The QualityStocks Daily Stock List
- JanOne (JAN)
- Hiru Corporation (HIRU)
- Mister Car Wash (MCW)
- ProKidney (PROK)
- Luminar Technologies (LAZR)
- Pacific Biosciences of California (PACB)
- Equinox Gold (EQX)
- OneMedNet Corporation (ONMD)
- Cresco Labs Inc. (CRLBF)
- Cenntro Electric Group Ltd. (CENN)
- Canopy Growth Corp. (CGC)
- Carrols Restaurant Group (TAST)
JanOne (JAN)
Broad Street, AwesomeStocks, StockMarketWatch, BUYINS.NET, StockStreetWire, Small Cap Firm, TradersPro, QualityStocks, StockWireNews, Fierce Analyst, MarketClub Analysis, MicroCapDaily, OTCtipReporter, Penny Stock 101, PennyStockLocks, PennyStockScholar, Schaeffer's, StockRockandRoll, StreetInsider, TopPennyStockMovers and PennyStockProphet reported earlier on JanOne (JAN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
JanOne Inc. (NASDAQ: JAN) (FRA: 5AR1) is a clinical-stage biopharmaceutical firm that is engaged in the identification, acquisition, licensing, development and commercialization of new, non-addictive, non-opioid treatments that address previously unmet medical needs to treat pain.
The firm has its headquarters in Las Vegas, Nevada and was incorporated in 1976 by Edward R. Cameron. Prior to its name change in September 2019, the firm was known as Appliance Recycling Centers of America Inc. It operates in the healthcare sector, under the biotech and pharma sub-industry and serves consumers in the United States.
The company operates in the Technology, Recycling and Biotechnology segments. Its recycling segment is engaged in the collection, recycling and installation of appliances for utilities and other consumers. It also focuses on the General Electric agreement. Its other segments are involved in selling appliance via Appliance Smart stores.
The enterprise’s product pipeline is made of a patented oral and sustained release pharmaceutical sodium nitrite composition which targets poor blood flow in patients suffering from peripheral artery disease and diabetes and is indicated for the treatment of pain. The enterprise also offers turnkey appliance replacement and recycling services for sponsors of efficiency programs. This is in addition to designing, developing and selling cellular transceiver modules and associated wireless services.
The firm recently appointed a world renowned expert on addiction to its advisory board and he brings with him decades of experience in drug addiction. This addition will be useful as the firm advances its treatment for PAD and will also be beneficial to their patients who will be utilizing their products.
JanOne (JAN), closed Thursday's trading session at $0.531, up 64.7533%, on 59,496,274 volume. The average volume for the last 3 months is 21.402M and the stock's 52-week low/high is $0.2204/$2.23.
Hiru Corporation (HIRU)
OTCReporter, Mina Mar Marketing Group, Willy Wizard, Stockpalooza, CRWEWallStreet, PennyOmega, Penny Invest, DrStockPick, StockHotTips, BestOtc, CRWEFinance, CoolPennyStocks, HotOTC, BullRally, Stock Rich, PennyToBuck, StockEgg, CRWEPicks, OTCPicks, Epic Stock Picks, StockRockandRoll, Penny Stock 101, MarketClub Analysis, PennyStockLocks, Stocks Gone Wild, Wise Alerts, Micro Cap Momentum, HEROSTOCKS, QualityStocks, Topgun stockpicks, Penny stock Profitz, The Stock Psycho, Stocktwiter, Purely Penny Stocks, Stock Exploder, Bull Warrior Stocks, StocksAlarm, StockMister, Penny Stocks Pushers, SmarTrend Newsletters, Penny Picks, Greenbackers, Pumps and Dumps, Light Speed Stocks, PennyStockRumors.net, Wise Penny Stocks, BeatPennyStocks and Damn Good Penny Picks reported earlier on Hiru Corporation (HIRU), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Hiru Corporation (OTC: HIRU) is engaged in the production of Chinese herbs for China’s naturopathic industry.
The firm has its headquarters in Phoenix, Arizona and was incorporated in 1989. Prior to its name change in November 2008, the firm was known as Phoenix Restaurant Group Inc.
The company distributes and sells its pharmaceuticals, dietary and herbal supplements, beauty and health products and other healthcare products via regional distributors, as well as directly to pharmacies, clinics and hospitals in China.
The enterprise manufactures herbal supplements that contain ginseng and an additional 120 extracts which are also utilized in traditional Chinese medicine. It also provides consumers in the state of Arizona with bottled water and bagged ice. In addition, it is involved in the development, manufacture and commercialization of various veterinary products for the agricultural market in China via its Jiangxi Shuangshi AHP Co. subsidiary whose objective is to protect both human and animal health. The enterprise is focused on expanding its research and development and is planning to introduce veterinary solutions and drugs to the Chinese market via its subsidiary. To improve the health of livestock, the firm produces premixes, loose powders, feed additives, liquid disinfectants, oral liquids, injections, volume injections and other injectables.
The company is focused on meeting all of its consumers’ demands having recently appointed a new CEO. This appointment will help bring in a lot of investment opportunities as well as business into the firm, which are bound to have a positive effect on the company’s growth.
Hiru Corporation (HIRU), closed Thursday's trading session at $0.0015, up 66.6667%, on 109,262,947 volume. The average volume for the last 3 months is 1.681M and the stock's 52-week low/high is $0.0007/$0.006.
Mister Car Wash (MCW)
MarketBeat, UndiscoveredEquities, Trades Of The Day, StocksEarning, Mina Mar Marketing Group, Daily Trade Alert, wyatt research newsletter, The Street and InvestorPlace reported earlier on Mister Car Wash (MCW), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Mister Car Wash, Inc. (NYSE: MCW) is a car wash firm focused on offering express interior and express exterior cleaning services in addition to related services to vehicle owners.
The firm is headquartered in Tuscan, Arizona and was founded in 1996. Prior to its name change in March 2021, the enterprise was known as Hotshine Holdings Inc. it operates in the other commercial services industry under the automotive sector. As of 2022, it had 6,350 employees. It serves customers in 21 U.S. states and is actively working to spread its footprint to more states.
Mister Car Wash offers a monthly subscription service to car owners through the UWC (Unlimited Wash Club). This service allows the firm’s clients to have access to cleaning services for their cars without worrying about having to pay each time they visit the carwash. The company uses RFID identification to ensure that clients are served quickly, conveniently and with minimal contact with staff, especially after the Covid-19 pandemic. The firm divides its locations into Express Exterior Locations, which specialize in offering exterior cleaning services only, as well as the Express Interior Locations, which offer both interior and exterior cleaning services.
The firm also offers the Mister Express Lube through which clients can access preventive maintenance services as well as engine oil changes. Its other products include Wheel Polish, Tire Shine, Repel Shield, HotShine, and N1 Protectant. The enterprise earns most of its revenue from the Unlimited Wash Club subscription service.
The company, which is the largest carwash brand in the U.S., remains focused on giving car owners an industry-leading “Mister Experience” by constantly improving how they serve their customers through innovations like dedicated lanes for UWC members to enable them get into and out of the carwash as quickly as possible.
Mister Car Wash (MCW), closed Thursday's trading session at $6.45, up 2.2187%, on 1,969,174 volume. The average volume for the last 3 months is 647,364 and the stock's 52-week low/high is $4.80/$11.14.
ProKidney (PROK)
MarketBeat, The Stock Dork and Money Wealth Matters reported earlier on ProKidney (PROK), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
ProKidney Corp (NASDAQ: PROK) (BMV: PROKN) is an American clinical-stage biotechnology firm that offers proprietary cell therapy platforms to treat various chronic kidney diseases using patient cells.
Its headquarters are in Winston-Salem, North Carolina and it was founded in 2005. The firm operates in the biotechnology industry, under the healthcare sector. Its main product candidate is Renal Autologous Cell Therapy (REACT) which is designed to preserve kidney function in patients with chronic kidney disease (CKD).
REACT is an autologous homologous cell admixture that is currently in Phase I trials for patients with Congenital Anomalies of the Kidney and Urinary Tract (CAKUT). The investigational cell therapy product candidate is also in Phase II and Phase III clinical trials for the treatment of moderate to severe kidney diseases as well. It comprises autologous renal cells formulated in a gelatin-based hydrogel and organ regeneration technology to develop the therapeutic candidate. The therapy uses selected renal cells (SCRs) which are collected from the patient’s renal biopsy tissue and reinjected into the patient’s kidney via a minimally invasive outpatient process (intra-renal injection). By leveraging a patient's cells rather than donor cells, REACT eliminates the need for immunosuppressive therapies which kidney transplant patients have to take for life to prevent transplant rejection. ProKidney Corp’s experimental therapy is currently under development for the treatment of type II diabetes, unspecified genito urinary disorders, and chronic kidney disease.
It estimates that around 4.5 million of the estimated 13.4 million patients with stage 3 or 4 CKD caused by diabetes will be eligible for REACT treatment if the therapy receives FDA approval.
ProKidney (PROK), closed Thursday's trading session at $1.67, off by 0.298507%, on 550,285 volume. The average volume for the last 3 months is 4.719M and the stock's 52-week low/high is $1.45/$14.19.
Luminar Technologies (LAZR)
Schaeffer's, InvestorPlace, MarketBeat, MarketClub Analysis, StocksEarning, INO Market Report, Daily Trade Alert, StockEarnings, Trades Of The Day, The Street, The Online Investor, Zacks and InsiderTrades reported earlier on Luminar Technologies (LAZR), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Luminar Technologies Inc. (NASDAQ: LAZR) (FRA: 9FS) (BVMF: L2AZ34) (BMV: LAZR) is an American automotive technology firm that produces vision-based machine perception and lidar technologies for commercial trucks and passenger cars.
The firm is headquartered in Orlando, Florida, and was incorporated in December 2012. It operates in the auto parts industry, under the consumer cyclical sector and has two segments; Advanced Technologies Services, and Autonomy Solutions. The auto technology company mainly serves carmakers in the U.S., Canada, Europe, the Middle East, and the Asia Pacific.
The company offers a wide range of technological solutions in the two segments it operates. In the Autonomy Solutions segment, it designs, produces, and sells laser imaging detection, and ranging lidars or sensors coupled with perception and autonomy software for OEMs (original equipment manufacturers) in the robo-taxi, commercial, automobile, commercial vehicle, and aeronautics, military, and defense markets. The Advanced Technologies and Services segment provides advanced lasers, ultra-sensitive pixel-based sensors, and application-specific integrated circuits. It also deals in design, testing, and consulting services for non-standard integrated circuits used in the aeronautics and automobile sectors as well as defense and military activities. Luminar Technologies develops Iris and Hydra commercial lidar sensors that use laser light to measure distance and generate 3D maps for enhancing vehicle safety and autonomy. The company’s technology has major applications in autonomous cars and Luminar has secured deals to supply lidar hardware and proprietary software to Mercedes-Benz, Volvo cars, and electric carmaker Polestar.
It remains committed to becoming the first technology firm to develop next-gen safety and autonomous capabilities for passenger cars, commercial trucks, and more.
Luminar Technologies (LAZR), closed Thursday's trading session at $3.13, off by 14.9457%, on 9,911,160 volume. The average volume for the last 3 months is 4.381M and the stock's 52-week low/high is $3.01/$10.55.
Pacific Biosciences of California (PACB)
MarketClub Analysis, Wall Street Resources, BUYINS.NET, MarketBeat, INO.com Market Report, StockEarnings, StreetInsider, The Street, TradersPro, Schaeffer's, StockOodles, StocksEarning, StockMarketWatch, Kiplinger Today, SmarTrend Newsletters, Barchart, Marketbeat.com, Hit and Run Candle Sticks, Zacks, Dynamic Wealth Report, Rick Saddler, FeedBlitz, Greenbackers, Investment U, CNBC Breaking News, INO Market Report, ChartPoppers, Wealthpire Inc., Investing Futures, Investment House, Daily Trade Alert, InvestmentHouse, InvestorPlace, Investors Underground, InvestorsUnderground, AllPennyStocks, Trades Of The Day, Money and Markets, PennyStockCrowd, QualityStocks, RedChip, Sling-Shot-Stocks, Stock Beast, Stock Market Watch, Top Secret Stocks and Jason Bond reported earlier on Pacific Biosciences of California (PACB), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Pacific Biosciences of California Inc. (NASDAQ: PACB) (FRA: P09) is an American biotechnology firm that focuses on developing and manufacturing systems for gene sequencing.
The firm is headquartered in Menlo Park, California. It was incorporated in 2000 under the name Nanofluidics, Inc. and changed its name to Pacific Biosciences of California Inc. in 2004. It operates as part of the medical devices industry, under the healthcare sector. Pacific Biosciences of California mostly serves corporate clients in the United States, Canada, Asia, Europe, Latin America, the Middle East, and Africa.
The company was created based on Cornell University research that combined biotechnology research with photonics and semiconductor processing. It leverages HiFi long read sequencing technology and short read Sequencing by Binding (SBB®) technology to create advanced sequencing systems for solving complex genetic problems. The publicly-held firm provides sequencing systems, consumable products such as single-molecule real-time (SMRT) cells, and a variety of reagent kits for specific workflows. The firm also offers binding kits to aid in genetic sequencing and sequencing kits with the reagents needed for real-time, on-instrument sequencing. The company’s products have applications in oncology, plant and animal sciences, and infectious disease and microbiology. Its primary clients are commercial laboratories, research institutions, hospitals, public health labs, contract research organizations, clinical research institutes, agricultural companies, pharmaceutical companies, and genome centers. PACB currently has a development and commercialization agreement with genetic information provider Invitae Corporation and has partnered with Radboud University Medical Center in Nijmegen to study the genetic causes of rare genetic disorders.
It remains committed to producing products that provide solutions for applications in oncology, infectious disease and microbiology, human germline sequencing, plant and animal sciences, and other emerging segments.
Pacific Biosciences of California (PACB), closed Thursday's trading session at $6.52, off by 2.5411%, on 3,522,035 volume. The average volume for the last 3 months is 2.201M and the stock's 52-week low/high is $5.74/$14.55.
Equinox Gold (EQX)
Zacks, MarketClub Analysis, TradersPro, Top Pros' Top Picks, StocksEarning, MarketBeat, StockMarketWatch, Money Wealth Matters, InvestorPlace, INO Market Report, Barchart and AllPennyStocks reported earlier on Equinox Gold (EQX), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Equinox Gold Corp (NYSE American: EQX) (TSE: EQX) (FRA: 1LRC) is a mining, mineral exploration and development firm that specializes in the exploration, acquisition, development, and operation of mineral properties with a focus on gold and silver.
Its headquarters are in Vancouver, British Columbia, Canada and it was incorporated in 2007 as Trek Mining Inc. The Canadian miner changed its name to Equinox Gold Corp in December 2017. It operates in the gold industry, under the basic materials sector, and holds an interest in several mining exploration projects in the Americas. Equinox Gold Corp operates in the U.S., Canada, Mexico, and Brazil.
The firm is involved in the acquisition, exploration, development, and production, of gold, silver, and copper mineral deposit properties. It fully owns and operates seven gold mines across the Americas; the Los Filos Mine Complex in Guerrero State, Mexico, the Castle Mountain Gold Mine and the Mesquite Gold Mine in California, the Fazenda Gold Mine and Santa Luz Gold Mine in Brazil’s Bahia State, the RDM Gold Mine in Mina Gerais State, Brazil, and the Aurizona Gold Mine, an open-pit gold mine in Maranhão State, Brazil. Current projects include the Los Filos Expansion, Greenstone Project, Castle Mountain Expansion, and Aurizona Expansion which will increase the miner’s annual gold output by close to 600,000 ounces. Equinox Gold Corp plans to nearly double its yearly gold production output by advancing expansion and development projects.
The company’s vision is to build a firm that will produce over one million ounces of gold annually responsibly and safely.
Equinox Gold (EQX), closed Thursday's trading session at $4.59, even for the day, on 1,449,912 volume. The average volume for the last 3 months is 43,201 and the stock's 52-week low/high is $3.12/$5.845.
OneMedNet Corporation (ONMD)
Real Pennies and MicroCap Gems reported earlier on OneMedNet Corporation (ONMD), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
OneMedNet (NASDAQ: ONMD, ONMDW), the leading curator of regulatory-grade Imaging Real World Data ("iRWD") through its proven OneMedNet iRWD(TM) solution, has completed its previously announced business combination with Data Knights Acquisition Corp. As a result, the company’s common stock began trading on NASDAQ under the ticker ONMD earlier this week. According to the announcement, the business combination was approved during a special meeting of Data Knights stockholders. The company offers innovation solutions that connect healthcare providers and patients, offering direct access to clinical images and associated contextual patient record. “Reaching this milestone represents a strong step forward for OneMedNet,” said OneMedNet CEO Paul Casey in the press release.
“The finalization of the merger with Data Knights is a direct testament to the diligence and hard work of the collective teams and the continued acknowledgement of OneMedNet’s unique and innovative regulatory grade iRWD data solution, which supplies the life sciences industry with an actionable component to earlier detection and monitoring in the discovery of therapeutics and treatments. The company has made significant strides in product and innovation capabilities and formed a solid operating structure required to effectively accelerate our growth strategy, including expanding our network through our active pipeline of partnership opportunities, and expanding OneMedNet’s team to include leading industry talent. The $400 billion market of clinical research and clinical trials is a minimally penetrated greenfield opportunity for the Company and serves as a sustainable and ongoing competitive advantage to accelerate our growth and ultimately unlock value for all of our stakeholders.”
To view the full press release, visit https://ibn.fm/ovKeN
About OneMedNet Corporation
Founded in 2009, OneMedNet provides innovative solutions that unlock the significant value contained within the clinical image archives of healthcare providers. Employing its proven OneMedNet iRWD solution, OneMedNet securely deidentifies, searches and curates a data archive locally, bringing a wealth of internal and third-party research opportunities to providers. By leveraging this extensive federated provider network, together with industry leading technology and in-house clinical expertise, OneMedNet successfully meets the most rigorous RWD Life Science requirements. For more information about the company, please visit www.OneMedNet.com.
OneMedNet Corporation (ONMD), closed Thursday's trading session at $4.49, off by 15.1229%, on 45,278 volume. The average volume for the last 3 months is 962,678 and the stock's 52-week low/high is $3.95/$13.51.
Cresco Labs Inc. (CRLBF)
InvestorPlace, QualityStocks, Kiplinger Today, Daily Trade Alert, MarketBeat, Cabot Wealth, Top Pros' Top Picks, The Street, The Wealth Report, The Online Investor, Wealth Insider Alert, Trading For Keeps, Trades Of The Day, Early Bird, StreetInsider, wyatt research newsletter, TradersPro and StocksEarning reported earlier on Cresco Labs Inc. (CRLBF), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Despite differing opinions among ardent supporters, cannabis legalization in Ohio could entail certain drawbacks. Nonetheless, an economic analysis published last week suggests that the advantages would surpass these drawbacks by a yearly sum of $250 million. Issue 2, a proposal to legalize recreational cannabis in Ohio, is set for the upcoming election on Tuesday.
A study conducted by Scioto Analysis sought to assess the potential benefits and drawbacks of such a step in a state where medical cannabis is already legal. To conduct the analysis, the research group drew on findings from states such as Colorado and Washington, where recreational cannabis has been legal for some time. To understand how the advantages and disadvantages identified in those states might apply to Ohio, the researchers used demographic and economic data, as well as crime statistics.
The most significant advantage they identified relates to the additional revenue that Issue 2’s passage would generate, thanks to its 10% excise tax in addition to Ohio’s regular sales tax. However, the report clarifies that the benefit doesn’t solely stem from the anticipated yearly tax revenue of $190 million but rather from how a substantial portion of these funds would be utilized.
According to the report, benefits are realized when services and goods procured by governments yield positive spillover effects. The researchers pinpointed positive spillovers from the 25% and 36% of Issue 2’s revenue allocated to the Substance Abuse Addiction and Marijuana Social Equity and Jobs Funds, respectively. Drawing from analyses of similar funds in other states, the group estimated that the Ohio equity and jobs fund would generate $5.76 in benefits for every dollar spent, and the substance abuse fund would yield $9.19. The report suggests that directing revenue to these two funds could potentially create $820 million worth of benefits for Ohioans annually.
Additionally, the new industry would directly create jobs. The report projects approximately $190 million in wage benefits during the first year of legalization for workers across the state, encompassing both full-time and part-time positions.
Recreational marijuana users would also enjoy benefits, as Issue 2 would reduce the annual marijuana expenditure of this group by $98 million compared to what they would pay otherwise. Furthermore, the legalization of recreational marijuana would likely lead to a reduction in arrests.
When it comes to the cons, one significant drawback is the decrease in worker productivity observed in states where legalization has already occurred. Citing a study from 2017, the report suggests that average productivity per worker dipped by just over 1% in certain industries in states that legalized recreational cannabis. Monetarily, this translates to roughly $900 of lost productivity per worker in Ohio. The report utilized federal employment data to estimate that legalization could cost Ohio workers approximately $760 million in lost productivity in the first year alone.
Moreover, concerns about public safety emerge with the possibility of more intoxicated drivers on the roads, leading to increased accidents causing injuries, property damage and even fatalities. The report estimates an additional 1,700 intoxicated-driving arrests annually in Ohio, with an associated cost of $130 million.
Considering these positives and negatives, the study suggests that if Issue 2 is passed, Ohioans could potentially gain annual benefits of $260 million.
This analysis of the forecast benefits of ending marijuana prohibition in Ohio goes to show what an impact companies such as Cresco Labs Inc. (CSE: CL) (OTCQX: CRLBF) might have once a legal cannabis market is launched and entrepreneurs are allowed to address the needs of customers.
Cresco Labs Inc. (CRLBF), closed Thursday's trading session at $1.26, up 3.2787%, on 379,026 volume. The average volume for the last 3 months is 1.506M and the stock's 52-week low/high is $1.00/$4.20.
Cenntro Electric Group Ltd. (CENN)
QualityStocks, GreenCarStocks, TradersPro, Penny Stock and InvestorPlace reported earlier on Cenntro Electric Group Ltd. (CENN), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Aside from price, electric vehicle range is one of the key metrics drivers consider before switching from gas-powered cars to battery electric vehicles (BEVs). Range refers to the distance an electric car can travel on a single charge and is affected by a battery’s maximum capacity as well as factors such as weather and driver habits.
The average electric vehicle driver is interested in optimizing their EV’s performance and maximizing the range, especially if they rely on public charging infrastructure. This allows them to travel further on a single charge and limits the need for constant stops at public charging stations.
Optimizing your car’s regenerative braking can help you feed energy into the battery while you drive. Regenerative braking is a feature that works much like a generator and converts an EV’s kinetic energy into electricity when it brakes. This allows you to feed a little energy into your EV’s battery whenever you brake, giving your EV a little extra range.
As there are several different levels of regenerative braking, you will have to experiment with each level to find the most efficient setting for your electric car.
Schedule your air conditioning usage. Since all your EV’s electricity-reliant features draw power from the battery, features such as air conditioning can reduce your capacity and eat into your range. The hotter the temperature gets, the more power your electric car’s AC will draw from the battery. You can mitigate this issue by setting your AC to kick in once your electric car is done charging.
This will allow you to heat your cabin with energy from the grid rather than the battery, limiting the pressure on your EV’s battery and maximizing the range.
Follow the EV’s consumption indicator. The majority of modern electric cars have consumption indicators that monitor power use in real time similar to a fuel gauge in internal combustion engine (ICE) cars. However electric car consumption indicators are much more efficient at tracking energy use and can allow you to adjust your driving habits and styles to maximize EV battery efficiency.
Charge at lower amperages to preserve your EV battery’s integrity for the long-term. While fast and super-fast charging is incredibly convenient because it cuts charging times significantly, regular fast charging can put a major strain on your EV battery. This can result in battery degradation and reduce your battery’s lifespan. Charging at a lower amperage is more efficient because it preserves your battery cells’ integrity and promotes efficiency.
Use driver-only mode if your electric car has this feature. It reduces energy consumption by lowering the air-conditioning load, allowing you to cut your EV’s energy consumption without impacting cabin comfort.
Coupled with proper EV driving habits such as avoiding rapid acceleration, high speed and aggressive driving, these tips can help you optimize EV power usage and maximize your range.
Motorists can also refer to the specific guidelines offered by companies such as Cenntro Electric Group Ltd. (NASDAQ: CENN) in order to maximize efficiency and range in their EVs.
Cenntro Electric Group Ltd. (CENN), closed Thursday's trading session at $0.175, off by 4.7878%, on 802,737 volume. The average volume for the last 3 months is 52.163M and the stock's 52-week low/high is $0.15/$0.955.
Canopy Growth Corp. (CGC)
InvestorPlace, Schaeffer's, The Street, Trades Of The Day, MarketClub Analysis, MarketBeat, Daily Trade Alert, Kiplinger Today, StocksEarning, StockEarnings, The Online Investor, Wealth Insider Alert, Streetwise Reports, StreetInsider, CFN Media Group, QualityStocks, Market Intelligence Center Alert, Investopedia, Zacks, StreetAuthority Daily, Stock Up Featured, The Wealth Report, Daily Profit, Top Pros' Top Picks, SmallCapVoice, Lebed.biz, SeriousTraders, StockMarketWatch, Wall Street Grand, Profit Trends, Early Bird, Money Morning, INO Market Report, Inside Trading, Jim Cramer, CNBC Breaking News, Cannabis Financial Network News, Louis Navellier, BUYINS.NET, StocksToBuyNow, Outsider Club, Trading For Keeps, MarketClub, AllPennyStocks, Beat The Street, Wealth Daily, Cabot Wealth, VectorVest, Trading Concepts, TradersPro, TheTradingReport, Profit Confidential, Stock Gumshoe, Insider Wealth Advice, Investment U, InvestmentHouse, Rick Saddler, Raging Bull All Access, Investors Alley, 24/7 Trader, Money and Markets and Technology Profits Daily reported earlier on Canopy Growth Corp. (CGC), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Beginning in December, adults in the state of Connecticut will be allowed to buy up to one-half an ounce of cannabis in one retail transaction. This comes after the state doubled its purchase limit from quarter of an ounce of marijuana.
The state’s Department of Consumer Protection announced the scheduled increase last week, explaining that it was the result of a continuing analysis of demand and supply and was designed to make certain businesses maintain sufficient supply for both medical cannabis patients and adult-use consumers.
It should be noted, however, that the limits imposed on medical cannabis remain unchanged.
The state department’s commissioner, Bryan T. Cafferelli, added that as more production companies, retailers and other licensees in the supply chain came online, the industry’s capacity had increased. Cafferelli noted that they were confident the measured approach to recreational sales had created a healthy market for businesses as well as a fair and safe marketplace for both medical cannabis patients and adult-use marijuana consumers.
Currently, possession of marijuana in the state is capped at one and a half ounces for adults. Adults are also allowed to store no more than five ounces in a locked trunk, glovebox or a locked container in their homes. Cafferelli also highlighted that purchased marijuana needed to remain in its original packaging and be out of reach of children.
The state legalized the recreational use of cannabis via the legislature in 2021. Since then, the state has recorded steady sales. Thus far, the adult-use market has seen a number of consecutive record-setting months of legal purchases. For instance, the state recorded $10.8 million worth of medical marijuana sales and $14.4 million in adult-use sales in September.
It is important to remember that the limits imposed on the purchase of products besides raw marijuana flower are set by equivalence.
Earlier in August, the state also launched a fund to provide support to social equity cannabis businesses. The established loan program will offer financial assistance to help individuals who have been disproportionately affected by the war on drugs to expand their businesses in this burgeoning industry.
In addition, in July of this year, the state legalized home cultivation of cannabis for personal use. It also adopted tax breaks for legal marijuana businesses in the same month. This move to help businesses that cannot make federal deductions under the IRS 280E code is expected to bring in $4.7 million in industry relief for the 2024 fiscal year. Projections from the office of the governor expect this figure to increase in the 2025 fiscal year, reaching $6.2 million.
The growth that is likely to result from this recent adjustment to how much recreational marijuana adults can buy could create opportunities for many local companies to grow to the levels experienced by companies that are licensed in other jurisdictions, such as Canopy Growth Corp. (NASDAQ: CGC) (TSX: WEED).
Canopy Growth Corp. (CGC), closed Thursday's trading session at $0.523, off by 10.0138%, on 36,286,088 volume. The average volume for the last 3 months is 555,865 and the stock's 52-week low/high is $0.346/$4.77.
Carrols Restaurant Group (TAST)
Zacks, MarketBeat, TradersPro, Market Intelligence Center Alert, Trades Of The Day, INO.com Market Report, The Street, Daily Trade Alert, StreetInsider, InvestorPlace, SmarTrend Newsletters, QualityStocks, Marketbeat.com, BUYINS.NET, OTCPicks, Street Insider, One Hot Stock, SmallCapNetwork, StockOodles, StocksEarning, InvestorGuide, Investing Lab, Top Pros' Top Picks, Greenbackers, Wealth Insider Alert and Cabot Wealth reported earlier on Carrols Restaurant Group (TAST), and today we highlight the Company, here at the QualityStocks Daily Newsletter.
Carrols Restaurant Group Inc. (NASDAQ: TAST) (FRA: XQC) is a restaurant firm that is focused on operating restaurants under the Popeyes and Burger King brands.
The firm has its headquarters in Syracuse, New York and was incorporated in 1960. It operates as part of the restaurants industry, under the consumer cyclical sector. The firm serves consumers around the globe.
The company helps deliver training individuals need to fulfill their career ambitions, starting with its workforce, which is trained using state-of-the-art equipment. It operates through its subsidiaries, which include Carrols LLC, Carrols Corporation, Carrols Holdco Inc., Cambridge Franchise Real Estate LLC and Republic Foods Inc.
The enterprise’s Burger King restaurants are fast-food hamburger restaurants that feature the flame-broiled Whopper sandwich, as well as a range of hamburgers, chicken and other specialty sandwiches, salads, French fries, breakfast items, snacks, soft drinks and more. It operates roughly 1,025 Burger King restaurants located in more than 23 Midwestern, Northeastern, Southeastern and Southcentral states. On the other hand, its Popeyes restaurants are quick-service chicken restaurants that feature a Louisiana-style menu, including fried chicken, chicken tenders, fried shrimp and other seafood, red beans and rice and other regional offerings. It operates about 65 Popeyes restaurants in over 7 Southeastern states.
The company, whose latest results show increases in its restaurant sales, is committed to making progress on its growth drivers, strengthening its operations and boosting brand awareness. This will increase traffic, allow it to extend it consumer reach and help drive long-term shareholder value, all while bolstering its overall growth.
Carrols Restaurant Group (TAST), closed Thursday's trading session at $7.63, up 22.6688%, on 3,822,492 volume. The average volume for the last 3 months is 282 and the stock's 52-week low/high is $1.31/$7.98.
The QualityStocks Company Corner
- Diamond Lake Minerals Inc. (OTC: DLMI)
- SenesTech Inc. (NASDAQ: SNES)
- Freight Technologies Inc. (NASDAQ: FRGT)
- Electronic Servitor Publication Network Inc. (OTCQB: XESP)
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF)
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF)
- PaxMedica Inc. (NASDAQ: PXMD)
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
- HeartBeam Inc. (NASDAQ: BEAT)
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI)
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF)
- SOHM Inc. (OTC: SHMN)
Diamond Lake Minerals Inc. (OTC: DLMI)
The QualityStocks Daily Newsletter would like to spotlight Diamond Lake Minerals Inc. (OTC: DLMI).
Diamond Lake Minerals (OTC: DLMI), a leading multistrategy operating company specializing in the development and support of digital assets and SEC-registered security tokens, is spotlighted in a recent Benzinga article noting the importance of integrating traditional investments with more dynamic technology. Titled "How Diamond Lake Minerals Is Spearheading Digital Asset Accessibility for Traditional Investors," the article notes that the company is leveraging decades of traditional business acumen to support investors as they move into the digital asset and security token market. The article observes that DLMI has entered a "fascinating transition" from mineral exploration to spearheading innovation in digital assets and security tokens. With that transition in mind, the company has a strategic PR plan in place to promote digital assets and educate the public about those assets while also working to make such investments more approachable to the general public. "With a vision that has evolved since its founding in 1954, Diamond Lake Minerals now focuses on the development and support of SEC-registered security tokens under the guidance of CEO Brian J. Esposito and his 20 years of industry experience," states the article. "In addition, big-name advisors such as Anthony Scaramucci, Larry Namer, Andrew Fromm, Brandon Fugal, Michael Malik Sr., Raul Leal, Agnes Budzyn and David Meltzer have joined the company, bringing years of expertise and success. Through blockchain innovations unlocking asset tokenization, Diamond Lake Minerals is shaping the landscape of investment for generations accustomed to the tangibility of assets — but with a modern twist that embraces new technologies."
To view the full article, visit https://ibn.fm/xY8hY
Diamond Lake Minerals Inc. (OTC: DLMI) is a multi-strategy operating company offering traditional investors an entry point to the future of digital securities. The company’s goal, through its established M&A roadmap, is to responsibly innovate and develop promising businesses that are likely to benefit from the ongoing shift toward digital assets. Through this approach, Diamond Lake Minerals provides traditional investors an opportunity to gain exposure to the emergence of regulated digital securities through a more familiar investment vehicle – the purchase of stock.
Founded in 1954 and headquartered in Salt Lake City, Diamond Lake Minerals is positioning itself as a leader in the digital asset and security token space. The company’s mission is to bring back to the public markets timeless business principles focused on healthy, sustainable growth and strong earnings with a goal of creating value for stakeholders in the modern digital world.
Diamond Lake Minerals believes the future of financial markets is set to be revolutionized by tokenization. Tokenization refers to the use of digital assets that can be traded via protocols with instantaneous settlement and reduced fees, eliminating the need for traditional clearing or settlement processes. Beyond efficiency, the emerging landscape emphasizes transparency, liquidity and security in asset management and investment.
With the backing of Esposito Intellectual Enterprises and its 20+ years of experience, Diamond Lake Minerals has access to the expertise of 110+ companies and 200+ joint ventures, along with knowledge spanning 25+ industries. The company is creating a vertically integrated ecosystem that encompasses various high-growth sectors. This integration aims to maximize operational efficiencies and profitability across all business units.
Products & Services Portfolio
Diamond Lake Minerals, guided by its strategic partnerships and future roadmap, envisions a diverse portfolio across multiple industries, as shown in the overview below. The company is poised to redefine the conglomerate model for the 21st century, with a focus on vertical integration, digital securities and sustainable growth.
Its target market segments include:
- Fashion: DLMI seeks stakes in brands blending timeless aesthetics with tech influences.
- Beauty: DLMI eyes partnerships with innovators elevating beauty through sustainable practices.
- Real Estate: DLMI aims for interests in ventures modernizing property transactions via blockchain.
- Hospitality: DLMI’s vision includes associations with enterprises enhancing guest experiences via tech integration.
- Liquor: DLMI aspires to collaborate with unique distillers merging tradition and innovation.
- IoT: DLMI intends to invest in solutions seamlessly connecting the digital and physical worlds.
- Wireless: DLMI envisions stakes in wireless tech optimizing global communication.
- Technology: DLMI plans to back pioneers driving the next tech revolution.
- Maritime: DLMI seeks partnerships in maritime solutions emphasizing green initiatives.
- Aviation: DLMI’s strategy includes holdings in aviation innovators focusing on efficiency.
- Aerospace: DLMI aims to support ventures pushing boundaries in space exploration.
- Education: DLMI collaborates with platforms revolutionizing learning through tech.
- Charity: DLMI eyes alliances with charitable entities leveraging transparency via blockchain.
- Healthcare: DLMI foresees investments in healthcare tech personalizing patient care.
- TV: DLMI intends stakes in TV platforms innovating content delivery.
- Film: DLMI aspires to support filmmakers merging storytelling with immersive tech.
- Music: DLMI plans interests in music ventures amplifying artists through digital platforms.
- Entertainment: DLMI targets stakes in platforms redefining entertainment paradigms.
- IP: DLMI envisions collaborations safeguarding intellectual properties via tech solutions.
- Data Management: DLMI seeks ventures optimizing data utilization and insights.
- Data Storage: DLMI’s roadmap includes alliances with secure data storage solutions.
- Streaming: DLMI intends to back streaming platforms prioritizing user experience.
- Real World Assets: DLMI eyes investments translating tangible assets into digital value.
- Gold & Silver: DLMI aims for stakes in platforms digitizing precious metal trading.
- Sports: DLMI envisions collaborations enhancing sports experiences via tech integration.
- Sports Technology: DLMI seeks ventures revolutionizing athlete performance and fan engagement.
- Water: DLMI plans to back solutions ensuring water sustainability and accessibility.
- Water Treatment: DLMI targets investments in eco-friendly water purification technologies.
- Animation: DLMI eyes stakes in animation houses blending art with cutting-edge tech.
- Studio Production: DLMI’s vision includes support for studios transforming content creation.
- Consumer Products: DLMI seeks partnerships with brands prioritizing consumer-centric innovations.
- Collectables: DLMI envisions collaborations with platforms digitizing unique collectibles.
- Digital Assets: DLMI aims to invest in ventures maximizing the potential of digital ownership.
- Web3: DLMI aspires to back pioneers ushering in the decentralized web era.
- Identity Management: DLMI eyes solutions prioritizing user identity security in the digital space.
- Media & Journalists: DLMI seeks alliances promoting unbiased reporting and content democratization.
- Metaverse: DLMI envisions stakes in ventures crafting immersive virtual universes.
- Space Economy: DLMI targets investments in ventures monetizing space exploration.
- Modular Homes: DLMI plans interests in solutions revolutionizing home construction.
- Financial Technology: DLMI seeks partnerships modernizing financial transactions.
- Gaming: DLMI aims to back game developers enhancing user immersion.
- Travel: DLMI eyes collaborations transforming travel experiences through tech.
- Health & Wellness: DLMI’s strategy includes investments in holistic health tech solutions.
- Augmented Reality: DLMI envisions stakes in AR platforms blurring reality and digital.
- AI: DLMI seeks to support AI innovations humanizing tech interactions.
- Esports: DLMI targets investments in platforms amplifying esports experiences.
- Construction: DLMI plans to back ventures modernizing construction practices.
- Virtual Reality: DLMI intends stakes in VR platforms offering alternate realities.
- Retail Tech: DLMI envisions collaborations digitizing retail experiences.
- Biotechnology: DLMI seeks ventures pushing boundaries in biotech innovations.
Market Opportunity
According to Diamond Lake Minerals’ business plan executive summary, the market for digital securities is projected to grow from $10 billion in 2022 to $1 trillion by 2028, a CAGR of 45% for the forecast period.
The global blockchain market value is expected to grow from an estimated $3 billion in 2020 to $39.7 billion by 2025, marking a CAGR of 67.3% for the period. Valued at $2.28 billion in 2021, the Security Token Offerings market is projected to grow at a CAGR of 19%. This growth is expected to be driven by the rising adoption of tokenization and the increasing prominence of STOs, especially in North America.
In addition, the global investment management market is projected to grow from a value of $100 trillion in 2020 to $178 trillion by 2025, recording a CAGR of 7.2% over the period.
Management Team
Brian J. Esposito is CEO of Diamond Lake Minerals. As founder and CEO of Esposito Intellectual Enterprises LLC, he brings over 20 years of diverse experience in sectors like manufacturing, technology, music and real estate, and is known for his global executive networking and balance sheet optimization skills.
Michael Reynolds is President and Director of Diamond Lake Minerals. With 35 years in private finance and M&A, he has been instrumental in growing companies like Herbalife through reverse acquisition, as well as elevating JB Oxford to $120 million in revenue. His expertise in operational management and business development ensures professional solutions for clients’ business interests.
Jon Karas is DLMI’s senior transaction and investment executive. As the CEO and co-founder of Akon Legacy Ventures, he structured, negotiated and closed numerous transactions focused on innovation and social impact in smart cities, blockchain, agriculture, mining and technology. He co-founded and led multiple companies in media and entertainment and was the driving force behind the development, financing and production of a broad range of film and television content.
Advisory Board
Anthony Scaramucci, Founder and Managing Partner of SkyBridge Capital and Chairman of SALT, brings to Diamond Lake Minerals unparalleled expertise in finance, technology and business strategy. He is expected to be instrumental in shaping DLMI’s strategic direction as the company continues to redefine the future of traditional and digital securities.
Larry Namer, Founder of E! Entertainment TV and President of Metan Global, boasts a remarkable career spanning more than half a century. He is an esteemed veteran of the entertainment industry, renowned for his influential contributions to cable television, live events, music and new media. He also leads LJN Media, a consulting firm known for its cross-industry expertise in technology, business and finance.
Andrew Fromm is a seasoned CEO and consultant with a focus on music publishing. He is known for his expertise in asset sales, songwriting and artist development. His extensive network extends beyond the music industry, showcasing his versatility and authority in the field.
Brandon Fugal is the Chairman of Colliers International in Utah and a former EY Entrepreneur of the Year. He has co-founded multiple ventures, including Coldwell Banker Commercial Advisors, Cypher, Axcend and Texas Growth Fund, and he is a recognized authority in real estate and entrepreneurship.
Michael Malik Sr. is a Detroit-based entrepreneur with a $750 million net worth, known for his pivotal role in legalizing gambling and developing major casino projects across the U.S., including Detroit’s MotorCity Casino and various Native American gaming ventures. He brings to Diamond Lake Minerals a wealth of experience and a proven track record in the gaming, sporting and entertainment industries spanning over five decades.
Raul Leal is an experienced CEO in the hospitality sector, known for his visionary leadership at SH Hotels & Resorts and former role at Virgin Hotels, where he secured over $500 million in funding and revolutionized guest experiences.
Agnes Budzyn, an accomplished entrepreneur and CEO of Bluedge Ventures, brings to the company a rich history in traditional finance and blockchain technology, serving on various global boards and committees. She has been recognized by the World Economic Forum and numerous institutions for her expertise and contributions to bridging legacy finance with emerging digital asset infrastructure.
Diamond Lake Minerals Inc. (OTC: DLMI), closed Thursday's trading session at $3.38, up 4%, on 440 volume. The average volume for the last 3 months is 123,778 and the stock's 52-week low/high is $0.35/$3.50.
Recent News
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks - Diamond Lake Minerals Inc. (DLMI) Featured in Recent Article Noting Focus on Helping Investors Transition to Digital Asset Accessibility
- Diamond Lake Minerals Inc. (DLMI) Represents an Attractive Option For Those Looking To Enter The World of Digital Assets
- InvestorNewsBreaks - Diamond Lake Minerals Inc. (DLMI) Featured in Bell2Bell Podcast
SenesTech Inc. (NASDAQ: SNES)
The QualityStocks Daily Newsletter would like to spotlight SenesTech Inc. (NASDAQ: SNES).
SenesTech, Inc. (NASDAQ: SNES, "SenesTech" or the "Company"), ( www.senestech.com ) the leader in fertility control to manage animal pest populations, today announced financial results for the third quarter of 2023. "We accelerated our growth trajectory during the third quarter with strong 44% year-over-year growth in revenues driven by the initiatives we have undertaken to drive sales, improve our product quality, and expand our product options," commented Joel Fruendt , SenesTech's President and Chief Executive Officer. "We expect this growth to further accelerate as we launch our Evolve Soft Bait, the first and only soft bait developed to control pest populations using a breakthrough technology that targets the rat population where it starts, by restricting fertility through nonlethal methods. We have received pre-orders and expect to start shipping product next week." Sales growth during the third quarter was led by increases in nearly every key market segment, including agribusiness, zoos and sanctuaries, and commercial. The Company also continued to experience sequential revenue growth and an improvement of its return on marketing investment within its e-commerce platform following the successful reconfiguration of those operations earlier this year. Overall, the various initiatives led to an improvement of nearly $700,000 in its net loss and Adjusted EBITDA loss compared to the year-ago period.
SenesTech Inc. (NASDAQ: SNES) is the rodent fertility control expert and the inventor of the only EPA-registered contraceptive for male and female rats. The company’s technology provides an innovative and humane method for managing rat populations.
SenesTech is focused on developing effective solutions that are grounded in science and proven through research, all while providing value to people, communities and the environment. The company’s passion is to create a healthier world by better controlling rat pest populations. This aim is critical, as, if left unchecked, a breeding pair of rats and their descendants can produce up to 15,000 pups after just one year.
The company strives for clean cities, efficient businesses and happy households – with a product that was scientifically designed to be effective without killing rats. SenesTech is committed to the sustainable, humane treatment of animals, improving the quality of all human life and enhancing environmental stewardship through the global application of its effective solution in fertility control technology.
SenesTech is headquartered in Phoenix, Arizona.
ContraPest®
SenesTech’s first product, ContraPest®, applies revolutionary technology to a global challenge that has persisted since the Middle Ages – the proliferation of rats in urban and agricultural settings. ContraPest® targets the reproductive capabilities of Norway and roof rats. As a highly palatable liquid, the formulation promotes sustained consumption, helping to reduce fertility in both male and female rats, bringing populations down and keeping them down.
The company’s flagship offering can be used as part of integrated pest management (IPM) programs – fitting seamlessly into all IPM programs – to help reduce reproduction and magnify the success of these protocols, or as a standalone solution for customers who want to reduce or eliminate the use of lethal rodent control methods.
In multiple, independent field deployments, ContraPest was shown to reduce rat activity over 90% when added to an existing IPM program.
ContraPest® is registered federally as a General Use Product.
Delivery Systems and New Products
In July 2023, SenesTech began to distribute a new delivery system for ContraPest®, the Isolate Bait System™. This new delivery system brings to market a simple design that enables more efficient deployment, incorporates an enhanced formulation of ContraPest® that is expected to provide improved performance of the fertility control bait in the field and is paired with a new bait station that is more space-efficient and economical.
The other delivery systems available for ContraPest include the Ultimate Bait System™, a tank and tray in a larger format for use with more severe infestations, and the Elevate Bait System™, a unique delivery system that targets above ground infestations, as with roof rats.
SenesTech, as of August 2023, is also in the final stages of releasing a soft bait formulation, which provides the unique attributes of proven fertility control in an industry-familiar format demanded by big box retailers, key e-commerce channels and leading industry pest management professionals.
Market Opportunity
According to SenesTech’s figures, rats cause over $27 billion in damage to public and private infrastructure annually in the United States. Rats also destroy 20% of the global stored food supply every year by consuming or contaminating it.
Rats are known to spread at least 35 diseases, globally posing a dangerous risk to public health and safety. Not only does this age-old problem persist despite extensive campaigns to eradicate it, but multiple sources have reported that post-COVID rat populations have boomed.
Poison-based control methods sicken rats, and they typically die slowly. An animal that eats a poisoned rat may also sicken or die. The global rodenticide market is projected to be worth $1.7 billion by 2026.
In one case study, results reported by the customer showed a $5,000 investment in ContraPest® saved more than $500,000 annually in reduced labor, loss and damage.
Management Team
Joel Fruendt is SenesTech’s President and CEO. He has 15 years of executive leadership in the vector and pest control industries as Vice President and General Manager of Clarke Environmental Inc., a leading vector and pest control products and services company. He has extensive expertise in the development and manufacturing of EPA-registered chemical control products, and the commercialization and sale of those products. He received the ‘Smart Leaders’ award from Smart Business Magazine and holds a bachelor’s degree in business from Illinois Wesleyan University.
Tom Chesterman is CFO at SenesTech. He has over 20 years of experience as the CFO of public companies in the life science, tech and telecommunications industries. Most recently, he was the Vice President and Treasurer of GCI, a telecommunications company. Previous to that, he was the CFO of life science companies Bio-Rad Laboratories, Aradigm and Bionovo. He has a bachelor’s degree from Harvard University and an MBA from the University of California at Davis.
Dan Palasky is Chief Technical Officer at SenesTech. Previously he held the title of Vice President of Research & Development at PLZ Corp., a manufacturer of chemical consumer products, serving as the technical expert for its entire product portfolio. He started his career with Camie-Campbell, Inc., as a chemist in the R&D department. Mr. Palasky received his bachelor’s degree in chemical engineering from the Missouri University of Science & Technology and his MBA in Project Management from Aspen University.
SenesTech Inc. (NASDAQ: SNES), closed Thursday's trading session at $0.347, up 21.7544%, on 603,462 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $3.50/$.
Recent News
- SenesTech Inc. (NASDAQ: SNES) - SenesTech Announces Third Quarter 2023 Financial Results
- SenesTech Inc. (NASDAQ: SNES) Webcast at Lytham Fall Investor Conference Highlights Company's Innovation and Growth with New Solution for Rodent Control
- InvestorNewsBreaks - SenesTech Inc. (NASDAQ: SNES) Now Taking Orders for Evolve(TM) Soft Bait
Freight Technologies Inc. (NASDAQ: FRGT)
The QualityStocks Daily Newsletter would like to spotlight Freight Technologies Inc. (NASDAQ: FRGT).
Freight Technologies announced securing a Request for Quotation from a leading global pharmaceutical giant that awards the company 33 lanes of transport pharmaceutical goods from the United States to Canada and Mexico
The Request for Quotation extends over seven months, and Fr8App will provide uninterrupted, end-to-end logistics solutions, including secure transport, tracking, and reporting for each load
The company's dedicated team of professionals will oversee this vital operation to guarantee the smooth and efficient delivery of the pharmaceutical company's critical products
Freight Technologies (NASDAQ: FRGT) ("Fr8Tech"), a technology company developing solutions to optimize and automate the supply chain process through its custom-developed, industry-leading freight-matching platform powered by AI and machine learning, recently announced securing a Request for Quotation (RFQ) from a leading global pharmaceutical giant. The pharmaceutical conglomerate is known for its commitment to global health and life-changing innovations and has chosen Fr8App as its trusted partner for crucial logistical operations (https://ibn.fm/vyOgV).
Freight Technologies Inc. (NASDAQ: FRGT) (“Fr8Tech”) is a technology company developing solutions to optimize and automate the supply chain process, providing a platform for B2B cross-border shipping in the NAFTA region. The company’s mission is to revolutionize cross-border shipping by providing carriers with increased growth opportunities and shippers with flexibility, visibility and simplicity for the once-complex process of international over-the-road shipping.
Freight Technologies, formerly known as Hudson Capital Inc., assumed its current name and ticker symbol on May 27, 2022. Its primary operating subsidiary and its marketplace are known as Fr8App, and it conducts operations throughout North America under the names of Fr8App and/or Freight App. The company is headquartered in Houston, Texas, with multiple locations across the U.S. and Mexico.
The Fr8Tech Solutions Suite
Fr8Tech leverages artificial intelligence to provide cloud-based platforms aimed at automating the over-the-road transportation process, effectively reducing human touch points and expediting load booking times. The company’s suite of solutions includes:
- Fr8app – A B2B marketplace powered by AI and Machine Learning offering a real-time broker portal to connect shippers with qualified carriers
- Fr8Radar – A tracking solution providing shippers and carriers real-time locational data via Fr8app’s mobile solution or through integration with third-party GPS alternatives
- Fr8TMS – A transportation management system designed to help shippers manage their freight and all of the documents involved in shipping transactions, including invoices, customs documents, confirmation rates and proof of deliveries
- Fr8FMS – A fleet management system allowing transportation companies to better manage their fleets, reduce operational costs and provide better service to their customers
- Fr8Data – A data solution offering real-time dashboards and reports to shippers and carriers in an effort to increase visibility and control while supporting better business decisions
- Fr8Fleet – A platform that provides private fleet management, enabling large corporate shippers to purchase dedicated capacity secured by Fr8app in exchange for a fixed fee
Commitment to the Environment
Through its core focus on technology, Fr8Tech seeks to reduce the carbon footprint of the logistics industry. Its solutions aim to minimize empty miles for transportation firms and reduce overall paper consumption.
Fr8University
Fr8University is an educational program offering classroom and on-the-job training for Fr8Tech team members. Through the program, employees learn in-depth business fundamentals and applications along the truckload freight industry value chain.
Led by corporate educator Mario Mena, Fr8University is designed as an investment in the company’s human capital, providing an opportunity to communicate Fr8Tech’s corporate culture while accelerating operational growth.
Market Outlook
Fr8Tech’s established foothold in Mexico is key to its current efforts to promote sustainable growth in the cross-border shipping industry. Ongoing disruption in U.S.-Chinese trade relations have strengthened Mexico’s status as the largest trading partner of the U.S., with cross-border annual freight spending estimated at $385 billion according to data from the U.S. Department of Transportation. Annual domestic shipping in Mexico is estimated at $34 billion, while annual domestic shipping in the U.S. is estimated to total $732 billion.
Despite the size of this industry, fragmentation and inefficiencies prevail in the space. Thousands of legacy brokers, tens of thousands of shippers and hundreds of thousands of carriers still rely on outdated systems to arrange transport, spending hours on the phone negotiating pricing, waiting days to find trucks and drivers, preparing and printing forms, and operating without tracking or visibility. Add in cross-border complexity relating to customs and additional paperwork, and you have an industry ripe for technological disruption.
Fr8Tech’s recent revenue growth trends have highlighted the company’s efforts to capitalize on this opportunity. In 2021, Fr8Tech achieved revenues of $21.5 million, marking a year-over-year increase of 134%. The company issued revenue guidance for fiscal 2022 of $40 million in a February 9, 2022, press release, which would account for a further 86% year-over-year increase.
Management Team
Javier Selgas is CEO and a Director of Freight Technologies Inc. and Freight App Inc. He brings to the company over 15 years of experience developing technology and digital marketing strategies, including serving as Country Manager for Osigu, Spain, and as head of AJEgroup’s IT division for the Asia-Pacific region. Prior to joining Fr8Tech, Mr. Selgas founded digital marketing agency Lanzadera Online. He has also served as an IT consultant to major corporations, including Endesa and Ibermatica.
Mike Flinker is President of Fr8Tech. He has over four decades of experience in the transportation industry, with 30+ years focused on cross-border logistics. Prior to joining Fr8Tech, Mr. Flinker founded FLS Transportation, the largest cross-border logistics company in Canada. He also previously held positions with Clarke Transport Inc., Canadian Pacific and Reimer Express Inc. (a division of Roadway Express).
Paul Freudenthaler is the company’s CFO and Secretary to the company Board. He has over 30 years of financial expertise, having previously served as CFO for several leading companies across multiple countries, including Macquarie in Mexico, Old Mutual in Latin America and Ascentium Capital in the U.S. Mr. Freudenthaler’s experience include leadership roles from which he guided IPOs and M&A transactions.
Luisa Lopez is COO of Fr8Tech. She brings to the company 25+ years of management experience in logistics, supply chain, operations and customer service. Ms. Lopez previously served as a Director of Landstar, where she was responsible for commercial and client development strategies in the Mexican market. Additionally, she managed more than 2,000 transport units specialized in staff and school mobility while with Traxion in Mexico.
Freight Technologies Inc. (NASDAQ: FRGT), closed Thursday's trading session at $0.318899, up 7.7361%, on 160,184 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.252/$6.70.
Recent News
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Secures Request for Quotation with Global Pharmaceutical Conglomerate for Secure Product Transport
- InvestorNewsBreaks - Freight Technologies Inc. (NASDAQ: FRGT) CEO Shares Open Letter to Shareholders
- Freight Technologies Inc. (NASDAQ: FRGT) Taking Part in the Trucking Industry's Ongoing Paradigm Shift with Its Fr8App Platform
Electronic Servitor Publication Network Inc. (OTCQB: XESP)
The QualityStocks Daily Newsletter would like to spotlight Electronic Servitor Publication Network Inc. (OTCQB: XESP).
Electronic Servitor Publication Network (OTCQB: XESP), a digital engagement company and market disruptor, today announced that its Board of Directors authorized and approved a merger with Pointward Inc. for its channel activation and customer engagement technology. XESP aims to leverage the intellectual property ("IP") holding company's IP to further the technological expansion of XESP's business model and to solidify itself as a leader in creating meaningful digital pathways. Among other highlights, the merger also broadens XESP's capacity to serve both unregulated and highly regulated markets.
To view the full press release, visit https://ibn.fm/YyyyC
Electronic Servitor Publication Network Inc. (OTCQB: XESP) is a digital engagement company offering a managed service which provides digital activation and engagement solutions to companies that seek to optimize their growth. Its managed service is powered by a proven, proprietary technology – the Digital Engagement Engine™. This technology provides intelligent interaction management, dynamic content provisioning, and a logic-driven workflow, which creates digital experiences that accelerate an audience from awareness to action – driving growth.
Electronic Servitor Publication Network’s services are designed to drive growth for both established and developing organizations. Through the optimization of digital interactions within current and new communities, the Digital Engagement Engine™ ensures that client content is relevant, reaches the right audience, and connects with the intended person at the right time.
The company calls it ‘Growth as a Service’.
Client implementation is nearly effortless, since the solution is completely managed by the Electronic Servitor Publication Network team. This business model allows clients to focus on their brands, core product offerings, and content creation, while the company manages the technology and outcome.
The company is headquartered in Minneapolis, Minnesota.
Technology
Electronic Servitor Publication Network’s Digital Engagement Engine™ utilizes a combination of automation, unique data management, and a modern workflow built on a microservices architecture to achieve greater reach and lift. Using sophisticated data analysis and smart technology, the Digital Engagement Engine™ provides companies with the ability to maintain complete control of their content while creating meaningful relationships with new customers and revenue streams.
The Digital Engagement Engine™ isn’t just another marketing or technology tool; it’s a way to develop real connections with target markets.
Market Outlook
According to a report by ReportLinker.com, an award-winning market research firm, the global customer engagement solutions market was estimated at $19.3 billion in 2022 and is forecast to grow to $32.2 billion by 2027, achieving a CAGR of 10.8% during the forecast period.
The report notes that these engagement solutions are vital to companies seeking to widen their customer bases, reduce customer churn rates and increase customer retention. These perceived benefits of customer engagement solutions are likely to drive their growing adoption around the globe during the forecast period, according to the report.
Management Team
Peter Hager is President and CEO of Electronic Servitor. He joined the company from Pointward Inc., a medtech customer engagement agency that provided solutions to drive market entry, growth, and commercialization for Fortune 500 health care brands and medtech startups. He has founded and managed multiple technology, professional services and medtech organizations throughout his career. Mr. Hager holds a bachelor’s degree from Macalester College in St. Paul, Minnesota, with concentrations in economics and psychology.
Jim Kellogg is CFO of Electronic Servitor. He has served as the principal of J. Kellogg & Company Inc., a business and tax consultant, since 2005. He has provided legal support to clients’ business valuations, business interruption and divorce property valuations. He has worked as a professional tax adviser since 1983. Mr. Kellogg obtained his JD with emphasis on taxation from Western State University College of Law and was certified as a financial planner by the College for Financial Planning in 1990.
Thomas (Denny) Spruce, RPh, is COO of Electronic Servitor. He oversees company infrastructure, regulatory reporting, and strategic partner relationships, among other roles and responsibilities. He joined the company in March 2022 and, since that time, has implemented foundational support processes, developed contractual relationships with service providers, managed financial and regulatory reporting and overseen contract development and management with the legal team. Mr. Spruce obtained a BS in Pharmacy from the University of Arkansas.
Electronic Servitor Publication Network Inc. (XESP), closed Thursday's trading session at $0.13, up 49.6489%, on 18,507 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.03/$0.22.
Recent News
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Acquires Pointward Inc. Technologies
- InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) to Acquire Assets of PhiTech Management LLC
- InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Announces Participation at Emerging Growth Conference
First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF)
The QualityStocks Daily Newsletter would like to spotlight First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) .
Tellurium is a rare and mildly toxic element that has applications in solar cells, tinting glass, vulcanizing rubber, semiconductor applications and rewritable CDs, among others. Battery manufacturers are also developing new lithium-tellurium battery designs that replace the liquid electrolytes in EV batteries with solid electrolytes to reduce fire risks. With the metal's market predicted to grow over the next decade amid a global transition to electric cars and renewable energy, researchers are becoming increasingly interested in the silvery-white metalloid. According to research from Lithuanian and Japanese scientists, tellurium may be one of the heavy elements that form when neutron stars collide and merge. Forming from the cores of massive stars that collapse into themselves when they reach the end of their lives, neutron stars are among the smallest and densest objects in the universe. They are so dense that a "tablespoon" of material from a neutron star would weigh more than a billion tons. In the meantime, entities such as First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) remain focused on ensuring that as the demand for this heavy metal grows, there are ample supplies to address the growing market.
First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) is committed to exploring for and providing essential and critical metals, including tellurium, gold, silver, copper and tungsten, for North American markets. This objective is anchored by the company’s Deer Horn tellurium-gold-silver-copper project in British Columbia, Canada, and further enhanced by its property option on the Klondike tellurium-gold prospect located in Colorado, USA.
First Tellurium’s unique business model is to generate revenue and value through mineral discovery, project development, project generation and cooperative access to untapped mineral regions in indigenous territory with sustainable exploration potential.
The company is headquartered in Vancouver, British Columbia.
Tellurium and the Green Energy Revolution
Tellurium has a key role to play in the ongoing green energy revolution. It is widely used in the manufacturing of photovoltaic cells for solar panels.

Despite this utility, ongoing trade tensions between China and the U.S. create implications for both tellurium and the production of cadmium-tellurium solar cells. Earlier this year, China announced plans to restrict exports of critical metals gallium and germanium, both essential for the production of semiconductors. For reference, China produces around 80% of the world’s gallium and approximately 60% of the world’s germanium.
China’s recent trade restrictions amplify the fragility of the North American tellurium supply, as the Asian nation currently produces about 60% of the world’s tellurium. This sustained supply vulnerability is why First Solar, the United States’ largest solar panel producer, set up a worldwide search for tellurium deposits in the mid-2000s.
“In North America alone, our understanding is that First Solar looked at over a hundred tellurium properties,” First Tellurium CEO Tyrone Docherty stated in a news release. “Their number one property by far, which they acquired, was the Colorado Klondike which we now control.”
The U.S. is now looking to secure safe, domestic sources of tellurium and many other critical metals to pre-empt potential shortages. The Biden administration has instituted a stream of policies, particularly the U.S. Inflation Reduction Act, to source solar components from North America and other “friendly” jurisdictions.
As the only junior mining company in the world focused on tellurium exploration, First Tellurium is ahead of the curve in capitalizing on these initiatives to establish strategic, domestic supplies of key resources for solar panel manufacturers.
First Tellurium’s ESG Initiatives
Through its exploration and partnerships with Fenix Advanced Materials, Cheona Metals and IRMA, First Tellurium strives to generate a measurable, beneficial social or environmental impact alongside a financial return. The company conducts a diversified search for metals, working in alliance with indigenous peoples, NGOs, governments and leading metals buyers. First Tellurium believes this is the future of mineral exploration — generating revenue by exploring responsibly and leveraging diverse partnerships.
First Tellurium proudly adheres to, and supports, the principles and rights set out in the United Nations Declaration on the Rights of Indigenous Peoples and, in particular, the fundamental proposition of free, prior and informed consent.
Projects
Deer Horn Tellurium-Gold-Silver-Copper Project
Deer Horn is located on 51.33 square kilometers (km) in west-central British Columbia, 36 km south of the prolific Huckleberry copper-molybdenum mine and 135 km southwest of the community of Burns Lake. It is one of few significant tellurium discoveries outside Asia and includes a 2.4 km-long vein system of high-grade gold, silver and tellurium, as well as broader zones of bulk-tonnage gold, silver and tellurium mineralization. The company completed a positive Preliminary Economic Estimate and has begun permitting for a 10,000-tonne bulk sample program to advance the project toward mine feasibility. It is North America’s only silver-gold-tellurium property with an NI 43-101 compliant tellurium resource, and it hosts a number of other mineralized targets and zone containing critical metals such as copper, tungsten and zinc.
First Tellurium owns 50% of the property, with an option to acquire up to a 75% interest. The company has engaged Dias Geophysical of Saskatoon, Saskatchewan, to conduct induced polarization (IP) geophysics on the Deer Horn Project in summer 2023. The program is designed to help develop drill targets for a subsequent drilling program.
Klondike Gold-Tellurium Project
The Klondike property is located in Saguache County, Colorado, southwest of Buena Vista in the state’s historical mining district. The company reports it has engaged Burgex Mining Consultants of Sandy, Utah, to stake additional claims around the Klondike property. The claims have been filed with the Bureau of Land Management.
Klondike demonstrates exceptional tellurium grades. Tellurium, used in high-efficiency cadmium telluride (Cd-Te) solar panels, next-generation lithium-ion batteries and thermoelectric devices to change heat into energy, is an essential element for the world’s transition to green energy.
The Klondike property was a top tellurium prospect owned previously by First Solar Inc., one of the world’s largest solar panel producers. First Solar terminated its worldwide raw materials exploration program in 2012 and sold the property to Colorado Klondike LLC, which optioned the project to First Tellurium. Colorado Klondike, led by First Solar’s former Exploration Manager in North America, is managing the upcoming exploration program.
The Colorado Geological Survey (CGS), in partnership with the Colorado School of Mines, reported on First Solar’s exploration at Klondike in 2015, noting: “Surface sampling by First Solar, Inc. in 2006 found very high tellurium grades of up to 3.3% (33,000 ppm), along with locally high gold grades. Tellurium grades at Klondike were the highest encountered in the company’s nationwide exploration program.”
Market Outlook
First Tellurium in spring 2023 referenced recent forecasts by the International Energy Agency (IEA) pointing to rapid growth in solar photovoltaic (solar PV) deployment worldwide. According to the agency, solar PV installations will generate more power by 2027 than any other energy source, including coal, natural gas and hydro. To meet this demand, consumption of both silver and tellurium, key components of solar panels, is expected to surge in coming years.
Chen Lin, founder of Lin Asset Management, has written in his investment newsletter for clients that solar PV is now the largest industrial usage of silver. He said that in 2022 solar PV production used about 12% of total silver demand, or about 120 million ounces of silver. Lin expects this number to rise dramatically in the coming years, and that is likely to lead to silver supply deficits for decades to come.
Lin points out that solar power is now the cheapest source of energy in many parts of the world and that all forecasts point to dramatic expansion of solar PV in the coming two decades. Conservative estimates forecast 300 gigawatts of solar PV production by 2027, up from the current level of about 200 gigawatts.
Management Team
Tyrone Docherty is President, Director and CEO of First Tellurium Corp. He previously served as President and CEO of Quinto Mining Inc., taking over when it had a market cap of $4 million. With limited resources in a difficult market environment, he raised more than $30 million and advanced Quinto’s Quebec iron ore property to a viable project. Quinto later sold for $175 million, with Quinto management taking shares of the purchaser, Consolidated Thompson Iron Mines, amounting to approximately 20-21% of that company. Consolidated Thompson Iron Mines sold two years later for $4.9 billion, giving the former Quinto team an enterprise value of approximately $1 billion. From 2012 to 2018, Mr. Docherty was Director and Chairman of Mason Graphite Inc. He has worked in the financial and minerals markets for more than 30 years.
Tony Fogarassy, M.Sc. LL.M., is Chairman of First Tellurium Corp. He is a lawyer and a geologist. His extensive legal and technical expertise includes minerals, oil and gas, coal and renewable energy projects and environmental and aboriginal/indigenous law in North America, Africa and Asia. He graduated as gold medalist in geological sciences from the University of British Columbia and in law from the London School of Economics.
First Tellurium Corp. (OTCQB: FSTTF), closed Thursday's trading session at $0.0551, up 5.0124%, on 23,046 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.04935/$0.1765.
Recent News
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - Researchers Find Evidence of Tellurium Production When Neutron Stars Merge
- First Tellurium Provides Update on Sampling and IP Survey Work at Deer Horn Project
- First Tellurium Closes First Tranche of Private Placement
Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF)
The QualityStocks Daily Newsletter would like to spotlight Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) .
Prospera Energy (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B), a public oil and gas exploration, exploitation and development company focusing on conventional oil and gas reservoirs in western Canada, announced that it has resumed phase 2 development of its horizontal infill drilling program. The company anticipates the horizontal infill drilling transformation from vertical wells will continue through winter. By working to bring this specific horizontal production online as quickly as possible, the company can sustain momentum and capitalize on the current strong oil price environment. According to the announcement, the first five horizontal wells that have been drilled performed higher than the estimated type curve with the IP60 exceeding Prospera's expectations. The company reports that permits have been received for the development of light oil slanted wells and the lease has been constructed, with development expected to begin in the next few weeks, depending on weather. The company noted that it will continue the phase 2 development program to increase production, aiming to achieve year-end targets and enhance share value. The update also observed that, in addition to the first closing of debt financing, Prospera has received a favorable response to its nonbrokered private placement of debt financing up to $3 million, with that financing designed to accommodate joint venture partner deficit and to extend drilling.
To view the full press release, visit https://ibn.fm/13Zff
Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) is a public oil and gas exploration, exploitation and development company focusing on conventional oil and gas reservoirs in Western Canada. The company uses its experience to develop, acquire and drill assets with potential for primary and secondary recovery.
Prospera is primarily focused on optimizing hydrocarbon recovery from legacy fields through environmentally safe and efficient reservoir development methods and production practices. It is in the midst of a three-stage restructuring process aimed at prioritizing cost effective operations while appreciating production capacity and reducing liabilities.
The company is based in Calgary, Alberta, Canada.
Operations
Prospera’s core properties include more than 42,000 cumulative acres across Cuthbert, Luseland and Heart Hills in Saskatchewan and Red Earth and Pouce Coupe in Alberta. In total, the company estimates that there are half a billion barrels of oil in place at these sites accounting for 20+ years of forward project lifespan, with as little as 8% of total reserves having been recovered via legacy vertical well technology.
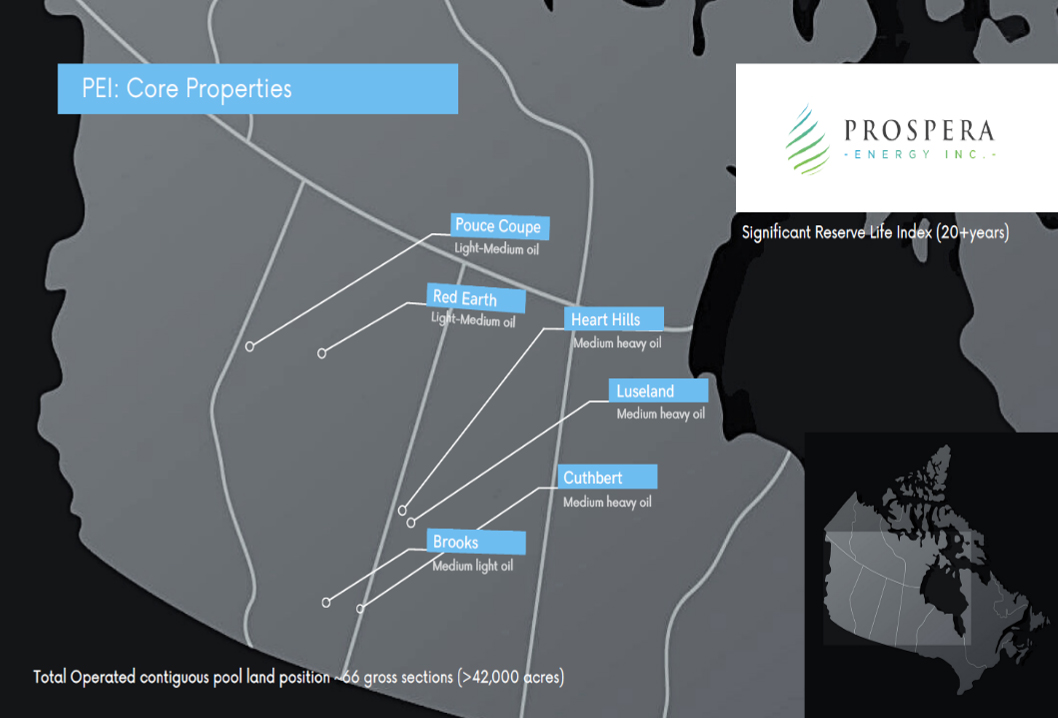
Restructuring Initiative
In 2021, Prospera enacted a top-down reorganization. The early results of these efforts were on display in May 2023, when the company reported a three-fold year-over-year increase in annual revenue for 2022 alongside drastically reduced operating costs and record-high cash flow from operations.
Prospera noted in the news release that it has positioned itself in 2023 to execute the second phase of its development plan aimed at increasing production through medium-oil development in Alberta and leveraging horizontal wells to capture the significant remaining reserves in Saskatchewan.
During the company’s investor summit in August 2023, Prospera CEO Samuel David provided more information regarding this three-phase strategy:
Phase I
Prospera completed the first phase of its restructuring by optimizing operations at its existing assets and addressing legacy arrears and non-compliance issues.
At the beginning of this transformation, the company was producing just 80 barrels of oil equivalent (BOE) per day. In Q4 of last year, Prospera peaked at nearly 1,200 BOE per day. Its breakeven is around 500 barrels per day, illustrating the opportunity for free cash flow. This prospect has driven Prospera’s capital development and optimization in recent quarters.
After a temporary slowdown in production due to harsh winter conditions, Prospera is currently producing about 800 BOE per day and anticipates an additional 300-500 barrels of daily production following the completion of ongoing site maintenance work.
This sustained increase has pushed the company’s NPV from roughly $3 million prior to the restructuring efforts to approximately $72 million today.
In an effort to build on this progress and maximize its available resources, Prospera piloted two horizontal reentries to assess a potential horizontal well transformation at its properties.
Phase II
Following up on the optimization efforts of Phase I, Prospera aims to commence a horizontal well transformation at its properties in the coming months. Based on its pilot wells from Phase I, the company has proposed 10 horizontal well locations at its Cuthbert and Heart Hills properties.
Prospera has likewise proposed eight medium light oil direction wells at its Alberta property, and it is exploring strategic acquisitions aimed at expanding its core area and diversifying its product mix.
Other facets of Phase II include piloting an enhanced oil recovery (EOR) application and continuing to execute its liability management goals and ESG initiatives. Prospera has already abandoned 60 vertical wells as part of its three-year LMR plan to reclaim surface land and reduce the environmental footprint of its operations.
Phase III
Beginning next year, Phase III of Prospera’s corporate redevelopment strategy will focus on continuing the company’s horizontal modular development to appreciate production and optimize recovery of remaining reserves. Prospera intends to implement full-scale EOR applications based on the results of its Phase II pilot program, which is forecast to optimize recovery by greater than 10%.
Prospera also intends to continue its acquisition strategy to diversify its product mix. Its goal, as detailed by in August 2023 investor summit, is to attain 50% light oil, 40% heavy oil and 10% gas – all while continuing to eliminate carbon emissions as part of its existing ESG initiatives.
Poised for Growth
Following its transformational efforts in 2022, Prospera is poised to achieve record growth in 2023. The company has forecast significant reductions in production costs through 2024, alongside sizable increases in daily production.
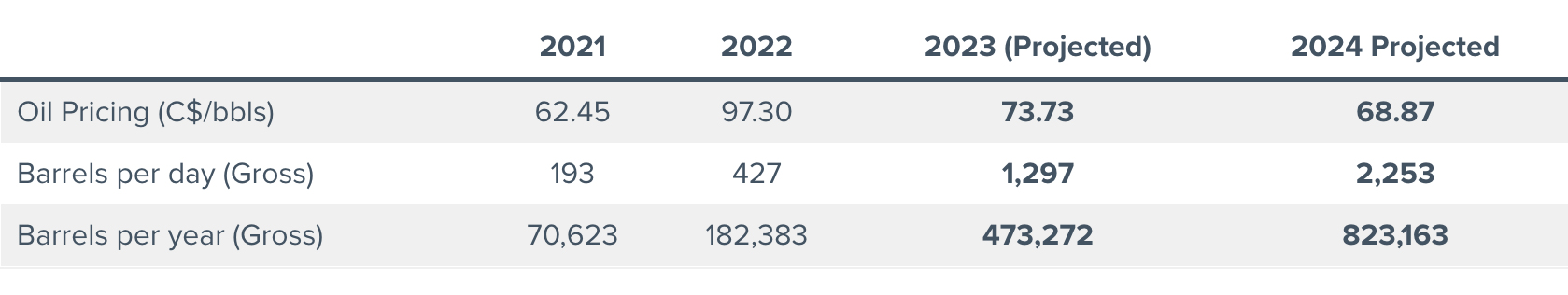
Prospera is currently exploring strategic acquisition targets to potentially increase its production beyond 5,000 BPD while expanding its reserve base to a billion barrels.
Market Opportunity
While the oil and gas industry faces long-term geopolitical and macroeconomic uncertainty, there is a clear trend to secure supply in the short term. According to Deloitte, the global upstream industry ended 2022 with some of the highest free cash flows on record, driving reinvestment in hydrocarbons and overall investment in clean energy.
The Energy Information Administration recently forecast a dip in global oil inventories over each of the next five quarters, placing upward pressure on oil prices. The agency further forecasts a YoY increase in fuel consumption, exacerbating the effects of OPEC+ production cuts that are set to remain in place through 2024.
For Prospera, these forecasts are promising. The company aims to build on its recent financial growth in the coming months (Prospera reported a three-fold YoY increase in revenue to $13.9 million in 2022), hitting a projected $57 million in total revenue by the end of 2024 while working to expand its core area holdings through accretive M&A transactions.
Leadership Team
Prospera is led by a team with extensive, diverse petroleum industry experience spanning both reservoir management and operations of oil and gas assets. The team boasts a proven track record of reorganizing companies, structuring financing arrangements and positioning for growth.
Samuel David is the company’s President and CEO. He brings to Prospera over 32 years of experience in operation, development and management of oil and gas assets and companies. Mr. David holds a B.Sc. in Mechanical Engineering and a B.A. in Economics from the University of Calgary. His background consists of both engineering and executive management experience with majors Petro-Canada, AEC Oil & Gas (now EnCana / Cenovus) and Husky Energy, as well as founding and operating juniors Ventura Energy and First West Petroleum. Mr. David has proven expertise in corporate planning, production, reservoir engineering, depletion strategies, EOR, property evaluations, acquisitions and divestitures.
George Magarian is VP Subsurface for Prospera. He is a professional petroleum geologist (APEGA) with over 36 years’ experience in the Western Canada Sedimentary Basin. After graduating with an Honors B.Sc. degree in Earth Science from the University of Waterloo, Mr. Magarian spearheaded many successful exploration programs, conducted evaluations for improved recovery schemes and assessed/exploited unconventional oil reservoir opportunities. He has held roles of increasing responsibility, from exploration geologist at oil industry major Petro-Canada and intermediates Anderson Exploration and Jordan Petroleum, to geoscience manager and VP exploration at junior companies Ionic Energy, Gentry Resources and Westfire Energy.
Chris Ludtke is the company’s VP Finance & Accounting. He is a high functioning finance leader with extensive expertise in finance, budgets and planning, accounting, economic evaluation, management, governance and sound decision making. Mr. Ludtke has 20 years of experience within the oil and gas, clean energy and renewables industries, including 12+ years working for Husky Energy before moving into an executive role in the junior oil and gas and hydrogen space. He graduated from the University of Lethbridge (Bachelor of Management) and is a Chartered Professional Accountant in the Province of Alberta.
Matthew Kenna is the CFO of Prospera. He has over 30 years’ experience leading organizations and helping them expand, drive efficiencies and grow profitability. Mr. Kenna is a professional accountant (CPA, CMA) and spent 15 years heading up the financial and operating departments at KUDU Industries, where he fostered financing arrangements, client relationships and manufacturing teams to take the organization from $35M to $150M in revenue. He has extensive experience turning companies around, growing them and building efficient organizations.
Prospera Energy Inc. (OTC: GXRFF), closed Thursday's trading session at $0.076, even for the day, on 11,090 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.059/$0.134.
Recent News
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Releases Status Update on Development Update
- InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Reports on Q3 2023 Performance, Debt Financing
- Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Provides Update on Drill Program; Announces Low-Dilutive Capital Raise
PaxMedica Inc. (NASDAQ: PXMD)
The QualityStocks Daily Newsletter would like to spotlight PaxMedica Inc. (NASDAQ: PXMD).
PaxMedica (NASDAQ: PXMD), a biopharmaceutical company focused on advancing treatments for neurological disorders, is in the spotlight during a recent episode of The Bell2Bell podcast. During the podcast, PaxMedica chief medical officer David Hough and host Stuart Smith discuss key company achievements, including the company's recent meeting with the U.S. Food and Drug Administration ("FDA"). Highlights mentioned during the podcast include the discussion with the FDA of data gathered from the company's PAX-HAT 301 study and other comprehensive nonclinical programs. Hough explained that the study provided positive evidence regarding the company's treatment for African Sleeping Sickness treatment; he also noted that the FDA acknowledged PaxMedica's comprehensive nonclinical program, including safety pharmacology and toxicology studies. Hough discussed waivers granted for the studies, affirmed data relevance to the patient population and stated that the anticipated 2024 NDA submission underscores PaxMedica's commitment to neglected tropical disease treatments. The Bell2Bell podcast is produced by IBN, a leading communications organization focused on linking public companies to the investment community.
To listen to the full podcast, visit https://ibn.fm/lA5ZR
To view the full press release, visit https://ibn.fm/hjW7L
PaxMedica Inc. (NASDAQ: PXMD) is a clinical stage biopharmaceutical company focusing on the development of novel anti-purinergic therapies (APTs) for the treatment of Autism Spectrum Disorder (ASD) and other serious conditions with intractable neurologic symptoms.
The company’s lead programs are focused on ASD, for which there are currently no approved pharmacologic treatments that target its cause and symptoms. Currently used treatments only address the symptoms of the condition, rather than targeting the pathophysiology itself.
PaxMedica is on a promising path to address these unmet medical needs, bringing hope to millions. Anti-purinergic therapies target the excess production of purines in cells. An overexpression of purines can offset homeostasis and result in an overproduction of cellular adenosine triphosphate, the main energy molecule in all living cells.
The company is headquartered in Tarrytown, New York.
Product Pipeline
PaxMedica is building a robust pipeline of products targeting ASD and related neurodevelopmental conditions. The company’s lead product in development may help eliminate, reduce or modulate some of the more troublesome aspects of ASD. That would open the potential for people with autism to integrate their behavior with others more successfully and improve their lives.
PaxMedica’s lead programs, PAX-101 and PAX-102, utilize the company’s proprietary source of suramin sodium, a broadly acting anti-purinergic therapy that has been known for over 100 years. Its current pipeline includes:
- PAX-101 (IV Suramin) for ASD – PAX-101 completed a Phase 2B study for ASD in 2021. Suramin is a broadly acting APT and has reported positive results from a dose range study. The results of PaxMedica’s Phase 2B study, which targeted 52 subjects across six sites in South Africa, were presented to AACAP in October 2021.
- PAX-102 (Intranasal Suramin) – PaxMedica has developed a proprietary intranasal formulation of suramin that is currently being evaluated in ASD and other neurodevelopmental conditions.
- PAX-101 for HAT – Given suramin’s historical use as a treatment for Human African Trypanosomiasis (HAT), or African Sleeping Sickness, the company is also developing PAX-101 as a treatment for HAT. PaxMedica’s most advanced program is the pursuit of PAX-101 for early-stage East African HAT.
- Selective APTs – PaxMedica has conducted several preclinical studies to evaluate other APTs that are more selective to specific purinergic receptors and may offer additional benefits over suramin.
Market Opportunity
According to a report by Fortune Business Insights, a leading global market research company, the global ASD therapeutics market was estimated at $1.93 billion in 2022 and is projected to grow from $2.01 billion in 2023 to $3.42 billion by 2030, a CAGR of 7.9% over the forecast period. As there is no current treatment for the core symptoms of autism, PaxMedica believes the addressable market for PAX-101, if approved, could greatly exceed these forecasts.
Autistic disorder, Asperger’s Syndrome and Pervasive Development Disorder are the three main types of ASD, affecting millions of people globally. A 2020 report by the U.S. Centers for Disease Control & Prevention estimated that one in 36 children in the U.S. have been diagnosed with autism disorder.
Several factors are expected to contribute to market growth prospects. A growing prevalence of the condition globally and rising awareness coupled with available treatment options are key factors expected to drive ASD therapeutics market growth during the forecast period. Growing investment in R&D to find effective treatments is also expected to fuel global market growth.
Management Team
Howard Weisman is Chairman and CEO of PaxMedica. He has been a founder and CEO of several specialty pharma and medical device companies. Most recently, he was executive chairman and co-founder of Sofregen, a biotech company. He also served as CEO and president of Seventh Sense Biosystems, a medical device development company. He also was founder, chairman and CEO of EKR Therapeutics, a specialty pharmaceutical company, and founder and COO of ESP Pharma, a company focused on cardio and neurovascular products. He has a bachelor’s degree in chemistry from Rutgers University.
David Hough, M.D., is Chief Medical Officer at PaxMedica. He is a neuroscience clinical development consultant who previously served as vice president at Janssen Research and Development and in various leadership roles over 17 years. Most recently, he was the compound development team leader for SPRAVATO® for treatment-resistant depression. Prior to that, he was the schizophrenia disease area leader. He played a pivotal role in the development programs for oral INVEGA®, INVEGA SUSTENNA® and XEPLION® for schizophrenia. He is a graduate of West Point and is board certified in psychiatry.
Stephen Sheldon is COO and CFO at PaxMedica. He has served as CEO of Thailand-based specialty healthcare company Indochina Healthcare Co. Ltd. since 2015. Previously, he was a consultant for PricewaterhouseCoopers Healthcare Advisory in the Chicago office. He was responsible for developing specialty pharmacy patient programs, strategy development for specialty products and compliance programs. He has an MBA from Thunderbird School of Global Management and a bachelor’s degree in computer science and visual arts from Bowdoin College.
PaxMedica Inc. (NASDAQ: PXMD), closed Thursday's trading session at $2.39, off by 30.9249%, on 1,098,568 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $2.30/$69.19.
Recent News
- PaxMedica Inc. (NASDAQ: PXMD) - InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) CMO Featured in Latest Bell2Bell Podcast Release
- Study Finds Autism Compromises Sleep Quality, Physical Activity in Kids
- InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) Phase 2 Study Results Published in Peer-Reviewed Journal
CNS Pharmaceuticals Inc. (NASDAQ: CNSP)
The QualityStocks Daily Newsletter would like to spotlight CNS Pharmaceuticals Inc. (NASDAQ: CNSP).
A team of researchers from Nottingham Trent University and the University of Sheffield is developing a first-of-its-kind finger-prick test to test for brain cancer. The research team says the simple test could make it easier to spot recurring brain tumors and even extend the lives of tens of thousands of cancer patients globally. More than 300,000 people worldwide are diagnosed with brain tumors every year, and experts predict that slightly more than 18,000 Americans will lose their lives to brain cancer and central nervous system tumors this year. In addition, even though brain cancer treatments have gotten significantly better over the decades, brain tumors often recur unpredictably after treatment. Brain tumors can return to their original location or different parts of the brain, sometimes weeks or even years after treatment, making it incredibly difficult for doctors to predict if and when their patients will experience brain tumor recurrence. This unpredictability makes it harder to detect brain cancer when it recurs early enough and significantly reduces the chances of positive treatment outcomes. As such, regular monitoring is necessary to detect brain tumors when they recur so treatment can begin as soon as possible. With this rapid detection of recurrence, novel drugs such as those being developed by CNS Pharmaceuticals Inc. (NASDAQ: CNSP) can be used to address those tumors before they spread.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP) is a clinical stage biotechnology company specializing in the development of novel treatments for primary and metastatic cancers of the brain and central nervous system.
The company was founded in 2017 and is headquartered in Houston, Texas.
Organ Targeted Therapeutics
The company’s lead drug candidate, Berubicin, is proposed for the treatment of glioblastoma multiforme (“GBM”), an aggressive and incurable form of brain cancer. Berubicin also has potential to treat other central nervous system malignancies. Based on limited clinical data, Berubicin appears to be the first anthracycline to cross the blood brain barrier in the adult brain, and it was the subject of a successful Phase 1 study which found the MDT and produced efficacy data as well.
CNS holds a worldwide exclusive license to the Berubicin chemical compound. The company has acquired all requisite data and know-how from Reata Pharmaceuticals Inc. related to a completed Phase I clinical trial of Berubicin in malignant brain tumors. In this trial, 44% of patients experienced a statistically significant improvement in clinical benefit. In 2017, CNS entered into a collaboration and asset purchase agreement with Reata.
CNS intends to explore the potential of Berubicin to treat other diseases, including pancreatic and ovarian cancers and lymphoma. The company is also examining plans to develop combination therapies that include Berubicin.
CNS estimates that more than $25 million in private capital and grants were invested in Berubicin prior to the company’s $9.8 million IPO in November 2019.
CNS intends to submit an IND for Berubicin during the fourth quarter of 2020 and expects to commence a Phase II clinical trial of Berubicin for the treatment of GBM in the U.S. in Q1 2021. A sub-licensee partner was awarded a $6 million EU/Polish National Center for Research and Development grant to undertake a Phase II trial of Berubicin in adults and a first-ever Phase I trial in pediatric GBM patients in Poland in 2021.
The company’s second drug candidate, WP1244, is a novel DNA binding agent licensed from the MD Anderson Cancer Center. In preclinical studies, WP1244 proved to be 500-times more potent than the chemotherapeutic agent, daunorubicin, in inhibiting tumor cell proliferation. The company has entered into a sponsored research agreement with the MD Anderson Cancer Center to further the development of WP1244.
CNS Pharmaceuticals recently engaged U.S.-based Pharmaceutics International Inc. and Italian BSP Pharmaceuticals SpA for the production of the Berubicin drug product. The company has implemented a dual-track manufacturing strategy to mitigate COVID-19-related risks, diversify its supply chain and provide for localized availability of Berubicin. CNS has already completed synthesis of Berubicin’s active pharmaceutical ingredient (API) and has shipped the API to both manufacturers in order to prepare an injectable form of Berubicin for clinical use.
Global Brain Tumor Therapeutics Market
The high recurrence rate of malignant brain tumors is due to reappearance of focal masses, indicating that a sub-population of tumor cells in these cancers may be insensitive to current therapies and may be responsible for reinitiating tumor growth. This necessitates the development of newer drugs in the market that demonstrate greater efficacy in treating such aggressive cancers.
A global increase in neurological disorders has placed increased attention on cancers of the brain over the past decade. Neurological disorders are becoming one of the most prevalent types of disorders, due to longer life expectancy, greater exposure to infection and an increasingly sedentary lifestyle. Because few treatments for primary and metastatic cancers of the brain exist, costs are high and have acted as a restraint for the brain tumor therapeutics market.
Despite progress in surgery, radiotherapy and chemotherapeutic strategies, effective treatments for brain cancer are limited by a lack of specific therapies for the brain and the difficulty in transporting therapeutic compounds across the blood brain barrier. Therefore, there is a significant need for novel and effective therapeutic drugs and strategies that prolong survival and improve quality of life for brain tumor patients.
Several companies are making significant investments into R&D, which is expected to bring more treatment options to the market in the near future. Industry reports consistently project continued growth in the market.
One report estimates that the global brain tumor therapeutics market will reach a valuation of $2.74 billion in 2023, with the market expected to register a CAGR of 11% during the forecast period from 2018 to 2023. Another report projects that the global brain tumor therapeutics market will reach $3.4 billion by 2025, up from $2.25 billion in 2019 (http://nnw.fm/eDUjp).
Management Team
John M. Climaco is the CEO of CNS Pharmaceuticals. For 15 years, Climaco has served in leadership roles for a variety of health care companies. Recently, Climaco served as the Executive Vice President of Perma-Fix Medical S.A, where he managed the development of a novel method to produce Technitium-99. Climaco also served as President and CEO of Axial Biotech Inc., a DNA diagnostics company. In the process of taking Axial from inception to product development to commercialization, Climaco forged strategic partnerships with Medtronic, Johnson & Johnson and Smith & Nephew.
Christopher Downs, CPA, is the company’s Chief Financial Officer. Downs previously served as Interim Chief Financial Officer and Executive Vice President of InfuSystem Holdings Inc. (NYSE: INFU), a supplier of infusion services to oncologists in the United States. Downs holds a Bachelor of Science from the United States Military Academy at West Point, an MBA from Columbia Business School and a Master of Science in Accounting from the University of Houston-Clear Lake.
Dr. Donald Picker is the Chief Scientific Officer of CNS. Picker has over 35 years of drug development experience. Prior to joining CNS, Picker worked at Johnson Matthey, where he was responsible for the development of Carboplatin, one of the world’s leading cancer drugs, which was acquired by Bristol-Myers Squibb with annual sales of over $500 million. In addition, he oversaw the development of Satraplatin and Picoplatin, third-generation platinum drugs currently in late-stage clinical development.
Sandra L. Silberman, M.D., Ph.D., is the Chief Medical Officer of CNS Pharmaceuticals. Silberman is a hematologist/oncologist who earned her B.A., Sc.M. and Ph.D. from the Johns Hopkins University School of Arts and Sciences, School of Public Health and School of Medicine, respectively, and her M.D. from Cornell University Medical College. She then completed both a clinical fellowship in hematology/oncology and a research fellowship in tumor immunology at the Brigham & Women’s Hospital and the Dana Farber Cancer Institute in Boston, Massachusetts. Silberman has played key roles in the development of many drugs, including Gleevec(TM), for which she led the global clinical development at Novartis. Silberman advanced several original, proprietary compounds into Phases I through III during her work with leading biopharmaceutical companies, including Bristol-Myers Squibb, AstraZeneca, Imclone and Roche.
CNS Pharmaceuticals Inc. (NASDAQ: CNSP), closed Thursday's trading session at $1.46, off by 9.8765%, on 38,542 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $0.6105/$6.597.
Recent News
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Brain Cancer Reemergence Could Soon Be Detected Using a Finger-Prick Test
- Stanford Study Finds Lung Cancer Cells Hide, Thrive in the Brain
- Spanish Clinical Trial to Explore Marijuana as Glioblastoma Treatment
HeartBeam Inc. (NASDAQ: BEAT)
The QualityStocks Daily Newsletter would like to spotlight HeartBeam Inc. (NASDAQ: BEAT) .
HeartBeam (NASDAQ: BEAT), a cardiac technology company that has developed the first and only credit card-size 3D-vector electrocardiogram ("VECG") platform for patient use at home, today announced the expansion of its scientific advisory board ("SAB") to include several leading cardiologists. New members include Charles L. Brown III, MD, CEO of the Piedmont Healthcare Physician Enterprise, Tony Das, MD, a nationally recognized board-certified interventional cardiology specialist, leader and educator in the field of cardiovascular diseases, Robert Harrington, MD, dean of Weill Cornell Medicine and provost for medical affairs of Cornell University, Campbell Rogers, MD, EVP and chief medical officer at HeartFlow Inc., and Niraj Varma, MD, Ph.D., professor of medicine and consultant electrophysiologist at the Cleveland Clinic. "We are honored and pleased to have such a distinguished group of physicians joining our scientific advisory board," said Branislav Vajdic, Ph.D., CEO and founder of HeartBeam. "HeartBeam will benefit tremendously from the input of these leaders in interventional cardiology, electrophysiology, clinical research, and new technologies. We look forward to working closely with all the SAB members as we advance our product development, clinical and commercial efforts."
To view the full press release, visit https://ibn.fm/TAGsh
HeartBeam Inc. (NASDAQ: BEAT) is a cardiac technology company that has developed the first and only 3D-vector 12-lead electrocardiogram (ECG) platform for heart attack detection anytime, anywhere. The company’s proprietary ECG telehealth technology aims to redefine the way high risk cardiovascular patients are diagnosed in ambulatory and acute care settings. HeartBeam’s initial focus is on providing diagnostic data to help physicians with care management of patients with cardiovascular disease.
In August 2022, HeartBeam announced that it submitted its HeartBeam AIMI™ software for approval from the U.S. Food and Drug Administration (FDA). HeartBeam AIMI is a platform technology to improve the speed and accuracy of heart attack detection in acute care settings. The company expects FDA approval by the end of 2022, and a full commercial roll-out of HeartBeam AIMI is targeted for Q1 2023.
HeartBeam sees submission of its first product based on its platform technology as an important milestone toward commercialization, which underscores the company’s continued progress toward making the HeartBeam AIMI platform widely available to help emergency department physicians quickly and accurately identify a heart attack.
While the FDA conducts its regulatory review, HeartBeam will focus on executing key components of its commercialization plan and subscription revenue model. It will also continue to engage in discussions with strategic institutions, including academic centers, regional healthcare systems and regional community hospital systems that can utilize HeartBeam products.
The company is based in Santa Clara, California.
Products
HeartBeam’s development portfolio includes two products:
- HeartBeam AIMI is software that provides a 3D comparison of baseline and symptomatic 12-lead ECG to more accurately identify a heart attack in acute care settings and, as noted above, has been submitted for FDA approval; and
- HeartBeam AIMIGo™, the first and only credit card-sized 12-lead output ECG device coupled with a smartphone app and cloud-based diagnostic software system for remote heart attack detection.
HeartBeam is developing AIMIGo, a medical-grade detection and monitoring technology for use in remote heart attack detection, thereby allowing physicians to diagnose a patient’s heart attack as it occurs, even if the patient is not at a medical facility. The company’s system, once approved by the FDA, can be used by patients at home or almost anywhere and anytime to help their physicians assess whether chest pain is the result of a heart attack or another cause. While approximately 82% of chest pain ED visits are unnecessary, patients delay approximately 3 to 4 hours after symptoms begin, increasing mortality rates by 40%. The company’s goal is to shorten the time to treatment outside of the medical facility to improve patients’ well-being.
HeartBeam’s AIMIGo is a powerful, portable and easy-to-use prescription-based product. It comprises a smartphone app, a credit card-sized ECG device placed on a patient’s chest, the HeartBeam cloud platform, and a digital portal for the physician to view ECG results and direct patient action. For the first time outside of a medical setting, HeartBeam AIMIGo enables patients and their clinicians to determine if symptoms are due to a heart attack, quickly and easily, so care can be expedited, if needed.
Pending FDA clearance, AIMIGo is initially intended to be available by prescription, and is reimbursable under existing remote patient monitoring codes (RPM codes). This provides a new revenue stream to physicians who before did not have a way to monitor these high-risk patients. The RPM codes provide a monthly reoccurring revenue stream to the company, as well. On average, at current reimbursement rates, the practice will receive $1,300+ per year per patient they monitor, and the company will receive $600 per year per patient from this RPM reimbursement.
Market Overview
Adoption rates of telehealth services increased dramatically in recent years, with the COVID-19 pandemic serving as a major driver of growth. Among the areas seeing the greatest expansion are cardiology, radiology, behavioral health and online consultation.
Encouraging this growth, governments are actively developing new policies and reimbursement guidelines to promote the use of digital health platforms. The U.S. Centers for Medicare & Medicaid Services (CMS), for example, has recently expanded reimbursement for telehealth services. U.S. market growth is also being driven by the rising prevalence of chronic conditions and the growing geriatric population.
Remote heart attack detection is a previously unsolved problem with a massive and underserved market that is several times larger than the $2 billion total addressable market (TAM) in the U.S. for ECG cardiac arrhythmia monitoring.
Approximately 8 million Americans have suffered at least one heart attack, and a total of 18 million have been diagnosed with coronary artery disease (CAD). Based on these figures, HeartBeam projects a total addressable U.S. market TAM valued at $10 billion annually for its AIMIGo solution for remote heart attack monitoring of CAD.
Management Team
Branislav Vajdic, Ph.D., Chief Executive Officer and Founder of HeartBeam, Inc, combines over 30 years of experience in technology development and senior management positions. Dr. Vajdic has been deeply involved with the development of HeartBeam’s technology to fit his vision for the company. Prior to HeartBeam, from 2007 to 2010, Dr. Vajdic was CEO and Founder of NewCardio, a publicly traded company in the cardiovascular devices space. From 1984 to 2007, Dr. Vajdic was at Intel, where he held various senior management position. At Intel, Dr. Vajdic was the designer of first Flash memory and two key inventions that enabled Flash as a product and led engineering groups responsible for Pentium 1 through Pentium 4 designs. Dr. Vajdic was awarded two Intel Achievement Awards, the highest level of award for outstanding contributions to Intel. Dr. Vajdic is author of numerous patents and publications in the fields of cardiovascular devices, as well as chip design. Dr. Vajdic holds a Ph.D. in Electrical Engineering from the University of Minnesota.
Jon Hunt, Ph.D., has over 35 years’ experience in the medical/medical device industry with extensive domestic and international experience in general management, clinical/regulatory, sales and marketing. He also has diverse experience in Fortune 500 companies, as well as start-up environments. Dr. Hunt was the Vice President of Clinical Science and Technology, Medical Device Innovation Consortium, from July 2019 to July 2021, and Vice President of Clinical and Regulatory Affairs, Cryterion Medical from January 2018 to June 2019 (acquired by Boston Scientific Corporation in July 2018 for $202M). Dr. Hunt was the Founding President and CEO of Bardy Diagnostics, Inc. from October 2013 to November 2017 (acquired by Hill-Rom Holdings, Inc.). Prior to joining Bardy Diagnostics, Dr. Hunt spent the previous 11 years as the Vice President of Clinical & Regulatory Affairs with Cameron Health, Inc. (acquired by Boston Scientific Corporation). Dr. Hunt spent the previous 10 years with Cardiac Pacemakers, Inc., St. Jude Medical and Cardiac Pathways Corporation. Dr. Hunt began his career with Cardiac Pacemakers, Inc. (now Boston Scientific Corporation) as the Director of Clinical Programs. He subsequently held positions at St. Jude Medical in Clinical Affairs and as the Business Unit Director for the Cardiac Rhythm Management division for Europe, the Middle East and Africa. At Cardiac Pathways Corporation, Dr. Hunt held various executive positions as Vice President of International Sales and Marketing and Vice President of Worldwide Sales and Marketing (acquired by Boston Scientific Corporation). Dr. Hunt received his Ph.D. in Motor Control from The Pennsylvania State University, his Master’s from California State University, Long Beach and his undergraduate degree from Keele University in the United Kingdom.
Rick Brounstein, HeartBeam’s Chief Financial Officer, combines over 30 years of experience in health technology senior management. Since 2017, Mr. Brounstein has been and is currently a partner of Hardesty, LLC, a financial services firm, and Mr. Brounstein is currently a managing director of CTRLCFO, LLC, a firm Mr. Brounstein founded in 2016 to support funded start-ups in life science and technology. From 2008 to 2011, Mr. Brounstein was Chief Financial Officer of NewCardio, Inc., a microcap public company in the cardiology space, and, over his career, he has been with nine other companies in life science or technology, holding positions including Chief Financial Officer, Chief Operating Officer, Treasurer and Accounting Manager. From June 2001 through November 2007, Mr. Brounstein held several positions at Calypte Biomedical Corporation, a publicly traded medical device company, including Chief Financial Officer and Executive Vice President. In January 2007, Mr. Brounstein was appointed as the National Member Representative for the 2007 COSO Monitoring Project, which published new guidelines for monitoring internal financial controls in February 2009; Mr. Brounstein subsequently was a member of the FEI task force that issued the updated COSO Internal Control Framework in 2013. In March 2005, Mr. Brounstein was appointed to the SEC Advisory Committee on Smaller Public Companies. Mr. Brounstein earned his Certified Public Accountant (CPA) certification while working at Arthur Andersen LLP, formerly a public accounting firm. Mr. Brounstein holds a B.A. in accounting and an M.B.A. in finance, both from Michigan State University.
Ken Persen, HeartBeam’s Chief Technology Officer, combines over 28 years of experience in the medical device and digital health industries in engineering and senior management positions. Mr. Persen has been involved in several companies in Cardiac Rhythm Management, holding positions including Chief Executive Officer, Chief Technology Officer, Executive Vice President and Director of Engineering. Since 2016 and prior to joining HeartBeam, Mr. Persen was the Chief Technology Officer at LIVMOR, Inc., a digital health company. In addition, from 2016 through November 2021, he was also Chief Executive Officer of LIVMOR. Prior roles included Director of Engineering at Cameron Health (acquired by Boston Scientific), a late-stage medical device start up, and engineering and management positions at Guidant Corp. (acquired by Boston Scientific), a large medical device manufacturer. He has an undergraduate degree from University of Minnesota, Duluth, with a BA in Computer Science.
HeartBeam Inc. (NASDAQ: BEAT), closed Thursday's trading session at $1.24, off by 0.8%, on 31,803 volume. The average volume for the last 3 months is and the stock's 52-week low/high is $1.11/$6.00.
Recent News
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Expands Scientific Advisory Board as It Advances Product Development, Clinical and Commercial Efforts
- InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Granted 3rd US Patent on Novel VECG Platform
- InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) to Host Third Quarter 2023 Results Conference Call
Lucy Scientific Discovery Inc. (NASDAQ: LSDI)
The QualityStocks Daily Newsletter would like to spotlight Lucy Scientific Discovery Inc. (NASDAQ: LSDI).
Psychedelic-assisted therapy has the potential to revolutionize mental-health treatments over the next couple of decades. This is an entirely new mode of treatment that involves putting patients under the influence of various hallucinogens to trigger psychedelic experiences, followed by talk therapy. A recent study has found that the combination of psychedelics and psychotherapy can induce rapid changes in the brain, potentially resulting in the treatment's high efficacy. As psychedelic research is still a relatively new field, scientists are constantly studying hallucinogens such as psilocybin (magic mushrooms), LSD and ayahuasca to try and understand how they deliver their benefits at a biological and molecular level. Research has found that psychedelic-assisted therapy can be incredibly effective against a myriad of mental-health disorders, offering long-term benefits at relatively low doses and with barely any side effects. The recent study found that the combination of psychedelics and psychotherapy can induce rapid changes in the brain, potentially resulting in the treatment's high efficacy. Human brains have an ability called neuroplasticity that allows them to change in response to intrinsic and extrinsic stimuli, albeit slowly and over a long time. Learning new skills, such as foreign languages or sports, as well as recovering from a stroke, are all activities that trigger the brain's inherent changing ability. The therapeutic formulations that are being developed by numerous companies such as Lucy Scientific Discovery Inc. (NASDAQ: LSDI) all seek to leverage this ability of psychedelics to trigger rapid beneficial changes in the brain.
Lucy Scientific Discovery Inc. (NASDAQ: LSDI) is an early-stage psychotropics manufacturing company focused on becoming the premier contract research, development and manufacturing organization for the emerging psychotropics-based medicines industry.
The company holds a Controlled Drugs and Substances Dealer’s License granted by Health Canada’s Office of Controlled Substances. This specialized license authorizes LSDI to develop, sell, deliver and manufacture pharmaceutical-grade active pharmaceutical ingredients (APIs) used in controlled substances and their raw material precursors. Lucy Scientific Discovery and its wholly owned subsidiary, LSDI Manufacturing Inc., operate under Part J of the Food and Drug Regulations promulgated under the Food and Drugs Act (Canada).
The company’s mission is to make its products and research services available for the development of medicines and experimental therapies to address certain psychiatric health disorders and other medical needs, including various mental health and addiction disorders. LSDI targets customers that include an increasing number of the leading universities, hospitals and other public, private and government institutions throughout the world that have launched research programs aimed at understanding the therapeutic potential of a range of psychedelic substances.
The company is headquartered in Victoria, British Columbia, Canada.
Products
LSDI produces a variety of high-quality natural, synthetic and biosynthetic products to meet the needs of the rapidly growing psychotropics-based medicines market. The company believes the emerging psychotropics industry will pave the way to a brighter future in mental health and overall wellness. LSDI is dedicated to advancing the frontiers of mind science and facilitating the development of psychotropic and psychedelic treatment therapies.
In Canada, the psychedelic compounds that LSDI is approved to produce under its Dealer’s License are regulated under the Controlled Drugs and Substances Act, or CDSA. Those compounds include:
- Psilocybin
- Psilocin
- Lysergic acid diethylamide, or LSD
- N,N-Dimethyltryptamine, or N,N-DMT
- 3,4-Methylenedioxymethamphetamine, or MDMA
- 4-Bromo-2,5-Dimethoxybenzeneethanamine, or 2C-B
The company also sells its consumer psychotropic products directly online and through retailers. Those products, described as microdose mushroom formulations, include a sleep aid, Twilight, and a mindfulness enhancer, Mindful.
Market Opportunity
According to a report from Global Market Insights, the psychotropics drug market had an estimated value of $20.2 billion in 2022 and is projected to reach a value of nearly $37.6 billion by 2032. That represents a CAGR of 6.4% for the forecast period. Factors driving market growth include the increasing prevalence of mental disorders, technological advancements in drug development, a rising geriatric population and increasing healthcare expenditures, the report states.
A growing awareness of mental health issues and an effort to reduce the stigma surrounding psychiatric disorders have encouraged more individuals to seek help, which in turn boosts the market. In addition, advancements in neuroscience, pharmacology and drug development have led to the discovery of new and more effective central nervous system therapeutics.
Innovative treatments offering better outcomes with fewer side effects attract patients and healthcare providers, also driving market growth.
Management Team
LSDI’s executive team brings deep experience in the development and commercialization of products featuring controlled substances, as well as the navigation of regulatory structures applicable to these products.
Richard Nanula is Chairman and CEO of LSDI. He has more than 35 years of leadership experience at several of the largest companies in the world, having been a senior executive at The Walt Disney Company, Amgen, Colony Capital and Starwood Hotels and Resorts. He has also served as a board member for Boeing Corporation and Starwood Capital, where he provided corporate guidance and oversight. He holds an MBA from Harvard Business School.
Assad J. Kazeminy, Ph.D., is Chief Scientific Officer at LSDI. He previously served as CEO of Irvine Pharmaceutical Services Inc. and Avrio Biopharmaceutical LLC, and he has founded several drug development companies. He has over 30 years of research and development experience in the biopharmaceutical industry. He received his Ph.D. in Pharmaceutical Science and Biochemistry from Esfahan University in Iran. He completed a Post Doctorate course of study at the University of Southern California Medical School, Department of Pharmacology.
Brian Zasitko, CPA, CA, is the company’s CFO. He is a Director of Invictus Accounting Group LLP, a professional services firm providing finance, advisory and accounting services. He also serves as CFO of Lobe Sciences Ltd., a company developing psychedelic compounds as therapeutics for the treatment of mild traumatic brain injuries and post-traumatic stress disorder. He has an undergraduate degree from Simon Fraser University and a CPA (CA) from Certified Professional Accountants, British Columbia.
Lucy Scientific Discovery Inc. (NASDAQ: LSDI), closed Thursday's trading session at $0.255, off by 0.507218%, on 107,989 volume. The average volume for the last 3 months is 226,981 and the stock's 52-week low/high is $0.2312/$4.00.
Recent News
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- DEA Announces Major Increase to 2023 Production Quotas for Psilocybin, THC and Ibogaine
- Study Suggests Methamphetamine More Helpful for Social Bonding Compared to MDMA
Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF)
The QualityStocks Daily Newsletter would like to spotlight Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF).
Appia Rare Earths & Uranium (CSE: API) (OTCQX: APAAF) (FSE: A0I0), a Canadian company in the rare earth element and uranium sectors, is reporting on the outstanding assay results from the latest 39 drill holes during its reverse circulation ("RC") drilling campaign at the PCH Ionic Clay Project in Goiás State, Brazil. The 39 holes are part of a comprehensive 147-hole drilling program. According to the report, the total weighted average across 57 RC drill holes reported to date is 2,287 parts per million ("PPM") or 0.23% total rare earth oxides ("TREO") with the project continuing to exhibit impressive distribution, width and grades, underscoring its significant potential. The report indicated that the highest-grade intercepts were PCH-RC-051 from 0 to 11 meters End of Hole ("EOH") and PCH-RC-063 from 0 to 24 meters EOH. Company officials noted that Appia has doubled the average depth of the mineralized zones to more than 13 meters, with a total weighted average of 2,287 PPM TREO across all reported drill holes each with significant magnet elements present; the report noted that the potential of Target IV in terms of distribution, grade and thickness of mineralization is well beyond Appia's initial expectations. "It is very encouraging for Appia to see more high-grade results occurring within the Target IV area," said Appia Rare Earths & Uranium CEO Tom Drivas in the press release. "Appia will be working with SGS Geological Services to complete a comprehensive NI 43-101 technical report and Mineral Resource Estimate (‘MRE') on Target IV over the coming months, capitalizing on the promising results we are getting from our recent drilling."
To view the full press release, visit https://ibn.fm/VL8FC
Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) is a mineral exploration company focused on exploration activities at its newly acquired Cachoeirinha rare earths project (“PCH Project”) in Brazil, as well as delineating high-grade critical rare earth elements (REE) and gallium at its Alces Lake property in Saskatchewan. Other properties in Appia’s portfolio include its Elliot Lake Property in Ontario’s historic mining camp, with a large NI 43-101 uranium and rare earths resource. Fully funded with over $5 million (CDN) in cash, no debt, aggressive exploration currently underway, and experienced management, Appia is progressing rapidly on multiple fronts in highly desired market sectors.
The company is headquartered in Toronto, Canada.
Projects
PCH Project-Brazil
The PCH project hosts REE mineralization in both ionic clays developed from the weathering of alkaline granites and in-situ rare earth mineralization associated with the underlying granite and a carbonatite intrusion to depths greater than 100 meters. Sampling data shows enrichment in rare earth minerals to depths of between eight meters and +30 meters.
In early 2023, Appia announced a definitive agreement to acquire a 70% interest in the PCH Project, which is 17,551 hectares in size and located in the Tocantins Structural Province of the Brasília Fold Belt, Goiás State, Brazil. It is classified as an alkaline intrusive rock occurrence with the potential for highly anomalous REE and Niobium mineralization.
The region around Iporá, a city located roughly 30 km from the PCH Project, has significant mineral exploration and mining activity and well-developed infrastructure.
In July 2023, Appia commenced an aggressive auger and reverse circulation (RC) drill campaign to delineate a potential resource estimate at the PCH project. Initial results at the site revealed significant exploration potential with impressive values that often surpass known ionic clay deposits in Brazil, particularly for the highly valuable heavy rare earths Terbium and Dysprosium.
The auger holes drilled at Target 4 have exhibited a range of total REE grades, ranging from 274 ppm to 16,648 ppm (1.66%), with an average of 1,291 ppm total REE. The valuable rare earths used in magnet applications – praseodymium, neodymium, terbium and dysprosium (Pr, Nd, Tb, and Dy) plus yttrium (Y) accounted for approximately 14% of total rare earths, reaching a maximum of 28.4%. Notably, the deposit also contains anomalous values of niobium and scandium, with average values of 736 ppm for Nb and 62 ppm for scandium in a composite sample from Target 4.
Heavy rare earths (HREEs) show maximum values of 1,624 ppm and average values of 1,291 ppm, primarily as terbium and dysprosium. Light rare earths (LREEs) show maximum values of 14,024 ppm (1.54%) with an average of 1,145 ppm. Neodymium and praseodymium, the main magnetic light rare earths, show respective maximum values of 3,131 ppm (Nd) and 885 ppm (Pr) and average values of 216 ppm (Nd) and 61.7 ppm (Pr). The overall HRRE/LREE ratio has a maximum of 39.5% and an average value of 16.67%.
“Appia is thrilled with the progress made and the promising results thus far,” CEO Tom Drivas stated in a news release. “The company remains committed to advancing its exploration plans, aiming to promptly gather significant data throughout the year, and to work towards estimating a maiden mineral resource in the coming months.”
Alces Lake Project – Saskatchewan
Appia’s Alces Lake project, located in northern Saskatchewan, encompasses some of the highest-grade total and critical REEs and gallium mineralization in the world, hosted within several surface and near-surface monazite occurrences that remain open at depth and along strike.
Following the company’s acquisition of additional new mineral claims in the area in February 2023, Appia’s Alces Lake claim block now totals 38,522 contiguous hectares (95,191 acres) – 100% owned by the Company.
Appia announced the completion of a NI43-101 technical report on the property in June 2023, providing an update on exploration previously reported in March 2021.The report is available on SEDAR under the company’s profile.
Extensive diamond drilling and geophysics surveys are underway to explore a more than 25-kilometer structural corridor. In July 2023, the company issued an update on its diamond drill program having completed the first phase of drilling at the project’s Magnet Ridge Zone to further test the extent of the mineralization to the south south-east (SSE). President Stephen Burega noted the presence of “continued mineralization at significantly thicker intercepts.”
As part of its 2023 exploration program at Alces Lake, Appia plans to target priority areas that extend SSE from the Wilson, Richard, Charles, Bell, Ivan, Dylan, Dante and AMP zones through the Magnet Ridge Zone and beyond, covering an area extending approximately 20 kilometers in length and 5 to 7 km in width. Appia will also undertake reconnaissance drilling on priority regional geological and geophysical targets in the Western Anomaly area.
Other Projects
- Appia holds a total of 75,314 hectares (186,106 acres) of land on four uranium claim blocks in the prolific Athabasca Basin (Loranger, North Wollaston, Eastside and Otherside). Exploration plans for these properties are expected to be announced once permits are in hand.
- Appia also has a 100% interest in 12,545 hectares (31,000 acres), with rare earth element and uranium deposits over five mineralized zones, in the Elliot Lake Camp, Ontario.
Market Opportunity
A report from Mordor Intelligence forecasts the global REE market is expected to grow from 168 million tons in 2023 to 206.25 million tons by 2028, marking a CAGR of 4.19% during the forecast period. The market is gradually improving following the economic and production restrictions of the COVID-19 pandemic.
Factors driving the market’s growth include high demand from emerging economies and the dependency of environmentally friendly technologies on rare earth elements.
According to UxC, one of the nuclear industry’s leading market research and analysis companies, the uranium market is rapidly becoming production-driven, where spot and long-term prices more closely correlate to the marginal cost of uranium production.
Although global reactor requirements are projected to be flat through 2024, UxC forecasts that significant demand growth from 2025 to 2040 will necessitate new production as resources are exhausted at several uranium projects. In addition, a large percentage of production exists in regions of the world with high geopolitical risk, which makes the market vulnerable to future disruptions and price volatility.
Management Team
Tom Drivas is CEO of Appia Rare Earths & Uranium Corp. He is an entrepreneur with over 30 years of experience in various industries, including over 20 years in the mineral resource industry. He is also currently a director of Romios Gold Resources Inc., a publicly traded company he founded in 1995.
Stephen Burega is President of Appia. He brings 16 years of management and operations experience in the mining and natural resources sectors. His extensive emerging markets background, along with a deep understanding of stakeholder management, social development and structured community engagement, position him well to lead Appia’s First Nations community engagements. He is also President and CEO of Romios Gold Resources which is focused on base and precious metal exploration in North America.
Frank van de Water is the company’s CFO. He holds CPA and CA designations and has been involved with international mining, metals and resource companies in North America, Latin America, Europe and Africa for more than 40 years.
Dr. Irvine R. Annesley, Ph.D., is VP Exploration at Appia. He is a licensed geoscientist (P.GEO.) and Professor in Economic (Mining and Mineral Exploration) Geology at École Nationale Supérieure de Géologie in France and an Adjunct Professor in Geology at the University of Saskatchewan. He has over 35 years of global exploration and applied research experience in uranium, gold and base metals exploration, most recently with Athabasca uranium explorer JNR Resources Inc.
Don Hains, P.Geo., is the company’s Consulting Geologist and Qualified Person Consulting Industrial Minerals Expert.
Antonio Vitor is Appia’s Country Manager, Brazil. He has a track record as a portfolio manager and board member. He has held multiple significant positions, including Territory Manager at Shell, as well as Senior Project Planning and Consulting roles at PwC and Petrobras.
Jack Lifton is the company’s Senior Technical Advisor and Consultant. He is an author and lecturer on the market fundamentals of technology metals.
Appia Rare Earths & Uranium Corp. (OTCQX: APAAF), closed Thursday's trading session at $0.151, off by 8.6288%, on 103,267 volume. The average volume for the last 3 months is 173,885 and the stock's 52-week low/high is $0.075/$0.35685.
Recent News
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Releases New Drilling Results from PCH Ionic Clay Project
- InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Announces Significant Milestone for Its PCH Project in Brazil
- InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Announces Exceptional Assay Drill Results from Its PCH Ionic Clay Project
SOHM Inc. (OTC: SHMN)
The QualityStocks Daily Newsletter would like to spotlight SOHM Inc. (OTC: SHMN).
SOHM (OTC: SHMN), a global generic drug innovator, recently completed the acquisition of a disruptive stem cell technology ABBIE (A Binding Based Integrating Enzyme) and patents from CGA Intellectual Holdings Inc. "[The acquisition] will allow the company to create regenerative medicine and cosmeceutical products for commercialization during the next year. SOHM's acquisition of ABBIE grants it the ability to edit genes of a large number of cell types at different stages in their life cycles, overcoming the limitations of current cell editing and cell engineering technologies, according to the company… SOHM intends to use ABBIE to edit the genes of stem cells that are recognized as the body's foundational building blocks with the ability to change themselves into various function-specific genes. The edited genes would then be inserted into patients' bodies using non-viral vectors to treat damaged organs in conditions such as ischemic heart disease or heart failure… SOHM expects to be able generating revenues immediately through licensing and gene-editing kits that are expected to be available in the third or fourth quarter of next year," a recent article explains. "ABBIE will soon become a reality and make a difference in the lives of millions of people who suffer from heart disease," SOHM COO David Aguilar, PhD, is quoted as saying.
To view the full article, visit https://ibn.fm/cBQMO
SOHM Inc. (OTC: SHMN) is a generic pharmaceutical manufacturing and marketing company with a vision of “Globalè Prospèro” (Global Prosperity). SOHM was founded in 1998 and is headquartered in Chino Hills, California.
The company’s primary goal is to create and produce cutting-edge generic medications that span a wide range of treatment areas, all while ensuring top-tier quality and keeping prices affordable. SOHM is dedicated to fully complying with all relevant regulatory prerequisites and upholding the most rigorous industry benchmarks, including the guidelines set forth by WHO-CGMP and USFDA.
Achievements and Milestones
SOHM is a recognized generic pharmaceutical manufacturer, with production and marketing of generic drugs covering all major treatment categories. SOHM also markets innovative formulations and packaging for various therapeutic segments, such as cosmeceuticals, nutraceuticals and OTC oral dosage formulations, with operations spanning India, the Philippines, Uganda, the U.S., the UK and the EU.
SOHM successfully launched a unique and innovative Salic-2 face wash, FōHM by SOHM, during the Oscar after party in Hollywood. The innovative Salic-2 offering in translucent gel form is marketed as an acne medication in the U.S. cosmeceutical market.
With proficiency in both manufacturing and marketing, SOHM stands out. The company holds licenses for producing over 300 products and has established distribution partnerships with firms in the United States, the Philippines and Uganda. Additionally, SOHM’s repertoire includes the launch of an innovative protein supplement, I-Prolec, featuring a distinct composition—a first-of-its-kind in India.
In 2012, SOHM gained recognition as “the most emerging company in the recent past” at the National Integrated Medical Association Conference. The company’s growth was underscored by its inclusion in the roster of ‘Fastest Growing Public Companies’ according to the Orange County Business Journal.
SOHM Today
SOHM brings all of its expertise and market knowledge toward a new vision. The company continues to develop, manufacture and market generic pharmaceutical drugs for various treatment categories. It offers its products in various dosage forms, including tablets and capsules, creams and topicals, ointments and liquids. The company also provides anti-arthritic/analgesics, dermatological drugs, gastrointestinal and respiratory drugs, biotechnology products, anesthetics, immunosuppressive agents and other various treatments. In addition, it offers a skincare line that includes dry dermatoses, mixed skin infection, acne vulgaris and seborrheic dermatitis products.
SOHM markets its products directly and through partner alliance agreements to drug wholesalers, mass merchandisers, chain drug stores and mail-order pharmacies primarily in the U.S. and has previously done business in the Far East, Africa and Southeast Asia. The company is working with its alliance partner in the African continent and Latin American countries.
SOHM has developed a comprehensive marketing strategy encompassing a diverse range of tactics to promote all products. SOHM uses the power of digital marketing channels, social media campaigns and targeted advertising to significantly enhance awareness and recognition of product offerings.
All distribution networks are strengthened through valuable partnerships. SOHM has gained access to the extensive U.S. market through a strategic alliance with different wholesalers catering to C-stores and retailers. The company has likewise partnered with a distribution firm that holds a remarkable network of more than 4,500 independent pharmacy accounts.
Additionally, a strong partnership with a prominent distribution network in New Jersey enables SOHM to facilitate nationwide distribution to big distribution houses, hospitals and retail chain stores which include but are not limited to Walmart, Publix, Sam’s and many more retail giants, thus extending the company’s market presence.
SOHM Long-Term
A report by Grand View Research estimated the global nutraceuticals market at $291.33 billion in 2022 and forecasts expansion at a compound annual growth rate (CAGR) of 9.4% from 2023 to 2030. The report states primary factors driving the market growth are preventive health care, increasing instances of lifestyle-related disorders, and rising consumer focus on health-promoting diets. Additionally, increasing consumer spending power in high-growth economies is projected to contribute to the growing demand for nutraceutical products.
Grand View valued the global NSAID market at $19.55 billion in 2021 and forecast it would expand to nearly $30 billion by 2030, marking a CAGR of 5.36% for the period. Projected growth is attributed to factors like the rising prevalence of chronic pain across the world, coupled with a growing global geriatric population. In addition, increasing demand for OTC NSAIDs and the rising adoption of NSAIDs in treating headaches, migraine, toothaches and menstrual pain is expected to boost market growth.
Fortune Business Insights estimated that the global cosmeceuticals market was worth $54.57 billion in 2022 and projects the market will grow to a value of $96.23 billion by 2029, marking a CAGR of 8.4% during the forecast period. The report credits the projected growth to the prevalence of skin disorders around the world and the inclination of dermatologists to prescribe or recommend these products as compared to other treatments.
SOHM envisions a future where it evolves into a prominent global corporation, expanding its reach across international borders while upholding its fundamental core values. The company aspires to extend its export portfolio to encompass 11 countries, showcasing a robust international presence.
Aiming for financial stability, SOHM is committed to maintaining sufficient working capital to support its growth endeavors. The company’s forward trajectory involves strategic collaborations, mergers with diverse brands and a focused approach to business expansion through vertical integration and a balanced mix of organic and inorganic strategies.
In this pursuit, SOHM is dedicated to establishing its proprietary network of partners within the over the counter (OTC) sector. Furthermore, the company seeks heightened recognition within crucial therapeutic domains, including oncology, HIV, cardiovascular health, diabetes care and skincare-dermatology, solidifying its prominent standing in these pivotal segments.
Management Team
Baron Night is CEO, President, and Director at SOHM Inc. He has over 40 years of experience in various industries with extensive contacts in emerging markets. His leadership and track record are great assets to the company as SOHM continues to strengthen its position and develop large-scale distribution of generic drug lines.
David Aguliar, Ph.D., is the COO of SOHM. He has 22 years of experience in the pharmaceutical industry, including multiple research positions and scientific publications. He has an extensive background in pharmaceutical Chemistry Manufacturing and Controls (CMC), as well as quality assurance experience in preclinical and Investigational New Drug (IND) application filings of allogeneic cell-based therapies. He has a deep understanding of regulatory and clinical pathways, coupled with an extensive scientific and technical background in the fields of pharmaceuticals, biopharmaceuticals and gene editing tools research.
Dr. Krishna Bhat, MD PHD, FACC, has a cardiology practice of over 35 years in the field of Clinical and Interventional Cardiology. He is a recipient of the 2021 Hall of Fame Award from the American Heart Association, which was awarded in recognition of his commitment to excellence in the field of Cardiovascular Care through his leadership as an outstanding physician, researcher, and educator. He is also a recipient of the Miles Canada Fellowship Award and the J. Louis Levesque Fellowship Award from Montreal Heart Institute in Montreal, Canada.
Dewey Rushing is a Senior Compliance Remediation and Quality Professional with over 30 years of experience in Quality Assurance and cGMP Compliance for products regulated by the U.S. Food and Drug Administration (FDA). He served as a trained Consumer Safety Investigator at the FDA and Instructor at the Los Angeles District. He has in-depth knowledge in technology transfer of biologics and pharmaceutical products, as well as validation of manufacturing equipment, facility cleaning and critical utility systems maintenance. He has an extensive background in auditing GMP facilities, implementing quality systems and performing gap assessments of manufacturing processes and facilities. He has also directed remediation projects in response to federal compliance audit observations.
Sowmya Jacob, MBA-PGP, possesses over a decade of accomplished and evolving expertise in human resources management, along with manufacturing and operations management. She earned an MBA, complemented by advanced marketing certifications. Demonstrating a track record of achievement, she excels in cultivating collaborative work environments and orchestrating transformative changes that lead to heightened productivity. With adeptness in business analysis, she has occupied senior managerial roles, showcasing her mastery. An engaged participant in professional circles, she maintains active memberships in SPHR and CHRP.
SOHM Inc. (OTC: SHMN), closed Thursday's trading session at $0.00115, off by 4.1667%, on 1,337,500 volume. The average volume for the last 3 months is 4.051M and the stock's 52-week low/high is $0.0005/$0.0015.
Recent News
- SOHM Inc. (OTC: SHMN) - BioMedNewsBreaks - SOHM Inc.'s (SHMN) ABBIE Technology Poised to Make a Difference in the Lives of Millions
- Meta-Analysis Says Nutraceuticals Show Potential in Acne Treatment
- SOHM, Inc. (SHMN) Completes Acquisition of ABBIE Gene-editing Tech, Begins Preparations to Commercialize Solutions for Heart Disease
The QualityStocks Numbers Report
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Expands Partnership with Benedict College through NAFEO-Amesite Alliance
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Releases New Drilling Results from PCH Ionic Clay Project
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Intercepts 83.7 m at 3.6 g/t AuEq (incl. 15.3 m at 6.3 g/t AuEq) and 21.9 m at 1.3% CuEq (incl. 1.4 m at 5.7% CuEq) at the Kay Deposit
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Delivers 50% Revenue Growth and Expanded Gross Margins for Third Quarter of 2023
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Reports on Second Closing of Unsecured Convertible Debentures
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Receive $900K IIA Grant for New CDMO Business Unit
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Reports on Barcelona CPHI Conference
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Featured in Bell2Bell Podcast
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - InvestorNewsBreaks - Clene Inc. (NASDAQ: CLNN) Announces Q3 2023 Financial Results, Operating Highlights
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Brain Cancer Reemergence Could Soon Be Detected Using a Finger-Prick Test
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Spotlighted in New Harbinger Research Report
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc.'s (NEO: CYBN) (NYSE American: CYBN) Announces Participation at Upcoming Milken Institute Future of Health Summit
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Q3 2023 Results
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks - Diamond Lake Minerals Inc. (DLMI) Featured in Recent Article Noting Focus on Helping Investors Transition to Digital Asset Accessibility
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Reports Third Quarter Results
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Acquires Pointward Inc. Technologies
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd. (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Publishes NI 43-101 Technical Report; Elaborates on Iska Iska Project's Economic Potential
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q3 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Announces 10-Year Organic-Waste Processing Agreement, $7M Senior Term Loan
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Issues Operations Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Filing of Federal Lawsuit Against Research Firm
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - Researchers Find Evidence of Tellurium Production When Neutron Stars Merge
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Expands with New Project in James Bay Lithium Region
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Completes Phase 1 of Cross-Collision Prevention Project in Japan
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Secures Request for Quotation with Global Pharmaceutical Conglomerate for Secure Product Transport
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Releases Mining Update Report, Plans to Expand Capacity
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - Government of Canada Invites FuelPositive to Join Rebuild Ukraine Delegation
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) to Present at BIO-Europe 2023
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Reports Record Preliminary Q3 2023 Revenues, Announces Earnings Conference Call
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Expands Scientific Advisory Board as It Advances Product Development, Clinical and Commercial Efforts
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) CEO to Present at Inaugural Transpacific Investment Summit
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Featured in Blog Talking about Winning Strategy of Dual Listing
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Cannabinoid-Based Topical Pain-Relief Treatment Receives European Patent
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) to Present Phase 2 Study Results for INM-755 at 12th Annual WCI
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Granted Second U.S. Patent for JW-700 Treatment Designed for $1.5B Hair-Growth Market
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Continues Expansion in Education and Commercial Real Estate Sectors
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Sex Hormones Could Influence Alzheimer's Development, Researchers Say
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Exploring Patented DehydraTECH(TM) Technology's Effectiveness on GLP-1 Drugs for Potential Diabetes Treatment and Weight Loss
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Schedules Q3 2023 Earnings Conference Call
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (CSE: MYCO) (AQSE: MYIG) (OTC: MYCOF) Files Prospectus Supplement, Closes Share Subscription Agreements
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Appointment of Strategic AI Advisor
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Brand Metrics e-Commerce Companies Need to Watch
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - Rich Millennials View EVs as a Status Symbol
- PaxMedica Inc. (NASDAQ: PXMD) - InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) CMO Featured in Latest Bell2Bell Podcast Release
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Announces Addition of Meek's Vegan Pizza to Chicago Foodhall Offerings
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Q3 Player Activity Milestones
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Bi-Weekly Status Report
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) BaroFold Technology Platform Featured at International Bioprocess Conference
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Creates Advisory Board, Names Renowned Experts as Members
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Releases Status Update on Development Update
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Releases Corporate Update, Including Aleafia Health Agreement
- Renovaro BioSciences Inc. (NASDAQ: RENB) - BioMedNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB), GEDi Cube Combine to Transform Medicine Through AI and Biotherapeutics
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Swedish Scientists Find Promising Way to Recover Minerals from Discarded EV Batteries
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Reports on Strong Drill Results at Oko West Project in Guyana
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Becomes Largest Shareholder of Plantify Foods Inc.
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters into Acquisition Agreement, Asset Purchase Agreement
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Positive Results of Preclinical Study to Advance SPU-21
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Charting the Path for Digital Identity Solutions Adoption and Cybersecurity Advancement Globally
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Closing of $1.6M Public Offering, $750K Order for Use by First Responders in Israel
- SOHM Inc. (OTC: SHMN) - BioMedNewsBreaks - SOHM Inc.'s (SHMN) ABBIE Technology Poised to Make a Difference in the Lives of Millions
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Exclusive Single-Serve Wine Brand to Be Featured by 130-Plus Circle K Locations
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Makes 'Significant Move' with Exercise of SWA Project Option Agreement
- Starco Brands Inc. (OTCQB: STCB) - Whipshots® Launches Festive New Holiday Campaign with Patti's Good Life
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Subsidiary Meets with Jordan Minister of Water in Round Table Gathering
- SenesTech Inc. (NASDAQ: SNES) - SenesTech Announces Third Quarter 2023 Financial Results
- Sekur Private Data Ltd. (FRA: GTD0) (CSE: SKUR) (OTCQB: SWISF) - New to The Street TV Announces Five Corporate Interviews on its 527th Episode, Airs on the Fox Business Network Tonight, Monday, November 6, 2023, at 10:30 PM PT
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Receives Full Compliance Notification from NASDAQ
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Enters MOU to Launch Business, Service Offerings in Pakistan
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Anticipates Gross Proceeds of AUD$3M on Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TrxADE Health, Inc. affirms its commitment to the pharmaceutical space via its subsidiary Trxade, Inc. which operates the pharmaceutical marketplace and added more members in the third quarter of 2023.
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - InvestorNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) to Present at Red Cloud's 2023 Fall Mining Showcase
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Releases FY 2023 Report
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Schedules Release of Q2 Results, Corporate Update Conference Call
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Closing of Million-Dollar Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Giles Tipsword Joins Wrap Technologies as Senior Vice President of North American Sales
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Expands Partnership with Benedict College through NAFEO-Amesite Alliance
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Releases New Drilling Results from PCH Ionic Clay Project
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Intercepts 83.7 m at 3.6 g/t AuEq (incl. 15.3 m at 6.3 g/t AuEq) and 21.9 m at 1.3% CuEq (incl. 1.4 m at 5.7% CuEq) at the Kay Deposit
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Delivers 50% Revenue Growth and Expanded Gross Margins for Third Quarter of 2023
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Reports on Second Closing of Unsecured Convertible Debentures
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Receive $900K IIA Grant for New CDMO Business Unit
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Reports on Barcelona CPHI Conference
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Featured in Bell2Bell Podcast
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - InvestorNewsBreaks - Clene Inc. (NASDAQ: CLNN) Announces Q3 2023 Financial Results, Operating Highlights
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Brain Cancer Reemergence Could Soon Be Detected Using a Finger-Prick Test
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Spotlighted in New Harbinger Research Report
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc.'s (NEO: CYBN) (NYSE American: CYBN) Announces Participation at Upcoming Milken Institute Future of Health Summit
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Q3 2023 Results
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks - Diamond Lake Minerals Inc. (DLMI) Featured in Recent Article Noting Focus on Helping Investors Transition to Digital Asset Accessibility
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Reports Third Quarter Results
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Acquires Pointward Inc. Technologies
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd. (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Publishes NI 43-101 Technical Report; Elaborates on Iska Iska Project's Economic Potential
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q3 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Announces 10-Year Organic-Waste Processing Agreement, $7M Senior Term Loan
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Issues Operations Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Filing of Federal Lawsuit Against Research Firm
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - Researchers Find Evidence of Tellurium Production When Neutron Stars Merge
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Expands with New Project in James Bay Lithium Region
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Completes Phase 1 of Cross-Collision Prevention Project in Japan
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Secures Request for Quotation with Global Pharmaceutical Conglomerate for Secure Product Transport
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Releases Mining Update Report, Plans to Expand Capacity
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - Government of Canada Invites FuelPositive to Join Rebuild Ukraine Delegation
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) to Present at BIO-Europe 2023
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Reports Record Preliminary Q3 2023 Revenues, Announces Earnings Conference Call
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Expands Scientific Advisory Board as It Advances Product Development, Clinical and Commercial Efforts
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) CEO to Present at Inaugural Transpacific Investment Summit
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Featured in Blog Talking about Winning Strategy of Dual Listing
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Cannabinoid-Based Topical Pain-Relief Treatment Receives European Patent
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) to Present Phase 2 Study Results for INM-755 at 12th Annual WCI
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Granted Second U.S. Patent for JW-700 Treatment Designed for $1.5B Hair-Growth Market
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Continues Expansion in Education and Commercial Real Estate Sectors
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Sex Hormones Could Influence Alzheimer's Development, Researchers Say
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Exploring Patented DehydraTECH(TM) Technology's Effectiveness on GLP-1 Drugs for Potential Diabetes Treatment and Weight Loss
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Schedules Q3 2023 Earnings Conference Call
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (CSE: MYCO) (AQSE: MYIG) (OTC: MYCOF) Files Prospectus Supplement, Closes Share Subscription Agreements
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Appointment of Strategic AI Advisor
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Brand Metrics e-Commerce Companies Need to Watch
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - Rich Millennials View EVs as a Status Symbol
- PaxMedica Inc. (NASDAQ: PXMD) - InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) CMO Featured in Latest Bell2Bell Podcast Release
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Announces Addition of Meek's Vegan Pizza to Chicago Foodhall Offerings
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Q3 Player Activity Milestones
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Bi-Weekly Status Report
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) BaroFold Technology Platform Featured at International Bioprocess Conference
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Creates Advisory Board, Names Renowned Experts as Members
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Releases Status Update on Development Update
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Releases Corporate Update, Including Aleafia Health Agreement
- Renovaro BioSciences Inc. (NASDAQ: RENB) - BioMedNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB), GEDi Cube Combine to Transform Medicine Through AI and Biotherapeutics
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Swedish Scientists Find Promising Way to Recover Minerals from Discarded EV Batteries
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Reports on Strong Drill Results at Oko West Project in Guyana
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Becomes Largest Shareholder of Plantify Foods Inc.
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters into Acquisition Agreement, Asset Purchase Agreement
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Positive Results of Preclinical Study to Advance SPU-21
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Charting the Path for Digital Identity Solutions Adoption and Cybersecurity Advancement Globally
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Closing of $1.6M Public Offering, $750K Order for Use by First Responders in Israel
- SOHM Inc. (OTC: SHMN) - BioMedNewsBreaks - SOHM Inc.'s (SHMN) ABBIE Technology Poised to Make a Difference in the Lives of Millions
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Exclusive Single-Serve Wine Brand to Be Featured by 130-Plus Circle K Locations
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Makes 'Significant Move' with Exercise of SWA Project Option Agreement
- Starco Brands Inc. (OTCQB: STCB) - Whipshots® Launches Festive New Holiday Campaign with Patti's Good Life
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Subsidiary Meets with Jordan Minister of Water in Round Table Gathering
- SenesTech Inc. (NASDAQ: SNES) - SenesTech Announces Third Quarter 2023 Financial Results
- Sekur Private Data Ltd. (FRA: GTD0) (CSE: SKUR) (OTCQB: SWISF) - New to The Street TV Announces Five Corporate Interviews on its 527th Episode, Airs on the Fox Business Network Tonight, Monday, November 6, 2023, at 10:30 PM PT
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Receives Full Compliance Notification from NASDAQ
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Enters MOU to Launch Business, Service Offerings in Pakistan
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Anticipates Gross Proceeds of AUD$3M on Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TrxADE Health, Inc. affirms its commitment to the pharmaceutical space via its subsidiary Trxade, Inc. which operates the pharmaceutical marketplace and added more members in the third quarter of 2023.
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - InvestorNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) to Present at Red Cloud's 2023 Fall Mining Showcase
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Releases FY 2023 Report
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Schedules Release of Q2 Results, Corporate Update Conference Call
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Closing of Million-Dollar Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Giles Tipsword Joins Wrap Technologies as Senior Vice President of North American Sales
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.
The QualityStocks Numbers Report
QualityStocksTwits is your stock tracking service portal to Twitter's universe of stock picks, commentary and research.
The QualityStocks Sponsored News
- 180 Life Sciences Corp. (NASDAQ: ATNF) - InvestorNewsBreaks - 180 Life Sciences Corp. (NASDAQ: ATNF) Releases Public Offering Pricing, Offering Could Reach $3M
- Aditxt Inc. (NASDAQ: ADTX) - InvestorNewsBreaks - Aditxt Inc. (NASDAQ: ADTX) Subsidiary Inks Stiff Person Syndrome Clinical Trial Agreement with Mayo Clinic
- Advanced Container Technologies Inc. (OTC: ACTX) - 420 with CNW - Study Finds Psychosis-Prone Young People Registered Surprising Symptom Improvement After Using Marijuana
- Amesite Inc. (NASDAQ: AMST) - InvestorNewsBreaks - Amesite Inc. (NASDAQ: AMST) Expands Partnership with Benedict College through NAFEO-Amesite Alliance
- Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) - InvestorNewsBreaks - Appia Rare Earths & Uranium Corp. (CSE: API) (OTCQX: APAAF) (FSE: A0I0) Releases New Drilling Results from PCH Ionic Clay Project
- Fresh2 Group Ltd. (NASDAQ: FRES) - Fresh2 Group Ltd. Completes Acquisition of SpeedIn INC, a Delivery Service Provider
- Arizona Metals Corp. (TSX: AMC) (OTCQX: AZMCF) - Arizona Metals Intercepts 83.7 m at 3.6 g/t AuEq (incl. 15.3 m at 6.3 g/t AuEq) and 21.9 m at 1.3% CuEq (incl. 1.4 m at 5.7% CuEq) at the Kay Deposit
- Augmedix Inc. (NASDAQ: AUGX) - Augmedix Delivers 50% Revenue Growth and Expanded Gross Margins for Third Quarter of 2023
- Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) - InvestorNewsBreaks - Battery Mineral Resources Corp. (TSX.V: BMR) (OTCQB: BTRMF) Reports on Second Closing of Unsecured Convertible Debentures
- BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) - InvestorNewsBreaks - BevCanna Enterprises Inc. (CSE: BEV) (OTCQB: BVNNF) (FSE: 7BC) Releases Shareholder Update with Corporate, Operational Highlights
- Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) - InvestorNewsBreaks - Scinai Immunotherapeutics Ltd. (NASDAQ: SCNI) Receive $900K IIA Grant for New CDMO Business Unit
- BioNxt Solutions Inc. (CSE: BNXT) (OTCQB: BNXTF) (FSE: 4XT) - BioNxt Solutions Reports on Barcelona CPHI Conference
- Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) - InvestorNewsBreaks - Blue Hat Interactive Entertainment Technology (NASDAQ: BHAT) Inks Gold Purchasing Agreement with Precious Metals Group
- Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) - InvestorNewsBreaks - Canada Nickel Company Inc. (TSX.V: CNC) (OTCQX: CNIKF) Featured in Bell2Bell Podcast
- CISO Global, Inc. (NASDAQ: CISO) - InvestorNewsBreaks - CISO Global Inc.'s (NASDAQ: CISO) Appoints Security Expert as New Chief Information Security Officer
- Clene Inc. (NASDAQ: CLNN) - InvestorNewsBreaks - Clene Inc. (NASDAQ: CLNN) Announces Q3 2023 Financial Results, Operating Highlights
- CNS Pharmaceuticals Inc. (NASDAQ: CNSP) - Brain Cancer Reemergence Could Soon Be Detected Using a Finger-Prick Test
- Correlate Energy Corp. (OTCQB: CIPI) - InvestorNewsBreaks - Correlate Energy Corp. (CIPI) Spotlighted in New Harbinger Research Report
- Coyuchi Inc. - InvestorNewsBreaks – Coyuchi Inc. Pushing Organic Textile Market Forward Through Its Circular Initiatives
- Cub Crafters Inc. - InvestorNewsBreaks – CubCrafters Inc. CEO Eyes Bright Future for Fleet Sales Program
- Cybin Inc. (NEO: CYBN) (NYSE American: CYBN) - InvestorNewsBreaks - Cybin Inc.'s (NEO: CYBN) (NYSE American: CYBN) Announces Participation at Upcoming Milken Institute Future of Health Summit
- DarioHealth Corp. (NASDAQ: DRIO) - InvestorNewsBreaks - DarioHealth Corp. (NASDAQ: DRIO) Releases Q3 2023 Results
- Diamond Lake Minerals Inc. (OTC: DLMI) - InvestorNewsBreaks - Diamond Lake Minerals Inc. (DLMI) Featured in Recent Article Noting Focus on Helping Investors Transition to Digital Asset Accessibility
- Data443 Risk Mitigation Inc. (OTC: ATDS) - InvestorNewsBreaks - Data443 Risk Mitigation Inc. (ATDS) Announces Q2, FH 2023 Results, Issues Corporate Update
- D-Wave Quantum Inc. (NYSE: QBTS) - D-Wave Reports Third Quarter Results
- DSG Global Inc. (OTCQB: DSGT) - InvestorNewsBreaks - DSG Global Inc.'s (DSGT) Vantage Tag Systems Announces Selection of Its Infinity GPS Fleet Management System at Two Signature Facilities
- Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) - InvestorNewsBreaks - Eat Well Investment Group Inc. (CSE: EWG) (OTC: EWGFF) Inks MIPA in $10M NASDAQ Merger
- Electronic Servitor Publication Network Inc. (OTCQB: XESP) - InvestorNewsBreaks - Electronic Servitor Publication Network Inc. (XESP) Acquires Pointward Inc. Technologies
- Eloro Resources Ltd. (TSX.V: ELO) (OTCQX: ELRRF) - Eloro Resources Ltd. (TSX: ELO) (OTCQX: ELRRF) (FSE: P2QM) Publishes NI 43-101 Technical Report; Elaborates on Iska Iska Project's Economic Potential
- Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) - InvestorNewsBreaks - Energy Fuels Inc. (NYSE American: UUUU) (TSX: EFR) Releases Q3 2023 Financial Report, Corporate Updates
- EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQB: EVGIF) - InvestorNewsBreaks - EverGen Infrastructure Corp. (TSX.V: EVGN) (OTCQX: EVGIF) Announces 10-Year Organic-Waste Processing Agreement, $7M Senior Term Loan
- Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) - InvestorNewsBreaks - Exro Technologies Inc. (TSX: EXRO) (OTCQB: EXROF) Issues Operations Update
- FingerMotion Inc. (NASDAQ: FNGR) - InvestorNewsBreaks - FingerMotion Inc. (NASDAQ: FNGR) Announces Filing of Federal Lawsuit Against Research Firm
- Fintech Ecosystem Development Corp. (NASDAQ: FEXD) - InvestorNewsBreaks - Fintech Ecosystem Development Corp. (NASDAQ: FEXD) Carving out Share in Challenger Bank, Crypto Markets
- First Tellurium Corp. (CSE: FTEL) (OTCQB: FSTTF) - Researchers Find Evidence of Tellurium Production When Neutron Stars Merge
- FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) - InvestorNewsBreaks - FE Battery Metals Corp. (CSE: FE) (OTCQB: FEMFF) (WKN: A2JC89) Expands with New Project in James Bay Lithium Region
- Flora Growth Corp. (NASDAQ: FLGC) - 420 with CNW - Connecticut Retailers See Brisk Business During First Week of Legal Cannabis Sales
- Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) - InvestorNewsBreaks - Foresight Autonomous Holdings Ltd. (NASDAQ: FRSX) (TASE: FRSX) Subsidiary Completes Phase 1 of Cross-Collision Prevention Project in Japan
- Freight Technologies Inc. (NASDAQ: FRGT) - Freight Technologies Inc. (NASDAQ: FRGT) Secures Request for Quotation with Global Pharmaceutical Conglomerate for Secure Product Transport
- Friendable Inc. (FDBL) - InvestorNewsBreaks - Friendable Inc. (FDBL) Announces Acquisition of OnlyFanPass.com Domain as 'Exciting New Business Opportunities' Develop
- GEMXX Corp. (OTC: GEMZ) - InvestorNewsBreaks - GEMXX Corporation (GEMZ) Releases Mining Update Report, Plans to Expand Capacity
- FuelPositive Corp. (TSX.V: NHHH) (OTC: NHHHF) - Government of Canada Invites FuelPositive to Join Rebuild Ukraine Delegation
- GeoSolar Technologies Inc. - QualityStocksNewsBreaks – GeoSolar Technologies Inc. Taking Part in Shift Toward a Greener Future
- Golden Matrix Group Inc. (NASDAQ: GMGI) - InvestorNewsBreaks - Golden Matrix Group Inc. (NASDAQ: GMGI) Releases FY 2022 Financial Numbers, Reports Record Revenue
- Golden Triangle Ventures Inc. (OTC: GTVH) - InvestorNewsBreaks - Golden Triangle Ventures Inc. (GTVH) Launches Sales of Le Pragma CBD Product Line; Outlines Le Pragma Development Plan
- GolfLync Inc. - GolfLync Inc. Unveils Exciting Updates to Enhance Your Golfing Experience
- Genprex Inc. (NASDAQ: GNPX) - InvestorNewsBreaks - Genprex Inc. (NASDAQ: GNPX) to Present at BIO-Europe 2023
- RYVYL Inc. (NASDAQ: RVYL) - InvestorNewsBreaks - RYVYL Inc. (NASDAQ: RVYL) Reports Record Preliminary Q3 2023 Revenues, Announces Earnings Conference Call
- HeartBeam Inc. (NASDAQ: BEAT) - InvestorNewsBreaks - HeartBeam Inc. (NASDAQ: BEAT) Expands Scientific Advisory Board as It Advances Product Development, Clinical and Commercial Efforts
- Cepton Inc. (NASDAQ: CPTN) - InvestorNewsBreaks - Cepton Inc. (NASDAQ: CPTN) Management Announces Participation at Upcoming J.P. Morgan Auto Conference
- Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTD) (FRA: 7HIA.F) - InvestorNewsBreaks - Hillcrest Energy Technologies Ltd. (CSE: HEAT) (OTCQB: HLRTF) (FRA: 7HIA.F) CEO to Present at Inaugural Transpacific Investment Summit
- Home Bistro Inc. (OTC: HBIS) - Home Bistro CEO Letter to Shareholders and Stakeholders
- Horizon Fintex | Upstream - InvestorNewsBreaks – Upstream Featured in Blog Talking about Winning Strategy of Dual Listing
- iClick Interactive Asia Group Ltd. (NASDAQ: ICLK) - InvestorNewsBreaks - iClick Interactive Asia Group Limited (NASDAQ: ICLK) to Help Overseas Brands Unlock New Opportunities
- Ideanomics Inc. (NASDAQ: IDEX) - InvestorNewsBreaks - Ideanomics Inc. (NASDAQ: IDEX) Releases Q2 2023 Financial Report
- IGC Pharma Inc. (NYSE American: IGC) - InvestorNewsBreaks - IGC Pharma Inc. (NYSE American: IGC) Cannabinoid-Based Topical Pain-Relief Treatment Receives European Patent
- Infobird Co., Ltd (NASDAQ: IFBD) - Infobird Co., Ltd Announces Receipt of Delinquency Notification Letter from Nasdaq
- InMed Pharmaceuticals Inc. (NASDAQ: INM) - InvestorNewsBreaks - InMed Pharmaceuticals Inc. (NASDAQ: INM) to Present Phase 2 Study Results for INM-755 at 12th Annual WCI
- Safety Shot Inc. (NASDAQ: SHOT) - InvestorNewsBreaks - Safety Shot (NASDAQ: SHOT) Granted Second U.S. Patent for JW-700 Treatment Designed for $1.5B Hair-Growth Market
- BlockQuarry Corp. (OTC: BLQC) - Interoperability Crucial to Mass Adoption of Blockchain Technology
- Knightscope (NASDAQ: KSCP) - InvestorNewsBreaks - Knightscope, Inc. (NASDAQ: KSCP) Continues Expansion in Education and Commercial Real Estate Sectors
- Kronos Advanced Technologies Inc. (OTC: KNOS) - InvestorNewsBreaks - Kronos Advanced Technologies Inc.'s (KNOS) Products Provide Relief from Canadian Wildfire Smoke
- Laredo Oil Inc. (OTC: LRDC) - ESGBreaks - Laredo Oil Inc. (LRDC) Ideally Poised as Bullish 2023 Forecast Suggests Pricing Uptrend
- Longeveron Inc. (NASDAQ: LGVN) - Sex Hormones Could Influence Alzheimer's Development, Researchers Say
- Lexaria Bioscience Corp. (NASDAQ: LEXX) - Lexaria Bioscience Corp. (NASDAQ: LEXX) Exploring Patented DehydraTECH(TM) Technology's Effectiveness on GLP-1 Drugs for Potential Diabetes Treatment and Weight Loss
- Lottery.com Inc. (NASDAQ: LTRY) - Lottery.com, Inc subsidiary, Sports.com, Inc. to enter partnership with Data Sports Group
- LQwD FinTech Corp. (TSX.V: LQWD) (OTCQB: LQWDF) - Mastercard Zeroes in On Five Factors to Facilitate Use of Cryptos as Payments
- Lucy Scientific Discovery Inc. (NASDAQ: LSDI) - Neuroscience Study Shows Psychedelic-Assisted Therapy Causes Rapid Changes in Brain
- McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) - InvestorNewsBreaks - McEwen Mining Inc. (NYSE: MUX) (TSX: MUX) Schedules Q3 2023 Earnings Conference Call
- MetAlert Inc. (OTC: MLRT) - Researchers Find Balancing Ability May Predict Risk of Cognitive Decline
- Mind Cure Health Inc. (CSE: MCUR) (OTCQX: MCURF) - MIND CURE HEALTH INC. ANNOUNCES CLOSING OF THE FIRST TRANCHE OF LNG ENERGY GROUP'S SUBSCRIPTION RECEIPT FINANCING
- Mydecine Innovations Group Inc. (NEO: MYCO) (OTC: MYCOF) - InvestorNewsBreaks - Mydecine Innovations Group Inc. (CSE: MYCO) (AQSE: MYIG) (OTC: MYCOF) Files Prospectus Supplement, Closes Share Subscription Agreements
- Mountain Top Properties Inc. (OTC: MTPP) - Mountain Top Properties Inc. (MTPP) Is 'One to Watch'
- Near Intelligence Inc. (NASDAQ: NIR) - InvestorNewsBreaks - Near Intelligence Inc. (NASDAQ: NIR) Partners with IBN to Strengthen Industry Awareness
- Nemaura Medical Inc. (NASDAQ: NMRD) - InvestorNewsBreaks - Nemaura Medical Inc. (NASDAQ: NMRD) Announces Appointment of Strategic AI Advisor
- NextPlat Corp. (NASDAQ: NXPL) (NASDAQ: NXPLW) - Brand Metrics e-Commerce Companies Need to Watch
- Nowigence Inc. (OTCQB: NOWG) - Nowigence Inc. (NOWG) Starts Trading on OTC
- OK Stone Engineering Inc. - OK Stone Engineering Inc. Company Profile
- Mullen Automotive Inc. (NASDAQ: MULN) - Rich Millennials View EVs as a Status Symbol
- PaxMedica Inc. (NASDAQ: PXMD) - InvestorNewsBreaks - PaxMedica Inc. (NASDAQ: PXMD) CMO Featured in Latest Bell2Bell Podcast Release
- PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTCQB: PLTXF) - InvestorNewsBreaks - PlantX Life Inc. (CSE: VEGA) (Frankfurt: WNT1) (OTC: PLTXF) Announces Addition of Meek's Vegan Pizza to Chicago Foodhall Offerings
- Playgon Games Inc. (TSX.V: DEAL) (OTCQB: PLGNF) - InvestorNewsBreaks - Playgon Games Inc. (TSX.V: DEAL) (OTC: PLGNF) (FSE: 7CR) Announces Q3 Player Activity Milestones
- PowerTap Hydrogen Capital Corp. (NEO: MOVE) (FWB: 2K6A) (OTC: MOTNF) - PowerTap Provides Bi-Weekly Status Report
- Predictive Oncology (NASDAQ: POAI) - InvestorNewsBreaks - Predictive Oncology Inc. (NASDAQ: POAI) Releases Q2 2023 Financial Results, Corporate Update
- Pressure BioSciences Inc. (PBIO) - InvestorNewsBreaks - Pressure BioSciences Inc. (PBIO) BaroFold Technology Platform Featured at International Bioprocess Conference
- Prime Harvest Inc. - InvestorNewsBreaks – Prime Harvest Inc. Eyes Continued Growth and Expansion with Recently Launched Reg A+ Offering
- Processa Pharmaceuticals Inc. (NASDAQ: PCSA) - InvestorNewsBreaks - Processa Pharmaceuticals Inc. (NASDAQ: PCSA) Creates Advisory Board, Names Renowned Experts as Members
- Progressive Care Inc. (OTCQB: RXMD) - InvestorNewsBreaks - Progressive Care Inc. (RXMD) Releases Record breaking Q1 2023 Financial Results
- QSAM Biosciences Inc. (OTCQB: QSAM) - InvestorNewsBreaks - QSAM Biosciences Inc. (QSAM) Issues Update on Progress, Preliminary Clinical Data for Phase 1 CycloSam(R) Trial
- Prospera Energy Inc. (TSX.V: PEI) (FRA: OF6B) (OTC: GXRFF) - InvestorNewsBreaks - Prospera Energy Inc. (TSX.V: PEI) (OTC: GXRFF) (FRA: OF6B) Releases Status Update on Development Update
- Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) - InvestorNewsBreaks - Red White & Bloom Brands Inc. (CSE: RWB) (OTCQX: RWBYF) Releases Corporate Update, Including Aleafia Health Agreement
- Renovaro BioSciences Inc. (NASDAQ: RENB) - BioMedNewsBreaks - Renovaro BioSciences Inc. (NASDAQ: RENB), GEDi Cube Combine to Transform Medicine Through AI and Biotherapeutics
- Reflex Advanced Materials Corp. (CSE: RFLX) (OTCQB: RFLXF) - Swedish Scientists Find Promising Way to Recover Minerals from Discarded EV Batteries
- Reunion Gold Corp. (TSX.V: RGD) (OTCQX: RGDFF) - InvestorNewsBreaks - Reunion Gold Corporation (TSX.V: RGD) (OTCQX: RGDFF) Reports on Strong Drill Results at Oko West Project in Guyana
- REZYFi, Inc. - InvestorNewsBreaks – REZYFi Inc. Using Diversified Approach to Capitalize on Growth in Multiple Verticals
- RJD Green Inc. (OTC: RJDG) - InvestorNewsBreaks - RJD Green Inc. (RJDG) Division Inks Definitive Purchase Agreement with Midwest Fireplace, Wood Stove Company
- RVL Pharmaceuticals plc (NASDAQ: RVLP) - InvestorNewsBreaks - RVL Pharmaceuticals plc (NASDAQ: RVLP) Reaches Agreement Regarding Reorganization of Three US Subsidiaries
- Sanwire Corp. (SNWR) - InvestorNewsBreaks - Sanwire Corporation's (SNWR) Intercept Music Incorporates Cutting-Edge AI in Latest Software Launch
- Save Foods Inc. (NASDAQ: SVFD) - InvestorNewsBreaks - Save Foods Inc. (NASDAQ: SVFD) Becomes Largest Shareholder of Plantify Foods Inc.
- Sharing Services Global Corporation (SHRG) - InvestorNewsBreaks - Sharing Services Global Corporation (SHRG) Inks LOI for Acquisition of MojiLife Brand Products
- Sigma Additive Solutions (NASDAQ: SASI) - InvestorNewsBreaks - Sigma Additive Solutions Inc. (NASDAQ: SASI) Enters into Acquisition Agreement, Asset Purchase Agreement
- Silo Pharma Inc. (OTCQB: SILO) - InvestorNewsBreaks - Silo Pharma Inc. (NASDAQ: SILO) Announces Positive Results of Preclinical Study to Advance SPU-21
- SuperCom Ltd. (NASDAQ: SPCB) - SuperCom Ltd. (NASDAQ: SPCB) Charting the Path for Digital Identity Solutions Adoption and Cybersecurity Advancement Globally
- Siyata Mobile Inc. (NASDAQ: SYTA) (TSXV: SIM) - InvestorNewsBreaks - Siyata Mobile Inc. (NASDAQ: SYTA) Announces Closing of $1.6M Public Offering, $750K Order for Use by First Responders in Israel
- SOHM Inc. (OTC: SHMN) - BioMedNewsBreaks - SOHM Inc.'s (SHMN) ABBIE Technology Poised to Make a Difference in the Lives of Millions
- Splash Beverage Group Inc. (NYSE American: SBEV) - InvestorNewsBreaks - Splash Beverage Group Inc. (NYSE American: SBEV) Exclusive Single-Serve Wine Brand to Be Featured by 130-Plus Circle K Locations
- SPYR Inc. (OTCQB: SPYR) - InvestorNewsBreaks - SPYR Inc. (SPYR) Subsidiary Patented Platform Provides Smart Solutions for Simple IoT Problems
- Standard Lithium Ltd. (NYSE American: SLI) - InvestorNewsBreaks - Standard Lithium Ltd. (TSX.V: SLI) (NYSE American: SLI) (FRA: S5L) Makes 'Significant Move' with Exercise of SWA Project Option Agreement
- Starco Brands Inc. (OTCQB: STCB) - Whipshots® Launches Festive New Holiday Campaign with Patti's Good Life
- StorEn Technologies Inc. - InvestorNewsBreaks – StorEn Technologies Inc. Recognized for Bringing Real Innovation to Vanadium Flow Batteries
- Sustainable Green Team Ltd. (OTC: SGTM) - InvestorNewsBreaks - Sustainable Green Team Ltd. (SGTM) Subsidiary Meets with Jordan Minister of Water in Round Table Gathering
- SenesTech Inc. (NASDAQ: SNES) - SenesTech Announces Third Quarter 2023 Financial Results
- Sekur Private Data Ltd. (FRA: GTD0) (CSE: SKUR) (OTCQB: SWISF) - New to The Street TV Announces Five Corporate Interviews on its 527th Episode, Airs on the Fox Business Network Tonight, Monday, November 6, 2023, at 10:30 PM PT
- TAAT Global Alternatives Inc Com (TOBAD) - InvestorNewsBreaks - TAAT(TM) Global Alternatives Inc. (CSE: TAAT) (OTCQB: TOBAF) (FRANKFURT: 2TP2) Plans 10-to-One Share Consolidation
- The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) - InvestorNewsBreaks - The Alkaline Water Company Inc. (NASDAQ: WTER) (CSE: WTER) Receives Full Compliance Notification from NASDAQ
- Tingo Group Inc. (NASDAQ: TIO) - InvestorNewsBreaks - Tingo Group Inc. (NASDAQ: TIO) Enters MOU to Launch Business, Service Offerings in Pakistan
- Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) - InvestorNewsBreaks - Tryp Therapeutics Inc. (CSE: TRYP) (OTCQB: TRYPF) Anticipates Gross Proceeds of AUD$3M on Private Placement
- TRxADE HEALTH, INC. (NASDAQ: MEDS) - TrxADE Health, Inc. affirms its commitment to the pharmaceutical space via its subsidiary Trxade, Inc. which operates the pharmaceutical marketplace and added more members in the third quarter of 2023.
- Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) - InvestorNewsBreaks - Ucore Rare Metals Inc. (TSX.V: UCU) (OTCQX: UURAF) to Present at Red Cloud's 2023 Fall Mining Showcase
- Uranium Energy Corp. (NYSE American: UEC) - InvestorNewsBreaks - Uranium Energy Corp. (NYSE American: UEC) Releases FY 2023 Report
- Utopia VR - InvestorNewsBreaks – Genius Group Limited. (NYSE American: GNS) Announces Dual Listing, Commences Trading on Upstream in April
- VistaGen Therapeutics Inc. (NASDAQ: VTGN) - InvestorNewsBreaks - VistaGen Therapeutics Inc. (NASDAQ: VTGN) Schedules Release of Q2 Results, Corporate Update Conference Call
- Vivos Therapeutics Inc. (NASDAQ: VVOS) - InvestorNewsBreaks - Vivos Therapeutics Inc. (NASDAQ: VVOS) Announces Closing of Million-Dollar Private Placement
- Wrap Technologies Inc. (NASDAQ: WRAP) - Giles Tipsword Joins Wrap Technologies as Senior Vice President of North American Sales
The QualityStocks DailyNetwork Sponsors
About The QualityStocks Daily
The QualityStocks Daily Newsletter brings you the latest company News and Profiles featuring the "Top Movers and Shakers" from the Small Cap Market each trading day. QualityStocks is committed to bring our subscribers Public companies in our Newsletter Section "Free of Charge" based on Percentage gained, Momentum, Press, and or Company Fundamentals.
Why do we spotlight companies for Free?
We Want To bring our subscribers the top movers in an unbiased setting.
"Homework Eliminates Mistakes"
Please never invest in a company anyone profiles unless you do the proper research and due diligence.
QualityStocks is compensated by the companies in The QS Company Corner. These companies will include a disclaimer with the amount and term of compensation.
Please consult the QualityStocks Market Basics Section on our site.







































