The QualityStocks Daily Stock List
- Secoo Holding (SECO)
- SoundHound AI (SOUN)
- First Pacific (FPAFF)
- Ausnutria Dairy (AUDYF)
- Arafura Rare Earths (ARAFF)
- Sappi Ltd (SPPJY)
- Wesdome Gold Mines (WDOFF)
- Coinbase Global Inc. (COIN)
- Lordstown Motors Corp. (RIDE)
- Arch Resources Inc. (ARCH)
- Field Trip Health Ltd. (FTHWF)
- Sidus Space Inc. (SIDU)
Secoo Holding (SECO)
StreetInsider, StockMarketWatch, MarketBeat, InvestorPlace, Trading Concepts, The Stock Dork, StocksEarning, SmallCapVoice, QualityStocks, FreeRealTime and BUYINS.NET reported earlier on Secoo Holding (SECO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Secoo Holding Ltd. (NASDAQ: SECO) (FRA: 1S8) is an integrated offline and online shopping and upscale products and services platform.
The firm has its headquarters in Beijing, the People’s Republic of China and was incorporated in 2011, on January 4th by Jiang Xiang Xun, Zhao Hui Huang and Ri Xue Li. It operates as part of the internet retail industry, under the consumer cyclical sector. The firm has 697 companies in its corporate family and serves consumers around the word, with a primary focus on Hong Kong and China.
The company is party to a strategic alliance with Parkson Group. Its offerings can be accessed on its platform, which comprises of its Secoo.com website covering more than 3,800 domestic and international brands.
The enterprise offers upscale brand products, which include accessories, jewelry, footwear, clothing, watches, handbags, skincare and cosmetics, home goods, kitchen equipment, computer peripherals, cleaning supplies, sportswear, home accessories, beverages and fine food products, 3C electronic devices, arts and Chinese original products. It also provides various services, including lifestyle services, through offline experience centers, mobile applications and its website. The enterprise also provides its website as a marketplace to 3rd party merchants to facilitate their sale of upscale services and products.
The company remains committed to serving high-end individuals across the globe who are pursuing the ultimate life experience, through the provision of the latest trendy items. This not only brings in additional revenue into the company but also helps extend its reach, which is good for investments.
Secoo Holding (SECO), closed Monday's trading session at $3.77, up 124.4048%, on 80,440,769 volume. The average volume for the last 3 months is 96.671M and the stock's 52-week low/high is $1.50/$6.90.
SoundHound AI (SOUN)
MarketBeat, TipRanks, The Stock Dork, QualityStocks and InsiderTrades reported earlier on SoundHound AI (SOUN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
SoundHound AI Inc. (NASDAQ: SOUN) is a software firm that is focused on developing an independent voice artificial intelligence (AI) platform that enables businesses across industries to deliver conversational experiences to their consumers.
The firm has its headquarters in Santa Clara, California and was incorporated in 2005, on September 2nd. It operates as part of the software-application industry, under the technology sector. The firm serves consumers across the globe.
The company’s vision is to create a voice AI platform that exceeds human capabilities and brings delight as well as value through an ecosystem of billions of products enhanced by innovation and monetization opportunities.
The enterprise’s advanced voice AI platform, dubbed Houndify, is built on proprietary Speech-to-Meaning and Deep Meaning Understanding technologies and offers speed and accuracy while also allowing people to interact with products and services as they interact with each other; by speaking naturally. Through its Deep Meaning Understanding technology, the user's voice assistant addresses multiple questions and filters results simultaneously while its Speech-to-Meaning technology tracks speech in real-time and understands it, even before an individual finishes speaking. Its other products include its SoundHound App, Midomi.com and Hound App.
The company recently entered into an agreement with Harman to deliver effortless voice AI experience to the automotive market. This move not only facilitates the company’s entrance in a new market but will also open it up to new growth and investments opportunities that will in turn, create value for its shareholders.
SoundHound AI (SOUN), closed Monday's trading session at $4.03, up 42.9078%, on 96,671,156 volume. The average volume for the last 3 months is 10,000 and the stock's 52-week low/high is $0.93/$18.14.
First Pacific (FPAFF)
We reported earlier on First Pacific (FPAFF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
First Pacific Co. Ltd. (OTCMKTS: FPAFF) (HKG: 0142) (BMV: FPAFYN) (FRA: FPC) is an investment management and holding firm engaged in the consumer food products, infrastructure, telecommunications and natural resource businesses globally.
The firm has its headquarters in Central, Hong Kong and was incorporated in May 1981 by Manuel Velez Pangilinan. It operates as part of the packaged foods industry, under the consumer defensive sector. The firm serves consumers globally, with a focus on those in the Philippines, Indonesia, Singapore, Africa and the Middle East.
The company operates through the Telecommunications, Infrastructure, Consumer Food Products and Natural Resources segments. It generates maximum revenue from the consumer food products segment. Geographically, the company derives a majority of revenue from Indonesia.
The enterprise manufactures and distributes a range of food products, including noodles, dairy products, snack foods, food seasonings, nutrition and special foods, beverages, wheat flour, and pasta; breeds seeds; cultivates oil palm, rubber, sugar cane, and other crops; and offers cooking oils, margarines, and shortenings. It also provides a range of telecommunications and digital services, including fiber optic backbone, and fixed line and mobile networks. This is in addition to exploring for, mining and producing gold, copper, and silver; and producing sugar and ethanol. Further, the enterprise is involved in the operation of a gas-fired power plant; operation of toll roads, hospitals, and rail lines; provision of water distribution, sewerage, and sanitation services; and provision of logistics services.
The firm remains committed to delivering returns to its shareholders and making further investments in value-enhancing businesses. This will in turn open it up to new growth and investment opportunities.
First Pacific (FPAFF), closed Monday's trading session at $0.35033, up 0.467451%, on 10,000 volume. The average volume for the last 3 months is 1,000 and the stock's 52-week low/high is $0.2624/$0.4605.
Ausnutria Dairy (AUDYF)
We reported earlier on Ausnutria Dairy (AUDYF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Ausnutria Dairy Corp. Ltd (OTC: AUDYF) (HKG: 1717) (FRA: 6AH) is an investment holding firm that is focused on the research and development, milk collection, processing, production, packaging, marketing and distribution of infant formula and other dairy products.
The firm has its headquarters in Sheung Wan, Hong Kong and was incorporated in 2009, on June 8th by Wei Bin Yan and Yue Shi Wu. It operates as part of the packaged foods industry, under the consumer defensive sector. The firm serves consumers around the globe.
The company operates through the Dairy and Related Products, and Nutrition Products segments. The Dairy and Related Products segment is involved in the manufacture and sale of dairy and related products, particularly on formula milk powder products, to its customers globally. On the other hand, the Nutrition Products segment is involved in the manufacture and sale of nutrition products. It derives most of its revenue from the Dairy and Related Products segment. Geographically, it generates a majority of revenue from PRC and also has a presence in the European Union; Middle East; North and South America; Australia; New Zealand and other countries. The company operates as a subsidiary of Hong Kong Jingang Trade Holding Company Ltd.
The enterprise provides cow milk-based formula products under the Allnutria, HyprocaHollary, HyprocaHypure, Neolac, and Puredo brand names; and goat milk-based infant formula products under the Kabrita brand. It also produces formula milk powder products on an original equipment manufacturing basis. In addition, it develops, distributes and sells probiotic products.
The company remains committed to expanding its corporate position globally, which will extend its consumer reach and open it up to new growth and investment opportunities. This will, in turn, generate significant value for its shareholders.
Ausnutria Dairy (AUDYF), closed Monday's trading session at $0.53, even for the day. The average volume for the last 3 months is 38,589 and the stock's 52-week low/high is $0.05/$0.53.
Arafura Rare Earths (ARAFF)
SmarTrend Newsletters, QualityStocks and PennyStockPower reported earlier on Arafura Rare Earths (ARAFF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Arafura Rare Earths Ltd (OTC: ARAFF) (ASX: ARU) (FRA: REB) is a mineral exploration and development firm that is focused on exploring for and developing mineral properties.
The firm has its headquarters in Pert, Australia and was incorporated in 1997, on December 3rd. Prior to its name change in October 2022, the firm was known as Arafura Resources Ltd. It operates as part of the other industrial metals and mining industry, under the basic materials sector. The firm serves consumers around the globe.
The company primarily explores for rare earth metals like neodymium-praseodymium (NdPr) oxide and mixed middle-heavy rare earths (SEG/HRE) oxide, phosphate, gold, nickel, and vanadium. NdPr oxide is its flagship product, which is used by magnet and magnet alloy customers. Rare earth products are also used in catalytic converters in automobiles, consumer electronics, energy efficiency lighting, main drive motors in electric vehicles and others.
The enterprise's principal property is the Nolans project, a rare earths-phosphate-uranium-thorium deposit that supplies neodymium and praseodymium products located in Northern Territory, Australia. This project comprises of a mine, process plant, including beneficiation, extraction and separation plants, and related infrastructure to be constructed and located at the Nolans site, roughly 135 km north of Alice Springs in Australia's Northern Territory. It is also engaged in other projects, such as Aileron-Reynolds, Northern Territory (NT) and Bonya JV, NT.
The firm remains focused on developing and growing its long-term prospects and advancing its engagements with other parties aligned with its investment strategies. This will positively influence revenues and investments into the firm as well as its overall growth.
Arafura Rare Earths (ARAFF), closed Monday's trading session at $0.4225, off by 2.8736%, on 38,589 volume. The average volume for the last 3 months is 1,800 and the stock's 52-week low/high is $0.1225/$0.44.
Sappi Ltd (SPPJY)
MarketBeat, Trades Of The Day, DividendStocks, Daily Trade Alert and Zacks reported earlier on Sappi Ltd (SPPJY), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Sappi Ltd (OTC: SPPJY) (JSE: SAP) (FRA: SPI) is a holding firm that is engaged in the provision of materials from woodfiber-based renewable resources.
The firm has its headquarters in Johannesburg, South Africa and was incorporated in 1936, on December 17th. It operates as part of the paper and paper products industry, under the basic materials sector. The firm serves consumers in Europe, South Africa and North America, through its energy self-sufficient operations. The majority of its revenue is generated from Europe.
The enterprise’s raw material offerings include dissolving pulp, wood pulp, biomaterials and timber, and end-use products, such as packaging and specialty papers, graphic papers, casting and release papers and forestry products, which are manufactured from wood fiber sourced from sustainably managed forests and plantations. Its Graphic papers include Galerie, Magno, GalerieArt, Quatro, Vantage, Raw and heaven 42. The enterprise also offers flexible packaging papers, label papers, functional paper packaging products, containerboards, paperboards, silicone base papers, dye sublimation papers, and inkjet papers. Its coated and uncoated graphic paper products are used by printers in the production of brochures, books,magazines, catalogs, direct mail, newspapers, and other print applications.The enterprise produces biomaterials and biochemicals to customers based across more than 150 countries.
The company, which is working towards building a thriving world by acting boldly to support the planet, people and prosperity, is focused on delivering sustained value to its stakeholders. This will positively influence its growth and bring in additional investments into the company.
Sappi Ltd (SPPJY), closed Monday's trading session at $3.0199, off by 5.0346%, on 1,800 volume. The average volume for the last 3 months is 611,556 and the stock's 52-week low/high is $2.244539/$4.20.
Wesdome Gold Mines (WDOFF)
MarketClub Analysis, MarketBeat, Trades Of The Day, TradersPro and Daily Trade Alert reported earlier on Wesdome Gold Mines (WDOFF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Wesdome Gold Mines Ltd (OTCQX: WDOFF) (TSE: WDO) (LON: 0VOA) (FRA: RKVA) is a mining exploration firm that is focused on exploring for, extracting, processing and reclaiming gold.
The firm has its headquarters in Toronto, Canada and was incorporated in 1980, on October 21st. It operates as part of the gold industry, under the basic materials sector. The firm serves consumers in Canada.
The company principally produces gold in the form of doré bars, as well as silver as a by-product. Its activities are primarily based in Canada, with the company generating most of its revenue from the sale of gold and silver bullion.
The enterprise’s properties include the Eagle River Complex, which comprises of the Eagle River Mine, the Mishi Mine and the Eagle River Mill. The Eagle River Underground Mine is located roughly 50 km west of Wawa, Ontario. The Eagle River Underground Mine in Wawa, Ontario produces gold at a rate of about 92,000 to 105,000 ounces annually. It also holds interest in the Kiena Mine Complex, which includes the Kiena Mine concession and Kiena Mill situated in Val-d'Or, Québec. The Kiena Complex is a fully permitted mine with an approximately 930-metre shaft and a 2,000 ton-per-day mill. The enterprise also retains exposure to the Moss Lake gold deposit in Ontario through Goldshore Resources Inc.
The company recently announced its latest production results, with its CEO noting that they remained focused on increased gold production and preparing the Presqu’ile zone for future development. This will positively influence the company’s revenues and investments.
Wesdome Gold Mines (WDOFF), closed Monday's trading session at $4.62, up 1.3158%, on 611,956 volume. The average volume for the last 3 months is 17.998M and the stock's 52-week low/high is $4.486/$13.23.
Coinbase Global Inc. (COIN)
InvestorPlace, Schaeffer's, Prfmonline, The Street, Greenbackers, MarketClub Analysis, QualityStocks, Kiplinger Today, MarketBeat, SmallCapVoice, OTCPicks, Ceocast News, Investopedia, CoolPennyStocks, HotOTC, Daily Trade Alert, The Online Investor, Trades Of The Day, StockEgg, Stock Stars, Penny Invest, Stock Rich, StocksEarning, The Wealth Report, InsiderTrades, CNBC Breaking News, The Stock Psycho, BestOtc, Top Pros' Top Picks, Top Gun, BullRally, HotShotStocks, StockHotTips, Zacks, Wealth Daily, Today's Financial News, Early Bird, MadPennyStocks, Profit Confidential, StockRich, Stockpalooza, PennyInvest, PennyStockVille, PennyTrader Publisher, FeedBlitz, Cabot Wealth, Dynamic Wealth Report, Blaque Capital Stocks, CRWEWallStreet, Early Investing, BloomMoney, Atomic Trades, Energy and Capital, Eagle Financial Publications, Pennybuster, wyatt research newsletter, WiseAlerts, StockMister, Stock Traders Chat, Stock Fortune Teller, Stock Analyzer, Penny Stock Finder, Round Up the Bulls, Green Chip Stocks, Penny Stock Rumble, AllPennyStocks, Momentum Traders, MicrocapVoice, Louis Navellier, INO Market Report and Standout Stocks reported earlier on Coinbase Global Inc. (COIN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
California’s Department of Motor Vehicles (DMV) has plans underway to apply blockchain technology to help in its recordkeeping, including car ownership transfers as well as licensing and registration of new cars and drivers. The DMV has collaborated with an open-source company, Tezos blockchain, to execute peer-to-peer business undertakings and be a platform used to deploy smart contracts. These sentiments were mentioned by Fortune magazine.
The DMV in California, plus a blockchain software enterprise called the Oxhead Alpha and Tezos, announced a proof-of-concept program that will enable the California DMV to title vehicles as of Jan. 25, 2023, this year.
Ajay Gupta, one of the senior digital officials at the Department of Motor Vehicles, hoped that the plans to create the blockchain title data base would mature within three months. “After that, the DMV aims to work on consumer facing applications like digital wallets which hold the title of the car via an NFT,” said Gupta.
With the many blockchain applications that maintain a decentralized, secure data record, car titles pose a possible use case for this technology. A Cleveland startup mobilized $5 million to digitize the title of cars, and California reported in 2020 that it was conducting some possible blockchain trial projects, with the California DMV included as one of these options.
Gupta said the DMV had started on the project but had to suspend it due to the COVID-19 pandemic. “We plan to utilize the opportunity to use digital ledgers to generate and transfer car titles, in a bid to digitize our DMV processes,” said Gupta. Andrew Smith, Oxhead Alpha’s president said, “Leveraging the blockchain tech would address transaction fraud and car tracking issues.”
Blockchain is a mutual and well-established ledger facilitating processes of transaction recording and tracking of assets in a professional business network. A great blockchain advantage is data record fidelity and security. Congressman French Hill was quoted saying he will promote a digital asset regulatory framework that promotes America as a place of innovation in financial technology and other blockchain technologies.
In a report this month, the World Economic Forum (WEF) said it trusts that the blockchain technology will be a fundamental part of today’s economy. WEF called attention to the extensive cryptography and blockchain tech applications, saying they have an impact in the financial services sector.
Such actions by the DMV in California create positive energy around the blockchain industry. Other actors such as Coinbase Global Inc. (NASDAQ: COIN) may see an uptick in the demand for their services as more people gain confidence in these new technologies.
Coinbase Global Inc. (COIN), closed Monday's trading session at $74.59, off by 0.053598%, on 17,998,386 volume. The average volume for the last 3 months is 3.396M and the stock's 52-week low/high is $31.55/$217.49.
Lordstown Motors Corp. (RIDE)
Green Car Stocks, Schaeffer's, InvestorPlace, QualityStocks, StocksEarning, The Street, MarketBeat, MarketClub Analysis, Trades Of The Day, Early Bird, Daily Trade Alert, StockEarnings, The Online Investor, CNBC Breaking News, Kiplinger Today, StreetInsider, BUYINS.NET, The Stock Dork and Cabot Wealth reported earlier on Lordstown Motors Corp. (RIDE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Norway is known for its frequent below-freezing weather, rocky terrain and lengthy outstretched lonely roads; the country might not appear to be the best spot to operate an electric vehicle, which has a battery that drains quickly during cold weather. However, the country has been a global leader in electric vehicles.
The amount of range electric cars lose varies depending on the vehicle model and the strength of the cold temperatures. Whenever an EV is left parked outdoors in cold (below 15°C) weather, the battery usage is substantially higher and requires a long time before it resumes regular consumption.
Long trips should be scheduled ahead by drivers; however, this process is simplified by vehicle applications as well as the extensive network of more than 5,600 quick and speedy charging points in Norway.
Norway, which is a significant oil producer, plans to phase out the sales of fuel oil vehicles by 2025, 10 years ahead of the planned prohibition by the European Union. In fact, a remarkable four of every five brand-new vehicles sold in the country in 2022 were electrical.
As per the figures released on Wednesday by ACEA, in contrast, the sales of new electric vehicles in the European Union increased from 9.1% in 2021 to 12.1% in 2022.
Finmark, a region in the north of Norway where temperatures have occasionally drop to 51C, is where electric cars made up 54% of newly registered vehicles last year, demonstrating that the cold is not such a deterrent. In fact, Nordic countries, which frequently experience cold weather, are ranked among the first in the world for electric vehicles. About 33% of Sweden and Iceland’s new automobile sales in 2022 were electric, for example.
According to Norwegian EV Association president Christina Bu, preheating systems are now being added to an increasing number of new electric cars; the systems warm the vehicle before it is charged, therefore accelerating charging and also extending range. She added that the cold is a concern for many people, not just those driving electric vehicles. In fact, fuel-powered engine vehicles occasionally have trouble starting in very cold conditions.
It is evident that the Norwegians are unfazed about the impact temperatures have on EV usage; more than 20% of the vehicles on the roads in Norway today are electric as well as environmentally friendly, with the electricity being used coming almost entirely from hydropower.
With this real-world evidence proving that EVs can operate in the colder regions of the world without any major problems, there is nothing that will stand in the way of models from automakers such as Lordstown Motors Corp. (NASDAQ: RIDE) reaching all corners of the world regardless of the regional climate.
Lordstown Motors Corp. (RIDE), closed Monday's trading session at $1.42, off by 3.4014%, on 3,396,493 volume. The average volume for the last 3 months is 204,411 and the stock's 52-week low/high is $0.87/$3.79.
Arch Resources Inc. (ARCH)
InvestorPlace, MarketBeat, Zacks, QualityStocks, The Online Investor, MarketClub Analysis, Kiplinger Today, TradersPro, Schaeffer's, The Street, Daily Wealth, StreetAuthority Daily, Cabot Wealth, Daily Trade Alert, FreeRealTime, Investing Daily, InvestorGuide, Barchart, Trades Of The Day, StreetInsider and Uncommon Wisdom reported earlier on Arch Resources Inc. (ARCH), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
As America looks to adopt green-energy sources into its energy mix over the next few decades, most of the benefits of this move have been described as environmental. After all, cutting out polluting sources of energy such as coal and oil would clean up our air and keep water bodies clean and fresh for human consumption.
Furthermore, a long-term and sustained green-energy drive could halt global warming and even undo some of the damage caused by burning dirty fossil fuels for centuries. With these goals in mind, the United States is planning on investing billions of dollars into researching, developing and building green-energy infrastructure.
However, switching to green and renewable energies could also result in significant savings for states that still overwhelmingly rely on coal. Although the fear is that shutting down coal-fired power plants in favor of renewables could impact local economies by causing innumerable job losses, the numbers show otherwise.
With a whopping 90% of coal power plants costing more to run compared to renewables, transitioning to green energy could save up to $589 billion in mostly Republican states. In fact, 209 out of America’s total 210 coal-fired power plants now cost more to run compared to the cost of replacing them with solar and wind energy.
In Nevada, for instance, the state’s last power plant is slated to shut down in 2025 after four decades of service due to increasing operating costs. In its place, local utility firm NV Energy plans on building two solar-plus-storage facilities next to the soon-to-be-closed power plant by 2025, creating hundreds of jobs and replacing the dozens that would be lost after the power plant’s closure.
The solar facilities would also result in long-term and sustainable revenue for the local and state governments. In fact, these two solar-plus-storage projects could end up creating more jobs over time as they continue to harvest solar energy in the region.
Given that the Inflation Reduction Act will invest billions of dollars into green-energy projects, utility firms across the country stand to gain close to $600 billion in investment by transitioning to renewables.
Coal use is already in decline across America, with the share of coal-generated electricity in the country’s energy mix dropping from 50% of the yearly supply to less than 22% in 2022. Although the decline of coal-fired electricity was initially caused by competition from natural gas and improvements in energy efficiency that reduced electricity demand, reducing green-energy costs coupled with smart government policy has accelerated the decline.
However, enterprises such as Arch Resources Inc. (NYSE: ARCH) will still see demand for the coal they extract because not all regions will be as quick to adopt renewable in lieu of coal and other fossil fuels.
Arch Resources Inc. (ARCH), closed Monday's trading session at $146.95, off by 0.716168%, on 207,238 volume. The average volume for the last 3 months is 110,065 and the stock's 52-week low/high is $102.26/$183.53.
Field Trip Health Ltd. (FTHWF)
We reported earlier on Field Trip Health Ltd. (FTHWF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Psychedelics are poised to revolutionize the psychiatric industry over the next few decades. This is because initial research into hallucinogenic drugs has revealed that they may be able to alleviate the symptoms of mental health disorders such as post-traumatic stress disorder.
Studies have found that even a single dose of a psychedelic, such as ketamine and psilocybin, can result in long-term benefits without most of the side effects typically seen in pharmaceutical mental health drugs. However, these hallucinogenic drugs are still psychoactive despite their therapeutic benefits, and patients usually have to go through a guided psychedelic experience, or “trip,” once they receive an infusion.
The hallucinogenic effects of these drugs have turned off some people who would like to gain the mental health benefits of psychedelics such as psilocybin without experiencing their psychoactive effects. A new research article published in the “ACS Chemical Neuroscience” journal has pointed to the possibility of a psychedelic that can deliver mental health benefits without the trip.
Researchers analyzed a non-hallucinogenic psychedelic called Ariadne to determine its safety and efficacy against psychiatric and neurological disorders. Although Ariadne has been around for a while, with pharmaceutical firm Bristol-Myers studying it in the 1970s, the research died down in the 1970s. The drug had exhibited a variety of effects in early unpublished trials such as remission of symptoms in schizophrenia and Parkinson’s disease patients as well as improvements in cognition in older individuals.
The recent research article used data from these unpublished clinical trials coupled with new research data to hypothesize how Ariadne and other compounds with similar chemical structures can be used to address psychiatric and mental disorders without causing any hallucinogenic effects. The article also highlighted an animal study carried out by the authors on a mouse that exhibited all the markers of Parkinson’s disease in humans, revealing that Ariadne quickly alleviated the symptoms after infusion.
However, more research is needed to determine if Ariadne and chemically similar drugs can be used to treat the disease in humans safely and effectively. Columbia University chemistry professor Dalibor Sames, who was one of the paper’s coauthors, noted that the research underlined the potential Ariadne and other chemically similar drugs can have in medical applications.
The study points to a possible future when patients can benefit from psychedelic-assisted therapies without having to go through the so-called psychedelic experience.
With investors and Big Pharma investing millions of dollars into the research and development of mass-market psychedelic treatments, drugs such as Ariadne may be able to bridge the gap for people who weren’t comfortable with psychedelic treatments due to their hallucinogenic effects.
At the moment, nothing is off the table as numerous startups such as Field Trip Health Ltd. (OTC: FTHWF) (TSX: FTHW) work on extensive drug-development pipelines that include various psychedelic compounds. As these R&D programs progress, a lot of new information about novel hallucinogenics could emerge.
Field Trip Health Ltd. (FTHWF), closed Monday's trading session at $0.07604, off by 3.7468%, on 110,065 volume. The average volume for the last 3 months is 7.693M and the stock's 52-week low/high is $0.0177/$0.50.
Sidus Space Inc. (SIDU)
MarketClub Analysis, RedChip, Wall St. Warrior, QualityStocks, Money Wealth Matters and INO Market Report reported earlier on Sidus Space Inc. (SIDU), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Sidus Space (NASDAQ: SIDU), a “Space-as-a-Service” satellite company, recently announced the closing of its underwritten public offering of 17,250,000 shares of its Class A common stock (or pre-funded warrants in lieu thereof), which included the full exercise of the underwriters’ over-allotment option. Each share of Class A common stock was sold to the public at a price of $0.30. Sidus Space secured approximately $5.2 million in gross proceeds from the offering and intends to use the net proceeds for sales and marketing, operational costs, product development, manufacturing expansion and for working capital and other general corporate purposes. Boustead Securities LLC and EF Hutton, division of Benchmark Investments LLC, acted as the representatives of the underwriters for the offering.
To view the full press release, visit https://ibn.fm/QjHGg
About Sidus Space Inc.
Sidus Space, located in Cape Canaveral, Florida, operates from a 35,000-square-foot manufacturing, assembly, integration and testing facility focused on commercial satellite design, manufacture, launch and data collection. The company’s rich heritage includes the design and manufacture of many flight and ground component parts and systems for various space-related customers and programs. Sidus Space has a broad range of “Space-as-a-Service” offerings including space-rated hardware manufacturing, design engineering, satellite manufacturing and platform development, launch and support services, data analytics services and satellite constellation management. Sidus Space has a mission of Bringing Space Down to Earth(TM) and a vision of enabling space flight heritage status for new technologies while delivering data and predictive analytics to domestic and global customers. Any corporation, industry or vertical can start their journey off-planet with Sidus Space’s rapidly scalable, low-cost satellite services, space-based solutions and testing alternatives. More than just a “Satellite-as-a-Service” provider, Sidus Space is a trusted mission partner–from concept to low Earth orbit and beyond. Sidus is ISO 9001:2015, AS9100 Rev. D certified and ITAR registered. For more information about the company, visit www.SidusSpace.com.
Sidus Space Inc. (SIDU), closed Monday's trading session at $0.5275, up 2.2287%, on 7,692,826 volume. The average volume for the last 3 months is 169,743 and the stock's 52-week low/high is $0.375/$11.23.
The QualityStocks Company Corner
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF)
- Coyuchi Inc.
- Utopia VR
- MetAlert Inc. (OTC: MLRT)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
- Knightscope Inc. (NASDAQ: KSCP)
- Cepton Inc. (NASDAQ: CPTN)
- India Globalization Capital Inc. (NYSE American: IGC)
- Advanced Container Technologies Inc. (OTC: ACTX)
- Data443 Risk Mitigation Inc. (OTC: ATDS)
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
The QualityStocks Daily Newsletter would like to spotlight BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV).
•BiondVax is a biotechnology company focused on developing,manufacturing, and commercializing innovative immunotherapeuticproducts
•The company is developing nanosized antibodies (“NanoAbs”)addressing large market diseases underserved by current treatments,with its lead candidate targeting COVID-19
•The FDA recently revoked the sole authorization of a COVIDprophylactic, Evusheld, citing the inability of the monoclonalantibody cocktail to neutralize emerging subvariants of the Omicronvariant
•BiondVax’s lead NanoAb candidate shows superior advantages tomonoclonal antibodies, with the data gathered strengthening thevalue proposition of the COVID-19 NanoAb as a safe, efficacious,and convenient therapeutic and prophylactic drug
The Food and Drug Administration (“FDA”) recently withdrew theauthorization of
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) is a biopharmaceutical company focused on developing, manufacturing and commercializing innovative products for the prevention and treatment of infectious diseases and other illnesses.
In collaboration with the prestigious Max Planck Institute for Multidisciplinary Sciences (MPG) and the University Medical Center Göttingen (UMG), both in Germany, BiondVax is developing a pipeline of innovative nanosized antibody (NanoAb) therapies addressing diseases underserved by current treatments and with large and growing markets, such as COVID-19, asthma and psoriasis.
NanoAbs, also known as VHH-antibodies or Nanobodies, are alpaca-derived nanosized antibodies that exhibit multiple significant competitive advantages over existing antibody therapies, including stability at high temperatures, superior binding affinity, more effective and convenient routes of administration and efficient production. BiondVax is uniquely positioned to advance nanosized antibody innovation from R&D through commercialization.
The company’s highly experienced and successful pharmaceutical industry leadership team includes former senior executives from Novartis, GSK and Bristol-Myers Squibb.
Since its founding, BiondVax has executed eight clinical trials, including a seven-country, 12,400-participant Phase 3 trial of a prior influenza vaccine candidate, and it built, owns and operates a 20,000 sq. ft. state-of-the-art GMP biologics manufacturing facility housing its laboratories, production facilities and offices.
Lead Candidate: Inhaled COVID-19 NanoAb
In December 2021, BiondVax signed definitive agreements with the Max Planck Society – parent organization of the Max Planck Institute for Multidisciplinary Sciences– and the UMG to enter a strategic collaboration for the development and commercialization of innovative COVID-19 NanoAbs.
The company is planning a rapid development path that leverages its expertise and capabilities in biological drug development and manufacturing. BiondVax anticipates preclinical proof-of-concept results for an inhaled COVID-19 NanoAb by the end of 2022, with initial Phase 1/2a human clinical trial results expected in 2023.
The intended inhaled mechanism of delivery of BiondVax’s COVID-19 NanoAb formulation may serve as a significant differentiator when compared to approved monoclonal antibodies, which are injected. Inhaled delivery has shown to be cheaper, more convenient and likely safer for patients and providers.
NanoAb Pipeline: Psoriasis, Asthma and More
The COVID-19 NanoAb development agreement is part of a broader five-year research collaboration agreement signed in March 2022 covering discovery, development and commercialization of NanoAbs for several other disease indications with large market medical needs, including asthma, psoriasis, macular degeneration and psoriatic arthritis.
BiondVax has an exclusive worldwide license for development and commercialization of COVID-19 NanoAbs and exclusive options for similar worldwide licenses for NanoAbs for the above mentioned additional large market disorders currently underserved by approved therapeutic antibodies.
Academic research teams from MPG and UMG have verified strong affinity by the new NanoAbs to their biological target molecules and high thermostability. They have also demonstrated strong neutralization by several NanoAb candidates of their respective target molecules. Neutralization studies of the other NanoAbs are expected to begin later in 2022.
Based on the promising results, BiondVax will focus development efforts beginning with the following NanoAbs:
- NanoAbs targeting IL-17 as drug candidates for the potential treatment of psoriasis and psoriatic arthritis
- NanoAbs targeting IL-13 and NanoAbs targeting TSLP as drug candidates for the potential treatment of asthma
These are conditions for which the antibody target is validated by existing treatments and the mechanism of action is well understood. Both represent large medical needs and growing markets. BiondVax anticipates preclinical proof-of-concept for at least one of these NanoAbs in 2023. This is in addition to the aforementioned human clinical Phase 1/2a for the inhaled COVID-19 NanoAb therapy, which is also anticipated in 2023.
Market Opportunity
COVID-19 treatment, target of the company’s lead NanoAb therapy candidate, had an estimated market size of $22 billion in 2021.
Future BiondVax drug candidates will target conditions with large markets growing at attractive CAGRs.
The global asthma treatment market was valued at $18.08 billion in 2019 and is projected to reach $26.01 billion by 2027, exhibiting a CAGR of 4.5% during the forecast period, according to Fortune Business Insights. The research firm predicts that the global psoriasis treatment market will grow from $26.37 billion in 2022 to $47.24 billion by 2029, exhibiting a CAGR of 8.7% over the forecast period.
Management Team
Amir Reichman is BiondVax’s CEO. He previously was Head of Global Vaccines Engineering Core Technologies at GSK Vaccines in Belgium. Prior to that, he held leadership roles at Novartis Vaccines’ Global Vaccines Supply Chain Management organization. He was the first employee of NeuroDerm Ltd., a company focused on transdermal drug delivery, and served as Chief Engineer and Senior Scientist until his departure in 2009. He earned a M.Sc. in Biotechnology Engineering from Ben-Gurion University and an MBA in Finance and Health Care Management from the University of Pennsylvania’s Wharton School.
Tamar Ben-Yedidia, Ph.D., is Chief Science Officer at BiondVax. She has more than 30 years of experience in immunology, with specific expertise in the development of vaccines. She began her career with Biotechnology General Ltd., working on development of a recombinant Hepatitis-B vaccine. She later joined the Weizmann Institute of Science, working on the design of a peptide-based vaccine against several pathogens. She is widely published, with numerous refereed articles and invited reviews in various scientific journals. She received her Ph.D. from the Weizmann Institute.
Elad Mark is COO at BiondVax. He has over 15 years of biotechnology industry experience encompassing diverse project stages including feasibility studies, conceptual and detailed design, commissioning, qualification and process validation. Prior to joining BiondVax, he led Novartis’s $800 million investment in a biologics facility in Singapore. With Biopharmax and Antero, both global pharmaceutical engineering companies, he successfully led projects in Israel, China and Singapore. He holds a BSc. in Engineering from the Afeka Tel Aviv Academic College of Engineering and an MBA from the Open University of Israel.
Uri Ben-Or is CFO at BiondVax. He has served as CFO with public life science companies traded on the TASE, OTC and Nasdaq. Ben-Or provides his services to BiondVax through CFO Direct, a company he founded and for which he serves as CEO. He served as the VP of Finance of Glycominds, a leading biotechnology company, and as CFO of a spin-off from Telrad Networks. He also served as a Corporate Controller at Menorah Capital Markets and as an Auditor at PWC. He holds a B.A. in Business from the College of Administration, an MBA from Bar-Ilan University, and is a CPA.
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV), closed Monday's trading session at $2.79, up 5.283%, on 195,149 volume. The average volume for the last 3 months is 41,271 and the stock's 52-week low/high is $2.37/$22.90.
Recent News
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Animal Study Results Featured in Aegis Capital Corp. Updated Price Target
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Aiming to Capture Significant Market Share for Treatment and Prevention of COVID-19 Illness with Innovative Inhaled NanoAb
- InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) to Present at BIO CEO & Investor Conference
Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF)
The QualityStocks Daily Newsletter would like to spotlight Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF).
•Eloro Resources announced the successful completion of its primaryshare issuance in late January, 2023
•The placement, which comprised of over 3.4 million primary sharesissued at a price of c$3.15, raised gross proceeds of approximatelyC$10.9mn
•The proceeds will be destined to Eloro’s ongoing Bolivian miningoperations as well as general corporate purposes
•Eloro Resources is set to disclose its initial resource estimatesfor its flagship Iska Iska Project in coming months
Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) is a publicly traded exploration and mine development company with a portfolio of gold and base-metal properties in Bolivia, Peru and Quebec.
The company has an option to acquire a 99% interest in the highly prospective Iska Iska Property, classified as a silver-tin polymetallic epithermal-porphyry complex, a significant mineral deposit type in the Potosi Department of southern Bolivia. Iska Iska is a road-accessible, royalty-free property.
Eloro also owns an 82% interest in the La Victoria Gold/Silver Project, located in the North-Central Mineral Belt of Peru, some 50 kilometers south of Barrick’s Lagunas Norte Gold Mine and Pan American Silver’s La Arena Gold Mine. La Victoria consists of eight mining concessions and eight mining claims encompassing approximately 89 square kilometers. La Victoria has good infrastructure, with access to road, water and electricity, and is located at an altitude that ranges from 3,150 meters to 4,400 meters above sea level.
The company has a strong management and technical team working diligently to uncover the value of both Iska Iska and La Victoria. Eloro is based in Toronto, Canada.
Projects
Iska Iska – Potosi, Bolivia
Iska Iska is associated with a Miocene possibly collapsed/resurgent caldera, emplaced on Ordovician age rocks with major breccia pipes, dacitic domes and hydrothermal breccias. The property is wholly controlled by the title holder, Empresa Minera Villegas S.R.L. It is located 48 kilometers north of Tupiza city, in the Sud Chichas Province of the Department of Potosi. This is an important mineral deposit type in the prolific South Mineral Belt of Bolivia. Eloro commissioned a NI 43-101 Technical Report on Iska Iska, which was completed by Micon International Limited and is available on Eloro’s website and under its filings on SEDAR.
A fully financed drill program is currently underway on the property, situated near world-class deposits including Silver Sand, San Bartolomé, Pulacayo, San Cristobal, San Vicente, Chorolque, Tasna, Choroma and Siete Suyos. Iska Iska is in the southwest part of the Eastern Cordillera, which hosts a number of major polymetallic mines and mineral deposits. Drilling and continuous channel sampling results have demonstrated some very high metal values, especially silver and tin, within an immense system, where mineralization has been encountered in every drill hole to date. The company believes there is excellent potential for world-class bulk mineable deposits.
La Victoria – Ancash, Peru
The La Victoria project, targeting gold and silver production, is situated near world-class, low-cost gold producers Pan American Silver and Barrick Gold Corporation. Located in Ancash Department, La Victoria sits on the western slopes of the Peruvian Andes. The property is located 12 hours from Lima, with a travel distance of 600 kilometers. The nearest road accessible population centers from La Victoria are Huandoval, Pallasca and Cabana. The project includes four principal mineralized zones in Peru’s prolific North-Central Mineral Belt – San Markito, Victoria, Victoria South and Ccori Orcco – with excellent potential for gold discovery. Operations at La Victoria are planned to proceed with a 2,000-meter diamond drilling program to test targets to outline potential resources at San Markito. Trenching and sampling confirmed high silver values and veins at San Markito in 2020.
Market Outlook
According to industry association The Silver Institute, the outlook for silver demand is exceptionally promising, with global demand forecast to rise to a record high of 1.112 billion ounces in 2022. The increase will be driven by record silver industrial fabrication, which is forecast to improve by 5%, as silver’s use expands primarily in solar energy and electric vehicle (EV) manufacturing. The institute states that government commitments to carbon neutrality have resulted in a rapid expansion of green energy projects, driving record photovoltaic panel installations which are expected to lift silver demand in this segment to an all-time high in 2022.
Rising demand in the electronics industry is also boosting the demand for tin, which is primarily used in solder. The electronics and electrical industries use solders containing 40-70% tin, which provide strong and reliable joints under a variety of environmental conditions. At present, the majority of the assemblers are using patented tin-and-copper-based solders. Mordor Intelligence estimated tin demand at 387 kilotons in 2021 and forecasts demand growth of 2.5% annually through 2027. Over the medium term, surging demand from the EV market and increasing applications in the electrical and electronics industry is expected to drive the market.
Management Team
Thomas G. Larsen is CEO of Eloro. He has more than 40 years of experience in the investment industry, specializing in corporate finance and management of junior resource companies, raising in excess of C$200 million. He previously held the position of President and Chief Executive Officer of Champion Iron Limited. Prior to that, he was President and Chief Executive Officer of Champion Iron Mines Limited.
Dr. Bill Pearson is Executive VP of Exploration for Eloro. He has more than 40 years of direct experience in the exploration and production of minerals worldwide. He played an integral role in the acquisitions of Desert Sun Mining Corp. by Yamana Gold in 2006 and Central Sun Mining by B2 Gold in 2009. He was formerly VP Exploration at Desert Sun Mining and Senior VP at Central Sun Mining.
Miles Nagamatsu, CPA, is CFO at Eloro. He has over 30 years of experience in accounting, management, lending, restructurings and turnarounds. Since 1993, he has acted as a CFO of public and private companies primarily in the mineral exploration and investment management sectors. He holds a Bachelor of Commerce degree from McMaster University.
Osvaldo Arce Burgoa is General Manager at Eloro. He is a geological and mineral processing engineer with 26 years of experience in Bolivia. He is a former President of the Bolivian Geological Society, Main Technical Advisor of the National Mining Corporation (COMIBOL) and has served as exploration manager and chief geologist at various mining and exploration companies. He has authored two books on Bolivian geology and holds a doctorate in mining engineering from Tohoku University in Sendai, Japan.
Eloro Resources Ltd. (OTCQX: ELRRF), closed Monday's trading session at $2.358, up 0.182691%, on 41,271 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $22.90/$.
Recent News
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Grants RSUs as Part of Long-Term Incentive Plan
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Announces Successful Capital Raising; Net Proceeds Will Be Destined Towards Ongoing Bolivian Mining Operations
- Eloro Resources Announces RSU Grants
Coyuchi Inc.
The QualityStocks Daily Newsletter would like to spotlight Coyuchi Inc.
To view the full article, visit https://ibn.fm/onlgJ
Coyuchi is the gold standard in sustainable luxury home goods. The company offers sustainably produced luxury organic bedding, sheets, towels, apparel, and other home goods for the environmentally conscious home. With a timeless, coastal-inspired aesthetic, Coyuchi uses only 100% organic cotton materials to manufacture all of its textiles.
The Company was built upon four foundational pillars: protect the planet, innovate circular design, live sustainably, and enrich the community. These guiding principles have proven an effective market strategy. In 2021, Coyuchi earned $33.3 million in net sales, amounting to 26% YoY growth (the industry average is only 5%). It also experienced 2x customer growth to 200,000 active customers, averaging a 35% customer repeat purchase rate.
With a seasoned leadership team, a robust e-commerce shopping experience, and a healthy customer base that drives the fast-growing organic luxury market, Coyuchi is prepared to propel a new phase of growth as the rest of the world finally awakens to sustainability at scale.
A Lucrative Market Ripe for the Taking
The global market for organic bedding, which was estimated at $814.3 million in 2020, is projected to reach $1.1 billion by 2027, growing at a CAGR of 4.9% over that period, according to Research and Markets. More specifically, the domestic organic bedding market is estimated at $240.1 million in 2020, according to Statista. Overall, the U.S. market for home textiles is currently valued at $25 billion annually, and, with a forecast annual growth rate of 5%, it is expected to reach $30 billion by the end of 2025.
Grand View Research reported in 2020 that shifting consumer preference toward high-end lifestyle products is a key factor driving the growth of the organic bedding market. Seventy-four percent of consumers are willing to pay more for sustainable products – a consumer preference that has steadily increased over the last few decades. Millennials especially favor ethical consumption over price when purchasing goods and services, with 83% of millennials reporting that they want the brands they purchase from to align with their beliefs and values (https://ibn.fm/PANNV). With a majority millennial customer base, Coyuchi is poised to capitalize on this trend.
Industry Defining Sustainability Practices
For 30 years, Coyuchi has explored organic farming and sustainable textiles and guarantees the highest environmental and ethical standards through a number of certifications such as The Global Organic Textile Standard (GOTS), Fair Trade Certified, and MADE SAFE®.
Coyuchi continues to push the organic textile market forward through its circularity initiatives and by supporting cross-industry sustainability advocates. Coyuchi’s mission to bring beauty and comfort to every home without sacrificing the health of our planet has resulted in a number of important sustainability checks and balances.
- A Circular Business Model: Coyuchi has cultivated a holistic 360-degree approach that contributes to the fight against climate change with its take back and recycling program, 2nd Home™. In 2017, it became the first luxury home brand to implement such an initiative, and, since then, the company has eliminated 68,758 lbs. of toxic chemicals from homes and renewed 6,000 lbs. of textiles.
- The Coyuchi Climate Council: In early 2022, Coyuchi introduced a cross-disciplinary council with a goal of Net Zero Emissions by 2025 and Net Positive Emissions by 2030. The Coyuchi Climate Council brings together influential minds across fashion, regenerative farming, and sustainability who have the knowledge and experience necessary to achieve climate change.
- C4: The California Cotton & Climate Coalition: Most recently, Coyuchi announced it is a founding member of C4, which includes innovative, sustainable fashion, apparel, and personal care brands like MATE the Label, Outerknown, Reformation, and Trace. Working together pre-competitively, C4 creates a structure for investing in regionally grown, Climate Beneficial™ cotton and directly supports the livelihoods of the farmers that grew it. Coyuchi is the only home industry brand currently involved in the project.
Omnichannel Business Model
Coyuchi differentiates itself through an omnichannel and circular business model, both of which have proven a clear draw for customers. It was an early adopter of an e-commerce sales and marketing approach (over 80% of its sales are directly through coyuchi.com), creating a distinct advantage over incumbents and start-up newcomers in the luxury space. This has resulted in a high lifetime value customer, luxury retail partners such as Nordstrom, and a flagship store in Marin County.
Coyuchi’s Organic Textile Products
Coyuchi’s product assortment consists of consciously designed bedding, bath, apparel, and lifestyle products spread across about 1,400 SKUs. The company believes that its product assortment, produced from 100% organic cotton with Global Organic Textile Standard (GOTS) certification, provides it with a significant competitive advantage. GOTS is the world’s leading textile processing standard for organic fibers, ensuring the organic status of textiles after harvesting raw materials through environmentally and socially responsible manufacturing all the way to labeling, a major environmental and social benefit over conventional cotton product production.
Coyuchi’s focused product assortment consists of four core categories:
- Bedding – A full suite of sustainable, organic, and high-quality sheets, duvet covers, blankets, and throws.
- Bath – A luxurious line of towels, bath rugs, and mats.
- Apparel – Premium apparel for men and women, including robes, sweaters, pants, and pajamas.
- Lifestyle – The lifestyle category offers 135 SKUs, from organic napkins to crossbody totes.
Management Team
Eileen Mockus is President and CEO at Coyuchi. She has more than 25 years of experience in retail, having held positions in textile development at Patagonia, Pottery Barn Teen, and The North Face. She earned a bachelor’s degree in textiles and clothing from UC Davis and an MSBA from San Francisco State University.
Sejal Solanki is Chief Marketing Officer at Coyuchi. She previously served as the company’s Vice President of E-Commerce. Before joining Coyuchi, she worked at teen clothing giant Charlotte Russe. She oversees the company’s digital marketing, site experience, brand marketing, and e-commerce strategy.
Marcus Chung is Coyuchi’s COO, overseeing supply chain, sourcing strategy, sustainability, and IT. He previously held positions at notable direct-to-consumer brands Third Love and Stitch Fix, as well as national retailer The Children’s Place. He holds a bachelor’s degree from Wesleyan University and an MBA from UC Berkeley’s Haas School of Business.
Margot Lyons is Director of Sustainability and Sourcing at Coyuchi, where she works with strategic partners to ensure all the company’s product sustainability standards are met. She received a master’s degree in textiles and clothing from UC Davis.
Use of Proceeds
This round of funding will be used to increase Coyuchi’s enterprise value through expanded marketing, product category expansion, continued physical presence, and B2B strategic partnerships with wholesalers, and online marketplaces.
Recent News
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Driving Growth While Focusing on Positive Environmental, Social Impacts
- Coyuchi Inc. Is Advancing the Organic Textiles Market Through Transparency and Circular Design
- InvestorNewsBreaks – Coyuchi Inc. CEO Featured in Recent Bell2Bell Podcast Release

Utopia VR
The QualityStocks Daily Newsletter would like to spotlight Utopia VR
•MetAlert, a developer of personal protective medical equipment andsupplies, owes much of its growth to acting on feedback fromreal-world caregivers who use its products for the monitoring andtracking of vulnerable patients
•This connection with caregivers has yielded advanced products suchas the award-winning, multi-patented GPS SmartSole, Invisabelt,Take-Along Tracker, and Rover Tracker, among others
•Attention to consumers has also led to its line of hearing aids,such as its flagship Hear IQ 4 rechargeable, app-controlled hearingaid, and its RoomMate wall-mounted 3D sensors for looking afterpatients without intruding on their personal space
Utopia VR is one of the world’s first ‘Metaverse-As-A-Service’ solutions for business. The company’s hosted and managed subscription software provides businesses a low barrier to entry, browser-based, device agnostic platform where they can manage their own private 3D metaverse meeting spaces. Users can host and attend Zoom-like virtual meetings in lifelike virtual reality – with no software downloads – engaging their audiences in a more collaborative and fun way.
Utopia VR has many technology and privacy advantages over solutions built on other metaverse marketplaces such as Horizon Worlds (META) or Decentraland. Utopia VR works on all devices – PC, mobile and virtual reality headsets – whereas many competitors only work in VR or on PC.
Utopia VR is headquartered in Kelowna, B.C.
Products
Utopia VR’s The Metaverse for Everyone™ is a one-click, web-based, avatar-driven, mobile-friendly audio- and video-conferencing platform that utilizes innovative 3D web technology. Utopia VR’s virtual platform works on digital devices including PCs, mobile phones and VR headsets such as Oculus Quest or HTC Vive. No software or proprietary hardware is needed.
Users navigate through the various VRoom environments by using avatars. Users can walk, talk and sit – just like they do in the real world. A user’s avatar can be controlled with a computer keyboard, smartphone or virtual reality headsets. Text chat, voice and video is ever-present and used to communicate with others in the VRoom. For important meetings and presentations, users can also import audio, video, 2D art and images, animated 3D objects, PDF files and their favorite NFTs by simply dragging and dropping files into a VRoom or pasting a video link from supported media platforms.
Organizations that have an existing website can transition their digital assets, including text, images, video, PDFs, slideshows and more, to VRoom environments with a simple copy and paste. This will allow their customers and audiences to experience their brand in a whole new, immersive environment.
Utopia VR’s mobile app enables users to personalize their own 3D environments and then schedule business meetings or social meetups in seconds through a proprietary link management system. The app is available for iPhone and iPad users. The company’s website mirrors the app, which means users can access Utopia VR directly from a PC, laptop, tablet, or VR headset without downloading the app.
Market Outlook
Regarded as the next iteration of the internet, the metaverse is a virtual space where the physical and digital worlds coexist and interact, encompassing virtual reality, augmented reality, extended reality and mixed reality, as well as making use of artificial intelligence and other technologies.
Data consolidator Statista estimated that the global metaverse market size stood at $38.85 billion in 2021 and projected the market would grow to be worth $47.48 billion in 2022. From there, Statista forecasts the value of the metaverse market will explode to reach $678.8 billion by 2030, achieving a CAGR of more than 39% over the period.
The metaverse could create $5 trillion in opportunity by 2030, according to McKinsey & Company.
Management Team
Stuart Gray, President, Co-Founder and director of Utopia VR, has been an officer and director for both private and publicly traded companies and has led public offerings for junior listed companies that have gone on to realize multibillion-dollar market valuations. He previously was a consultant and quarterbacked taking eXp World Holdings Inc. (NASDAQ: EXPI) public. eXp is a disruptive, no bricks and mortar, real estate brokerage firm with 85,000 agents worldwide using its virtual, software-based, metaverse platform for closing transactions, training and events.
Cory Braden, CTO and director of Utopia VR, is a forward-thinking strategic leader with over 20 years of experience in delivering software as a service. Recognized for a positive leadership style and excellent communication skills, he is well-versed in user experience, complex application architectures, cloud infrastructure and management of high-performance teams.
Terry Woloszyn, VP of Sales and Advisory at Utopia VR, brings vast technical and sales experience to the company. Before joining Utopia VR, he conceived and launched a data security startup and graduated from two startup accelerator programs. He has personally raised $20 million in equity venture funding.
Recent News
- Utopia VR - InvestorNewsBreaks – Why Utopia VR Is ‘One to Watch’
- InvestorNewsBreaks – Why Utopia VR Is ‘One to Watch’
- Utopia VR Is ‘One to Watch’

MetAlert Inc. (OTC: MLRT)
The QualityStocks Daily Newsletter would like to spotlight MetAlert Inc. (OTC: MLRT).
•2022 saw Lexaria introduce its patented DehydraTECH(TM) technologyto world-leading collaborators while also exploring DehydraTECH-CBDfor the potential treatment of hypertension
•It also saw success in its research for the potential treatment ofconditions such as epilepsy, making the year Lexaria’s most activeone yet
•Significant progress was made on key objectives in 2022, a featwhich the company plans to capitalize on and leverage to scale upthe commercial execution of its technology in 2023
•To facilitate this, Lexaria has brought on board Julian Gangolli,former President of GW Pharmaceuticals USA, as a strategic advisor.Chris Bunka, Lexaria’s CEO, anticipates 2023 to be the “best yearever”
MetAlert Inc. (OTC: MLRT) is a pioneer in location sensitive health monitoring devices (estimated $47 billion industry in 2021) and wearable technology products (industry forecast to reach $174 billion by 2030).
With over 20 years of experience and an extensive patent portfolio (30+), MetAlert is a leader for consumers/patients afflicted with Alzheimer’s, dementia, and autism (ADA). This market represents approximately 2.9% of the world’s population (approximately 34 million people in 24 developed countries). Due to specific behaviors (problems with memory, adversity to wearing unknown items, etc.) of consumers/patients in this market segment, traditional products, such as an iPhone or Fitbit, are not a practical solution. This has created a significant market with very few competitors for MetAlert.
MetAlert and its subsidiaries are engaged in designing, developing, manufacturing, distributing, and selling products and services in GPS/BLE wearable technology, personal location, wandering assistive technology, and health data collection and monitoring. The company offers a global end-to-end hardware, software, and connectivity solution, in addition to developing two-way tracking technologies, which seamlessly integrate with consumer products and enterprise applications.
Using its award-winning, patented GPS SmartSole® as a hub for collecting and transmitting data to the cloud in real-time, MetAlert is expanding its value proposition to consumers and increasing its revenue per user (RPU) while creating the largest database of health statistics for ADA consumers/patients. MetAlert generates revenue from product sales, recurring subscriptions, intellectual property (IP) licensing, and professional services. The company has international distributors servicing customers in over 35 countries and is an approved U.S. military government contractor. Its customers include public health authorities and municipalities, emergency and law enforcement, private schools, assisted living facilities, NGOs, small business enterprises, senior care homes and consumers.
The company is headquartered in Los Angeles, California, with a sales office in London, England, and distributors across the globe.
Products
- GPS SmartSoles® HUB (launched Q4 2022) is a GPS/BLE-equipped insole that allows remote monitoring, data collection, and encrypted data transmission to the cloud.
- Telehealth (available Q4 2022) allows access remotely to doctors and other health professionals on an as-needed basis. This service will also function as the prescribing doctor once Medicare reimbursement codes are established.
- Concierge (available Q4 2022) provides 24/7/365 enhanced emergency response that coordinates with all relevant parties to quickly detect false alarms and escalate response as needed.
- Bluetooth Enabled Devices (available Q1 2023) include third-party devices that collect vitals and other health data and connect with the GPS Smartsoles® HUB.
- Artificial Intelligence (available Q1 2023) software will evaluate the Teradata of health information identifying trends and respond to preestablished alert thresholds.
- Take-Along Tracker is a small GPS tracking device – less than three inches long – that works with 4G cellular service and will have the same “HUB” functionality as the GPS Smartsoles®. This versatile and affordable mini tracker boasts super long battery life, with up to 14 days of operation per charge.
- RoomMate™ is a wall-mounted alert system that detects and alerts caregivers about patient behavior that could lead to falls and injuries. The system features 3D infrared and wall-mounted sensors, eliminating the need for any other physical installation or wearables. RoomMate™ offers patient privacy by design. Images are not stored, but all actions are logged. It’s a unique solution for looking after patients without intruding on their personal space.
Market Outlook
According to Grand View Research (Patient Monitoring Devices Market Size & Share Report, 2030), the global patient monitoring devices market size was valued at $47.0 billion in 2021 and is expected to expand at a compound annual growth rate (CAGR) of 7.8% from 2022 to 2030. The expansion of the industry can be attributed to the rise in demand for monitoring devices used to measure, distribute, record, and display a variety of biometric data, including blood pressure, temperature, and blood oxygen saturation level.
The growing number of chronic disorders, such as diabetes, stroke, and kidney disease, are driving the demand for patient monitoring devices. For instance, according to the World Health Organization (WHO), about 422 million people globally have diabetes. Likewise, the number of asthma and chronic obstructive pulmonary disease patients (COPD) is increasing rapidly.
According to the WHO, around 235 million people suffer from asthma. As a result, peak flow meters, which are used to gauge respiration rate, are increasingly used. The market for patient monitoring devices is driven by the simplicity with which it is handled, transported, and remotely accessible. Major market players are engaging in a variety of tactics to expand the industry, including partnerships, cooperation, innovation, launches, and mergers.
During the COVID-19 outbreak, social segregation and quarantining procedures were put into place worldwide. Many people avoided regular hospital visits as a result. Many people now need routine home temperature and oxygen level monitoring to maintain track of their health, thereby demanding monitoring devices at home.
Various government programs are supporting the pandemic outbreak. The FDA has granted Emergency Use Authorizations (EUAs) for a few wearables and patient monitoring devices to improve access to medicines, monitor patients more closely, and lessen the risk of SARS-CoV-2 exposure to medical professionals during the COVID-19 pandemic.
The growing popularity of wearable and remote patient monitoring devices is another factor fueling the market’s expansion. By fusing clinical symptomology with vital indicators, wearable technology helps in the diagnosis of many chronic diseases. Thus, there has been a dramatic rise in the usage of wearable technology to combat COVID-19.
The wearable medical device market is anticipated to reach $174.48 Billion by 2030, expanding at a 27.1% CAGR during the forecast period (2022-2030), according to Market Research Future.
MetAlert identifies the total addressable market for its wearable patient monitoring tech for those with Alzheimer’s, dementia, and autism at more than 34 million potential patients in North America, Europe, South Africa, and Asia.
Management Team
Patrick E. Bertagna is Founder, CEO and Chairman at MetAlert. He began his career in apparel sales in 1983 and was promoted to national sales manager within two years. In 1986, he founded his first company importing apparel from Europe and selling to U.S. retailers from JCPenney to Neiman Marcus. He has founded several technology and apparel companies, including MetAlert in 2002, which he took public in 2008. He attended Cal State University Northridge with a business major and a psychology minor.
Louis Rosenbaum is COO of MetAlert. He co-founded Global Trek Xploration and was an initial investor in MetAlert. He has successfully started companies in multiple industries, including apparel, environmental services, and the music industry, achieving annual revenues in the multi-millions of dollars. He previously was president of Elements, a women’s apparel company, and of Advanced Environmental Services.
Alex McKean is CFO at MetAlert. He is also the CFO of Encore Brands Inc., a position he has held since 2009. He has held positions as Controller and VP of Finance at 24:7 Film and InternetStudios.com, Director of FP&A/SVP at Franchise Mortgage Acceptance Company, Corporate Accounting Manager/Treasurer of Polygram Filmed Entertainment and Assistant Treasurer/Controller for State Street Bank. He holds an International MBA from Thunderbird School of Global Management and undergraduate degrees in business and political science from Trinity University.
MetAlert Inc. (OTC: MLRT), closed Monday's trading session at $0.1797, even for the day, on 31 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.06/$1.00.
Recent News
- MetAlert Inc. (OTC: MLRT) - MetAlert, Inc. (MLRT) - Developing New and Practical GPS Tracking and Other Health Tech Solutions for Real-World Caregivers and Businesses
- MetAlert, Inc. (MLRT) Patient Tracking Technology To Gain User Access in Sweden's Cognitive Care Centers
- Developing Heart Disease Earlier in Life Increases Cognitive Decline Later in Life
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
Glioblastoma is an
Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing.
DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future.
Lexaria has a collaborative research agreement with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has filed for patent protection for specific delivery of nicotine, vitamins, NSAIDs, testosterone, estrogen, cannabinoids, terpenes, PDE5 inhibitors (with brand names like Viagra), tobacco and more.
Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats.
The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products.
DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions.
DehydraTECH Technology
Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following:
- Speeds up delivery – the effects of the product are felt by the subject in just minutes.
- Increases bioavailability – the technology is much more effective at delivering a drug or product into the bloodstream.
- Increases brain absorption – animal testing suggests significant improvement in the quantity of drug delivered across the blood-brain barrier.
- Improves drug potency – more of the ingested product is made available to the body, so lower doses are required to achieve the desired effect.
- Reduces drug administration cost – lower doses mean lower overall drug costs.
- Masks unwanted taste – the technology eliminates or reduces the need for sweeteners.
Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery.
Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine.
Market Outlook
Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments:
- Oral Cannabinoids – a market estimated to be worth $18.4 billion in 2021 and expected to reach $46.2 billion by 2025.
- Antivirals – an estimated $52.1 billion market in 2021 that’s expected to grow to $66.7 billion by 2025.
- Oral Mucosal Nicotine – smokeless tobacco products, a $13.6 billion market in 2018, is forecast to grow at 7.2 percent annually through 2025.
- Human Hormones – estrogen and testosterone replacement therapies represented a $21.9 billion market in 2019, with a forecast CAGR of 7.7 percent through 2027.
- Ibuprofen and Naproxen – NSAID sales totaled $15.6 billion globally in 2019 and are projected to reach $24.4 billion by 2027.
- Vitamin D3 – the global market size was $1.1 billion in 2021, growing at 7 percent per year and expected to reach $1.7 billion in 2026.
Management Team
Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations.
John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents.
Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations.
Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch.
Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia.
Lexaria Bioscience Corp. (LEXX), closed Monday's trading session at $2.91, off by 3.3223%, on 33,365 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.80/$4.83.
Recent News
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) To Intensify Focus on Commercial Execution of its DehydraTECH Technology for the 2023 Calendar year
- CannabisNewsBreaks - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) DehydraTECH(TM) Shown to Elevate Drug Delivery Across Blood-Brain Barrier by ~1,900%
- Lexaria Bioscience Corp. (NASDAQ: LEXX) CEO Anticipating 'Best Year Ever' in 2023
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
The announcement reads, “The Las Vegas Roadshow landing will beguided by Knightscope experts, and visitors will be able tointeract directly with each of the ASRs and see the KnightscopeSecurity Operations Center (‘KSOC’) user interface in action.Clients, investors and the media are all welcome to attend to learnmore about Knightscope.”
There is no charge to participate in, or visit, the Roadshow.However, available slots fill up quickly. Appointments arerecommended and can be made by visiting the following link: https://calendly.com/robotroadshow/
To view a short video of a past event hosted by the Los AngelesPolice Department, visit https://ibn.fm/n69H9
To view the full press release, visit https://ibn.fm/3l6fW
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Monday's trading session at $2.345, off by 4.2857%, on 46,378 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.05/$16.50.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Study Finds Boosting White Blood Cell Counts May Improve Treatment Outcomes
- BioMedNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Pleased with Early Results from Global Clinical Trial
- Study Determines That How Proteins Move May Influence Brain Tumor Growth
Knightscope, Inc. (NASDAQ: KSCP)
The QualityStocks Daily Newsletter would like to spotlight Knightscope, Inc. (NASDAQ: KSCP).
To view the full press release, visit https://ibn.fm/bFNC7
Knightscope, Inc. (NASDAQ: KSCP), founded in 2013 and based in Mountain View, California, is a leader in the development of autonomous security capabilities targeting to disrupt the $500 billion security industry. Knightscope’s technology uniquely combines self-driving technology, robotics, artificial intelligence and electric vehicles.
Knightscope designs and builds Autonomous Security Robots (ASRs) that provide 24/7/365 security to the places you live, work, visit and study. The company’s client list covers public institutions and commercial business operations, including multiple Fortune 1000 companies to date. These ASRs have been proven to enhance safety at hospitals, logistics facilities, manufacturing plants, schools and corporations. ASRs act as highly cost-effective complementary systems to traditional security and law enforcement officials, providing an additional advantage by continuing to offer uninterrupted patrolling capabilities across the country.
The company’s ASRs have assisted in the arrest of suspects involved in crimes ranging from armed robbery to hit-and-runs. Their machine-embedded thermal scanning capability even aided in preventing the breakout of a major fire. You can learn more about the crime fighting wins at www.knightscope.com/crime
The company has achieved several milestones since its creation in 2013, including:
- Establishing itself in a 15,000-square-foot facility located in Mountain View, California, in the heart of Silicon Valley, where Knightscope designs, engineers and builds its technology (Made in the USA)
- Operating for more than 1 million hours in the field and securing contracts across five time zones, from Hawaii to Rhode Island
- Raising over $100 million since inception to build its technology from scratch and generating over $13 million in lifetime revenue, validating both the market opportunity and the technology
Growth Capital & Proposed Nasdaq Listing
With backing from more than 28,000 investors and four major corporations and over $100 million raised since inception, Knightscope is poised to be an industry leader in the future of public safety and security.
On December 1, 2021, Knightscope announced the commencement of an offering of up to $40 million of its Class A common stock, with shares to be listed immediately following closing on the Nasdaq Global Market under the ticker symbol ‘KSCP’. The offering is for up to 4 million shares priced at $10 per share. Learn more at www.knightscope.com/investors
Company Mission – Reimagining Public Safety
Knightscope’s long-term vision has an eye on the greater good. The company’s mission is to make the United States of America the safest nation in the world while supporting the 2+ million law enforcement and security professionals across the country.
Crime has an estimated negative economic impact in excess of $2 trillion annually. As crime is reduced, positive impacts will likely be realized across several aspects of society, including housing, financial markets, insurance, municipal budgets, local business and safety in general.
Knightscope CEO William Santana Li was interviewed by Kevin O’Leary, more commonly known as Shark Tank’s Mr. Wonderful. When asked to explain how the benefits provided by the ASRs outrank a human doing the same job, Li said, “First, just the simple presence of a physical deterrent causes criminal behavior to change. Second, the machines are self-driving cars that patrol all around and recharge themselves. They also generate 90 terabytes of data per year. No human would ever be able to process that. The robots are intended to be eyes and ears for the humans, not a one-to-one replacement.”
The Knightscope solution to reduce crime combines the physical presence of ASRs, sometimes referred to as proprietary Autonomous Data Machines, with real-time onsite data collection and analysis. The ASRs are fitted with eye-level 360° cameras, thermal scanning, public address announcements and various other features that work in tandem with humans to provide law enforcement officers and security guards unprecedented situational awareness.

Those 90 terabytes of data are then formatted in a useable way, so law enforcement can leverage that information and execute their responsibilities more effectively.
Public Safety Innovation
The company’s recurring revenue business model is set up to mimic the recurring societal problem of crime, and it takes into consideration the fact that innovation in the security and public safety industry has been stagnant for decades. Because the traditional practices of the sector have remained unchanged for years, automation has potential to drive substantial cost savings – and significant improvement in capabilities.
Human security guards are one of both the largest expenses and the largest liabilities for companies. Knightscope’s robots are offered at an effective price of $3 to $9 per hour, compared with approximately $85 for an armed off-duty law enforcement officer and $15 to $35 for an unarmed security guard.
This innovation has the potential to drive considerable cost savings. Based on these estimates, manufacturing costs can be recovered as soon as the first year of operation.
Product Offerings
The company has nine patents and a framework of unique intellectual property. Knightscope currently offers a K1 stationary machine, a K3 indoor machine and a K5 outdoor machine. A K7 multi-terrain four-wheel version is in development.

The ASRs autonomously patrol client sites without the need for remote control, providing a visible, force multiplying, physical security presence to help protect assets, monitor changes in the area and deter crime. The data is accessible through the Knightscope Security Operations Center (KSOC), an intuitive, browser-based interface that enables security professionals to review events generated by the ASRs providing effectively ‘mobile smart eyes and ears’. Learn more at www.knightscope.com/ksoc
The ASRs and the related technologies were developed ground up by the company and are Made in the USA.
The Robot Roadshow

Knightscope has created the ultimate hybrid physical and virtual event, bringing its Autonomous Security Robot technologies to cities across the country for interactive and in-person demonstrations.
Each roadshow landing is hosted virtually by a Knightscope expert, and visitors can interact directly with each of the company’s ASRs and see the Knightscope Security Operations Center (KSOC) user interface in action. Learn more at www.knightscope.com/roadshow
Management Team
Chief Executive Officer William Santana Li is a veteran entrepreneur, a former executive at Ford Motor Company and the founder of GreenLeaf, a company that grew to be the world’s second-largest automotive recycler and is now part of LKQ Corporation (NASDAQ: LKQ).
Chief Client Officer Stacy Dean Stephens brings his experience as a former Dallas law enforcement officer, as well as his skills as a seasoned entrepreneur, to assist on the client acquisition side.
Chief Intelligence Officer Mercedes Soria is an award-winning technologist and former Deloitte software engineer.
Chief Design Officer Aaron Lehnhardt brings over two decades of two- and three-dimensional product and industrial design in modeling and VR to the table, on top of his experience as a senior designer at Ford Motor Company.
Chief Financial Officer Mallorie Burke is a seasoned financial executive and strategic advisor for both private and publicly traded technology companies with a successful track record of mergers & acquisitions, corporate growth and exit strategies, including public listings.
General Counsel Peter Weinberg leverages 30 years of diverse corporate counsel experience, spanning from startups to well-established companies, private and public. He has significant experience training personnel at all levels in critical areas to improve corporate compliance and productivity.
Knightscope, Inc. (NASDAQ: KSCP), closed Monday's trading session at $1.62, off by 2.4096%, on 272,694 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.35/$9.95.
Recent News
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Robot Roadshow to Take the Spotlight in Vegas
- InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Inks Five New Contracts for More than a Dozen Machines
- InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Continues Momentum with Five New Contracts
Cepton Inc. (NASDAQ: CPTN)
The QualityStocks Daily Newsletter would like to spotlight Cepton Inc. (NASDAQ: CPTN).
Despite legislative requests from stakeholders, legislators andadvocates, the
Cepton Inc. (NASDAQ: CPTN) is a provider of state-of-the-art, intelligent, lidar-based solutions serving a range of markets, including automotive (ADAS/AV), smart cities, smart spaces and smart industrial applications. General Motors (NYSE:GM) has granted a series production award for Cepton’s lidar, the biggest such award to date in the automotive space. Cepton’s is the lidar component of GM’s Ultra Cruise autonomous driving platform. By leveraging its patented Micro Motion Technology (MMT®) lidar platform, the company develops reliable, scalable and cost-effective solutions that deliver long-range, high-resolution 3D perception for smart applications.
Cepton was established in 2016 by co-founders Dr. Jun Pei and Dr. Mark McCord. The company is headquartered in San Jose, California, and serves a fast-growing customer base through an international presence spanning North America, Germany, Japan, India and China.
Micro Motion Technology (MMT®)
Cepton was built from the ground up to meet key lidar industry challenges for mass market adoption. This company’s portfolio of proprietary technology is uniquely aimed at facilitating this industry growth through a combination of performance, reliability, affordability and design integration.
Key among its innovations is MMT®, a mirrorless, frictionless, rotation-free 3D imaging platform designed specifically for lidars. Its benefits for OEMs and system integrators include:
- Reliability – The durable design uses common, easily attainable materials.
- Versatility – The platform is capable of achieving near- to ultra-long range with a wide field of view.
- Efficiency – MMT® features a compact form factor, low power usage and inexpensive components.
- Scalability – Its simple design means that scale-up to high manufacturing volumes is easily attainable.
Because of their compact form factor, Cepton lidars are embeddable and ideally suited for advanced driver-assistance system (ADAS) integration, whether behind windshield, in headlamp or in fascia.
Agreement with KOITO
KOITO Manufacturing Co. Ltd., the world’s premier Tier 1 auto lighting supplier, originally started an evaluation of Cepton’s MMT® based lidars in 2018. In 2020, KOITO made an investment in Cepton aimed at accelerating the company’s development and enabling KOITO’s industrialization of high-performance and high reliability lidar sensors for ADAS and autonomous vehicle (AV) applications.
Through this collaboration, Cepton was able to secure the largest ADAS lidar series production award[1] with General Motors as a sole source in the automotive space. The award covers GM vehicles for the initial period of 2023-2027.
On August 5, 2021, the two companies deepened their relationship when KOITO committed to invest a further $50 million in Cepton’s business through its participation in a Private Investment in Public Equity (PIPE) offering of shares of common stock of Growth Capital Acquisition Corp. in connection with Cepton’s recent merger.
Collaboration with GM
On July 13, 2021, Cepton announced that it had secured an ADAS lidar series production award from a leading, Detroit-based global automotive OEM – the biggest lidar production award by any OEM to any lidar company. It was later clarified that the OEM was General Motors, and Cepton’s lidar is part of GM’s ADAS Ultra Cruise system.
GM is “expected to deploy Cepton lidars in its next generation of advanced driver assistance systems (ADAS) across multiple vehicle classes and models – not just luxury cars.” As such, the agreement marks the potential for “an industry-first, mass-market adoption of lidar technology for automotive ADAS, with an anticipated deployment in consumer vehicles starting in 2023.”
On July 28, 2021, Ford Motor Company (NYSE: F) distributed an article on Medium noting, “Ford has been engaged with Cepton almost since their inception in 2016, both for R&D collaboration and small-scale deployments. Cepton LiDAR are deployed in some of [Ford’s] smart city projects. Based on Ford’s guidance, Cepton delivered a custom version of their LiDAR to enable R&D on advanced ADAS features.”
Market Outlook
Driven by increasing development and adoption in automobile safety applications, environmental mapping and 3D-modeling, the global lidar market is forecast to experience considerable growth over the coming years. A research report published by MarketsAndMarkets suggests that the sector will grow to an estimated $3.4 billion by 2026, achieving a CAGR of 21.6% over the next five years.
The report further highlights increasing investments in lidar startups by automotive giants as a driver of growth opportunities in the sector, particularly in North America.
In 2020, ground-based lidar accounted for the lion’s share of the overall lidar market, and this trend is expected to continue as the automotive sector continues to rapidly advance adoption across the full spectrum of vehicle classes. One factor not to be underestimated is the high barrier of entry and the exceptionally long time required for automotive OEMs to vet and award a production win to a lidar company. It is a commonly held view that the over 50 lidar companies will inevitably coalesce into a handful serving all OEMs.
Cepton, having a head start through its established partnership with leading global OEM GM, is uniquely positioned to capitalize on this market growth in the years to come.
Management Team
Cepton’s founder-led team is made up of lidar industry pioneers with decades of collective experience across advanced lidar and imaging technologies.
Jun Pei, Ph.D., is the company’s CEO and Co-Founder. He is a technology specialist with a focus in optics and electronics. Prior to founding Cepton, Dr. Pei founded AEP Technology, a firm focused on developing advanced 3D optical instruments. He received his Ph.D. in electrical engineering from Stanford University.
Mark McCord, Ph.D., is Cepton’s CTO and Co-Founder. Prior to founding Cepton, he led advanced development at KLA-Tencor. Dr. McCord also formerly served as an associate professor at Stanford University, where he earned his Ph.D. in electrical engineering.
Winston Fu, Ph.D., is the company’s CFO. Dr. Fu is the founder of Silicon Valley venture capital firm LDV Partners. Prior to joining Cepton, he served as CFO and Chairman of Active-Semi before its acquisition. Dr. Fu has also helped to build many technology companies as an entrepreneur and/or board member. He received his Ph.D. in applied physics from Stanford University, as well as an MBA from the Kellogg School of Management at Northwestern University.
[1] Largest known ADAS lidar series production award based on number of vehicle models awarded
Cepton Inc. (NASDAQ: CPTN), closed Monday's trading session at $1.26, off by 3.8168%, on 1,415,809 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.01/$80.16.
Recent News
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Wins Largest Commercial Lidar Deployment Contract in Tolling Sector to Date
- InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Announces Participation at Upcoming Conferences
- InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Completed $100M Koito Manufacturing Investment
India Globalization Capital Inc. (NYSE American: IGC)
The QualityStocks Daily Newsletter would like to spotlight India Globalization Capital Inc. (NYSE American: IGC).
Aurora, Colorado, celebrated the official opening of
India Globalization Capital Inc. (NYSE American: IGC), through subsidiary IGC Pharma, develops, patents, and markets advanced THC-based drug formulations for the treatment of symptoms related to various diseases including but not limited to Alzheimer’s disease, Tourette syndrome, chronic pain, and pet seizures.
IGC’s leading drug candidate, IGC-AD1, has completed Phase 1 of a safety and tolerability trial and entered Phase 2 trials for treating agitation in patients with Alzheimer’s dementia, the first study in humans of a natural tetrahydrocannabinol (THC) compound plus another molecule (www.clinicaltrials.gov). As of September 2022, the IGC trial is the only ongoing Phase 2 trial of a natural THC-based formulation on Alzheimer’s patients.
The company’s other drug candidate, TGR-63, is an enzyme inhibitor that has shown in preclinical trials the potential to reduce neurotoxicity in Alzheimer’s cell lines. Both drug candidates have shown their ability to ameliorate beta amyloid plaques in Alzheimer’s cell lines and improve memory in Alzheimer’s mouse models. Beta amyloid plaques are a key hallmark of Alzheimer’s and an important target of Alzheimer’s pharmaceutical drug development.
Neuro Psychiatric Symptoms (NPS) are not only debilitating for Alzheimer’s patients; they also place an immense emotional burden on their caregivers. Beyond reducing symptoms, IGC-AD1’s active molecules and TGR-63 have also shown promise in preclinical trials to reduce important hallmarks of Alzheimer’s including plaques and tangles, as well as improving the treatment of memory loss.
Over the past eight years, the IGC team has amassed a deep knowledge of cannabinoid science, including extraction, isolation, purification, and development. The company’s strategy is to leverage its unique end-to-end capabilities, platform, and expertise to develop a class-leading program and bring it to market quickly and cost efficiently to treat neurodegenerative diseases such as Alzheimer’s.
The company also has a family of cannabidiol (CBD)-based consumer products (www.Holief.com) such as pain relief creams, pain relief gels, purpose gummies, tinctures, and capsules targeting women’s wellness, with a particular focus on premenstrual syndrome (PMS) and dysmenorrhea (period cramps). In addition, the company targets individuals that need sleep-aids with its specially formulated low melatonin cannabinoid gummies.
IGC has also introduced a low-calorie CBD- and caffeine-infused energy beverage brand (www.SundaySeltzer.com) that is currently available for purchase. The company’s brands are founded on the belief that effective natural solutions should be affordable and accessible to everyone. As the demand for natural products targeting women’s wellness and energy drinks continue to grow, these products are seeing strong traction in the market.
The company operates three facilities – a large GMP (Good Manufacturing Production Standards) certified facility that includes extraction, distillation, and manufacturing, in Washington State; a GMP-211 (pharmaceutical) grade facility in Maryland; and a facility licensed for controlled substances including cannabis in Bogota, Colombia, with complete access to legal licensed cannabis where the company conducts its testing.
In addition, the company’s development under Magistral Formulations is approved by INVIMA (Colombia National Food and Drug Surveillance Institute) to treat neurological disorders, non-oncological chronic pain, and mental disorders.
IGC’s intellectual property (IP) portfolio comprises of eight patents that it controls and seven patent applications. The portfolio includes #11,446,276, a patent for extreme low dose THC treatment of Alzheimer’s that was granted in September 2022.
The company is headquartered in Potomac, Maryland.
IGC-AD1
IGC-AD1 is the company’s leading drug candidate for the treatment and relief of Alzheimer’s symptoms. A significant amount of research on Alzheimer’s cell lines has shown that the active agents in IGC-AD1 reduce plaques and neurofibrillary tangles that are the hallmarks of Alzheimer’s. Further, micro-dosing of THC, as shown in cell lines, could increase the functioning of mitochondria and potentially promote the growth of new neural pathways (neurogenesis). The research shows that micro-dosing of THC affects the brain radically differently from the normal higher dosing of THC.
While there is a significant body of research showing that THC is neuro-toxic at normal levels of dosing, micro-dosing of THC has been shown to be non-toxic to neurons. With the results of these preclinical studies, the company developed an oral formulation, IGC-AD1. The company recently completed a safety and tolerability Phase 1 trial on Alzheimer’s patients and has initiated a Phase 2, multi-site, double-blind, randomized, placebo-controlled trial of the safety and efficacy of IGC-AD1 on agitation in participants with dementia due to Alzheimer’s disease at sites in the U.S. and Canada. IGC expects the Phase 2 trial to take between 9 and 12 months to complete, barring unknown factors such as, for example, a resurgence of COVID and the enforcement of lockdowns and travel restrictions.
With further successful trials and FDA approvals, IGC hopes to bring a drug based on natural THC as an effective treatment for agitation in Alzheimer’s to market.
TGR-63
The company’s other molecule, TGR-63, has been shown to reduce the neurotoxicity that impacts memory loss in preclinical trials with mice. On a dose dependent manner, transgenic Alzheimer’s mice treated with TGR-63 showed improvement in memory relative to control.
Both drug candidates, IGC-AD1 and TGR-63, have shown their ability to reduce the brain plaques associated with memory loss in Alzheimer’s in mice.
With further successful trials and FDA approvals, IGC hopes to bring TGR-63 as a treatment for Alzheimer’s disease to market.
Market Opportunity
Alzheimer’s disease impacts over 55 million people worldwide and about 5.5 million individuals in the U.S. Over 70% of these patients face debilitating symptoms, including anxiety, depression, and agitation (Mendez, 2021). Agitation in dementia patients can include excessive physical movement and verbal activity, restlessness, pacing, belligerence, aggression, screaming, crying, and wandering.
In 2020, the estimated healthcare costs for Alzheimer’s disease in the U.S. were $305 billion. Medicare and Medicaid covered about 70% of those costs, leaving considerable burden on patients and families. At the current rate of growth of Alzheimer’s and other dementia diagnoses, those costs are estimated to reach over $1 trillion by 2050.
Currently, there are no FDA-approved medications to alleviate the symptoms of dementia due to Alzheimer’s disease, providing a tremendous opportunity for formulations that can have an impact on quality of life and disease progression.
Management Team
Richard Prins has been chairman at IGC since 2012 and served as an independent director since 2007. From March 1996 to 2008, he was the Director of Investment Banking at Ferris, Baker Watts, Incorporated. Prins served in a consulting role to RBC until January 2009. He currently volunteers full time with a non-profit organization, Advancing Native Missions, and is a private investor. Since February 2003, he has been on the board of Amphastar Pharmaceuticals Inc. He holds a bachelor’s degree from Colgate University and an MBA from Oral Roberts University.
Ram Mukunda is CEO and President of IGC. He has been the chief inventor and architect of most of the company’s patent filings and is responsible for the company’s strategic positioning. Prior to IGC, he was founder and CEO of Startec Global Communications, which he took public in 1997. He served as Strategic Planning Advisor at Intelsat, a communications satellite services provider. From 2001 to 2003, he was a Council Member at Harvard’s Kennedy School of Government, Belfer Center of Science and International Affairs. He was named the 1998 Ernst & Young Entrepreneur of the Year. He holds bachelor’s degrees in electrical engineering and mathematics, and a master’s degree in engineering from the University of Maryland.
Dr. Jagadeesh Rao is the company’s Principal Scientist. His career spans two decades in the public sector and product R&D for Johnson & Johnson. He leads IGC’s scientists in the development of pharmaceutical and OTC products. He worked for the federal National Institutes of Health, and for the National Institute on Drug Abuse. His Ph.D. in Neurochemistry is from the National Institute of Mental Health & Neurosciences in India. He did postdoctoral training at the University of Illinois-Chicago.
Claudia Grimaldi is a Director, Vice President, Principal Financial Officer, and Chief Compliance Officer for IGC. She also serves as a Director/Manager Director for some of the company’s subsidiaries. She graduated with highest honors from Javeriana University in Colombia with a bachelor’s degree in psychology. She holds an MBA, graduating with highest honors, from Meredith College in North Carolina. In addition, she has attended the Darden School of Business Financial Management Executives program and the Corporate Governance Program at Columbia Business School. She is currently pursuing her Directorship Certification with the National Association of Corporate Directors. She is fluent in both English and Spanish.
India Globalization Capital Inc. (NYSE American: IGC), closed Monday's trading session at $0.4101, off by 1.0854%, on 151,899 volume. The average volume for the last 3 months is 144,032 and the stock's 52-week low/high is $0.2785/$1.16.
Recent News
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - FDA Throws CBD Supplements Rulemaking Duty to Congress
- 420 with CNW - New Poll Finds Americans Favor Looser Cannabis Regulation, Tighter Regulation of EVs and Cryptos
- How Emotional Support, Service Dogs Differ
Advanced Container Technologies Inc. (OTC: ACTX)
The QualityStocks Daily Newsletter would like to spotlight Advanced Container Technologies Inc. (OTC: ACTX).
•Data443 Risk Mitigation recently revealed it had submitted andfiled its 2022 Form 10-K with the SEC
•The filing will mark the company’s fourth full year of auditedfinancial statements filed with the SEC
•In turn, this would exempt the company from meeting minimum shareprice requirements imposed by The Nasdaq Capital Market oncompanies created by way of a reverser merger and seeking to uplist on to the index
•A potential uplisting would mark the latest achievement forData443, with the company recently revealing a new contract win aswell as a capital raising designed to fund its future growthendeavors
Advanced Container Technologies Inc. (OTC: ACTX) is in the business of selling and distributing self-contained, automated, indoor “micro-farms” called Grow Pods, along with related equipment and supplies. Additionally, the company designs and sells patented proprietary medical-grade plastic containers, known as the Medtainer®, that store and grind pharmaceuticals, herbs, teas and other solids or liquids.
ACTX is the leading distributor of Grow Pods. With a controlled environment, food and herbs can be grown without pesticides, harmful chemicals or risk of pathogen contamination, and with low energy consumption. Restaurants, grocery stores, non-profits, MSOs and entrepreneurs can use Grow Pods to ensure a fresh supply of ultra-clean produce year-round.
The company entered the Grow Pod business in October 2020 with its acquisition of all shares of Advanced Container Technologies Inc., a California corporation. As of February 28, 2022, ACTX is exploring the acquisition of the assets and the assumption of some or all of the liabilities of GP Solutions Inc., the developer and manufacturer of Grow Pods, for which ACTX is currently the sole U.S. distributor.
Because Grow Pods can be located almost anywhere, produce can be grown closer to the point of consumption and harvested at its peak, providing nutritious fruits and vegetables where needed. Indoor micro-farms, utilizing a practice known as vertical farming, have attracted the attention of governments and universities, which are now promoting vertical farming as a way to combat food insecurity and inequities.
The United States Department of Agriculture (USDA) has stated that vertical farming “is no longer a futuristic concept.” The department is enthusiastic about vertical farming, particularly those utilizing repurposed shipping containers, such as Grow Pods. Arizona State University reports that vertical farming reduces water use by 90 percent compared to conventional farming but produces 10 times the crop yield.
Products
Grow Pods
One of the company’s main business units is focused on selling advanced, self-contained hydroponic containers called Grow Pods. These unique and innovative automated systems are essentially micro-farms that can be placed virtually anywhere and, with their controlled and specially filtered environment, allow cultivation of a wide variety of crops, 365 days a year. The Grow Pod controlled environment offers major advantages for the production of high-value crops. The ability to grow year-round and the ability to cultivate in a smaller footprint using less water and power are some of the primary advantages of the system. Grow Pods offer constant temperature, humidity and airflow control, as well as automated watering and lighting schedules for optimal growth and minimal labor requirements, regardless of crop.
Containers
ACTX meets the needs of the pharmaceutical and medical markets, including the cannabis and hemp industries, with patented packaging systems. The company designs, customizes, brands and sells proprietary medical grade plastic containers that can store pharmaceuticals, herbs, teas and other solids or liquids, with a special built-in feature that can grind solids and shred herbs. The company’s flagship container product is the patented Medtainer®, a child resistant, medical-grade herb container and grinder that is water-tight, air-tight and smell proof. Packaging in the cannabis industry is critical, with numerous stringent regulations about how cannabis products must be packaged and labeled. ACTX also offers custom-branded, compliant vacuum seal bags and other retail container solutions.
Equipment and Supplies
ACTX markets and sells two principal products: Grow Pods, which are specially modified insulated shipping containers manufactured by GP Solutions Inc., in which plants, herbs and spices may be grown hydroponically in a controlled environment, and Medtainers®, which may be used to store pharmaceuticals, herbs, teas and other solids or liquids and can grind solids and shred herbs. The company also markets and sells various products related to Grow Pods and the Medtainer®, as well as providing private labeling and branding services for purchasers of Medtainers® and certain related products.
GP Solutions manufactures and sells other products, such as humidity controllers and LED lighting systems for vertical farming. The company’s specially designed lighting panels are programmed to emit the exact wavelength of light that each crop requires. The system has a daybreak-to-nightfall feature that gives plants the proper chromatic signals to grow rapidly and fruitfully. High efficiency LED light strips supply the crops with a red and blue light spectrum required for photosynthesis in the spectrum that plants need most.
Market Overview
The global vertical farming market is expected to reach $33.02 billion by 2030, according to a new report by Grand View Research. The market is forecast to expand at a CAGR of 25.5 percent from 2022 to 2030, according to Grand View. Escalating production of biopharmaceutical products, including cannabis, is anticipated to drive the market. The building-based segment of the market is expected to register a significant CAGR of 27.8 percent over the projected period. In addition, the climate control segment is expected to see high growth.
The global cannabis packaging market is expected to reach $14.34 billion by 2028, according to analysis by Reports and Data. The analysis forecasts 1,700 percent growth in cannabis users by the end of 2026, with packaging likely observing a whopping 26.42 percent growth in the forecast period. There are significant barriers to entry in the cannabis packaging market, giving an advantage to companies already established in the sector. These barriers include developing a thorough knowledge of the myriad regulations that govern cannabis packaging (which differ in each state), and child-resistance requirements.
Management Team
Douglas P. Heldoorn is the Founder and Chairman of Advanced Container Technologies Inc. He also holds the positions of President, CEO and COO at the company. Mr. Heldoorn has served on the Board of Directors since its inception in 2013. He has also previously held the position of Executive General Manager at Nissan Motor Corp.
Jeffory A. Carlson is CFO and Treasurer of ACTX. Mr. Carlson has also served as the company’s Corporate Controller since 2014.
Advanced Container Technologies Inc. (OTC: ACTX), closed Monday's trading session at $0.375, off by 1.3158%, on 203 volume. The average volume for the last 3 months is 203 and the stock's 52-week low/high is $0.2005/$1.25.
Recent News
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Colorado Opens Recreational Center Built Using Cannabis Taxes
- 420 with CNW - State Data Shows Massachusetts Adult-Use Cannabis Sales Have Reached $4B
- 420 with CNW - Oklahoma Voters to Decide on Cannabis Legalization in March
Data443 Risk Mitigation Inc. (OTC: ATDS)
The QualityStocks Daily Newsletter would like to spotlight Data443 Risk Mitigation Inc. (OTC: ATDS).
rong>Data443 Risk Mitigation Inc. (OTC: ATDS) is a data security and privacy software company for ALL THINGS DATA SECURITY™. The company is committed to organizing the world’s information by identifying and protecting all sensitive data regardless of location, platform or format.
Data443 provides software and services to enable secure data across devices and databases – at rest and in transit – locally, on a network, or in the cloud. With over 10,000 customers in more than 100 countries, Data443 provides a modern approach to data governance and security. The company’s framework helps customers prioritize risk, identify security gaps, and implement effective data protection and privacy management strategies.
Data443 derives revenue primarily from contracts for subscriptions to access its SaaS platforms, and ancillary services provided in connection with its subscription services. In today’s ever-changing environment with unique and complex requirements for data privacy, governance and hybrid workforces, every organization needs to know where all their data is, who has access to it and how sensitive it is. Data443 provides the tools needed to give companies control over their data processing activities, with capabilities for identifying, reporting and migrating or deleting sensitive data.
The company is headquartered in Research Triangle Park, North Carolina.
Products
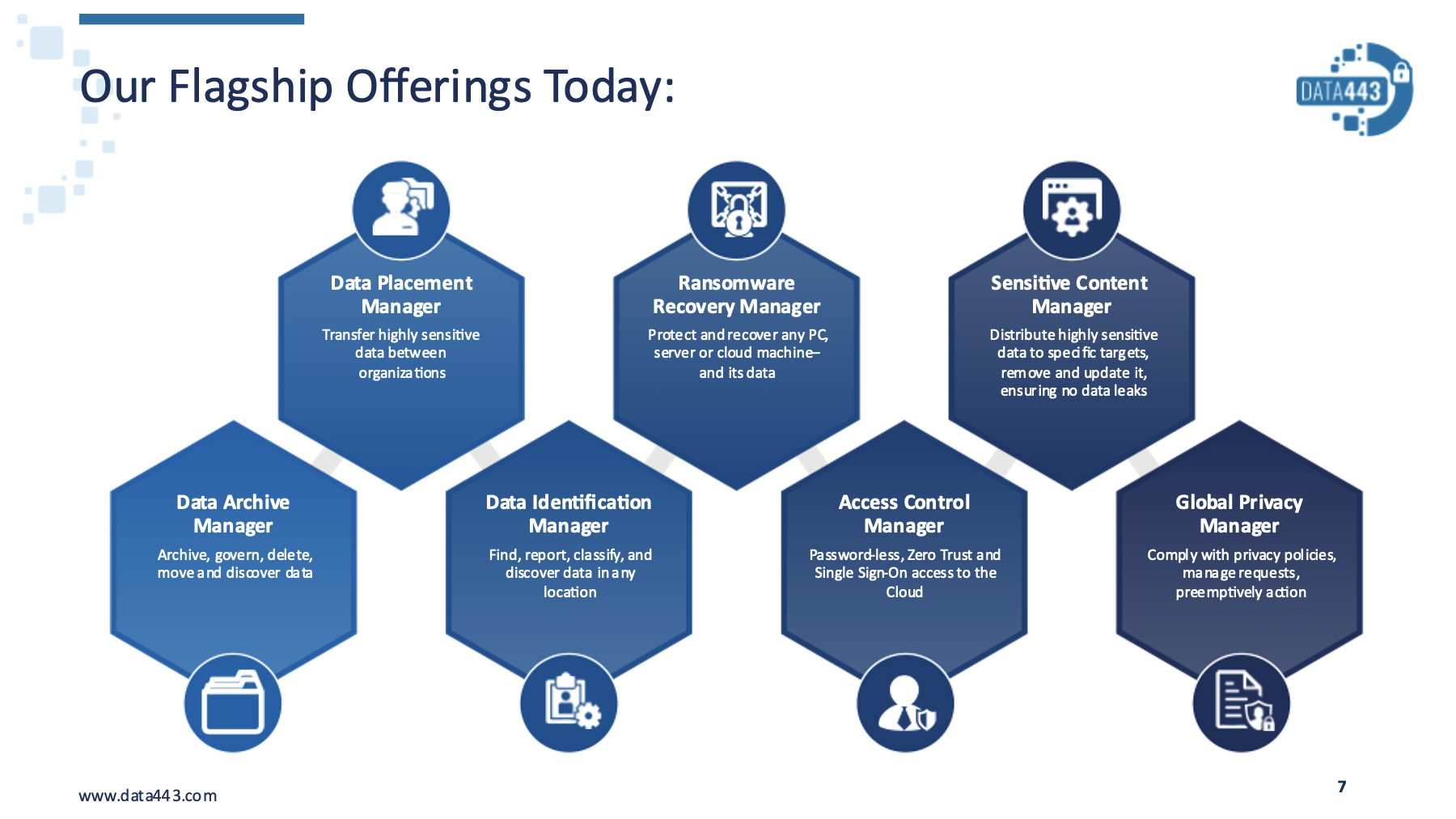
Focused on data security with a privacy-forward methodology, the Data443 product suite delivers solutions designed to securely manage data and data privacy needs on-premises, in the cloud and in hybrid environments. Offerings include:
- Data Identification Manager reduces risk by shining a light on dark data across cloud, on-premises and hybrid environments. From a centralized dashboard, Data Identification Manager provides the ability to automatically inventory all data repositories, classify and tag all data, and enable global search and discovery – all through an agentless deployment.
- Data Placement Manager quickly and securely transfers sensitive data over any public or private network. Available as an HP Nonstop server-based application and for Windows, Linux or any public cloud provider, Data Placement Manager enables the scheduling, routing, formatting and transfer of business-critical data.
- Data Archive Manager is an “all information, anywhere” archiving solution designed to handle and manage all types of privacy requests across cloud, on-premises and hybrid environments. With over 15 years operational history and hundreds of clients managing millions of mailboxes, the platform is purpose-built for information archiving, retention and privacy request management.
- Data Hound™ is a data discovery, classification and capture toolset that enables organizations to perform quick scans, detailed reporting and subsequent data actions based on policy.
- Ransomware Recovery Manager is the only industry solution that actively recovers the device, operating system and data with a simple reboot. Using patented, proven technology, the product produces 100% effectiveness for the whole device and datasets.
- Access Control Manager provides user ID and passwordless access to quickly enable trust across an organization’s entire ecosystem. Its unique architecture allows it to leverage multiple distributed authoritative sources to understand and resolve a typical access request – with the ability to enable or deny the action on the fly.
- Global Privacy Manager provides organizations one comprehensive view, for all privacy requirements, across all enterprise data, all at once. This unmatched visibility into an organization’s data assets ensures that all private and sensitive data can be identified and protected and that enterprises can obey all relevant privacy laws in any jurisdiction.
- Sensitive Content Manager is a security-centric collaboration service designed to give organizations the tools needed for successful content sharing, collaboration and safe distribution with full enterprise management in mind. With a continuous sync feature, encrypted data is automatically downloaded and updated in real time – regardless of location – ensuring that users have the most accurate data available.
Market Outlook
A report from Allied Market Research estimates that the global data security market was worth about $19 billion in 2021 and is projected to reach a value of $54.23 billion by 2027. That represents a CAGR of more than 18% for the forecast period, making data security one of the hottest areas within IT.
Separately, Fortune Business Insights estimates the global data privacy software market is valued at $2.36 billion in 2022 and projects it will grow to $25.85 billion by 2029. That represents a CAGR of 40.8% over the forecast period.
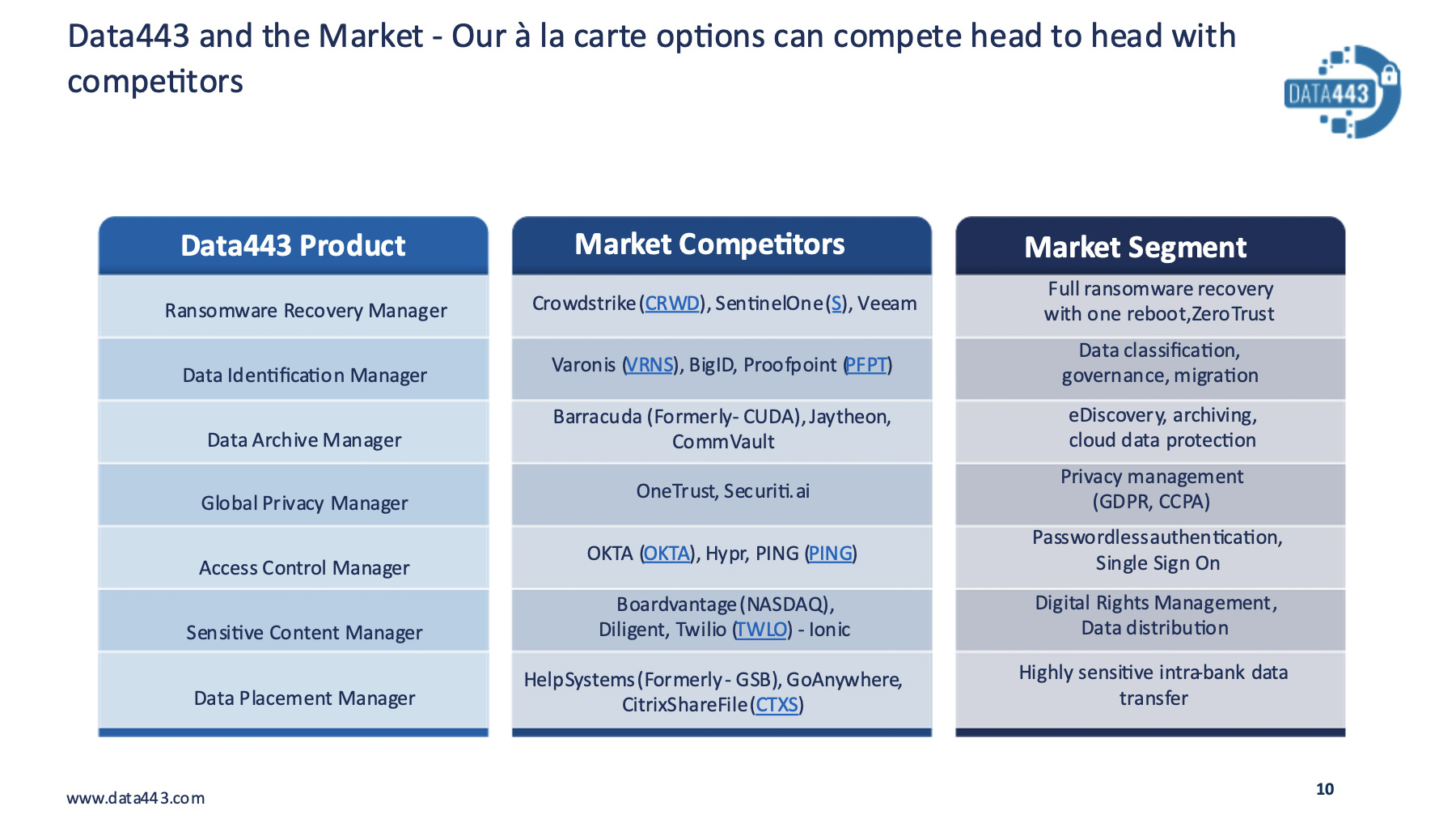
Management Team
Jason Remillard is President, CEO and Founder of Data443. He is responsible for overseeing global expansion, management, execution and corporate development. With over 25 years in global enterprise and B2C software sales and marketing, he brings deep leadership and technical experience, having spent previous time at Fortune 500 companies such as Deutsche Bank, TD Bank, IBM & Merrill Lynch.
Greg McCraw is CFO at Data443. He has over 25 years of experience helping businesses strengthen their accounting and finance operations. He previously served as Vice President of Finance for a dental services organization active in acquisitions, and, prior to that, he was managing director of a boutique accounting and finance consulting firm advising Fortune 500 clients in pharmaceutical, financial services, and private equity sectors on how to execute on regulatory and compliance solutions.
Bennett Pursell is Data443’s Chief Technology Officer. He has over 20 years of experience in IT architecture, security governance and systems integration. Prior to his role at Data443, he served as Head of Technology Architecture at Moody’s Investor Services and was Vice President and Technical Architect of Cloud Computing at Deutsche Bank, along with a host of technical and project management roles dating back to 2006, after starting his career as a web developer with a few startups and running research labs.
Kirill Kashigin is Chief Software Architect at Data443. He leads the development and quality teams, and serves as technical adviser and subject matter expert, bringing vast technical knowledge on privacy management and data security. Formerly the CTO of FileFacets, he has nearly 20 years in development of high-performance systems and deployment.
Data443 Risk Mitigation Inc. (OTC: ATDS), closed Monday's trading session at $0.1255, off by 13.4483%, on 65,820 volume. The average volume for the last 3 months is 53,794 and the stock's 52-week low/high is $0.1255/$6.99.
Recent News
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Files Form 10-K, Announces Plan to Potentially Upgrade Listing to the Nasdaq Capital Market
- InvestorNewsBreaks - Data443 Risk Mitigation Inc.'s (ATDS) Product Suite Supporting Impressive Slate of Clients
- Data443 Risk Mitigation Inc. (ATDS) Announces New Contract Wins; Raises Capital to Fund Future Growth Agenda
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Medical Advisory Board Chair Coauthors Review of Early Stage Dupuytren's Disease Treatment
- Aditxt Inc. (NASDAQ: ADTX) - BioMedNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Reaches Pivotal Milestone to Advance ADI (TM) Technology
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Colorado Opens Recreational Center Built Using Cannabis Taxes
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Releases Video Featuring Successful Global Enterprise Learning Solution Case Study
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Signs $5.2M of Definitive Investment Agreements
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Norway Discovers Huge Mineral Deposits On Seabed
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Announces Participation in Maxim Group Healthcare IT Investor Conference on January 25
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Animal Study Results Featured in Aegis Capital Corp. Updated Price Target
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Partners with Leading CRO after Proprietary NeuroCap Performs Well in Extensive Testing
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - MiningNewsBreaks - Why Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Is 'One to Watch'
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Announces Company First with Kivu Consulting Partnership
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Study Finds Boosting White Blood Cell Counts May Improve Treatment Outcomes
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Helping Companies to Affordably Transition from Dependence on Carbon Fuel
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Driving Growth While Focusing on Positive Environmental, Social Impacts
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc.’s Long Relationship with US Government Demonstrates Its Exceptional Abilities in Backcountry Aviation
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Receives Approval for First-in-Human Dosing of Proprietary DMT Molecule
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Preliminary 2022 Operating Results, Eyes Continued Growth of B2B Revenue
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Files Form 10-K, Announces Plan to Potentially Upgrade Listing to the Nasdaq Capital Market
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Announces New Commercial Customer Engagements, Cross-Platform Product Enhancements at Qubits 2023
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Study Looks into Effects of Hallucinogenic Mushrooms on Bipolar Disorder
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) VTS Debuts New Products, Now Has 'Complete Portfolio of Proven Solutions'
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Grants RSUs as Part of Long-Term Incentive Plan
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Bill Gates Lauds Green Energy Industries in West Virginia
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) at Epicenter of Green-Energy Transformation
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Reports on Company Status in Q1 2023 Shareholder Letter
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Q3 2023 Financial Results
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Enters Option, JV Agreement Regarding Lithium Claims
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Participation at Upcoming A.G.P. Conference
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT), Fr8App Partners with Leading Mexico-Based Trucking Company to Expand Services
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc.’s SmartGreen(TM) Helping Homeowners Realize Unparalleled Energy Efficiency, Lower Utility Bills
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Issues Update on Restatement of Previously Issued Financial Statements
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc.'s (NASDAQ: BEAT) Distinctive Solutions Positioned to Transform the Way Patients, Doctors Detect Heart Attacks
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Wins Largest Commercial Lidar Deployment Contract in Tolling Sector to Date
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) at Forefront of Ushering in Next-Gen Inverter Tech
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Names New Key Execs
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - FDA Throws CBD Supplements Rulemaking Duty to Congress
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Key Milestones, 2023 Business Outlook
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Announces Reappointment of Dr. Hector Alila to Establish Advisory Group
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Robot Roadshow to Take the Spotlight in Vegas
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) To Intensify Focus on Commercial Execution of its DehydraTECH Technology for the 2023 Calendar year
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - MiningNewsBreaks - Why McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Is 'One to Watch'
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - MetAlert, Inc. (MLRT) - Developing New and Practical GPS Tracking and Other Health Tech Solutions for Real-World Caregivers and Businesses
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - MYDECINE INNOVATIONS GROUP FILES PROSPECTUS SUPPLEMENT AND ANNOUNCES CLOSING UNDER SHARE SUBSCRIPTION AGREEMENT
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures ~$8.4M in Registered Direct Offering and Concurrent Private Placement
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Partners with Loop Global, Menzies Aviation on LAX Pilot Program
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) Partners with Minor Figures at Flagship Venice Beach Location
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) to Showcase iGaming Software Solutions at ICE-London
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Joins California's Public-Private Hydrogen Hub Consortium
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Names Seasoned Veteran as New Chief Business Officer
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces Record Order, Eyes Strong Commercial Traction for UltraShear Nanoemulsions Technology Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Receives Approval Recommendation for Retail Cannabis Outlet by San Diego Planning Group
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Mucus-Eating Gut Bacteria May Cause Fever Following Cancer Treatment
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Are Marijuana Penny Stocks A Buy In 2023? 3 To Watch Now
- REZYFi, Inc. - 420 with CNW — Predictions, Trends for Cannabis Industry This Year
- RYAH Group Inc. (CSE: RYAH) - SEDAR filing of the unaudited Interim Financial Statements to September 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - Are These Relatively Little-Known Organic And Earth-Friendly Food Stocks On Your Watchlist?
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Leveraging Innovation, Creativity to Capture Profitable Market Share
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) Notes Importance of Cybersecurity for Cannabis Industry Operational Technology as Hybrid Work Models Expand
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Expands IP Portfolio with New USPTO Patent
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Enters New Vertical Market with MCPTT Order from Hospitality Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Targets Ideal Timing for TapouT Energy Launch
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Installs Carbon Capture Pilot Plant
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Appoints Seasoned Executive as CFO
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - Beyond Alternatives Company of TAAT® to Launch "Jayvees" Brand at CHAMPS Las Vegas Trade Show
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces Additions Sprouts Farmers Market Offerings
- Tingo Inc. (OTC: TMNA) - Five Industries Making Use of Blockchain Tech
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Is 'One to Watch'
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Paid $17.85M from DOE to Supply Strategic Uranium Reserve Program
- Utopia VR - InvestorNewsBreaks – Why Utopia VR Is ‘One to Watch’
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) to Host Q3 Fiscal 2023 Results Call and Webcast
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Regains Compliance with Nasdaq Listing Requirements
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies to Host an Investor Webinar
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Medical Advisory Board Chair Coauthors Review of Early Stage Dupuytren's Disease Treatment
- Aditxt Inc. (NASDAQ: ADTX) - BioMedNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Reaches Pivotal Milestone to Advance ADI (TM) Technology
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Colorado Opens Recreational Center Built Using Cannabis Taxes
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Releases Video Featuring Successful Global Enterprise Learning Solution Case Study
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Signs $5.2M of Definitive Investment Agreements
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Norway Discovers Huge Mineral Deposits On Seabed
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Announces Participation in Maxim Group Healthcare IT Investor Conference on January 25
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Animal Study Results Featured in Aegis Capital Corp. Updated Price Target
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Partners with Leading CRO after Proprietary NeuroCap Performs Well in Extensive Testing
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - MiningNewsBreaks - Why Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Is 'One to Watch'
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Announces Company First with Kivu Consulting Partnership
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Study Finds Boosting White Blood Cell Counts May Improve Treatment Outcomes
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Helping Companies to Affordably Transition from Dependence on Carbon Fuel
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Driving Growth While Focusing on Positive Environmental, Social Impacts
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc.’s Long Relationship with US Government Demonstrates Its Exceptional Abilities in Backcountry Aviation
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Receives Approval for First-in-Human Dosing of Proprietary DMT Molecule
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Preliminary 2022 Operating Results, Eyes Continued Growth of B2B Revenue
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Files Form 10-K, Announces Plan to Potentially Upgrade Listing to the Nasdaq Capital Market
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Announces New Commercial Customer Engagements, Cross-Platform Product Enhancements at Qubits 2023
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Study Looks into Effects of Hallucinogenic Mushrooms on Bipolar Disorder
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) VTS Debuts New Products, Now Has 'Complete Portfolio of Proven Solutions'
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Grants RSUs as Part of Long-Term Incentive Plan
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Bill Gates Lauds Green Energy Industries in West Virginia
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) at Epicenter of Green-Energy Transformation
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Reports on Company Status in Q1 2023 Shareholder Letter
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Q3 2023 Financial Results
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Enters Option, JV Agreement Regarding Lithium Claims
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Participation at Upcoming A.G.P. Conference
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT), Fr8App Partners with Leading Mexico-Based Trucking Company to Expand Services
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc.’s SmartGreen(TM) Helping Homeowners Realize Unparalleled Energy Efficiency, Lower Utility Bills
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Issues Update on Restatement of Previously Issued Financial Statements
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc.'s (NASDAQ: BEAT) Distinctive Solutions Positioned to Transform the Way Patients, Doctors Detect Heart Attacks
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Wins Largest Commercial Lidar Deployment Contract in Tolling Sector to Date
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) at Forefront of Ushering in Next-Gen Inverter Tech
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Names New Key Execs
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - FDA Throws CBD Supplements Rulemaking Duty to Congress
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Key Milestones, 2023 Business Outlook
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Announces Reappointment of Dr. Hector Alila to Establish Advisory Group
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Robot Roadshow to Take the Spotlight in Vegas
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) To Intensify Focus on Commercial Execution of its DehydraTECH Technology for the 2023 Calendar year
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - MiningNewsBreaks - Why McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Is 'One to Watch'
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - MetAlert, Inc. (MLRT) - Developing New and Practical GPS Tracking and Other Health Tech Solutions for Real-World Caregivers and Businesses
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - MYDECINE INNOVATIONS GROUP FILES PROSPECTUS SUPPLEMENT AND ANNOUNCES CLOSING UNDER SHARE SUBSCRIPTION AGREEMENT
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures ~$8.4M in Registered Direct Offering and Concurrent Private Placement
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Partners with Loop Global, Menzies Aviation on LAX Pilot Program
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) Partners with Minor Figures at Flagship Venice Beach Location
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) to Showcase iGaming Software Solutions at ICE-London
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Joins California's Public-Private Hydrogen Hub Consortium
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Names Seasoned Veteran as New Chief Business Officer
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces Record Order, Eyes Strong Commercial Traction for UltraShear Nanoemulsions Technology Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Receives Approval Recommendation for Retail Cannabis Outlet by San Diego Planning Group
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Mucus-Eating Gut Bacteria May Cause Fever Following Cancer Treatment
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Are Marijuana Penny Stocks A Buy In 2023? 3 To Watch Now
- REZYFi, Inc. - 420 with CNW — Predictions, Trends for Cannabis Industry This Year
- RYAH Group Inc. (CSE: RYAH) - SEDAR filing of the unaudited Interim Financial Statements to September 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - Are These Relatively Little-Known Organic And Earth-Friendly Food Stocks On Your Watchlist?
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Leveraging Innovation, Creativity to Capture Profitable Market Share
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) Notes Importance of Cybersecurity for Cannabis Industry Operational Technology as Hybrid Work Models Expand
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Expands IP Portfolio with New USPTO Patent
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Enters New Vertical Market with MCPTT Order from Hospitality Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Targets Ideal Timing for TapouT Energy Launch
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Installs Carbon Capture Pilot Plant
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Appoints Seasoned Executive as CFO
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - Beyond Alternatives Company of TAAT® to Launch "Jayvees" Brand at CHAMPS Las Vegas Trade Show
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces Additions Sprouts Farmers Market Offerings
- Tingo Inc. (OTC: TMNA) - Five Industries Making Use of Blockchain Tech
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Is 'One to Watch'
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Paid $17.85M from DOE to Supply Strategic Uranium Reserve Program
- Utopia VR - InvestorNewsBreaks – Why Utopia VR Is ‘One to Watch’
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) to Host Q3 Fiscal 2023 Results Call and Webcast
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Regains Compliance with Nasdaq Listing Requirements
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies to Host an Investor Webinar
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Medical Advisory Board Chair Coauthors Review of Early Stage Dupuytren's Disease Treatment
- Aditxt Inc. (NASDAQ: ADTX) - BioMedNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Reaches Pivotal Milestone to Advance ADI (TM) Technology
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Colorado Opens Recreational Center Built Using Cannabis Taxes
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Releases Video Featuring Successful Global Enterprise Learning Solution Case Study
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Signs $5.2M of Definitive Investment Agreements
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Norway Discovers Huge Mineral Deposits On Seabed
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Announces Participation in Maxim Group Healthcare IT Investor Conference on January 25
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - InvestorNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Animal Study Results Featured in Aegis Capital Corp. Updated Price Target
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Partners with Leading CRO after Proprietary NeuroCap Performs Well in Extensive Testing
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - MiningNewsBreaks - Why Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Is 'One to Watch'
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Announces Company First with Kivu Consulting Partnership
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Study Finds Boosting White Blood Cell Counts May Improve Treatment Outcomes
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Helping Companies to Affordably Transition from Dependence on Carbon Fuel
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Driving Growth While Focusing on Positive Environmental, Social Impacts
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc.’s Long Relationship with US Government Demonstrates Its Exceptional Abilities in Backcountry Aviation
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Receives Approval for First-in-Human Dosing of Proprietary DMT Molecule
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Announces Preliminary 2022 Operating Results, Eyes Continued Growth of B2B Revenue
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc. (ATDS) Files Form 10-K, Announces Plan to Potentially Upgrade Listing to the Nasdaq Capital Market
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Announces New Commercial Customer Engagements, Cross-Platform Product Enhancements at Qubits 2023
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Study Looks into Effects of Hallucinogenic Mushrooms on Bipolar Disorder
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) VTS Debuts New Products, Now Has 'Complete Portfolio of Proven Solutions'
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Grants RSUs as Part of Long-Term Incentive Plan
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Bill Gates Lauds Green Energy Industries in West Virginia
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) at Epicenter of Green-Energy Transformation
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Reports on Company Status in Q1 2023 Shareholder Letter
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Q3 2023 Financial Results
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Enters Option, JV Agreement Regarding Lithium Claims
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Participation at Upcoming A.G.P. Conference
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT), Fr8App Partners with Leading Mexico-Based Trucking Company to Expand Services
- Friendable Inc. (FDBL) - Video Sizzle Reel from Friendable Inc. (FDBL) Highlights 360-Degree Music Artist Platform Offering for Independent Artists Looking to Break Record and Manager Control
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - GreenEnergyBreaks - FuelPositive Corp.'s (TSX.V: NHHH) (OTCQB: NHHHF) Onsite System Poised to Disrupt Traditional Ammonia and Green Hydrogen Sectors
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc.’s SmartGreen(TM) Helping Homeowners Realize Unparalleled Energy Efficiency, Lower Utility Bills
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Inks Deal with University of Pittsburgh for Exclusive License to Diabetes Technology
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Issues Update on Restatement of Previously Issued Financial Statements
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc.'s (NASDAQ: BEAT) Distinctive Solutions Positioned to Transform the Way Patients, Doctors Detect Heart Attacks
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Wins Largest Commercial Lidar Deployment Contract in Tolling Sector to Date
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Completes Purchase of Michigan Property for Cannabis Operations
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - GreenEnergyBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) at Forefront of Ushering in Next-Gen Inverter Tech
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Names New Key Execs
- India Globalization Capital Inc. (NYSE American: IGC) - 420 with CNW - FDA Throws CBD Supplements Rulemaking Duty to Congress
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Announces Key Milestones, 2023 Business Outlook
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks – InnerScope Hearing Technologies Inc. (INND) Announces Latest CVS Launch of HearingAssist Brand
- Jupiter Wellness Inc. (NASDAQ: JUPW) - InvestorNewsBreaks - Jupiter Wellness Inc. (NASDAQ: JUPW) Announces Reappointment of Dr. Hector Alila to Establish Advisory Group
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Robot Roadshow to Take the Spotlight in Vegas
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) To Intensify Focus on Commercial Execution of its DehydraTECH Technology for the 2023 Calendar year
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - MiningNewsBreaks - Why McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Is 'One to Watch'
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- MetAlert Inc. (OTC: MLRT) - MetAlert, Inc. (MLRT) - Developing New and Practical GPS Tracking and Other Health Tech Solutions for Real-World Caregivers and Businesses
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - MYDECINE INNOVATIONS GROUP FILES PROSPECTUS SUPPLEMENT AND ANNOUNCES CLOSING UNDER SHARE SUBSCRIPTION AGREEMENT
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Secures ~$8.4M in Registered Direct Offering and Concurrent Private Placement
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Partners with Loop Global, Menzies Aviation on LAX Pilot Program
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) Partners with Minor Figures at Flagship Venice Beach Location
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) to Showcase iGaming Software Solutions at ICE-London
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6B) (OTC: MOTNF) Joins California's Public-Private Hydrogen Hub Consortium
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Names Seasoned Veteran as New Chief Business Officer
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces Record Order, Eyes Strong Commercial Traction for UltraShear Nanoemulsions Technology Platform
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Receives Approval Recommendation for Retail Cannabis Outlet by San Diego Planning Group
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) to Present at Biotech Showcase
- QSAM Biosciences Inc. (OTCQB: QSAM) - Study Finds Mucus-Eating Gut Bacteria May Cause Fever Following Cancer Treatment
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Are Marijuana Penny Stocks A Buy In 2023? 3 To Watch Now
- REZYFi, Inc. - 420 with CNW — Predictions, Trends for Cannabis Industry This Year
- RYAH Group Inc. (CSE: RYAH) - SEDAR filing of the unaudited Interim Financial Statements to September 30, 2022, and Update on the failure-to-file cease trade order by the Ontario Securities Commission
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - Are These Relatively Little-Known Organic And Earth-Friendly Food Stocks On Your Watchlist?
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Leveraging Innovation, Creativity to Capture Profitable Market Share
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) Notes Importance of Cybersecurity for Cannabis Industry Operational Technology as Hybrid Work Models Expand
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Marks New Step in Supporting Customer Quality Throughout Additive Journey
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Expands IP Portfolio with New USPTO Patent
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Enters New Vertical Market with MCPTT Order from Hospitality Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Targets Ideal Timing for TapouT Energy Launch
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Installs Carbon Capture Pilot Plant
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - CannabisNewsBreaks - Sugarmade Inc. (SGMD) Leveraging Portfolio Data to Improve Efficiency, Profit Margins, Top-Line Sales
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Appoints Seasoned Executive as CFO
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - Beyond Alternatives Company of TAAT® to Launch "Jayvees" Brand at CHAMPS Las Vegas Trade Show
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Announces Additions Sprouts Farmers Market Offerings
- Tingo Inc. (OTC: TMNA) - Five Industries Making Use of Blockchain Tech
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - Tryp Therapeutics Sets Stage for IV-Channeled Psilocybin Trials
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Subsidiary Signs New Telemedicine Services Agreement
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Is 'One to Watch'
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Paid $17.85M from DOE to Supply Strategic Uranium Reserve Program
- Utopia VR - InvestorNewsBreaks – Why Utopia VR Is ‘One to Watch’
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) to Host Q3 Fiscal 2023 Results Call and Webcast
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Regains Compliance with Nasdaq Listing Requirements
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Technologies to Host an Investor Webinar
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































