s traction inverter have been confirmed via testing and shared in a technical white paper, published in April 2022, that confirmed the following results:
- Significant efficiency gains – 99%-plus inverter efficiency
- Increased power density targeting 50kW/L+
- Significantly increased motor efficiency
- Lower stress on mechanical and electrical parts, enhancing reliability
- Improved thermal management
Hillcrest has also filed a patent for an enhanced powertrain solution that offers the potential to simplify EV charging and redefine how the industry envisions charging infrastructure. The company believes the most exciting benefit of the enhanced powertrain solution is the ability to eliminate the onboard charger and booster from an EV, as well as faster, anywhere charging including direct DC, wireless, and bidirectional charging across current and future power levels. Hillcrest sees this as a true EV charging game changer.
The company’s technology applies to nearly every clean energy industry segment:
- Wind power – an inverter is deployed at a wind turbine generator to convert the AC output, with at least one additional inverter used to deliver the power to the grid/battery.
- Solar power – an inverter is used to convert the DC output from the photovoltaic panels into the AC power that flows to the grid/battery/home.
- Energy storage – an inverter is deployed to convert the DC output from the storage system or batteries to the AC power that flows to the grid/home/EV.
- EV fast chargers – an inverter converts the AC input from the grid/storage system to the DC output needed to charge an EV’s battery.
Market Outlook
According to an April 2022 market analysis by Vantage Market Research (VMR), the global power inverter market is expected to reach a value of $95 billion by 2028, driven by increasing demand for EVs, energy generating wind turbines and solar-powered photovoltaic systems. That jump is forecast from an estimated $70.5 billion market value in 2021 and represents a compound annual growth rate of more than 5%.
According to the VMR report, many governments in countries around the world are supporting alternative options for efficient and nonpolluting energy generation. This has boosted demand for wind energy and solar energy systems. Hillcrest is aiming to capture a share of this future market growth across nearly every segment of the clean energy industry.
Management Team
Don Currie is the founding CEO of Hillcrest Energy Technologies. He has led the company’s successful transition from fossil fuels into clean energy technologies. Earlier in his career, he held various senior level positions, including director, officer and vice president of corporate communications with Enhanced Oil Resources Inc., an oil and gas exploration and production company based in Houston. Prior to that, he worked in other private and public ventures spanning the mining, gaming and technology sectors.
Jamie L. Hogue is the COO of Hillcrest. She brings more than two decades of progressive policy leadership, economic analysis and organizational development experience to Hillcrest. She builds collaborative processes and solutions that drive growing organizations toward a more resilient future. She previously served as the director of operations for Arizona State University’s Ten Across initiative – a compelling observatory positioned on the front lines of economic, social and climate change. She earned a master’s degree in public administration and a bachelor’s degree in economics from Arizona State University.
Ari Berger is Chief Technology Officer at Hillcrest. He brings over a decade of commercial experience with a track record of deploying new electrification technologies and go-to-market strategies. In 2015, he founded NIG Systems Ltd. in Israel, which specializes in custom high performance control systems design. Prior to this, he previously worked for Bental Industries, a leading motor manufacturer. He holds a master’s degree in system control engineering from the Technion – Israel Institute of Technology.
Raj Clair is CFO at Hillcrest. She is a CPA who began her career at Deloitte and has served in advanced finance positions in the energy and resources sector. She has been responsible for reporting, audits and internal controls, as well as working on budgeting and forecasting. She has worked with various publicly listed companies, including SEC registrants, and has both Canadian and U.S. experience. She holds a bachelor’s degree in accounting from Simon Fraser University.
Hillcrest Energy Technologies Ltd. (NASDAQ: HLRTF), closed Thursday's trading session at $0.0725, off by 7.9949%, on 21,145 volume. The average volume for the last 3 months is 21,145 and the stock's 52-week low/high is $0.0682/$0.1512.
Recent News
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) at Inflection Point with Cutting-Edge Inverter Prototype
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Eyes Entry into Grid-Tied Applications; Continues Development of Its Enhanced Powertrain Solution
- InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Recaps 2022 as a Tremendous Year of Technology and Milestones
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Is Notified by NASDAQ Exchange of Full Compliance
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Forms Subsidiary with Focus on Immune Modulation Therapies
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Minnesota Medical Association Backs Statewide Drug Decriminalization
- Amesite Inc. (NASDAQ: AMST) - NASDAQ: $AMST Amesite Announces Response Regarding Potential Semiconductor Manufacturing Institutes
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Releases Report Noting 48.8% Decrease in 2022 Net Loss
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Taps into Arizona's Rich Mining History
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix to Attend the Benchmark 2022 Discovery One-on-One Investor Conference
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Announces Participation in Upcoming Biotech Showcase
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - Brain Scientific Signs Agreement with MVAP Medical Supplies to Go After Rapidly Growing Pediatric Neurodiagnostic Space
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corporation (NASDAQ: CISO) Announces Corporate Rebrand to Reflect Global Organization
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Four Developments in Cancer Research That Occurred in 2022
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Encouraged by Growing Government Moves to Reduce Emissions
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Poised to Provide Greater Access to Sustainable Living
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc.’s Distinctiveness in Backcountry Aviation Increases Demand for Its Aircraft
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) CEO Highlights 2022 as 'Momentous Year'
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Validates Position as Leading Provider of Integrated Solutions in Employer and Payer Markets
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Seeks to Capitalize on Expanding Data Protection Market
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave and Davidson Technologies Enter Multi-Year Reseller Agreement
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Schools Step Up Training for Psychedelic Therapy Professionals
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Subsidiary Shares Statement on 2023 Golf Cart, LSV Markets
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Analysts Discuss Gold 2023 Outlook
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Three US Firms Win Contracts to Supply Uranium Strategic Reserve
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - ESGBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) at Forefront to Supply North American Grid with Clean Energy
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces Major Project and Corporate Milestone
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Looks for Strong Numbers in Upcoming Quarter
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Announces Acquisition to Expand of Quebec Lithium Base
- Flora Growth Corp. (NASDAQ: FLGC) - InvestorNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Releases Status Update on Commercial Cannabis Export Business
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Unveils Virtual Sensor for Auto Industry at CES 2023
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Stream of Steady Business on Fr8App Platform
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Positioned to Benefit from Electrification as Rising Natural Gas Prices Show No Sign of Slowing Down
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Exclusive Distribution Agreement
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - Green Hygienics Holdings Inc. Provides Corporate Update
- RYVYL Inc. (NASDAQ: GBOX) - RYVYL Inc Issues Shareholder Letter
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Receives Key Patent from European Patent Office
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Unveils New Lidar That Embodies Auto Industry's Biggest Trends, Next-Gen Innovations
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) at Inflection Point with Cutting-Edge Inverter Prototype
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces US$5 Million Share Repurchase Program
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Expanded Collaboration for Hydrogen Conversion of Port Drayage Operations
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - Schumer Promises to Fight for Federal Cannabis Decriminalization in 2023
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks – InMed Pharmaceuticals Inc. (NASDAQ: INM) Shares Annual General Meeting Results
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Jupiter Wellness Announces Redeemable Digital Coupon Awarded to all Shareholders of Record
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) to Deploy Seven ASRs for Multiple New Contracts
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Dosing Begins in Lexaria Bioscience Corp. (NASDAQ: LEXX) Human Clinical Study, Evaluating DehydraTECH(TM)-Nicotine Against Leading Nicotine Oral Pouch Brands
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - InvestorNewsBreaks - MetAlert Inc. (MLRT) Issues Shareholder Update
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - Mind Cure Health to Acquire South American Utility Company
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Closes Sale of Mindleap Health Inc.
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) CEO to Present at Planet MicroCap Showcase: Virtual 2022
- Mullen Automotive Inc. (NASDAQ: MULN) - Why Mullen (MULN) Stock Is Shaking Off Tesla's EV Plunge TodayMore From InvestorPlace
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) Files Key Interim Financial Documents
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Anticipates Completion of Private Placement of Unsecured Convertible Debentures, Shares
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Issues Corporate Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Expands Scientific Advisory Board to Appoint Genomics Expert
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Releases Q4 2022 Guidance, Projections for 2023
- Prime Harvest Inc. - Prime Harvest Inc. Positioned to Lead California’s Cannabis Market with Direct-to-Consumer Expansion
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - New Treatment Approach for Cancer Combines Bacterial, Pharmaceutical Therapy
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Board Votes to Eliminate Millions in Debt
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Announces New Financing Round; Looking to Raise up to $1 Million
- REZYFi, Inc. - 420 with CNW — SAFE Banking Act Sponsor Retires from Congress
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - SEDAR filing of the unaudited Interim Financial Statements to March 31, and June 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Inc. Joins Digital DJ Pool
- Save Foods Inc. (NASDAQ: SVFD) - This Company Anticipates Massive Results As It Works To Address Food Waste, Loss And Food Safety Challenges
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks – Sharing Services Global Corporation’s (SHRG) Hapi Travel Destinations Providing Max Savings, Travel Perks
- SideChannel Inc. (OTCQB: SDCH) - InvestorNewsBreaks - SideChannel Inc. (SDCH) Featured on 'Cyber for Hire' Podcast
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Milestone in IND-Enabling Study of Time-Released Ketamine Formulation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Adds UK, Ireland Supplier to Distribution Network
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Issues Corporate Update
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Receives Notice of Allowance for Final US Patent Covering DLE Technology
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) CEO Presents at Global Unity Summit & Gala
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Released End-of-Year Status Update
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces New DSD Partnerships, Expanded Distribution for Alkaline88(R)
- Tingo Inc. (OTC: TMNA) - Four Possible Crypto Trends to Watch in 2023
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Wins Strategic Uranium Reserve Order from DOE
- Vision Energy Corp. (OTCQB: VENG) - Vision Energy Corp. (VENG) Expands Project Management and Development Team, Advancing Green Energy Hub Project in North Sea Port of Vlissingen
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Takes Key Steps to Protect, Enhance IP Program
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Enters into Private Placement Agreement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Technology Acquisition and Partnership with Lumeto, Inc. to Expand its Wrap Reality Training Platform
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Is Notified by NASDAQ Exchange of Full Compliance
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Forms Subsidiary with Focus on Immune Modulation Therapies
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Minnesota Medical Association Backs Statewide Drug Decriminalization
- Amesite Inc. (NASDAQ: AMST) - NASDAQ: $AMST Amesite Announces Response Regarding Potential Semiconductor Manufacturing Institutes
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Releases Report Noting 48.8% Decrease in 2022 Net Loss
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Taps into Arizona's Rich Mining History
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix to Attend the Benchmark 2022 Discovery One-on-One Investor Conference
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Announces Participation in Upcoming Biotech Showcase
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - Brain Scientific Signs Agreement with MVAP Medical Supplies to Go After Rapidly Growing Pediatric Neurodiagnostic Space
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corporation (NASDAQ: CISO) Announces Corporate Rebrand to Reflect Global Organization
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Four Developments in Cancer Research That Occurred in 2022
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Encouraged by Growing Government Moves to Reduce Emissions
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Poised to Provide Greater Access to Sustainable Living
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc.’s Distinctiveness in Backcountry Aviation Increases Demand for Its Aircraft
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) CEO Highlights 2022 as 'Momentous Year'
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Validates Position as Leading Provider of Integrated Solutions in Employer and Payer Markets
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Seeks to Capitalize on Expanding Data Protection Market
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave and Davidson Technologies Enter Multi-Year Reseller Agreement
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Schools Step Up Training for Psychedelic Therapy Professionals
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Subsidiary Shares Statement on 2023 Golf Cart, LSV Markets
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Analysts Discuss Gold 2023 Outlook
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Three US Firms Win Contracts to Supply Uranium Strategic Reserve
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - ESGBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) at Forefront to Supply North American Grid with Clean Energy
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces Major Project and Corporate Milestone
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Looks for Strong Numbers in Upcoming Quarter
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Announces Acquisition to Expand of Quebec Lithium Base
- Flora Growth Corp. (NASDAQ: FLGC) - InvestorNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Releases Status Update on Commercial Cannabis Export Business
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Unveils Virtual Sensor for Auto Industry at CES 2023
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Stream of Steady Business on Fr8App Platform
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Positioned to Benefit from Electrification as Rising Natural Gas Prices Show No Sign of Slowing Down
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Exclusive Distribution Agreement
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - Green Hygienics Holdings Inc. Provides Corporate Update
- RYVYL Inc. (NASDAQ: GBOX) - RYVYL Inc Issues Shareholder Letter
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Receives Key Patent from European Patent Office
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Unveils New Lidar That Embodies Auto Industry's Biggest Trends, Next-Gen Innovations
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) at Inflection Point with Cutting-Edge Inverter Prototype
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces US$5 Million Share Repurchase Program
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Expanded Collaboration for Hydrogen Conversion of Port Drayage Operations
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - Schumer Promises to Fight for Federal Cannabis Decriminalization in 2023
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks – InMed Pharmaceuticals Inc. (NASDAQ: INM) Shares Annual General Meeting Results
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Jupiter Wellness Announces Redeemable Digital Coupon Awarded to all Shareholders of Record
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) to Deploy Seven ASRs for Multiple New Contracts
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Dosing Begins in Lexaria Bioscience Corp. (NASDAQ: LEXX) Human Clinical Study, Evaluating DehydraTECH(TM)-Nicotine Against Leading Nicotine Oral Pouch Brands
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - InvestorNewsBreaks - MetAlert Inc. (MLRT) Issues Shareholder Update
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - Mind Cure Health to Acquire South American Utility Company
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Closes Sale of Mindleap Health Inc.
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) CEO to Present at Planet MicroCap Showcase: Virtual 2022
- Mullen Automotive Inc. (NASDAQ: MULN) - Why Mullen (MULN) Stock Is Shaking Off Tesla's EV Plunge TodayMore From InvestorPlace
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) Files Key Interim Financial Documents
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Anticipates Completion of Private Placement of Unsecured Convertible Debentures, Shares
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Issues Corporate Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Expands Scientific Advisory Board to Appoint Genomics Expert
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Releases Q4 2022 Guidance, Projections for 2023
- Prime Harvest Inc. - Prime Harvest Inc. Positioned to Lead California’s Cannabis Market with Direct-to-Consumer Expansion
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - New Treatment Approach for Cancer Combines Bacterial, Pharmaceutical Therapy
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Board Votes to Eliminate Millions in Debt
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Announces New Financing Round; Looking to Raise up to $1 Million
- REZYFi, Inc. - 420 with CNW — SAFE Banking Act Sponsor Retires from Congress
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - SEDAR filing of the unaudited Interim Financial Statements to March 31, and June 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Inc. Joins Digital DJ Pool
- Save Foods Inc. (NASDAQ: SVFD) - This Company Anticipates Massive Results As It Works To Address Food Waste, Loss And Food Safety Challenges
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks – Sharing Services Global Corporation’s (SHRG) Hapi Travel Destinations Providing Max Savings, Travel Perks
- SideChannel Inc. (OTCQB: SDCH) - InvestorNewsBreaks - SideChannel Inc. (SDCH) Featured on 'Cyber for Hire' Podcast
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Milestone in IND-Enabling Study of Time-Released Ketamine Formulation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Adds UK, Ireland Supplier to Distribution Network
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Issues Corporate Update
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Receives Notice of Allowance for Final US Patent Covering DLE Technology
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) CEO Presents at Global Unity Summit & Gala
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Released End-of-Year Status Update
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces New DSD Partnerships, Expanded Distribution for Alkaline88(R)
- Tingo Inc. (OTC: TMNA) - Four Possible Crypto Trends to Watch in 2023
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Wins Strategic Uranium Reserve Order from DOE
- Vision Energy Corp. (OTCQB: VENG) - Vision Energy Corp. (VENG) Expands Project Management and Development Team, Advancing Green Energy Hub Project in North Sea Port of Vlissingen
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Takes Key Steps to Protect, Enhance IP Program
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Enters into Private Placement Agreement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Technology Acquisition and Partnership with Lumeto, Inc. to Expand its Wrap Reality Training Platform
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Is Notified by NASDAQ Exchange of Full Compliance
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Forms Subsidiary with Focus on Immune Modulation Therapies
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Minnesota Medical Association Backs Statewide Drug Decriminalization
- Amesite Inc. (NASDAQ: AMST) - NASDAQ: $AMST Amesite Announces Response Regarding Potential Semiconductor Manufacturing Institutes
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Releases Report Noting 48.8% Decrease in 2022 Net Loss
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) Taps into Arizona's Rich Mining History
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix to Attend the Benchmark 2022 Discovery One-on-One Investor Conference
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Announces Participation in Upcoming Biotech Showcase
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - Brain Scientific Signs Agreement with MVAP Medical Supplies to Go After Rapidly Growing Pediatric Neurodiagnostic Space
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corporation (NASDAQ: CISO) Announces Corporate Rebrand to Reflect Global Organization
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Four Developments in Cancer Research That Occurred in 2022
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Encouraged by Growing Government Moves to Reduce Emissions
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Poised to Provide Greater Access to Sustainable Living
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc.’s Distinctiveness in Backcountry Aviation Increases Demand for Its Aircraft
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) CEO Highlights 2022 as 'Momentous Year'
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Validates Position as Leading Provider of Integrated Solutions in Employer and Payer Markets
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Seeks to Capitalize on Expanding Data Protection Market
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave and Davidson Technologies Enter Multi-Year Reseller Agreement
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Schools Step Up Training for Psychedelic Therapy Professionals
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Subsidiary Shares Statement on 2023 Golf Cart, LSV Markets
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Analysts Discuss Gold 2023 Outlook
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Three US Firms Win Contracts to Supply Uranium Strategic Reserve
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - ESGBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) at Forefront to Supply North American Grid with Clean Energy
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Announces Major Project and Corporate Milestone
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Looks for Strong Numbers in Upcoming Quarter
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Announces Acquisition to Expand of Quebec Lithium Base
- Flora Growth Corp. (NASDAQ: FLGC) - InvestorNewsBreaks - Flora Growth Corp. (NASDAQ: FLGC) Releases Status Update on Commercial Cannabis Export Business
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Unveils Virtual Sensor for Auto Industry at CES 2023
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Secures Stream of Steady Business on Fr8App Platform
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc. Positioned to Benefit from Electrification as Rising Natural Gas Prices Show No Sign of Slowing Down
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Announces Exclusive Distribution Agreement
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - Green Hygienics Holdings Inc. Provides Corporate Update
- RYVYL Inc. (NASDAQ: GBOX) - RYVYL Inc Issues Shareholder Letter
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Receives Key Patent from European Patent Office
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Unveils New Lidar That Embodies Auto Industry's Biggest Trends, Next-Gen Innovations
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) at Inflection Point with Cutting-Edge Inverter Prototype
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces US$5 Million Share Repurchase Program
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Announces Expanded Collaboration for Hydrogen Conversion of Port Drayage Operations
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - Schumer Promises to Fight for Federal Cannabis Decriminalization in 2023
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks – InMed Pharmaceuticals Inc. (NASDAQ: INM) Shares Annual General Meeting Results
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Jupiter Wellness Announces Redeemable Digital Coupon Awarded to all Shareholders of Record
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) to Deploy Seven ASRs for Multiple New Contracts
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Dosing Begins in Lexaria Bioscience Corp. (NASDAQ: LEXX) Human Clinical Study, Evaluating DehydraTECH(TM)-Nicotine Against Leading Nicotine Oral Pouch Brands
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - InvestorNewsBreaks - MetAlert Inc. (MLRT) Issues Shareholder Update
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - Mind Cure Health to Acquire South American Utility Company
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Closes Sale of Mindleap Health Inc.
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) CEO to Present at Planet MicroCap Showcase: Virtual 2022
- Mullen Automotive Inc. (NASDAQ: MULN) - Why Mullen (MULN) Stock Is Shaking Off Tesla's EV Plunge TodayMore From InvestorPlace
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) Files Key Interim Financial Documents
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Anticipates Completion of Private Placement of Unsecured Convertible Debentures, Shares
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Issues Corporate Update
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Expands Scientific Advisory Board to Appoint Genomics Expert
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Releases Q4 2022 Guidance, Projections for 2023
- Prime Harvest Inc. - Prime Harvest Inc. Positioned to Lead California’s Cannabis Market with Direct-to-Consumer Expansion
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - New Treatment Approach for Cancer Combines Bacterial, Pharmaceutical Therapy
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Board Votes to Eliminate Millions in Debt
- Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) - Reklaim Ltd. (TSX.V: MYID) (OTCQB: MYIDF) Announces New Financing Round; Looking to Raise up to $1 Million
- REZYFi, Inc. - 420 with CNW — SAFE Banking Act Sponsor Retires from Congress
- RYAH Group Inc. (CSE: RYAH) - RYAH GROUP INC. - SEDAR filing of the unaudited Interim Financial Statements to March 31, and June 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp.'s (SNWR) Intercept Music Inc. Joins Digital DJ Pool
- Save Foods Inc. (NASDAQ: SVFD) - This Company Anticipates Massive Results As It Works To Address Food Waste, Loss And Food Safety Challenges
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks – Sharing Services Global Corporation’s (SHRG) Hapi Travel Destinations Providing Max Savings, Travel Perks
- SideChannel Inc. (OTCQB: SDCH) - InvestorNewsBreaks - SideChannel Inc. (SDCH) Featured on 'Cyber for Hire' Podcast
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Milestone in IND-Enabling Study of Time-Released Ketamine Formulation
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Adds UK, Ireland Supplier to Distribution Network
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Issues Corporate Update
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Receives Notice of Allowance for Final US Patent Covering DLE Technology
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) CEO Presents at Global Unity Summit & Gala
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Released End-of-Year Status Update
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces New DSD Partnerships, Expanded Distribution for Alkaline88(R)
- Tingo Inc. (OTC: TMNA) - Four Possible Crypto Trends to Watch in 2023
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Wins Strategic Uranium Reserve Order from DOE
- Vision Energy Corp. (OTCQB: VENG) - Vision Energy Corp. (VENG) Expands Project Management and Development Team, Advancing Green Energy Hub Project in North Sea Port of Vlissingen
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Takes Key Steps to Protect, Enhance IP Program
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Enters into Private Placement Agreement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Technology Acquisition and Partnership with Lumeto, Inc. to Expand its Wrap Reality Training Platform
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































