The QualityStocks Daily Stock List
- TDH Holdings (PETZ)
- NewAge (NBEV)
- TMC The Metals Company (TMC)
- Biolife Sciences (BLFE)
- Nutranomics Inc. (NNRX)
- Danimer Scientific (DNMR)
- Alpha Teknova (TKNO)
- Okyo Pharma (OKYO)
- Qilian International (QLI)
- Retractable Technologies (RVP)
- ElectraMeccanica Vehicles Corp. Ltd. (SOLO)
- Freeport-McMoRan Inc. (FCX)
TDH Holdings (PETZ)
MarketClub Analysis, QualityStocks, StockMarketWatch, TradersPro, BUYINS.NET, PoliticsAndMyPortfolio, InvestorPlace, TopPennyStockMovers and The Online Investor reported earlier on TDH Holdings (PETZ), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
TDH Holdings Inc. (NASDAQ: PETZ) is a holding firm that is engaged in the development, manufacture and sale of high quality pet food products for pet owners in North America, Europe, Asia and China.
The firm has its headquarters in Qingdao, the People’s Republic of China and was incorporated in April 2002 by Rong Feng Cui. It operates in the consumer staples sector, under the food sub-industry and serves consumers across the globe.
The enterprise provides 6 product lines, i.e. baked pet biscuits; dental health snack foods; wet canned pet foods including jerkies, strips and fillets made of lamb, pork, duck, chicken, etc.; dried pet snacks including jerkies, strips and fillets made of lamb, pork, duck, chicken, etc.; pet chew products which include rawhide, different bones and similar products; and other pet foods. The enterprise’s wet foods are mainly suited for cats while dry food can be eaten by both cats and dogs. Its products come in various forms which include tubs, strips, serve rolls and slice rolls. The enterprise’s products are sold under the following brands; Dog Zone Sasami, Tiandihui, TDH, Like, Hum and Cheer and Pet Cuisine, among others.
In addition to this, the company also sells pet toys, dog leashes, vegetarian pet food, fish pet food and dentifrice products. It provides its products via an e-commerce platform as well as through wholesalers and retailers.
The firm is focused on improving its profitability and is also working on identifying acquisition opportunities which will be useful in helping the company grow, while also bringing in more investments.
TDH Holdings (PETZ), closed Monday's trading session at $4.81, up 29.3011%, on 17,569,438 volume. The average volume for the last 3 months is 17.569M and the stock's 52-week low/high is $1.70/$188.00.
NewAge (NBEV)
StocksEarning, MarketClub Analysis, Schaeffer's, MarketBeat, StockMarketWatch, StreetInsider, QualityStocks, InvestorPlace, The Street, BUYINS.NET, TraderPower, PoliticsAndMyPortfolio, TopPennyStockMovers, Promotion Stock Secrets, StockEarnings, SmallCapVoice, TradersPro, TheTradingReport, Market Intelligence Center Alert, Jason Bond, InvestmentHouse and Inside Trading reported earlier on NewAge (NBEV), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
NewAge Inc. (NASDAQ: NBEV) (FRA: N1K) is an organic and healthy products firm that is engaged in the development, marketing, sale and distribution of healthy products.
The firm has its headquarters in Denver, Colorado and was incorporated in 2010, on April 26th by Neil Fallon. Prior to its name change in July 2020, the firm was known as New Age Beverages Corporation. It serves consumers in China, Japan, the United States and internationally.
The company operates through the Direct Store, New Age and the Direct/Social selling segments. The social selling segment is engaged in developing, manufacturing and marketing healthy products in three major category platforms, namely nutritional performance, healthy appearance and health and wellness. On the other hand, the New Age segment is involved in the marketing and sale of various healthy beverage brands like Volvi, Nestea, Evian, Bucha Live Kombucha and XingTea. The company provides its products under the MaVie, Puritii, Reviive, Lucim, TeMana, Hiro Natural, LIMU Blue Frog, Zennoa, LIMU and Tahitian Noni brands. Its products are sold mainly via a direct route to market and e-commerce.
The enterprise offers personal care, water and air filtration, snacks, beverages, cosmetics, anti-aging and essential oil skincare, energy drink and health and wellness products; CBD products; diagnostic products like DNA testing and diagnostic kits; and slenderize, nutraceutical, nutritional supplement and weight management products.
The firm recently entered into a partnership agreement with Verb Technology Inc. to launch its sales app. This move will not only boost the company’s sales and bring in more revenue but also extend its consumer reach, which will have a positive effect on its investments and growth.
NewAge (NBEV), closed Monday's trading session at $0.3632, up 74.1966%, on 166,944,642 volume. The average volume for the last 3 months is 153.335M and the stock's 52-week low/high is $0.196/$1.80.
TMC The Metals Company (TMC)
StocksEarning, MarketBeat, QualityStocks, Trades Of The Day, The Street, Schaeffer's and MarketClub Analysis reported earlier on TMC The Metals Company (TMC), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
TMC The Metals Company Inc. (NASDAQ: TMC) is an electric car battery metal firm that is engaged in the collection, processing and refining of polymetallic nodules.
The firm has its headquarters in Vancouver, Canada. Prior to its name change, the firm was known as Sustainable Opportunities Acquisition Corp. It operates as part of the metals and mining industry and serves consumers around the globe.
The company is focused on recovering metals from nodules in order to support demand from electric car and battery production. Its collection of polymetallic nodules decreases the social and environmental impact of primary metal production, which it hopes could lead to a significant reduction of or total elimination of tailings.
The enterprise’s polymetallic nodules are obtained from the Clarion Clipperton Zone seafloor. This zone is found in the eastern part of the Pacific Ocean and is made up of abyssal plains and other formations. It occupies about 4500 miles and is known to not only contain a diversity and abundance of life but also manganese nodule resources. The enterprise holds commercial and exploration rights to 3 contract areas which host nodules that contain manganese, cobalt sulfate, copper and nickel sulfate. It produces these metals to power electric cars and store clean energy.
The company is well-positioned to not only grow but also bring in huge returns for its shareholders given the expectation that the demand for battery metals will continue to increase as more countries and people adopt the use of electric cars in order to reduce emissions. The adoption of electric vehicles will not only bring in additional revenue into the firm but also encourage more investments, which will boost its growth.
TMC The Metals Company (TMC), closed Monday's trading session at $1.12, up 33.3333%, on 17,687,390 volume. The average volume for the last 3 months is 17.687M and the stock's 52-week low/high is $0.790101/$15.39.
Biolife Sciences (BLFE)
QualityStocks, OTCtipReporter, PennyStockProphet, PennyStockScholar and AllPennyStocks reported earlier on Biolife Sciences (BLFE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Biolife Sciences Inc. (OTC: BLFE) operates as a developer and licensor of technology for various sectors, including the food and beverage, material science, beauty and healthcare sectors.
The firm has its headquarters in Mississauga, Canada and was incorporated in 2000, on March 31st. It serves consumers around the globe, with a focus on Canada.
At its core though, the company operates as a biotech firm that is focused on the development, licensing and distribution of touchless vending products, non-contact human temperature screening technology and antibacterial products. It is focused on innovative antimicrobial solutions which protect against pathogens, bacteria, viruses and fungus. The company specializes in moving innovative products from small scale production or the laboratory into wider market adoption.
The enterprise’s solutions include next-generation vending machines, ultraviolet sterilization, non-contact human temperature screening and antimicrobial copper air filters. Its non-contact temperature screening technology detects the body temperature of a subject. Its copper air filter captures and kills bacterial that is passed through the air vents, then releases clean air that has copper ion deposits. On the other hand, its touchless vending marketplaces offer digital vending machines and digital advertising, serving a range of antimicrobial products and personal protective equipment.
Projections show that the global temperature sensor market size will grow to almost $9 billion by the year 2027. This means that the firm is well-positioned to not only grow significantly but also occupy a larger market share. This will be good for the firm’s revenues as well as investments into the firm.
Biolife Sciences (BLFE), closed Monday's trading session at $0.055605, up 32.7088%, on 2,051,045 volume. The average volume for the last 3 months is 2.051M and the stock's 52-week low/high is $0.0365/$2.54.
Nutranomics Inc. (NNRX)
QualityStocks, PennyStocks24, Pumps and Dumps, PennyStocks Forever, PennyOmega, PennyToBuck, Real Pennies, StockHotTips, Stock Stars, BestOtc, MicrocapAlliance, Global Equity Alert, DrStockPick, CRWEWallStreet, CRWEPicks, CRWEFinance and MonsterStocksPicks reported earlier on Nutranomics Inc. (NNRX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Nutranomics Inc. (OTC: NNRX) is focused on researching, developing and selling nutritional food products.
The firm has its headquarters in Salt Lake City, Utah and was incorporated in 1995 by Tracy K. Gibbs. The firm serves consumers around the globe.
The company is party to a strategic partnership agreement with Terra-Formance LLC. Its aim is to expand its intellectual property to include a pre-digestive delivery system poised to revolutionize the way individuals ingest supplements.
The enterprise provides about 480 nutritional supplements, which include probiotic supplements, herbal supplements, mineral supplements and formulating vitamin supplements. These nutritional products include Mobility and Flexibility Complex, Pre Pro-Biotic, PhytoNutrient, Immune Modulating Complex, South American Herbal Blend, Glucozyme, All Natural Vitamin C Complex, The Works, Total Body Detox and Viral Guard. It also offers the Assimilation Enhancing System, a patented system which improves the absorption of nutrients in the body; and a tool that examines a consumer’s blood cells in real time, live, on a video monitor dubbed Nutritional Blood Analysis. In addition to this, the enterprise provides educational services, which include different types of trainings, certifications, analysis and group presentations to industry professionals and individuals. It distributes its products through retail stores, as well as through sales representatives throughout Asia and North America.
The firm plans to acquire DHS Development Inc., which will focus on the production of its core products like tablets, capsules and gummies, among others. In addition to producing top quality products for its consumers, this move will allow the firm to improve its production facility, which will have a positive effect on its efficiency and product quality.
Nutranomics Inc. (NNRX), closed Monday's trading session at $0.0003, up 50%, on 508,394,342 volume. The average volume for the last 3 months is 508.394M and the stock's 52-week low/high is $0.0001/$0.00111.
Danimer Scientific (DNMR)
Schaeffer's, Kiplinger Today, MarketClub Analysis and MarketBeat reported earlier on Danimer Scientific (DNMR), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Danimer Scientific Inc. (NYSE: DNMR) (FR: 6CU) (BMV: DNMR) is a performance polymer firm that is focused on the development and production of bioplastic replacements for traditional plastics that are petroleum-based.
The firm has its headquarters in Bainbridge, Georgia and was incorporated in 2014, on January 13th. It operates as part of the specialty chemicals industry, under the basic materials sector. The firm serves consumers in the United States.
The company’s objective is to provide individuals and communities at large with environmentally friendly and sustainable products which are biodegradable and compostable. This means that they easily decompose, without leaving a harmful footprint or a trace of particulate matter. It holds over 400 patents and pending patent applications for various biopolymer formulations and manufacturing processes.
The enterprise produces a biodegradable plastic feedstock alternative known as polyhydroxyalkanoate, which is used in various plastic applications, including food containers, straws, films and other things; and polylactic acid-based resins, which are used to coat disposable paper cups. It provides its products for biopolymers, including thermoforming, filaments, fibers, aqueous coatings, additives and injection-molded articles. The enterprise also markets its products to manufacturers and converters in the plastics industry, as well as consumer packaging brand owners.
The firm recently announced its latest financial results, which show increases in its revenues. The CEO noted that its business development efforts had progressed significantly and they remain focused on capturing long-term opportunities to transform the plastics market. This will bring in additional revenues and investments into the firm and create shareholder value.
Danimer Scientific (DNMR), closed Monday's trading session at $4.29, off by 2.5%, on 942,368 volume. The average volume for the last 3 months is 938,690 and the stock's 52-week low/high is $3.0934/$21.38.
Alpha Teknova (TKNO)
MarketBeat reported earlier on Alpha Teknova (TKNO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Alpha Teknova Inc. (NASDAQ: TKNO) is a biotechnology firm that is engaged in the provision of critical reagents for the global life sciences market.
The firm has its headquarters in Hollister, California and was incorporated in 1996 by Thomas E. Davis. It operates as part of the drug manufacturers-specialty and generic industry, under the healthcare sector. The firm serves consumers around the world, with a focus on those in the United States.
The company operates through the clinical solutions and laboratory essentials business lines. Its processes have been designed to handle various customer-requested inputs, which vary by quality specifications, chemical formulation, volume, transportation requirements and types of containers. This enables the use of these products across the scope of the life sciences market.
The enterprise’s reagents enable the development and production of bio-pharmaceutical products like molecular diagnostics, new vaccines and drug therapies. It provides pre-poured media plates for cloning and cell growth; molecular biology reagents for sample re-suspension, manipulation and purification; and liquid cell culture supplements and media for cellular expansion. The enterprise’s clients include academic and government research institutions, biotechnology and pharmaceutical firms, in vitro diagnostic franchises, as well as contract development and manufacturing companies.
The firm recently released financial and operational results, which show increases in its revenues and significant progress in the construction of its new manufacturing facility. Its CEO noted that they had also observed healthy demand across its customer base, noting that they remained focused on improving its position in the market and taking advantage of new growth opportunities.
Alpha Teknova (TKNO), closed Monday's trading session at $4.77, up 1.0593%, on 50,797 volume. The average volume for the last 3 months is 50,463 and the stock's 52-week low/high is $4.23/$28.64.
Okyo Pharma (OKYO)
We reported earlier on Okyo Pharma (OKYO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Okyo Pharma Ltd (NASDAQ: OKYO) (OTCQB: EMMLF) (LON: OKYO) (FRA: OK10) is a pre-clinical bio-pharmaceutical firm that is focused on the development of therapies for ocular pain and inflammatory eye ailments.
The firm has its headquarters in London, the United Kingdom and was incorporated in 2007, on July 4th. It operates as part of the biotechnology industry, under the healthcare sector. The firm serves consumers around the globe.
The company’s objective is to improve the lives of patients suffering from ocular pain and inflammatory eye illnesses by developing effective treatments and cures. It does so by targeting GPCRs (G protein-coupled receptors). The company operates through its Okyo Pharma U.S. Inc. subsidiary.
The enterprise’s focus is primarily on conditions like allergic conjunctivitis, non-infectious anterior uveitis, ocular pain and dry eye disease. Its product pipeline consists of a formulation dubbed OK-101, which comprises of an anchoring lipid domain, a linker component and a 10-mer C-terminal chemerin peptide sequence. This candidate has been developed to treat dry eye disease by targeting an ocular receptor which controls inflammation and ocular pain. It also develops a lipidated-peptide, bovine adrenal medulla preclinical analogue candidate known as OK-201, to help treat neuropathic ocular pain.
The firm remains focused on advancing its ongoing Investigational New Drug application for its OK-101 candidate. The success and approval of this candidate for the treatment of dry eye will not only benefit patients with this indication but also bring in significant revenues and investments into the firm.
Okyo Pharma (OKYO), closed Monday's trading session at $2.4361, up 22.4171%, on 84,050 volume. The average volume for the last 3 months is 84,039 and the stock's 52-week low/high is $1.81/$6.11.
Qilian International (QLI)
MarketBeat reported earlier on Qilian International (QLI), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Qilian International Holding Group Limited (NASDAQ: QLI) is a pharmaceutical and chemical firm that is focused on conducting research on, developing and producing traditional Chinese medicine derivatives, active pharmaceutical ingredients and other by-products.
The firm has its headquarters in Jiuquan, China and was incorporated in 2019, on February 7th. It operates as part of the drug manufacturers-specialty and generic industry, under the healthcare sector. The firm serves consumers in the People’s Republic of China.
The enterprise’s products include a primary ingredient used in the production of compound licorice oral solutions known as Qilian Shan licorice liquid extract; an ingredient used to manufacture traditional licorice tablets by pharmaceutical firms dubbed Qilian Shan Licorice Extract; and an antitussive and expectorant medicine known as Gan Di Xin. It also offers Qilian Shan oxytetracycline active pharmaceutical ingredients for pharmaceutical firms to manufacture drugs; and Qilian Shan oxytetracycline tablets to treat and prevent various illnesses in humans as well as swine, cattle, turkeys and chickens. The enterprise also provides heparin products, including heparin sodium preparations used to produce drugs for hemodialysis, cerebrovascular illnesses and cardiovascular ailments; as well as traditional Chinese medicine derivatives, including Ahan antibacterial paste for the treatment of refractory chronic skin ailments.
The company recently announced its latest financial results, with its CEO noting that they remained focused on expanding the company’s production capacity and fueling its growth. This will bring in additional revenues and investments into the company while also helping create value for its shareholders.
Qilian International (QLI), closed Monday's trading session at $1.28, up 7.563%, on 962 volume. The average volume for the last 3 months is 874 and the stock's 52-week low/high is $1.01/$4.19.
Retractable Technologies (RVP)
TradersPro, Wall Street Resources, StockMarketWatch, Kiplinger Today, SmallCapVoice, MarketClub Analysis, MarketBeat, Zacks, The Street, StreetInsider, StreetAuthority Daily, StockOodles, SmallCapInvestor.com, QualityStocks and Marketbeat.com reported earlier on Retractable Technologies (RVP), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Retractable Technologies Inc. (NYSE American: RVP) is a company focused on designing, developing, manufacturing and marketing safety medical products like safety syringes for healthcare markets globally.
The firm has its headquarters in Little Elm, Texas and was incorporated in 1994, on May 9th by Thomas J. Shaw. It operates as part of the medical instruments and supplies industry, under the healthcare sector. The firm serves consumers in the United States and the rest of South and North America, as well as other regions across the globe.
The company is focused on becoming a catalyst in the reduction of the spread of infectious illnesses globally through education, innovation and the development of reliable and safe medical devices.
The enterprise’s products include 0.5mL, 1mL, 2mL, 3mL, 5mL, and 10mL syringes; VanishPoint insulin syringes; 1mL allergy antigen, insulin and tuberculin syringes; allergy trays; the VanishPoint Blood Collection Set; blood collection tube holders; small diameter tube adapters; the Patient Safe Luer cap; EasyPoint needles as well as standard 3mL syringes which come with EasyPoint needles; catheters which are IV safe; and VanishPoint autodisable syringes, which are sold in the global markets. It distributes its products via specialty distributors and general line, as well as a direct marketing network and global distributors.
The company, which recently released its latest financial results, remains focused on improving its revenues both domestically as well as at a global scale. This will help create shareholder value and bolster the company’s overall growth.
Retractable Technologies (RVP), closed Monday's trading session at $2.44, off by 4.3137%, on 153,236 volume. The average volume for the last 3 months is 153,065 and the stock's 52-week low/high is $2.29/$13.91.
ElectraMeccanica Vehicles Corp. Ltd. (SOLO)
Green Car Stocks, InvestorPlace, QualityStocks, StocksEarning, Kiplinger Today, Schaeffer's, MarketClub Analysis, StockMarketWatch, TradersPro, BUYINS.NET, The Street, MarketBeat, Trades Of The Day, TopPennyStockMovers, Daily Trade Alert, SmallCapVoice, Small Cap Firm, VectorVest, Eagle Financial Publications, Cabot Wealth and PoliticsAndMyPortfolio reported earlier on ElectraMeccanica Vehicles Corp. Ltd. (SOLO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Although they are slated to replace internal combustion engine cars over the next few decades, electric vehicles still have to overcome a few challenges on their way to the top of the automotive space. One major challenge so far has been charging. While you can fill up a conventional gas-powered car with fuel and be on your way within minutes, it can take an hour or longer to recharge an electric vehicle.
If you own a Tesla or have access to a supercharger network, you could bring this time down to around half an hour or less. Even though this is faster than most electric vehicle drivers can recharge, it is still a long way from the speed and convenience of pulling into a gas station and filling up a gas-powered car. As such, car and battery makers are working to develop charging technology that is fast and reliable while being safe for EV battery health.
Researchers have now revealed that a slight tweak in the way EV owners charge their cars could allow them to fill up their batteries from empty in only 10 minutes. According to Gil Tal from the University of California, the standard charging protocol that usually fills up an EV in around half an hour via supercharging tends to start at low power before ramping up and then reducing power at around 60% to 70%.
This is meant to prevent the battery from experiencing unnecessary stress and wearing down faster. Experts have been trying to ramp up charging speeds by optimizing the charging curve to give the battery more time with high power, Tal said. Researchers from the Idaho National Laboratory managed to do this by leveraging artificial intelligence algorithms to analyze how changing factors such as voltage and current affect battery aging over repeated charging cycles.
Erik Dufek from the laboratory says that he and his colleagues used data gathered from computer models and tested on actual batteries to develop a new charging protocol that is capable of charging a standard EV battery from 0% to 90% in only 10 minutes while preserving its long-term health. This protocol could revolutionize EV charging because it overcomes a major challenge to fast charging: high-voltage charging wears down EV batteries faster and negatively impacts a battery’s long-term life.
Virginia Tech’s Feng Lin says fast charging and battery health are balancing acts. While Virginia Tech was not involved in this new development, Lin notes that the risk with fast charging is that it can either lower battery capacity or damage the chemicals within the battery, thereby reducing its lifespan. But with this new charging protocol, EV drivers will be able to recharge their EVs in only 10 minutes without damaging their batteries.
As the technology to collapse EV battery charging time becomes commonplace in the years to come, electric vehicle makers such as ElectraMeccanica Vehicles Corp. Ltd. (NASDAQ: SOLO) are likely to use the technology so that motorists no longer have to stop for long periods of time during trips in order to recharge their vehicles.
ElectraMeccanica Vehicles Corp. Ltd. (SOLO), closed Monday's trading session at $1.49, off by 1.9737%, on 473,218 volume. The average volume for the last 3 months is 468,924 and the stock's 52-week low/high is $1.22/$4.10.
Freeport-McMoRan Inc. (FCX)
MarketClub Analysis, The Street, InvestorPlace, Schaeffer's, Kiplinger Today, SmarTrend Newsletters, StocksEarning, MarketBeat, Barchart, StreetAuthority Daily, Trades Of The Day, StreetInsider, TopStockAnalysts, Money Morning, Investopedia, Daily Trade Alert, Louis Navellier, VectorVest, Zacks, Trading Markets, Marketbeat.com, The Growth Stock Wire, ProfitableTrading, Daily Wealth, The Wealth Report, TheStockAdvisors, Top Pros' Top Picks, StreetAlerts, Wall Street Elite, All about trends, Leeb's Market Forecast, TradingMarkets, Streetwise Reports, The Online Investor, Market FN, INO.com Market Report, TheStockAdvisor, Dividend Opportunities, QualityStocks, InvestmentHouse, StockTwits, Investor Guide, Investors Alley, Trading Tips, Wealth Insider Alert, Investment House, Trading Concepts, InvestorGuide, Money and Markets, Market Intelligence Center Alert, Options Trader Elite, The Best Newsletters, Energy and Capital, Darwin Investing Network, Buttonwood Research, The Motley Fool, Trade of the Week, AnotherWinningTrade, Wealth Daily, Investing Signal, Wyatt Investment Research, Bourbon and Bayonets, MarketWatch, Market Authority, Early Bird, Investor Update, Profit Confidential, Wealthpire Inc., Street Insider, TradingAuthority Daily, Wall Street Daily, Cabot Wealth, Stock Research Newsletter, Super Stock Investor, InvestorsUnderground, AllPennyStocks, CNBC Breaking News, Daily Markets, Uncommon Wisdom, FeedBlitz and Normandy Investment Research reported earlier on Freeport-McMoRan Inc. (FCX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The second quarter of the year has been rough on the mining industry. With production costs on the rise and the global economy faltering in recent months, fortunes have dimmed substantially in the mining sector in recent months.
Global mining activity has slowed down since June, and according to analysts from S&P Global Market Intelligence, lower metal prices and an increasingly negative global economic outlook are to blame.
Senior analyst Sean DeCoff said in a quarterly report, titled “State of the Market; Mining Q2,” that the valuations of metals and the companies that mine them have been impacted by worsening economic conditions around the globe as well as low metal prices. This is despite the fact that metal and mining stocks outperformed the S&P 500 through the three months since June.
Speaking during a webcast, S&P Global Commodity Insights’ research director Mark Ferguson stated that certain indices faired much poorer in the first half of the year; he attributed this to an increase in market volatility in the macroeconomic outlook. In fact, the past couple of months were so rough on the metals and mining industry that the S&P 500 saw the worst first half in half a century, he noted.
Ferguson added that reduced metal prices during the first half of the year resulted in limited capital support in the mining industry as well in the first half of 2022. He added that the ASX Metals and Minings and the TSX Global Mining indexes weathered the past few months better, achieving high prices and maintaining them well into April. However, Ferguson observes that those indices have also begun to struggle alongside the rest of the mining market.
The June quarter saw especially depressed equity market support for the mining industry, Ferguson stated, arguing that the drop in metal prices further weakened capital support for the industry.
S&P Global tracks and compiles the aggregate market caps of nearly 2,800 public listed companies worldwide, the analysts noted. And when they reviewed the data from this group of companies, they found that June saw especially low equity market support levels. Although the analysts noted that equity support levels hadn’t fallen off the cliff, they acknowledged that the levels were still down compared to last year and the second half of 2020.
In addition to this, Ferguson pointed out gold as a standout, observing that it held sway through the period while other metal prices were impacted by geopolitical factors. The solid performance of gold during this turbulent period boosted the balance sheets of giant mining companies such as Freeport-McMoRan Inc. (NYSE: FCX).
Freeport-McMoRan Inc. (FCX), closed Monday's trading session at $29.76, off by 5.5238%, on 19,734,757 volume. The average volume for the last 3 months is 19.439M and the stock's 52-week low/high is $24.80/$51.99.
The QualityStocks Company Corner
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN)
- Mullen Automotive Inc. (NASDAQ: MULN)
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF)
- American Cannabis Partners
- GeoSolar Technologies Inc.
- D-Wave Quantum Inc. (NYSE: QBTS)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
- Aditxt Inc. (NASDAQ: ADTX)
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF)
- Cannabis Strategic Ventures Inc. (OTC: NUGS)
Cybin Inc. (NEO: CYBN) (OTC: CYBN)
The QualityStocks Daily Newsletter would like to spotlight Cybin Inc. (NEO: CYBN) (NYSE American: CYBN).
Cybin (NEO: CYBN) (NYSE American: CYBN), a biopharmaceutical company focused on progressing Psychedelics to Therapeutics(TM), has announced a clinical milestone: the first two participants in its phase 1/2a trial evaluating CYB003 for the treatment of major depressive disorder (“MDD”) have been dosed. According to the announcement, CYB003 is the first-ever novel psilocybin analog to enter clinical development. The company is conducting a randomized, double-blind, placebo-controlled study evaluating people with moderate to severe MDD in which participants will receive two administrations (placebo/active and active/active) with a response assessed at week 3 after first dose and at week 6 after second dose. “To commence dosing in our first-in-human phase 1/2a trial is a tremendous milestone for Cybin, especially having reached the clinic within just 18 months,” said Cybin CEO Doug Drysdale in the press release. “Our goal continues to focus on becoming a leader in creating the best psychedelic therapies for patients and today we have moved one step closer. Through our rigorous preclinical work and ongoing clinical development of CYB003, we believe we have the potential to unlock the powerful benefits of psilocybin for the treatment of MDD without its well-known limitations. . . . The high level of participant interest in our study serves to validate the significant unmet need for alternative and better treatment options to improve mental health conditions. We expect that this phase 1/2a trial will provide valuable insights and data. These findings will be critical in establishing a safe and efficacious treatment profile for CYB003 so we can continue to progress our mission to help revolutionize the treatment landscape for people suffering from depression.” To view the full press release, visit https://ibn.fm/CftJi
Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) is a Canada-based life sciences company focused on the pharmaceutical development of psychedelic products, as well as the functional mushroom market.
The early-stage company boasts an experienced management team featuring industry veterans from pharmaceutical and consumer product backgrounds who have run multiple clinical trials and collectively helped facilitate billions of dollars in product revenues. The team is dedicated to the development of products and protocols within the psychedelic, pharmaceutical and nutraceutical industries.
In particular, Cybin aims to further build upon and expand its intellectual property (IP) portfolio, which is structured around unique psilocybin delivery mechanisms that target a number of different therapeutic indications. In addition, the company has dedicated itself toward furthering its research and IP within the fields of synthetic compounds, extraction methods, the isolation of chemical compounds, new drug formulations and protocol regimes.
Serenity Life Sciences & Natures Journey Inc.
The company’s business model is centered around its two core subsidiaries, Serenity Life Sciences and Natures Journey Inc., which comprise Cybin’s two-pronged approach toward delivering fungi-derived psychedelic and medicinal products.
Serenity Life Sciences is focused on furthering research and development of psilocybin-based medications. Psilocybin is found in certain species of mushrooms and is a non-habit forming, naturally occurring psychedelic compound. Research into psilocybin has shown positive results for the treatment of depression, anxiety, PTSD, addiction, eating disorders, ADHD and other indications.
Natures Journey Inc. operates the Journey brand, which specializes in developing proprietary medicinal mushroom products that target and promote mental wellness, immune boosting detoxification and overall general health and wellbeing.
Partnership with the Toronto Centre for Psychedelic Science (TCPS)
Staying true to its axiom of being a research-first medicinal mushroom life sciences company, Cybin recently announced its entry into a strategic partnership with the Toronto Centre for Psychedelic Science (TCPS), with the goal of furthering its ongoing psilocybin research efforts and expanding Cybin’s psilocybin IP portfolio (http://nnw.fm/9EUkI).
“While there is evidence to support psilocybin as a treatment for certain indications, the Toronto Centre for Psychedelic Science is taking a clinical approach to prove or disprove the safety and efficacy of psilocybin-based microdosing through an open science approach,” Paul Glavine, CEO of Cybin, stated in a news release.
“We are excited to join forces with Cybin and to offer our expertise. A number of firms had approached TCPS, but Cybin demonstrated a superior commitment to high-quality research and integrity in product development. Our high standards for scientific rigor and transparency will find a fitting home within the culture Cybin is cultivating in Canada and abroad,” Thomas Anderson, co-founder of the Toronto Centre for Psychedelic Science, added.
Journey’s Product Monetization & Market Potential for Nutraceutical Supplements
Although Cybin is at the forefront of companies seeking to conduct clinical trials aimed at gaining regulatory approval for psilocybin and other psychedelic products, the company has also placed a great deal of emphasis on generating meaningful revenue from its very outset.
Cybin’s Journey brand has is launching a range of supplements comprised of popular fungi-derived ingredients such as Reishi, Lion’s Mane and Cordyceps. Purported to aid focus and concentration while promoting neurogenesis, Journey’s range of nutraceutical products provides Cybin with a crucial foothold within the non-psychedelic legal supplement market, which is valued at over $25 billion globally and growing at a 9% year-over-year rate.
Pharmaceutical Psychedelics
In addition to the company’s range of non-psychedelic supplements, Cybin has plans to carry out a clinical trial with a new delivery system for its psilocybin-based medications later this year. Ultimately, the company aims to enter into technology transfer agreements with global pharmaceutical companies after phase 1 & phase 2 clinical trials are complete in order to accelerate regulatory approvals in major indications in global markets with entire lifecycle product management.
With products such as psilocybin truffles already legal in nations such as the Netherlands, Jamaica and Bulgaria, Cybin has positioned itself to capitalize on an eventual legalization of psychedelic mushroom-derived products in the future. Working within a regulatory environment with strong similarities to that which dealt with cannabis prior to the industry’s eventual legalization by the Canadian government in 2018, Cybin is laying the groundwork for the moment pharmaceutical psychedelics gain acceptance in North America and abroad.
Amalgamation Agreement and Financing
Cybin recently announced its entry into an amalgamation agreement dated June 26, 2020, with Clarmin Explorations Inc. (TSX.V: CX) and 2762898 Ontario Inc., a wholly owned subsidiary of Clarmin (http://nnw.fm/w04LH). Completion of the transactions contemplated in the amalgamation agreement will result in the reverse takeover of Clarmin by Cybin.
In connection with the proposed transaction, Cybin plans to complete a “best-efforts” brokered private placement of subscription receipts of Cybin, with a syndicate of agents co-led by Stifel Nicolaus Canada Inc. (Stifel GMP) and Eight Capital, to raise a minimum of C$14 million ($10 million) and a maximum of C$21 million ($15 million), with a 15% agents’ option.
To date, Cybin has raised approximately C$10,400,000 through an initial financing round and its series A financing round.
Cybin Inc. (NEO: CYBN) (NYSE American: CYBN), closed Monday's trading session at $1.09, up 2.8302%, on 1,512,322 volume. The average volume for the last 3 months is 1.477M and the stock's 52-week low/high is $0.3903/$2.88.
Recent News
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Begins Dosing Participants in CYB003 Trial for Treatment of MDD
- How Psychedelics Startups Are Improving Their Resilience
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Leading Out in Transformation of Psychedelic Sector
Mullen Automotive Inc. (NASDAQ: MULN)
The QualityStocks Daily Newsletter would like to spotlight Mullen Automotive Inc. (MULN).
Mullen Automotive (NASDAQ: MULN), an emerging electric vehicle (“EV”) manufacturer, owns and partners with several synergistic businesses to create clean and scalable EVs and energy solutions. “The company’s ability to reach this goal can perhaps be measured by its success, which was recently on display when Mullen inked a deal with DelPack Logistics LLC (‘DPL’) for up to 600 Mullen Class 2 Electric Cargo Vans. The two companies entered a binding agreement calling for DPL, an Amazon delivery service partner, to purchase the EVs over the next 18 months; the first 300 will be ready for delivery by Nov. 30, 2022,” reads a recent article. “This agreement is a milestone for Mullen Automotive,” said Mullen CEO and Chair David Michery. “DelPack is a leader in last-mile package delivery, and this agreement puts our Class 1 cargo van program front and center for last-mile delivery opportunities.” To view the full article, visit http://ibn.fm/dID9D
Mullen Automotive Inc. (NASDAQ: MULN) is a Southern California-based automotive company that owns and partners with several synergistic businesses working toward the unified goal of creating clean and scalable energy solutions. Mullen has evolved over the past decade in sync with consumers and technology trends. Today, the company is working diligently to provide exciting EV options built entirely in the United States and made to fit perfectly into the American consumer’s life. Mullen strives to make EVs more accessible than ever by building an end-to-end ecosystem that takes care of all aspects of EV ownership.
Commencement of Trading on Nasdaq
On November 5, 2021, Mullen announced its commencement of trading on the Nasdaq Capital Market.
“Today is a monumental day for Mullen Automotive. I am especially proud of our team, investors and all who have believed in Mullen and taken us to this point as a publicly traded company on the Nasdaq Capital Market,” David Michery, CEO and Chairman of Mullen Automotive, stated in the news release. “Trading on Nasdaq now opens us up to new investors, both institutional and retail shareholders, and broadens our awareness and company profile, while increasing awareness of Mullen and our technology platform and opening new opportunities in EV and beyond. The road ahead has never been brighter for Mullen, and I am proud to lead us into the future.”
The milestone came in the wake of the company’s stock-for-stock merger with Net Element Inc.
The Mullen FIVE
The Mullen FIVE EV Crossover, debuting at the Los Angeles International Auto Show (LAIAS) on November 17, 2021, embodies Mullen’s Southern California roots with an inspired design focused on two complementary Golden State themes – California landscape and California urban.
The FIVE is built on an EV Crossover skateboard platform that offers multiple powertrain configurations and trim levels in a svelte design that is Strikingly Different™ and exciting to experience in person.
Prior to the start of LAIAS, the Mullen FIVE was selected as a finalist by the LA Auto Show for Top EV SUV in the ZEVA “People’s Choice” Awards.
LAIAS provides Mullen an opportunity to display multiple variants of the FIVE model while also showcasing its powertrain, battery and charging technology. The company intends to bring the FIVE to market in 2024, and reservations are currently open here.
Mullen’s development portfolio also includes EV Fleet Vans, which it intends to bring to market in Q2 2022, and the pure electric, high performance Mullen DragonFLY.
Expansion of Manufacturing Capacity
On November 2, 2021, Mullen announced plans to expand its facility in Robinsonville, Mississippi.
Mullen’s Advanced Manufacturing and Engineering Facility (AMEC) currently occupies 124,000 square feet of manufacturing space. The total available land on the property is over 100 acres, and Mullen is moving ahead with plans to build out another 1.2 million square feet of manufacturing space to support class 1 and class 2 EV cargo vans and the Mullen FIVE EV Crossover.
On the expanded site, Mullen plans to build a body shop, a fully automated paint shop and a general assembly shop.
EV Market Outlook
The global EV market was reported to consist of 3,269,671 units in 2019, a figure that is expected to grow at a CAGR of 21.1% through 2030 to a total of 26,951,318 units worldwide. This market’s monetary value was estimated at $162.34 billion in 2019 and is expected to grow at a CAGR of 22.6%, resulting in an approximate value of $802.81 billion by 2027. The primary driver for this exponential growth is a worldwide increase in vehicle emissions regulations.
Management Team
David Michery is the CEO and Founder of Mullen and has been leading the company and its divisions since inception in 2014. With over 25 years of executive management, marketing, distressed assets, and business restructuring experience, Mr. Michery brings a wealth of relevant knowledge and expertise to the Mullen brand. He has notably created 12 trademarks so far to develop the company brand and vision.
Mr. Michery is working toward a sustainable future accessible to all by creating a suite of clean-energy electric vehicles at varied price points. With entirely U.S.-based manufacturing and operations, he is also determined to have Mullen Technologies play a role in shaping a self-sustaining local economy by creating more jobs in America.
Mr. Michery manages risks and company expectations as a pathway to success and has personally overseen several businesses that totaled over $1 billion in transactions. His key strength is the ability to be fiscally responsible and lead teams to complete projects on time and within budget. As a seasoned professional in this space, Mr. Michery has demonstrated skill in building businesses from the ground up and into successful entities that subsequently sold for hundreds of millions of dollars.
Mullen Automotive Inc. (MULN), closed Monday's trading session at $0.6355, up 3.0151%, on 78,287,142 volume. The average volume for the last 3 months is 78.287M and the stock's 52-week low/high is $0.52/$15.90.
Recent News
- Mullen Automotive Inc. (NASDAQ: MULN) - GreenCarNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Positioned 'Front and Center for Last-Mile Delivery Opportunities'
- Analysts Foresee Growing EV Sales in Europe, But Doubts Remain
- InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Announces New Strategic Collaboration, Rapid Progress on FIVE EV Crossover Program
Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF)
The QualityStocks Daily Newsletter would like to spotlight Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF).
Playgon Games (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR), a propriety SaaS technology company delivering mobile first live dealer technology to online gaming operators globally, is reporting on its second quarter financial results for the period ended June 30, 2022. Highlights of the report include revenues from the Live Dealer platform of $216,881 for the quarter with total revenues of $357,758 for the six-month period ended June 30, 2022. According to the announcement, revenues for the quarter increased by $76,004 from Q1 2022, representing a 54% increase, and operator net win for the quarter was $3 million, an 11% increase from the previous quarter, which totaled $2.7 million. The company reported a net loss for the quarter of $4,402,353 with cash on hand as of June 30, 2022, totaling $539,485. Operational highlights for the quarter included the onboarding of 14 additional operators, with average monthly players reaching 20,100, compared to the previous quarter of 17,500. During the quarter, Playgon applied for a Gaming Related Supplier–Manufactures License with the Alcohol and Gaming Commission of Ontario and signed a software licensing and distribution agreement with Markor Technology whereby Markor will add and market Playgon's proprietary mobile first live dealer and E-table game offerings. “Playgon experienced a strong Q2 as the company saw continued growth in revenue and player activity," said Playgon Games CFO Harry Nijjar in the press release. “Our existing customer base continue to show growth in these two key metrics, and we expect the same from the new operators added during Q2. We strongly believe that with the new corporate milestones achieved already this third quarter, industry continues to validate Playgon's Live Dealer offering.” To view the full press release, visit https://ibn.fm/ySZfT
Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) is a SaaS technology company focused on developing and licensing digital content for the growing global iGaming market. The company provides a multi-tenant gateway that allows online operators the ability to offer their customers innovative iGaming software solutions. Its current software platform includes Live Dealer Casino, E-Table Games and Daily Fantasy Sports. Seamless integration at the operator level allows customer access without requiring the sharing of any sensitive customer data. Playgon games run on any browser and any device as fast and secure as a native app, without requiring any app store download. All that’s needed is a stable internet connection. The gaming experience is identical across all mobile devices. As a true business-to-business digital content provider, the company’s products are scalable turnkey solutions for online casinos, sportsbook operators, location-based operators, media groups, and big database companies.

Playgon’s proprietary technology provides digital games for online gambling sites and mobile device apps, with the company licensing its mobile live-dealer technology to online gaming operators worldwide. Playgon combines high definition live streaming dealers with state-of-the-art augmented reality betting to provide the most authentic casino experience, live from Las Vegas. Playgon’s mobile platform features popular table games, all optimized for one-handed play on mobile devices.
The COVID-19 pandemic has accelerated an already existing shift away from location-based casinos to online gambling. At the same time, the proliferation of mobile devices has provided players with new access to betting. A younger, tech-savvy consumer demographic is driving adoption of digital gaming globally. To meet this demand, Playgon has launched a studio with 10 gaming tables from which its live dealer streaming video originates. The company’s platform is live with multiple online casino operators through four aggregator clients in South Africa and Europe, and commitments are coming in from more.
Playgon plans to expand the studio to 25 tables in the near term and is working to establish a U.S. strategy. The company will continue to expand licensing of its live dealer games to iGaming operators worldwide under a SaaS license agreement. As a B2B software supplier, Playgon avoids player acquisition costs.
Games

Live Dealer Casino
Playgon offers the first and only Live Dealer Casino streaming live from Las Vegas. The company brings cutting-edge handheld features and functionality to the mobile generation of gaming enthusiasts who demand a world-class gaming experience on all devices. Playgon’s Blackjack delivers the look and feel of location-based casino tables with features providing players with the most unique user experience. The company’s true-to-life Roulette offers players a clear and uninterrupted view of the dealer, wheel, ball, bets, results, trends and statistics. Players can strategize, place multiple bets, track results and review trends without ever losing focus of the game.
Playgon’s traditional Baccarat and proprietary Tiger Bonus Baccarat™ prove their worth by not only recognizing the need for a prominent product, but by adding elements which separate them from the pack without removing their authenticity. The games mix advances in technology with the traditional game attributes that have resonated and captivated players for hundreds of years.

eTable Games
To lead the rise of mobile-first gaming, Playgon developed a user experience perfected for one-handed play. Providing this next evolution in gaming technology ensures the company’s client operators loyalty from existing customers and is a powerful strategy to attract and retain new players. Playgon’s VEGAS LOUNGE™ brings together an innovative mix of games, technology and gameplay that offers players an authentic experience and real Las Vegas casino fun every time, everywhere.
Daily Fantasy Sports
Playgon’s Daily Fantasy Sports (DFS) are a subset of fantasy sport games which typically target a younger demographic. DFS provides iGaming operators a turnkey fantasy sports platform that can quickly go to market, integrate with the operator’s existing operations and services, and be customized to match and enhance the operator’s brand. The platform is mobile and desktop friendly, built for regulated market environments, and allows operators to monetize users through a network of shared liquidity.
Market Outlook
Online casinos and sports betting sites/apps are increasingly adding market share to traditional location-based casinos. This trend is only expected to accelerate as millennials reach their peak earning years and Gen Z youth begin to complete their education and move into careers. These generations are completely comfortable with online recreation, as well as tech like digital wallets and digital gameplay that underpins Playgon Games. The company has been described as “Netflix + Vegas, all in one.”
The online gambling market is slated to reach a value of $127.3 billion by 2027, according to Grand View Research, with much of the growth expected from the U.S. and Asia. Even Europe, the most mature gaming market, is expected to grow at a rate of 20-25 percent year over year. The current global online Live Casino TAM is estimated at about $6 billion annually, and revenue is forecast to reach more than $8 billion by 2023 and more than $13 billion by 2027.
Management Team
Darcy Krogh is CEO of Playgon Games. He is a veteran of the iGaming industry with over 20 years of experience. In 1999, he co-founded Chartwell Technology Inc., which pioneered the development of browser-based digital content for the iGaming industry. After that company was sold to Amaya Gaming Group, he served as VP Business Development with Amaya. In 2016, he started Playgon Games (formally Global Daily Fantasy Sports Inc.) as President and CEO. His experience in the online gaming industry includes sales and marketing, relationship management, corporate finance, M&A, and strategic corporate development.
Guido Ganschow is President of Playgon Interactive. He has more than 12 years of experience in creating real-time Live Dealer technology and platforms and was the co-founder and Creative Director for a Macau-based casino consortium. Between 2008 and 2014, he successfully created and established Live Dealer platform businesses in Asia and Europe, and executed commercial partnerships, sales, and integration of the Live Dealer solution with major global gaming brands, including Ho Gaming Group, Chartwell Technology and Amaya Gaming Group.
Steve Baker is COO of Playgon. He is a former VP Operations for Shaw Communications, where he was directly involved in video streaming, home entertainment, new products, sales and M&A. He oversaw revenue growth from $300 million to $2.8 billion and employee growth from 350 to 13,000. He has broad experience and a proven record in development and implementation of cost effective and efficient growth strategies transitioning businesses from development to operations.
Harry Nijjar is CFO of Playgon Games. He is currently a Managing Director with Malaspina Consultants Inc. and provides CFO and strategic financial advisory services to his clients across many industries. This experience has allowed him to help his clients successfully navigate the regulatory and financial environments within which they operate. Mr. Nijjar holds a CPA-CMA designation from the Chartered Professional Accountants of British Columbia.
Playgon Games Inc. (PLGNF), closed Monday's trading session at $0.0655, up 23.3522%, on 16,000 volume. The average volume for the last 3 months is 16,000 and the stock's 52-week low/high is $0.0299/$0.406.
Recent News
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Releases Q2 2022 Financial Report
- InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Announces Live Dealer Studio GLI Certification for Ontario and Isle of Man
- InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Shares Milestone Update
American Cannabis Partners
The QualityStocks Daily Newsletter would like to spotlight American Cannabis Partners.
Cannabis reform activists in Missouri have managed to get their legalization bill on the November ballot after being bogged down by setbacks and nearly forced to give up on their bill. According to unofficial tallies, the Legal Missouri campaign had not collected enough signatures to qualify for the upcoming ballot by late July, being 2,275 signatures short. With the ballot fast approaching, many feared the campaign’s hope of gathering enough signatures in time would be dashed. But activists behind the campaign managed to pull a major comeback, collecting the remaining 2,275 signatures by Aug. 9, 2022, and qualifying the legalization initiative for the upcoming ballot. Missouri officials announced in early August that the Legal Missouri campaign had collected the required number of signatures and that the organization’s cannabis legalization measure would appear on Missouri’s November ballot. The resilience displayed by the advocates to ensure that the ballot initiative qualifies for the November polls exemplifies how the entire marijuana industry and its actors, such as American Cannabis Partners, go to great lengths to make progress despite insurmountable odds.
American Cannabis Partners (ACP) is a multi-state cannabis company with 560,000 square feet of licensed canopy space for cultivation and one retail license. The company is nationally headquartered in Trinity County of Northern California’s Emerald Triangle.
ACP is focused on three complementary business segments: real estate, acquisition & development of proprietary assets, and ongoing cultivation operations. Led by a seasoned management team with 30+ years of canna-business experience, ACP’s strategy is to capture opportunities in real estate and licensing in states that have recently passed cannabis legalization legislation, thereby equipping the company to capitalize on Federal interstate commerce opportunities.

Through its current cultivation operations, ACP supplies approximately 80% of its whole flower products for manufacturing, distribution and retail licenses. With the remaining 20%, the company supplies its proprietary strains to select California distributors and its own Michigan retail location under its exclusive in-house brand, ZÜK.
History of American Cannabis Partners
In 2014, Stephen Jordan, President of ACP, took on the Director of Operations position for a U.S.-based company operating in the Jamaican cannabis space. Over the course of his three-year tenure in this role, Jordan developed a number of relationships that would help serve as the basis of American Cannabis Partners.
One such relationship was with Junior Gordon, a cultivation lead grower from Jamaica’s Westmoreland Parish. Jordan immediately saw the value of Gordon’s unique skillset and credentials, and Gordon recognized Jordan’s heartfelt vision of bringing Jamaican culture to the rapidly developing U.S. cannabis space.
Guided by that mission, ACP’s unchanging goal is to improve the lives of individuals through cannabis and business.
Current Operations
Since its founding in 2018, privately-owned American Cannabis Partners has established a foothold in two key U.S. cannabis markets – California and Michigan. In total, the company has acquired 12 cannabis licenses, including 20,000 sq. ft. of cultivation licenses in California and 540,000 sq. ft. of cultivation licenses & one retail license in Michigan.
ACP’s IP portfolio features three proprietary strains sold exclusively through the company’s wholly owned ZÜK brand, as well as proprietary data collection and mining systems supporting its cultivation and retail operations.
Plans for Expansion
American Cannabis Partners is pursuing additional growth in the cannabis sector through multiple planned initiatives. These include:
- Submitting applications for additional cultivation licenses at the company’s Trinity County, California, location;
- Planning land acquisition and project development strategies for expanding operations to its third U.S. state beginning in the second quarter of 2022; and
- Planning land acquisition and project development strategies for expanding operations to its fourth U.S. state beginning in the second quarter of 2024.
ACP is currently exploring expansion opportunities through partnerships and joint ventures in New Jersey, New York, Virginia, Nevada, Arizona, Missouri and Massachusetts.
Management Team
Stephen Jordan is the CEO of American Cannabis Partners. He is focused on the first and last steps of legal cannabis – cultivation and retail. To date, Mr. Jordan has provided the company with ownership of 12 licenses, three proprietary cannabis strains and multiple real estate assets. His background in cannabis operations and financial strategies has guided American Cannabis Partners’ efforts to produce consistently high-quality product for both the medical and recreational segments. Mr. Jordan has operated under cultivation, manufacturing, distribution, medical research (Univ. of West Indies), retail and exportation licenses in multiple countries, further strengthening his network within the cannabis industry.
Gary Coltek is the company’s Chief Operating Officer. He has credentials based in the culinary, hospitality and sustainability industries spanning over 40 years, including taking three companies public. Mr. Coltek has held management positions internationally with Ritz Carlton, Four Seasons, Trump Hospitality, Phymatrix and International Oncology Network. For 17 years, he was the founding member and partner of a private boutique consulting firm. He is currently a guest speaker and visiting professor at universities in Israel, China, Italy, the Netherlands and Peru, covering topics that include culinary sustainability, sustainable cannabis farming, organic sustainable farming and cannabis clinical studies.
Scot C. Crow is the Lead Corporate Counsel for American Cannabis Partners. He has extensive experience in corporate mergers & acquisitions and tax law. His clients rely on him to advise them with respect to their complex financial transactions and provide outside general counsel. Mr. Crow provides his clients proactive advice with respect to sensitive management matters, litigation management, day to day transactional needs and objective assessments for the development of successful business strategies. His experience includes serving as lead counsel for numerous mergers & acquisitions, private equity investments, private offerings, venture capital financings, mezzanine debt offerings, divestures and other related transactions, with an emphasis in the legalized marijuana segment.
Jacob Frenkel is the company’s Lead Compliance Counsel. He is the current Chair of Dickinson Wright’s Government Investigations and Securities Enforcement Practice. Mr. Frenkel’s solutions-minded approach to issues has earned him a reputation as an aggressive, tenacious, creative and proactive defense lawyer and litigator. After 14 years as a Senior Counsel in the SEC’s Division of Enforcement, U.S. federal criminal prosecutor and New Orleans Assistant District Attorney, Mr. Frenkel has practiced in the private sector for 20 years. His unique mix of corporate transactional, litigation and investigations defense clients extend well beyond the cannabis industry and cover a wide range of industries worldwide.
Junior Gordon is the Director of Cultivation for American Cannabis Partners. With 30 years of international cannabis cultivation experience in both the Caribbean and United States, Mr. Gordon is recognized as one of the top growers in the world. His skills span both controlled indoor and large volume outdoor harvest programs, giving him proficiency in nursery, propagation and indoor & outdoor grow strategies. As a winner of High Times and other notable Cannabis Cups, his focus is on connecting the dots between propagation, soil, irrigation, planting, harvesting, curing, processing and inventory control, bringing Jamaican cannabis cultivation best practices to American Cannabis Partners’ operations.
Recent News
- American Cannabis Partners - 420 with CNW — How Missouri Activists Overcame Setbacks to Put Cannabis Measure on Upcoming Ballot
- InvestorNewsBreaks – American Cannabis Partners Looking to Capitalizing on Possible Future Federal Interstate Commerce Opportunities
- American Cannabis Partners Has Vested Interest in Cannabis Act Just Passed by U.S. House

GeoSolar Technologies Inc.
The QualityStocks Daily Newsletter would like to spotlight GeoSolar Technologies Inc.
- GeoSolar goes beyond solar panel installation to provide complete home energy makeovers that can eliminate utility bills, provide energy independence
- GeoSolar’s SmartGreen(TM) technology produces energy through rooftop photovoltaic (“PV”) solar systems and geothermal ground loops
- Additional system customizations include tightening building envelope, insulation upgrades, LED lighting replacement, and EV charging infrastructure
- SEC recently qualified GeoSolar for Regulation A+ capital raise
GeoSolar Technologies (“GST”), a Colorado-based climate technology company, goes beyond solar panel installation to provide complete building “green energy” makeovers that redefine power generation, utility, and conservation. GeoSolar’s SmartGreen(TM) energy technology leverages the power of the earth and sun to heat, cool, power, and purify newly built and existing homes. A SmartGreen(TM) building produces energy in two ways: rooftop photovoltaic (“PV”) solar systems generate electricity year-round, and geothermal ground loops use the earth as a heat source or heat sink depending on the home’s heating and cooling needs.
GeoSolar Technologies Inc. (“GST”) is a Colorado-based climate technology company and the creator of the Smart Green Home® system for newly built and existing residences and commercial buildings. The company is focused on revolutionizing the way we heat, cool and power homes with 100% natural energy sources. Its patent-pending integrated system harnesses energy from the earth and sun to power and purify homes and automobiles without the use of fossil fuels.
In a GST home, the sun’s energy is captured on the roof to generate all of the electricity required. Additionally, the consistent climate of the earth is used to keep the home at a perfect temperature year-round, and the company’s proprietary air purifying unit ensures that the air inside the home is safe and healthy.
GST’s home technology has been installed in multiple test homes in Colorado and achieved exceptional results, including some of the most impressive energy efficiency ratings (HERS) in the industry.
GeoSolar Technologies is currently accepting investment as part of a Regulation A+ offering. Everyone* can invest now for as little as $300. For more information, visit the company’s profile on Manhattan Street Capital and review its Offering Circular.
GeoSolar Technologies Inc. (“GST”) has been qualified by the U.S. Securities and Exchange Commission (SEC) to conduct a Regulation A+ capital raise. GST is already a publicly traded company who makes quarterly and annual filings with the SEC and is subject to quarterly PCAOB audits. This is the first time shares of GeoSolar Technologies are being made available for public purchase. Upon completion of this Regulation A+ offering, the company intends to seek a listing of its stock.
The Decarbonization Movement
 Soaring and unstable energy/fuel costs continue to highlight the importance of rethinking the traditional approach to powering homes, from top to bottom. While most everyone is well aware of the remarkable, multi-trillion-dollar opportunity the electric vehicle transformation offers to investors (in addition to the benefits to the climate problem), few recognize that the all-electric home market is as large as electric vehicles and equally important to reducing carbon emissions.
Soaring and unstable energy/fuel costs continue to highlight the importance of rethinking the traditional approach to powering homes, from top to bottom. While most everyone is well aware of the remarkable, multi-trillion-dollar opportunity the electric vehicle transformation offers to investors (in addition to the benefits to the climate problem), few recognize that the all-electric home market is as large as electric vehicles and equally important to reducing carbon emissions.
U.S. energy expenditures clocked in at $3,891 per person in 2018, leading to estimated spending of $1.3 trillion on energy that year alone. Despite this, fewer than 3% of U.S. homes are currently powered by solar. This number is poised to increase exponentially as both new and existing residences transition to zero carbon models.
GST estimates that if all the homes in America were powered by its technology, carbon pollution could be reduced by an estimated 1.9 trillion pounds per year, greatly reducing the negative impacts on our climate.
GeoSolarPlus®
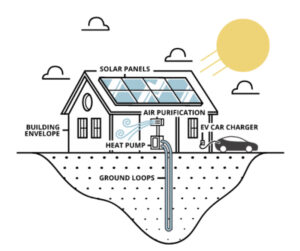 The GeoSolarPlus (“GSP”) system combines solar power, geothermal ground-sourced energy and other clean energy technologies into one fully integrated system.
The GeoSolarPlus (“GSP”) system combines solar power, geothermal ground-sourced energy and other clean energy technologies into one fully integrated system.
Key benefits of the GSP system include:
- Making a real planet-changing difference in reducing air pollution
- Eliminating or significantly reducing homeowners’ future utility bills
- Enjoying lifetime energy independence and protection from price escalation and energy shortages
- Eliminating greenhouse gas emissions from operation of home and daily life
- Increasing home value
- An integrated design for seamless operation of renewable energy systems
- Maintaining a significantly healthier living environment
- Leveraging existing renewable energy tax credits and electrification incentives
- Creating stable jobs capable of supporting families in the decarbonized future
Click here to learn more about how GeoSolarPlus works.
Management Team
The GST leadership and management team includes some of the world’s most experienced and respected leaders in the fields of decarbonization and sustainable homes.
Stone Douglass is the Chairman and CEO of GST. He is a seasoned, 30-year public company executive and former Chairman and CEO of the Piper Aircraft Company.
Brent Mosbarger is the company’s Co-Founder and leads its commercial operations. He is a highly respected solar engineer whose experience includes roles with Chevron Energy’s green operations and serving as project manager and executive for a $400 million solar/geothermal innovation project.
Peter Romenesko is a Senior Strategic Advisor with GST. He brings to the company considerable experience as an engineer and large-scale project manager for Johnson Controls and Siemens.
Dr. Norbert Klebl is the company’s Co-Founder and Development Director. Recognized as one of the world’s leading experts in the field of zero-carbon innovation, he is a former McKinsey partner of 16 years with an MBA from Columbia.
Dar-Lon Chang is GST’s Director of New Product Development. Prior to joining GST, he had a 16-year career with ExxonMobil Energy Research. He received his PhD in engineering from the University of Illinois.
* Must be over 18, certain states are not currently available and will be added soon.
Recent News
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc.’s SmartGreen(TM) Tech Transforms Homes, Enables Energy Independence
- QualityStocksNewsBreaks – GeoSolar Technologies Inc. Leveraging SmartGreen(TM) to Address Systemically Unsustainable Growth in Energy Demand
- GeoSolar Technologies Inc. Helps Residents and Businesses Achieve “Net-Zero” Ahead of Global Targets

D-Wave Quantum Inc. (NYSE: QBTS)
The QualityStocks Daily Newsletter would like to spotlight D-Wave Quantum Inc. (NYSE: QBTS).
D-Wave (NYSE: QBTS), a leader in quantum computing systems, software and services and the only company building both quantum annealing and gate-based quantum computers, has expanded its portfolio with a growing stream of customers. Through an innovative new program, D-Wave is helping companies from a variety of industries, including manufacturing and logistics, financial services and life sciences, as they show increasing interest in evaluating the benefits of quantum computing solutions. A recent article reads, “With a commitment to making quantum solution implementation as easy as possible for its customers, D-Wave designed the Launch(TM) program. The program’s goal is to help companies across a multitude of verticals get started with quantum computing by identifying the right types of business problems to run on a quantum computer and developing solutions to address these complex challenges through its proprietary annealing quantum computing and quantum hybrid technologies. The four-step D-Wave Launch(TM) program engages an in-house professional services team and technical domain experts to kick off quantum projects, provide the necessary quantum training for teams, and realize business value faster.” To view the full article, visit https://ibn.fm/b4GGL
D-Wave Quantum Inc. (NYSE: QBTS) is a leader in quantum computing systems, software and services focused on delivering customer value via practical quantum applications for problems such as logistics, artificial intelligence, materials sciences, drug discovery, scheduling, fault detection and financial modeling. As the only provider building both annealing and gate-model quantum computers, the company is unlocking commercial use cases in optimization today, while building the technologies that will enable new solutions tomorrow.
D-Wave is a pioneer in quantum computing, with a history of delivering the world’s first commercial quantum computer; the first real-time quantum cloud service; countless hardware and software product and research milestones; and the planned first cross-platform quantum solution which will deliver both annealing and gate-model quantum computers to customers via an integrated platform. Its current commercial product offerings include: Advantage™ (fifth generation quantum computer), Leap™ (quantum cloud service), Launch™ (quantum computing onboarding service) and Ocean™ (full suite of open-source programming tools).
D-Wave’s relentless pursuit of practical quantum computing has resulted in the technology being used today by some of the world’s most advanced enterprises – more than 25 of the Forbes Global 2000 use D-Wave.
D-Wave’s commercial customers include blue-chip industry leaders like Volkswagen, Accenture, BBVA, NEC Corporation, Save-On-Foods, DENSO and Lockheed Martin. The company boasts an extensive IP portfolio featuring more than 200 issued U.S. patents and over 100 peer-reviewed papers published in leading scientific journals.
Founded in 1999, D-Wave is the world’s first commercial supplier of quantum computers. With headquarters and the Quantum Engineering Center of Excellence based near Vancouver, Canada, D-Wave’s U.S. operations are based in Palo Alto, California.
Advantage™ Quantum Computer
With the Advantage™ Quantum Computer, D-Wave has incorporated two decades of experience and over 10 years of customer feedback to create the first and only quantum computer designed for business. The platform features a new processor architecture with over 5,000 qubits and 15-way qubit connectivity. This is 2.5x more connections and more than double the number of qubits than the company’s previous generation quantum computer.
D-Wave’s quantum computers, first located in its facilities in British Columbia, have been available to North American users through its Leap™ quantum cloud service since 2018. It has since introduced new Advantage systems in Julich, Germany, and most recently, Marina Del Rey, California, which marked the availability of the first Advantage quantum computer physically located in the United States.
That new deployment is part of the USC-Lockheed Martin Quantum Computing Center (QCC) hosted at USC’s Information Sciences Institute (ISI), a unit of the University of Southern California’s prestigious Viterbi School of Engineering. Additionally, Amazon Web Services (AWS) and D-Wave announced that the U.S.-based system is available for use in Amazon 2racket, expanding the number to three different D-Wave quantum systems available to AWS users.
Leap Quantum Cloud Service
D-Wave’s customers interface with its systems through the Leap™ quantum cloud service. Leap delivers immediate, real-time access to the company’s Advantage quantum computer and quantum hybrid solver service, all with enterprise-class performance and scalability.
Leap allows developers proficient in Python to get started building and running quantum applications. Through a seamless and secure cloud-based connection, users can easily start solving complex problems of up to 1 million variables and 100,000 constraints.
Using Leap, D-Wave customers have developed quantum hybrid applications for use cases in manufacturing, logistics, financial services, life sciences, materials science, retail and transportation. By eliminating the need to wait hours, days or weeks to get good answers to a broad array of problems, D-Wave is helping businesses move forward.
D-Wave Launch
D-Wave Launch™ is the company’s onboarding platform aimed at helping businesses easily start their quantum journey. Through this program, D-Wave’s team of experts and partners aid enterprises in identifying best use cases for quantum and work with them to develop a proof of concept and production pilot.
From there, the team coordinates with customers to get their hybrid quantum applications up and running, providing ongoing Leap quantum cloud access to ensure the application is operating smoothly and delivering real business value.
Target Verticals
While the potential applications for quantum computing are effectively limitless, D-Wave has identified a number of industry verticals as key areas of focus for its quantum architecture, providing case studies for each. These include:
- Manufacturing – D-Wave worked with Volkswagen to identify a commercial optimization application, the binary paint shop problem, which was run on D-Wave’s hybrid solver service. The solver outperformed four purely classical methods on problem sizes at commercial scale (N=3,000). In a separate project, similar inputs were tested using a leading ion trap system, which failed to find any commercial solution.
- Life Sciences – Menten AI makes use of D-Wave quantum computing to assist in the design of novel therapeutic peptides—short strings of amino acids that can act as potent drugs. With the rise of COVID-19, D-Wave’s Advantage system made it possible to identify molecules that might be especially well-suited for binding and inhibiting the related spike protein, producing several promising peptide designs.
- Finance – Multiverse Computing, a leader in developing quantum solutions for the financial sector, leveraged D-Wave’s hybrid solver service in a collaboration with BBVA, one of the world’s largest financial institutions. Multiverse demonstrated management strategies that far exceeded the granularity of traditional returns in a fraction of the time, helping BBVA identify a low-risk portfolio for investment.
Market Opportunity
The quantum computing total addressable market is projected to grow between $450 billion and $850 billion over the next 15 to 30 years, with between $5 billion and $10 billion of anticipated TAM growth coming in the next three to five years, according to Boston Consulting Group. Driving factors behind this growth include rising investments in quantum computing tech by governments and an increasing number of commercial use-cases.
Forward-thinking organizations see quantum as an opportunity to move ahead of the competition. From finding efficiencies and reducing waste to decreasing time to solution and solving problems abandoned due to complexity, the business value is real. According to data from 451 Research, 40% of large enterprises are already experimenting with quantum computing.
D-Wave is strategically positioned – in an industry with significant barriers to entry – as evident by a decades-long track record serving a roster of blue-chip customers. The company is singularly focused on helping its customers achieve clear value by leveraging quantum computing in practical business applications. With a full stack of systems, software, developer tools and services, D-Wave is working to enable enterprises, governments, developers and researchers to access the power of quantum computing, thereby providing an intriguing opportunity for prospective investors.
D-Wave’s current investor base includes PSP Investments, Goldman Sachs, BDC Capital, NEC Corporation, Aegis Group Partners and In-Q-Tel.
Leadership Team
Dr. Alan Baratz has served as the CEO of D-Wave since 2020. Previously, as Executive Vice President of R&D and Chief Product Officer, he drove the development, delivery, and support of all of D-Wave’s products, technologies, and applications. Dr. Baratz has over 25 years of experience in product development and bringing new products to market at leading technology companies and software startups. As the first president of JavaSoft at Sun Microsystems, he oversaw the growth and adoption of the Java platform from its infancy to a robust platform supporting mission-critical applications in nearly 80 percent of Fortune 1000 companies. He has also held executive positions at Symphony, Avaya, Cisco, and IBM. Dr. Baratz holds a doctorate in computer science from the Massachusetts Institute of Technology.
John Markovich is the company’s CFO. He brings to D-Wave over three decades of experience working with rapidly growing private and public technology companies across all stages of development. Mr. Markovich has directed the finance, accounting, tax, treasury, M&A, legal, operations, customer service, IR, HR, and IT functions for companies ranging from privately held pre-revenue startups to an NYSE-listed Fortune 500 multi-national company with over $1.2 billion in annual revenue. During his career, he has negotiated and closed over 150 debt, equity, M&A, and joint venture transactions exceeding $2.5 billion in value; over a dozen private placements; nearly a dozen M&A transactions; and several international joint ventures. Mr. Markovich holds a BS in Business from Miami University and an MBA from the Michigan State Graduate School of Business.
D-Wave Quantum Inc. (NYSE: QBTS), closed Monday's trading session at $7.05, off by 9.6154%, on 184,528 volume. The average volume for the last 3 months is 183,405 and the stock's 52-week low/high is $7.00/$13.23.
Recent News
- D-Wave Quantum Inc. (NYSE: QBTS) - BillionDollarBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Helping Companies Across Various Sectors Implement Quantum Computing Solutions
- D-Wave Quantum Inc. (NYSE: QBTS) Quantum Computing Technology Is Providing Solutions to Complex Problems Experienced Across Multiple Industries
- D-Wave Quantum Inc. (NYSE: QBTS) Launch(TM) Program Provides Onboarding Professional Services for Quantum Computing Across a Variety of Industries, including Manufacturing and Logistics, Financial Services, and Life Sciences
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
- Lexaria’s latest broadcast highlights the company’s progress in finding a potential solution for hypertension treatment while also pointing out why such a solution is much needed at this point
- Since the company began developing its patented DehydraTECH(TM) technology in 2014, it has made significant progress in its clinical studies, even as it currently focuses on human clinical research to evaluate the tolerability, safety, and efficacy of its solution for hypertension treatment
- Lexaria expressed its confidence in its proposed solution, noting that it will not only be a safer but also better-tolerated drug candidate, unlike current alternatives in the market whose side effects far outweigh their benefits
- The company looks to capitalize on the growing global anti-hypertensive drugs market, which is projected to hit $27.81 billion in value by 2025, and maintains that should it sustain the results achieved in recent hypertension studies, then it could be looking at mega-drug status ($1 billion per year) in the hypertension market
Lexaria Bioscience (NASDAQ: LEXX), a global innovator in drug delivery platforms, just released its latest addition to a growing body of corporate communication pieces in its line-up. Titled “The Power of Science to Stop a Killer,” this recent broadcast highlights the company’s progress in finding a potential solution for hypertension treatment, while also pointing out why a solution is much needed at this point (https://cnw.fm/6E6Dy).
Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing.
DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future.
Lexaria has a collaborative research agreement with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has filed for patent protection for specific delivery of nicotine, vitamins, NSAIDs, testosterone, estrogen, cannabinoids, terpenes, PDE5 inhibitors (with brand names like Viagra), tobacco and more.
Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats.
The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products.
DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions.
DehydraTECH Technology
Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following:
- Speeds up delivery – the effects of the product are felt by the subject in just minutes.
- Increases bioavailability – the technology is much more effective at delivering a drug or product into the bloodstream.
- Increases brain absorption – animal testing suggests significant improvement in the quantity of drug delivered across the blood-brain barrier.
- Improves drug potency – more of the ingested product is made available to the body, so lower doses are required to achieve the desired effect.
- Reduces drug administration cost – lower doses mean lower overall drug costs.
- Masks unwanted taste – the technology eliminates or reduces the need for sweeteners.
Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery.
Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine.
Market Outlook
Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments:
- Oral Cannabinoids – a market estimated to be worth $18.4 billion in 2021 and expected to reach $46.2 billion by 2025.
- Antivirals – an estimated $52.1 billion market in 2021 that’s expected to grow to $66.7 billion by 2025.
- Oral Mucosal Nicotine – smokeless tobacco products, a $13.6 billion market in 2018, is forecast to grow at 7.2 percent annually through 2025.
- Human Hormones – estrogen and testosterone replacement therapies represented a $21.9 billion market in 2019, with a forecast CAGR of 7.7 percent through 2027.
- Ibuprofen and Naproxen – NSAID sales totaled $15.6 billion globally in 2019 and are projected to reach $24.4 billion by 2027.
- Vitamin D3 – the global market size was $1.1 billion in 2021, growing at 7 percent per year and expected to reach $1.7 billion in 2026.
Management Team
Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations.
John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents.
Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations.
Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch.
Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia.
Lexaria Bioscience Corp. (LEXX), closed Monday's trading session at $2.99, off by 0.664452%, on 17,845 volume. The average volume for the last 3 months is 17,825 and the stock's 52-week low/high is $1.85/$7.20.
Recent News
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces the Availability of a Broadcast titled, "The Power of Science to Stop a Killer"
- Lexaria Bioscience Corp.'s (NASDAQ: LEXX) Drug Development Program and Recent FDA Confirmation Position It for Favorable Valuation
- CannabisNewsBreaks - Lexaria Bioscience Corp. (NASDAQ: LEXX) Stands to Gain Substantial Revenue Through Licensing of DehydraTECH(TM)
Cerberus Cyber Sentinel Corp. (NASDAQ: CISO)
The QualityStocks Daily Newsletter would like to spotlight Cerberus Cyber Sentinel Corp. (NASDAQ: CISO).
Cerberus Cyber Sentinel (NASDAQ: CISO), an industry leader as a managed cybersecurity and compliance provider, has selected InvestorBrandNetwork (“IBN”), a multifaceted financial news and publishing company for private and public entities, as its communications partner. Cerberus Sentinel has developed an integrated approach to help clients enjoy a simpler and more successful journey to cyber resilience. The company is rapidly expanding by acquiring world-class cybersecurity and compliance businesses with top-tier talent that offer the latest technology to create innovative protection solutions. IBN offers an investor-based distribution network of more than 5,000 syndication outlets along with newsletters, social media channels, wire services, blogs and other outreach tools all designed to create a greater awareness of its clients. “Cerberus Sentinel has invested in enterprise solutions and executive talent to integrate its different organizations. We see IBN as an important addition to our team as we move into the next level of cybersecurity and compliance leadership for our customers,” said Cerberus Sentinel founder and CEO David Jemmett in the press release. “Our ecosystem works together to provide complete cybersecurity through cross-pollination of solutions that begin at the network level and extend through technologies, people, policy, and practices. This ecosystem is intended to foster additional growth opportunities and drive overall recurring revenue. Once engaged, our company strives to become trusted advisors for our customers’ cybersecurity and compliance demands by providing tailored security solutions based upon their organizational needs.” To view the full press release, visit https://ibn.fm/M3vjD
Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) is an industry leader in cybersecurity and compliance services. The company leverages an integrated approach to reduce noise and bridge common silos that often limit the effectiveness of cybersecurity programs. Pulling disparate technologies, teams, and vendors together, Cerberus helps its clients enjoy a simpler and more successful journey to cyber resilience. Since 2019, Cerberus Sentinel has worked to rapidly expand by acquiring world-class cybersecurity and compliance businesses with top-tier talent who utilize the latest technology to create innovative protection solutions.
The Cerberus Sentinel workforce is comprised of cybersecurity experts spanning not only global geographies, but also specialties, industries, regulatory frameworks and focus areas. Its team includes audit and compliance specialists, certified forensics experts, ethical hackers, IEEE® certified biometric professionals, security engineers, around-the-clock analysts, and more – all backed by the most respected credentials in the industry. On an ongoing basis, the company works to identify cyber talent that is culturally aligned and that offers operating leverage through both existing customer revenue and relationships.
Cerberus Sentinel has invested in enterprise solutions and executive talent to integrate its different organizations into an ecosystem that works together to provide complete cybersecurity through cross-pollination of solutions that begin at the network level and extend through technologies, people, policy, and practices. This ecosystem is intended to foster additional growth opportunities and drive overall recurring revenue. Once engaged, the company strives to become trusted advisors for customers’ cybersecurity and compliance demands by providing tailored security solutions based upon their organizational needs.
While cyber resilience requires cycles of continuous improvement, it is a journey that few in the current business and security climate seem to understand. With its deep bench of seasoned experts, Cerberus Sentinel works to simplify that journey for its growing customer base, straightening out the curves and speeding up the process to resilience along the way.
Cybersecurity is a Culture, Not a Product
Integrating compliance and security, including principles of security by design, Cerberus Sentinel helps its clients create an organization-wide culture of cybersecurity. Its offerings include audit and compliance, security operations center services, security engineering, virtual Chief Information Security Officer services, incident response, certified forensics, technical assessments and cybersecurity training.
In contrast to the majority of cybersecurity firms that specialize in a specific technology or service, Cerberus Sentinel seeks to differentiate itself by remaining technology agnostic, focusing on accumulating highly sought-after subject matter experts. Cerberus Sentinel believes that bringing together a world-class team of technological experts with multi-faceted proficiency in the critical aspects of cybersecurity is key to providing technology agnostic solutions to its clients in a business ecosystem that suffers from a chronic lack of highly skilled professionals.
Cerberus Sentinel’s goal is to create a culture of security and to help quantify, define and capture a return on investment from information technology and cybersecurity spending. Its end-to-end, holistic process covers every aspect of clients’ cybersecurity and compliance requirements in an effort to promote greater efficiency and strengthen awareness about the integral role of internal team members in the cybersecurity culture of an organization.
As a result of this strategy, Cerberus Sentinel customers receive an efficient engagement from a single partner that covers a wide range of their needs – addressing challenges more thoroughly and resolving problems more rapidly when compared to working with a host of vendors.
Market Outlook
According to an analysis by the firm Research and Markets, the global managed security services market was valued at $22.45 billion in 2020 and is projected to reach $77.01 billion by 2030, growing at a CAGR of 12.8% through the forecast period.
An expected increase in cybercrime, cost effectiveness of provided solutions and stringent mandatory government regulations aimed at protecting corporate data will drive the global managed security services market for the foreseeable future.
In addition, the documented and growing use of mobile devices in the workplace and the rise in captured and stored digital data serve to fuel market growth. Moreover, growing awareness about the critical nature of data security, the growing importance of e-business and demand for customized services is expected to offer ample opportunities for expansion of the market during the forecast period.
Management Team
David Jemmett is CEO and founder of Cerberus Sentinel. He has more than 35 years of executive management and technology experience with telecommunications, managed services, and cybersecurity consulting services. He previously held positions as CEO of GenResults, a leading provider of security consulting services and technology solutions, and as CTO and founder at ClearData Networks, a HIPAA-compliant HealthDATA cloud hosting platform.
Dave Bennett is COO at Cerberus Sentinel. Since 2015, he has served on the President’s STEM Advisory Board of Grand Canyon University. Before joining Cerberus Sentinel, he served as Chief Product Officer at Experian Health and as Senior Vice President, Product for Gainwell Technologies. He has also held positions as Vice President and Worldwide Head of Build, Healthcare and Life Sciences at DXC Technology, and as EVP, Product and Strategy at Orion Health.
Ashley Devoto is President and Chief Information Security Officer at Cerberus Sentinel. Over the past 17 years, Devoto has worked with the cybersecurity elite to design, build, and operate world-class cybersecurity programs for large, diverse organizations in both government and commercial enterprises. Prior to joining Cerberus, Devoto served as CISO for Booz Allen Hamilton, as business information security officer (BISO) at Bank of America, and as a cyberspace operations officer in the United States Air Force.
Deb Smith is CFO at Cerberus Sentinel. Prior to assuming that position, she was the company’s EVP, Finance and Accounting. She has also served as SVP, Global Accounting at International Cruise and Excursions Inc., and as Chief Accounting Officer for BeyondTrust, an information security software company. She has also held the positions of Corporate Controller at Aspect Software and Assistant Controller at JDA Software.
Cerberus Cyber Sentinel Corp. (NASDAQ: CISO), closed Monday's trading session at $2.66, off by 5.3381%, on 151,876 volume. The average volume for the last 3 months is 151,876 and the stock's 52-week low/high is $2.61/$49.00.
Recent News
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Partners with IBN for Corporate Communications
- InvestorNewsBreaks - Cerberus Cyber Sentinel Corporation (NASDAQ: CISO) Acquires Santiago, Chile-Based CUATROi
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Is 'One to Watch'
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
- CNS Pharmaceuticals is currently undertaking a potentially pivotal global study evaluating the efficacy and safety of Berubicin compared with Lomustine administered after first-line therapy for the treatment of recurrent glioblastoma multiforme (“GBM”)
- GBM is an aggressive form of cancer that occurs in the brain or spinal cord
- In the U.S., it affects between 12,000-15,000 new patients annually with a median survival rate of only 14.6 months from its diagnosis
- CNS’s drug development program is targeting the GBM drug market which is expected to expand at a CAGR of more than 4% between 2020 and 2030
On July 24, 2021, pre-clinical stage biotechnology company CNS Pharmaceuticals (NASDAQ: CNSP) received Fast Track Designation from the U.S. Food and Drug Administration (“FDA”) for its Berubicin drug product (https://ibn.fm/fFjF8). Granted to help facilitate the development and expedite the review of drugs being developed to treat serious conditions and fill unmet medical needs, the designation showed the seriousness of Glioblastoma Multiforme (“GBM”), an aggressive form of cancer that occurs in the brain or spinal cord. The Fast Track status came just over a year after the company had received Orphan Drug Designation (“ODD”), which is intended to support the development and review of novel treatments for rare diseases.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Monday's trading session at $0.2521, off by 2.702%, on 99,326 volume. The average volume for the last 3 months is 99,070 and the stock's 52-week low/high is $0.20/$1.70.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Looking to Fill Glioblastoma Multiforme Treatment Gap with Berubicin Drug Product
- Study Looks into Why Deadly Brain Tumors Continue to Grow After Treatment
- BioMedNewsBreaks - CNS Pharmaceuticals Inc. (NASDAQ: CNSP) Continues Priority to Advance Clinical Development Program for Berubicin
Aditxt Inc. (NASDAQ: ADTX)
The QualityStocks Daily Newsletter would like to spotlight Aditxt Inc. (NASDAQ: ADTX).
A new study has found that pediatric patients who undergo kidney transplants may have better outcomes if the donated kidney comes from a biologically unrelated and living donor in comparison to a deceased donor. For their research, the scientists reviewed data obtained from the Organ Procurement and Transplantation Network database during the period between January 2001 and September 2021. The investigators examined more than 12,000 children who had received kidney transplants during the study period. Of the total number of children, 61% received kidneys from deceased donors, 36% received kidneys from living related donors and less than 3% received kidneys from a living unrelated donor. The majority of the living related donors were parents and other relatives. Several companies, including Aditxt Inc. (NASDAQ: ADTX), are studying various approaches, such as modulating immune system function, with the goal of decreasing the rates of transplant organ rejections and helping patients enjoy a better quality of life with the new organs they have received.
Aditxt Inc. (NASDAQ: ADTX) is a biotech innovation company developing technologies focused on mapping and reprogramming the immune system. Aditxt’s immune mapping technologies are designed to provide a personalized immune profile. Aditxt’s immune reprogramming technologies, currently preclinical, are being developed to retrain the immune system to induce tolerance to address rejection of transplanted organs, autoimmune diseases, and allergies.
As further discussed below, the company’s first commercial product is an immune mapping technology, AditxtScore™, which is designed to provide a personalized profile of the immune system.
The company’s preclinical immune reprogramming technology, Apoptotic DNA Immunotherapy™ (“ADi™”), aims to retrain the immune system to induce tolerance, with the goal of addressing vast unmet needs in transplanted organ rejection, autoimmune diseases, and allergies. The company is developing specific ADi™ products for psoriasis, type 1 diabetes, and skin grafting.
Headquartered in Richmond, Virginia, Aditxt also operates locations in Silicon Valley and New York.
AditxtScore™
AditxtScore™ is a proprietary platform designed to provide a personalized, comprehensive profile of an individual’s immune system. The underlying technology, licensed from Stanford University through an exclusive worldwide agreement, offers a highly sensitive and accurate method of detecting and quantifying cellular responses, allowing greater specificity, quantification, and amplification of both clinical and commercial opportunities.
The company’s first commercial application of the platform, AditxtScore™ for COVID-19, delivers timely reports on vulnerability and immune status relating to SARS-CoV-2 and its known variants, giving consumers and physicians the data needed to make informed health decisions. Potential future applications will offer early detection of an array of conditions, including diabetes, cardio-metabolic maladies and hormonal imbalances.
Aditxt’s AditxtScore™ immune monitoring center in Richmond, Virginia, is operational and designed to support the anticipated increased demand for AditxtScore™ as well as related products and services. The company is currently scaling its capabilities at this location, with a goal of processing up to 10 million immune system tests/reports annually.
ADi™
ADi™ is Aditxt’s immune reprogramming platform addressing disease-causing immune responses while maintaining the immune system’s ability to combat pathogenic infection. The company is commercializing a nucleic acid-based technology called Apoptotic DNA Immunotherapy™ (ADi™) which utilizes a novel approach that mimics the way our bodies naturally induce tolerance to our own tissues (therapeutically induced immune tolerance). Aditxt believes its ADi™ technology platform can be engineered to address a wide variety of indications.
Aditxt is currently developing ADi™ products for psoriasis, type 1 diabetes and skin grafting.
Currently, immuno-tolerance is achievable through chimerism and cell-based therapy, but there is a clinical need for a more practical and cost-effective approach which:
- Can be made into a product
- Does not require additional hospitalization
- Is simple to produce and ship
Preclinical studies have demonstrated that ADi™ treatment significantly and substantially prolongs graft survival, in addition to successfully “reversing” other established immune-mediated inflammatory processes. ADi™ treatment is not expected to require hospitalization, instead being delivered as an injection in minute amounts into the skin.
IP Portfolio
Both AditxtScore™ and ADi™ are supported by a strong IP portfolio.
AditxtScore™, built upon initial technology invented, licensed from and used at Stanford University, is protected by U.S. patents encompassing methods, systems, and kits for detection and measurement of specific immune responses.
ADi™ technology is protected by seven patent families, including:
- 8 U.S. patents
- 4 pending U.S. patent applications
- 86 foreign patents and 14 pending foreign patent applications spanning the EU, Australia, Canada, Japan, China, India and Hong Kong
These patents are broadly categorized into three groups:
- Autoimmune diseases and Type 1 Diabetes
- Organ transplantation and a method of producing plasmid DNA to prevent immune activation
- Composition of matter for a tolerance delivery system for antigens of interest
Aditxt also possesses and/or in-licenses substantial know-how and trade secrets relating to the development and commercialization of its product candidates, including related manufacturing processes and technologies.
Market Overview
The potential market opportunities presented by immune monitoring and reprogramming are extensive, particularly as Aditxt continues to evaluate additional applications for the platforms.
The company’s initial focus on organ transplantation and related autoimmune response provides some insight into the potential of its approach. According to BCC Research, the global organ and tissue transplantation and alternatives market is on course to reach $120.3 billion by 2024, recording a CAGR of 7.4% from 2019. Industry data suggest that approximately 50% of all transplanted organs are rejected within 10-12 years, further highlighting the critical need for a practical, cost-effective solution to harmful autoimmune responses.
Through its focus on the COVID-19 testing market with AditxtScore™, Aditxt demonstrated the wide-ranging potential of its portfolio. Fortune Business Insights estimated the global COVID-19 diagnostics market at $48.64 billion for 2022. While demand for COVID-19 diagnostics is expected to lessen in the coming years, Aditxt will be uniquely positioned to leverage its existing infrastructure stemming from these operations as the company works to advance broader applications for the AditxtScore™ platform.
Leadership Team
Amro Albanna is the Co-Founder, Chairman, and CEO of Aditxt. He has founded multiple startups to commercialize innovations in various industries, including healthcare, enterprise software, telecommunications, nano technology, consumer health, and biotech. Mr. Albanna has led numerous M&A and going-public transactions as a founder, co-founder, and senior executive.
Shahrokh Shabahang, D.D.S., MS, Ph.D., is the company’s Co-Founder, Chief Innovation Officer, and a member of its board. He brings to the team more than 20 years of experience in developing and commercializing life science technologies focused on product and clinical development in the fields of microbiology and immunology.
Corinne Pankovcin, CPA, MBA, is the President of Aditxt. Prior to joining Aditxt, Ms. Pankovcin served as CFO for several world class organizations, including Business Development Corporation of America, Blackrock Kelso Capital and AIG Capital Partners. In these roles, Ms. Pankovcin was responsible for executing portfolio investments and managing significant M&A transactions.
Thomas Farley is the Chief Financial Officer of Aditxt. From December 2015 to June 2020, Mr. Farley was the Controller and Treasurer of Business Development Corporation of America (“BDCA”), a publicly listed business development company. Prior thereto, from January 2011 to August 2015, Mr. Farley was the Senior Controller of Blackrock Capital Investment Corporation (NASDAQ: BKCC). Prior to joining BlackRock Capital Investment Corporation, Mr. Farley was a Senior Controller for PineBridge Investments Emerging Markets practice. Mr. Farley was also an Accounting Manager for Bessemer Venture Partners prior to his tenue at PineBridge. Mr. Farley began his career with PricewaterhouseCoopers LLP, from 1996 to 2001. Mr. Farley earned his B.S. in Accounting from Long Island University and is a Certified Public Accountant.
Rowena Albanna is the company’s Chief Operating Officer. Ms. Albanna has over two decades of experience in senior leadership roles for both technology startups and public companies. Ms. Albanna’s experience spans a wide variety of industries, including biotechnology, insect control, nanotechnology, consumer electronics, financials, telecommunications, e-commerce, online marketing, medical, and defense.
Matthew Shatzkes is the Chief Legal Officer and General Counsel of Aditxt. As a former partner at an AM Law 50 law firm, Mr. Shatzkes advised a wide variety of healthcare related entities, including biotech companies, on corporate, regulatory, and strategic business matters. Mr. Shatzkes will oversee all aspects of the legal functions at Aditxt, including, providing advice and counsel on governance, regulatory matters, strategic alliances, mergers and acquisitions, and commercial transactions.
Aditxt Inc. (NASDAQ: ADTX), closed Monday's trading session at $0.154, off by 1.2821%, on 420,441 volume. The average volume for the last 3 months is 420,441 and the stock's 52-week low/high is $0.1242/$2.72.
Recent News
- Aditxt Inc. (NASDAQ: ADTX) - Study Finds Children Have Better Outcomes with Kidney Transplants from Living Donors
- Study Finds That Early Exposure to Antibiotics May Trigger Asthma, Permanent Allergies
- Senate Review Reveals Screening Mistakes Caused Organ Transplant Failures, Illnesses, Deaths
Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F)
The QualityStocks Daily Newsletter would like to spotlight Hillcrest Energy Technologies Ltd. (OTCQB: HLRTF).
- Electric vehicle charging infrastructure can be notorious for its efficiency losses, with onboard car chargers habitually losing 14% or more of the energy put into the vehicle
- Hillcrest Energy Technologies has looked to address that issue through the release of their simplified EV charger solution
- The traction inverter market is projected to reach $7 billion by 2025 at a CAGR of 17.57% due to increased demand for EV’s
Electric vehicle (“EV”) owners are currently living through a scenario not entirely dissimilar to that being borne by their internal combustion engine vehicle driving counterparts. Over the past year, several car-charging operations have shifted away from EV owners paying per charging session, rather opting to have customer pay per kWh of energy input. However, and unlike gasoline-powered vehicles, paying per unit of electric power for an EV can be confounding. In one test, an EV using a commercial charging point was revealed to be taking on 27.83 kW – despite the battery pack of the vehicle boasting a total capacity of 27 kWh and still possessing a slight level of charge (https://ibn.fm/34S25). The key difference comes down to efficiency losses, a paradigm which Hillcrest Energy Technologies (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F), a clean technology company developing transformative power conversion technologies, seeks to address.
Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) is a clean technology company based in Vancouver, British Columbia, engaged in developing high-value, high-performance power conversion technologies and digital control systems for next-generation powertrains and grid-connected renewable energy systems.
From concept to commercialization, Hillcrest invests in the development of energy solutions that power a more sustainable and electrified future. Hillcrest power inverter technology helps produce efficiencies in electrification and maximize the performance of electric systems, including electric vehicles (EV), motors and generators.
The company offers a flexible, single-inverter architecture that can be applied at nearly every stage of the electrification ecosystem, from renewable energy generation through the charging and operation of an EV, to provide full-cycle efficiency and performance improvements.
As momentum to electrify and decarbonize energy systems accelerates, Hillcrest believes the power inverter is increasingly emerging as a key component. While system cohorts such as battery packs, PV panels and electric motors are often in the spotlight, the inverter holds the key to unlocking efficiency and performance improvements.
Hillcrest power inverter technology is:
- REVOLUTIONARY: high-efficiency inverter technology has the potential to revolutionize how motors respond and how efficiency is gained.
- AGILE: able to deliver and deploy high-efficiency inverter solutions purpose-designed to meet specific customer needs.
- INNOVATIVE: technology-forward, clean-energy experts who are focused on advancing and optimizing efficient alternative energy use across all electric vehicle and charging platforms.
- A MARKET LEADER: a next-generation technology provider to the automotive industry’s top suppliers and manufacturers.
Technology & Applications
Hillcrest’s first application for its inverter technology – a 250 kW|800V Hillcrest SiC high efficiency traction inverter – is focused on the growing EV market. Hillcrest technology eliminates traditional design trade-offs faced across the power industry – deploying higher switching frequencies has historically meant a greater increase in losses, lower system efficiency and higher heat. Through a combination of hardware and software expertise, Hillcrest enables power applications to leverage higher switching frequencies AND
- Realize improved power system performance and reliability
- Operate at higher power levels without compromising efficiency
The expected benefits of Hillcrest’s traction inverter have been confirmed via testing and shared in a technical white paper, published in April 2022, that confirmed the following results:
- Significant efficiency gains – 99%-plus inverter efficiency
- Increased power density targeting 50kW/L+
- Significantly increased motor efficiency
- Lower stress on mechanical and electrical parts, enhancing reliability
- Improved thermal management
Hillcrest has also filed a patent for an enhanced powertrain solution that offers the potential to simplify EV charging and redefine how the industry envisions charging infrastructure. The company believes the most exciting benefit of the enhanced powertrain solution is the ability to eliminate the onboard charger and booster from an EV, as well as faster, anywhere charging including direct DC, wireless, and bidirectional charging across current and future power levels. Hillcrest sees this as a true EV charging game changer.
The company’s technology applies to nearly every clean energy industry segment:
- Wind power – an inverter is deployed at a wind turbine generator to convert the AC output, with at least one additional inverter used to deliver the power to the grid/battery.
- Solar power – an inverter is used to convert the DC output from the photovoltaic panels into the AC power that flows to the grid/battery/home.
- Energy storage – an inverter is deployed to convert the DC output from the storage system or batteries to the AC power that flows to the grid/home/EV.
- EV fast chargers – an inverter converts the AC input from the grid/storage system to the DC output needed to charge an EV’s battery.
Market Outlook
According to an April 2022 market analysis by Vantage Market Research (VMR), the global power inverter market is expected to reach a value of $95 billion by 2028, driven by increasing demand for EVs, energy generating wind turbines and solar-powered photovoltaic systems. That jump is forecast from an estimated $70.5 billion market value in 2021 and represents a compound annual growth rate of more than 5%.
According to the VMR report, many governments in countries around the world are supporting alternative options for efficient and nonpolluting energy generation. This has boosted demand for wind energy and solar energy systems. Hillcrest is aiming to capture a share of this future market growth across nearly every segment of the clean energy industry.
Management Team
Don Currie is the founding CEO of Hillcrest Energy Technologies. He has led the company’s successful transition from fossil fuels into clean energy technologies. Earlier in his career, he held various senior level positions, including director, officer and vice president of corporate communications with Enhanced Oil Resources Inc., an oil and gas exploration and production company based in Houston. Prior to that, he worked in other private and public ventures spanning the mining, gaming and technology sectors.
Jamie L. Hogue is the COO of Hillcrest. She brings more than two decades of progressive policy leadership, economic analysis and organizational development experience to Hillcrest. She builds collaborative processes and solutions that drive growing organizations toward a more resilient future. She previously served as the director of operations for Arizona State University’s Ten Across initiative – a compelling observatory positioned on the front lines of economic, social and climate change. She earned a master’s degree in public administration and a bachelor’s degree in economics from Arizona State University.
Ari Berger is Chief Technology Officer at Hillcrest. He brings over a decade of commercial experience with a track record of deploying new electrification technologies and go-to-market strategies. In 2015, he founded NIG Systems Ltd. in Israel, which specializes in custom high performance control systems design. Prior to this, he previously worked for Bental Industries, a leading motor manufacturer. He holds a master’s degree in system control engineering from the Technion – Israel Institute of Technology.
Raj Clair is CFO at Hillcrest. She is a CPA who began her career at Deloitte and has served in advanced finance positions in the energy and resources sector. She has been responsible for reporting, audits and internal controls, as well as working on budgeting and forecasting. She has worked with various publicly listed companies, including SEC registrants, and has both Canadian and U.S. experience. She holds a bachelor’s degree in accounting from Simon Fraser University.
Hillcrest Energy Technologies Ltd. (NASDAQ: HLRTF), closed Monday's trading session at $0.088, off by 1.8952%, on 7,600 volume. The average volume for the last 3 months is 7,600 and the stock's 52-week low/high is $0.075/$0.1826.
Recent News
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Seeks to Address Electric Vehicle Energy Efficiency Issues Through Proprietary Powertrain Solution
- Hillcrest Energy Technologies Ltd.'s (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Power Inverter Platform Boosts Performance and Efficiency, Enables Multiple End-Uses
- GreenEnergyBreaks - Hillcrest Energy Technologies Ltd.'s (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Inverter Combining Innovation to Improve System Performance, Reliability
Cannabis Strategic Ventures Inc. (OTC: NUGS)
The QualityStocks Daily Newsletter would like to spotlight Cannabis Strategic Ventures Inc. (NUGS).
Over the past couple of years, a surge in cannabis research has revealed that the controversial plant has a variety of medical applications. Despite being classified as a Schedule I drug with no medical application, studies have found that cannabis can be effective at alleviating conditions such as chronic pain, depression and insomnia. Furthermore, the U.S. Food and Drug Administration has also approved a CBD-based drug called Epidiolex to treat seizures in pediatric epilepsy. In recent months, research into the impact of cannabis on cancer treatment has found that the plant can have a positive impact on cancer patients. Cancer is one of the deadliest killers in the country, taking more than 500,000 lives per year and costing billions of dollars for treatment. Although there are treatments that can stop cancers from growing and significantly increase patients’ quality of life, no cure for cancer currently exists. Researchers concluded that medical cannabis seems to play a role in alleviating symptoms of advanced cancer with minimal side effects and called for more research to determine optimal delivery methods and doses for patients. In this regard, industry players such as Cannabis Strategic Ventures Inc. (OTC: NUGS) are leading the way by coming up with different innovations that optimize consumers’ experience while using marijuana products.
Cannabis Strategic Ventures Inc. (OTC: NUGS) is an emerging leader in the U.S. cannabis marketplace as a publicly traded cannabis cultivator. The company is based in Los Angeles, with a 6-acre cannabis farm in Northern California called NUGS Farm North. The company’s vision is to acquire and scale assets in the legal cannabis market while achieving efficiencies through economies of scale and vertical integration.
Cannabis Strategic Ventures recently expanded its portfolio by completing the transfer process for cultivation, retail, distribution and manufacturing licenses issued by the City of Los Angeles and the State of California, and it is now working toward taking operational control of each license. The company also recently announced the upcoming grand opening of its cannabis dispensary, MDRN Tree. Following that launch, Cannabis Strategic Ventures intends to deploy another of its new licenses to establish an indoor cultivation facility with capacity to produce two to three pounds of premium exotic cannabis flower per light per harvest. The facility will have up to 1,200 grow lights and is anticipated to yield 5.75 harvests per year, bringing it to a total production capacity of over 15,000 pounds of cannabis flower annually.
Brand Portfolio
The company owns multiple brands under the Cannabis Strategic Ventures umbrella. The firm’s NUGS brand provides operational and financial strategic partnerships and a range of essential services to emerging and existing cannabis consumer brands.
The NUGS Farm North brand operates as a six-and-a-half-acre cannabis cultivation property located in northern California. The company believes that the key to success in its business is consistent quality and reliable supply to fit growing consumer demand. Cannabis Strategic Ventures addressed these consumer needs by building NUGS Farm North. At NUGS Farm North, the company’s process is customized, and its product is consistent. Located in the heart of an agricultural mecca for globally distributed produce, NUGS Farm North finds power in its product, not in its size. Decades of agricultural experience and a dedication to consistency ensure quality cannabis.
MDRN Tree is Cannabis Strategic Ventures’ customer-facing dispensary brand. MDRN Tree will open its first Los Angeles location sometime in the fall of 2021. MDRN Tree will be the company’s factory retail store – a direct interface with the end-market community – where Cannabis Strategic Ventures plans on showcasing the cannabis flower produced at its NUGS Farm North cultivation site. This farm-to-sale model offers the potential to drive simultaneous gains in quality control and profitability.
Market Outlook
The demand for legal marijuana is expected to surge due to ongoing changes in U.S. state government policies toward cannabis. In addition, the number of indications for which medical marijuana is prescribed continues to increase steadily. These factors are expected to rapidly boost legal sales of cannabis products, opening new revenue channels for producers and retailers. Furthermore, an anticipated federal legalization of medical marijuana in the U.S. will only present more high growth opportunities for this market.
According to a report from Grand View Research, the global legal marijuana market was valued at $9.1 billion in 2020. Market size is forecast to grow at a compound annual growth rate of 26.7 percent from 2021 to 2028. That CAGR would put the market value at roughly $30 billion as soon as 2025.
According to the report, “One of the major factors fueling market growth is the expanding demand for legal marijuana owing to the growing number of legal cannabis countries. (Due) to recent legalizations in different countries, the use of medical marijuana for various ailments is gaining momentum worldwide. Patients suffering from chronic illnesses such as Parkinson’s, cancer, Alzheimer’s, and many neurological disorders are administered medical marijuana. The demand for cannabis oil is increasing rapidly, especially among countries with legalized medical marijuana.”
Management Team
Simon Yu is CEO, President, CFO and Secretary of Cannabis Strategic Ventures. He is also a co-founder, former COO and board member of Clubhouse Media Group Inc., a publicly traded social media company. Mr. Yu holds an MBA from the University of Southern California.
Cannabis Strategic Ventures Inc. (NUGS), closed Monday's trading session at $0.01283, off by 4.963%, on 641,018 volume. The average volume for the last 3 months is 641,018 and the stock's 52-week low/high is $0.0099/$0.058.
Recent News
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - Recent Studies Show Advanced Cancer Sufferers Could Benefit from Using Marijuana
- 420 with CNW - Gallup Poll Finds Americans Think Cannabis Better than Alcohol for Society
- 420 with CNW - New York to Start Receiving Social Equity Cannabis Business Applications
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Enrolls First Patient in Early-Stage Frozen Shoulder Trial
- Aditxt Inc. (NASDAQ: ADTX) - Study Finds Children Have Better Outcomes with Kidney Transplants from Living Donors
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Mothers in Indonesia Fight for Their Children to Have Access to Medical Cannabis
- American Cannabis Partners - 420 with CNW — How Missouri Activists Overcame Setbacks to Put Cannabis Measure on Upcoming Ballot
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Announces $2.3M Registered Direct Offering
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - Today's Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on MondayMore From InvestorPlace
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- AREV Life Sciences Global Corp. (CSE: AREV) (OTC: AREVF) - SUSTAINN, AREV's Ready to Use Therapeutic Foods discussed in leading article in HealthHIV
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix to Present at the H.C. Wainwright 24th Annual Global Investment Conference
- Avricore Health Inc. (TSX.V: AVCR) (OTCQB: AVCRF) - InvestorNewsBreaks - Avricore Health Inc. (TSX.V: AVCR) (OTCQB: AVCRF) Releases Q2 2022 Report
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - BevCanna Enterprises Inc.'s (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Journey: From Runners' Knee to Canada's 101 Top F&B Start-up List
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Enters MOU to Acquire 100% Equity of Shengruihao
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Secures CE Mark Approval for NeuroCap(TM)
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - Recent Studies Show Advanced Cancer Sufferers Could Benefit from Using Marijuana
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Partners with IBN for Corporate Communications
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Looking to Fill Glioblastoma Multiforme Treatment Gap with Berubicin Drug Product
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Helps Companies Boost Profitability Through Energy Improvements as ESG Gains Weight
- Cub Crafters Inc. - InvestorNewsBreaks – Cub Crafters Inc. Looking to Expand Operations Amid Increasing Popularity of Backcountry Aviation
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Begins Dosing Participants in CYB003 Trial for Treatment of MDD
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Q2 2022 Financial Report, Corporate Update
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - BillionDollarBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Helping Companies Across Various Sectors Implement Quantum Computing Solutions
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - DEA Delay Leaves Synthetic Psychedelics in Legal Limbo
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Secures ICON Capital Group as Financial Advisor to Pursue Strategic Spinout
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Supplying Options Worldwide to Improve Food Security
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - MiningNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) on Target to Own, Control 'the Elements' to Supply EV, Renewable Energy, Other Technology Manufacturers
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - Exro Announces a Public and Private Financing of up to approximately $10mm
- FingerMotion Inc. (NASDAQ: FNGR) - ChineseNewsBreaks - FingerMotion Inc.'s (NASDAQ: FNGR) Relationship with China's Largest Telcos Providing 'Strong Footing' in Market
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Options Two Lithium Claims Directly Adjacent to 'Electric Avenue'
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Experts Say Federal Court Ruling in Maine Could Pave Way for Interstate Cannabis Commerce
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Releases Q2 2022 Financial, Corporate Report
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Streamlining NAFTA Region's Cross-Border Shipping Process
- Friendable Inc. (FDBL) - Friendable Inc. (FDBL) Addresses Churn in the Music Streaming Industry by Offering Fan Pass Live and Branded Products to Enhance Listener Experience
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Announces Filing of Quarterly Report and Provides Early Pre-Sales Update
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc.’s SmartGreen(TM) Tech Transforms Homes, Enables Energy Independence
- Golden Matrix Group Inc. (NASDAQ: GMGI) - Golden Matrix Group Inc. (NASDAQ: GMGI) Scores Big Q3 Win on International Expansion
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH), Electryone Advisors Expand Partnership with Advancement of Exceptional Technology
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Secures New Patent, Critical Protection for REQORSA(TM) Combination Therapy
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- GreenBox POS (NASDAQ: GBOX) - InvestorNewsBreaks - GreenBox POS (NASDAQ: GBOX) Announces Restructuring of Convertible Note Financing Terms
- Cepton Inc. (NASDAQ: CPTN) - Cepton, Inc. Announces New Investment Intent for Up To $100 Million from Koito Manufacturing
- Hemptown USA - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Common Stock Upgraded to Mid-Tier OTC Equity Market
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) Reports on Growth Potential of US Cannabis Markets
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Seeks to Address Electric Vehicle Energy Efficiency Issues Through Proprietary Powertrain Solution
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Unveils Vegan + Sustainable Line from Celebrity Chef Priyanka
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Q2 2022 Unaudited Financial Results
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) to Participate in Energica Week 2022
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd.'s (NASDAQ: IFBD) Shanghai Qishuo to Leverage 'Retail Cube' Solution for World-Renowned Brand
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InMed Announces Share Consolidation to Meet Nasdaq Listing Criteria
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Notes FDA Final Rule on OTC Hearing Aids
- Innovative Payment Solutions Inc. (OTCQB: IPSI) - Innovative Payment Solutions, Inc. Appoints Richard Rosenblum as President and Chief Financial Officer
- BlockQuarry Corp. (OTC: BLQC) - Cryptocurrencies Appear to Have Seen the Worst of Crypto Winter
- Knightscope (NASDAQ: KSCP) - Oregon Healthcare Provider is Latest Knightscope (Nasdaq: KSCP) Client
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - Laredo Oil Inc. (LRDC) Progressing Toward Drilling as 2023 Oil Demand Projected to Increase
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces the Availability of a Broadcast titled, "The Power of Science to Stop a Killer"
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Releases Q1 Financial, Operational Results
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - InvestorNewsBreaks - LQwD Fintech Corp. (TSX.V: LQWD) (OTC: LQWDF) Releases Results from Annual General Meeting
- LQwD Announces Annual General Meeting Voting Results
- CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging LN to Enable Faster, More Secure Micropayments
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Third Quarter of Fiscal 2022
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Enters Common Share Subscription Agreement
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Releases Q1 2023 Report, Notes Purchase Agreement Highlight
- Mullen Automotive Inc. (NASDAQ: MULN) - GreenCarNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Positioned 'Front and Center for Last-Mile Delivery Opportunities'
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (OTCQB: NEXCF) (NEO: NTAR) (CSE: NTAR) (FSE: N29) CEO Talks ARway News in Power Play Interview
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - InvestorNewsBreaks - Odyssey Health Inc. (ODYY) Featured in NeurologyLive(R) Article
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - PlantX Conducts Additional Live Retail Events, Driving Store Traffic and Increasing Sales Velocity
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Releases Q2 2022 Financial Report
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Poised for US Expansion Due to 'Attractive Incentives to Build Hydrogen Infrastructure'
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 Financial Report
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Partners with Skincare Leader, Icon and Pioneer
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Subsidiary Receives Adult-Use Edibles Sales, Expansion Rights
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Names New Key Board Appointments
- QSAM Biosciences Inc. (OTCQB: QSAM) - BioMedNewsBreaks - QSAM Biosciences Inc. (QSAM) Receives 'Marketable Advantage' Toward Manufacturing, Supply Chain of CycloSam(R)
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Red White & Bloom Reports Financial Results for Second Quarter 2022 and Six Months Ended June 30th 2022
- REZYFi, Inc. - REZYFi, Inc. is "One to Watch"
- RYAH Group Inc. (CSE: RYAH) - InvestorNewsBreaks - RYAH Group Inc. (CSE: RYAH) Names New CFO, Corporate Secretary
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation (SNWR) Subsidiary Releases Q2 Financial Results, Corporate Update
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Receives Notice of Patent Allowance for Tech Designed to Protect Produce from Decay
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Has Created a Community, Distinction to Build Powerful Brand
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions (NASDAQ: SASI) Announces Q2 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Psychedelics Company Raises More than $10M in Seed Funding to Advance Ketamine Therapy
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Ramps up Sales for SD7, Companion Devices Across Multiple Verticals
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc.'s (NYSE American: SBEV) Selected for 7-Eleven's Highly Competitive Brands with Heart Campaign
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - InvestorNewsBreaks - SRAX Inc. (NASDAQ: SRAX) Announces Entry into Revolving Credit Facility, Launch of Stock Portfolio Tracker
- Standard Lithium Ltd. (NYSE American: SLI) - Bullish Sentiments Continue for Global Lithium Mining Industry as Demand Soars
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Subsidiary Inks MSA for Manufacturing, Distribution Expansion
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Secures Purchase Renewal Contract with Louisville Division of The Kroger Company (NYSE: KR)
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT(R) Global Alternatives (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Announces UK Distributor Places Third Reorder
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Expands Akaline88(R) to 200+ Midwest, Mid-Atlantic Stores
- Tingo Inc. (OTC: TMNA) - World Bank Affiliate Looks to Blockchain to Facilitate Carbon Trading
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Expands IP Portfolio with New Provisional Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Launches TRxADE Prime Plus to Meet Customers' Needs of On-Demand Inventory
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Completes Acquisition to Create Largest Diversified North American Focused Uranium Company
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - 4 Cheap Robinhood Stocks To Buy According To Insiders In August 2022
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Extension of LAPD BolaWrap Pilot Program
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - InvestorNewsBreaks - XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) Exploring Expanded Application of ODF Screening Tests
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Enrolls First Patient in Early-Stage Frozen Shoulder Trial
- Aditxt Inc. (NASDAQ: ADTX) - Study Finds Children Have Better Outcomes with Kidney Transplants from Living Donors
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Mothers in Indonesia Fight for Their Children to Have Access to Medical Cannabis
- American Cannabis Partners - 420 with CNW — How Missouri Activists Overcame Setbacks to Put Cannabis Measure on Upcoming Ballot
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Announces $2.3M Registered Direct Offering
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - Today's Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on MondayMore From InvestorPlace
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- AREV Life Sciences Global Corp. (CSE: AREV) (OTC: AREVF) - SUSTAINN, AREV's Ready to Use Therapeutic Foods discussed in leading article in HealthHIV
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix to Present at the H.C. Wainwright 24th Annual Global Investment Conference
- Avricore Health Inc. (TSX.V: AVCR) (OTCQB: AVCRF) - InvestorNewsBreaks - Avricore Health Inc. (TSX.V: AVCR) (OTCQB: AVCRF) Releases Q2 2022 Report
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - BevCanna Enterprises Inc.'s (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Journey: From Runners' Knee to Canada's 101 Top F&B Start-up List
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Enters MOU to Acquire 100% Equity of Shengruihao
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Secures CE Mark Approval for NeuroCap(TM)
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - Recent Studies Show Advanced Cancer Sufferers Could Benefit from Using Marijuana
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Partners with IBN for Corporate Communications
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Looking to Fill Glioblastoma Multiforme Treatment Gap with Berubicin Drug Product
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Helps Companies Boost Profitability Through Energy Improvements as ESG Gains Weight
- Cub Crafters Inc. - InvestorNewsBreaks – Cub Crafters Inc. Looking to Expand Operations Amid Increasing Popularity of Backcountry Aviation
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Begins Dosing Participants in CYB003 Trial for Treatment of MDD
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Q2 2022 Financial Report, Corporate Update
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - BillionDollarBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Helping Companies Across Various Sectors Implement Quantum Computing Solutions
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - DEA Delay Leaves Synthetic Psychedelics in Legal Limbo
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Secures ICON Capital Group as Financial Advisor to Pursue Strategic Spinout
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Supplying Options Worldwide to Improve Food Security
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - MiningNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) on Target to Own, Control 'the Elements' to Supply EV, Renewable Energy, Other Technology Manufacturers
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - Exro Announces a Public and Private Financing of up to approximately $10mm
- FingerMotion Inc. (NASDAQ: FNGR) - ChineseNewsBreaks - FingerMotion Inc.'s (NASDAQ: FNGR) Relationship with China's Largest Telcos Providing 'Strong Footing' in Market
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Options Two Lithium Claims Directly Adjacent to 'Electric Avenue'
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Experts Say Federal Court Ruling in Maine Could Pave Way for Interstate Cannabis Commerce
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Releases Q2 2022 Financial, Corporate Report
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Streamlining NAFTA Region's Cross-Border Shipping Process
- Friendable Inc. (FDBL) - Friendable Inc. (FDBL) Addresses Churn in the Music Streaming Industry by Offering Fan Pass Live and Branded Products to Enhance Listener Experience
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Announces Filing of Quarterly Report and Provides Early Pre-Sales Update
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc.’s SmartGreen(TM) Tech Transforms Homes, Enables Energy Independence
- Golden Matrix Group Inc. (NASDAQ: GMGI) - Golden Matrix Group Inc. (NASDAQ: GMGI) Scores Big Q3 Win on International Expansion
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH), Electryone Advisors Expand Partnership with Advancement of Exceptional Technology
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Secures New Patent, Critical Protection for REQORSA(TM) Combination Therapy
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- GreenBox POS (NASDAQ: GBOX) - InvestorNewsBreaks - GreenBox POS (NASDAQ: GBOX) Announces Restructuring of Convertible Note Financing Terms
- Cepton Inc. (NASDAQ: CPTN) - Cepton, Inc. Announces New Investment Intent for Up To $100 Million from Koito Manufacturing
- Hemptown USA - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Common Stock Upgraded to Mid-Tier OTC Equity Market
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) Reports on Growth Potential of US Cannabis Markets
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Seeks to Address Electric Vehicle Energy Efficiency Issues Through Proprietary Powertrain Solution
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Unveils Vegan + Sustainable Line from Celebrity Chef Priyanka
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Q2 2022 Unaudited Financial Results
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) to Participate in Energica Week 2022
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd.'s (NASDAQ: IFBD) Shanghai Qishuo to Leverage 'Retail Cube' Solution for World-Renowned Brand
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InMed Announces Share Consolidation to Meet Nasdaq Listing Criteria
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Notes FDA Final Rule on OTC Hearing Aids
- Innovative Payment Solutions Inc. (OTCQB: IPSI) - Innovative Payment Solutions, Inc. Appoints Richard Rosenblum as President and Chief Financial Officer
- BlockQuarry Corp. (OTC: BLQC) - Cryptocurrencies Appear to Have Seen the Worst of Crypto Winter
- Knightscope (NASDAQ: KSCP) - Oregon Healthcare Provider is Latest Knightscope (Nasdaq: KSCP) Client
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - Laredo Oil Inc. (LRDC) Progressing Toward Drilling as 2023 Oil Demand Projected to Increase
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces the Availability of a Broadcast titled, "The Power of Science to Stop a Killer"
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Releases Q1 Financial, Operational Results
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - InvestorNewsBreaks - LQwD Fintech Corp. (TSX.V: LQWD) (OTC: LQWDF) Releases Results from Annual General Meeting
- LQwD Announces Annual General Meeting Voting Results
- CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging LN to Enable Faster, More Secure Micropayments
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Third Quarter of Fiscal 2022
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Enters Common Share Subscription Agreement
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Releases Q1 2023 Report, Notes Purchase Agreement Highlight
- Mullen Automotive Inc. (NASDAQ: MULN) - GreenCarNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Positioned 'Front and Center for Last-Mile Delivery Opportunities'
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (OTCQB: NEXCF) (NEO: NTAR) (CSE: NTAR) (FSE: N29) CEO Talks ARway News in Power Play Interview
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - InvestorNewsBreaks - Odyssey Health Inc. (ODYY) Featured in NeurologyLive(R) Article
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - PlantX Conducts Additional Live Retail Events, Driving Store Traffic and Increasing Sales Velocity
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Releases Q2 2022 Financial Report
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Poised for US Expansion Due to 'Attractive Incentives to Build Hydrogen Infrastructure'
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 Financial Report
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Partners with Skincare Leader, Icon and Pioneer
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Subsidiary Receives Adult-Use Edibles Sales, Expansion Rights
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Names New Key Board Appointments
- QSAM Biosciences Inc. (OTCQB: QSAM) - BioMedNewsBreaks - QSAM Biosciences Inc. (QSAM) Receives 'Marketable Advantage' Toward Manufacturing, Supply Chain of CycloSam(R)
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Red White & Bloom Reports Financial Results for Second Quarter 2022 and Six Months Ended June 30th 2022
- REZYFi, Inc. - REZYFi, Inc. is "One to Watch"
- RYAH Group Inc. (CSE: RYAH) - InvestorNewsBreaks - RYAH Group Inc. (CSE: RYAH) Names New CFO, Corporate Secretary
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation (SNWR) Subsidiary Releases Q2 Financial Results, Corporate Update
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Receives Notice of Patent Allowance for Tech Designed to Protect Produce from Decay
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Has Created a Community, Distinction to Build Powerful Brand
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions (NASDAQ: SASI) Announces Q2 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Psychedelics Company Raises More than $10M in Seed Funding to Advance Ketamine Therapy
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Ramps up Sales for SD7, Companion Devices Across Multiple Verticals
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc.'s (NYSE American: SBEV) Selected for 7-Eleven's Highly Competitive Brands with Heart Campaign
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - InvestorNewsBreaks - SRAX Inc. (NASDAQ: SRAX) Announces Entry into Revolving Credit Facility, Launch of Stock Portfolio Tracker
- Standard Lithium Ltd. (NYSE American: SLI) - Bullish Sentiments Continue for Global Lithium Mining Industry as Demand Soars
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Subsidiary Inks MSA for Manufacturing, Distribution Expansion
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Secures Purchase Renewal Contract with Louisville Division of The Kroger Company (NYSE: KR)
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT(R) Global Alternatives (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Announces UK Distributor Places Third Reorder
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Expands Akaline88(R) to 200+ Midwest, Mid-Atlantic Stores
- Tingo Inc. (OTC: TMNA) - World Bank Affiliate Looks to Blockchain to Facilitate Carbon Trading
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Expands IP Portfolio with New Provisional Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Launches TRxADE Prime Plus to Meet Customers' Needs of On-Demand Inventory
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Completes Acquisition to Create Largest Diversified North American Focused Uranium Company
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - 4 Cheap Robinhood Stocks To Buy According To Insiders In August 2022
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Extension of LAPD BolaWrap Pilot Program
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - InvestorNewsBreaks - XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) Exploring Expanded Application of ODF Screening Tests
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Enrolls First Patient in Early-Stage Frozen Shoulder Trial
- Aditxt Inc. (NASDAQ: ADTX) - Study Finds Children Have Better Outcomes with Kidney Transplants from Living Donors
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Mothers in Indonesia Fight for Their Children to Have Access to Medical Cannabis
- American Cannabis Partners - 420 with CNW — How Missouri Activists Overcame Setbacks to Put Cannabis Measure on Upcoming Ballot
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Announces $2.3M Registered Direct Offering
- AnPac Bio-Medical Science Co. Ltd. (NASDAQ: ANPC) - Today's Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on MondayMore From InvestorPlace
- AmpliTech Group Inc. (NASDAQ: AMPG) - InvestorNewsBreaks - AmpliTech Group Inc. (NASDAQ: AMPG) Releases Acquisition Update, Anticipated Results
- AREV Life Sciences Global Corp. (CSE: AREV) (OTC: AREVF) - SUSTAINN, AREV's Ready to Use Therapeutic Foods discussed in leading article in HealthHIV
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix to Present at the H.C. Wainwright 24th Annual Global Investment Conference
- Avricore Health Inc. (TSX.V: AVCR) (OTCQB: AVCRF) - InvestorNewsBreaks - Avricore Health Inc. (TSX.V: AVCR) (OTCQB: AVCRF) Releases Q2 2022 Report
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - BevCanna Enterprises Inc.'s (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Journey: From Runners' Knee to Canada's 101 Top F&B Start-up List
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Enters MOU to Acquire 100% Equity of Shengruihao
- Brain Scientific Inc. (OTCQB: BRSF) - InvestorNewsBreaks - Brain Scientific Inc. (BRSF) Secures CE Mark Approval for NeuroCap(TM)
- Cannabis Strategic Ventures Inc. (OTC: NUGS) - 420 with CNW - Recent Studies Show Advanced Cancer Sufferers Could Benefit from Using Marijuana
- Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) - InvestorNewsBreaks - Cerberus Cyber Sentinel Corp. (NASDAQ: CISO) Partners with IBN for Corporate Communications
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - CNS Pharmaceuticals, Inc. (NASDAQ: CNSP) Looking to Fill Glioblastoma Multiforme Treatment Gap with Berubicin Drug Product
- Correlate Infrastructure Partners Inc. (OTCQB: CIPI) - Correlate Infrastructure Partners Inc. (CIPI) Helps Companies Boost Profitability Through Energy Improvements as ESG Gains Weight
- Cub Crafters Inc. - InvestorNewsBreaks – Cub Crafters Inc. Looking to Expand Operations Amid Increasing Popularity of Backcountry Aviation
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) Begins Dosing Participants in CYB003 Trial for Treatment of MDD
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Q2 2022 Financial Report, Corporate Update
- DealMaker - InvestorNewsBreaks – DealMaker CEO Featured on ‘Gamechangers LIVE’
- D-Wave Quantum Inc. (NYSE: QBTS) - BillionDollarBreaks - D-Wave Quantum Inc. (NYSE: QBTS) Helping Companies Across Various Sectors Implement Quantum Computing Solutions
- Delic Holdings Corp. (CSE: DELC) (OTCQB: DELCF) - DEA Delay Leaves Synthetic Psychedelics in Legal Limbo
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc. (DSGT) Secures ICON Capital Group as Financial Advisor to Pursue Strategic Spinout
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - Strong Earnings, Revenue Reports from Plant-based Foods Maker Belle Pulses, Drives Cheery Assessment for Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF)
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Lays Strong Foundation in 'Seed-to-Market' Approach
- ESGBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Supplying Options Worldwide to Improve Food Security
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - MiningNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) on Target to Own, Control 'the Elements' to Supply EV, Renewable Energy, Other Technology Manufacturers
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - Exro Announces a Public and Private Financing of up to approximately $10mm
- FingerMotion Inc. (NASDAQ: FNGR) - ChineseNewsBreaks - FingerMotion Inc.'s (NASDAQ: FNGR) Relationship with China's Largest Telcos Providing 'Strong Footing' in Market
- First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - First Energy Metals Ltd. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Options Two Lithium Claims Directly Adjacent to 'Electric Avenue'
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Experts Say Federal Court Ruling in Maine Could Pave Way for Interstate Cannabis Commerce
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Releases Q2 2022 Financial, Corporate Report
- Freight Technologies Inc. (NASDAQ: FRGT) - TinyGemsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) Streamlining NAFTA Region's Cross-Border Shipping Process
- Friendable Inc. (FDBL) - Friendable Inc. (FDBL) Addresses Churn in the Music Streaming Industry by Offering Fan Pass Live and Branded Products to Enhance Listener Experience
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - FuelPositive Announces Filing of Quarterly Report and Provides Early Pre-Sales Update
- GeoSolar Technologies Inc. - GeoSolar Technologies Inc.’s SmartGreen(TM) Tech Transforms Homes, Enables Energy Independence
- Golden Matrix Group Inc. (NASDAQ: GMGI) - Golden Matrix Group Inc. (NASDAQ: GMGI) Scores Big Q3 Win on International Expansion
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH), Electryone Advisors Expand Partnership with Advancement of Exceptional Technology
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Secures New Patent, Critical Protection for REQORSA(TM) Combination Therapy
- Green Hygienics Holdings Inc. (OTCQB: GRYN) - InvestorNewsBreaks - Green Hygienics Holdings Inc. (GRYN) Enters US Plant-Based Food Market with Superior Hemp Protein Product
- GreenBox POS (NASDAQ: GBOX) - InvestorNewsBreaks - GreenBox POS (NASDAQ: GBOX) Announces Restructuring of Convertible Note Financing Terms
- Cepton Inc. (NASDAQ: CPTN) - Cepton, Inc. Announces New Investment Intent for Up To $100 Million from Koito Manufacturing
- Hemptown USA - InvestorNewsBreaks - Hero Technologies Inc. (HENC) Common Stock Upgraded to Mid-Tier OTC Equity Market
- Hero Technologies Inc. (OTC: HENC) - InvestorNewsBreaks - Hero Technologies (HENC) Reports on Growth Potential of US Cannabis Markets
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA) Seeks to Address Electric Vehicle Energy Efficiency Issues Through Proprietary Powertrain Solution
- Home Bistro Inc. (OTC: HBIS) - InvestorNewsBreaks - Home Bistro Inc. (HBIS) Unveils Vegan + Sustainable Line from Celebrity Chef Priyanka
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) Announces Q2 2022 Unaudited Financial Results
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) to Participate in Energica Week 2022
- Infobird Co., Ltd (NASDAQ: IFBD) - InvestorNewsBreaks - Infobird Co. Ltd.'s (NASDAQ: IFBD) Shanghai Qishuo to Leverage 'Retail Cube' Solution for World-Renowned Brand
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InMed Announces Share Consolidation to Meet Nasdaq Listing Criteria
- InnerScope Hearing Technologies Inc. (OTC: INND) - InvestorNewsBreaks - InnerScope Hearing Technologies Inc. (INND) Notes FDA Final Rule on OTC Hearing Aids
- Innovative Payment Solutions Inc. (OTCQB: IPSI) - Innovative Payment Solutions, Inc. Appoints Richard Rosenblum as President and Chief Financial Officer
- BlockQuarry Corp. (OTC: BLQC) - Cryptocurrencies Appear to Have Seen the Worst of Crypto Winter
- Knightscope (NASDAQ: KSCP) - Oregon Healthcare Provider is Latest Knightscope (Nasdaq: KSCP) Client
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc. (KNOS) Positioned Amid Expected Growth of Indoor Air Quality Market
- Laredo Oil Inc. (OTC: LRDC) - Laredo Oil Inc. (LRDC) Progressing Toward Drilling as 2023 Oil Demand Projected to Increase
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Announces the Availability of a Broadcast titled, "The Power of Science to Stop a Killer"
- Lottery.com Inc. (NASDAQ: LTRY) - InvestorNewsBreaks - Lottery.com (NASDAQ: LTRY) Releases Q1 Financial, Operational Results
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - InvestorNewsBreaks - LQwD Fintech Corp. (TSX.V: LQWD) (OTC: LQWDF) Releases Results from Annual General Meeting
- LQwD Announces Annual General Meeting Voting Results
- CryptoNewsBreaks - LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) Leveraging LN to Enable Faster, More Secure Micropayments
- MedSmart Group Inc. (OTC: MSGP) - InvestorNewsBreaks - MedSmart Group Inc. (MSGP) Announces Milanion Limited's Inroads into Ukraine Through Strategic MOU
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MINDCURE Announces Financial Results for the Third Quarter of Fiscal 2022
- Mobius Interactive Ltd. - InvestorNewsBreaks – Mobius Interactive Ltd.’s Mobius.bet Poised in Lucrative Emerging Market
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) (FSE: 0NFA) Enters Common Share Subscription Agreement
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Releases Q1 2023 Report, Notes Purchase Agreement Highlight
- Mullen Automotive Inc. (NASDAQ: MULN) - GreenCarNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Positioned 'Front and Center for Last-Mile Delivery Opportunities'
- Nexstar Media Group Inc. (NASDAQ: NXST) - InvestorNewsBreaks - Nexstar Media Group Inc. (NASDAQ: NXST) Announces 17th Market to Launch ATSC 3.0 This Year
- Nextech AR Solutions Corp. (CSE: NTAR) (OTCQB: NEXCF) - InvestorNewsBreaks - Nextech AR Solutions Corp. (OTCQB: NEXCF) (NEO: NTAR) (CSE: NTAR) (FSE: N29) CEO Talks ARway News in Power Play Interview
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- Odyssey Group International Inc. (OTC: ODYY) - InvestorNewsBreaks - Odyssey Health Inc. (ODYY) Featured in NeurologyLive(R) Article
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - PlantX Conducts Additional Live Retail Events, Driving Store Traffic and Increasing Sales Velocity
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) (FSE: 7CR) Releases Q2 2022 Financial Report
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - InvestorNewsBreaks - PowerTap Hydrogen Capital Corp. (NEO: MOVE) (OTC: MOTNF) (FWB: 2K6) Poised for US Expansion Due to 'Attractive Incentives to Build Hydrogen Infrastructure'
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 Financial Report
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) Partners with Skincare Leader, Icon and Pioneer
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Subsidiary Receives Adult-Use Edibles Sales, Expansion Rights
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Names New Key Board Appointments
- QSAM Biosciences Inc. (OTCQB: QSAM) - BioMedNewsBreaks - QSAM Biosciences Inc. (QSAM) Receives 'Marketable Advantage' Toward Manufacturing, Supply Chain of CycloSam(R)
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - Red White & Bloom Reports Financial Results for Second Quarter 2022 and Six Months Ended June 30th 2022
- REZYFi, Inc. - REZYFi, Inc. is "One to Watch"
- RYAH Group Inc. (CSE: RYAH) - InvestorNewsBreaks - RYAH Group Inc. (CSE: RYAH) Names New CFO, Corporate Secretary
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation (SNWR) Subsidiary Releases Q2 Financial Results, Corporate Update
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Receives Notice of Patent Allowance for Tech Designed to Protect Produce from Decay
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Has Created a Community, Distinction to Build Powerful Brand
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions (NASDAQ: SASI) Announces Q2 2022 Financial Results
- Silo Pharma Inc. (OTCQB: SILO) - Psychedelics Company Raises More than $10M in Seed Funding to Advance Ketamine Therapy
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Ramps up Sales for SD7, Companion Devices Across Multiple Verticals
- Simply Sonoma Inc. - InvestorNewsBreaks – Simply Sonoma Enters Growing CBD-Infused Beverages Space
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc.'s (NYSE American: SBEV) Selected for 7-Eleven's Highly Competitive Brands with Heart Campaign
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- SRAX Inc. (NASDAQ: SRAX) - InvestorNewsBreaks - SRAX Inc. (NASDAQ: SRAX) Announces Entry into Revolving Credit Facility, Launch of Stock Portfolio Tracker
- Standard Lithium Ltd. (NYSE American: SLI) - Bullish Sentiments Continue for Global Lithium Mining Industry as Demand Soars
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- StraightUp Resources Inc. (CSE: ST) (OTCQB: STUPF) - StraightUp Resources Extends Warrant Term
- Streamlytics - InvestorNewsBreaks – Streamlytics Joins World’s Most-Trusted Enterprise Cloud Marketplace
- Sugarmade Inc. (OTC: SGMD) - InvestorNewsBreaks - Sugarmade Inc. (SGMD) Subsidiary Inks MSA for Manufacturing, Distribution Expansion
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Secures Purchase Renewal Contract with Louisville Division of The Kroger Company (NYSE: KR)
- Sycamore Entertainment Group Inc. (OTC: SEGI) - InvestorNewsBreaks - Sycamore Entertainment Group Inc. (SEGI) Featured in Bell2Bell Podcast
- TAAT Lifestyle & Wellness Ltd. (CSE: TAAT) (OTCQX: TOBAF) - InvestorNewsBreaks - TAAT(R) Global Alternatives (CSE: TAAT) (OTCQX: TOBAF) (FRANKFURT: 2TP) Announces UK Distributor Places Third Reorder
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Expands Akaline88(R) to 200+ Midwest, Mid-Atlantic Stores
- Tingo Inc. (OTC: TMNA) - World Bank Affiliate Looks to Blockchain to Facilitate Carbon Trading
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Expands IP Portfolio with New Provisional Patent Application
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - InvestorNewsBreaks - TRxADE Health Inc. (NASDAQ: MEDS) Launches TRxADE Prime Plus to Meet Customers' Needs of On-Demand Inventory
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Completes Acquisition to Create Largest Diversified North American Focused Uranium Company
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - 4 Cheap Robinhood Stocks To Buy According To Insiders In August 2022
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - Vivos Therapeutics Announces Receipt of Notice from Nasdaq Regarding Late Filing of Quarterly Report on Form 10-Q
- Wrap Technologies Inc. (NASDAQ: WRAP) - Wrap Announces Extension of LAPD BolaWrap Pilot Program
- XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) - InvestorNewsBreaks - XPhyto Therapeutics Corp. (CSE: XPHY) (OTCQB: XPHYF) (FSE: 4XT) Exploring Expanded Application of ODF Screening Tests
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































