The QualityStocks Daily Stock List
- Aldeyra Therapeutics (ALDX)
- YaSheng (HERB)
- Independence Gold (IEGCF)
- Thai Beverage (TBVPF)
- US Nuclear (UCLE)
- Webcentral (MLBEF)
- NIO Inc. (NIO)
- Trulieve Cannabis Corp. (TCNNF)
- Newmont Corporation (NEM)
- atai Life Sciences N.V. (ATAI)
- Marathon Digital Holdings Inc. (MARA)
- Aurora Cannabis Inc. (ACB)
Aldeyra Therapeutics (ALDX)
MarketClub Analysis, MarketBeat, StockMarketWatch, BUYINS.NET, RedChip, TradersPro, TraderPower, Kiplinger Today, Marketbeat.com, Promotion Stock Secrets, InvestorPlace, Schaeffer's, Wall Street Mover, PoliticsAndMyPortfolio, FreeRealTime, Barchart, Red Chip, Short Term Wealth, StockOodles, StreetInsider, TopPennyStockMovers, Trades Of The Day and QualityStocks reported earlier on Aldeyra Therapeutics (ALDX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Aldeyra Therapeutics Inc. (NASDAQ: ALDX) (FRA: 137) is a biotechnology firm that is focused on the development and commercialization of medicines for systemic and immune-mediated ocular ailments.
The firm has its headquarters in Lexington, Massachusetts and was incorporated in 2004, on August 13th by John E. Dowling and Thomas A. Jordan. Prior to its name change in March 2014, the firm was known as Aldexa Therapeutics Inc. It serves consumers in the United States.
The company’s objective is to develop new products for orphan, inflammatory, immune-mediated and other illnesses that may be caused partly by toxic chemical species which occur naturally, known as free aldehydes.
The enterprise’s product pipeline is made up of a di-hydrofolatereductase inhibitor known as ADX-2191, which is undergoing phase 2 clinical trials testing its effectiveness in treating primary vitreo-retinal lymphoma and in phase III trials for the prevention of proliferative vitreo-retinopathy. It also develops an ophthalmic solution dubbed reproxalap, which is in a phase 2 clinical trial evaluating its efficacy in treating allergic conjunctivitis and dry eye ailments. In addition to this, it develops a small molecule heat shock protein 90 inhibitor known as ADX-1612, to treat lymphoproliferative illnesses and reactive aldehyde species that are pro-inflammatory scavengers. It also develops ADX-103 to treat retinal ailments; and ADX-629 to treat allergies and autoimmune ailments.
The firm recently announced its latest financial results for 2021, with its CEO noting that they were focused on advancing the development of effective and safe treatments for systemic and ocular ailments that will improve the lives of patients whose needs haven’t been met.
Aldeyra Therapeutics (ALDX), closed Wednesday's trading session at $2.54, up 48.538%, on 66,680,191 volume. The average volume for the last 3 months is 6,252 and the stock's 52-week low/high is $1.42/$11.97.
YaSheng (HERB)
Zacks and QualityStocks reported earlier on YaSheng (HERB), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
YaSheng Group (OTC: HERB) is a company focused on designing, developing, manufacturing and marketing farming and sideline products; chemical materials and products; construction materials; textiles; and livestock and poultry products.
The firm has its headquarters in Lanzhou, China and was incorporated in 1998 by Changsheng Zhou. It operates as part of the farm products industry, under the consumer defensive sector. The firm serves consumers around the globe, with a focus on those in the People’s Republic of China.
The company operates through the Agriculture & Fisheries, Biotechnology, Blockchain and Mining segments. It operates as a subsidiary of Gansu Yasheng Salt Chemical Industrial Group Limited.
The enterprise cultivates, processes, markets and distributes a range of food and agro-byproducts. Its products include cotton, wheat, corns, barley, flax, and alfalfa; vegetables comprising onions, beets, potatoes, and peas; fruits, including pears, apples, and apricots; specialty crops consisting of hops, cumin, wolfberries, hemp, and liquorices; seeds, such as black melon, hemp, sunflower, corn, and flax seeds; and eggs. It also offers construction materials, such as cement; and designs, develops and markets new technologies related to agriculture and genetic biology. The enterprise sells its products to food processors, supermarkets, and wholesale stores through distributors, as well as to direct customers. This is in addition to exporting its products to 15 countries, including the United States and the Philippines.
The firm is poised for further expansion into the global market through organic growth, mergers and acquisitions. This may positively influence investments into the firm as well as its overall growth.
YaSheng (HERB), closed Wednesday's trading session at $0.0849, up 39.1803%, on 400 volume. The average volume for the last 3 months is 104,574 and the stock's 52-week low/high is $0.053/$0.237.
Independence Gold (IEGCF)
We reported earlier on Independence Gold (IEGCF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Independence Gold Corp. (OTCQB: IEGCF) (CVE: IGO) is a mineral exploration firm focused on acquiring, exploring for and evaluating precious metal properties.
The firm has its headquarters in Vancouver, Canada and was incorporated in 2011, on November 1st. It operates as part of the gold industry, under the basic materials sector. The firm serves consumers in Canada and North America.
The company primarily explores for gold, silver, copper, cobalt, nickel and zinc deposits. It holds 100% interest in the 3Ts project, which comprises of 15 mineral claims that cover roughly 5,200 hectares located in the Nechako Plateau region of central British Columbia; and Boulevard project, which holds about 958 quartz mining claims that cover about 19,960 hectares situated in the Whitehorse mining district, Yukon. The company also holds interests in the Moosehorn property, which comprises of 82 quartz mining claims that cover an area of approximately 1,720 hectares located in the Whitehorse mining district, Yukon. In addition to this, it has an option to acquire 60% interests in the Nicoamen property, which comprises of 9 mineral claims covering approximately 3,332 hectares located in the Spences Bridge Gold Belt, British Columbia; and the Merit property, which is made up of 4 mineral claims that cover an area of approximately 1,907 hectares and is located in the Spences Bridge Gold Belt, British Columbia.
The company, which recently discovered a new copper-silver target dubbed Ootsa at its 100% owned 3Ts Project, remains focused on advancing exploration efforts at its projects and creating additional value for its shareholders.
Independence Gold (IEGCF), closed Wednesday's trading session at $0.1447, off by 2.3617%, on 97,899 volume. The average volume for the last 3 months is 58,411 and the stock's 52-week low/high is $0.03/$0.175.
Thai Beverage (TBVPF)
MarketBeat, QualityStocks and InvestorPlace reported earlier on Thai Beverage (TBVPF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Thai Beverage Public Co. Ltd (OTC: TBVPF) (OTC: TBVPY) (SGX: Y92) is a company focused on producing and distributing alcoholic and non-alcoholic beverages, and food products.
The firm has its headquarters in Bangkok, Thailand and was incorporated in 2003. It operates as part of the beverages-wineries and distilleries industry, under the consumer defensive sector. The firm serves consumers across the globe.
The enterprise operates through Beer, Spirits, Non-Alcoholic Beverages and Food segments. It provides malt, liquor, beer and yeast products; spirits, including white spirits, brown spirits, herbs, and other products; and non-alcoholic beverages comprising drinking and soda water, electrolyte beverages, energy drinks, green and herbal tea, ready-to-drink coffee, fruit flavored drinks, and soft drinks; carbonated soft drinks, isotonics, tea, soya drinks, Asian and sparkling drinks, ready-to-drink juice, cordials, UHT milk, water, pasteurized milk, yoghurt, sterilized milk, ice cream, cereal bars, canned milk and pasteurized juice. It also offers chilled and frozen food products; and fertilizers, feeds, bricks, and oak barrels. In addition to this, the enterprise is involved in the trading of molasses; operation of bakeries, restaurants and social enterprise; provision of transportation and distribution, logistics, advertising and marketing, consultancy, asset and brands management, public cold storage, and human resources and organization development services; and production and distribution of biogas. Further, it trades bottles and supplies; produces metal and plastic packaging for businesses; trades chemical and packaging materials; and manufactures equipment used in food manufacturing.
The company remains committed to strengthening its core business, building new capabilities and unlocking its potential to create maximum value. This may open it up to new growth and investment opportunities to generate shareholder value.
Thai Beverage (TBVPF), closed Wednesday's trading session at $0.39, even for the day, on 19,000 volume. The average volume for the last 3 months is 69,711 and the stock's 52-week low/high is $0.355/$0.5486.
US Nuclear (UCLE)
QualityStocks, Innovative Marketing, Stock News Now, PoliticsAndMyPortfolio, Wall Street Mover, TopPennyStockMovers, The Online Investor, Pennystockmania, PennyPickGains, MarketBeat and InvestorPlace reported earlier on US Nuclear (UCLE), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
US Nuclear Corp. (OTCQB: UCLE) is a company focused on the development, manufacture and sale of radiation detection and measuring equipment.
The firm has its headquarters in Canoga Park, California and was incorporated in 2012, on February 14th. It operates as part of the scientific and technical instruments industry, under the technology sector. The firm serves consumers globally.
The company is party to a strategic alliance with Arbok-Nuclear for the introduction of desalination and rad-wastewater treatment techniques. It operates through the Optron and Overhoff segments.
The enterprise provides radiation water monitors that allow the detection of radioactive materials in drinking water, lakes, ground water, rivers and rainfall; alpha, beta, gamma and Tritium monitors; air and water monitors; DroneRAD aerial radiation detection; and nano-second X-ray monitors. It also offers personnel, vehicle, room and exit monitors; radon air monitors and radon switch products to determine the radon content in the air in mines, basements, buildings and mills; handheld survey meters, as well as personal dosimeters and pocket micro-R meters; and port security equipment. In addition to this, the enterprise provides software which measures airborne radioactivity levels; gamma and neutron radiation levels; temperature and humidity in the facility; barometric pressure; status of security doors; and wind speed and direction. Its products and services are used in nuclear reactor plants, universities, local and state hospitals, government agencies, and emergency medical technicians/first responders, as well as in airports, cargo screening at borders and ports, and other critical infrastructure.
The firm recently appointed a new member to the board, a move that strengthens it as it moves ahead towards its goals. This new addition, who has extensive experience and skills, will guide the firm as it pursues mergers and acquisitions that may benefit its stakeholders.
US Nuclear (UCLE), closed Wednesday's trading session at $0.11, up 3.4078%, on 47,034 volume. The average volume for the last 3 months is 6,531 and the stock's 52-week low/high is $0.04525/$0.187.
Webcentral (MLBEF)
We reported earlier on Webcentral (MLBEF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Webcentral Ltd (OTC: MLBEF) (ASX: WCG) is a digital services firm engaged in the provision of a full product suite of innovative and scalable digital services.
The firm has its headquarters in Melbourne, Australia and was incorporated in 1996. Prior to its name change in November 2021, the firm was known as Webcentral Group Limited. It operates as part of the information technology services industry, under the technology sector. The firm primarily serves consumers in Australia and New Zealand.
The company operates through the Small Medium Business Solutions and Enterprise Services segments. The Small Medium Business Solutions segment provides website and email hosting, domain name registrations and renewals, website development, search engine optimization, and analysis for businesses in New Zealand and Australia. On the other hand, the Enterprise Services segment offers cloud, mobile application development, data, and analytics to Australian enterprise and government organizations.
The enterprise provides cloud and data center, data connectivity, and managed services. It offers cloud-based solutions and network services; and operates fiber and wireless infrastructure, manages cloud the computing environment, and operates data center facilities. In addition to this, it is involved in domain name registrations and renewals, website development, social advertising campaigns, website and email hosting and search engine marketing for businesses. The enterprise serves small and medium businesses, enterprises, and wholesale and government customers.
The firm remains dedicated to leading online success for its clients through the delivery of successful business outcomes and maintaining its market-leading position. This may translate to higher returns for the business as well as its shareholders.
Webcentral (MLBEF), closed Wednesday's trading session at $0.25, even for the day, on 43,640 volume. The average volume for the last 3 months is 47.766M and the stock's 52-week low/high is $0.0015/$2.21.
NIO Inc. (NIO)
Green Car Stocks, InvestorPlace, Schaeffer's, MarketClub Analysis, The Street, MarketBeat, StocksEarning, Daily Trade Alert, Trades Of The Day, Kiplinger Today, The Online Investor, QualityStocks, StockEarnings, INO Market Report, Zacks, Early Bird, StreetInsider, StockMarketWatch, BUYINS.NET, Cabot Wealth, Wealth Insider Alert, GreenCarStocks, InvestorsUnderground, FreeRealTime, Money Wealth Matters, The Wealth Report, TipRanks, CNBC Breaking News, Daily Wealth, wyatt research newsletter, TradersPro, Investopedia, Energy and Capital, CRWEWallStreet, Green Energy Stocks, InvestorIntel, Investors Alley, InvestorsObserver Team, MarketClub, Louis Navellier, TopPennyStockMovers, AllPennyStocks, Wealth Daily, Smartmoneytrading, Stock Market Watch, The Night Owl, Top Pros' Top Picks, Top Pros’ Top Picks and Jim Cramer reported earlier on NIO Inc. (NIO), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
German automaker Mercedes says the electric vehicle market is becoming increasingly “brutal” for automakers, forcing many to cut vehicle prices amid intense competition. While Tesla has been the most dominant player in the global EV industry for almost a decade, with established automakers and startups are quickly catching up to the Texas-based automaker.
China and Europe have seen a surge in EV sales in recent months, primarily thanks to a major influx of affordable Chinese electric cars. Massive subsidies from the Chinese government have allowed EV makers in China to significantly reduce production costs and pass the savings on to their customers, resulting in incredibly affordable electric cars.
Tesla has cut prices for best-selling EVs such as the Model 3 and Y in a bid to remain competitive, resulting in an intense price discount war in China that even attracted the attention of local authorities. Affordable Chinese EVs have also become incredibly popular in the European market in recent months, undercutting European automakers and triggering an EU probe into Chinese EV subsidies.
EV sales in China and Europe, currently the largest EV markets on the globe, have surged in recent months, leaving the U.S. market behind. The market is becoming especially hot for European carmakers that have been forced to contend with cheap electric car imports from China. Mercedes-Benz’s chief financial officer Harald Wilhelm says the current EV market is a “pretty brutal space” for many players.
Even though the cost of EV production is higher, automakers are pricing their EV offerings at internal combustion engine (ICE) levels just to get them off their lots, severely limiting their profitability. According to Wilhelm, the current status quo in the EV market cannot be sustainable for most players in the automotive industry.
With the exception of a few companies such as Toyota, most established automakers are in the process of transitioning from ICE car production to electric vehicles. On top of national and global pledges to transition to electric cars, the Chinese EV industry is also forcing European automakers to transition much faster than they anticipated.
Chinese companies such as NIO Inc. (NYSE: NIO) are flooding the European market with heavily subsidized EVs and threatening to take over the entire space before local automakers have a chance to compete. The recently launched EU probe could lead to the EU levying tariffs on Chinese EV imports in an effort to protect local automakers. Beijing has condemned the EU probe, calling it a “naked protectionist act” that could negatively impact the nascent EV industry.
NIO Inc. (NIO), closed Wednesday's trading session at $7.45, up 2.0548%, on 33,335,132 volume. The average volume for the last 3 months is 458,087 and the stock's 52-week low/high is $7.00/$16.18.
Trulieve Cannabis Corp. (TCNNF)
InvestorPlace, MarketBeat, Wealth Insider Alert, Daily Trade Alert, QualityStocks, Top Pros' Top Picks, Cabot Wealth, The Street, Trades Of The Day, Profit Trends, TradersPro, The Online Investor and StreetInsider reported earlier on Trulieve Cannabis Corp. (TCNNF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The Bank of America, MasterCard and the National Rifle Association are among a group of institutions that have disclosed lobbying activities related to legislation that would grant the cannabis industry access to banking services. Despite generating billions of dollars in sales, America’s state-legal cannabis industry has had no access to financial services for most of its existence.
Cannabis businesses cannot access financial aid, and they are forced to operate on a cash-only basis, making them prime targets for criminals looking for liquid assets to steal.
After lawmakers failed to advance cannabis banking past the U.S. Senate several times, an amended version of the bill titled the Secure and Fair Enforcement Regulation (SAFER) Banking Act has gained bipartisan support and has a high chance of reaching President Joe Biden’s desk.
The three institutions recently included the SAFER Banking Act in their lobbying disclosure for Q3 2023. This measure would finally allow financial institutions such as banks to serve state-legal cannabis businesses without fear of reprisal from federal authorities. The measure was recently subject to a historic vote at the Senate Banking Committee, representing the first time the U.S. Senate addressed cannabis banking legislation. Although the bill’s previous iteration passed the House multiple times, it failed in the Senate each time.
While the Bank of America has disclosed lobbying activities for both the SAFE Banking Act and the SAFER Banking Act, which recently received the Senate’s approval, the National Rifle Association (NRA) has only disclosed lobbying activities related to the SAFER Banking Act. Since neither institution has discussed its position on the legislation, it still isn’t clear if they intend to lobby against or in favor of cannabis banking legislation.
Even so, we can use their past positions on the controversial drug to surmise whether they will lobby for or against expanding the marijuana industry’s access to financial services.
The Bank of America has shown interest in the cannabis industry in recent years, with Bank of America Merrill Lynch initiating equity research coverage for Canadian cannabis companies in 2019.
While the National Rifle Association hasn’t expressed an official stance on cannabis, former NRA president David Keene has noted that federal restrictions that criminalize Americans for medical marijuana use are causing major issues. The former president said in a 2018 op-ed that the dissonance between federal and state cannabis laws was creating significant problems for gun owners across the country.
MasterCard is actively lobbying in support of the SAFER Banking Act and lobbied for cannabis banking in 2019 and 2020.
These efforts are a welcome development to marijuana enterprises such as Trulieve Cannabis Corp. (CSE: TRUL) (OTCQX: TCNNF) that are invested in seeing marijuana policies reformed not just at state level but preferably federally.
Trulieve Cannabis Corp. (TCNNF), closed Wednesday's trading session at $4.4, off by 2.2222%, on 291,811 volume. The average volume for the last 3 months is 10.173M and the stock's 52-week low/high is $3.42/$16.11.
Newmont Corporation (NEM)
MarketClub Analysis, InvestorPlace, The Street, Schaeffer's, Kiplinger Today, StocksEarning, MarketBeat, The Online Investor, INO.com Market Report, Investopedia, Barchart, StreetAuthority Daily, Daily Trade Alert, TopStockAnalysts, Top Pros' Top Picks, StreetInsider, Louis Navellier, Streetwise Reports, Daily Wealth, SmarTrend Newsletters, Zacks, QualityStocks, Uncommon Wisdom, TradingMarkets, Money Morning, Marketbeat.com, PROFIT CONFIDENTIAL, Trades Of The Day, Wealth Daily, The Growth Stock Wire, TheStockAdvisor, The Wealth Report, Lebed.biz, ProfitableTrading, Wall Street Grand, StockEarnings, Cabot Wealth, Investing Signal, Market Intelligence Center Alert, Dividend Opportunities, Trading Tips, The Best Newsletters, TheStockAdvisors, All about trends, Trading Markets, National Inflation Association, InvestorIntel, AllPennyStocks, Market FN, Energy and Capital, DividendStocks, Wyatt Investment Research, InvestmentHouse, Darwin Investing Network, Money and Markets, Stockhouse, Investment House, Daily Markets, Wall Street Daily, OTC Stock Pick, Buttonwood Research, Street Insider, InvestorsObserver Team, MiningNewsWire, TradingAuthority Daily, Wall Street Greek, Investors Alley, Eagle Financial Publications, Dynamic Wealth Report, Investiv, The Tycoon Report, StreetAlerts, Market Authority, Investing Futures, Investment U, Wealth Insider Alert, FNNO Newsletters, MarketWatch, Trader Jack, Money Wealth Matters, The Stock Enthusiast, 24/7 Trader, StockTwits, Daily Profit, Short Term Wealth, FutureMoneyTrends.com, Trading Concepts, Early Bird, CrushTheStreet.com, CNBC Breaking News, ChartAdvisor, INO Market Report, Investing Daily, Candle Stick Forum, Insider Wealth Alert, Global Equity Alert, Global Equity Report, equities Canada, Total Wealth, SmallCap Network, Super Stock Picker, The Motley Fool, The Street Report, The Wall Street Transcript, One Hot Stock, TipRanks, Rick Saddler, Trade of the Week, UndiscoveredEquities, Vantage Wire, VectorVest, Wall Street Window, Weekly Wizards, The Weekly Options Trader, Penny Detectives, Investor Update, Investors Insights, Jim Cramer, Leeb's Market Forecast, MarketClub, Navellier Growth, Small Cap Firm, OTCPicks, Seeking Alpha, Penny Stock Buzz, PennyStockProphet, Power Profit Trades, Profitable Trader Authority, Profits Run, Investor Guide and Normandy Investment Research reported earlier on Newmont Corporation (NEM), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
After experiencing a continuous decline in prices for nearly six months, gold prices have started to swing in the other direction. Kitco reported that the precious metal was trading at more than $1,970 per ounce earlier this week before closing at $1,922 an ounce, a notable feat after months of decline.
With gold prices rallying in recent weeks, gold-backed exchange-traded funds (ETF) have seen increasingly high returns in the past week as well.
The Sprott Gold Miners ETF had the highest return at 2.81% followed by the Sprott Junior Gold Miners ETF, which posted a 2.07% return. Data from LOGICLY show that the Sprott Gold Miners ETF (SGDM) ranked fourth in the top ten gold ETFs for the week while the Sprott Junior Gold Miners ETF was ninth.
With $257 million in assets and a 0.50 expense ratio, SGDM is a relatively new ETF that has been around for less than a decade. It tracks the Sprott Zacks Gold Miners Index and provides exposure to large-cap firms, or companies with more than $10 billion capitalization, in the gold industry in three economies: the United States, South Africa and Canada.
Canadian companies represent more than 85% of SGDM’s portfolio. The ETF had a return of close to 9% for the 30-day period beyond last week, a 24.03% return over the past 12 months and a 3.19% return in the last four years.
Launched on March 31, 2015, the Sprott Junior Gold Miners ETF (SGDJ) is even younger than SGDM. It is currently managing assets worth $100 million with a 0.50% expense ratio and tracks the Sprott Zacks Junior Gold Miners index to expose investors to small-cap firms in the gold mining ecosystem.
SGDJ tracks gold mining companies in five countries and has an overwhelming presence in Australia. Australian companies account for 50% of SGDJ’s portfolio followed by Canada at 31.16%, the United States at 9.30%, the United Kingdom at 9.01% and Indonesia at 0.53%.
The gold ETF saw its performance surge in October, posting a 7.62% return for the month and outperforming SGDM slightly for the past year.
However, SGDM surpassed Sprott Junior Gold Miners ETF in all the other time frames.
Additionally, both ETFs have experienced negative outflows over the past year but a continued rally in gold prices could reverse the trend. Experts predict that such a continued rally would contribute to sustainable growth among gold ETFs and could potentially reawaken investor interest in gold mining equities.
The rally witnessed in the gold market has undoubtedly had a positive impact on the stocks of major gold mining firms like Newmont Corporation (NYSE: NEM) (TSX: NGT) since share prices usually go up when the commodity enjoys favorable market conditions.
Newmont Corporation (NEM), closed Wednesday's trading session at $37.27, off by 0.53376%, on 13,935,940 volume. The average volume for the last 3 months is 540,738 and the stock's 52-week low/high is $34.81/$60.08.
atai Life Sciences N.V. (ATAI)
QualityStocks, MarketBeat, The Online Investor, StockMarketWatch, Dynamic Wealth Report, StreetInsider, MarketClub Analysis, Small Caps, Uncommon Wisdom, Marketbeat.com, CRWEWallStreet, PennyToBuck, CRWEPicks, InsiderTrades, CRWEFinance, BestOtc, DrStockPick, PennyOmega, Schaeffer's, StockHotTips, TraderPower, Awareness Stocks, StockOodles, Street Insider, The Street, TopPennyStockMovers, Broad Street and ProTrader reported earlier on atai Life Sciences N.V. (ATAI), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Recent advancements in virtual technology may have applications in psychedelic-assisted therapy. Both are relatively new fields that have attracted lots of attention in recent years thanks to their potentially game-changing applications.
Psychedelic-assisted therapy has shown it has the potential to offer long-term relief against a variety of mental-health disorders with few if any side effects. Researchers have found that virtual technology may enhance psychedelic-assisted therapy by simulating the visual phenomenology of psychedelic states. With both fields in their infancy, scientists are focusing their efforts on using virtual reality (VR) as a companion to psychedelic-assisted therapy or a potential nondrug alternative.
The Isness-D study found that interactive group virtual-reality experiences have shown that they can simulate effects similar to psychedelics, including psilocybin (magic mushrooms) and LSD. These results drew the interest of scientists who were already interested in the efficacy of combining VR with psychedelic therapy as well as the ability of VR to mitigate bad trips.
Aside from individuals who simply don’t want to go through a psychedelic experience as part of their treatment, the possibility of bad trips is perhaps the most adverse side effect of psychedelic-assisted therapy.
Isness-D was the brainchild of digital artist, scientist and Intangible Realities Laboratory (IRL) founder David Glowacki, a specialist in scientific stimulation and interactive multiperson virtual reality. After having a life-altering, near-death experience during a mountain hike, Glowacki created Isness-D and used scientific stimulation to recreate the near-death experience.
There is growing evidence that group VR can produce psychedelic-like effects, such as feelings of self-actualization, transcendence and ego death. Participants also reported experiencing feelings of connectedness and epiphanies, feelings that are also induced by numerous psychedelics.
But with most of this research still in its infancy, we still don’t understand how interactive group VR produces psychedelic-like effects or whether it can trigger similar brain mechanisms. In fact, researchers still don’t fully understand the brain mechanisms that allow psychedelics to alleviate mental disorders such as anxiety, treatment-resistant depression, eating disorders and post-traumatic stress disorder (PTSD).
The prevailing theory is that psychedelics stimulate neuroplasticity, the brain’s ability to change and adapt to extrinsic and intrinsic stimuli, and boost brain entropy. Psychologist and chief science officer at VR program Healium Jeff Tarrant says that virtual reality experiences can increase entropy, or the brain’s collective knowledge and information processing capacity. He says that while virtual reality may not replace psychedelic-assisted therapy completely, it may offer alternative approaches that could enhance mental-health treatments.
The public is closely watching these developments as it also keeps an eye on psychedelics startups such as atai Life Sciences N.V. (NASDAQ: ATAI) that are making headway in developing therapeutic formulations from psychedelic compounds.
atai Life Sciences N.V. (ATAI), closed Wednesday's trading session at $1.19, off by 3.252%, on 334,320 volume. The average volume for the last 3 months is 29.637M and the stock's 52-week low/high is $1.05/$3.65.
Marathon Digital Holdings Inc. (MARA)
MarketClub Analysis, InvestorPlace, Schaeffer's, QualityStocks, INO Market Report, StockMarketWatch, MarketBeat, StocksEarning, StockEarnings, TradersPro, BUYINS.NET, The Online Investor, Lebed.biz, Trades Of The Day, Zacks, Marketbeat.com, InvestorsUnderground, Early Bird, Daily Trade Alert, The Street, TraderPower, BillionDollarClub, PoliticsAndMyPortfolio, TopPennyStockMovers, Wall Street Mover, StreetAuthority Daily, FeedBlitz, Kiplinger Today, Wealth Insider Alert, Eagle Financial Publications, Barchart, DreamTeamNetwork, Promotion Stock Secrets, AllPennyStocks, Rick Saddler, Stock Analyzer, Stock Beast, StockOodles, Street Insider, StreetInsider and RedChip reported earlier on Marathon Digital Holdings Inc. (MARA), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
The number of cryptocurrency ATMs worldwide has significantly decreased this year, reaching levels not seen since 2021. According to data gleaned from CoinATMRadar.com, the biggest decline was in July, when an astounding 4,123 ATMs stopped working, seriously disrupting the market.
Presently, thousands of ATMs have become nonfunctional, the majority of which have faded into obscurity this year. CoinATMRadar.com listed 32,704 cryptocurrency ATMs globally as of mid-October. This figure is in sharp contrast to the all-time high of 39,353 units that was recorded in August 2022, indicating a sharp drop in the number of crypto ATMs — roughly 6,649 — that were formerly operational.
The first significant decline occurred in March 2023, which led to 3,568 units becoming obsolete. However, July witnessed an even steeper decline, with 4,123 machines being discontinued. A minor uptick in installations was observed in April and May following the March decline, and this pattern persisted after July’s setback. However, even with more new arrivals in October, the losses from July have not been made up for.
The count as of right now is 6,649 units below the August 2022 peak, or about in line with the November 2021 numbers. When it comes to the leading players in the crypto ATM market, Bitcoin Depot leads the pack with 6,306 ATMs, or 19.3% of all ATMs worldwide. Coinflip comes in second with 4,017 machines, while Bitstop comes in third with 2,753 units. Other players at the top include Athena Bitcoin, Coinhub and Rockitcoin.
More than 32,500 of the world’s crypto ATMs are built to accept Bitcoin, and 18,141 of those can also accept other digital currencies, including Bitcoin cash, litecoin, and ethereum. Furthermore, 2,208 units handle transactions in Circle’s USDC, while 3,586 units serve Tether (USDT) users.
One contributing factor to this decline is the controversy surrounding the potential misuse of these machines for illicit activities. For example, there have been cases of con artists using websites such as eBay to trick customers into using crypto ATMs to deposit money into prearranged cryptocurrency wallets. The con artists disappear as soon as the money is deposited, leaving the customers in the dark.
According to Bitcoin Depot CEO and founder, Brandon Mintz, operators pulling down unproductive ATMs or going out of business altogether may also be to blame for the decline in cryptocurrency ATMs. Mintz sees this trend as an opportunity for his business to increase its market share through the acquisition of more ATMs and a greater presence in more retail spaces.
Given the recent crypto winter, it isn’t surprising that ATMs serving crypto users have declined. But, as entities such as Marathon Digital Holdings Inc. (NASDAQ: MARA) see BTC and other major cryptocurrencies start climbing in price, it is only a matter of time before ATM activity grows again.
Marathon Digital Holdings Inc. (MARA), closed Wednesday's trading session at $8.92, up 1.2486%, on 31,215,338 volume. The average volume for the last 3 months is 21.264M and the stock's 52-week low/high is $3.11/$19.875.
Aurora Cannabis Inc. (ACB)
InvestorPlace, Schaeffer's, MarketBeat, MarketClub Analysis, StocksEarning, The Street, Trades Of The Day, Daily Trade Alert, StockEarnings, StreetInsider, The Online Investor, Wealth Insider Alert, QualityStocks, Market Intelligence Center Alert, Kiplinger Today, StockMarketWatch, CFN Media Group, Investopedia, Stock Up Featured, Profit Trends, BUYINS.NET, BlackSwanAlert, StreetAuthority Daily, The Rich Investor, Jim Cramer, Early Bird, Investors Alley, Cannabis Financial Network News, Wall Street Window, CNBC Breaking News, Daily Profit, Tradespoon, Inside Trading, Outsider Club, TheTradingReport, Zacks, The Wealth Report, Market Intelligence Center, Technology Profits Daily, Money and Markets and Top Pros' Top Picks reported earlier on Aurora Cannabis Inc. (ACB), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
A recently published study shows that engaging in yoga after using cannabis can lead to improved mystical experiences and mindfulness. The study suggests that the surrounding environment and activities can significantly influence a person’s cannabis experience. The study, which forms part of a psychology dissertation submitted at the University of British Columbia, was intended to investigate how contextual factors during marijuana consumption impact overall well-being.
Study author Sarah Elizabeth A. Daniels stated that when using psychoactive substances intended for mental-health treatment, researchers frequently take into account contextual factors outside the direct effects of the drug, such as one’s behavior, mindset and setting. These factors are known to have a substantial impact on therapeutic outcomes, a principle well-recognized in psychedelic research but less so in the context of therapeutic marijuana use. The study’s findings suggest that, similar to psychedelics, the concept of “set & setting” during marijuana use may profoundly influence the therapeutic benefits of the drug.
To assess how context affects the marijuana experience, Daniels conducted a study with 47 participants who self-administered cannabis on two separate occasions, one week apart. In one session, the subjects practiced yoga, and in the other, they took part in their routine activities, which often involved doing housework, eating, socializing or watching TV. The subjects were evaluated on measures including state effect, mystical experience, as well as state mindfulness.
The results showed a significant increase in mystical experiences and mindfulness when participants practiced yoga while using cannabis. On the other hand, there wasn’t a noticeable difference in the participants’ mood and emotional states between the two sessions.
The study findings have potential implications for enhancing the therapeutic use of cannabis as well as recommendations for clinical practice. Daniels pointed out that providing specific behavioral guidelines and psychoeducation on the role of set & setting may be beneficial. The study further suggested that the altered state of consciousness experienced while using cannabis should not be disregarded as it holds significant therapeutic value.
The U.S. government has previously acknowledged the consciousness-changing potential of hallucinogens as a significant side effect, which led to research into pharmaceutical products that mimic their effects without hallucinations. Meanwhile, the combination of yoga plus marijuana has been a long-standing practice within the marijuana community, with classes offering this pairing emerging as early as the initial stages of state-level legalization. These activities have often been praised anecdotally for their benefits.
The findings of this study can help marijuana industry players, such as Aurora Cannabis Inc. (NASDAQ: ACB) (TSX: ACB), to find novel ways in which to position their products in a way that shows customers how best they can get maximum benefits while using those products for different purposes.
Aurora Cannabis Inc. (ACB), closed Wednesday's trading session at $0.4528, up 2.6059%, on 5,745,836 volume. The average volume for the last 3 months is 877,735 and the stock's 52-week low/high is $0.41/$1.62.
The QualityStocks Company Corner
- Renovaro BioSciences Inc. (NASDAQ: RENB)
- Lexaria Bioscience Corp. (NASDAQ: LEXX)
- Genprex Inc. (NASDAQ: GNPX)
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF)
- SuperCom Ltd. (NASDAQ: SPCB)
- SenesTech Inc. (NASDAQ: SNES)
- Sekur Private Data Ltd. (FRA: GTD0) (CSE: SKUR) (OTCQB: SWISF)
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF)
- Mountain Top Properties Inc. (OTC: MTPP)
- PaxMedica Inc. (NASDAQ: PXMD)
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI)
- Longeveron Inc. (NASDAQ: LGVN)
Renovaro BioSciences Inc. (NASDAQ: RENB)
The QualityStocks Daily Newsletter would like to spotlight Renovaro BioSciences Inc. (NASDAQ: RENB) .
Hepatitis B is a liver infection caused by the hepatitis B virus. This disease, which can be prevented via vaccine, spreads when blood or other body fluids from an infected individual enter the body of another. Having chronic hepatitis B increases an individual's risk of developing cirrhosis, liver cancer or liver failure. New research, which evaluated a cohort of adult veterans in America, has determined that only about one-half started on antiviral therapy. The study was led by clinical associate professor Robert J. Wong of the Stanford University School of Medicine. The researchers' aim was to understand chronic hepatitis B-related cirrhosis better and determine if there were certain predictors that made patients susceptible to holdups in treatment of said infection. For their study, Wong and his colleagues identified 2,550 veterans with chronic hepatitis B cirrhosis via data from the Veterans Affairs Corporate Data Warehouse, for the period between 2010–2022. Enterprises such as Renovaro BioSciences Inc. (NASDAQ: RENB) are working to develop effective treatments that can eliminate HBV so that patients don't have to live with the long-term complications that often result from this infection.
Renovaro BioSciences Inc. (NASDAQ: RENB), formerly Enochian BioSciences Inc., is an advanced, pre-clinical biotechnology firm in cell, gene and immunotherapy focused on solid tumors with short life expectancy. The company aims to unlock potentially long-term or life-long cancer remission in some of the deadliest cancers, and to potentially treat or cure serious infectious diseases such as Human Immunodeficiency Virus (HIV) and Hepatitis B Virus (HBV) infection.
The oncology platform is now at the forefront of Renovaro’s development activities. While Renovaro’s current efforts focus primarily on pancreatic cancer, it plans to include other solid tumors with short life-expectancy in the first in human Phase I/IIa studies that are on track to start by mid-2024. The company’s Pre-Investigational New Drug (pre-IND) submission included a human study plan covering pancreatic cancer, as well as other cancers that are difficult to treat, potentially including triple-negative breast cancer, head and neck cancers and mesothelioma.
Renovaro’s proprietary, novel technology uses cell- and gene-therapy to promote a renewed immune response against solid tumors. Important confirmatory results from two humanized mouse models using the company’s novel dendritic cell-based therapy, independently conducted by Dr. Anahid Jewett, a renowned cancer researcher in the field of immunotherapy at UCLA, were presented previously at two scientific conferences and were the foundation supporting a pre-IND submission to the U.S. Food and Drug Administration. Notably, Dr. Jewett’s findings from these studies consistently demonstrated 80% to 90% pancreatic tumor reduction in size and weight that was correlated with significant enhancement of key aspects of the immune response.
Renovaro is headquartered in Los Angeles, California.
RENB-DC11
Renovaro’s product development strategy is anchored in the use of “non-self” or allogeneic cells that enhance targeted immune response. Its lead candidate, RENB-DC11, is an innovative therapeutic vaccination platform that could potentially be used to induce life-long remissions from some of the deadliest solid tumors.
Treatment with RENB-DC11 has now been shown to significantly reduce the size of human pancreatic tumors in humanized mice in three independent studies. The reduction in tumor size correlated with statistically significant increases in key components of an immune response.
Pre-IND was completed in June 2023, with IND filing forecast for first half of 2024. First in-human Phase I/IIa trials are predicted shortly after in H1 of 2024, including pancreatic and other solid tumors with poor treatment options and life-expectancy.
Renovaro believes that RENB-DC11 could represent the most promising and effective strategy to achieve life-long remission for a number of common and deadly tumors.
Other Development Candidates
In addition to its lead oncology platform, Renovaro’s development pipeline includes a platform targeting infectious diseases, including:
- RENB-HV12 – An engineered allogeneic T-Cell vaccine, this therapeutic HIV vaccine candidate enhances immune infiltration, immune killing and immune surveillance. Potential pre-IND submission is planned for first half of 2024, with IND-submission expected in second half of 2024.
- RENB-HV21 – Leveraging allogeneic NK plus Gamma Delta T (GDT) cells as potential therapy for HIV, ENOB-HV21 shows promising preliminary results without confounding factors. Renovaro owns an exclusive license and has completed the Pre-IND submission, with a potential IND submission and human trials expected in 2024.
- RENB-HV01 – Caring Cross, a non-profit corporation, has shown that its proprietary CAR-T cells cure HIV in a mouse model. Studies in humans have begun. Renovaro has entered into a profit-sharing sublicense with Caring Cross and would share in profits if the product is commercialized.
- RENB-HB01 – This therapeutic approach aims to eliminate all HBV rapidly (“seek and kill”) with a two to three dose treatment regimen. It is expected to be applicable for early disease to maximize impact with low risk of toxicity. Pre-IND comments have been received from the FDA for its AAV-delivery system.
LOI to Merge with GEDi Cube International Ltd.
On August 9, 2023, Renovaro announced its execution of a binding, exclusive letter of intent to merge a subsidiary with cutting-edge health AI company GEDi Cube International Ltd. The combined company is expected to create a potential multiplier effect to accelerate earlier diagnosis, more effective therapy, and precision in silico drug discovery.
GEDi Cube’s innovative technology, developed over nearly a decade, has already validated earlier diagnoses of lung cancer in humans at a leading university hospital. GEDi Cube has likewise created the early diagnosis technology for 12 additional cancers, including pancreatic and breast cancer.
“I believe joining forces with GEDi Cube could enhance the efficacy of our upcoming trials and speed up the discovery of novel treatment approaches, thereby extending our life-saving technology to more cancer patients and renewing hope for them and their families,” Dr. Mark Dybul, CEO of Renovaro, stated in the news release.
GEDi Cube is led by CEO Craig Rhodes, who brings to that company tremendous industry experience leading life sciences groups at industry leaders Intel, Oracle and NVIDIA.
Market Opportunity
Pancreatic cancer alone is diagnosed globally in approximately 495,000 people each year, including roughly 64,000 in the U.S. Nearly 466,000 of those patients die annually, including approximately 51,000 in the U.S. Because of limited treatment options, life expectancy is very poor – with an approximately 10% patient survival rate at five years after diagnosis.
The global pancreatic cancer treatment market was valued at $2.15 billion in 2021 and is projected to grow from $2.48 billion in 2022 to $6.85 billion by 2029, according to Fortune Business Insights. That growth represents a CAGR of 15.7% for the forecast period.
A separate report from Fortune Business Insights projects that the global HIV drug market will grow from $30.46 billion in 2021 to $45.58 billion in 2028, recording a CAGR of 5.9% over the forecast period.
According to GlobalData, the value of the market for hepatitis B treatment is forecast to experience a significant increase in the coming years, with revenues expected to grow from $1.6 billion in 2022 to $10.5 billion in 2029. That represents a very rapid CAGR of 30% over the period. An estimated 296 million people suffer from the condition worldwide.
Management Team
Dr. Mark Dybul is the CEO of Renovaro. He has served as a tenured professor in the Department of Medicine at Georgetown University Medical Center since June 2017. He also served as Faculty Co-Director of the Center for Global Health and Quality from 2017-2021. Dr. Dybul has worked on HIV and public health for nearly 30 years as a clinician, scientist, teacher and administrator, including as an architect and eventually the Global Ambassador of the U.S. President’s Emergency Plan for AIDS Relief and the Executive Director of the Global Fund to Fight AIDS, Tuberculosis and Malaria from 2013 through May of 2017, and as the co-director of the Global Health Law Program at the O’Neill Institute for National and Global Health Law from 2009 through 2012. He is a member of the U.S. National Academy of Medicine.
Luisa Puche is the company’s CFO. She has served as a senior accounting and financial advisor and president of Puche Group LLC from 2015-2019. She served in various key executive roles, including Interim Chief Accounting Officer, at Brightstar Corp., a $10 billion global wireless device services provider. Ms. Puche began her career at Ernst & Young, where she served for approximately 10 years. Leveraging her broad global audit, advisory and corporate expertise, she has provided strong cross-functional leadership experience managing small and large projects for both publicly traded and privately held companies in various industries, including a global implementation of the latest revenue recognition accounting standard for Del Monte, as well as the global implementation of their SOX-404 program.
Francois Binette, Ph.D., is the Chief Operating Officer and Executive Vice President of Research & Development at Renovaro. He has over 25 years of product development expertise in Advanced Therapies and Regenerative Medicine. His broad industry experience spans a wide range of serious medical conditions, from orthopedics to ophthalmology, CNS and immuno-oncology. His career includes positions at Genzyme, Biosyntech, the DePuy Franchise of Johnson and Johnson, Medtronic and Lineage Cell Therapeutics. He received his Ph.D. from Laval University in Québec, followed by post-doctoral training at the Sanford-Burnham Institute in La Jolla and Harvard Medical School in Boston.
Renovaro BioSciences Inc. (NASDAQ: RENB), closed Wednesday's trading session at $2.9, up 2.1127%, on 71,816 volume. The average volume for the last 3 months is 94,592 and the stock's 52-week low/high is $0.3928/$5.25.
Recent News
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Alarming Data Highlights Gaps in Treating People with Chronic HBV
- Biotech Firms Register Successes in Quest for Better Pancreatic Cancer Treatments
- Renovaro BioSciences Inc. (NASDAQ: RENB) CEO Provides Insights on Definitive Agreement to Combine with GEDi Cube to Transform Medicine
Lexaria Bioscience Corp. (NASDAQ: LEXX)
The QualityStocks Daily Newsletter would like to spotlight Lexaria Bioscience Corp. (NASDAQ: LEXX).
NetworkNewsWire Editorial Coverage: GLP-1 agonists have become incredibly popular obesity drugs, reshaping the standard of care as people increasingly turn to the new class of drugs in lieu of conventional weight-loss procedures. There is a growing body of clinical evidence showing GLP-1 agonists safely control blood sugar (glucose) levels and deliver tremendous positive outcomes, such as a recent New England Journal of Medicine study reporting weight loss of 49 to 52 pounds. As good as GLP-1 agonist are, they can get better, which is part of the mission of Lexaria Bioscience Corp. (NASDAQ: LEXX) (Profile), a global innovator in drug-delivery platforms. The company started diabetes-related formal studies in 2022 evaluating its DehydraTECH(TM) platform, which showed lower blood-sugar levels and decreased body weight. This compelling data is undergirding the next phase of research, new human and animal studies for the purpose of demonstrating DehydraTECH processing can make GLP-1 drugs even more effective and more accessible. Positive developments on DehydraTECH could catch the attention of companies dedicated to developing drugs that support better health— companies such as Johnson & Johnson (NYSE: JNJ), Sanofi (NASDAQ: SNY), Amgen (NASDAQ: AMGN), and Novartis AG (NYSE: NVS).
Lexaria's DehydraTECH is a platform technology that seamlessly integrates in manufacturing processes to enhance delivery of API payloads, subsequently improving pharmacokinetics and reduce side effects
Lexaria is researching the effect of DehydraTECH on oral performance of GLP-1 drugs for diabetes and weight loss, markets expected to climb to $200 billion annually
DehydraTECH is scalable and shown to have utility for a multitude of applications, including oral nicotine products, for which Altria has secured rights from Lexaria
DehydraTECH promotes rapid uptake and better efficacy through less expensive, oral administration, as validated through laboratory and clinical testing
Lexaria Bioscience Corp. (NASDAQ: LEXX) is a global innovator in drug delivery platforms. The company’s patented technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules. DehydraTECH promotes fast-acting, less expensive and more effective oral drug delivery and has been thoroughly evaluated through in vivo, in vitro and human clinical testing.
DehydraTECH is covered by 21 issued and more than 50 pending patents in over 40 countries around the world. Lexaria’s first patent was issued by the U.S. Patent and Trademark Office in October 2016 (US 9,474,725 B1), providing 20 years of patent protection expiring June 2034. Multiple patents have been awarded since then and are expected in the future.
Lexaria has also collaborated with the National Research Council (NRC), the Canadian government’s premier research and technology organization. The company has been granted patent protection for specific delivery of nicotine, vitamins, NSAIDs, antiviral drugs, cannabinoids and more.
Lexaria began developing DehydraTECH in 2014 and has since continued to strengthen and broaden the technology. The company has no plans to create or sell Lexaria-branded products containing controlled substances. Instead, Lexaria licenses its technology to other companies around the world to offer consumers the best possible performance across an array of ingestible product formats.
The company’s technology is best thought of as an additional layer that providers of consumer supplements, prescription and non-prescription drugs, nicotine and CBD products can utilize to improve the effectiveness of their own existing or planned new offerings. Lexaria has licensed DehydraTECH to multiple companies, including a world-leading tobacco producer for the research and development of smokeless, oral-based nicotine products, and for use in industries that produce cannabinoid beverages, edibles and oral products.
DehydraTECH is suitable for use with a wide range of product formats including pharmaceuticals, nutraceuticals, consumer packaged goods and over-the-counter capsules, pills, tablets and oral suspensions.
DehydraTECH Technology
Lexaria’s DehydraTECH is designed specifically for formulating and delivering lipophilic (fat-soluble) drugs and active ingredients. DehydraTECH increases their effectiveness and improves the way active pharmaceutical ingredients enter the bloodstream. The major benefits to a subject ingesting a DehydraTECH-enabled drug or consumer product can be summarized by the following:
- Speeds up delivery – the effects of the product are felt by the subject in just minutes.
- Increases bioavailability – the technology is much more effective at delivering a drug or product into the bloodstream.
- Increases brain absorption – animal testing suggests significant improvement in the quantity of drug delivered across the blood-brain barrier.
- Improves drug potency – more of the ingested product is made available to the body, so lower doses are required to achieve the desired effect.
- Reduces drug administration cost – lower doses mean lower overall drug costs.
- Masks unwanted taste – the technology eliminates or reduces the need for sweeteners.
Lexaria has demonstrated in animal studies a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain barrier by as much as 1,900 percent, initiating additional new patent applications and opening possibilities for improved drug delivery.
Since 2016, DehydraTECH has repeatedly demonstrated, with cannabinoids and nicotine, the ability to increase bio-absorption by up to five to 10 times, reduce time of onset from one to two hours to just minutes, and mask unwanted tastes. The technology is to be further evaluated for additional orally administered bioactive molecules, including antivirals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs) and nicotine.
Market Outlook
Lexaria’s ongoing research and development efforts are mainly focused on development of product candidates across several key segments:
- Oral Cannabinoids – a market estimated to be worth $18.4 billion in 2021 and expected to reach $46.2 billion by 2025.
- Antivirals – an estimated $52.1 billion market in 2021 that’s expected to grow to $66.7 billion by 2025.
- Oral Mucosal Nicotine – smokeless tobacco products, a $13.6 billion market in 2018, is forecast to grow at 7.2 percent annually through 2025.
- Human Hormones – estrogen and testosterone replacement therapies represented a $21.9 billion market in 2019, with a forecast CAGR of 7.7 percent through 2027.
- Ibuprofen and Naproxen – NSAID sales totaled $15.6 billion globally in 2019 and are projected to reach $24.4 billion by 2027.
- Vitamin D3 – the global market size was $1.1 billion in 2021, growing at 7 percent per year and expected to reach $1.7 billion in 2026.
Management Team
Chris Bunka is Chairman and CEO of Lexaria Bioscience Corp. He is a serial entrepreneur who has been involved in several private and public companies since the late 1980s. He has extensive experience in the capital markets, corporate governance, mergers and acquisitions, as well as corporate finance. He is named as an inventor on multiple patent innovations.
John Docherty, M.Sc., is the President of Lexaria. He is a pharmacologist and toxicologist, and a specialist in the development of drug delivery technologies. He is the former president and COO of Helix BioPharma Corp. (TSX: HBP). He is named as an inventor on multiple issued and pending patents.
Greg Downey is Lexaria’s CFO. He has more than 35 years of diverse financial experience in the mining, oil and gas, manufacturing, and construction industries, and in the public sector. He served for eight years as CFO for several public companies and has provided business advisory and financial accounting services to many large organizations.
Gregg Smith is a strategic advisor to Lexaria. He is a founder and private investor with Evolution VC Partners. He is a member of the Sand Hill Angels and held previous investment banking roles with Cowen and Company and Bank of America Merrill Lynch.
Dr. Philip Ainslie serves as a scientific and medical advisor to Lexaria. He is co-director for the Centre for Heart, Lung and Vascular Health, Canada. He is also Research Chair in Cerebrovascular Physiology and Professor at the School of Health and Exercise Sciences, Faculty of Health and Social Development at the University of British Columbia.
Lexaria Bioscience Corp. (LEXX), closed Wednesday's trading session at $1.45, up 5.0725%, on 153,868 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $5.25/$.
Recent News
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Get Ready for Next Generation of Diabetes, Weight-Management Drugs
- Lexaria Bioscience Corp. (NASDAQ: LEXX) Grows List of Licensed Partners Following Exclusive Collaboration and License Agreement with SulfoSyn
- Can Diabetes, Weight-Loss Drugs Get Even Better?
Genprex Inc. (NASDAQ: GNPX)
The QualityStocks Daily Newsletter would like to spotlight Genprex Inc. (NASDAQ: GNPX).
Genprex (NASDAQ: GNPX), a clinical-stage gene therapy company focused on developing life-changing treatments for patients with cancer and diabetes, today announced that Catherine Vaczy, its EVP, general counsel and chief strategy officer, will be providing an overview of the company's gene therapies for cancer and diabetes at the upcoming BIO-Europe 2023 Conference taking place in Munich, Germany. Vaczy's presentation is slated to begin at 4:45 p.m UTC on Tuesday, Nov. 7, 2023. Several members of Genprex's executive leadership team will be in attendance at BIO-Europe 2023. Those interested in meeting Genprex management during the event should request a meeting through the conference portal or reach out to investor relations at investors@genprex.com.
To view the full press release, visit https://ibn.fm/8RGAC
Genprex Inc. (GNPX) is a clinical-stage gene therapy company developing potentially life-changing technologies for cancer patients based upon a unique proprietary technology platform, including Genprex’s initial product candidate, Oncoprex™ immunogene therapy for non-small cell lung cancer (NSCLC). Genprex’s platform technologies are designed to administer cancer-fighting genes by encapsulating them into nanoscale hollow spheres called nanovesicles, which are then administered intravenously and taken up by tumor cells where they express proteins that are missing or found in low quantities.
Research and Development
Genprex holds a portfolio of 30 issued and two pending patents covering its technologies and targeted molecular therapies. The company’s research and development program is focused on identifying and developing leading-edge gene therapies that can be used alone or in combination with other therapies for treatment of cancer.
Genprex’s initial product candidate is Oncoprex™, an immunogene therapy for the treatment of non-small cell lung cancer (NSCLC). Oncoprex works by interrupting cell signaling pathways that cause replication and proliferation of cancer cells, re-establishes pathways for apoptosis (or programmed cell death) in cancer cells, and modulates the immune response against cancer cells. Oncoprex has also been shown to block mechanisms that create drug resistance.
Preclinical research is being conducted with the goal of developing Oncoprex to be administered with targeted therapies in other solid tumors, and with immunotherapies in NSCLC and other solid tumors. In addition, Genprex has conducted and plans to continue research into other tumor suppressor genes associated with chromosome 3p21.3, as well as other potential applications of the company’s immunogene therapy platform.
Clinical Trials
Genprex is currently conducting the second phase of a phase I/II clinical trial at the University of Texas MD Anderson Cancer Center in Houston. The company plans to expand its clinical program by adding a new clinical study evaluating Oncoprex™ in combination with a checkpoint inhibitor for treatment of Stage IV or recurrent NSCLC. In research presented at the 2017 Annual Meeting of the American Association of Cancer Research in Washington, D.C., Genprex’s collaborators showed that TUSC2 in combination with PD-1 checkpoint inhibition has a significantly greater anti-tumor effect in lung cancer than either agent alone. The research also shows that TUSC2 in combination with PD-1 blockade has synergistic activity in upregulating natural killer (NK) cells, correlating with prolonged survival in mice.
TUSC2 (Tumor Suppressor Candidate 2) is a tumor suppressor gene that is absent or deficient in cancer cells of many different cancer types.
The Market
Genprex technologies seek to bridge a critical gap by combining with targeted therapies and immunotherapies to provide treatments to large patient populations who would otherwise not be candidates for those therapies or who have become resistant to them. Genprex technologies are being developed to overcome genomic limitations which are inherent in targeted therapies and immunotherapies in order to provide new treatment solutions to large cancer populations, such as those with lung cancer.
Each year, more people die of lung cancer than of colon, breast and prostate cancers combined. NSCLC is the most common type of lung cancer, accounting for about 85 percent of all lung cancers, according to the American Cancer Society (“ACS”). Despite radical advances in drug development and novel therapeutic standards, survival for late stage lung cancer has not improved significantly in the past 25 years.
Senior Management
Chairman and Chief Executive Officer J. Rodney Varner, JD, is a co-founder of Genprex and has served in these roles since August 2012. He has more than 35 years of legal experience with large and small law firms and as outside general counsel of a Nasdaq-listed company. Varner has served as counsel in company formation, mergers and acquisitions, capital raising, other business transactions, protection of trade secrets and other intellectual property, real estate, and business litigation. He is a member of the State Bar of Texas and has been admitted to practice before the U.S. Court of Appeals for the Fifth Court and the U.S. Tax Court.
Julien L. Pham, M.D., MPH, is president and chief operating officer of Genprex. In March 2013, Dr. Pham co-founded RubiconMD, a healthcare IT company that connects primary care providers to specialists for additional guidance and opinions on medical cases and served as its chief medical officer. He has served on the faculty at Harvard Medical School’s Brigham and Women’s Hospital and is a board-certified internal medicine doctor and nephrologist.
Ryan M. Confer, MS, has served as Genprex chief financial officer since September 2016. Confer has more than 10 years of executive experience in planning, launching, developing, and growing emerging technology companies and has served in the chief operating and chief financial roles for non-profit and for-profit entities since 2008. Confer has also served as an international business development consultant for the University of Texas at Austin’s IC2 Institute, where he focused on evaluating the commercialization potential of nascent technologies in domestic and international markets applicable to technology incubator programs associated with the University. Confer holds a BS in finance and legal studies from Bloomsburg University of Pennsylvania and an MS in technology commercialization from the McCombs School of Business at the University of Texas at Austin.
Jan Stevens, RN, is vice president of Clinical Operations. Stevens has nearly 20 years of comprehensive clinical operations experience in the biopharma industry and a specialization in early-to-late stage oncology companies. Stevens joined the company to help support the various clinical development programs for Oncoprex™.
Genprex Inc. (NASDAQ: GNPX), closed Wednesday's trading session at $0.224, up 1.3575%, on 316,511 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.1903/$1.94.
Recent News
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) to Present at BIO-Europe 2023
- InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) Schedules, Hosts Virtual Key Opinion Leader Event
- InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) to Unveil Encouraging Data on REQORSA(R) for Lung Cancer Treatment at 2023 AACR-NIH-EORTC Conference
Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF)
The QualityStocks Daily Newsletter would like to spotlight Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF).
Eloro Resources (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM), an exploration and mine-development company with a portfolio of gold and base-metal properties in Bolivia, Peru and Quebec, has completed a metallurgical drill program at its Iska Iska silver-tin polymetallic project in the Potosi Department of southwestern Bolivia. The company is now conducting a definition drill program designed to fill-in major gaps in the block model as well as expand the higher-grade zone both along and across strike. According to the announcement, work will also be completed on improving tin recovery for future use, which is a major, very underexplored, exploration target at Iska Iska. "This new drilling program will be an important part of our PEA, which commenced with a site visit by the preliminary economic assessment (‘PEA') team in late September," said Eloro Resources CEO Tom Larsen in the press release. "The cascade ‘ore-sorting' tests at TOMRA will be a more thorough investigation of the applicability of the Santa Barbara mineralization for ‘XRT ore sorting' and provide additional preconcentrated products for further metallurgical tests to optimize recoveries. Definition drilling is in progress to expand the shallow higher-grade mineral resource which has the potential to provide an early payback in the PEA."
To view the full press release, visit https://ibn.fm/sr0bf
Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) is a publicly traded exploration and mine development company with a portfolio of gold and base-metal properties in Bolivia, Peru and Quebec.
The company has an option to acquire a 99% interest in the highly prospective Iska Iska Property, classified as a silver-tin polymetallic epithermal-porphyry complex, a significant mineral deposit type in the Potosi Department of southern Bolivia. Iska Iska is a road-accessible, royalty-free property.
Eloro also owns an 82% interest in the La Victoria Gold/Silver Project, located in the North-Central Mineral Belt of Peru, some 50 kilometers south of Barrick’s Lagunas Norte Gold Mine and Pan American Silver’s La Arena Gold Mine. La Victoria consists of eight mining concessions and eight mining claims encompassing approximately 89 square kilometers. La Victoria has good infrastructure, with access to road, water and electricity, and is located at an altitude that ranges from 3,150 meters to 4,400 meters above sea level.
The company has a strong management and technical team working diligently to uncover the value of both Iska Iska and La Victoria. Eloro is based in Toronto, Canada.
Projects
Iska Iska – Potosi, Bolivia
Iska Iska is associated with a Miocene possibly collapsed/resurgent caldera, emplaced on Ordovician age rocks with major breccia pipes, dacitic domes and hydrothermal breccias. The property is wholly controlled by the title holder, Empresa Minera Villegas S.R.L. It is located 48 kilometers north of Tupiza city, in the Sud Chichas Province of the Department of Potosi. This is an important mineral deposit type in the prolific South Mineral Belt of Bolivia. Eloro commissioned a NI 43-101 Technical Report on Iska Iska, which was completed by Micon International Limited and is available on Eloro’s website and under its filings on SEDAR.
A fully financed drill program is currently underway on the property, situated near world-class deposits including Silver Sand, San Bartolomé, Pulacayo, San Cristobal, San Vicente, Chorolque, Tasna, Choroma and Siete Suyos. Iska Iska is in the southwest part of the Eastern Cordillera, which hosts a number of major polymetallic mines and mineral deposits. Drilling and continuous channel sampling results have demonstrated some very high metal values, especially silver and tin, within an immense system, where mineralization has been encountered in every drill hole to date. The company believes there is excellent potential for world-class bulk mineable deposits.
La Victoria – Ancash, Peru
The La Victoria project, targeting gold and silver production, is situated near world-class, low-cost gold producers Pan American Silver and Barrick Gold Corporation. Located in Ancash Department, La Victoria sits on the western slopes of the Peruvian Andes. The property is located 12 hours from Lima, with a travel distance of 600 kilometers. The nearest road accessible population centers from La Victoria are Huandoval, Pallasca and Cabana. The project includes four principal mineralized zones in Peru’s prolific North-Central Mineral Belt – San Markito, Victoria, Victoria South and Ccori Orcco – with excellent potential for gold discovery. Operations at La Victoria are planned to proceed with a 2,000-meter diamond drilling program to test targets to outline potential resources at San Markito. Trenching and sampling confirmed high silver values and veins at San Markito in 2020.
Market Outlook
According to industry association The Silver Institute, the outlook for silver demand is exceptionally promising, with global demand forecast to rise to a record high of 1.112 billion ounces in 2022. The increase will be driven by record silver industrial fabrication, which is forecast to improve by 5%, as silver’s use expands primarily in solar energy and electric vehicle (EV) manufacturing. The institute states that government commitments to carbon neutrality have resulted in a rapid expansion of green energy projects, driving record photovoltaic panel installations which are expected to lift silver demand in this segment to an all-time high in 2022.
Rising demand in the electronics industry is also boosting the demand for tin, which is primarily used in solder. The electronics and electrical industries use solders containing 40-70% tin, which provide strong and reliable joints under a variety of environmental conditions. At present, the majority of the assemblers are using patented tin-and-copper-based solders. Mordor Intelligence estimated tin demand at 387 kilotons in 2021 and forecasts demand growth of 2.5% annually through 2027. Over the medium term, surging demand from the EV market and increasing applications in the electrical and electronics industry is expected to drive the market.
Management Team
Thomas G. Larsen is CEO of Eloro. He has more than 40 years of experience in the investment industry, specializing in corporate finance and management of junior resource companies, raising in excess of C$200 million. He previously held the position of President and Chief Executive Officer of Champion Iron Limited. Prior to that, he was President and Chief Executive Officer of Champion Iron Mines Limited.
Dr. Bill Pearson is Executive VP of Exploration for Eloro. He has more than 40 years of direct experience in the exploration and production of minerals worldwide. He played an integral role in the acquisitions of Desert Sun Mining Corp. by Yamana Gold in 2006 and Central Sun Mining by B2 Gold in 2009. He was formerly VP Exploration at Desert Sun Mining and Senior VP at Central Sun Mining.
Miles Nagamatsu, CPA, is CFO at Eloro. He has over 30 years of experience in accounting, management, lending, restructurings and turnarounds. Since 1993, he has acted as a CFO of public and private companies primarily in the mineral exploration and investment management sectors. He holds a Bachelor of Commerce degree from McMaster University.
Osvaldo Arce Burgoa is General Manager at Eloro. He is a geological and mineral processing engineer with 26 years of experience in Bolivia. He is a former President of the Bolivian Geological Society, Main Technical Advisor of the National Mining Corporation (COMIBOL) and has served as exploration manager and chief geologist at various mining and exploration companies. He has authored two books on Bolivian geology and holds a doctorate in mining engineering from Tohoku University in Sendai, Japan.
Eloro Resources Ltd. (OTCQX: ELRRF), closed Wednesday's trading session at $1.09, off by 3.5398%, on 10,178 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.089/$3.135.
Recent News
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Reports on Progress at Bolivian Iska Iska Project
- Eloro Resources Announces Completion of Metallurgical Holes and Updates Progress on Definition Drill Program to Expand Higher Grade Mineral Resource for PEA at Iska Iska Project, Potosi Department, Southwestern Bolivia
- How Junior Mining Firms Can Leverage Current Market Conditions
SuperCom Ltd. (NASDAQ: SPCB)
The QualityStocks Daily Newsletter would like to spotlight SuperCom Ltd. (NASDAQ: SPCB) .
The UK's criminal justice department recently announced that domestic abusers leaving prison will be wearing electronic monitoring tags for better victim protection
Up to 500 offenders will be forced to wear GPS and curfew tags to monitor their movements, with those who breach the stipulated license conditions being returned to prison
This move by the UK validates digital monitoring solutions such as SuperCom's Electronic Monitoring ("EM") products
It also shows the technology's viability in solving emerging issues while also validating SuperCom's research and development efforts as targeting an active and growing world market
SuperCom (NASDAQ: SPCB), a global provider of traditional and digital identity solutions offering advanced safety, identification, and security products and solutions to governments, is constantly pushing the envelope with innovation and exploring new markets for its products. This ambition has been driven by its mission and commitment to "creating safer communities". More nations globally are beginning to realize the need for more advanced technologies to manage offender populations, representing a significant growth opportunity for SuperCom.
SuperCom Ltd. (NASDAQ: SPCB) provides secured solutions for the e-government, IoT and cybersecurity sectors. Since 1988, the company has been a trusted global provider of traditional and digital identity offerings, providing cutting-edge electronic and digital security solutions to governments and organizations, both private and public, around the world.
SuperCom’s mission is to revolutionize the public safety sector worldwide through proprietary electronic monitoring technology, data intelligence, and complementary services.
The company is headquartered in Tel Aviv, Israel, with offices in California and other regions in the U.S.
Business Units
IoT and Connectivity
SuperCom IoT products and solutions provide advanced electronic monitoring solutions and services to criminal justice agencies, enabling customers to detect unauthorized movement of people, vehicles, and other monitored objects. The company provides an all-in-one, field-proven PureSecurity offender monitoring suite, accompanied by services such as GPS monitoring, home detention, domestic violence prevention, and more. The company’s services are specifically tailored to meet each client’s needs.

SuperCom’s proprietary Puresecurity suite of hardware, connectivity, and software components is the foundation for its criminal justice services and offerings. SuperCom is leveraging its extensive technology expertise to implement groundbreaking artificial intelligence (AI) technologies into various parts of its core offerings. By leveraging the power of AI, SuperCom’s PureSecurity platform can offer new abilities, such as amplified data analysis, predictive modeling, and streamlined automation – all geared toward optimizing decision-making and operational efficiency.
Competitive advantages of SuperCom’s technology include:
- Long Battery Life (No Tag Charging Required)
- Ultra Lightweight Form Factor
- Next-Gen Location Tech
- Protection of Domestic Violence Victims
- And More
Cybersecurity
In 2015, SuperCom identified the cybersecurity market as a fast-growing space with significant advantages due to synergistic technologies and a shared customer base with its e-Gov and IoT business units. Consequently, SuperCom strategically acquired Prevision Ltd., a company with a strong presence in the market and a broad range of competitive cybersecurity services.
During the first quarter of 2016, SuperCom acquired Safend Ltd., an international provider of cutting-edge endpoint data protection guarding against corporate data loss and theft through content discovery and inspection, encryption methodologies, and comprehensive device and port control.
Both acquisitions significantly expanded the breadth of the company’s global cybersecurity capabilities.
e-Gov
Through proprietary e-government platforms and innovative solutions for traditional and biometrics enrollment, personalization, issuance, and border control services, SuperCom has helped governments, and national agencies design and issue secured multi-identification, or Multi-ID, documents and robust digital identity solutions to their citizens, visitors, and lands.
The company has focused on expanding its activities in the traditional identification, or ID, and electronic identification, or e-Gov, markets, including the design, development, and marketing of identification technologies and solutions to governments in Europe, Asia, America, and Africa using SuperCom’s e-Government platforms.
Market Opportunity
Data from Berg Insight estimates the market for electronic monitoring solutions will grow from $1.2 billion in 2021 to $2.1 billion in 2026, marking a CAGR of 10.8% for the forecast period.
High recidivism rates, prison overcrowding, and soaring incarceration costs are some factors that are driving the electronic monitoring of offenders’ market growth.
An analysis by ReportLinker forecasts that the global cybersecurity market will grow from an estimated value of $173.5 billion in 2022 to $266.2 billion by 2027, achieving a CAGR of 8.9% for the period.
The increased number of data breaches worldwide, the ability of malicious actors to operate from anywhere in the world, the links between cyberspace and physical systems, and the difficulty of reducing vulnerabilities and consequences in complex cyber networks are some factors driving the cybersecurity market growth.
Management Team
Ordan Trabelsi is President and CEO of SuperCom. He has over 15 years of experience as CEO, growing high-tech companies globally. He also has experience in research and development and product innovation, as well as hands-on experience in cybersecurity, encryption, advanced mathematics, and mobile and internet network technologies. Prior to joining SuperCom, he served as co-founder and CEO of Klikot Inc., a global social networking company. He holds an MBA from Columbia University and a B.Sc. in Computer Engineering from The Technion: Israel Institute of Technology.
Barak Trabelsi is COO of SuperCom. He has expertise in big data, cyber, mobile, and internet network technologies, as well as extensive experience in product development and strategies. Prior to joining SuperCom, he served as Senior Product Manager at Equinox Ltd. Before that, he served for four years as VP of R&D at Sigma Wave, a wireless, security, and internet-focused company. He holds a B.Sc. in Computer Science and Business, as well as an MBA from Tel Aviv University.
Gil Alfi is VP of Sales at Safend Ltd., SuperCom’s cybersecurity subsidiary. He joined SuperCom in 2016 as VP of Business Development for Safend. He has more than 18 years of experience in technology companies. He served as an R&D team technology lead for more than seven years and as Director of Product Management for various telecom and wireless companies for more than 10 years. Prior to joining SuperCom, he served as Regional Sales Director at Safend, managing sales regions in Europe and Africa. He holds a B.Sc. in Computer Science and Mathematics and an M.Sc. in Computer Science from Bar-Ilan University.
SuperCom Ltd. (NASDAQ: SPCB), closed Wednesday's trading session at $0.46, off by 1.0753%, on 345,984 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.3416/$2.695.
Recent News
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Eyes the Growing World Market for Domestic Violence Offender Monitoring Solutions
- InvestorNewsBreaks - SuperCom Ltd. (NASDAQ: SPCB) Extends Collaboration with Romanian Government on Pivotal EM Initiative
- SuperCom Ltd. (NASDAQ: SPCB) to Deploy Its Domestic Violence Monitoring Solution in Finland, Supporting the Country's Recently Strengthened Law on Restraining Orders
SenesTech Inc. (NASDAQ: SNES)
The QualityStocks Daily Newsletter would like to spotlight SenesTech Inc. (NASDAQ: SNES).
SenesTech (NASDAQ: SNES), the leader in fertility control to manage animal pest populations, announced that it is now taking orders from commercial customers for Evolve(TM) Soft Bait ("Evolve(TM)"), a solution designed to reduce fertility in rats, for shipment on or before Nov. 13. According to the announcement, the company already has received advanced purchase commitments from key customers. "Evolve was developed to offer customers a soft bait product that has similar efficacy to ContraPest(R), but in a format that is easier to deploy and that they can use daily in their integrated pest management programs," said Joel Fruendt, SenesTech's president and CEO. "Evolve is also suited for use by consumers, and we expect to announce partnerships with e-commerce providers and big box retailers by the end of the year."
To view the full press release, visit https://ibn.fm/rNT4Q
SenesTech Inc. (NASDAQ: SNES) is the rodent fertility control expert and the inventor of the only EPA-registered contraceptive for male and female rats. The company’s technology provides an innovative and humane method for managing rat populations.
SenesTech is focused on developing effective solutions that are grounded in science and proven through research, all while providing value to people, communities and the environment. The company’s passion is to create a healthier world by better controlling rat pest populations. This aim is critical, as, if left unchecked, a breeding pair of rats and their descendants can produce up to 15,000 pups after just one year.
The company strives for clean cities, efficient businesses and happy households – with a product that was scientifically designed to be effective without killing rats. SenesTech is committed to the sustainable, humane treatment of animals, improving the quality of all human life and enhancing environmental stewardship through the global application of its effective solution in fertility control technology.
SenesTech is headquartered in Phoenix, Arizona.
ContraPest®
SenesTech’s first product, ContraPest®, applies revolutionary technology to a global challenge that has persisted since the Middle Ages – the proliferation of rats in urban and agricultural settings. ContraPest® targets the reproductive capabilities of Norway and roof rats. As a highly palatable liquid, the formulation promotes sustained consumption, helping to reduce fertility in both male and female rats, bringing populations down and keeping them down.
The company’s flagship offering can be used as part of integrated pest management (IPM) programs – fitting seamlessly into all IPM programs – to help reduce reproduction and magnify the success of these protocols, or as a standalone solution for customers who want to reduce or eliminate the use of lethal rodent control methods.
In multiple, independent field deployments, ContraPest was shown to reduce rat activity over 90% when added to an existing IPM program.
ContraPest® is registered federally as a General Use Product.
Delivery Systems and New Products
In July 2023, SenesTech began to distribute a new delivery system for ContraPest®, the Isolate Bait System™. This new delivery system brings to market a simple design that enables more efficient deployment, incorporates an enhanced formulation of ContraPest® that is expected to provide improved performance of the fertility control bait in the field and is paired with a new bait station that is more space-efficient and economical.
The other delivery systems available for ContraPest include the Ultimate Bait System™, a tank and tray in a larger format for use with more severe infestations, and the Elevate Bait System™, a unique delivery system that targets above ground infestations, as with roof rats.
SenesTech, as of August 2023, is also in the final stages of releasing a soft bait formulation, which provides the unique attributes of proven fertility control in an industry-familiar format demanded by big box retailers, key e-commerce channels and leading industry pest management professionals.
Market Opportunity
According to SenesTech’s figures, rats cause over $27 billion in damage to public and private infrastructure annually in the United States. Rats also destroy 20% of the global stored food supply every year by consuming or contaminating it.
Rats are known to spread at least 35 diseases, globally posing a dangerous risk to public health and safety. Not only does this age-old problem persist despite extensive campaigns to eradicate it, but multiple sources have reported that post-COVID rat populations have boomed.
Poison-based control methods sicken rats, and they typically die slowly. An animal that eats a poisoned rat may also sicken or die. The global rodenticide market is projected to be worth $1.7 billion by 2026.
In one case study, results reported by the customer showed a $5,000 investment in ContraPest® saved more than $500,000 annually in reduced labor, loss and damage.
Management Team
Joel Fruendt is SenesTech’s President and CEO. He has 15 years of executive leadership in the vector and pest control industries as Vice President and General Manager of Clarke Environmental Inc., a leading vector and pest control products and services company. He has extensive expertise in the development and manufacturing of EPA-registered chemical control products, and the commercialization and sale of those products. He received the ‘Smart Leaders’ award from Smart Business Magazine and holds a bachelor’s degree in business from Illinois Wesleyan University.
Tom Chesterman is CFO at SenesTech. He has over 20 years of experience as the CFO of public companies in the life science, tech and telecommunications industries. Most recently, he was the Vice President and Treasurer of GCI, a telecommunications company. Previous to that, he was the CFO of life science companies Bio-Rad Laboratories, Aradigm and Bionovo. He has a bachelor’s degree from Harvard University and an MBA from the University of California at Davis.
Dan Palasky is Chief Technical Officer at SenesTech. Previously he held the title of Vice President of Research & Development at PLZ Corp., a manufacturer of chemical consumer products, serving as the technical expert for its entire product portfolio. He started his career with Camie-Campbell, Inc., as a chemist in the R&D department. Mr. Palasky received his bachelor’s degree in chemical engineering from the Missouri University of Science & Technology and his MBA in Project Management from Aspen University.
SenesTech Inc. (NASDAQ: SNES), closed Wednesday's trading session at $0.2945, off by 1.8333%, on 20,873 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.2801/$7.718.
Recent News
- SenesTech Inc. (NASDAQ: SNES) - InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Now Taking Orders for Evolve(TM) Soft Bait
- SenesTech Inc. (NASDAQ: SNES) Geometrically Increases its Addressable Market with the Addition of Evolve(TM) Soft Bait to its Portfolio
- InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Brings Sustainable Solution for Rising Demand in 'Rattiest Cities'
Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FRA: GTD0)
The QualityStocks Daily Newsletter would like to spotlight Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FRA: GTD0).
Sekur Private Data (CSE: SKUR) (OTCQB: SWISF) (FSE: GDT0), a leading Swiss hosted secure and private communications platform, today announced that it has signed a major distribution agreement with a distributor in the Hashemite Kingdom of Jordan and the Republic of Iraq in order to distribute its entire Sekur Swiss-hosted private and encrypted communications platform. According to the announcement, Sekur's distribution agreement was signed with Solutions Communications & Security Systems ("SOLCOM"), a Jordanian corporation from Amman, Jordan, and is valid for a period of 12 months, with renewal options for additional 12 months.
"We are thrilled to have signed this distribution agreement for Jordan and Iraq with SOLCOM, and we expect more agreements coming, covering the rest of the Middle East and the Gulf Cooperation Council ("GCC") countries," said Alain Ghiai, CEO of Sekur Private Data. "The region is unfortunately a hotbed of instability and cyber warfare is now part of that instability and is showing signs of increased activity, since the Israel-Hamas conflict broke out. Fortunately, we can offer the only Swiss-hosted, proprietary, private and secure communications platform that does not rely on big tech infrastructure, therefore reducing exponentially the risk of cyber penetration for businesses and governments in the region. From our discussions with our partners in Jordan, we believe that our SekurMessenger and SekurMail and SekurVPN for enterprises will be a success with governments, telecom operators and enterprises, such as banks and energy enterprises. SekurVoice, our encrypted voice solution, and our video conferencing solution SekurPRO will come in very handy to enterprises and governments looking for an alternative to existing big tech hosted solutions."
To view the full press release, visit https://ibn.fm/DRVYI
Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FRA: GTD0) is a Cybersecurity and Internet privacy provider of Swiss hosted solutions for secure and private communications. The company distributes a suite of encrypted e-mails, secure messengers and secure communication tools. Sekur Private Data Ltd. sells its products through its own website at www.Sekur.com, approved distributors, and telecommunications companies. Sekur Private Data Ltd. serves consumers, businesses and governments worldwide.
Customer information is completely confidential and safely stored in Switzerland using military grade security. All data, whether physical, network-based or encryption security, is stored in bank-approved, state-of-the-art ISO-certified data centers used by Swiss and global banks and most United Nations organizations, as well as many corporations and governmental organizations. All user data is protected by the Swiss Federal Data Protection Act and the Swiss Federal Data Protection Ordinance, which offer some of the strongest privacy protection in the world for both individuals and organizations.
The company owns 100% of its own infrastructure and, unlike its competitors, does not rely on third party cloud services like Amazon Web Services, Microsoft Azure Cloud or Google cloud infrastructure.
Sekur Private Data has chosen Switzerland to locate its data storage because of the country’s neutrality, independence, strong privacy laws, long standing political stability and excellent international relations. Switzerland is also home to several large multinational corporations and is ranked as having one of the strongest and most competitive economies in the world.
The company is headquartered in Toronto, Ontario.
Products
Sekur Private Data distributes a privacy communications suite offering encrypted and private email, the only Swiss-hosted privacy VPN, and a secure and private messaging application. All solutions cater to consumers, SMBs, enterprises and governments.
- SekurMail® is an encrypted email service offering a private, safe and powerful tool to communicate with everyone, either within the Sekur ecosystem or outside. SekurMail protects personal information and communications from being accessed by unauthorized parties. Its encryption and other security measures prevent messages from being intercepted, modified or tampered with, either in transit or while stored. SekurMail empowers the client to access information and communicate with anyone in the world, regardless of geographical or political barriers.
- SekurVPN® creates a secure, encrypted connection between the client’s device and the Internet, giving clients access to the web safely and privately by routing their connections through a server and hiding their online actions. All the data sent and received is hidden from prying eyes. This includes the clients’ Internet Service Providers, as well as potential hackers and even government surveillance agencies. It can also help clients bypass geographical restrictions and censorship.
- SekurMessenger® is a Swiss-hosted private and secure messaging communications app providing secure and private chat, self-deleting chat, voice recording and file transfer via any mobile device, tablet or desktop computer. Communications are transmitted only within secure servers. It’s designed for organizations that need to protect their flow of information and secure their communications with customers and partners. SekurMessenger is designed to provide military-grade encryption and privacy by ensuring that only the sender and intended recipient can read the messages exchanged. It works for both licensed users of the app and intended message recipients who do not have the app.
Market Opportunity
An analysis from ReportLinker forecasts that the global cybersecurity market will grow from an estimated $173.5 billion in 2022 to $266.2 billion by 2027, recording a CAGR of 8.9% for the period.
The increased number of data breaches worldwide, the ability of malicious actors to operate from anywhere in the world, the links between cyberspace and physical systems and the difficulty of reducing vulnerabilities and consequences in complex cyber networks are some factors that are driving cybersecurity market growth, according to the report.
The global data privacy software market was estimated to be worth $1.68 billion in 2021 and is expected to grow from $2.36 billion in 2022 to $25.85 billion by 2029, achieving an eye-popping 40.8% CAGR during the forecast period, according to a Fortune Business Insights report titled ‘Data Privacy Software Market 2022-2029’.
The widespread shift toward remote working culture, evolving government data privacy regulations and the rapidly increasing adoption of Internet-of-Things devices are among the major factors propelling market growth, per the report.
Management Team
Alain Ghiai is founder, CEO and Director at Sekur Private Data. He also founded GlobeX Data S.A. (GDSA) in 2007 and has served as Director and CEO since then. He founded GlobeX Data Inc. (GlobeX US) in August 2012 and has served as Director and CEO since that time. He attended the California College of Arts in San Francisco, where he earned a Bachelor of Architecture. He has over 15 years of experience in the software industry and was instrumental in taking Sekur Private Data public in July 2019.
Scott Davis, CPA, CGA, is CFO at Sekur Private Data. He is also a partner at Cross Davis & Company LLP Chartered Professional Accountants. His experience includes CFO positions at several companies listed on the TSX Venture Exchange. He spent four years at Appleby as an Assistant Financial Controller. Prior to that, he spent two years at Davison & Company Chartered Professional Accountants as Auditor, five years with Pacific Opportunity Capital as Accounting Manager and two years at Jacobson Soda and Hosak, Chartered Professional Accountants. He obtained his CPA, CGA in 2003.
Learn more about the company’s management team by visiting its corporate page.
Sekur Private Data Ltd. (OTCQB: SWISF), closed Wednesday's trading session at $0.078, off by 0.319489%, on 183,876 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.027/$0.1576.
Recent News
- Sekur Private Data Ltd. (FRA: GTD0) (CSE: SKUR) (OTCQB: SWISF) - InvestorNewsBreaks - Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FSE: GDT0) Announces Entry into Multinational Distribution Agreement in the Middle East
- Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FRA: GTD0) Eyes Growth on New Pricing for SekurVPN Packages, Launch of Communication Suite in German Language
- New to The Street Announces Five Corporate Interviews on its 525th Show, Airing on the Fox Business Network, Monday, October 30, 2023, at 10:30 PM PT
Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF)
The QualityStocks Daily Newsletter would like to spotlight Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) .
Prospera Energy (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B), a public oil and gas exploration, exploitation and development company focusing on conventional oil and gas reservoirs in western Canada, is reporting on its third-quarter production and financial results. According to the announcement, the company is steadily recovering production rates that had been impacted by adverse weather in earlier quarters, with PEI reaching gross peak rates of 770+ boe/d, with another 350 boe/d behind the pipe to be optimized. In addition, the report noted that phase 2 development drilling had begun during the quarter, which necessitated the temporary shut-in of the adjacent production of approximately 120 boe/d for smoother drilling operations; the report stated that the second phase of restructuring development includes the transformation from vertical wells to horizontal wells to access the significant remaining reserves in Prospera's Saskatchewan heavy oil fields.
Financial numbers of note included positive operating income of $1,101,827 in Q3 2023 compared to $783,084 in the same quarter of 2022, and net income of $71,011 in Q3 2023 compared to a ($779,438) net loss in Q2 2023. The company saw average sales prices of $82.15/boe during the quarter, compared to $85.09/boe in Q3 2022, with operating expenses coming in at $41.45/boe for Q3 2023 compared to $36.24/boe in Q3 2022. The company is involved in restructuring efforts that continue to improve its balance sheet. Additionally, Prospera Energy announced the first closing of private placement financing of debt with an equity bonus, resulting in C$580,000, which the company anticipates using for development capital (covering default partner portion) – drilling, completion and tie-in, well abandonment and reclamation costs, and ongoing ESG initiatives.
To view the full press release, visit https://ibn.fm/rAsEP
Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) is a public oil and gas exploration, exploitation and development company focusing on conventional oil and gas reservoirs in Western Canada. The company uses its experience to develop, acquire and drill assets with potential for primary and secondary recovery.
Prospera is primarily focused on optimizing hydrocarbon recovery from legacy fields through environmentally safe and efficient reservoir development methods and production practices. It is in the midst of a three-stage restructuring process aimed at prioritizing cost effective operations while appreciating production capacity and reducing liabilities.
The company is based in Calgary, Alberta, Canada.
Operations
Prospera’s core properties include more than 42,000 cumulative acres across Cuthbert, Luseland and Heart Hills in Saskatchewan and Red Earth and Pouce Coupe in Alberta. In total, the company estimates that there are half a billion barrels of oil in place at these sites accounting for 20+ years of forward project lifespan, with as little as 8% of total reserves having been recovered via legacy vertical well technology.
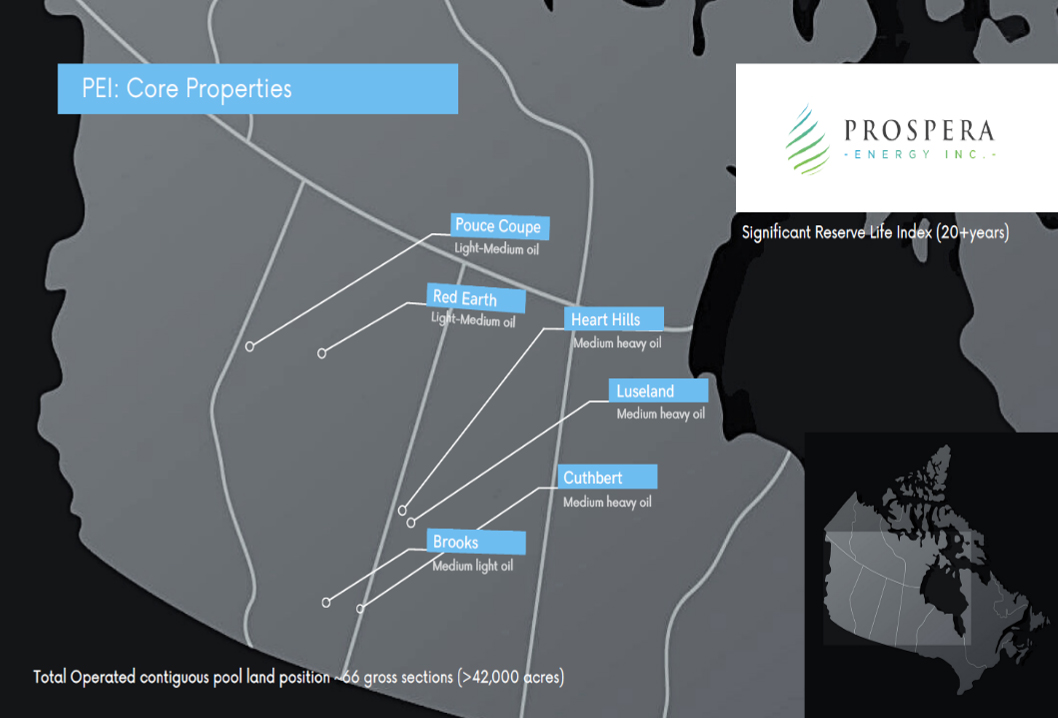
Restructuring Initiative
In 2021, Prospera enacted a top-down reorganization. The early results of these efforts were on display in May 2023, when the company reported a three-fold year-over-year increase in annual revenue for 2022 alongside drastically reduced operating costs and record-high cash flow from operations.
Prospera noted in the news release that it has positioned itself in 2023 to execute the second phase of its development plan aimed at increasing production through medium-oil development in Alberta and leveraging horizontal wells to capture the significant remaining reserves in Saskatchewan.
During the company’s investor summit in August 2023, Prospera CEO Samuel David provided more information regarding this three-phase strategy:
Phase I
Prospera completed the first phase of its restructuring by optimizing operations at its existing assets and addressing legacy arrears and non-compliance issues.
At the beginning of this transformation, the company was producing just 80 barrels of oil equivalent (BOE) per day. In Q4 of last year, Prospera peaked at nearly 1,200 BOE per day. Its breakeven is around 500 barrels per day, illustrating the opportunity for free cash flow. This prospect has driven Prospera’s capital development and optimization in recent quarters.
After a temporary slowdown in production due to harsh winter conditions, Prospera is currently producing about 800 BOE per day and anticipates an additional 300-500 barrels of daily production following the completion of ongoing site maintenance work.
This sustained increase has pushed the company’s NPV from roughly $3 million prior to the restructuring efforts to approximately $72 million today.
In an effort to build on this progress and maximize its available resources, Prospera piloted two horizontal reentries to assess a potential horizontal well transformation at its properties.
Phase II
Following up on the optimization efforts of Phase I, Prospera aims to commence a horizontal well transformation at its properties in the coming months. Based on its pilot wells from Phase I, the company has proposed 10 horizontal well locations at its Cuthbert and Heart Hills properties.
Prospera has likewise proposed eight medium light oil direction wells at its Alberta property, and it is exploring strategic acquisitions aimed at expanding its core area and diversifying its product mix.
Other facets of Phase II include piloting an enhanced oil recovery (EOR) application and continuing to execute its liability management goals and ESG initiatives. Prospera has already abandoned 60 vertical wells as part of its three-year LMR plan to reclaim surface land and reduce the environmental footprint of its operations.
Phase III
Beginning next year, Phase III of Prospera’s corporate redevelopment strategy will focus on continuing the company’s horizontal modular development to appreciate production and optimize recovery of remaining reserves. Prospera intends to implement full-scale EOR applications based on the results of its Phase II pilot program, which is forecast to optimize recovery by greater than 10%.
Prospera also intends to continue its acquisition strategy to diversify its product mix. Its goal, as detailed by in August 2023 investor summit, is to attain 50% light oil, 40% heavy oil and 10% gas – all while continuing to eliminate carbon emissions as part of its existing ESG initiatives.
Poised for Growth
Following its transformational efforts in 2022, Prospera is poised to achieve record growth in 2023. The company has forecast significant reductions in production costs through 2024, alongside sizable increases in daily production.
Prospera is currently exploring strategic acquisition targets to potentially increase its production beyond 5,000 BPD while expanding its reserve base to a billion barrels.
Market Opportunity
While the oil and gas industry faces long-term geopolitical and macroeconomic uncertainty, there is a clear trend to secure supply in the short term. According to Deloitte, the global upstream industry ended 2022 with some of the highest free cash flows on record, driving reinvestment in hydrocarbons and overall investment in clean energy.
The Energy Information Administration recently forecast a dip in global oil inventories over each of the next five quarters, placing upward pressure on oil prices. The agency further forecasts a YoY increase in fuel consumption, exacerbating the effects of OPEC+ production cuts that are set to remain in place through 2024.
For Prospera, these forecasts are promising. The company aims to build on its recent financial growth in the coming months (Prospera reported a three-fold YoY increase in revenue to $13.9 million in 2022), hitting a projected $57 million in total revenue by the end of 2024 while working to expand its core area holdings through accretive M&A transactions.
Leadership Team
Prospera is led by a team with extensive, diverse petroleum industry experience spanning both reservoir management and operations of oil and gas assets. The team boasts a proven track record of reorganizing companies, structuring financing arrangements and positioning for growth.
Samuel David is the company’s President and CEO. He brings to Prospera over 32 years of experience in operation, development and management of oil and gas assets and companies. Mr. David holds a B.Sc. in Mechanical Engineering and a B.A. in Economics from the University of Calgary. His background consists of both engineering and executive management experience with majors Petro-Canada, AEC Oil & Gas (now EnCana / Cenovus) and Husky Energy, as well as founding and operating juniors Ventura Energy and First West Petroleum. Mr. David has proven expertise in corporate planning, production, reservoir engineering, depletion strategies, EOR, property evaluations, acquisitions and divestitures.
George Magarian is VP Subsurface for Prospera. He is a professional petroleum geologist (APEGA) with over 36 years’ experience in the Western Canada Sedimentary Basin. After graduating with an Honors B.Sc. degree in Earth Science from the University of Waterloo, Mr. Magarian spearheaded many successful exploration programs, conducted evaluations for improved recovery schemes and assessed/exploited unconventional oil reservoir opportunities. He has held roles of increasing responsibility, from exploration geologist at oil industry major Petro-Canada and intermediates Anderson Exploration and Jordan Petroleum, to geoscience manager and VP exploration at junior companies Ionic Energy, Gentry Resources and Westfire Energy.
Chris Ludtke is the company’s VP Finance & Accounting. He is a high functioning finance leader with extensive expertise in finance, budgets and planning, accounting, economic evaluation, management, governance and sound decision making. Mr. Ludtke has 20 years of experience within the oil and gas, clean energy and renewables industries, including 12+ years working for Husky Energy before moving into an executive role in the junior oil and gas and hydrogen space. He graduated from the University of Lethbridge (Bachelor of Management) and is a Chartered Professional Accountant in the Province of Alberta.
Matthew Kenna is the CFO of Prospera. He has over 30 years’ experience leading organizations and helping them expand, drive efficiencies and grow profitability. Mr. Kenna is a professional accountant (CPA, CMA) and spent 15 years heading up the financial and operating departments at KUDU Industries, where he fostered financing arrangements, client relationships and manufacturing teams to take the organization from $35M to $150M in revenue. He has extensive experience turning companies around, growing them and building efficient organizations.
Prospera Energy Inc. (OTC: GXRFF), closed Wednesday's trading session at $0.07495, off by 2.2816%, on 23,700 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.059/$0.134.
Recent News
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Reports on Q3 2023 Performance, Debt Financing
- Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Provides Update on Drill Program; Announces Low-Dilutive Capital Raise
- Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Increases Output as Canadian Oil Production Hits Record High
Mountain Top Properties Inc. (OTC: MTPP)
The QualityStocks Daily Newsletter would like to spotlight Mountain Top Properties Inc. (OTC: MTPP).
Mountain Top Properties Inc. (OTC: MTPP) is a diversified real estate holding company that acquires, sells and operates assets through its wholly owned subsidiaries and limited partnerships. The company specializes in property management, property technology (“PropTech”) and real estate redevelopment.
Mountain Top Properties was incorporated in 1990 and is based in Liverpool, New York, with offices in Sag Harbor, New York.
Organization
The company’s flagship subsidiary is Mountain Top Realty Inc., the managing partner of its first real estate fund focused on residential redevelopment in the prestigious and storied Hamptons, New York, beachfront communities.
Mountain Top Properties is also the lead investor in blockchain-enabled industrial and warehouse flex space HQXpress, which services the warehousing, reverse logistics and liquidation markets.
The company is in negotiations for the addition of AI-powered technologies that promise to simplify real estate services, including purchasing and sales.
Mountain Top Capital Fund I LLC
Mountain Top Capital Fund I LLC is a New York limited liability company recently organized by affiliates of Mountain Top Realty, manager of the fund. Through this fund, Mountain Top Realty will leverage the company’s experience, market conditions and industry relationships to capitalize on real estate projects as they arise.
This partnership will be focused on waterfront or water view properties in the Hamptons. The Hamptons market has historically remained strong and continues to set new highs year over year.
The fund has partnered with On Site Builder Construction Co. Inc., which is managed by Joseph Kelley, who, for over 40 years, has continued to build the highest quality architecturally designed custom houses, varying in style from classic to ultra-modern, in the Hamptons. Several houses he has built are showcased in books and magazines and featured across various forms of digital and social media. His assembled team of skilled subcontractors are among the finest skilled craftsmen in their various fields of expertise.
Mr. Kelley has built over 60 custom homes in this market and has the unique distinction of building the most expensive house sold in Sag Harbor in 2014, which sold for $31,750,000; the most expensive house in the Hamptons in 2019, which first sold for $27,500,000 in 2017 and later was renovated and re-traded for $39,250,000; and the most expensive house sold in the Hamptons in 2022, which sold with the neighboring house for $118,500,000. Mr. Kelley’s portfolio of projects is valued at over $400,000,000. Although he has historically worked as a custom home builder, he would like to shift from providing a service to now providing a finished product in this market.
Market Opportunity
Mountain Top Capital Fund I has a target to raise $75 million to acquire, renovate and remarket Hamptons waterfront or water view properties. The fund has secured debt capital commitments for 70% of the acquisition costs and 100% of the construction costs and will use $10 million to leverage strategic waterfront opportunities in and around the Hamptons.
Profits from each project will be distributed upon the sale of the project, which is anticipated every 15 to 18 months, with a target of a minimum return on investment of 20% to 30% per transaction.
The company anticipates the fund’s Hamptons projects will be followed up by several other funds targeting additional high-end markets.
Management Team
Beau Kelley is CEO, President, CFO and Director of Mountain Top Properties.
Mountain Top Properties Inc. (OTC: MTPP), closed Wednesday's trading session at $0.064, up 64.1026%, on 240,198 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.0064/$0.0695.
Recent News
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
- Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
- Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
PaxMedica Inc. (NASDAQ: PXMD)
The QualityStocks Daily Newsletter would like to spotlight PaxMedica Inc. (NASDAQ: PXMD).
PaxMedica Inc. (NASDAQ: PXMD) is a clinical stage biopharmaceutical company focusing on the development of novel anti-purinergic therapies (APTs) for the treatment of Autism Spectrum Disorder (ASD) and other serious conditions with intractable neurologic symptoms.
The company’s lead programs are focused on ASD, for which there are currently no approved pharmacologic treatments that target its cause and symptoms. Currently used treatments only address the symptoms of the condition, rather than targeting the pathophysiology itself.
PaxMedica is on a promising path to address these unmet medical needs, bringing hope to millions. Anti-purinergic therapies target the excess production of purines in cells. An overexpression of purines can offset homeostasis and result in an overproduction of cellular adenosine triphosphate, the main energy molecule in all living cells.
The company is headquartered in Tarrytown, New York.
Product Pipeline
PaxMedica is building a robust pipeline of products targeting ASD and related neurodevelopmental conditions. The company’s lead product in development may help eliminate, reduce or modulate some of the more troublesome aspects of ASD. That would open the potential for people with autism to integrate their behavior with others more successfully and improve their lives.
PaxMedica’s lead programs, PAX-101 and PAX-102, utilize the company’s proprietary source of suramin sodium, a broadly acting anti-purinergic therapy that has been known for over 100 years. Its current pipeline includes:
- PAX-101 (IV Suramin) for ASD – PAX-101 completed a Phase 2B study for ASD in 2021. Suramin is a broadly acting APT and has reported positive results from a dose range study. The results of PaxMedica’s Phase 2B study, which targeted 52 subjects across six sites in South Africa, were presented to AACAP in October 2021.
- PAX-102 (Intranasal Suramin) – PaxMedica has developed a proprietary intranasal formulation of suramin that is currently being evaluated in ASD and other neurodevelopmental conditions.
- PAX-101 for HAT – Given suramin’s historical use as a treatment for Human African Trypanosomiasis (HAT), or African Sleeping Sickness, the company is also developing PAX-101 as a treatment for HAT. PaxMedica’s most advanced program is the pursuit of PAX-101 for early-stage East African HAT.
- Selective APTs – PaxMedica has conducted several preclinical studies to evaluate other APTs that are more selective to specific purinergic receptors and may offer additional benefits over suramin.
Market Opportunity
According to a report by Fortune Business Insights, a leading global market research company, the global ASD therapeutics market was estimated at $1.93 billion in 2022 and is projected to grow from $2.01 billion in 2023 to $3.42 billion by 2030, a CAGR of 7.9% over the forecast period. As there is no current treatment for the core symptoms of autism, PaxMedica believes the addressable market for PAX-101, if approved, could greatly exceed these forecasts.
Autistic disorder, Asperger’s Syndrome and Pervasive Development Disorder are the three main types of ASD, affecting millions of people globally. A 2020 report by the U.S. Centers for Disease Control & Prevention estimated that one in 36 children in the U.S. have been diagnosed with autism disorder.
Several factors are expected to contribute to market growth prospects. A growing prevalence of the condition globally and rising awareness coupled with available treatment options are key factors expected to drive ASD therapeutics market growth during the forecast period. Growing investment in R&D to find effective treatments is also expected to fuel global market growth.
Management Team
Howard Weisman is Chairman and CEO of PaxMedica. He has been a founder and CEO of several specialty pharma and medical device companies. Most recently, he was executive chairman and co-founder of Sofregen, a biotech company. He also served as CEO and president of Seventh Sense Biosystems, a medical device development company. He also was founder, chairman and CEO of EKR Therapeutics, a specialty pharmaceutical company, and founder and COO of ESP Pharma, a company focused on cardio and neurovascular products. He has a bachelor’s degree in chemistry from Rutgers University.
David Hough, M.D., is Chief Medical Officer at PaxMedica. He is a neuroscience clinical development consultant who previously served as vice president at Janssen Research and Development and in various leadership roles over 17 years. Most recently, he was the compound development team leader for SPRAVATO® for treatment-resistant depression. Prior to that, he was the schizophrenia disease area leader. He played a pivotal role in the development programs for oral INVEGA®, INVEGA SUSTENNA® and XEPLION® for schizophrenia. He is a graduate of West Point and is board certified in psychiatry.
Stephen Sheldon is COO and CFO at PaxMedica. He has served as CEO of Thailand-based specialty healthcare company Indochina Healthcare Co. Ltd. since 2015. Previously, he was a consultant for PricewaterhouseCoopers Healthcare Advisory in the Chicago office. He was responsible for developing specialty pharmacy patient programs, strategy development for specialty products and compliance programs. He has an MBA from Thunderbird School of Global Management and a bachelor’s degree in computer science and visual arts from Bowdoin College.
PaxMedica Inc. (NASDAQ: PXMD), closed Wednesday's trading session at $3.12, off by 0.952381%, on 175,262 volume. The average volume for the last 3 months is 130,600 and the stock's 52-week low/high is $2.50/$69.19.
Recent News
- PaxMedica Inc. (NASDAQ: PXMD) - InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Acquires Research Assets to Accelerate PAX-101 NDA Submission
- InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Reports on 1-for-17 Reverse Stock Split
- InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Reports on Publication of Low-Dose Suramin for ASD Research
Lucy Scientific Discovery Inc. (NASDAQ: LSDI)
The QualityStocks Daily Newsletter would like to spotlight Lucy Scientific Discovery Inc. (NASDAQ: LSDI).
Lucy Scientific Discovery Inc. (NASDAQ: LSDI) is an early-stage psychotropics manufacturing company focused on becoming the premier contract research, development and manufacturing organization for the emerging psychotropics-based medicines industry.
The company holds a Controlled Drugs and Substances Dealer’s License granted by Health Canada’s Office of Controlled Substances. This specialized license authorizes LSDI to develop, sell, deliver and manufacture pharmaceutical-grade active pharmaceutical ingredients (APIs) used in controlled substances and their raw material precursors. Lucy Scientific Discovery and its wholly owned subsidiary, LSDI Manufacturing Inc., operate under Part J of the Food and Drug Regulations promulgated under the Food and Drugs Act (Canada).
The company’s mission is to make its products and research services available for the development of medicines and experimental therapies to address certain psychiatric health disorders and other medical needs, including various mental health and addiction disorders. LSDI targets customers that include an increasing number of the leading universities, hospitals and other public, private and government institutions throughout the world that have launched research programs aimed at understanding the therapeutic potential of a range of psychedelic substances.
The company is headquartered in Victoria, British Columbia, Canada.
Products
LSDI produces a variety of high-quality natural, synthetic and biosynthetic products to meet the needs of the rapidly growing psychotropics-based medicines market. The company believes the emerging psychotropics industry will pave the way to a brighter future in mental health and overall wellness. LSDI is dedicated to advancing the frontiers of mind science and facilitating the development of psychotropic and psychedelic treatment therapies.
In Canada, the psychedelic compounds that LSDI is approved to produce under its Dealer’s License are regulated under the Controlled Drugs and Substances Act, or CDSA. Those compounds include:
- Psilocybin
- Psilocin
- Lysergic acid diethylamide, or LSD
- N,N-Dimethyltryptamine, or N,N-DMT
- 3,4-Methylenedioxymethamphetamine, or MDMA
- 4-Bromo-2,5-Dimethoxybenzeneethanamine, or 2C-B
The company also sells its consumer psychotropic products directly online and through retailers. Those products, described as microdose mushroom formulations, include a sleep aid, Twilight, and a mindfulness enhancer, Mindful.
Market Opportunity
According to a report from Global Market Insights, the psychotropics drug market had an estimated value of $20.2 billion in 2022 and is projected to reach a value of nearly $37.6 billion by 2032. That represents a CAGR of 6.4% for the forecast period. Factors driving market growth include the increasing prevalence of mental disorders, technological advancements in drug development, a rising geriatric population and increasing healthcare expenditures, the report states.
A growing awareness of mental health issues and an effort to reduce the stigma surrounding psychiatric disorders have encouraged more individuals to seek help, which in turn boosts the market. In addition, advancements in neuroscience, pharmacology and drug development have led to the discovery of new and more effective central nervous system therapeutics.
Innovative treatments offering better outcomes with fewer side effects attract patients and healthcare providers, also driving market growth.
Management Team
LSDI’s executive team brings deep experience in the development and commercialization of products featuring controlled substances, as well as the navigation of regulatory structures applicable to these products.
Richard Nanula is Chairman and CEO of LSDI. He has more than 35 years of leadership experience at several of the largest companies in the world, having been a senior executive at The Walt Disney Company, Amgen, Colony Capital and Starwood Hotels and Resorts. He has also served as a board member for Boeing Corporation and Starwood Capital, where he provided corporate guidance and oversight. He holds an MBA from Harvard Business School.
Assad J. Kazeminy, Ph.D., is Chief Scientific Officer at LSDI. He previously served as CEO of Irvine Pharmaceutical Services Inc. and Avrio Biopharmaceutical LLC, and he has founded several drug development companies. He has over 30 years of research and development experience in the biopharmaceutical industry. He received his Ph.D. in Pharmaceutical Science and Biochemistry from Esfahan University in Iran. He completed a Post Doctorate course of study at the University of Southern California Medical School, Department of Pharmacology.
Brian Zasitko, CPA, CA, is the company’s CFO. He is a Director of Invictus Accounting Group LLP, a professional services firm providing finance, advisory and accounting services. He also serves as CFO of Lobe Sciences Ltd., a company developing psychedelic compounds as therapeutics for the treatment of mild traumatic brain injuries and post-traumatic stress disorder. He has an undergraduate degree from Simon Fraser University and a CPA (CA) from Certified Professional Accountants, British Columbia.
Lucy Scientific Discovery Inc. (NASDAQ: LSDI), closed Wednesday's trading session at $0.28, off by 3.4483%, on 141,200 volume. The average volume for the last 3 months is 193,566 and the stock's 52-week low/high is $0.2511/$4.00.
Recent News
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Study Suggests Methamphetamine More Helpful for Social Bonding Compared to MDMA
- InvestorNewsBreaks - Lucy Scientific Discovery Inc. (NASDAQ: LSDI) Strengthen Position in Alternative Mental-Health Treatments Sector
- Lucy Scientific Discovery (NASDAQ: LSDI) Acquisitions Create 'Golden Opportunity' for Growth, Success
Longeveron Inc. (NASDAQ: LGVN)
The QualityStocks Daily Newsletter would like to spotlight Longeveron Inc. (NASDAQ: LGVN) .
Longeveron Inc. (NASDAQ: LGVN) is a clinical-stage biotechnology company developing regenerative medicines to address unmet medical needs for specific aging-related and life-threatening conditions. The Company’s research and therapies are aimed at improving the outcome of infants born with a life-threatening heart condition, as well as improving the healthspan for the aging population – the number of years a person is expected to live in relatively good health, free of chronic disease and disabilities of aging, with function and ability to perform activities of daily living.
Longeveron is involved in clinical trials in the following indications: Hypoplastic left heart syndrome (HLHS), Alzheimer’s disease, and Aging-related Frailty.
The Company’s philosophy revolves around the idea that regenerative medicine may hold the potential to improve certain rare medical conditions and contribute to healthy aging. While there has been a remarkable rise in life expectancy over the last century due to medical and public health advancements, this increase in longevity has not been paralleled by the number of years a person is expected to live in relatively good health, free of chronic disease and disabilities of aging.
Longeveron’s lead investigational product is Lomecel-B™, an allogeneic Medicinal Signaling Cell therapy product isolated from the bone marrow of young, healthy adult donors. As humans age, they experience a decrease in immune system function, a decline in blood vessel functioning, chronic inflammation, and other issues. Clinical data has suggested that Lomecel-B™ may address these conditions through multiple mechanisms of action (MOA) that simultaneously target key aging-related processes.
The Company is headquartered in Miami, Florida.
Lomecel-B™
Lomecel-B™ is being evaluated in multiple clinical trials for aging-related chronic diseases and other life-threatening conditions under U.S. FDA-approved Investigational New Drug applications. Lomecel-B™ has multiple potential mechanisms of action encompassing pro-vascular, pro-regenerative, anti-inflammatory, and tissue repair and healing effects with broad potential applications across a spectrum of disease areas.
The drug is made from special living cells called Medicinal Signaling Cells (MSCs) that are isolated from fresh bone marrow tissue that has been donated by adult donors aged 18 to 45. Once the MSCs have been isolated from the fresh bone marrow through a careful selection process, the cells are culture-expanded (allowed to replicate under controlled laboratory conditions) into the billions using specialized techniques and processes. After a specific number of expansion cycles, called “passages,” the cells are harvested, separated into specific doses (e.g., 50 million cells), and cryopreserved until future use.
These cells have been shown to have characteristics that allow them to be transplanted from a donor to host without triggering a harmful immune response in the recipient, and they can be administered on an outpatient basis in as little as 40 minutes after thawing. Because of these characteristics, Lomecel-B™ is considered an “off-the-shelf” product.
In some trials, such as for Alzheimer’s disease and Aging-related Frailty, Lomecel-B™ is administered via peripheral intravenous infusion, while, in the Company’s HLHS trial, Lomecel-B™ is administered via direct injection into the heart tissue.
Market Opportunity
Longeveron estimates the potential market size for Lomecel-B™ in the treatment of HLHS to be up to $1 billion annually, globally.
U.S. patients suffering from Aging-related Frailty are estimated using U.S. Census Bureau statistics to be approximately 8.1 million. That population potentially represents a market for Lomecel-B™ of between $4 billion and $8 billion globally per year, according to Company estimates.
Additionally, the Alzheimer’s Association puts the number of Americans with that disease at 5.1 million, highlighting another potentially addressable market for Lomecel-B™, that’s worth $5 billion to $10 billion annually.
Management Team
Wa’el Hashad is CEO of Longeveron. He has more than 35 years of experience in the pharmaceutical and biotech industries. He has launched several successful brands in the U.S. and worldwide markets. Prior to joining Longeveron, he was president and CEO of Avanir Pharmaceuticals. Before Avanir, he was the chief commercial officer of Seres Therapeutics. He also has held senior leadership positions at Amgen, Boehringer Ingelheim, and Eli Lilly and Company. He holds a bachelor’s degree in pharmacy from Cairo University and an MBA from the University of Akron.
Joshua M. Hare, M.D., FACC, FAHA, is Co-Founder, Chief Science Officer and Chairman of Longeveron. He is a double board-certified cardiologist and is the founding director of the Interdisciplinary Stem Cell Institute at the University of Miami’s Miller School of Medicine. He is a recipient of the Paul Beeson Physician Faculty Scholar in Aging Research Award and is an elected member of the American Association of Physicians and The American Society for Clinical Investigation. He is also an elected Fellow of the American Heart Association. He received a bachelor’s degree from the University of Pennsylvania and his M.D. from The Johns Hopkins University School of Medicine.
Lisa Locklear is CFO at Longeveron. She previously served as the senior vice president and CFO for Avanir Pharmaceuticals. Prior to Avanir, she held senior financial roles at GSN Games, CoreLogic, Ingram Micro, the Walt Disney Company, and Price Waterhouse, with assignments in Paris and London. She holds a bachelor’s degree in plant science from the University of California, Davis, and an MBA from the University of California, Irvine. She is a licensed CPA (inactive) and is a member of the American Institute of Certified Public Accountants, the California Society of CPAs, and Financial Executives International.
Dr. Nataliya Agafonova, M.D., is the Chief Medical Officer at Longeveron. She previously served as clinical development lead, senior medical director, and product development chair at Otsuka Pharmaceuticals. Before that, she was the clinical development lead and senior medical director at Bristol-Myers Squibb. She previously held senior leadership positions at Ardea Bioscience, Biogen, Amgen, and Genzyme Corporation. She earned an M.D. from the Ukrainian National Medical University and completed her internal medicine residency at Kharkov State University Hospital in Ukraine.
Certain statements in this corporate profile that are not historical facts are forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, which reflect management’s current expectations, assumptions, and estimates of future operations, performance and economic conditions, and involve risks and uncertainties that could cause actual results to differ materially from those anticipated by the statements made herein. Forward-looking statements are generally identifiable by the use of forward-looking terminology such as “believe,” “expects,” “may,” “looks to,” “will,” “should,” “plan,” “intend,” “on condition,” “target,” “see,” “potential,” “estimates,” “preliminary,” or “anticipates” or the negative thereof or comparable terminology, or by discussion of strategy or goals or other future events, circumstances, or effects. Factors that could cause actual results to differ materially from those expressed or implied in any forward-looking statements in this release include, but are not limited to, statements regarding the offer and sale of securities, the terms of the offering, about the ability of Longeveron’s clinical trials to demonstrate safety and efficacy of the Company’s product candidates, and other positive results; the timing and focus of the Company’s ongoing and future preclinical studies and clinical trials and the reporting of data from those studies and trials; the size of the market opportunity for the Company’s product candidates, including its estimates of the number of patients who suffer from the diseases being targeted; the success of competing therapies that are or may become available; the beneficial characteristics, safety, efficacy and therapeutic effects of the Company’s product candidates; the Company’s ability to obtain and maintain regulatory approval of its product candidates in the U.S., Japan and other jurisdictions; the Company’s plans relating to the further development of its product candidates, including additional disease states or indications it may pursue; the Company’s plans and ability to obtain or protect intellectual property rights, including extensions of existing patent terms where available and its ability to avoid infringing the intellectual property rights of others; the need to hire additional personnel and the Company’s ability to attract and retain such personnel; the Company’s estimates regarding expenses, future revenue, capital requirements and needs for additional financing; the Company’s need to raise additional capital, and the difficulties it may face in obtaining access to capital, and the dilutive impact it may have on its investors; the Company’s financial performance and ability to continue as a going concern, and the period over which it estimates its existing cash and cash equivalents will be sufficient to fund its future operating expenses and capital expenditure requirements. Additionally, Longeveron makes no assurance that any public offering of its securities as described herein will occur on the timelines, in the manner or on the terms anticipated due to numerous factors. Further information relating to factors that may impact the Company’s results and forward-looking statements are disclosed in the Company’s filings with the Securities and Exchange Commission, including Longeveron’s Annual Report on Form 10-K for the year ended December 31, 2022, filed with the Securities and Exchange Commission on March 14, 2023 and its Quarterly Report on Form 10-Q for the second quarter of 2023 filed with the SEC on August 11, 2023. The forward-looking statements contained in this corporate profile are made as of the date of this corporate profile, and the Company disclaims any intention or obligation, other than imposed by law, to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise.
Investor Contact
Mike Moyer
LifeSci Advisors
Tel: 617-308-4306
Email: mmoyer@lifesciadvisors.com
Date prepared: August 31, 2023
Longeveron Inc. (NASDAQ: LGVN), closed Wednesday's trading session at $2.06, up 4.5685%, on 90,549 volume. The average volume for the last 3 months is 347,830 and the stock's 52-week low/high is $1.50/$4.5793.
Recent News
- Longeveron Inc. (NASDAQ: LGVN) - Longeveron Inc. (NASDAQ: LGVN) Validates Safety and Therapeutic Potential of its Lomecel-B(TM) Lead Investigational Product in its CLEAR MIND Phase 2a Clinical Trial
- InvestorNewsBreaks - Longeveron Inc. (NASDAQ: LGVN; LGVNR) Top-Line Results from Phase 2a Trial Spotlighted in Latest NNA Release
- InvestorNewsBreaks - Longeveron Inc. (NASDAQ: LGVN; LGVNR) Closes on $4M Registered Direct Offering
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Collaborates with WSU Grad School to Produce eLearning Programs
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Announces Exceptional Assay Drill Results from Its PCH Ionic Clay Project
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Intercepts 83.7 m at 3.6 g/t AuEq (incl. 15.3 m at 6.3 g/t AuEq) and 21.9 m at 1.3% CuEq (incl. 1.4 m at 5.7% CuEq) at the Kay Deposit
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Forging Strategic Partnerships for an Open Network of Digital Health Solutions
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) to Present at Upcoming Emerging Growth Conference
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Releases Q3 2023 Financial Report, Reports Possible Adjustment of Facility Terms by EIB
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Announces Closing of Third and Final Tranche of Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Featured in Bell2Bell Podcast
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - Clene Inc. (NASDAQ: CLNN) Reports Improved Survival Status in ALS Patients Treated with CNM-Au8(R) on Follow-Up Analysis; Announces Peer-Reviewed Study
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Stanford Study Finds Lung Cancer Cells Hide, Thrive in the Brain
- Correlate Energy Corp. (OTCQB: CIPI) - Harbinger Research Publishes New Research Report on Correlate Energy Corp., a Rapidly Growing Provider of Sustainable Energy Solutions Based on Solar Generation and Battery Storage Systems
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc.'s (NEO: CYBN) (NYSE American: CYBN) Releases Update, Notes Upcoming Pipeline Milestones
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Launches Innovative Smart Blood Glucose Meter
- Diamond Lake Minerals Inc. (OTC: DLMI) - Diamond Lake Minerals Inc. (DLMI) Welcomes Brian J. Esposito As Company CEO In Addition To Seven New Strategic Advisors
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS), QuantumBasel Announce Two-Year Agreement Extension to Fuel Quantum Commercial Adoption
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Announces Participation at Emerging Growth Conference
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Reports on Progress at Bolivian Iska Iska Project
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Announces 10-Year Organic-Waste Processing Agreement, $7M Senior Term Loan
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Issues Operations Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Filing of Federal Lawsuit Against Research Firm
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Closes First Tranche of Private Placement
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Expands with New Project in James Bay Lithium Region
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Releases Business Update, Roadmap
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Taking Part in the Trucking Industry's Ongoing Paradigm Shift with Its Fr8App Platform
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Reports on Key Milestones for 2023, Provides Update on Targets Moving Forward
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Receives $1.9M Funding Grant from Canadian Government ACT Program
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) to Present at BIO-Europe 2023
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Reports Record Preliminary Q3 2023 Revenues, Announces Earnings Conference Call
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) to Host Third Quarter 2023 Results Conference Call
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Sees Ongoing Traction in Customer Demonstrations; Nears Proof-of-Concept for New Technology Offering
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream’s Investing App Signifies Potential of Blockchain Technology, Next Phase of Capital Markets
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Cannabinoid-Based Topical Pain-Relief Treatment Receives European Patent
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Secures $5.2M in Private Placement and Preferred Investment Option Offerings
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Granted Second U.S. Patent for JW-700 Treatment Designed for $1.5B Hair-Growth Market
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Secures Expanded and New Contracts for K1 Technologies
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Longeveron Inc. (NASDAQ: LGVN) Validates Safety and Therapeutic Potential of its Lomecel-B(TM) Lead Investigational Product in its CLEAR MIND Phase 2a Clinical Trial
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Get Ready for Next Generation of Diabetes, Weight-Management Drugs
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Study Suggests Methamphetamine More Helpful for Social Bonding Compared to MDMA
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Releases Update on Q3 Production
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (CSE: MYCO) (AQSE: MYIG) (OTC: MYCOF) Files Prospectus Supplement, Closes Share Subscription Agreements
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Appointment of Strategic AI Advisor
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Brand Metrics e-Commerce Companies Need to Watch
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Request to Continue Listing Is Granted by NASDAQ Hearings Panel
- PaxMedica Inc. (NASDAQ: PXMD) - InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Acquires Research Assets to Accelerate PAX-101 NDA Submission
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Announces Addition of Meek's Vegan Pizza to Chicago Foodhall Offerings
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Q3 Player Activity Milestones
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Bi-Weekly Status Report
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - New to The Street Announces Four Corporate Interviews on its 525th Show, Airing on the Fox Business Network, Monday, October 30, 2023, at 10:30 PM PT
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Creates Advisory Board, Names Renowned Experts as Members
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Reports on Q3 2023 Performance, Debt Financing
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Releases Corporate Update, Including Aleafia Health Agreement
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Alarming Data Highlights Gaps in Treating People with Chronic HBV
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Swedish Scientists Find Promising Way to Recover Minerals from Discarded EV Batteries
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Reports on Strong Drill Results at Oko West Project in Guyana
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Becomes Largest Shareholder of Plantify Foods Inc.
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters into Acquisition Agreement, Asset Purchase Agreement
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Positive Results of Preclinical Study to Advance SPU-21
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Eyes the Growing World Market for Domestic Violence Offender Monitoring Solutions
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Closing of $1.6M Public Offering, $750K Order for Use by First Responders in Israel
- SOHM Inc. (OTC: SHMN) - SOHM, Inc. (SHMN) Completes Acquisition of ABBIE Gene-editing Tech, Begins Preparations to Commercialize Solutions for Heart Disease
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Exclusive Single-Serve Wine Brand to Be Featured by 130-Plus Circle K Locations
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Makes 'Significant Move' with Exercise of SWA Project Option Agreement
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands to Present at LD Micro Main Event XVI
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Subsidiary Meets with Jordan Minister of Water in Round Table Gathering
- SenesTech Inc. (NASDAQ: SNES) - InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Now Taking Orders for Evolve(TM) Soft Bait
- Sekur Private Data Ltd. (FRA: GTD0) (CSE: SKUR) (OTCQB: SWISF) - InvestorNewsBreaks - Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FSE: GDT0) Announces Entry into Multinational Distribution Agreement in the Middle East
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Receives Full Compliance Notification from NASDAQ
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Enters MOU to Launch Business, Service Offerings in Pakistan
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Closes on AUD$175,000 Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TrxADE Health, Inc. affirms its commitment to the pharmaceutical space via its subsidiary Trxade, Inc. which operates the pharmaceutical marketplace and added more members in the third quarter of 2023.
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Executing Louisiana Project with Potential to Significantly Enhance North America's Manufacturing Landscape
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Releases FY 2023 Report
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Announces Participation in Inaugural Biotech CNS/Neuro Summit
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Pricing of $4M Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Giles Tipsword Joins Wrap Technologies as Senior Vice President of North American Sales
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Collaborates with WSU Grad School to Produce eLearning Programs
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Announces Exceptional Assay Drill Results from Its PCH Ionic Clay Project
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Intercepts 83.7 m at 3.6 g/t AuEq (incl. 15.3 m at 6.3 g/t AuEq) and 21.9 m at 1.3% CuEq (incl. 1.4 m at 5.7% CuEq) at the Kay Deposit
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Forging Strategic Partnerships for an Open Network of Digital Health Solutions
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) to Present at Upcoming Emerging Growth Conference
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Releases Q3 2023 Financial Report, Reports Possible Adjustment of Facility Terms by EIB
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Announces Closing of Third and Final Tranche of Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Featured in Bell2Bell Podcast
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - Clene Inc. (NASDAQ: CLNN) Reports Improved Survival Status in ALS Patients Treated with CNM-Au8(R) on Follow-Up Analysis; Announces Peer-Reviewed Study
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Stanford Study Finds Lung Cancer Cells Hide, Thrive in the Brain
- Correlate Energy Corp. (OTCQB: CIPI) - Harbinger Research Publishes New Research Report on Correlate Energy Corp., a Rapidly Growing Provider of Sustainable Energy Solutions Based on Solar Generation and Battery Storage Systems
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc.'s (NEO: CYBN) (NYSE American: CYBN) Releases Update, Notes Upcoming Pipeline Milestones
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Launches Innovative Smart Blood Glucose Meter
- Diamond Lake Minerals Inc. (OTC: DLMI) - Diamond Lake Minerals Inc. (DLMI) Welcomes Brian J. Esposito As Company CEO In Addition To Seven New Strategic Advisors
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS), QuantumBasel Announce Two-Year Agreement Extension to Fuel Quantum Commercial Adoption
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Announces Participation at Emerging Growth Conference
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Reports on Progress at Bolivian Iska Iska Project
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Announces 10-Year Organic-Waste Processing Agreement, $7M Senior Term Loan
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Issues Operations Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Filing of Federal Lawsuit Against Research Firm
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Closes First Tranche of Private Placement
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Expands with New Project in James Bay Lithium Region
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Releases Business Update, Roadmap
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Taking Part in the Trucking Industry's Ongoing Paradigm Shift with Its Fr8App Platform
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Reports on Key Milestones for 2023, Provides Update on Targets Moving Forward
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Receives $1.9M Funding Grant from Canadian Government ACT Program
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) to Present at BIO-Europe 2023
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Reports Record Preliminary Q3 2023 Revenues, Announces Earnings Conference Call
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) to Host Third Quarter 2023 Results Conference Call
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Sees Ongoing Traction in Customer Demonstrations; Nears Proof-of-Concept for New Technology Offering
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream’s Investing App Signifies Potential of Blockchain Technology, Next Phase of Capital Markets
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Cannabinoid-Based Topical Pain-Relief Treatment Receives European Patent
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Secures $5.2M in Private Placement and Preferred Investment Option Offerings
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Granted Second U.S. Patent for JW-700 Treatment Designed for $1.5B Hair-Growth Market
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Secures Expanded and New Contracts for K1 Technologies
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Longeveron Inc. (NASDAQ: LGVN) Validates Safety and Therapeutic Potential of its Lomecel-B(TM) Lead Investigational Product in its CLEAR MIND Phase 2a Clinical Trial
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Get Ready for Next Generation of Diabetes, Weight-Management Drugs
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Study Suggests Methamphetamine More Helpful for Social Bonding Compared to MDMA
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Releases Update on Q3 Production
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (CSE: MYCO) (AQSE: MYIG) (OTC: MYCOF) Files Prospectus Supplement, Closes Share Subscription Agreements
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Appointment of Strategic AI Advisor
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Brand Metrics e-Commerce Companies Need to Watch
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Request to Continue Listing Is Granted by NASDAQ Hearings Panel
- PaxMedica Inc. (NASDAQ: PXMD) - InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Acquires Research Assets to Accelerate PAX-101 NDA Submission
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Announces Addition of Meek's Vegan Pizza to Chicago Foodhall Offerings
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Q3 Player Activity Milestones
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Bi-Weekly Status Report
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - New to The Street Announces Four Corporate Interviews on its 525th Show, Airing on the Fox Business Network, Monday, October 30, 2023, at 10:30 PM PT
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Creates Advisory Board, Names Renowned Experts as Members
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Reports on Q3 2023 Performance, Debt Financing
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Releases Corporate Update, Including Aleafia Health Agreement
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Alarming Data Highlights Gaps in Treating People with Chronic HBV
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Swedish Scientists Find Promising Way to Recover Minerals from Discarded EV Batteries
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Reports on Strong Drill Results at Oko West Project in Guyana
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Becomes Largest Shareholder of Plantify Foods Inc.
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters into Acquisition Agreement, Asset Purchase Agreement
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Positive Results of Preclinical Study to Advance SPU-21
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Eyes the Growing World Market for Domestic Violence Offender Monitoring Solutions
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Closing of $1.6M Public Offering, $750K Order for Use by First Responders in Israel
- SOHM Inc. (OTC: SHMN) - SOHM, Inc. (SHMN) Completes Acquisition of ABBIE Gene-editing Tech, Begins Preparations to Commercialize Solutions for Heart Disease
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Exclusive Single-Serve Wine Brand to Be Featured by 130-Plus Circle K Locations
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Makes 'Significant Move' with Exercise of SWA Project Option Agreement
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands to Present at LD Micro Main Event XVI
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Subsidiary Meets with Jordan Minister of Water in Round Table Gathering
- SenesTech Inc. (NASDAQ: SNES) - InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Now Taking Orders for Evolve(TM) Soft Bait
- Sekur Private Data Ltd. (FRA: GTD0) (CSE: SKUR) (OTCQB: SWISF) - InvestorNewsBreaks - Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FSE: GDT0) Announces Entry into Multinational Distribution Agreement in the Middle East
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Receives Full Compliance Notification from NASDAQ
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Enters MOU to Launch Business, Service Offerings in Pakistan
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Closes on AUD$175,000 Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TrxADE Health, Inc. affirms its commitment to the pharmaceutical space via its subsidiary Trxade, Inc. which operates the pharmaceutical marketplace and added more members in the third quarter of 2023.
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Executing Louisiana Project with Potential to Significantly Enhance North America's Manufacturing Landscape
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Releases FY 2023 Report
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Announces Participation in Inaugural Biotech CNS/Neuro Summit
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Pricing of $4M Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Giles Tipsword Joins Wrap Technologies as Senior Vice President of North American Sales
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Collaborates with WSU Grad School to Produce eLearning Programs
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Announces Exceptional Assay Drill Results from Its PCH Ionic Clay Project
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Intercepts 83.7 m at 3.6 g/t AuEq (incl. 15.3 m at 6.3 g/t AuEq) and 21.9 m at 1.3% CuEq (incl. 1.4 m at 5.7% CuEq) at the Kay Deposit
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Forging Strategic Partnerships for an Open Network of Digital Health Solutions
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) to Present at Upcoming Emerging Growth Conference
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Releases Q3 2023 Financial Report, Reports Possible Adjustment of Facility Terms by EIB
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Announces Closing of Third and Final Tranche of Non-Brokered Private Placement
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Featured in Bell2Bell Podcast
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - Clene Inc. (NASDAQ: CLNN) Reports Improved Survival Status in ALS Patients Treated with CNM-Au8(R) on Follow-Up Analysis; Announces Peer-Reviewed Study
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Stanford Study Finds Lung Cancer Cells Hide, Thrive in the Brain
- Correlate Energy Corp. (OTCQB: CIPI) - Harbinger Research Publishes New Research Report on Correlate Energy Corp., a Rapidly Growing Provider of Sustainable Energy Solutions Based on Solar Generation and Battery Storage Systems
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc.'s (NEO: CYBN) (NYSE American: CYBN) Releases Update, Notes Upcoming Pipeline Milestones
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Launches Innovative Smart Blood Glucose Meter
- Diamond Lake Minerals Inc. (OTC: DLMI) - Diamond Lake Minerals Inc. (DLMI) Welcomes Brian J. Esposito As Company CEO In Addition To Seven New Strategic Advisors
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - InvestorNewsBreaks - D-Wave Quantum Inc. (NYSE: QBTS), QuantumBasel Announce Two-Year Agreement Extension to Fuel Quantum Commercial Adoption
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Announces Participation at Emerging Growth Conference
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - InvestorNewsBreaks - Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) (FSE: P2QM) Reports on Progress at Bolivian Iska Iska Project
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q2 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Announces 10-Year Organic-Waste Processing Agreement, $7M Senior Term Loan
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Issues Operations Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Filing of Federal Lawsuit Against Research Firm
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - First Tellurium Closes First Tranche of Private Placement
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Expands with New Project in James Bay Lithium Region
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Releases Business Update, Roadmap
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Taking Part in the Trucking Industry's Ongoing Paradigm Shift with Its Fr8App Platform
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Reports on Key Milestones for 2023, Provides Update on Targets Moving Forward
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - InvestorNewsBreaks - FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) Receives $1.9M Funding Grant from Canadian Government ACT Program
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) to Present at BIO-Europe 2023
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Reports Record Preliminary Q3 2023 Revenues, Announces Earnings Conference Call
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) to Host Third Quarter 2023 Results Conference Call
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) Sees Ongoing Traction in Customer Demonstrations; Nears Proof-of-Concept for New Technology Offering
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream’s Investing App Signifies Potential of Blockchain Technology, Next Phase of Capital Markets
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Cannabinoid-Based Topical Pain-Relief Treatment Receives European Patent
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) Secures $5.2M in Private Placement and Preferred Investment Option Offerings
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Granted Second U.S. Patent for JW-700 Treatment Designed for $1.5B Hair-Growth Market
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Secures Expanded and New Contracts for K1 Technologies
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Longeveron Inc. (NASDAQ: LGVN) Validates Safety and Therapeutic Potential of its Lomecel-B(TM) Lead Investigational Product in its CLEAR MIND Phase 2a Clinical Trial
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Get Ready for Next Generation of Diabetes, Weight-Management Drugs
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Study Suggests Methamphetamine More Helpful for Social Bonding Compared to MDMA
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Releases Update on Q3 Production
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (CSE: MYCO) (AQSE: MYIG) (OTC: MYCOF) Files Prospectus Supplement, Closes Share Subscription Agreements
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Appointment of Strategic AI Advisor
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Brand Metrics e-Commerce Companies Need to Watch
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - InvestorNewsBreaks - Mullen Automotive Inc. (NASDAQ: MULN) Request to Continue Listing Is Granted by NASDAQ Hearings Panel
- PaxMedica Inc. (NASDAQ: PXMD) - InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Acquires Research Assets to Accelerate PAX-101 NDA Submission
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Announces Addition of Meek's Vegan Pizza to Chicago Foodhall Offerings
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Q3 Player Activity Milestones
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Bi-Weekly Status Report
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - New to The Street Announces Four Corporate Interviews on its 525th Show, Airing on the Fox Business Network, Monday, October 30, 2023, at 10:30 PM PT
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Creates Advisory Board, Names Renowned Experts as Members
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Reports on Q3 2023 Performance, Debt Financing
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Releases Corporate Update, Including Aleafia Health Agreement
- Renovaro BioSciences Inc. (NASDAQ: RENB) - Alarming Data Highlights Gaps in Treating People with Chronic HBV
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Swedish Scientists Find Promising Way to Recover Minerals from Discarded EV Batteries
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Reports on Strong Drill Results at Oko West Project in Guyana
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Becomes Largest Shareholder of Plantify Foods Inc.
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters into Acquisition Agreement, Asset Purchase Agreement
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Positive Results of Preclinical Study to Advance SPU-21
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Eyes the Growing World Market for Domestic Violence Offender Monitoring Solutions
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Closing of $1.6M Public Offering, $750K Order for Use by First Responders in Israel
- SOHM Inc. (OTC: SHMN) - SOHM, Inc. (SHMN) Completes Acquisition of ABBIE Gene-editing Tech, Begins Preparations to Commercialize Solutions for Heart Disease
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Exclusive Single-Serve Wine Brand to Be Featured by 130-Plus Circle K Locations
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Makes 'Significant Move' with Exercise of SWA Project Option Agreement
- Starco Brands Inc. (OTCQB: STCB) - Starco Brands to Present at LD Micro Main Event XVI
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Subsidiary Meets with Jordan Minister of Water in Round Table Gathering
- SenesTech Inc. (NASDAQ: SNES) - InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Now Taking Orders for Evolve(TM) Soft Bait
- Sekur Private Data Ltd. (FRA: GTD0) (CSE: SKUR) (OTCQB: SWISF) - InvestorNewsBreaks - Sekur Private Data Ltd. (CSE: SKUR) (OTCQB: SWISF) (FSE: GDT0) Announces Entry into Multinational Distribution Agreement in the Middle East
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Receives Full Compliance Notification from NASDAQ
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Enters MOU to Launch Business, Service Offerings in Pakistan
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Closes on AUD$175,000 Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TrxADE Health, Inc. affirms its commitment to the pharmaceutical space via its subsidiary Trxade, Inc. which operates the pharmaceutical marketplace and added more members in the third quarter of 2023.
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - MiningNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) Executing Louisiana Project with Potential to Significantly Enhance North America's Manufacturing Landscape
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Releases FY 2023 Report
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - Vistagen Therapeutics Inc. (NASDAQ: VTGN) Announces Participation in Inaugural Biotech CNS/Neuro Summit
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Pricing of $4M Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Giles Tipsword Joins Wrap Technologies as Senior Vice President of North American Sales
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































