The QualityStocks Daily Stock List
- ContraFect (CFRX)
- Windtree Therapeutics (WINT)
- Gamida Cell Ltd (GMDA)
- CaixaBank (CIXPF)
- Manhattan Bridge Capital (LOAN)
- Groupon (GRPN)
- Altisource Portfolio Solutions (ASPS)
- Near Intelligence (NIR)
- Marathon Digital Holdings Inc. (MARA)
- atai Life Sciences N.V. (ATAI)
- NIO Inc. (NIO)
- FSD Pharma Inc. (HUGE)
ContraFect (CFRX)
QualityStocks, MarketBeat, StockMarketWatch, TraderPower, Marketbeat.com, BUYINS.NET, 247 Market News, Money Wealth Matters, Wall Street Resources, StockOodles, StreetInsider, Streetwise Reports, INO.com Market Report, The Stock Dork, AllPennyStocks and Schaeffer's reported earlier on ContraFect (CFRX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
ContraFect Corporation (NASDAQ: CFRX) (FRA: 22R1) is a clinical-stage biotechnology firm that is engaged in discovering and developing protein and antibody products for treating drug-resistant infectious and life-threating ailments in the U.S.
The firm has its headquarters in Yonkers, New York and was incorporated in 2008, on March 5th by Robert Nowinski. It operates as part of the biotechnology research services industry under the health care sector, in the biotech and pharma sub-industry.
The company serves consumers in the U.S. and is party to a collaboration research agreement with the Rockefeller University which entails identifying new lysin therapy candidates that target gram-negative pathogens. It also established an expanded access program to offer exebacase for treating persistent bacteremia brought about by methicillin-resistant staphylococcus aureus in coronavirus patients.
The enterprise’s product pipeline is made up of an investigational anti-bacterial therapy candidate dubbed CF-370 which is currently undergoing preclinical trials evaluating its effectiveness in treating pseudomonas aeruginosa infections that include surgery carry infections, complicated urinary tract infections, blood stream infections and ventilator associated pneumonia. It also develops an engineered lysin termed CF-296, which is used to treat invasive infections brought about by staph aureus, like osteomyelitis and biofilm-related infections in indwelling devices and prosthetic joints. Furthermore, it also develops an investigational new lysin dubbed CF-301, which is currently undergoing phase 3 clinical trials assessing its efficacy in treating staphylococcus aureus bacteremia, which include endocarditis caused by methicillin-susceptible or methicillin-resistant.
The company recently appointed a new Senior Vice President who brings with him depth of knowledge and experience, which will be helpful in the advancement of the firm’s programs. This move may be good for the company’s growth, which will bring in more investors into the firm.
ContraFect (CFRX), closed Monday's trading session at $2.29, up 154.4444%, on 120,499,976 volume. The average volume for the last 3 months is 12.067M and the stock's 52-week low/high is $0.90/$363.20.
Windtree Therapeutics (WINT)
QualityStocks, TraderPower, MarketBeat, The Stock Dork, StockMarketWatch, StreetInsider, MarketClub Analysis, InvestorsUnderground and InvestorPlace reported earlier on Windtree Therapeutics (WINT), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Windtree Therapeutics Inc. (NASDAQ: WINT) is a medical device and clinical-stage biotechnology firm that is engaged in developing therapies for treating cardiovascular and acute pulmonary ailments.
The firm has its headquarters in Warrington, Pennsylvania and was founded in 1992, on November 6th by James S. Kuo and Evan Myrianthopoulos. Prior to its name change in April 2016, the firm was known as Discovery Laboratories Inc. The firm operates in the healthcare sector, under the biotech and pharma sub-industry and serves consumers in the U.S.
The enterprise is party to a collaboration agreement with the University of Milan-Bicocca, which entails discovering and developing new SERCA2a compounds for treating acute and chronic human heart failure. The enterprise is a subsidiary of Lee’s Pharmaceutical Holdings Ltd.
Windtree Therapeutics’ product pipeline is made up of a formulation developed for treating genetically associated hypertension which is known as Rostafuroxin and is currently undergoing phase 2 clinical trials; a lyophilized surfactant indicated for treating lung injuries caused by the coronavirus infection; an aerosolized surfactant indicated for the treatment of respiratory distress syndrome in premature infants dubbed Aerosurf, which is currently undergoing phase 2 clinical trials; and a drug formulation developed for treating early cardiogenic shock and acute decompensated heart failure dubbed Istaroxime, which is in phase 2a and 2b clinical trials.
The firm recently reported their financial results for the 2021 first quarter, which show that it expanded the participating cites and countries for its istaroxime clinical study and extended their collaboration with the University of Milan-Bicocca. The firm’s CEO noted that they were also focused on developing the business and would continue to advance their current and planned activities, which is bound to bring in more investors into the firm.
Windtree Therapeutics (WINT), closed Monday's trading session at $8.27, up 88.8128%, on 12,066,667 volume. The average volume for the last 3 months is 32.419M and the stock's 52-week low/high is $4.28/$71.50.
Gamida Cell Ltd (GMDA)
StreetInsider, MarketBeat, StockMarketWatch, MarketClub Analysis, Trades Of The Day, QualityStocks and BUYINS.NET reported earlier on Gamida Cell Ltd (GMDA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Gamida Cell Ltd (NASDAQ: GMDA) is a clinical-stage biopharmaceutical firm that is focused on the development of cell therapies to cure severe hematologic illnesses and blood cancers.
The firm has its headquarters in Jerusalem, Israel and was incorporated in 1998 by Menashe Levy and Tony Peled. It operates as part of the scientific research and development services industry, under the health care sector. The firm has two companies in its corporate family and serves consumers in Israel and United States.
The company uses its NAM-based (nicotinamide-based) cell expansion technology to develop next-generation cell therapies for patients with cancer and other debilitating illnesses who need other treatment options. It strives to ensure access to its therapies for patients who can benefit from them.
The enterprise’s product pipeline is comprised of a natural killer cell-based cancer immunotherapy dubbed GDA-201, which is undergoing a phase 2 clinical trial evaluating its effectiveness in treating relapsed or refractory multiple myeloma and non-Hodgkin lymphoma. It is also developing a cell therapy dubbed omidubicel, which is undergoing phase 3 clinical trials testing its efficacy in treating high-risk hematologic malignancies and phase 2 clinical trials evaluating its effectiveness in treating severe aplastic anemia.
The firm plans to seek approval for its omidubicel formulation as an allogeneic hematopoietic stem cell transplant solution, after the FDA agreed to initiating rolling submission for a BLA. The formulation’s approval will not only benefit patients with blood cancers but also bring in more investors into the firm, which will be good for its growth.
Gamida Cell Ltd (GMDA), closed Monday's trading session at $1.12, up 38.7856%, on 32,418,567 volume. The average volume for the last 3 months is 10 and the stock's 52-week low/high is $0.56/$3.66.
CaixaBank (CIXPF)
We reported earlier on CaixaBank (CIXPF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
CaixaBank S.A. (OTC: CIXPF) (BME: CABK) (FRA: 4BCA) is an integrated financial group engaged in the provision of a range of banking products and financial services in Spain and internationally.
The firm has its headquarters in Valencia, Spain and was incorporated in 1904. It operates as part of the banks-regional industry, under the financial services sector. The firm serves consumers around the globe.
The company operates through the Banking & Insurance, Equity Investments and BPI segments. The Banking & Insurance segment core business includes the entire banking business, including corporate banking, retail banking, cash and markets, and the insurance business, primarily carried out in Spain through the branch network and the other complementary channels. It also provides life insurance, pension plans and general insurance products. On the other hand, the Equity Investment segment engages in the equity investment business while the BPI segment comprises all businesses under BPI group. Most of the bank's revenue is derived from traditional banking net interest income, with the remainder coming from fees and commissions.
The enterprise also offers solutions related to security, protection, internationalization, and financing; traditional financial advice, independent advice, and broker services; and distributes non-life and life risk insurance policies; and private banking services. In addition, the enterprise is involved in the real estate business. It has branches and ATMs in Spain as well as in Portugal.
The firm remains committed to generating value for its shareholders while also improving people's lives and contributing to the well-being of society.
CaixaBank (CIXPF), closed Monday's trading session at $3.866786, even for the day, on 10 volume. The average volume for the last 3 months is 37,279 and the stock's 52-week low/high is $2.84389/$4.43.
Manhattan Bridge Capital (LOAN)
Wall Street Resources, MarketBeat, The Bowser Report, Marketbeat.com, StreetInsider, QualityStocks, The Online Investor, StreetAuthority Daily, ProfitableTrading, Market FN, One Hot Stock, Dividend Opportunities, StockOodles, The Best Newsletters, The Street, TopStockAnalysts and Stock Research Newsletter reported earlier on Manhattan Bridge Capital (LOAN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Manhattan Bridge Capital Inc. (NASDAQ: LOAN) is a real estate finance firm that is focused on originating, servicing, and managing a portfolio of first mortgage loans.
The firm has its headquarters in Great Neck, New York and was incorporated in 1989 by Assaf N. Ran. It operates as part of the REIT-Mortgage industry, under the real estate sector. The firm primarily serves consumers in the United States.
The company’s strategy is to generate above average, consistent returns while maintaining low risk. It seeks unique and time sensitive opportunities that are underserved by existing lenders. The company remains committed to preserving capital through all steps of the investment and asset management process.
The enterprise provides short-term, secured, and non-banking loans to real estate investors to fund their renovation, acquisition, development or rehabilitation of residential or commercial properties in the New York metropolitan area, including New Jersey and Connecticut, as well as in Florida. Its loans are primarily secured by collateral consisting of real estate and accompanied by personal guarantees from the principals of the borrowers. The enterprise qualifies as a real estate investment trust for federal income tax purposes. It generally would not be subject to federal corporate income taxes if it distributes at least 90% of its taxable income to its stockholders.
The company, whose latest financial results show significant increases in its revenues, remains focused on generating consistent and stable returns for its shareholders. This will positively influence investments into the company as well as boost its overall growth.
Manhattan Bridge Capital (LOAN), closed Monday's trading session at $5.03, even for the day, on 37,292 volume. The average volume for the last 3 months is 1.646M and the stock's 52-week low/high is $4.93/$6.14.
Groupon (GRPN)
The Street, InvestorPlace, MarketClub Analysis, StreetInsider, StocksEarning, Schaeffer's, Jason Bond, StreetAuthority Daily, Barchart, Real Pennies, MarketBeat, TopStockAnalysts, INO.com Market Report, Zacks, StockEarnings, Street Insider, Greenbackers, Cabot Wealth, Hit and Run Candle Sticks, Market Intelligence Center Alert, Profit Confidential, Daily Trade Alert, Dividend Opportunities, ProfitableTrading, Trades Of The Day, Wealth Insider Alert, Marketbeat.com, The Motley Fool, Wealth Daily, All about trends, Money Morning, Wyatt Investment Research, Investing Futures, Louis Navellier, The Online Investor, SmallCap Network, Uncommon Wisdom, Trading Concepts, Investopedia, SmarTrend Newsletters, Daily Wealth, MarketWatch, StockTwits, Penny Stock Rumble, BUYINS.NET, The Growth Stock Wire, WStreet Market Commentary, Wall Street Daily, InvestorGuide, Trade of the Week, StockOrange, AllPennyStocks, The Weekly Options Trader, Stockhouse, QualityStocks, Quant Ratings Team, The Best Newsletters, Energy and Capital, Daily Market Beat, CustomerService, Kiplinger Today, CNBC Breaking News, Market FN, ChartPoppers, Investor Update, Investment Contrarians, Insider Wealth Alert, Dynamic Wealth Report, Penny Sleuth, Top Pros' Top Picks, MarketTamer, Stock Beast, SmallCapVoice, SmallCapNetwork, Super Stock Investor, TradingAuthority Daily, Stock Stars, Penny Detectives, Money and Markets, Millennium-Traders, WiseAlerts, MonsterStocksPicks, Eagle Financial Publications, The Momentum Traders Network, Top Stock Picks, YOLOTraderAlerts, Trader Prep, TradingMarkets, Traders For Cash Flow, Wall Street Mover, AnotherWinningTrade, BestChartNow, WhisperFromWallStreet, Weekly Wizards, Daily Profit, ShazamStocks, MarketClub, MarketArmor.com, Market Wrap Daily, National Inflation Association, OTC Showcase, OTCJournal, StockMarketWatch, Investors Alley, FeedBlitz, SpeculatingStocks, Investing Lab, Stock Market Media Group, Stock Research Newsletter, Penny Stock Buzz, Goldman Small Cap Research, FNNO Newsletters and Otcstockexchange reported earlier on Groupon (GRPN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Groupon Inc. (NASDAQ: GRPN) (LON: 0R1H) (BMV: GRPN) is a global scaled two-sided marketplace that connects consumers to merchants.
The firm has its headquarters in Chicago, Illinois and was incorporated in 2008 by Bradley A. Keywell, Eric Paul Lefkofsky and Andrew D. Mason. Prior to its name change in October 2008, the firm was known as ThePoint.com Inc. It operates as part of the internet content and information industry, under the communication services sector. The firm serves consumers across the globe.
The company operates through the North America and International segments. It allows consumers to access its marketplace through its mobile applications and its websites, through localized groupon.com sites in approximately thirteen countries. The company generates revenue from the take rate on the vouchers' purchase and/or usage. Geographically, more than 60% of its revenue comes from North America.
The enterprise provides goods and services through its online marketplaces in 3 primary categories: Goods, Travel and Local. Its Goods category includes merchandise across multiple product lines, such as electronics, sporting goods, jewelry, toys, household items and apparel. Its Travel category features travel experiences at both discounted and market rates, including hotels, airfare and package deals covering both domestic and international travel. On the other hand, the enterprise’s Local category includes services from local and national merchants, including things to do, beauty and wellness and dining.
The firm, which recently announced its latest financial results, remains committed to meeting consumer needs and creating shareholder value. This will be good for investments as well as its overall growth.
Groupon (GRPN), closed Monday's trading session at $4.16, up 10.9333%, on 1,645,983 volume. The average volume for the last 3 months is 116,107 and the stock's 52-week low/high is $3.37/$21.175.
Altisource Portfolio Solutions (ASPS)
The Street, Marketbeat, BUYINS.NET, Louis Navellier, Marketbeat.com, Hit and Run Candle Sticks, StreetInsider, Street Insider, StockMarketWatch, Zacks, Market Intelligence Center Alert, VectorVest, CrashTrade, Daily Trade Alert, FreeRealTime, InsiderTrades, Investment House, Microcapmillionaires, TradersPro, AllPennyStocks, The Trading Report, Rick Saddler, StockTwits, StreetAuthority Daily and InvestorPlace reported earlier on Altisource Portfolio Solutions (ASPS), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Altisource Portfolio Solutions S.A. (NASDAQ: ASPS) is an integrated service provider and marketplace for the real estate and mortgage industries.
The firm has its headquarters in Luxembourg City, Luxembourg and was incorporated in 1999, on November 4th. It operates as part of the real estate services industry, under the real estate sector. The firm serves consumers in the United States and globally.
The enterprise offers property preservation and inspection services, payment management technologies, and a vendor management oversight software-as-a-service (SaaS) platform. It also provides Equator, a SaaS-based technology to manage real estate owned, short sales, foreclosure, bankruptcy, and eviction processes; and Hubzu, an online real estate auction platform, as well as real estate auction, real estate brokerage, and asset management services. It offers loan fulfillment, certification and certification insurance, title insurance agent, settlement, real estate valuation, residential and commercial construction inspection and risk mitigation, and foreclosure trustee services. The enterprise also operates RentRange, a single family rental data, analytics, and rent-based valuation solution; Vendorly Invoice, a vendor invoicing and payment system; NestRange, an automated valuation model and analytics solution; and REALSynergy, a commercial loan servicing platform. This is in addition to operating ADMS, a document management and data analytics delivery platform; and TrelixAI technology to manage the workflow and automate components of the loan fulfillment, pre and post-close quality control and service transfer processes. It serves financial institutions, banks, government-sponsored enterprises, asset managers, property management firms, investors, real estate brokerages, mortgage bankers, insurance firms, originators, and correspondent and private money lenders.
The company recently released its latest financial results, with its CEO noting that they remained focused on growing the sales pipeline and improving operational efficiencies. This will bring in additional revenues into the company while also improving overall performance.
Altisource Portfolio Solutions (ASPS), closed Monday's trading session at $3.84, even for the day, on 116,107 volume. The average volume for the last 3 months is 2.133M and the stock's 52-week low/high is $3.78/$17.67.
Near Intelligence (NIR)
We reported earlier on Near Intelligence (NIR), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Near Intelligence Inc. (NASDAQ: NIR) is a data intelligence platform that offers privacy-led data intelligence solutions.
The firm has its headquarters in Pasadena, California. It operates as part of the software-application industry, under the technology sector. The firm serves consumers around the globe, with a focus on those in the United States.
The company is focused on enabling firms to make power strategic decision-making using consumer behavior data. It does so by bringing together online and offline consumer data from disparate sources and turning that data into actionable insights that have a real business impact.
The enterprise has designed a data-driven SaaS platform dubbed the Near Identity Graph, which offers information on people, places, and products by generating marketing and operational intelligence on consumer behavior using multiple consumer identifiers that enable enterprises to make informed and rapid strategic decisions. The identifiers are enriched using its CARBON software function with attributes such as age range, gender, income range, behavior profiles and online interests. The enterprise’s other products include Allspark, an AI audience assistant for marketing RoI, customer acquisition, customer retention, and life-time value solutions as well as Pinnacle that provides data intelligence for places. It caters to retail, restaurants, real estate, government, financial services, media and tech, tourism, and automotive industries.
The firm, whose customer and market demand has increased greatly, remains focused on heightening focus on its business. This will positively influence its expansion strategy while also opening it up to new growth and investment opportunities.
Near Intelligence (NIR), closed Monday's trading session at $4.28, off by 28.3082%, on 2,132,820 volume. The average volume for the last 3 months is 29.147M and the stock's 52-week low/high is $2.18/$18.65.
Marathon Digital Holdings Inc. (MARA)
InvestorPlace, MarketClub Analysis, Schaeffer's, QualityStocks, StockMarketWatch, MarketBeat, StocksEarning, TradersPro, Lebed.biz, The Online Investor, BUYINS.NET, INO Market Report, Trades Of The Day, Marketbeat.com, StockEarnings, The Street, TraderPower, Daily Trade Alert, Wall Street Mover, PoliticsAndMyPortfolio, BillionDollarClub, TopPennyStockMovers, FeedBlitz, Early Bird, StreetAuthority Daily, Wealth Insider Alert, Kiplinger Today, InvestorsUnderground, Eagle Financial Publications, Barchart, DreamTeamNetwork, Promotion Stock Secrets, AllPennyStocks, Rick Saddler, Zacks, Stock Analyzer, Stock Beast, StockOodles, Street Insider, StreetInsider and RedChip reported earlier on Marathon Digital Holdings Inc. (MARA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Bitcoin is currently trading at more than $30,000 — last week, the dominant crypto hit a new record price for 2023. This is the first time Bitcoin has surpassed the $30,000 mark since last June. As of now, Bitcoin has increased by around 84%, making it the best-performing asset in the relevant market for the first quarter of 2023.
After a calm Easter weekend, the price of BTC jumped by 3.37% to above $29,300 on April 10, 2023. The momentary increases in Bitcoin were interestingly accompanied by a decline in U.S. stocks, an unusual decoupling that emphasizes the coin’s waning risk-on qualities. In the past month, BTC has increased by nearly 46%, reaching its most recent high on Tuesday .
The Crypto Greed and Fear Index has been consistently in the “Greed” zone since last week, with the most recent update on Tuesday, April 11, 2023, showing a score of 68 out of 100. The Index’s objective is to statistically depict the current “sentiments and emotions” regarding the crypto market, with 100 being the highest possible score.
On March 21, 2023, the index also reached a score of 68, which is the highest level it had reached since Bitcoin’s all-time high of almost $69,000 was registered on November 10, 2021.
According to CoinDCX, the increase can be due to a number of causes, including rising U.S. inflation, unrest in the banking industry and unpredictability around stablecoin. Many investors are placing bets against the U.S. dollar’s deteriorating value and purchasing Bitcoin as an inflation hedge.
Due to this, institutional adoption has increased dramatically, leading major companies such as MicroStrategy, one of the largest Bitcoin corporate holders as of today, to amass a total of about 140,000 Bitcoins, totaling $4.16 billion.
Michael Saylor, the creator of MicroStrategy, has been a vocal supporter of Bitcoin and has led the charge for companies to exploit cryptocurrency as a major asset. Since making its initial investment in 2020, MicroStrategy has consistently increased its BTC holdings despite the ongoing bear market in cryptocurrencies.
CoinDCX Research also noted that the narrative surrounding the upcoming Bitcoin halving, set for April 2024, is beginning to gain momentum, which has historically been a bullish indication for the price of Bitcoin and other cryptocurrencies.
A close eye will be kept on Bitcoin’s future performance. However, for the time being, the $30,000 threshold is considered a positive development for the global cryptocurrency markets, and industry companies such as Marathon Digital Holdings Inc. (NASDAQ: MARA) could see increased activity as a result of this positive spike in the price of the leading cryptocurrency.
Marathon Digital Holdings Inc. (MARA), closed Monday's trading session at $11.45, off by 4.3442%, on 29,146,918 volume. The average volume for the last 3 months is 843,547 and the stock's 52-week low/high is $3.11/$22.09.
atai Life Sciences N.V. (ATAI)
QualityStocks, MarketBeat, The Online Investor, StockMarketWatch, StreetInsider, Dynamic Wealth Report, Marketbeat.com, Uncommon Wisdom, MarketClub Analysis, CRWEFinance, CRWEPicks, CRWEWallStreet, DrStockPick, InsiderTrades, PennyOmega, BestOtc, PennyToBuck, Schaeffer's, Small Caps, StockHotTips, TraderPower, StockOodles, Street Insider, The Street, TopPennyStockMovers and ProTrader reported earlier on atai Life Sciences N.V. (ATAI), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The psychedelic renaissance is here, with numerous studies showing that psychedelics, including LSD, psilocybin, MDMA and ketamine. may be useful in the treatment of mental health conditions, such as addiction, depression, anxiety and psychological distress.
Veteran religion journalist Don Lattin recently wrote a book describing how these drugs may prompt a spiritual awakening. In his latest book, “God on Psychedelics,” Lattin looks into how various religious groups are encouraging experiences of human unity and interconnectedness through the substances.
He starts by discussing entheogens, which refer to psychoactive drugs that are consumed for spiritual or religious purposes. Lattin talks about priests, rabbis and other clergy who view using entheogens as a way to renew their faith, noting that there are also individuals who don’t subscribe to a particular faith but also use the drugs consciously as a spiritual practice. He gives the example of the Sacred Garden Church, which uses different psychedelics to divinely commune.
Lattin also covers how psychedelics may not always induce positive feelings such as compassion, unity and awe, talking about how some individuals who use these substances may experience bad trips. He explains that during bad trips, individuals may experience paranoia and existential dread among other negative feelings; he also highlights the importance of having someone help you through feelings that come up in an environment which feels safe.
Despite these bad trips, Lattin reaffirms that these substances open one up to greater psychological insight and spiritual awareness.
Lattin also talks about how the upcoming psychedelic churches retain some Christian elements, giving an example of Santo Daime. Santo Daime, as profiled in his book, is a mixture of spiritualism, folk Catholicism, Indigenous and African religion. He notes that while most individuals in the church may not consider themselves Christian, they believe that they’re connected to Christ’s consciousness.
Interestingly, he adds, a good number of individuals involved in these groups in the United States were raised Jewish. Lattin also discusses the legal psychedelic landscape, highlighting how states such as Colorado and Oregon have legalized the use of psilocybin mushrooms and are regulating supervised psychedelic therapy. He expects that the legal situation will continue to change rapidly, as more cities pass laws restricting arrest of individuals who use certain plant medicines and drugs.
In addition to this, Lattin describes the meditation group he belongs to, which applies Buddhist meditation and contemplative prayer practices, noting that it has nothing to do with psychedelics.
With psychedelic use for religious purposes dating back hundreds of years, it is little wonder that start-ups such as atai Life Sciences N.V. (NASDAQ: ATAI) are confident that the formulations they are developing will find a receptive market once they are approved by regulators like the U.S. FDA.
atai Life Sciences N.V. (ATAI), closed Monday's trading session at $1.95, up 3.7234%, on 845,497 volume. The average volume for the last 3 months is 48.155M and the stock's 52-week low/high is $1.14/$5.41.
NIO Inc. (NIO)
Green Car Stocks, InvestorPlace, Schaeffer's, The Street, MarketClub Analysis, MarketBeat, Daily Trade Alert, StocksEarning, Trades Of The Day, The Online Investor, Kiplinger Today, QualityStocks, Zacks, StreetInsider, INO Market Report, StockEarnings, Early Bird, StockMarketWatch, BUYINS.NET, Cabot Wealth, Wealth Insider Alert, CNBC Breaking News, InvestorsUnderground, The Wealth Report, Investopedia, TradersPro, wyatt research newsletter, Daily Wealth, CRWEWallStreet, Energy and Capital, FreeRealTime, Green Energy Stocks, InvestorIntel, Investors Alley, InvestorsObserver Team, Jim Cramer, AllPennyStocks, MarketClub, Money Wealth Matters, Smartmoneytrading, Stock Market Watch, Top Pros' Top Picks, TopPennyStockMovers, Wealth Daily and Louis Navellier reported earlier on NIO Inc. (NIO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
According to a recent survey, almost 50% of Americans feel it’s doubtful that the next car they buy will be an electric vehicle, citing the cost and scarcity of charging alternatives as the major deterrents. The Biden government’s ambitions to significantly increase American sales of electric vehicles may encounter customer pushback, according to an AP-NORC/EPIC poll. Just 8% of American people claim ownership of or leases on an electric car, and just 8% claim to have a PHEV in their homes.
Despite the $7,500 in tax incentives to purchase a new electric vehicle, it could be challenging to convince people to trade in their gas-guzzling automobiles and trucks for ones with no exhaust emissions.
Automakers are spending billions on manufacturing and battery technologies in order to hasten the transition to electric vehicles in an effort to reduce pollution and combat global warming. However, only 19% of American adults feel that it is extremely likely that when they next purchase a car, they will get an electric one, and 22% say it is fairly likely, based on the poll. The majority, 47%, said it is unlikely that they will switch to electricity.
High price was seen as a significant reason by 6 out of 10 people and a moderate reason by nearly a quarter of the people. Just 16% of respondents claimed that the high price of an electric vehicle wouldn’t be an obstacle.
Around 75% of people think there aren’t enough charging stations; 50% of them say it is a significant reason why they won’t transition to electric vehicles. And two-thirds identify their “desire for conventional cars” as a significant or minor barrier to doing so.
There is a party gap in the way Americans feel about electric cars, with at least 50% of Republicans and Democrats mentioning the high costs of electric vehicles and scarcity of chargers as the biggest deterrents from purchasing one. Republicans are more likely than Democrats to claim that their preference for gasoline or diesel cars is a key deterrent to purchasing an electric vehicle (54% vs. 29%, respectively).
A promising result for advocates of electric vehicles is that 55% of persons below the age of 30 and 49% of adults between the ages of 30 and 44 claim they’re considerably more inclined to purchase an electric car at the right moment, in contrast to only 31% of persons above the age of 45.
Reducing one’s personal influence on the environment is cited by 35% of respondents as a significant cause for purchasing an electric vehicle and 31% as a moderate reason. Automakers such as NIO Inc. (NYSE: NIO) appear to have their work cut out to convince more members of the public to choose electric vehicles when they next purchase an automobile.
NIO Inc. (NIO), closed Monday's trading session at $9.83, up 6.041%, on 48,549,523 volume. The average volume for the last 3 months is 211,902 and the stock's 52-week low/high is $8.03/$24.43.
FSD Pharma Inc. (HUGE)
QualityStocks, Schaeffer's, BUYINS.NET, StockMarketWatch, MarketClub Analysis, Penny Dreamers, InvestorPlace, CFN Media Group and AwesomeStocks reported earlier on FSD Pharma Inc. (HUGE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
FSD Pharma (NASDAQ: HUGE) (CSE: HUGE) (FRA: 0K9A) is a biopharmaceutical company dedicated to building a portfolio of innovative assets and biotech solutions for the treatment of challenging neurodegenerative, inflammatory and metabolic disorders. The company today announced completion of the first-in-human (“FIH”) sentinel dosing of Lucid-21-302 (“Lucid-MS”) in its phase I clinical trial evaluating its novel drug candidate as an orally administered treatment for multiple sclerosis (“MS”). “Dosing the sentinel subject is a major achievement for our team and culmination of more than a decade of very promising research by a seasoned development team passionate about changing the future treatment paradigm for patients dealing with the debilitating effects of MS,” said Dr. Lakshmi Kotra, CEO of Lucid Psycheceuticals, a wholly owned subsidiary of FSD Pharma. “Current MS treatments are immunomodulatory and include repeated subcutaneous or intramuscular injections for treating the symptoms of MS. We envision a day where an oral medication will protect or even help repair myelin in the central nervous system, a hallmark feature of the disease. We are optimistic Lucid-MS has this type of paradigm-shifting potential and the sentinel dosing is a critical step in advancing this pipeline forward.”
To view the full press release, visit https://ibn.fm/AFKIk
About FSD Pharma Inc.
FSD Pharma is a biotechnology company with three drug candidates in different stages of development. FSD BioSciences Inc., a wholly owned subsidiary, is focused on pharmaceutical research and development of its lead compound, FSD201, a proprietary ultra-micronized PEA formulation, for the treatment of inflammatory diseases. Lucid Psychss Inc., a wholly owned subsidiary, is focused on the research and development of its lead compounds, Lucid-Psych and Lucid-MS. Lucid-Psych is a molecular compound identified for the potential treatment of mental health disorders, and expanding this category, the company is investigating other products addressing acute medical needs due to the abuse of drugs such as alcohol. Lucid-MS is a molecular compound identified for the potential treatment of neurodegenerative disorders. For more information, visit the company’s website at www.FSDPharma.com.
FSD Pharma Inc. (HUGE), closed Monday's trading session at $1.75, off by 5.914%, on 211,902 volume. The average volume for the last 3 months is 689 and the stock's 52-week low/high is $0.6181/$2.10.
The QualityStocks Company Corner
- Progressive Care Inc. (OTCQB: RXMD)
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
- Aditxt Inc. (NASDAQ: ADTX)
- IGC Pharma Inc. (NYSE American: IGC)
- Data443 Risk Mitigation Inc. (OTC: ATDS)
- Advanced Container Technologies Inc. (OTC: ACTX)
- Prime Harvest Inc.
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF)
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF)
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF)
Progressive Care Inc. (OTCQB: RXMD)
The QualityStocks Daily Newsletter would like to spotlight Progressive Care Inc. (OTCQB: RXMD).
To view the full article, visit https://ibn.fm/PRSKF
Progressive Care Inc. (OTCQB: RXMD) is a health services organization based in Florida that offers personalized healthcare services and technology that supports the managed healthcare industry. Through its subsidiaries, Progressive Care provides Third-Party Administration (TPA), data management and analytics, COVID-19 diagnostics and vaccinations, 340B contracted pharmacy services, compounded medications, tele-pharmacy services, dispensing of anti-retroviral medications, medication therapy management (MTM), long-term care facility-targeted prescription medications, and health practice risk management.
The company collaborates with various healthcare organizations such as managed care organizations (MCOs), management services organizations (MSOs), accountable care organizations (ACOs), primary care providers, Medicare Advantage plans, Medicaid, commercial payors, pharmaceutical manufacturers, and distributors to enhance patient and provider engagement while improving the lives of patients with chronic diseases. Progressive Care offers a wide range of innovative solutions to address the dispensing, delivery, dosing, and reimbursement of clinically intensive, high-cost drugs.
Progressive Care currently operates four pharmacies in Florida, which generate the majority of its revenue. Pharmacy revenue is derived from dispensing medications, third-party administrative services to 340B-covered entities, and MTM services. The company also provides customized management, patient health risk reviews, and free same- and next-day delivery. Its focus is on complex chronic diseases that require multiyear or lifelong therapy, driving recurring revenue and sustainable growth. Progressive Care’s pharmacy revenue growth stems from its expanding breadth of services, new drugs coming to market, new indications for existing drugs, volume growth with current clients, and addition of new customers resulting from its emphasis on higher patient engagement, free delivery to the patient, and clinical expertise.
With licenses in 14 states, Progressive Care is poised for national expansion. The company anticipates revenue growth by signing new contract pharmacy service and data management contracts with 340B-covered entities, expanding data management and analytics services to healthcare organizations, and potential acquisitions.
Subsidiaries
Progressive Care’s wholly-owned subsidiaries provide services to client organizations and patients.
PharmcoRx Pharmacy
PharmcoRx, a full-service pharmacy, provides a complete healthcare ecosystem with services such as medication therapy management, rapid COVID-19 testing and vaccines, contactless medication delivery, Smart-Pack Unit Dosing packaging, custom compound medications, specialty medications, hospital transition pharmacy services, medication adherence monitoring, medication adherence risk management, and drug cost containment. PharmcoRx Pharmacy is a contracted pharmacy services provider for 340B-covered entities under the 340B Drug Discount Pricing Program.
ClearMetrX
ClearMetrX, a wholly-owned data management company, offers services that support healthcare organizations across the country. In September 2022, ClearMetrX launched the 340MetrX Platform, a software product developed by ClearMetrX that provides 340B-covered entities with data insights to effectively operate and maximize the benefits of the 340B program. 340MetrX supplies data access and delivers actionable insights that providers and support organizations can use to improve their practices and patient care. Its TPA services include management of wholesale accounts and contract pharmacies, patient eligibility with regard to the 340B drug program, development and review of 340B policies and procedures, and management of receivables.
Market Opportunity
According to an industry report by global consulting firm Berkeley Research Group, gross sales across the 340B drug program were valued at $116 billion in 2021 and are projected to grow to $280 billion by 2026, achieving a CAGR of more than 19% over the period.
The 340B drug pricing program allows eligible healthcare clinics and hospitals (the covered entities) to purchase outpatient drugs at a 20-50 percent discount to treat low-income, uninsured, or underinsured populations. The program’s forecast growth is expected to benefit Progressive care’s business of providing 340B program services to covered entities through the nationwide expansion of ClearMetrX, its third-party administration and data-management business.
Management Team
Charles M. Fernandez is CEO and Chairman of the Board of Directors of Progressive Care. Mr. Fernandez is also the Executive Chairman and CEO of NextPlat Corp. (NASDAQ: NXPL) and has over 30 years of experience in identifying profitable start-up and dislocation opportunities, building significant value and executing exit strategies as an entrepreneur and global investor. In 2008, he joined Fairholme Capital Management. As president, he co-managed all three Fairholme funds and was commended for bringing in a $2 billion gain for shareholders. Throughout his impressive career in media, pharmaceuticals, healthcare, finance and technology, Mr. Fernandez has participated in more than 100 significant mergers, acquisitions and product development projects. He was the founder, chairman and CEO of eApeiron Solutions LLC, a brand protection and e-commerce company in partnership with Alibaba (NYSE: BABA) and Eastman Kodak (NYSE: KODK) which was successfully sold to Smartrac, a leading developer, manufacturer and supplier of RFID and Internet of Things (“IoT”) solutions and a unit of Avery Dennison Corporation (NYSE: AVY).
Other top management team members include Chief Operating Officer Birute Norkute, Chief Financial Officer Cecile Munnik, and Pamela Roberts, who serves as the company’s Pharmacist in Charge.
FingerMotion Inc. (RXMD), closed Monday's trading session at $3.05, up 5.1724%, on 689 volume. The average volume for the last 3 months is 351,947 and the stock's 52-week low/high is $2.00/$10.40.
Recent News
- Progressive Care Inc. (OTCQB: RXMD) - BioMedNewsBreaks - Progressive Care Inc. (RXMD) Identified as a High-Growth Opportunity
- Electronic Health Data Could Boost Chronic Disease Surveillance
- InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases 2022 Annual Financial Results, Reports $40.6M Total Revenue
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV)
The QualityStocks Daily Newsletter would like to spotlight BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV).
Earlier this week, President Joseph Biden sig ned a billthat would end the COVID-19 national emergency with immediateeffect. The national emergency was
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) is a biopharmaceutical company focused on developing, manufacturing and commercializing innovative products for the prevention and treatment of infectious diseases and other illnesses.
In collaboration with the prestigious Max Planck Institute for Multidisciplinary Sciences (MPG) and the University Medical Center Göttingen (UMG), both in Germany, BiondVax is developing a pipeline of innovative nanosized antibody (NanoAb) therapies addressing diseases underserved by current treatments and with large and growing markets, such as COVID-19, asthma and psoriasis.
NanoAbs, also known as VHH-antibodies or Nanobodies, are alpaca-derived nanosized antibodies that exhibit multiple significant competitive advantages over existing antibody therapies, including stability at high temperatures, superior binding affinity, more effective and convenient routes of administration and efficient production. BiondVax is uniquely positioned to advance nanosized antibody innovation from R&D through commercialization.
The company’s highly experienced and successful pharmaceutical industry leadership team includes former senior executives from Novartis, GSK and Bristol-Myers Squibb.
Since its founding, BiondVax has executed eight clinical trials, including a seven-country, 12,400-participant Phase 3 trial of a prior influenza vaccine candidate, and it built, owns and operates a 20,000 sq. ft. state-of-the-art GMP biologics manufacturing facility housing its laboratories, production facilities and offices.
Lead Candidate: Inhaled COVID-19 NanoAb
In December 2021, BiondVax signed definitive agreements with the Max Planck Society – parent organization of the Max Planck Institute for Multidisciplinary Sciences– and the UMG to enter a strategic collaboration for the development and commercialization of innovative COVID-19 NanoAbs.
The company is planning a rapid development path that leverages its expertise and capabilities in biological drug development and manufacturing. BiondVax anticipates preclinical proof-of-concept results for an inhaled COVID-19 NanoAb by the end of 2022, with initial Phase 1/2a human clinical trial results expected in 2023.
The intended inhaled mechanism of delivery of BiondVax’s COVID-19 NanoAb formulation may serve as a significant differentiator when compared to approved monoclonal antibodies, which are injected. Inhaled delivery has shown to be cheaper, more convenient and likely safer for patients and providers.
NanoAb Pipeline: Psoriasis, Asthma and More
The COVID-19 NanoAb development agreement is part of a broader five-year research collaboration agreement signed in March 2022 covering discovery, development and commercialization of NanoAbs for several other disease indications with large market medical needs, including asthma, psoriasis, macular degeneration and psoriatic arthritis.
BiondVax has an exclusive worldwide license for development and commercialization of COVID-19 NanoAbs and exclusive options for similar worldwide licenses for NanoAbs for the above mentioned additional large market disorders currently underserved by approved therapeutic antibodies.
Academic research teams from MPG and UMG have verified strong affinity by the new NanoAbs to their biological target molecules and high thermostability. They have also demonstrated strong neutralization by several NanoAb candidates of their respective target molecules. Neutralization studies of the other NanoAbs are expected to begin later in 2022.
Based on the promising results, BiondVax will focus development efforts beginning with the following NanoAbs:
- NanoAbs targeting IL-17 as drug candidates for the potential treatment of psoriasis and psoriatic arthritis
- NanoAbs targeting IL-13 and NanoAbs targeting TSLP as drug candidates for the potential treatment of asthma
These are conditions for which the antibody target is validated by existing treatments and the mechanism of action is well understood. Both represent large medical needs and growing markets. BiondVax anticipates preclinical proof-of-concept for at least one of these NanoAbs in 2023. This is in addition to the aforementioned human clinical Phase 1/2a for the inhaled COVID-19 NanoAb therapy, which is also anticipated in 2023.
Market Opportunity
COVID-19 treatment, target of the company’s lead NanoAb therapy candidate, had an estimated market size of $22 billion in 2021.
Future BiondVax drug candidates will target conditions with large markets growing at attractive CAGRs.
The global asthma treatment market was valued at $18.08 billion in 2019 and is projected to reach $26.01 billion by 2027, exhibiting a CAGR of 4.5% during the forecast period, according to Fortune Business Insights. The research firm predicts that the global psoriasis treatment market will grow from $26.37 billion in 2022 to $47.24 billion by 2029, exhibiting a CAGR of 8.7% over the forecast period.
Management Team
Amir Reichman is BiondVax’s CEO. He previously was Head of Global Vaccines Engineering Core Technologies at GSK Vaccines in Belgium. Prior to that, he held leadership roles at Novartis Vaccines’ Global Vaccines Supply Chain Management organization. He was the first employee of NeuroDerm Ltd., a company focused on transdermal drug delivery, and served as Chief Engineer and Senior Scientist until his departure in 2009. He earned a M.Sc. in Biotechnology Engineering from Ben-Gurion University and an MBA in Finance and Health Care Management from the University of Pennsylvania’s Wharton School.
Tamar Ben-Yedidia, Ph.D., is Chief Science Officer at BiondVax. She has more than 30 years of experience in immunology, with specific expertise in the development of vaccines. She began her career with Biotechnology General Ltd., working on development of a recombinant Hepatitis-B vaccine. She later joined the Weizmann Institute of Science, working on the design of a peptide-based vaccine against several pathogens. She is widely published, with numerous refereed articles and invited reviews in various scientific journals. She received her Ph.D. from the Weizmann Institute.
Elad Mark is COO at BiondVax. He has over 15 years of biotechnology industry experience encompassing diverse project stages including feasibility studies, conceptual and detailed design, commissioning, qualification and process validation. Prior to joining BiondVax, he led Novartis’s $800 million investment in a biologics facility in Singapore. With Biopharmax and Antero, both global pharmaceutical engineering companies, he successfully led projects in Israel, China and Singapore. He holds a BSc. in Engineering from the Afeka Tel Aviv Academic College of Engineering and an MBA from the Open University of Israel.
Uri Ben-Or is CFO at BiondVax. He has served as CFO with public life science companies traded on the TASE, OTC and Nasdaq. Ben-Or provides his services to BiondVax through CFO Direct, a company he founded and for which he serves as CEO. He served as the VP of Finance of Glycominds, a leading biotechnology company, and as CFO of a spin-off from Telrad Networks. He also served as a Corporate Controller at Menorah Capital Markets and as an Auditor at PWC. He holds a B.A. in Business from the College of Administration, an MBA from Bar-Ilan University, and is a CPA.
BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV), closed Monday's trading session at $2.15, up 20.1117%, on 355,189 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $10.40/$.
Recent News
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Biden Signs Measure to End National COVID-19 Emergency
- BioMedNewsBreaks - BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) Leveraging Innovative NanoAb Pipeline to Disrupt High-Value Market Opportunities
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) CEO Discusses Positive Results from In Vivo Proof-of-Concept Study of Its Anti-COVID-19 NanoAb Therapy and Additional Pipeline Plans at BIO-Europe Spring
Aditxt Inc. (NASDAQ: ADTX)
The QualityStocks Daily Newsletter would like to spotlight Aditxt Inc. (NASDAQ: ADTX).
To view the full press release, visit https://ibn.fm/50mNa
Aditxt Inc. (NASDAQ: ADTX) is a biotech innovation company developing technologies focused on mapping and reprogramming the immune system. Aditxt’s immune mapping technologies are designed to provide a personalized immune profile. Aditxt’s immune reprogramming technologies, currently preclinical, are being developed to retrain the immune system to induce tolerance to address rejection of transplanted organs, autoimmune diseases, and allergies.
As further discussed below, the company’s first commercial product is an immune mapping technology, AditxtScore™, which is designed to provide a personalized profile of the immune system.
The company’s preclinical immune reprogramming technology, Apoptotic DNA Immunotherapy™ (“ADi™”), aims to retrain the immune system to induce tolerance, with the goal of addressing vast unmet needs in transplanted organ rejection, autoimmune diseases, and allergies. The company is developing specific ADi™ products for psoriasis, type 1 diabetes, and skin grafting.
Headquartered in Richmond, Virginia, Aditxt also operates locations in Silicon Valley and New York.
AditxtScore™
AditxtScore™ is a proprietary platform designed to provide a personalized, comprehensive profile of an individual’s immune system. The underlying technology, licensed from Stanford University through an exclusive worldwide agreement, offers a highly sensitive and accurate method of detecting and quantifying cellular responses, allowing greater specificity, quantification, and amplification of both clinical and commercial opportunities.
The company’s first commercial application of the platform, AditxtScore™ for COVID-19, delivers timely reports on vulnerability and immune status relating to SARS-CoV-2 and its known variants, giving consumers and physicians the data needed to make informed health decisions. Potential future applications will offer early detection of an array of conditions, including diabetes, cardio-metabolic maladies and hormonal imbalances.
Aditxt’s AditxtScore™ immune monitoring center in Richmond, Virginia, is operational and designed to support the anticipated increased demand for AditxtScore™ as well as related products and services. The company is currently scaling its capabilities at this location, with a goal of processing up to 10 million immune system tests/reports annually.
ADi™
ADi™ is Aditxt’s immune reprogramming platform addressing disease-causing immune responses while maintaining the immune system’s ability to combat pathogenic infection. The company is commercializing a nucleic acid-based technology called Apoptotic DNA Immunotherapy™ (ADi™) which utilizes a novel approach that mimics the way our bodies naturally induce tolerance to our own tissues (therapeutically induced immune tolerance). Aditxt believes its ADi™ technology platform can be engineered to address a wide variety of indications.
Aditxt is currently developing ADi™ products for psoriasis, type 1 diabetes and skin grafting.
Currently, immuno-tolerance is achievable through chimerism and cell-based therapy, but there is a clinical need for a more practical and cost-effective approach which:
- Can be made into a product
- Does not require additional hospitalization
- Is simple to produce and ship
Preclinical studies have demonstrated that ADi™ treatment significantly and substantially prolongs graft survival, in addition to successfully “reversing” other established immune-mediated inflammatory processes. ADi™ treatment is not expected to require hospitalization, instead being delivered as an injection in minute amounts into the skin.
IP Portfolio
Both AditxtScore™ and ADi™ are supported by a strong IP portfolio.
AditxtScore™, built upon initial technology invented, licensed from and used at Stanford University, is protected by U.S. patents encompassing methods, systems, and kits for detection and measurement of specific immune responses.
ADi™ technology is protected by seven patent families, including:
- 8 U.S. patents
- 4 pending U.S. patent applications
- 86 foreign patents and 14 pending foreign patent applications spanning the EU, Australia, Canada, Japan, China, India and Hong Kong
These patents are broadly categorized into three groups:
- Autoimmune diseases and Type 1 Diabetes
- Organ transplantation and a method of producing plasmid DNA to prevent immune activation
- Composition of matter for a tolerance delivery system for antigens of interest
Aditxt also possesses and/or in-licenses substantial know-how and trade secrets relating to the development and commercialization of its product candidates, including related manufacturing processes and technologies.
Market Overview
The potential market opportunities presented by immune monitoring and reprogramming are extensive, particularly as Aditxt continues to evaluate additional applications for the platforms.
The company’s initial focus on organ transplantation and related autoimmune response provides some insight into the potential of its approach. According to BCC Research, the global organ and tissue transplantation and alternatives market is on course to reach $120.3 billion by 2024, recording a CAGR of 7.4% from 2019. Industry data suggest that approximately 50% of all transplanted organs are rejected within 10-12 years, further highlighting the critical need for a practical, cost-effective solution to harmful autoimmune responses.
Through its focus on the COVID-19 testing market with AditxtScore™, Aditxt demonstrated the wide-ranging potential of its portfolio. Fortune Business Insights estimated the global COVID-19 diagnostics market at $48.64 billion for 2022. While demand for COVID-19 diagnostics is expected to lessen in the coming years, Aditxt will be uniquely positioned to leverage its existing infrastructure stemming from these operations as the company works to advance broader applications for the AditxtScore™ platform.
Leadership Team
Amro Albanna is the Co-Founder, Chairman, and CEO of Aditxt. He has founded multiple startups to commercialize innovations in various industries, including healthcare, enterprise software, telecommunications, nano technology, consumer health, and biotech. Mr. Albanna has led numerous M&A and going-public transactions as a founder, co-founder, and senior executive.
Shahrokh Shabahang, D.D.S., MS, Ph.D., is the company’s Co-Founder, Chief Innovation Officer, and a member of its board. He brings to the team more than 20 years of experience in developing and commercializing life science technologies focused on product and clinical development in the fields of microbiology and immunology.
Corinne Pankovcin, CPA, MBA, is the President of Aditxt. Prior to joining Aditxt, Ms. Pankovcin served as CFO for several world class organizations, including Business Development Corporation of America, Blackrock Kelso Capital and AIG Capital Partners. In these roles, Ms. Pankovcin was responsible for executing portfolio investments and managing significant M&A transactions.
Thomas Farley is the Chief Financial Officer of Aditxt. From December 2015 to June 2020, Mr. Farley was the Controller and Treasurer of Business Development Corporation of America (“BDCA”), a publicly listed business development company. Prior thereto, from January 2011 to August 2015, Mr. Farley was the Senior Controller of Blackrock Capital Investment Corporation (NASDAQ: BKCC). Prior to joining BlackRock Capital Investment Corporation, Mr. Farley was a Senior Controller for PineBridge Investments Emerging Markets practice. Mr. Farley was also an Accounting Manager for Bessemer Venture Partners prior to his tenue at PineBridge. Mr. Farley began his career with PricewaterhouseCoopers LLP, from 1996 to 2001. Mr. Farley earned his B.S. in Accounting from Long Island University and is a Certified Public Accountant.
Rowena Albanna is the company’s Chief Operating Officer. Ms. Albanna has over two decades of experience in senior leadership roles for both technology startups and public companies. Ms. Albanna’s experience spans a wide variety of industries, including biotechnology, insect control, nanotechnology, consumer electronics, financials, telecommunications, e-commerce, online marketing, medical, and defense.
Matthew Shatzkes is the Chief Legal Officer and General Counsel of Aditxt. As a former partner at an AM Law 50 law firm, Mr. Shatzkes advised a wide variety of healthcare related entities, including biotech companies, on corporate, regulatory, and strategic business matters. Mr. Shatzkes will oversee all aspects of the legal functions at Aditxt, including, providing advice and counsel on governance, regulatory matters, strategic alliances, mergers and acquisitions, and commercial transactions.
Aditxt Inc. (NASDAQ: ADTX), closed Monday's trading session at $0.9159, up 4.6744%, on 63,005 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.8304/$28.49.
Recent News
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Releases Shareholder Update on Major Programs, M&A Plans
- US Federal Agency Plans to Overhaul Organ Transplant Network
- Study Describes Link Between Viral infections, Autoimmune Diseases
IGC Pharma Inc. (NYSE American: IGC)
The QualityStocks Daily Newsletter would like to spotlight IGC Pharma Inc. (NYSE American: IGC).
IGC Pharma Inc. (NYSE American: IGC), through subsidiary IGC Pharma, develops, patents, and markets advanced THC-based drug formulations for the treatment of symptoms related to various diseases including but not limited to Alzheimer’s disease, Tourette syndrome, chronic pain, and pet seizures.
IGC’s leading drug candidate, IGC-AD1, has completed Phase 1 of a safety and tolerability trial and entered Phase 2 trials for treating agitation in patients with Alzheimer’s dementia, the first study in humans of a natural tetrahydrocannabinol (THC) compound plus another molecule (www.clinicaltrials.gov). As of September 2022, the IGC trial is the only ongoing Phase 2 trial of a natural THC-based formulation on Alzheimer’s patients.
The company’s other drug candidate, TGR-63, is an enzyme inhibitor that has shown in preclinical trials the potential to reduce neurotoxicity in Alzheimer’s cell lines. Both drug candidates have shown their ability to ameliorate beta amyloid plaques in Alzheimer’s cell lines and improve memory in Alzheimer’s mouse models. Beta amyloid plaques are a key hallmark of Alzheimer’s and an important target of Alzheimer’s pharmaceutical drug development.
Neuro Psychiatric Symptoms (NPS) are not only debilitating for Alzheimer’s patients; they also place an immense emotional burden on their caregivers. Beyond reducing symptoms, IGC-AD1’s active molecules and TGR-63 have also shown promise in preclinical trials to reduce important hallmarks of Alzheimer’s including plaques and tangles, as well as improving the treatment of memory loss.
Over the past eight years, the IGC team has amassed a deep knowledge of cannabinoid science, including extraction, isolation, purification, and development. The company’s strategy is to leverage its unique end-to-end capabilities, platform, and expertise to develop a class-leading program and bring it to market quickly and cost efficiently to treat neurodegenerative diseases such as Alzheimer’s.
The company also has a family of cannabidiol (CBD)-based consumer products (www.Holief.com) such as pain relief creams, pain relief gels, purpose gummies, tinctures, and capsules targeting women’s wellness, with a particular focus on premenstrual syndrome (PMS) and dysmenorrhea (period cramps). In addition, the company targets individuals that need sleep-aids with its specially formulated low melatonin cannabinoid gummies.
IGC has also introduced a low-calorie CBD- and caffeine-infused energy beverage brand (www.SundaySeltzer.com) that is currently available for purchase. The company’s brands are founded on the belief that effective natural solutions should be affordable and accessible to everyone. As the demand for natural products targeting women’s wellness and energy drinks continue to grow, these products are seeing strong traction in the market.
The company operates three facilities – a large GMP (Good Manufacturing Production Standards) certified facility that includes extraction, distillation, and manufacturing, in Washington State; a GMP-211 (pharmaceutical) grade facility in Maryland; and a facility licensed for controlled substances including cannabis in Bogota, Colombia, with complete access to legal licensed cannabis where the company conducts its testing.
In addition, the company’s development under Magistral Formulations is approved by INVIMA (Colombia National Food and Drug Surveillance Institute) to treat neurological disorders, non-oncological chronic pain, and mental disorders.
IGC’s intellectual property (IP) portfolio comprises of eight patents that it controls and seven patent applications. The portfolio includes #11,446,276, a patent for extreme low dose THC treatment of Alzheimer’s that was granted in September 2022.
The company is headquartered in Potomac, Maryland.
IGC-AD1
IGC-AD1 is the company’s leading drug candidate for the treatment and relief of Alzheimer’s symptoms. A significant amount of research on Alzheimer’s cell lines has shown that the active agents in IGC-AD1 reduce plaques and neurofibrillary tangles that are the hallmarks of Alzheimer’s. Further, micro-dosing of THC, as shown in cell lines, could increase the functioning of mitochondria and potentially promote the growth of new neural pathways (neurogenesis). The research shows that micro-dosing of THC affects the brain radically differently from the normal higher dosing of THC.
While there is a significant body of research showing that THC is neuro-toxic at normal levels of dosing, micro-dosing of THC has been shown to be non-toxic to neurons. With the results of these preclinical studies, the company developed an oral formulation, IGC-AD1. The company recently completed a safety and tolerability Phase 1 trial on Alzheimer’s patients and has initiated a Phase 2, multi-site, double-blind, randomized, placebo-controlled trial of the safety and efficacy of IGC-AD1 on agitation in participants with dementia due to Alzheimer’s disease at sites in the U.S. and Canada. IGC expects the Phase 2 trial to take between 9 and 12 months to complete, barring unknown factors such as, for example, a resurgence of COVID and the enforcement of lockdowns and travel restrictions.
With further successful trials and FDA approvals, IGC hopes to bring a drug based on natural THC as an effective treatment for agitation in Alzheimer’s to market.
TGR-63
The company’s other molecule, TGR-63, has been shown to reduce the neurotoxicity that impacts memory loss in preclinical trials with mice. On a dose dependent manner, transgenic Alzheimer’s mice treated with TGR-63 showed improvement in memory relative to control.
Both drug candidates, IGC-AD1 and TGR-63, have shown their ability to reduce the brain plaques associated with memory loss in Alzheimer’s in mice.
With further successful trials and FDA approvals, IGC hopes to bring TGR-63 as a treatment for Alzheimer’s disease to market.
Market Opportunity
Alzheimer’s disease impacts over 55 million people worldwide and about 5.5 million individuals in the U.S. Over 70% of these patients face debilitating symptoms, including anxiety, depression, and agitation (Mendez, 2021). Agitation in dementia patients can include excessive physical movement and verbal activity, restlessness, pacing, belligerence, aggression, screaming, crying, and wandering.
In 2020, the estimated healthcare costs for Alzheimer’s disease in the U.S. were $305 billion. Medicare and Medicaid covered about 70% of those costs, leaving considerable burden on patients and families. At the current rate of growth of Alzheimer’s and other dementia diagnoses, those costs are estimated to reach over $1 trillion by 2050.
Currently, there are no FDA-approved medications to alleviate the symptoms of dementia due to Alzheimer’s disease, providing a tremendous opportunity for formulations that can have an impact on quality of life and disease progression.
Management Team
Richard Prins has been chairman at IGC since 2012 and served as an independent director since 2007. From March 1996 to 2008, he was the Director of Investment Banking at Ferris, Baker Watts, Incorporated. Prins served in a consulting role to RBC until January 2009. He currently volunteers full time with a non-profit organization, Advancing Native Missions, and is a private investor. Since February 2003, he has been on the board of Amphastar Pharmaceuticals Inc. He holds a bachelor’s degree from Colgate University and an MBA from Oral Roberts University.
Ram Mukunda is CEO and President of IGC. He has been the chief inventor and architect of most of the company’s patent filings and is responsible for the company’s strategic positioning. Prior to IGC, he was founder and CEO of Startec Global Communications, which he took public in 1997. He served as Strategic Planning Advisor at Intelsat, a communications satellite services provider. From 2001 to 2003, he was a Council Member at Harvard’s Kennedy School of Government, Belfer Center of Science and International Affairs. He was named the 1998 Ernst & Young Entrepreneur of the Year. He holds bachelor’s degrees in electrical engineering and mathematics, and a master’s degree in engineering from the University of Maryland.
Dr. Jagadeesh Rao is the company’s Principal Scientist. His career spans two decades in the public sector and product R&D for Johnson & Johnson. He leads IGC’s scientists in the development of pharmaceutical and OTC products. He worked for the federal National Institutes of Health, and for the National Institute on Drug Abuse. His Ph.D. in Neurochemistry is from the National Institute of Mental Health & Neurosciences in India. He did postdoctoral training at the University of Illinois-Chicago.
Claudia Grimaldi is a Director, Vice President, Principal Financial Officer, and Chief Compliance Officer for IGC. She also serves as a Director/Manager Director for some of the company’s subsidiaries. She graduated with highest honors from Javeriana University in Colombia with a bachelor’s degree in psychology. She holds an MBA, graduating with highest honors, from Meredith College in North Carolina. In addition, she has attended the Darden School of Business Financial Management Executives program and the Corporate Governance Program at Columbia Business School. She is currently pursuing her Directorship Certification with the National Association of Corporate Directors. She is fluent in both English and Spanish.
IGC Pharma Inc. (NYSE American: IGC), closed Monday's trading session at $0.3326, up 3.6137%, on 111,439 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.2785/$0.8505.
Recent News
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - New Certification Will Help Standardize Marijuana Product Testing by Labs
- 420 with CNW - German Cannabis Imports Grow to Record Levels
- 420 with CNW - Missouri Sees Uptick in Marijuana Jobs Shortly After Legalization Ballot
Data443 Risk Mitigation Inc. (OTC: ATDS)
The QualityStocks Daily Newsletter would like to spotlight Data443 Risk Mitigation Inc. (OTC: ATDS).
• As the world becomes increasingly digital, cyber threats aboundwith the
• The global cyber security solutions market is
• Data443 Risk Mitigation offers both comprehensive and a la carte solutions for companiesto mitigate cybersecurity risks
•
Our increasing global dependence on digital infrastructure is adouble-edged sword. While benefits like improved efficiencies,technological innovation and economic growth propel thetransformation forward, it has also created a myriad ofopportunities for cybercriminals. An expensive ecosystem isemerging, one where progressively digital enterprise and theiradversaries are in a relentless pursuit to develop new technologiesto defeat the other.
Data443 Risk Mitigation Inc. (OTC: ATDS) is a data security and privacy software company for ALL THINGS DATA SECURITY™. The company is committed to organizing the world’s information by identifying and protecting all sensitive data regardless of location, platform or format.
Data443 provides software and services to enable secure data across devices and databases – at rest and in transit – locally, on a network, or in the cloud. With over 10,000 customers in more than 100 countries, Data443 provides a modern approach to data governance and security. The company’s framework helps customers prioritize risk, identify security gaps, and implement effective data protection and privacy management strategies.
Data443 derives revenue primarily from contracts for subscriptions to access its SaaS platforms, and ancillary services provided in connection with its subscription services. In today’s ever-changing environment with unique and complex requirements for data privacy, governance and hybrid workforces, every organization needs to know where all their data is, who has access to it and how sensitive it is. Data443 provides the tools needed to give companies control over their data processing activities, with capabilities for identifying, reporting and migrating or deleting sensitive data.
The company is headquartered in Research Triangle Park, North Carolina.
Products
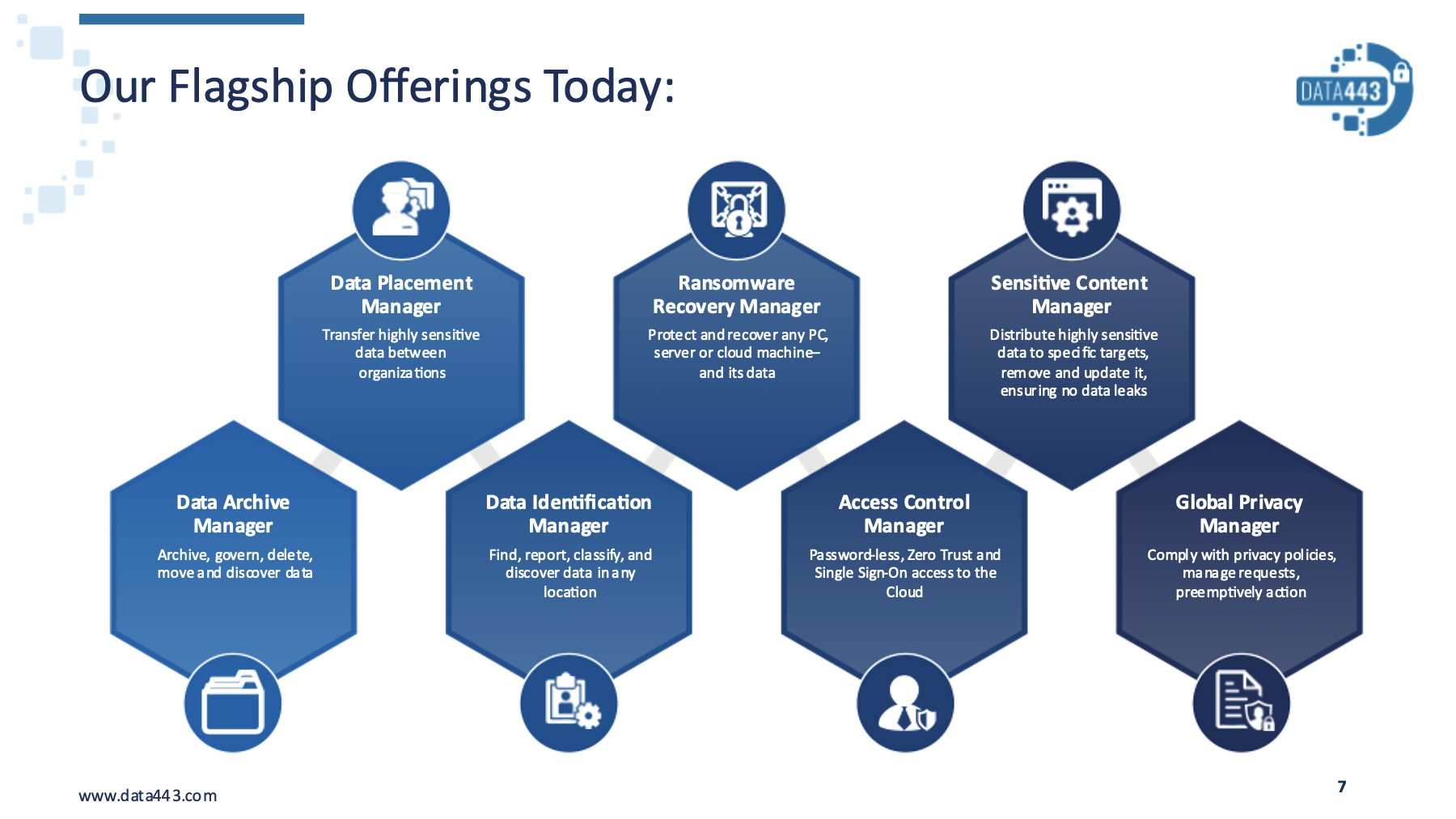
Focused on data security with a privacy-forward methodology, the Data443 product suite delivers solutions designed to securely manage data and data privacy needs on-premises, in the cloud and in hybrid environments. Offerings include:
- Data Identification Manager reduces risk by shining a light on dark data across cloud, on-premises and hybrid environments. From a centralized dashboard, Data Identification Manager provides the ability to automatically inventory all data repositories, classify and tag all data, and enable global search and discovery – all through an agentless deployment.
- Data Placement Manager quickly and securely transfers sensitive data over any public or private network. Available as an HP Nonstop server-based application and for Windows, Linux or any public cloud provider, Data Placement Manager enables the scheduling, routing, formatting and transfer of business-critical data.
- Data Archive Manager is an “all information, anywhere” archiving solution designed to handle and manage all types of privacy requests across cloud, on-premises and hybrid environments. With over 15 years operational history and hundreds of clients managing millions of mailboxes, the platform is purpose-built for information archiving, retention and privacy request management.
- Data Hound™ is a data discovery, classification and capture toolset that enables organizations to perform quick scans, detailed reporting and subsequent data actions based on policy.
- Ransomware Recovery Manager is the only industry solution that actively recovers the device, operating system and data with a simple reboot. Using patented, proven technology, the product produces 100% effectiveness for the whole device and datasets.
- Access Control Manager provides user ID and passwordless access to quickly enable trust across an organization’s entire ecosystem. Its unique architecture allows it to leverage multiple distributed authoritative sources to understand and resolve a typical access request – with the ability to enable or deny the action on the fly.
- Global Privacy Manager provides organizations one comprehensive view, for all privacy requirements, across all enterprise data, all at once. This unmatched visibility into an organization’s data assets ensures that all private and sensitive data can be identified and protected and that enterprises can obey all relevant privacy laws in any jurisdiction.
- Sensitive Content Manager is a security-centric collaboration service designed to give organizations the tools needed for successful content sharing, collaboration and safe distribution with full enterprise management in mind. With a continuous sync feature, encrypted data is automatically downloaded and updated in real time – regardless of location – ensuring that users have the most accurate data available.
Market Outlook
A report from Allied Market Research estimates that the global data security market was worth about $19 billion in 2021 and is projected to reach a value of $54.23 billion by 2027. That represents a CAGR of more than 18% for the forecast period, making data security one of the hottest areas within IT.
Separately, Fortune Business Insights estimates the global data privacy software market is valued at $2.36 billion in 2022 and projects it will grow to $25.85 billion by 2029. That represents a CAGR of 40.8% over the forecast period.
Management Team
Jason Remillard is President, CEO and Founder of Data443. He is responsible for overseeing global expansion, management, execution and corporate development. With over 25 years in global enterprise and B2C software sales and marketing, he brings deep leadership and technical experience, having spent previous time at Fortune 500 companies such as Deutsche Bank, TD Bank, IBM & Merrill Lynch.
Greg McCraw is CFO at Data443. He has over 25 years of experience helping businesses strengthen their accounting and finance operations. He previously served as Vice President of Finance for a dental services organization active in acquisitions, and, prior to that, he was managing director of a boutique accounting and finance consulting firm advising Fortune 500 clients in pharmaceutical, financial services, and private equity sectors on how to execute on regulatory and compliance solutions.
Bennett Pursell is Data443’s Chief Technology Officer. He has over 20 years of experience in IT architecture, security governance and systems integration. Prior to his role at Data443, he served as Head of Technology Architecture at Moody’s Investor Services and was Vice President and Technical Architect of Cloud Computing at Deutsche Bank, along with a host of technical and project management roles dating back to 2006, after starting his career as a web developer with a few startups and running research labs.
Kirill Kashigin is Chief Software Architect at Data443. He leads the development and quality teams, and serves as technical adviser and subject matter expert, bringing vast technical knowledge on privacy management and data security. Formerly the CTO of FileFacets, he has nearly 20 years in development of high-performance systems and deployment.
Data443 Risk Mitigation Inc. (OTC: ATDS), closed Monday's trading session at $0.08, up 23.0769%, on 33,006 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.025/$6.99.
Recent News
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc.'s (ATDS) Award-Winning Ransomware Defense Shines as Cybercrime Persists
- InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Secures Contract Win with Multinational Client
- InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Secures Another Contract Win from Leading Fintech Giant
Advanced Container Technologies Inc. (OTC: ACTX)
The QualityStocks Daily Newsletter would like to spotlight Advanced Container Technologies Inc. (OTC: ACTX).
Latest data shows that cannabis sales in the state of Missouri forthe month of March hit $126.2 million. This is month two sincerecreational retailers were launched in the state, after voterspassed a measure to legalize the recreational use of the drug last year. The revenue generated represents a 22.4% increase inmarijuana sales from the amount generated in February. Broken down,recreational and medical marijuana sales stood at $93,500,000 and$32,700,000 respectively. Projections expect that before the yearends, collective marijuana sales in Missouri
Advanced Container Technologies Inc. (OTC: ACTX) is in the business of selling and distributing self-contained, automated, indoor “micro-farms” called Grow Pods, along with related equipment and supplies. Additionally, the company designs and sells patented proprietary medical-grade plastic containers, known as the Medtainer®, that store and grind pharmaceuticals, herbs, teas and other solids or liquids.
ACTX is the leading distributor of Grow Pods. With a controlled environment, food and herbs can be grown without pesticides, harmful chemicals or risk of pathogen contamination, and with low energy consumption. Restaurants, grocery stores, non-profits, MSOs and entrepreneurs can use Grow Pods to ensure a fresh supply of ultra-clean produce year-round.
The company entered the Grow Pod business in October 2020 with its acquisition of all shares of Advanced Container Technologies Inc., a California corporation. As of February 28, 2022, ACTX is exploring the acquisition of the assets and the assumption of some or all of the liabilities of GP Solutions Inc., the developer and manufacturer of Grow Pods, for which ACTX is currently the sole U.S. distributor.
Because Grow Pods can be located almost anywhere, produce can be grown closer to the point of consumption and harvested at its peak, providing nutritious fruits and vegetables where needed. Indoor micro-farms, utilizing a practice known as vertical farming, have attracted the attention of governments and universities, which are now promoting vertical farming as a way to combat food insecurity and inequities.
The United States Department of Agriculture (USDA) has stated that vertical farming “is no longer a futuristic concept.” The department is enthusiastic about vertical farming, particularly those utilizing repurposed shipping containers, such as Grow Pods. Arizona State University reports that vertical farming reduces water use by 90 percent compared to conventional farming but produces 10 times the crop yield.
Products
Grow Pods
One of the company’s main business units is focused on selling advanced, self-contained hydroponic containers called Grow Pods. These unique and innovative automated systems are essentially micro-farms that can be placed virtually anywhere and, with their controlled and specially filtered environment, allow cultivation of a wide variety of crops, 365 days a year. The Grow Pod controlled environment offers major advantages for the production of high-value crops. The ability to grow year-round and the ability to cultivate in a smaller footprint using less water and power are some of the primary advantages of the system. Grow Pods offer constant temperature, humidity and airflow control, as well as automated watering and lighting schedules for optimal growth and minimal labor requirements, regardless of crop.
Containers
ACTX meets the needs of the pharmaceutical and medical markets, including the cannabis and hemp industries, with patented packaging systems. The company designs, customizes, brands and sells proprietary medical grade plastic containers that can store pharmaceuticals, herbs, teas and other solids or liquids, with a special built-in feature that can grind solids and shred herbs. The company’s flagship container product is the patented Medtainer®, a child resistant, medical-grade herb container and grinder that is water-tight, air-tight and smell proof. Packaging in the cannabis industry is critical, with numerous stringent regulations about how cannabis products must be packaged and labeled. ACTX also offers custom-branded, compliant vacuum seal bags and other retail container solutions.
Equipment and Supplies
ACTX markets and sells two principal products: Grow Pods, which are specially modified insulated shipping containers manufactured by GP Solutions Inc., in which plants, herbs and spices may be grown hydroponically in a controlled environment, and Medtainers®, which may be used to store pharmaceuticals, herbs, teas and other solids or liquids and can grind solids and shred herbs. The company also markets and sells various products related to Grow Pods and the Medtainer®, as well as providing private labeling and branding services for purchasers of Medtainers® and certain related products.
GP Solutions manufactures and sells other products, such as humidity controllers and LED lighting systems for vertical farming. The company’s specially designed lighting panels are programmed to emit the exact wavelength of light that each crop requires. The system has a daybreak-to-nightfall feature that gives plants the proper chromatic signals to grow rapidly and fruitfully. High efficiency LED light strips supply the crops with a red and blue light spectrum required for photosynthesis in the spectrum that plants need most.
Market Overview
The global vertical farming market is expected to reach $33.02 billion by 2030, according to a new report by Grand View Research. The market is forecast to expand at a CAGR of 25.5 percent from 2022 to 2030, according to Grand View. Escalating production of biopharmaceutical products, including cannabis, is anticipated to drive the market. The building-based segment of the market is expected to register a significant CAGR of 27.8 percent over the projected period. In addition, the climate control segment is expected to see high growth.
The global cannabis packaging market is expected to reach $14.34 billion by 2028, according to analysis by Reports and Data. The analysis forecasts 1,700 percent growth in cannabis users by the end of 2026, with packaging likely observing a whopping 26.42 percent growth in the forecast period. There are significant barriers to entry in the cannabis packaging market, giving an advantage to companies already established in the sector. These barriers include developing a thorough knowledge of the myriad regulations that govern cannabis packaging (which differ in each state), and child-resistance requirements.
Management Team
Douglas P. Heldoorn is the Founder and Chairman of Advanced Container Technologies Inc. He also holds the positions of President, CEO and COO at the company. Mr. Heldoorn has served on the Board of Directors since its inception in 2013. He has also previously held the position of Executive General Manager at Nissan Motor Corp.
Jeffory A. Carlson is CFO and Treasurer of ACTX. Mr. Carlson has also served as the company’s Corporate Controller since 2014.
Advanced Container Technologies Inc. (OTC: ACTX), closed Monday's trading session at $0.3, even for the day, on 350 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.073/$1.05.
Recent News
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - State Data Shows Marijuana Sales in Missouri Totaled $126M in March
- 420 with CNW - Maryland Legislators Finalize Details of Recreational Marijuana Sales Launch
- 420 with CNW - Cannabis Prices Slump in Massachusetts Amid Supply Glut
Prime Harvest Inc.
The QualityStocks Daily Newsletter would like to spotlight Prime Harvest Inc.
To view the full article, visit https://ibn.fm/0nTJu
Prime Harvest Inc., based in San Diego, California, is a technology-focused, full-service cannabis company with horizontally diversified operations spanning various segments of the cannabis value chain, from licensing acquisition and compliance management to direct-to-consumer operations. The company is leveraging a long-term strategy of investing in the growth and scale of licensed assets anchored by the power of data-driven technology to expand its footprint throughout California.
Sustainability is key to Prime Harvest’s corporate vision. The company aims to ensure that the communities it serves capture their fair share of the fruits of the industry’s growth, including financial profit, employment opportunities, environmental enrichment and impactful innovation through R&D and education.
The company’s mission is to appeal to the ethos of the cannabis consumer by setting a new operational standard emphasizing accountability, sustainability and community. With this commitment, Prime Harvest continues to work toward positively affecting millions of lives through the creation of a world-class platform that caters to strengthening the commercial cannabis pipeline.
Jaxx Cannabis
Jaxx Cannabis is the flagship brand in Prime Harvest’s portfolio. Through Jaxx Cannabis, the company aims to use technology to facilitate a true customer-centric culture while enhancing the overall craft cannabis experience. Jaxx features an expertly curated selection of premium products from some of the most respected brands in the thriving California market.
Key values serving as the foundation of Jaxx Cannabis include:
- Creating and nurturing a welcoming culture for all
- Unlocking the true potential of customer value
- Being innovative in uncovering new ways to grow both the company and the industry
- Meeting the wants and needs of consumers to promote profitability
- Remaining accountable for the results of its operations
It is these values that differentiate Prime Harvest and Jaxx Cannabis in the California cannabis sector.
Brand Partnerships
Prime Harvest works diligently to establish strong alliances with complementary brands that are in alignment with its culture and values. Through a combination of deliberate foresight and strategic action, the company seeks to grow existing cannabis brands and continuously discover new, high-potential performers that are primed for long-term success.
These partnerships enhance Prime Harvest’s efforts to transform the world’s cannabis access and bring its consumers high-quality products that are fair for both people and the planet.
Responsibility
Prime Harvest remains committed to the goal of creating a more sustainable environment, now and in the future. Concern for human beings and the environment can be observed in every facet of its operations, including its ongoing R&D activities dedicated to exploring methods of reducing and repurposing waste into composite materials and exploring the potential of the hemp plant for industrial and wellness contributions.
The company is a proud member of the Community Alliance Program, a foundation that seeks to make a difference in local communities by providing financial assistance for educational programs, housing homeless veterans, creating urban farms, and holding local arts initiatives for children and adults. The program also helps explore the natural healing attributes of medical cannabis through research, development, clinical trials, and advocating for the safe access of cannabis to those in need.
Market Overview
Ongoing changes in U.S. state government policies toward cannabis are expected to cause demand for legal marijuana to surge. In addition, the number of indications for which medical marijuana is prescribed continues to increase. These factors are expected to rapidly boost legal sales of cannabis products.
Legal sales across the U.S. hit a record of $17.5 billion in 2020, marking an increase of 46% over 2019, according to Forbes. This strong growth is expected to continue. According to a Grand View Research report, the global legal marijuana market is forecast to grow at a CAGR of 26.7 percent from 2021 to 2028.
California – Prime Harvest’s home state – has consistently led the pack in terms of U.S. cannabis sales. The Motley Fool pegged cannabis spending in the Golden State at $3.8 billion in 2020, more than doubling the second state on its list.
Leadership Team
The Prime Harvest team is composed of true experts in their respective fields focused on building a world-class organization capable of driving the cannabis industry and movement forward.
E. Duane Alexander is the company’s Founder and CEO. He brings to the team more than 25 years of real-world, hands-on cannabis retail, marketing and commercial operations experience. Mr. Alexander has championed 40+ cannabis license applications throughout the western U.S. to date.
John Wilczak is the COO of Prime Harvest. He has 30+ years of executive management, strategy development & configuration experience with GE, pharmaceutical and agriculture companies. Mr. Wilczak is a Brown & Columbia MBA with vast knowledge of technology driven intellectual properties.
Andrea Jenson is the Chief Financial Officer of Prime Harvest. As CFO, she is responsible for all the company’s financial functions, including accounting, corporate finance and investor relations. Her career spans more than 20 years of varied experience in financial management, business leadership and financial strategy.
John Kazanjian is the VP of Business Development of Prime Harvest. He has worked over 40 years in business operations, brand marketing, sales and investor/lender communications. Mr. Kazanjian earned his B.S. from Rutgers University and his MBA from Harvard University.
Johann Balbuena is the Chief Marketing Officer of Prime Harvest. She has more than six years of experience in California cannabis licensing acquisition and compliance management. Ms. Balbuena has led multimedia production and content marketing efforts for the likes of the Social Club TV app, The Emerald Cup, High Times, Weedmaps and Synergy.
Recent News
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Prime Harvest Inc. Launches Weed 4 The People Reg A+ Offering to Fund Cannabis Retail and Delivery Expansion Across California
- InvestorNewsBreaks – Prime Harvest Inc. Secures Reg A+ Qualification, Eyes New Heights of Opportunity

Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF)
The QualityStocks Daily Newsletter would like to spotlight Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF).
•Cartier Iron opted to change its name to Cartier Silver in October2022, part of a wider pivot within its business plan
•The revamped company, which is both a sister company and minorityshareholder in Eloro Resources (TSX: ELO), recently optioned 100%of the zinc-silver Los Chorrillos Project, within Bolivia’s CerroRico region
•The company, which boasts the same, experienced management team asEloro Resources’, recently completed their initial capital raise of$3 million
•Proceeds from the capital raise will be usedto finance initial exploration at theLos Chorillos site
In October 2022, when Cartier Iron made a dramatic pivot within itsbusiness and amended its name to
Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) is a publicly traded exploration and mine development company with a portfolio of gold and base-metal properties in Bolivia, Peru and Quebec.
The company has an option to acquire a 99% interest in the highly prospective Iska Iska Property, classified as a silver-tin polymetallic epithermal-porphyry complex, a significant mineral deposit type in the Potosi Department of southern Bolivia. Iska Iska is a road-accessible, royalty-free property.
Eloro also owns an 82% interest in the La Victoria Gold/Silver Project, located in the North-Central Mineral Belt of Peru, some 50 kilometers south of Barrick’s Lagunas Norte Gold Mine and Pan American Silver’s La Arena Gold Mine. La Victoria consists of eight mining concessions and eight mining claims encompassing approximately 89 square kilometers. La Victoria has good infrastructure, with access to road, water and electricity, and is located at an altitude that ranges from 3,150 meters to 4,400 meters above sea level.
The company has a strong management and technical team working diligently to uncover the value of both Iska Iska and La Victoria. Eloro is based in Toronto, Canada.
Projects
Iska Iska – Potosi, Bolivia
Iska Iska is associated with a Miocene possibly collapsed/resurgent caldera, emplaced on Ordovician age rocks with major breccia pipes, dacitic domes and hydrothermal breccias. The property is wholly controlled by the title holder, Empresa Minera Villegas S.R.L. It is located 48 kilometers north of Tupiza city, in the Sud Chichas Province of the Department of Potosi. This is an important mineral deposit type in the prolific South Mineral Belt of Bolivia. Eloro commissioned a NI 43-101 Technical Report on Iska Iska, which was completed by Micon International Limited and is available on Eloro’s website and under its filings on SEDAR.
A fully financed drill program is currently underway on the property, situated near world-class deposits including Silver Sand, San Bartolomé, Pulacayo, San Cristobal, San Vicente, Chorolque, Tasna, Choroma and Siete Suyos. Iska Iska is in the southwest part of the Eastern Cordillera, which hosts a number of major polymetallic mines and mineral deposits. Drilling and continuous channel sampling results have demonstrated some very high metal values, especially silver and tin, within an immense system, where mineralization has been encountered in every drill hole to date. The company believes there is excellent potential for world-class bulk mineable deposits.
La Victoria – Ancash, Peru
The La Victoria project, targeting gold and silver production, is situated near world-class, low-cost gold producers Pan American Silver and Barrick Gold Corporation. Located in Ancash Department, La Victoria sits on the western slopes of the Peruvian Andes. The property is located 12 hours from Lima, with a travel distance of 600 kilometers. The nearest road accessible population centers from La Victoria are Huandoval, Pallasca and Cabana. The project includes four principal mineralized zones in Peru’s prolific North-Central Mineral Belt – San Markito, Victoria, Victoria South and Ccori Orcco – with excellent potential for gold discovery. Operations at La Victoria are planned to proceed with a 2,000-meter diamond drilling program to test targets to outline potential resources at San Markito. Trenching and sampling confirmed high silver values and veins at San Markito in 2020.
Market Outlook
According to industry association The Silver Institute, the outlook for silver demand is exceptionally promising, with global demand forecast to rise to a record high of 1.112 billion ounces in 2022. The increase will be driven by record silver industrial fabrication, which is forecast to improve by 5%, as silver’s use expands primarily in solar energy and electric vehicle (EV) manufacturing. The institute states that government commitments to carbon neutrality have resulted in a rapid expansion of green energy projects, driving record photovoltaic panel installations which are expected to lift silver demand in this segment to an all-time high in 2022.
Rising demand in the electronics industry is also boosting the demand for tin, which is primarily used in solder. The electronics and electrical industries use solders containing 40-70% tin, which provide strong and reliable joints under a variety of environmental conditions. At present, the majority of the assemblers are using patented tin-and-copper-based solders. Mordor Intelligence estimated tin demand at 387 kilotons in 2021 and forecasts demand growth of 2.5% annually through 2027. Over the medium term, surging demand from the EV market and increasing applications in the electrical and electronics industry is expected to drive the market.
Management Team
Thomas G. Larsen is CEO of Eloro. He has more than 40 years of experience in the investment industry, specializing in corporate finance and management of junior resource companies, raising in excess of C$200 million. He previously held the position of President and Chief Executive Officer of Champion Iron Limited. Prior to that, he was President and Chief Executive Officer of Champion Iron Mines Limited.
Dr. Bill Pearson is Executive VP of Exploration for Eloro. He has more than 40 years of direct experience in the exploration and production of minerals worldwide. He played an integral role in the acquisitions of Desert Sun Mining Corp. by Yamana Gold in 2006 and Central Sun Mining by B2 Gold in 2009. He was formerly VP Exploration at Desert Sun Mining and Senior VP at Central Sun Mining.
Miles Nagamatsu, CPA, is CFO at Eloro. He has over 30 years of experience in accounting, management, lending, restructurings and turnarounds. Since 1993, he has acted as a CFO of public and private companies primarily in the mineral exploration and investment management sectors. He holds a Bachelor of Commerce degree from McMaster University.
Osvaldo Arce Burgoa is General Manager at Eloro. He is a geological and mineral processing engineer with 26 years of experience in Bolivia. He is a former President of the Bolivian Geological Society, Main Technical Advisor of the National Mining Corporation (COMIBOL) and has served as exploration manager and chief geologist at various mining and exploration companies. He has authored two books on Bolivian geology and holds a doctorate in mining engineering from Tohoku University in Sendai, Japan.
Eloro Resources Ltd. (OTCQX: ELRRF), closed Monday's trading session at $2.79, off by 1.4134%, on 42,667 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.09/$3.91.
Recent News
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Cartier Silver Corp. (CSE: CFE) Follows in Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) Footsteps Within Bolivia's Potosi Region
- InvestorNewsBreaks - Eloro Resources Ltd. (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Announces Latest Results, Completion of Santa Barbara Definition Drill Program at Iska Iska Project
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Releases Promising Update on Iska Iska Project at Metals Investor Forum
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
•Chris Bunka, Lexaria’s CEO, has reiterated the company’s commitmentto creating value for its shareholders with the licensed use of itspatented DehydraTECH(TM) technology
•Mr. Bunka has noted that Lexaria is already in discussions withinterested parties in Europe and North America, a move thatpresents a critical fiscal opportunity for the company
•He has emphasized how profitable new licensing agreements could befor the company, presenting additional avenues for Lexaria togenerate revenue and create value for its shareholders
•As the company continues to make a transition toward a morecommercial focus in 2023, Bunka notes that licensing will play anintegral part
Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing.
DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future.
Lexaria has a collaborative research agreement with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has filed for patent protection for specific delivery of nicotine, vitamins, NSAIDs, testosterone, estrogen, cannabinoids, terpenes, PDE5 inhibitors (with brand names like Viagra), tobacco and more.
Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats.
The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products.
DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions.
DehydraTECH Technology
Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following:
- Speeds up delivery – the effects of the product are felt by the subject in just minutes.
- Increases bioavailability – the technology is much more effective at delivering a drug or product into the bloodstream.
- Increases brain absorption – animal testing suggests significant improvement in the quantity of drug delivered across the blood-brain barrier.
- Improves drug potency – more of the ingested product is made available to the body, so lower doses are required to achieve the desired effect.
- Reduces drug administration cost – lower doses mean lower overall drug costs.
- Masks unwanted taste – the technology eliminates or reduces the need for sweeteners.
Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery.
Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine.
Market Outlook
Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments:
- Oral Cannabinoids – a market estimated to be worth $18.4 billion in 2021 and expected to reach $46.2 billion by 2025.
- Antivirals – an estimated $52.1 billion market in 2021 that’s expected to grow to $66.7 billion by 2025.
- Oral Mucosal Nicotine – smokeless tobacco products, a $13.6 billion market in 2018, is forecast to grow at 7.2 percent annually through 2025.
- Human Hormones – estrogen and testosterone replacement therapies represented a $21.9 billion market in 2019, with a forecast CAGR of 7.7 percent through 2027.
- Ibuprofen and Naproxen – NSAID sales totaled $15.6 billion globally in 2019 and are projected to reach $24.4 billion by 2027.
- Vitamin D3 – the global market size was $1.1 billion in 2021, growing at 7 percent per year and expected to reach $1.7 billion in 2026.
Management Team
Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations.
John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents.
Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations.
Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch.
Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia.
Lexaria Bioscience Corp. (LEXX), closed Monday's trading session at $2.17, off by 0.458716%, on 27,535 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.80/$4.24.
Recent News
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Eyes the Licensed Use of its DehydraTECH(TM) Technology to Complement its Royalty Earnings and Realize Revenue in Advance of Royalties
- TinyGemsBreaks - Lexaria Bioscience Corp.'s (NASDAQ: LEXX) DehydraTECH(TM) Demonstrates Positive Results in First Diabetes Study
- Lexaria Bioscience Corp. (NASDAQ: LEXX) Revolutionizing Drug Delivery Potential for Active Pharmaceutical Ingredients Using Patented DehydraTECH(TM) Technology
Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF)
The QualityStocks Daily Newsletter would like to spotlight Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF).
As the world
Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) is advancing the next generation of nickel-cobalt sulfide projects to deliver the metals needed to power the electric vehicle (EV) revolution and feed the high growth stainless steel market. The company is one of only a few new sources of potential supply outside Indonesia and China.
Canada Nickel possesses industry leading nickel expertise and is focused on low risk, well established mining jurisdictions. The company has launched wholly owned subsidiary NetZero Metals Inc. to develop zero-carbon production of nickel, cobalt and iron and has applied in multiple jurisdictions to trademark the terms NetZero Nickel, NetZero Cobalt and NetZero Iron. Canada Nickel is also pursuing development of processes to allow net zero carbon production of these elements.
Canada Nickel is currently anchored by its 100% owned flagship Crawford Nickel-Cobalt Sulfide Project with large-scale potential located in the heart of Ontario’s prolific Timmins-Cochrane mining camp, adjacent to major infrastructure.
The company believes the EV industry and many other consumer sectors have an urgent need for zero-carbon metal this decade, not in 20-25 years as contemplated by some resource companies. Canada Nickel also believes that nickel supplies from Indonesia and other Pacific island nations, typically controlled by Chinese-owned companies, are not the answer for batteries needed by GM, Ford and the European automakers working to develop and manufacture EV models.
The company is headquartered in Toronto.
Crawford Nickel-Cobalt Sulfide Project
The Crawford Nickel-Cobalt Sulfide Project is the largest sulfide discovery since the early 1970s and contains the fifth-largest nickel sulfide resource in the world, based on Measured & Indicated resources, according to the latest update. The Crawford project is expected to be one of the largest base metal mines in Canada based on results of a Preliminary Economic Assessment. Early projections by Canada Nickel estimate that the project has the potential to produce 50,000 tons of nickel per year. The company is now in the final stages of completing the project’s feasibility study.
The project is projected to produce 2.8 tons of CO2 per ton of nickel equivalent production, which is 89% lower than the industry average of 34 tons of CO2 per ton of nickel equivalent production.
The company is taking significant steps toward developing the Crawford project as a net zero carbon producer. In addition to harnessing the natural ability of the project’s geology to act as a carbon sink through spontaneous reaction of the host rock once exposed to atmospheric conditions called mineral carbonation, Canada Nickel has discovered a new way to enhance carbon capture, termed In Process Tailings (IPT) Carbonation. This act of conditioning the tailings with a concentrated stream of carbon dioxide before deposition has been demonstrated at lab scale to achieve carbon capture at a rate 8-12 times faster than naturally occurring sequestration, achieving more than 60% of the capture that had previously taken six days.
These latest results move the company further toward production of Net Zero Nickel™ and generation of 21 tonnes of CO2 credits per tonne of nickel, which would produce an estimated average of 710,000 tonnes of CO2 credits annually and 18 million total tonnes of CO2 credits over the expected life of mine. IPT Carbonation does not require complex new technologies and major process modifications and could encourage the development of a net zero carbon industrial cluster centered around the Crawford project.
Canada Nickel in January 2023 announced that its latest test work results support the incorporation of carbon capture and storage into the Crawford project. The company believes that utilization of existing process streams should allow IPT to be efficiently engineered and incorporated into the project’s flowsheet, with an integrated feasibility study for the project expected in the second quarter of 2023.
In December 2022, Canada Nickel announced its engagement on Deutsche Bank Securities Inc. (“Deutsche Bank”) and Scotiabank – two of the world’s leading investment banks with a broad base of mining and industrial expertise – as financial advisors for the equity component of the project financing for the Crawford project. In the same release, the company announced the completion of another significant permitting milestone by filing the detailed project description with the Impact Assessment Agency of Canada. Canada Nickel targets receipt of permits by mid-2025, with construction to immediately follow.
Additional Projects
The Reid Nickel Property is located just 16 kilometers southwest of Crawford, or 37 kilometers northwest of Timmins, and contains an ultramafic body with a target geophysical footprint of 3.9 square kilometers. Preliminary assay results from Canada Nickel’s summer/fall drilling program confirm the presence of mineralized dunite, as well as currently undefined higher-grade sections. Partial assay results confirm expected nickel grades. Nickel mineralization in serpentinized dunite was found in all 16 holes drilled to date.
The Sothman Nickel Property is located 70 kilometers south of Timmins. Five drill holes on the eastern half of the target anomaly confirmed the continuation of ultramafic lithologies, primarily peridotite, with moderate to strong serpentinization and variable amounts of mineralization throughout.
The company in December 2022 announced positive drilling results from its ongoing regional exploration campaign at its Reid and Sothman properties. These latest results continue to reinforce the success of Canada Nickel’s geophysical targeting approach and increase the probability of success at the company’s other 20-plus properties within its 42 square kilometers of geophysical targets.
Building on this momentum, Canada Nickel in December 2022 announced its entry into a deal to acquire a 100% interest in the past producing Texmont property situated between the company’s properties south of Timmins. As noted in the news release, the acquisition of the Texmont property provides near-term smaller scale production potential and is highly complementary to the company’s large-scale Crawford and regional nickel sulphide projects.
Market Opportunity
Global demand leaves the market fundamentally short of nickel in the medium- and long-term. Global primary nickel demand will likely reach 3 million tons in 2022, up from 2.4 million tons in 2020, according to the International Nickel Study Group (INSG).
The INSG says primary nickel production is forecast to hit 3.1 million tons in 2022. Indonesia, the world’s largest nickel miner, halted exports of unprocessed nickel ore in January 2020, due to a government-imposed ban. Indonesia has floated the concept of a nickel cartel whose member nations would exert influence over world nickel supply and prices, similar to OPEC’s pricing power over oil.
Benchmark Minerals, a leading EV supply chain research firm, projects that, by 2035, world demand for nickel will double from current levels to 6 million tons annually. That growing demand represents a need for new nickel production equivalent to 70 mines the size of Canada Nickel’s Crawford Project.
Management Team
Mark Selby is Chairman, CEO and Director of Canada Nickel. He was formerly President and CEO of RNC Minerals, where he led a team that successfully raised over $100 million and advanced the Dumont nickel-cobalt project from initial resource to a fully permitted, construction-ready project. He has held senior management roles with Quadra Mining, Inco and Purolator Courier, and was a partner at Mercer Management Consulting. Since 2001, he has been recognized as one of the leading authorities on the nickel market. He graduated from Queen’s University with a Bachelor of Commerce.
Wendy Kaufman is CFO of Canada Nickel. She has 25 years of experience leading publicly listed mining companies in project financing, capital structuring, capital markets, accounting and internal controls, tax, and financial reporting and public disclosure. She was also previously CFO at Khiron Life Sciences Corp. and held CFO and senior finance positions at Pasinex Resources Limited, Primero Mining Corporation and Inmet Mining Corporation. She holds a Bachelor of Business Administration from Wilfrid Laurier University and is a Chartered Professional Accountant.
Steve Balch is VP Exploration at Canada Nickel. He is an Ontario registered geoscientist with 32 years of experience in geophysics, specializing in magnetic and electromagnetic methods. He founded Triumph Instruments and developed the AirTEM system, a multi-coil helicopter-borne EM system that is in use worldwide. He has also been active in borehole geophysics and helped develop new technologies including north-seeking gyros, temperature compensated induction conductivity probes, UAV-based magnetometers and high sensitivity magnetic gradiometers.
Christian Brousseau is VP Capital Projects at Canada Nickel. He is a professional engineer (P.Eng) with over 30 years of experience in engineering, design and construction in the Canadian mining industry, including six years as Project Director for the Dumont Project and three years as the Engineering and Construction Manager for Detour Gold. Prior to Detour, he held various construction management positions at Osisko’s Malartic Project and at Goldcorp’s Éléonore Project. He also spent eight years at Falconbridge supervising and managing various capital projects.
Canada Nickel Company Inc. (OTCQX: CNIKF), closed Monday's trading session at $2.05, up 3.0151%, on 73,251 volume with 375 trades. The average volume for the last 3 months is 57,207 and the stock's 52-week low/high is $1.04999995/$5.63000011.
Recent News
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Copper and Nickel: Major Players of the Energy Transition
- InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Excited by Preliminary Mineralogy Results at Midlothian
- InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Commences Next Phase of Permitting, Appoints Project Debt Advisor
Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF)
The QualityStocks Daily Newsletter would like to spotlight Ucore Rare Metals Inc. (TSX.V: BMR) (OTCQB: BTRMF).
To view the full press release, visit https://ibn.fm/p1bSh
Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) is a battery minerals company providing shareholders exposure to the global mega-trend of electrification while being focused on growth through cash-flow, exploration and acquisitions in favorable mining jurisdictions.
The company’s mission is the discovery, acquisition and development of battery metals (namely copper, cobalt, lithium, and graphite) in North America, South America and South Korea. It aims to become a leading low-cost producer of high quality, ethically sourced battery metals from high-grade, low impact mines in stable jurisdictions that are close to major consumer industries.
BMR is headquartered in Vancouver, British Colombia, with a portfolio of projects spanning Canada, the U.S., Chile and South Korea.
Project Portfolio
BMR’s current focus is the restart of its Punitaqui copper mine in Chile, as well as the exploration and development of its cobalt, lithium and graphite assets in North America and South Korea. The company also continues to identify and evaluate new project opportunities in its operating jurisdictions.
Its current portfolio includes:
Chile – Copper
BMR’s 100%-owned Punitaqui copper mine, acquired in March 2021, has the potential to generate an annual EBITDA of up to $50 million at or above a copper price of $4.25/lb. The company’s flagship project, the Punitaqui mine has been the subject of numerous milestones in recent months, including:
- BMR funded and completed a successful 32,526m resource drill program in 2022. Metallurgical testwork has confirmed the ability to produce excellent copper concentrates from each of the five zones tested, including recoveries ranging from 81% on the low end at Cinabrio Norte up to 96.5% at the Dalmacia deposit.
- The company in August 2022 reported the results of its first ever NI 43-101-compliant resource estimate for the underground deposits at its Punitaqui copper mining complex of 6.2 million tonnes grading 1.14% Cu in indicated category, along with 3.1 million tonnes grading 0.93% Cu in the inferred category. This resource estimate greatly exceeded management goals.
- In September 2022, BMR announced the approval by the Chilean Environment Assessment Service for the Environmental Impact Declaration (“DIA”) pertaining to mining at the company’s Cinabrio mine and San Andres deposit. The approval of the DIA allows BMR to move forward with starting mining operations in 2023 and restarting the mill at its Punitaqui copper mining complex soon after.
- BMR is focused on securing the final funding for the restart of mining and resumption of copper concentrate production at Punitaqui. Once this funding is received, BMR aims to complete mine rehabilitation and development in four to six months, with the ramp up from first production to the full production rate of 20-25 million pounds of copper in concentrate per annum to require a further four to six months.
“From exploration, engineering, community and permitting successes to realizing several non-dilutive means of funding to allow BMR to advance the project, our team looks forward to taking advantage of the renewed positive market sentiment for near term copper pricing and placing ourselves in a strong position to participate in a robust copper sector in 2023,” CEO Martin Kostuik stated in a news release.
Canada – Cobalt/Silver
Between 2016 and April 2018, BMR acquired through claim staking, option, joint venture and direct purchase the largest regional land holding in the historic home of high-grade cobalt-silver veins in Canada known locally as the Cobalt Embayment.
As of February 2023, BMR controlled a land package totaling 9 properties with 4,086 tenements that encompass an area of 84,003.39Ha. The key projects within the land package include McAra, Gowganda, Elk Lake, Fabre and Wilder. From 2017-2022, a total of 412 holes/51,452.34m were drilled on eight projects/20 targets. In addition, a total of 26,709 Line-Km of airborne geophysical surveys & 1,324.84sqkm of LiDAR topography was flown. Follow-up ground geophysical surveys resulted in a total of 37 surveys (514.64 Line-Km) being completed.
Initial NI 43-101 compliant resource defined at McAra (M&I Resource of 1,124,000lbs Co) was detailed in a Technical Report on Cobalt Exploration Assets in Canada dated as of February 5, 2021, with an effective date of October 31, 2020, prepared by SRK Consulting – G Cole PGeo (APGO#1416).
Idaho – Cobalt
BMR holds the Bonanza and East Fork properties located in the historic cobalt-copper-gold Blackbird mining district (Blackbird Mine from 1902-1963 produced 17Mt grading 0.7% Co, 1.4% Cu, and 1 g/t Au) located about 30 kilometers west of Salmon, Idaho. The Bonanza project is immediately adjacent to Jervois Global’s Idaho Cobalt Operations, the United States’ only operating primary cobalt mine. At Bonanza, there are seven mineralized sites within an area over three kilometers wide that extends along a gabbro dyke striking continuously for over six kilometers northward from Noranda’s historic Blackbird Cobalt/Copper mine. The showings on the project are Bonanza Copper Tunnels, Tinker’s Pride, Bonanza Copper #25, Indian Creek, Gray Copper, Blackrock #4 and Papoose #’s 1-4.
From 2018-2021, BMR’s Bonanza Exploration included 550 line-km of airborne magnetics and radiometrics followed up by surface exploration that included rock sampling, soil sampling, channel sampling of historic workings and 3.6km of time domain induced polarization geophysics.
The two properties cover 12 significant cobalt-copper prospects within the known mineralized zone. Both of the BMR Idaho cobalt belt properties host excellent high-grade discovery potential.
South Korea – Graphite
BMR has 100% ownership of the Guemam and Taehwa graphite exploration projects containing high-purity flake graphite deposits. Both assets are past-producing mines with existing local infrastructure and near-term production potential.
Nevada – Lithium
The company’s Amargosa lithium project is in the southern Basin & Range province and central Mojave Desert of Nevada. It is an early-stage exploration opportunity in a favorable region that hosts numerous lithium occurrences, including the Clayton Valley lithium deposit owned by Cypress Development Corp., as well as a major nearby lithium brine mine currently in production called the Silver Peak mine held by Albermarle Corp., one of the world’s largest lithium producers.
Market Opportunity
Near-term forecasts for the copper sector are extremely bullish, with stalwart Wall Street firms such as Goldman Sachs and Bank of America projecting record highs in the coming months. A combination of short-term supply deficits and long-term energy transition demand are expected to buck the downward pressures that have impacted copper prices in recent years.
Goldman in December 2022 forecast a 178K metric ton deficit in the copper market in 2023, causing the firm to raise its 12-month target to $11K/ton and its average price for calendar 2023 to $9,750/ton.
With China likely to continue accelerating efforts to restock depleted inventories in the wake of its COVID-19 reopening and a sustained push toward electrification around the globe placing a strain on supply, BMR is uniquely positioned to capitalize through the anticipated restart of operations at its Punitaqui copper mine.
Management Team
JMartin Kostuik is CEO and a Director of BMR. He brings to the company nearly three decades of diversified experience in the mining industry as a mining engineer and senior executive. Prior to joining BMR, Mr. Kostuik served as president and director of Arizona Gold Corporation and as CEO and director of Rupert Resources Limited. He built a broad base of experience in operations, engineering, exploration and capital projects with various companies including Luna Gold (Equinox), Barrick Gold Corporation, Taseko Mines Limited and DMC Mining Services. Mr. Kostuik earned his B.S. in Mining Engineering from Queen’s University and his M.B.A. from the University of Tennessee.
JMax Satel is the company’s CFO. He has over 18 years of experience as a successful natural resources-focused executive, most recently serving as EVP Corporate Development & Investor Relations for Arrow Exploration Corp., a TSX Venture- and AIM-listed oil & gas company with operations in Colombia and Canada. Prior to joining Arrow, Mr. Satel was principal and co-founder of Bordeaux Capital Inc., a Toronto-based advisory firm focused on the capital needs of companies across the natural resources sector, where he led and executed project financing advisory mandates involving global financial institutions and private equity funds. He earned a Bachelor of Commerce in Finance and Economics from the University of Toronto.
Jacob Willoughby is VP Corporate Development & Strategy for BMR. He brings to the company nearly 17 years of diversified experience in mining capital markets, including over eight years as a mining analyst covering exploration and development companies globally in both precious and base metals. Mr. Willoughby was most recently Vice President of Research and Analyst at Red Cloud Securities in Toronto. He spent two years as President and Director of Aldridge Minerals, a former Canadian based public exploration and development company with assets in Turkey and Papua New Guinea. Mr. Willoughby earned both a B.S. in Geology and a Masters in Business Administration from the University of Windsor.
FingerMotion Inc. (BTRMF), closed Monday's trading session at $0.141925, off by 3.4195%, on 150 volume. The average volume for the last 3 months is 150 and the stock's 52-week low/high is $0.0355/$1.05.
Recent News
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) to Attend CESCO; Announces Interest Settlement Agreements
- Battery Mineral Resources Corp. announces attendance at CESCO week in Santiago Chile and interest settlement transactions with holders of convertible debentures
- InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Reports Community, Social Initiatives; Grants PSUs
Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF)
The QualityStocks Daily Newsletter would like to spotlight Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF).
•Ucore just announced the selection of an 80,800 square-footbrownfield facility located in England Airpark in Alexandria,Louisiana, that will serve as the company’s LSMC REE separation andoxide production facility
•The company is confident that the establishment will expand itsmaximum production rate from the current 5,000 TPA to 7,500 TPA inTREO throughout by 2027
•The facility is expected to create 298 indirect jobs, for a totalof 398 new jobs in Central Louisiana. It is also set to bring thetotal potential value of state grants, tax incentives, 1payrollrebates, and other benefits to US$15 million
Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) is engaged in Rare Earth Element (REE) resource development and in commercializing its critical metals separation technology, RapidSX™, for the mining and metals extraction industry. The company is guided by principles of environmental, social and corporate governance (ESG) with a focus on disrupting China’s current dominance of the U.S. REE supply chain.
Ucore’s vision is to become a leading advanced technology company providing best-in-class metal separation products and services to the mining and mineral extraction industry. It plans to aid in the development, through strategic partnerships, of a North American REE supply chain controlled by the U.S. and its allies.
The company intends to contribute to this initiative through the near-term development of a heavy and light rare-earth processing facility in Louisiana and subsequent development of Strategic Metals Complexes (SMCs) in Alaska and Canada, as well as through the longer-term development of its 100%-owned Heavy Rare Earth Element (HREE) mineral resource property at Bokan Mountain on Prince of Wales Island, Alaska.
Ucore is headquartered in Halifax, Nova Scotia.
Projects & Technology
RapidSX™ Demonstration Plant
The Kingston, Ontario, RapidSX™ Demonstration Plant commissioning process is underway. Once commissioned, the plant is designed to demonstrate the commercial capabilities of the RapidSX technology platform.
The RapidSX demo plant will show:
- The techno-economic advantages of the RapidSX technology platform
- The processing of tens of tons of heavy and light mixed rare earth element concentrates in a simulated production environment
- The platform’s ability to operate for thousands of semi-continuous run-time hours
- Production of high-purity NdPr, praseodymium, neodymium, terbium and dysprosium rare earth elements for early OEM product qualification trials
The demo plant is located within Ucore’s 5,000-square-foot RapidSX Commercialization and Demonstration Facility and is run by its laboratory partner, Kingston Process Metallurgy Inc. (“KPM”).
RapidSX™ Technology
Innovation Metals Corp., acquired by the company in 2020, developed the RapidSX separation technology platform with early-stage assistance from the United States Department of Defense, later resulting in the production of commercial-grade, separated rare earth elements at pilot scale.
RapidSX combines the time-proven chemistry of conventional solvent extraction (SX) with a new column-based platform that significantly reduces time to completion and plant footprint, as well as potentially lowering capital and operating costs. SX is the international REE industry’s standard commercial separation technology and is currently used by all REE producers worldwide for bulk commercial separation of both heavy and light REEs.
Utilizing similar chemistry to conventional SX, RapidSX is not a “new” technology, but it represents a significant improvement on the well-established, well-understood, proven conventional SX separation technology preferred by REE producers.
Strategic Metals Complex
Ucore, engineering partner Mech-Chem Associates Inc. and KPM are developing the full-scale engineering for the company’s first Strategic Metals Complex (SMC). The SMC is a planned REE separation and rare earth oxide production plant slated to commence construction in Louisiana in 2023. It is scheduled to initially process 2,000 tons of total rare earth oxides by the end of 2024, increasing to 5,000 tons in 2026.
Bokan-Dotson Ridge REE Deposit
Ucore has invested over C$35 million to establish and validate the Bokan-Dotson Ridge resource in preparation for mine design and permitting. Initial drilling is complete, and a Preliminary Economic Assessment has been issued. Next steps for the project include a feasibility study, detailed mine design and permit acquisition. The project can be “near shovel ready” for construction in less than 30 months after receipt of the next stage of development funding.
Market Opportunity
According to a report by Grand View Research, the global rare earth elements market was valued at $2.8 billion in 2018 and is forecast to reach a value of $5.6 billion by 2025, achieving a CAGR of 10.4% during the period. Market growth is driven by increasing demand for these elements in the manufacturing of magnets and catalysts for the automotive industry. Rising demand for electric vehicles to reduce CO2 emissions is expected to propel the use of permanent magnets in the production of EV batteries.
China is the major producer and consumer of REEs. To maintain self-sufficiency and to meet future demand, China has been raising the export tariffs on rare earth elements shipped to various countries, including the U.S., Japan, India, Brazil and the European Union. This led to the current supply-demand gap in these countries, as they rely on imports from China.
China reduced the exports of REEs by 72% in the second half of 2010 to preserve its reserves of these elements and continues to export REEs at reduced levels, thereby affecting industries such as automotive, oil and gas, and electronics, which require an ample amount of rare earth elements.
Management Team
Pat Ryan, P.Eng., is Chairman and CEO of Ucore Rare Metals. He began as a director with the company when he developed a heightened interest in critical metals. Before joining Ucore, he founded and led a multimillion-dollar automotive OEM design and lean manufacturing company. His understanding of complex supply chains across international markets has led to a prime positioning as the global auto industry transitions to vehicle electrification. He holds a Bachelor of Engineering degree from Dalhousie University.
Peter Manuel is Vice President and CFO of Ucore. Prior to joining the company, he practiced as a Chartered Accountant for more than 17 years, providing consulting services to companies in a range of industries, with a focus on the financial services and resource sectors. He spent 10 years in England and Ireland providing assurance, strategic planning, corporate finance and other consulting services to a portfolio of both public and private entities. He holds a Bachelor of Commerce Degree from Dalhousie University.
Michael Schrider, MEng, P.E., is Vice President and COO of Ucore. He is a multidisciplinary engineer who has been involved in manufacturing, engineering and managing complex structural and mechanical systems projects since 1989. He was the Founder, President and Chief Engineer of Schrider & Associates and Alton Bay Design, both engineering services firms. He holds a bachelor’s degree in naval architecture and marine engineering from the University of New Orleans and a master’s degree in mining, geological and geophysical engineering from the University of Arizona.
Mark MacDonald is Vice President of Investor Relations at Ucore. He has over 25 years of experience implementing award winning business development and marketing programs at regional and national levels. As Vice President of Sales, he was responsible for Mediapro Communication’s growth as AT&T Canada’s leading B2B sales partner. He subsequently became Atlantic Regional Vice President of AT&T Canada Corp. He holds a Bachelor of Commerce degree from Dalhousie University.
Ucore Rare Metals Inc. (UURAF), closed Monday's trading session at $0.9858, off by 4.2913%, on 22,234 volume. The average volume for the last 3 months is 22,034 and the stock's 52-week low/high is $0.40/$1.15.
Recent News
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Anticipates Putting RapidSX(TM) to Work at Commercial Scale
- InvestorNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Announces Location for First REE Processing Plant
- MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Focused on Recreating Mid-Market REE Supply Chain in North America
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Closes on $3M Registered Direct Offering, Concurrent Private Placement
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Releases Shareholder Update on Major Programs, M&A Plans
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - State Data Shows Marijuana Sales in Missouri Totaled $126M in March
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) CEO to Attend ASU+GSV Summit
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Closes on $3M Registered Direct Offering
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Zinc Production to Recover in 2023 Though Scheduled Mine Closures Could Dampen Results
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Reports Strong Commercial Traction and Sales Momentum; Reports Fourth Quarter and Full Year 2022 Financial Results
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) to Attend CESCO; Announces Interest Settlement Agreements
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Biden Signs Measure to End National COVID-19 Emergency
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF), IMAC Execute Strategic 'Merger-of-Equals' LOI
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Copper and Nickel: Major Players of the Energy Transition
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - TechMediaBreaks - CISO Global Inc. (NASDAQ: CISO) Achieves Widely Recognized SOC 2 Certification
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Global Trial Targets Unmet Medical Need in Arresting GBM Brain Cancer
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Committed to Being ‘Responsible Beyond the Thread’
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Launches EMBARK OA, a Cost-Free Facilitator Training Program for Psychedelic Therapy
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Appoints Digital Health Product Leader as Chief Product Officer
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc.'s (ATDS) Award-Winning Ransomware Defense Shines as Cybercrime Persists
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Announces Q4 and Year-End 2022 Results
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Why Psychedelic Trips Cause Geometric Visual Effects
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Inks New Sales Orders, Anticipates More Sales from New 'Orientable' System
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Cartier Silver Corp. (CSE: CFE) Follows in Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) Footsteps Within Bolivia's Potosi Region
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Nuclear Industry Calls on EU to Gain Prominence in Net-Zero Push
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) Releases Q4, FY 2022 Financial Results, Quarterly Update
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Names Interim Chair as Executive Chair Steps Down
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Retires All Outstanding Institutional Convertible Debt
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Reschedules Special Meeting of Stockholders, Extends Deadline for Share Redemption
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Announces Results of Drill Hole at Flagship Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Planned 1-for-30 ADS Ratio Change
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Receives NASDAQ Notification, Has Regained Compliance
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Potential to Develop App Designed to Complete with TikTok on New Opportunities, Reduced Operating Expenses
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Eyes Successful Results of Its Preorder Strategy
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Seeking to Combine Power of Solar, Geothermal on Path to a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Provides Statement Regarding FDA Initiative to Accelerate Approval Process for Gene Therapies
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Moving Forward with Spin-Off of Subsidiary, Value Creation Strategy
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Poised to Offer Timely Solution amid a 'Smoldering Public Health Crisis'
- Cepton Inc. (NASDAQ: CPTN) - Lidar Pioneer Cepton, Inc. (NASDAQ: CPTN) Bridges Technological Gaps to Advance Mass Market ADAS Adoption
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Partners with Top Investment Banking Firm
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Engaged to Help Combat Manipulative Trading and Market Activities
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc.'s (NASDAQ: IDEX) Solectrac Announces Key Upgrades to Its Industry Leading e25 Electric Tractor
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - New Certification Will Help Standardize Marijuana Product Testing by Labs
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces $5M Registered Direct Offering, Private Placement
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Concludes Enrollment for Phase 2 Trial Studying INM-755 Cannabinol Cream
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces YE 2022 Results Confirming Right Path to Scale, Support Continued Growth
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Study Finds That Tinnitus, Hearing Loss Are Common in Cancer Survivors
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Contracted by NYPD to Patrol New York Subway
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Integrates ChatGPT into Shopify Powered Online e-Commerce Site
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Eyes the Licensed Use of its DehydraTECH(TM) Technology to Complement its Royalty Earnings and Realize Revenue in Advance of Royalties
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - MiningNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Eyes 'Exciting Results' from Exploration and Delineation Program at Los Azules Copper Project
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Filing of Prospectus Supplement, First Tranche Closing
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participating in EF Hutton Fireside Chat
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Announces Completion of Pilot Program at LAX; Accelerates Integration of New Battery Tech
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Adds Good Planet Foods to eCommerce Partner Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Reports Record Month in Q1 2023 Numbers
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Enters Non-Exclusive Discussions with HTA Regarding Potential Supply of Hydrogen Fuel
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Names Oncology Translational Research Leader to Scientific Advisory Board
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces $1.5M Order for Proprietary UltraShear Nanoemulsified CBD
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Releases FY 2022 Financials, Corporate Update
- Progressive Care Inc. (OTCQB: RXMD) - BioMedNewsBreaks - Progressive Care Inc. (RXMD) Identified as a High-Growth Opportunity
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) to Provide Clinical Trial Updates at Emerging Growth Conference
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Names New CFO, Corporate Secretary
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) Is 'One to Watch'
- REZYFi, Inc. - 420 with CNW — Voters in Oklahoma to Decide Whether to Legalize Recreational Marijuana
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Acquires Direct Ownership of 30,004,349 Common Shares of Plantify Foods Inc. (TSX.V: PTFY)
- Sharing Services Global Corporation (SHRG) - DSS Announces Letter to Shareholders
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) CEO Brian Haugli Discusses Government TikTok Ban in Forum Daily Interview
- Sigma Additive Solutions (NASDAQ: SASI) - Sigma Additive Solutions Reports Fourth Quarter and Full Year 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Oregon Psilocybin Company Silo Wellness to Resume Trading on Canadian Securities Exchange
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Receives Single Largest SD7 Handset Order to Date from Education Vertical
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) to Host Q4 and FY 2022 Results Call and Webcast
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation at Upcoming Echelon Partners Lithium Summit
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces the Televised Lineups for Shows 452, 457, and 458, Airings on Three Networks Will Begin Tonight, Thursday, April 13, 2023, at 9:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Releases Q1 2023 Financial Results
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaline88(R) Earns Distribution Throughout All Banners of Nation's Largest Grocer
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Regains Compliance with Nasdaq Listing Rule
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Announces Private Placement for Minimum of AUD$2,000,000
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Releases Q4, Year-End 2022 Financial and Corporate Report
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Anticipates Putting RapidSX(TM) to Work at Commercial Scale
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Announces Re-Election of Scott Melbye as UPA President for Third Term
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Expands Portfolio with New European Patent for AV-101
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Schedules Release of Q4 and FY 2022 Results, Conference Call
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap® to Host Investor Insights Webcast on March 23, 2023
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Closes on $3M Registered Direct Offering, Concurrent Private Placement
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Releases Shareholder Update on Major Programs, M&A Plans
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - State Data Shows Marijuana Sales in Missouri Totaled $126M in March
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) CEO to Attend ASU+GSV Summit
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Closes on $3M Registered Direct Offering
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Zinc Production to Recover in 2023 Though Scheduled Mine Closures Could Dampen Results
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Reports Strong Commercial Traction and Sales Momentum; Reports Fourth Quarter and Full Year 2022 Financial Results
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) to Attend CESCO; Announces Interest Settlement Agreements
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Biden Signs Measure to End National COVID-19 Emergency
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF), IMAC Execute Strategic 'Merger-of-Equals' LOI
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Copper and Nickel: Major Players of the Energy Transition
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - TechMediaBreaks - CISO Global Inc. (NASDAQ: CISO) Achieves Widely Recognized SOC 2 Certification
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Global Trial Targets Unmet Medical Need in Arresting GBM Brain Cancer
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Committed to Being ‘Responsible Beyond the Thread’
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Launches EMBARK OA, a Cost-Free Facilitator Training Program for Psychedelic Therapy
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Appoints Digital Health Product Leader as Chief Product Officer
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc.'s (ATDS) Award-Winning Ransomware Defense Shines as Cybercrime Persists
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Announces Q4 and Year-End 2022 Results
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Why Psychedelic Trips Cause Geometric Visual Effects
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Inks New Sales Orders, Anticipates More Sales from New 'Orientable' System
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Cartier Silver Corp. (CSE: CFE) Follows in Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) Footsteps Within Bolivia's Potosi Region
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Nuclear Industry Calls on EU to Gain Prominence in Net-Zero Push
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) Releases Q4, FY 2022 Financial Results, Quarterly Update
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Names Interim Chair as Executive Chair Steps Down
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Retires All Outstanding Institutional Convertible Debt
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Reschedules Special Meeting of Stockholders, Extends Deadline for Share Redemption
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Announces Results of Drill Hole at Flagship Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Planned 1-for-30 ADS Ratio Change
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Receives NASDAQ Notification, Has Regained Compliance
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Potential to Develop App Designed to Complete with TikTok on New Opportunities, Reduced Operating Expenses
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Eyes Successful Results of Its Preorder Strategy
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Seeking to Combine Power of Solar, Geothermal on Path to a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Provides Statement Regarding FDA Initiative to Accelerate Approval Process for Gene Therapies
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Moving Forward with Spin-Off of Subsidiary, Value Creation Strategy
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Poised to Offer Timely Solution amid a 'Smoldering Public Health Crisis'
- Cepton Inc. (NASDAQ: CPTN) - Lidar Pioneer Cepton, Inc. (NASDAQ: CPTN) Bridges Technological Gaps to Advance Mass Market ADAS Adoption
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Partners with Top Investment Banking Firm
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Engaged to Help Combat Manipulative Trading and Market Activities
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc.'s (NASDAQ: IDEX) Solectrac Announces Key Upgrades to Its Industry Leading e25 Electric Tractor
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - New Certification Will Help Standardize Marijuana Product Testing by Labs
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces $5M Registered Direct Offering, Private Placement
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Concludes Enrollment for Phase 2 Trial Studying INM-755 Cannabinol Cream
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces YE 2022 Results Confirming Right Path to Scale, Support Continued Growth
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Study Finds That Tinnitus, Hearing Loss Are Common in Cancer Survivors
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Contracted by NYPD to Patrol New York Subway
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Integrates ChatGPT into Shopify Powered Online e-Commerce Site
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Eyes the Licensed Use of its DehydraTECH(TM) Technology to Complement its Royalty Earnings and Realize Revenue in Advance of Royalties
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - MiningNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Eyes 'Exciting Results' from Exploration and Delineation Program at Los Azules Copper Project
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Filing of Prospectus Supplement, First Tranche Closing
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participating in EF Hutton Fireside Chat
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Announces Completion of Pilot Program at LAX; Accelerates Integration of New Battery Tech
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Adds Good Planet Foods to eCommerce Partner Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Reports Record Month in Q1 2023 Numbers
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Enters Non-Exclusive Discussions with HTA Regarding Potential Supply of Hydrogen Fuel
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Names Oncology Translational Research Leader to Scientific Advisory Board
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces $1.5M Order for Proprietary UltraShear Nanoemulsified CBD
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Releases FY 2022 Financials, Corporate Update
- Progressive Care Inc. (OTCQB: RXMD) - BioMedNewsBreaks - Progressive Care Inc. (RXMD) Identified as a High-Growth Opportunity
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) to Provide Clinical Trial Updates at Emerging Growth Conference
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Names New CFO, Corporate Secretary
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) Is 'One to Watch'
- REZYFi, Inc. - 420 with CNW — Voters in Oklahoma to Decide Whether to Legalize Recreational Marijuana
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Acquires Direct Ownership of 30,004,349 Common Shares of Plantify Foods Inc. (TSX.V: PTFY)
- Sharing Services Global Corporation (SHRG) - DSS Announces Letter to Shareholders
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) CEO Brian Haugli Discusses Government TikTok Ban in Forum Daily Interview
- Sigma Additive Solutions (NASDAQ: SASI) - Sigma Additive Solutions Reports Fourth Quarter and Full Year 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Oregon Psilocybin Company Silo Wellness to Resume Trading on Canadian Securities Exchange
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Receives Single Largest SD7 Handset Order to Date from Education Vertical
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) to Host Q4 and FY 2022 Results Call and Webcast
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation at Upcoming Echelon Partners Lithium Summit
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces the Televised Lineups for Shows 452, 457, and 458, Airings on Three Networks Will Begin Tonight, Thursday, April 13, 2023, at 9:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Releases Q1 2023 Financial Results
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaline88(R) Earns Distribution Throughout All Banners of Nation's Largest Grocer
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Regains Compliance with Nasdaq Listing Rule
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Announces Private Placement for Minimum of AUD$2,000,000
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Releases Q4, Year-End 2022 Financial and Corporate Report
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Anticipates Putting RapidSX(TM) to Work at Commercial Scale
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Announces Re-Election of Scott Melbye as UPA President for Third Term
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Expands Portfolio with New European Patent for AV-101
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Schedules Release of Q4 and FY 2022 Results, Conference Call
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap® to Host Investor Insights Webcast on March 23, 2023
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Closes on $3M Registered Direct Offering, Concurrent Private Placement
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Releases Shareholder Update on Major Programs, M&A Plans
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - State Data Shows Marijuana Sales in Missouri Totaled $126M in March
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) CEO to Attend ASU+GSV Summit
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - InvestorNewsBreaks - AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) Closes on $3M Registered Direct Offering
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Zinc Production to Recover in 2023 Though Scheduled Mine Closures Could Dampen Results
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Reports Strong Commercial Traction and Sales Momentum; Reports Fourth Quarter and Full Year 2022 Financial Results
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) to Attend CESCO; Announces Interest Settlement Agreements
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- BiondVax Pharmaceuticals Ltd. (NASDAQ: BVXV) - Biden Signs Measure to End National COVID-19 Emergency
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Releases Financial Report for Q2, First Half of 2022
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF), IMAC Execute Strategic 'Merger-of-Equals' LOI
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - Copper and Nickel: Major Players of the Energy Transition
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - DC Mayor Enacts Measure Allowing Patients to Self-Certify as Medical Cannabis Patients
- CISO Global, Inc. (NASDAQ: CISO) - TechMediaBreaks - CISO Global Inc. (NASDAQ: CISO) Achieves Widely Recognized SOC 2 Certification
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Global Trial Targets Unmet Medical Need in Arresting GBM Brain Cancer
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - GreenEnergyBreaks - Correlate Infrastructure Partners Inc. (CIPI) Deploying Clean Energy Generation and Energy Efficiency Solutions at Scale
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Committed to Being ‘Responsible Beyond the Thread’
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) Launches EMBARK OA, a Cost-Free Facilitator Training Program for Psychedelic Therapy
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Appoints Digital Health Product Leader as Chief Product Officer
- Data443 Risk Mitigation Inc. (OTC: ATDS) - Data443 Risk Mitigation Inc.'s (ATDS) Award-Winning Ransomware Defense Shines as Cybercrime Persists
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Announces Q4 and Year-End 2022 Results
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - Why Psychedelic Trips Cause Geometric Visual Effects
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Inks New Sales Orders, Anticipates More Sales from New 'Orientable' System
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Welcomes Plant-based Industry Pioneer and former Director of Beyond Meat to Advisory Board Amid Ramp-up of Healthful Food Innovations, Distribution
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Cartier Silver Corp. (CSE: CFE) Follows in Eloro Resources Ltd.'s (TSX: ELO) (OTCQX: ELRRF) Footsteps Within Bolivia's Potosi Region
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - Nuclear Industry Calls on EU to Gain Prominence in Net-Zero Push
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) Releases Q4, FY 2022 Financial Results, Quarterly Update
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Names Interim Chair as Executive Chair Steps Down
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Retires All Outstanding Institutional Convertible Debt
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Reschedules Special Meeting of Stockholders, Extends Deadline for Share Redemption
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFD) Announces Results of Drill Hole at Flagship Property
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Announces Planned 1-for-30 ADS Ratio Change
- Freight Technologies Inc. (NASDAQ: FRGT) - InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Receives NASDAQ Notification, Has Regained Compliance
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Potential to Develop App Designed to Complete with TikTok on New Opportunities, Reduced Operating Expenses
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Eyes Successful Results of Its Preorder Strategy
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Seeking to Combine Power of Solar, Geothermal on Path to a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - Golden Triangle Ventures, Inc. Announces End of Year Corporate Update Letter
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Provides Statement Regarding FDA Initiative to Accelerate Approval Process for Gene Therapies
- RYVYL Inc. (NASDAQ: GBOX) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Moving Forward with Spin-Off of Subsidiary, Value Creation Strategy
- HeartBeam Inc. (NASDAQ: BEAT) - BioMedNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Poised to Offer Timely Solution amid a 'Smoldering Public Health Crisis'
- Cepton Inc. (NASDAQ: CPTN) - Lidar Pioneer Cepton, Inc. (NASDAQ: CPTN) Bridges Technological Gaps to Advance Mass Market ADAS Adoption
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) Partners with Top Investment Banking Firm
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Engaged to Help Combat Manipulative Trading and Market Activities
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Engages Financial Advisor, Legal Counsel for Proposal Evaluation
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc.'s (NASDAQ: IDEX) Solectrac Announces Key Upgrades to Its Industry Leading e25 Electric Tractor
- IGC Pharma Inc. (NYSE American: IGC) - 420 with CNW - New Certification Will Help Standardize Marijuana Product Testing by Labs
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd. (NASDAQ: IFBD) Announces $5M Registered Direct Offering, Private Placement
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Concludes Enrollment for Phase 2 Trial Studying INM-755 Cannabinol Cream
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Announces YE 2022 Results Confirming Right Path to Scale, Support Continued Growth
- Jupiter Wellness Inc. (NASDAQ: JUPW) - Study Finds That Tinnitus, Hearing Loss Are Common in Cancer Survivors
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Contracted by NYPD to Patrol New York Subway
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Integrates ChatGPT into Shopify Powered Online e-Commerce Site
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Eyes the Licensed Use of its DehydraTECH(TM) Technology to Complement its Royalty Earnings and Realize Revenue in Advance of Royalties
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Marijuana Company of America Inc. (OTC: MCOA) - InvestorNewsBreaks - Marijuana Company of America Inc. (MCOA) Releases Q3 2022 Financial Report
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - MiningNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Eyes 'Exciting Results' from Exploration and Delineation Program at Los Azules Copper Project
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Second Quarter of Fiscal 2023
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Announces Filing of Prospectus Supplement, First Tranche Closing
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Participating in EF Hutton Fireside Chat
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Announces Completion of Pilot Program at LAX; Accelerates Integration of New Battery Tech
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXD) Adds Good Planet Foods to eCommerce Partner Roster
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Reports Record Month in Q1 2023 Numbers
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Enters Non-Exclusive Discussions with HTA Regarding Potential Supply of Hydrogen Fuel
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Names Oncology Translational Research Leader to Scientific Advisory Board
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Announces $1.5M Order for Proprietary UltraShear Nanoemulsified CBD
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Releases FY 2022 Financials, Corporate Update
- Progressive Care Inc. (OTCQB: RXMD) - BioMedNewsBreaks - Progressive Care Inc. (RXMD) Identified as a High-Growth Opportunity
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) to Provide Clinical Trial Updates at Emerging Growth Conference
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Names New CFO, Corporate Secretary
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) Is 'One to Watch'
- REZYFi, Inc. - 420 with CNW — Voters in Oklahoma to Decide Whether to Legalize Recreational Marijuana
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corp. (SNWR) Subsidiary, Leading Global Music Video Network Ink Partnership Agreement
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) (FSE: 80W) Acquires Direct Ownership of 30,004,349 Common Shares of Plantify Foods Inc. (TSX.V: PTFY)
- Sharing Services Global Corporation (SHRG) - DSS Announces Letter to Shareholders
- SideChannel Inc. (OTCQB: SDCH) - SideChannel Inc. (SDCH) CEO Brian Haugli Discusses Government TikTok Ban in Forum Daily Interview
- Sigma Additive Solutions (NASDAQ: SASI) - Sigma Additive Solutions Reports Fourth Quarter and Full Year 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Oregon Psilocybin Company Silo Wellness to Resume Trading on Canadian Securities Exchange
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) (TSX.V: SIM) Receives Single Largest SD7 Handset Order to Date from Education Vertical
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) to Host Q4 and FY 2022 Results Call and Webcast
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Announces Participation at Upcoming Echelon Partners Lithium Summit
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Names New Board, Audit Committee Member
- Sustainable Green Team Ltd. (OTC: SGTM) - New to The Street Announces the Televised Lineups for Shows 452, 457, and 458, Airings on Three Networks Will Begin Tonight, Thursday, April 13, 2023, at 9:30 PM ET
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT Global Alternatives Inc. (CSE: TAAT) (OTCQX: TOBAF) Releases Q1 2023 Financial Results
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc.'s (NASDAQ: WTER) (CSE: WTER) Alkaline88(R) Earns Distribution Throughout All Banners of Nation's Largest Grocer
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Regains Compliance with Nasdaq Listing Rule
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Announces Private Placement for Minimum of AUD$2,000,000
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Releases Q4, Year-End 2022 Financial and Corporate Report
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Anticipates Putting RapidSX(TM) to Work at Commercial Scale
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Announces Re-Election of Scott Melbye as UPA President for Third Term
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Expands Portfolio with New European Patent for AV-101
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Schedules Release of Q4 and FY 2022 Results, Conference Call
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap® to Host Investor Insights Webcast on March 23, 2023
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































